Abstract
Dendritic cells (DCs) are antigen presenting cells of the immune system that play a crucial role in lymphocyte responses, host defense mechanisms, and pathogenesis of inflammation. Isolation and study of DCs have been important in biological research because of their distinctive features. Although they are essential key mediators of the immune system, DCs are very rare in blood, accounting for approximately 0.1 - 1% of total blood mononuclear cells. Therefore, alternatives for isolation methods rely on the differentiation of DCs from monocytes isolated from peripheral blood mononuclear cells (PBMCs). The utilization of proper isolation techniques that combine simplicity, affordability, high purity, and high yield of cells is imperative to consider. In the current study, two distinct methods for the generation of DCs will be compared. Monocytes were selected by adherence or negatively enriched using magnetic separation procedure followed by differentiation into DCs with IL-4 and GM-CSF. Monocyte and MDDC viability, proliferation, and phenotype were assessed using viability dyes, MTT assay, and CD11c/ CD14 surface marker analysis by imaging flow cytometry. Although the magnetic separation method yielded a significant higher percentage of monocytes with higher proliferative capacity when compared to the adhesion method, the findings have demonstrated the ability of both techniques to simultaneously generate monocytes that are capable of proliferating and differentiating into viable CD11c+ MDDCs after seven days in culture. Both methods yielded > 70% CD11c+ MDDCs. Therefore, our results provide insights that contribute to the development of reliable methods for isolation and characterization of human DCs.
Keywords: Immunology, Issue 116, Human, monocyte-derived dendritic cells, monocytes, isolation techniques, immune system, magnetic separation
Introduction
Dendritic cells (DCs) are essential mediators of the innate and adaptive immune systems. They function to induce primary immune responses and facilitate the development of immunological memory. These cells are primarily responsible for antigen capture, migration and T cell stimulation and are therefore referred to as professional antigen presenting cells (APCs) 1.Manipulation of DCs could be utilized across a wide variety of research fields and in the clinical setting to treat different inflammatory diseases such as HIV 6,7, cancer 8, autoimmune diseases 9, and allergic responses 10. DCs are also being used for substance abuse research in order to solve unknown mechanisms and pathways such as those associated with alcohol dependence 11-14, drug dependence 13,15, and the combination of HIV infection and substance abuse 16-19. These ongoing studies and future research studies in the field of immunology make in vitro generation of DCs extremely important for research. However, there are several difficulties associated with isolating DCs from human blood as they only constitute 0.1 - 1% of total blood mononuclear cells 20.
To date, some of the well-established methods for the generation of DCs in vitro consists of plastic or glass adherence of monocytes 21,22, density gradient centrifugation 23, specific marker based separation such as magnetic activated cell sorting 22, fluorescent activated cell sorting 24, positive selection of CD14+ monocytes using dextran-coated magnetic nanoparticles 25, and rapid isolation of highly purified monocytes using fully automated negative cell selection 26. However, the best method of choice remains controversial. Therefore, to improve DC generation techniques, several methods have been developed in which the purity of these cells can be greatly increased by differentiation from purified CD34+ progenitor cells and monocytes isolated from peripheral blood mononuclear cells (PBMCs) 27. As mentioned prior, a widely used and popular method for generating monocyte derived dendritic cells (MDDCs) is to explore the ability of monocytes to adhere to glass or plastic (adherence method) 21,22,27. The adherence method is a rapid and straightforward method that does not require the use of complex equipment. However, some disadvantages of this process include lymphocyte contamination, low flexibility, and monocyte transient manipulation 28. An alternative method to the adherence method is the magnetic isolation of monocytes from total PBMCs , particularly with the use of a human monocyte enrichment kit, which is designed to isolate monocytes from PBMCs by negative selection 26. During this procedure, unwanted cells are targeted for removal with tetrameric antibody complexes and dextran-coated magnetic particles. The advantage of this isolation method is that the unwanted labeled cells are separated using a magnet while target cells can be freely poured off into a new tube without the need for columns. To date, with the availability of specific monoclonal antibodies that can label unique cell populations, the magnetic separation technique has become not simply an additional method, but a necessity for the isolation of rare cells in the field of immunology. For instance, techniques such as magnetic cell sorting with commercially available paramagnetic MACS-nanoparticles have facilitated the development of new approaches for research and clinical applications 22,29. Furthermore, recent research studies comparing DC generation from monocyte adherence and MACS technology methods have demonstrated a higher DC purity and viability using MACS separated monocytes 22,30.
The current study presents a comparison between two methods for the generation of human DCs from monocytes isolated from PBMCs: 1) monocyte isolation by adherence and 2) monocyte isolation by negative selection using a commercial human monocyte enrichment kit. This study provides evidence to show that the negative selection magnetic separation procedure to isolate monocytes generates the highest yield of monocytes with no significant differences in monocyte viability when compared with monocytes isolated by adherence method. In turn, after seven days, the monocytes isolated by magnetic separation differentiated into MDDCs with significantly higher proliferative capacity and higher amount of cells expressing double positive (CD11c+/CD14+) phenotype without affecting MDDC viability. Overall, the current study differs from the previous studies referenced above since it demonstrates the ability of both techniques to simultaneously generate monocytes that are capable of proliferating and differentiating into CD11c+ MDDCs (> 70%) after seven days in culture without compromising their viability. In addition, the current approach provides for the first time characterization of different CD11c/CD14 MDDCs populations by imaging flow cytometry.
In summary, since DCs play a focal role regarding research in the field of immunology, different parameters must be taken into consideration when considering how they are derived and what methods are used to isolate and culture them in vitro. Therefore, this study aims to provide insights on two different methods of monocyte isolation and how these methods differentially affect monocyte viability and yield eventually affecting dendritic cell viability, proliferation, and phenotype. These findings will contribute greatly to the field of immunology and will provide a detailed protocol of DC isolation, purification, and characterization.
Protocol
Overall human blood studies have been reviewed and approved by the Institutional Review Board (IRB) of FIU, IRB protocol approval # IRB-13-0440. Human leukopaks were purchased from the community blood bank in Miami, FL.
1. Isolation of PBMCs by Standard Density Gradient Technique
Perform a 1:1 dilution of blood with 1x-phosphate-buffered saline in a T75 flask.
Pipette 15 ml of density gradient solution into 50 ml centrifuge tubes and carefully layer (25 - 30 ml/tube) of the diluted blood over this gradient.
Centrifuge for 20 min at 1,200 x g with acceleration of 1 and deceleration of 0.
After centrifugation, collect the interface layer (white blood cells) into a new 50 ml centrifuge tube and wash cells twice in PBS (3 min at 1,080 x g). Discard supernatant each time.
Treat cells with ammonium-chloride-potassium lysing (ACK) buffer (to lyse red blood cells). Add 10 ml of buffer and incubate for 15 min at 4 °C.
Wash cells twice with PBS, centrifuge for 3 min at 1,080 x g and discard supernatant each time.
Save the pellet, which will contain the PBMCs.
Proceed with monocyte purification method of choice.
2. Monocyte Purification by Adherence Method
Prepare complete cell culture medium containing L-glutamine (300 mg/ml) and supplemented with penicillin (50 U/ml)-streptomycin (50 µg/ml) and 10% fetal bovine serum.
Allow PBMCs to adhere for approximately 2 hr by culturing them in a T75 flask at a concentration of 5 x 107 cells per 10 ml of complete medium at 37 °C and 5% CO2 in a humidified incubator.
After incubation, remove non-adherent floating cells from culture flask and gently wash adherent cells twice with PBS.
Incubate adherent cells in complete cell culture medium supplemented with 2 µl/ml of human granulocyte-macrophage colony-stimulating factor (GM-CSF) and interleukin 4 (IL-4) stored at a stock concentration of 10 µg/ml.
Change half of the medium and replenish cytokines every 48 hr.
Allow 5 - 7 days for the differentiation of monocytes into MDDCs .
3. Monocyte Purification by Magnetic Separation Method
Collect PBMCs from standard density gradient technique, pour into a 5 ml polystyrene tube and re-suspend in PBS buffer at a concentration of cells/ml.
Add human monocyte enrichment cocktail using 50 µl/ml cells, mix well and incubate at 4 °C for 10 min.
After incubation, add magnetic particles using 50 µl/ml cells, mix well and incubate at 4 °C for 5 min.
Bring cell suspension up to a total volume of 2.5 ml by adding PBS buffer, mix well by pipetting up and down 2 - 3 times.
Place the polystyrene tube without a cap into the magnetic device and set aside at RT for 2.5 min.
After incubation and in one continuous motion, pick up the magnet and pour the desired purified monocyte fraction into a clean 50 ml centrifugation tube.
Incubate adherent cells in complete cell culture medium supplemented with 2 µl/ml of human granulocyte-macrophage colony-stimulating factor (GM-CSF) and interleukin 4 (IL-4) stored at a stock concentration of 10 µg/ml.
Every 48 hr, change half of the medium, spin down at 1,080 x g for 5 min and re-suspend pellet with new media (CRPMI) and cytokines.
Allow 5 - 7 days for the differentiation of monocytes into MDDCs.
4. Trypan Blue Exclusion Viability Assay
Note: Use this technique to obtain cell yield and viability of PBMCs, monocytes, and MDDCs.
Harvest cells using standard trypsin-EDTA and perform washes of the flasks and the cell pellet using PBS. Depending on the size of the pellet, resuspend in 5 to 10 ml of PBS to dissolve the pellet.
In a micro-centrifuge tube, perform a 1:1 dilution of trypan blue reagent with diluted cell pellet.
From this mix, aliquot 10 µl into a cell counting slide and read results using an automated cell counter according to manufacturer's protocol. If an automated cell counter is not available, a similar cell count is possible using a hemocytometer or a manual counter.
5. MTT Assay
Plate cells at a concentration of 4 x 104/100 µl of complete media into each well in a 96-well plate. Allow 24 hr for cells to adjust to culture.
Incubate and perform readings at 0 (day 0), 36 (day 3) and 84 (day 7) hr respectively.
At completion of desired incubation, remove media and replace with 100 µl of PBS alone.
Prepare 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide solution (MTT) (5 mg/ml) by adding 10 ml of dimethyl sulfoxide (DMSO) to 1 g of sodium dodecyl sulfate (SDS).
Add 10 µl (5 mg/ml) of MTT solution to each well and incubate at 37 °C for 2 additional hours.
After incubation, remove supernatants which contain PBS and unconverted MTT mixture (yellow-color solution).
Add 100 µl of SDS-DMSO solution to each well and incubate for one additional hour.
Read absorbance at 540 nm using a microplate reader.
6. Cell Surface Staining Analysis by Imaging Flow Cytometry
After 7 days of differentiation from purified monocytes, dispense 1x106 cell aliquots of the monocyte-derived dendritic cells into the appropriate number of 1.5 ml tubes.
Start cell surface staining by blocking cells using 50 µl of heat inactivated (HI) human serum, incubate at 4 °C for 10 min.
After incubation, centrifuge cells at 720 x g for 5 min, discard supernatant.
To cell pellet, add respective fluorochrome-conjugated antibodies (e.g., anti-CD11c or anti-CD14) and incubate at 4 °C for 20 min protected from light. In separate tubes, prepare single fluorochrome-stained samples/ml) since they are needed for compensation.
After incubation, wash cells twice in 1 ml of PBS buffer and centrifuge each time at 720 x g for 5 min.
Keeping cells protected from light, re-suspend in PBS buffer at a concentration of cells/100 µl for flow cytometry analysis.
In order to gate on viable cells, add 1 µl of DAPI to each tube prior to acquiring the cells on the single cell imaging flow cytometry instrument according to manufacturer's instructions.
7. Statistical Analysis
Input all data in a spreadsheet.
Compare results using the student's t-test and/or one-way ANOVA as appropriate.
Consider differences to be statistically significant if p < 0.05.
Representative Results
Monocyte Yield by Magnetic Separation is Higher Compared to Monocyte Yield by Adherence Method
Data presented in Figure 1 display PBMC and monocyte cell counts by the trypan blue exclusion method at the day of isolation of PBMCs and separation of monocytes. On average, monocytes isolated by the adherence method accounted for approximately 6.2 percent of total PBMCs while monocytes isolated by magnetic separation accounted for up to 25 percent of PBMCs. Statistical analysis revealed that the percentage of monocytes yielded by magnetic separation were significantly higher (≥ 4 fold). Data are expressed as mean ± SD of at least three independent experiments. Statistical analysis using student's t-test showed a significant difference of monocyte yield when comparing both methods (p ≤ 0.0005).
Monocyte Isolation by Adherence and by Magnetic Separation Do Not Affect Monocyte Viability
After monocyte isolation by the two different techniques described above, viability assays were performed to ensure that the methods used were not cytotoxic and affecting the viability of the cells. The data presented in Figure 2 display the average of the percent of viable monocytes isolated by either adherence method or by magnetic separation. Viable cells were counted using the trypan blue exclusion method at the day of isolation. Comparing percent viability of monocytes between the two isolation methods revealed that the difference between both groups was not statistically significant. Additionally, the average of the percent of viable monocytes isolated by adherence was 91.75% ± 1.66 and the average of the percent of viable monocytes isolated by magnetic separation was 87.4% ± 2.75. These viability data were assessed from at least three independent experiments. Statistical significance was determined using student's t-test.
Cells Derived from Monocytes Isolated by Magnetic Separation Show a Significant Increase in Cell Proliferation Compared to Cells Derived from Monocytes Acquired by Adherence Method
MTT was used as a colorimetric assay to measure cell proliferation. In living cells, the yellow tetrazole color of MTT is reduced to purple formazan, which is easily measured using a spectrophotometer. The data presented in Figure 3 demonstrate that both processes of monocyte isolation lead to higher cell proliferation over a period of 7 days as measured by an increased in absorbance at day 0 (adherence method: 0.22 ± 0.02 vs. magnetic method: 0.38 ± 0.02), day 3 (adherence method: 0.28 ± 0.02, magnetic method: 0.55 ± 0.05) and day 7 (adherence method: 0.36 ± 0.01, magnetic = 0.66 ± 0.006). However, unlike the XTT assay (data not shown), the MTT assay revealed a significant increase in cell proliferation of MDDCs from monocytes isolated by magnetic separation compared to MDDCs from monocytes acquired by adherence method at day 0 (p= 0.003), day 3 (p= 0.006), and day 7 (p= 0.002). Cell proliferation was also found to be significantly different between days 0 and 3 (p= 0.003), and between days 0 and 7 (p< 0.001) within the group of MDDCs from monocytes purified through magnetic separation. Furthermore, a significant difference in cell proliferation between day 0 and day 7 in MDDCs from monocytes purified by adhesion (p= 0.008) was observed. MTT assay was conducted using MDDCs acquired from three different experiments. Statistical significance was determined using ANOVA and student's t-test, p ≤ 0.05 was considered significant, and p values are noted on the graph unless no statistical significance was found.
Characterization of MDDCs by CD11c and CD14 Cell Surface Staining
After culturing monocytes for seven days with IL-4 and GM-CSF, MDDCs obtained from monocytes isolated by adherence and magnetic isolation methods were characterized in Figure 4. The characterization revealed low single CD14+/CD11c- phenotype or purely monocytic population in both methods, adherence = 1% ± 1.0 versus magnetic = 1% ± 0.0 and high total CD11c+ phenotype , adherence = 72% ± 11 versus magnetic = 79% ± 19. However, when the total CD14+ population was analyzed further, there was a high yield of cells double positive for CD11c and CD14 in both methods with magnetic separation giving a higher number of CD11c and CD14 double positive population when compared to the adherence method, adherence = 49% ± 7 versus magnetic = 73% ± 15. Although, there was a higher yield of MDDCs with double positive phenotype via magnetic separation, the CD11c positive and CD14 negative population of MDDCs was higher in the adherence method; adherence = 23% ± 7 versus magnetic = 6% ± 7. In Figure 4B, the mean fluorescence intensity (MFI) of each individual marker were looked at in the gated population and showed high intensity of CD14 in CD14+ population, equivalent intensity of both CD11c and CD14 in double positive population and high CD11c intensity in CD11c+ and CD14- population. In summary, even though magnetic isolation gives a higher percentage of CD11c+/CD14+ cells, which may be early stage differentiated MDDCs that have not yet dropped the monocytic marker (CD14), the adherence method gives a higher percentage of CD11c+/CD14-, which may be a late stage differentiated MDDC population. In addition, DAPI staining was performed to confirm viability results and to ensure characterization was performed on live MDDCs. The percent of DAPI negative/live cells (adherence = 76 ± 7 versus magnetic = 67 ± 1) are not significantly different between the two methods confirming both methods do not affect the viability of the MDDCs after seven days in culture.
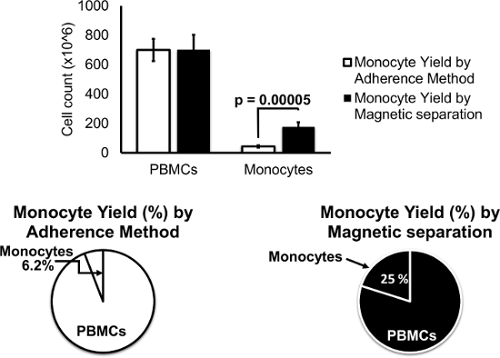 Figure 1.Monocyte Yield by Magnetic Separation is Higher Compared to Monocyte Yield by Adherence Method. About 6.2% of monocytes were isolated from PBMCs using the adherence method while ~ 25% of monocytes were obtained using the magnetic separation method. Data are expressed as mean ± SD of at least three independent experiments. Statistical analysis using student's t-test showed a significant difference of monocyte yield when comparing both methods (p = 0. 00005). Please click here to view a larger version of this figure.
Figure 1.Monocyte Yield by Magnetic Separation is Higher Compared to Monocyte Yield by Adherence Method. About 6.2% of monocytes were isolated from PBMCs using the adherence method while ~ 25% of monocytes were obtained using the magnetic separation method. Data are expressed as mean ± SD of at least three independent experiments. Statistical analysis using student's t-test showed a significant difference of monocyte yield when comparing both methods (p = 0. 00005). Please click here to view a larger version of this figure.
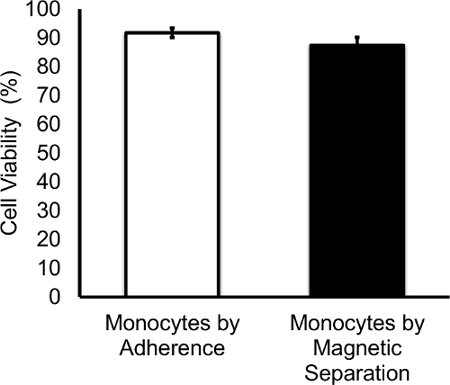 Figure 2.Monocyte Isolation by Adherence and by Magnetic Separation Do Not Affect Monocyte Viability. The average of the percent of viable monocytes isolated by adherence was 91.75% ± 1.66 and the average of the percent of viable monocytes isolated by magnetic separation was 87.4% ± 2.75. Data are expressed as mean ± SD percentage of viable cells of at least three independent experiments. No significant difference was observed in cell viability when comparing both purification methods. Please click here to view a larger version of this figure.
Figure 2.Monocyte Isolation by Adherence and by Magnetic Separation Do Not Affect Monocyte Viability. The average of the percent of viable monocytes isolated by adherence was 91.75% ± 1.66 and the average of the percent of viable monocytes isolated by magnetic separation was 87.4% ± 2.75. Data are expressed as mean ± SD percentage of viable cells of at least three independent experiments. No significant difference was observed in cell viability when comparing both purification methods. Please click here to view a larger version of this figure.
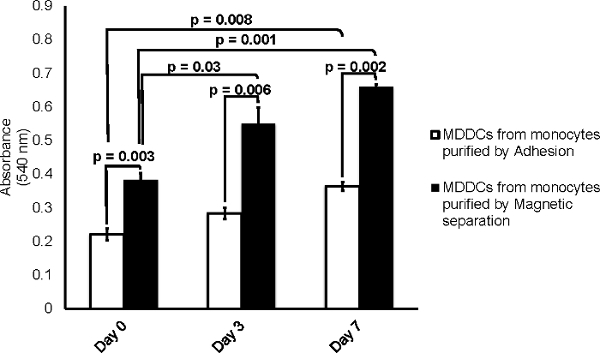 Figure 3.Cells Derived from Monocytes Isolated by Magnetic Separation Show a Significant Increase in Cell Proliferation Compared to Cells Derived from Monocytes Acquired by Adherence Method. A significant increase in absorbance was observed for both methods at both day 3 and day 7 when comparing the values against its respective controls of day 0. In addition, ANOVA and student's t-test also showed a significant difference in absorbance of MTT uptake when comparing both methods (p ≤ 0.05). Data are expressed as mean ± SD of three independent experiments. Please click here to view a larger version of this figure.
Figure 3.Cells Derived from Monocytes Isolated by Magnetic Separation Show a Significant Increase in Cell Proliferation Compared to Cells Derived from Monocytes Acquired by Adherence Method. A significant increase in absorbance was observed for both methods at both day 3 and day 7 when comparing the values against its respective controls of day 0. In addition, ANOVA and student's t-test also showed a significant difference in absorbance of MTT uptake when comparing both methods (p ≤ 0.05). Data are expressed as mean ± SD of three independent experiments. Please click here to view a larger version of this figure.
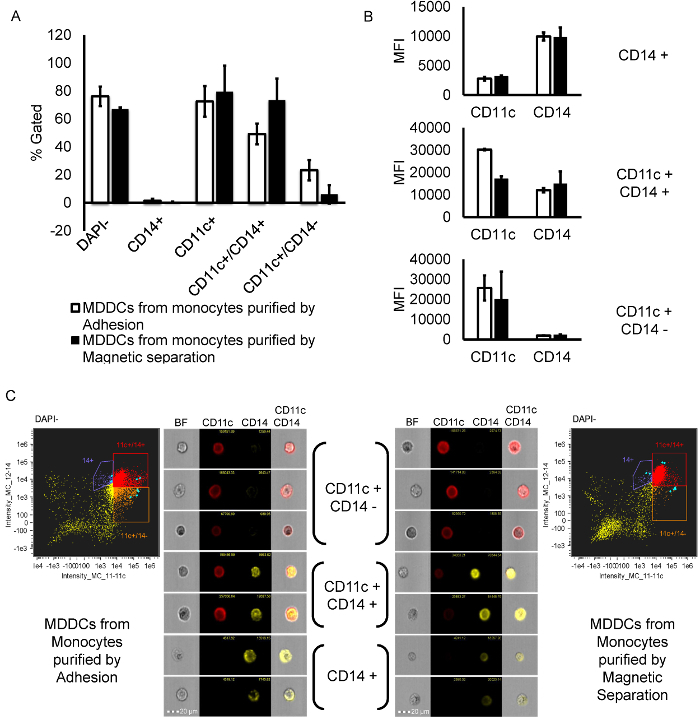 Figure 4.Characterization of MDDCs by CD11c and CD14 Cell Surface Staining.A) Bar graph representing the different populations (% gated out of single cells). First, DAPI-(alive) cells were gated from total single cells. Then, CD14+/CD11c-, CD11c+, CD11c+/CD14+ and CD11c+/CD14- were gated from DAPI- (alive) population. CD11c + population is the summation of two regions CD11c+/CD14+ and CD11c+/CD14-. B) Representative graphs showing the mean fluorescence intensity (MFI) values for both CD11c and CD14 in every subpopulation of cells. C) Representative scatter plots of cells labelled with CD11c-APC and CD14-APCcy7 gated on DAPI negative (alive) cell population for both magnetic and adherence methods. Representative image gallery of single cells (selected in blue in the scatter plots) belonging to each sub-population of cells, CD11c+/CD14- (gated in orange), CD11c+/CD14+ (gated in red), and CD14+ (gated in purple). In the image gallery of single cells, the red color represents CD11c labelled with APC and yellow color represents CD14 labelled with APCcy7. In the scatter plots, the colors chosen to gate the populations are random and do not correlate to the actual cell images. Please click here to view a larger version of this figure.
Figure 4.Characterization of MDDCs by CD11c and CD14 Cell Surface Staining.A) Bar graph representing the different populations (% gated out of single cells). First, DAPI-(alive) cells were gated from total single cells. Then, CD14+/CD11c-, CD11c+, CD11c+/CD14+ and CD11c+/CD14- were gated from DAPI- (alive) population. CD11c + population is the summation of two regions CD11c+/CD14+ and CD11c+/CD14-. B) Representative graphs showing the mean fluorescence intensity (MFI) values for both CD11c and CD14 in every subpopulation of cells. C) Representative scatter plots of cells labelled with CD11c-APC and CD14-APCcy7 gated on DAPI negative (alive) cell population for both magnetic and adherence methods. Representative image gallery of single cells (selected in blue in the scatter plots) belonging to each sub-population of cells, CD11c+/CD14- (gated in orange), CD11c+/CD14+ (gated in red), and CD14+ (gated in purple). In the image gallery of single cells, the red color represents CD11c labelled with APC and yellow color represents CD14 labelled with APCcy7. In the scatter plots, the colors chosen to gate the populations are random and do not correlate to the actual cell images. Please click here to view a larger version of this figure.
Discussion
Based on the known difficulties of isolating and generating MDDCs from human blood, the present study aimed to provide a comprehensive comparison of two well-established methods for the generation of MDDCs. The first method compared is a well-established traditional method for generating MDDCs by exploiting the ability of monocytes to adhere to glass or plastic (adherence method) 21,22,27. The adherence method is fast and cost effective, and does not require the use of complex equipment. However, some disadvantages of this process include lymphocyte contamination, low flexibility, monocyte transient manipulation 22,30,31. The second alternative method compared is the magnetic isolation of monocytes from total peripheral blood mononuclear cells (PBMCs) using a human monocyte enrichment kit 26, which is designed to isolate monocytes from PBMCs by negative selection. The advantage of this isolation method is that the unwanted cells are labeled and separated using a magnet while the target cells are freely poured off into a new tube without the need of columns and without directly targeting the cells with antibodies. Regardless of monocyte isolation method (adherent vs. magnetic separation), after both procedures, one of the critical steps within the protocol is the in vitro stimulation of monocytes with granulocyte macrophage-colony-stimulating factor (GM-CSF) and interleukin-4 (IL-4), which are required to generate MDDCs. This is a common and well established protocol to generate MDDCs from blood mononuclear cells without compromising antigen capturing and processing capacity characteristic of immature MDDCs 32. One of the major limitations of the techniques commonly used for the in vitro generation of DCs is the low yield of precursor cells such as the monocytes. As reported above, monocytes isolated by the adherence method accounted for approximately 6.2 percent of total PBMCs, while monocytes isolated by magnetic separation accounted for up to 25 percent of PBMCs. According to the literature, ranges for differential white blood cell count in normal adults for monocytes is from 2 - 10% 31 while DCs only constitute 0.1 - 1% of total blood mononuclear cells 20. Therefore, the high yield of monocytes (~ 25%) obtained after magnetic isolation could be due to impurity of cell population and lack of antibody and/or magnetic particle binding to unwanted cells.
To achieve a high yield and purity of the isolated monocytes, all the critical steps in the protocol should be carefully followed. During isolation of PBMCs from blood by standard density gradient centrifugation, there are a few critical steps. First, pipetting and carefully layering of blood over density gradient solution into 50 ml centrifuge tubes is a step that requires practice since pipetting harshly can cause the mixing of this solution and blood, which can lead to a weak PBMC layer that is hard to isolate, or it can result in the complete mixture of red blood cells with PBMCs. In this step, it is important to maintain a constant and relatively slow flow of pipetting to have a well-defined layer of blood and a distinct PBMC layer. Second, centrifugation in the next step with acceleration 1 and deceleration 0 is very critical. If these settings are not used for the first centrifugation step, it will lead to mixing of the blood with the density gradient solution erratically and will not separate the PBMCs from the rest of the blood components. Third, it is important to incubate the PBMCs with Ammonium-Chloride-Potassium (ACK) lysing buffer for no longer than the optimized time (10 - 15 min) since longer incubation with ACK can lead to reduction of viable PBMCs along with red blood cells. In addition, it is also important to carry out the PBS washes after ACK incubation. However, if the red blood cells are not lysed with the first ACK step and they still continue to appear after washes, another round of ACK lysing is highly recommended.
After isolation of white blood cells, there are a few critical steps to keep in mind when performing the adherence method to generate monocytes. First, washing off the floating lymphocytes after two hours of PBMCs incubation is very important since the presence of floating lymphocytes can cause less monocytes to adhere and can also result in lower MDDC purity. Although these washing steps are critical, excessive and harsh washing (more than recommended, i.e., twice) can cause washing off the adherent monocytes too, resulting in lower monocyte yield. Second, incubating the cells with complete medium and cytokines is critical since the cytokines are responsible for differentiation of monocytes into MDDCs. In addition,changing media and replenishing the cytokines every 48 hr is important for the differentiation and survival of the cells. While changing media, it is also crucial to save the floating MDDCs and return them back into culture along with fresh media and cytokines since there are some MDDCs that can detach from the surface during the process of differentiation. It also important to make sure that the monocytes are seeded at the recommended concentration, attached and closely spaced because they need cell to cell contact in order to grow. When performing the magnetic separation method to generate monocytes, there are a few critical steps in order to get a good yield and high purity of monocytes. First, it is critical to maintain the cell concentration recommended by the manufacturer. It is also important to not disturb the polystyrene tube when placed in the magnet, as it may lead to lesser yield and lesser purity. As presented above, this technique leads to a significantly higher monocyte yield (Figure 1) when compared to the adherence method, which may be related to the presence of other cells. Furthermore, when the cells were characterized by flow cytometry, the magnetic method shows a higher percentage of double positive cells (CD11C+/CD14+), which may correlate with the presence of other cells or may also be due to the presence of different stages of MDDC differentiation.
Another important aspect of in vitro MDDC generation regardless of yield and purity is the ability to generate viable and proliferative MDDCs. The current study provides evidence to demonstrate that both purification methods induce proliferation of MDDCs (Figure 3) as shown by the increase in cell metabolic activity during seven days in culture. In addition, the generation of MDDCs from monocytes purified by magnetic separation showed a significantly higher rate of proliferation compare to MDDCs generated from monocytes purified by adhesion (Figure 3). These findings are in accordance with previous literature demonstrating that generation of DCs from CD34+ cells is a proliferative process due to the presence of proliferative DC progenitors in human blood 27,33. However, there are controversial findings demonstrating that monocytes can also differentiate into DCs without any evidence of proliferation 34,35. It is relevant to point out that there is a lot of controversy about the in vitro generation of DCs and regarding whether DCs are generated from proliferating precursors or from differentiation of monocytes. For instance, in vitro production of DC from adherent peripheral blood cells have been shown to also enrich for progenitor cells that are capable of proliferation after exposure to GM-CSF 27 and there is also evidence demonstrating that the yield of DCs derived in the presence of GM-CSF and IL-4 cannot be expanded beyond the number of starting monocytes 33. Overall, the current study demonstrates an increased in proliferation of monocytic cells in culture that by seven days result in a total MDDCs population with > 70% CD11c+ phenotype (adherence = 72% ± 11 versus magnetic = 79% ± 19). It is relevant to point out that when further phenotypic analysis was performed, although not significant, the monocytes isolated by the magnetic separation method resulted in MDDCs with a higher double positive phenotype (adherence = 49% ± 7 CD11c+/CD14+ versus magnetic = 73% ± 15 CD11c+/CD14+) while the monocytes isolated by adhesion resulted in MDDCs with a higher single positive phenotype (adherence = 23% ± 7 CD11c+/CD14- versus magnetic = 6% ± 7 CD11c+/CD14-). MDDCs with a higher double positive phenotype (CD11c+/CD14+) may be under early stages of differentiation while MDDCs with a higher single positive phenotype may be under late stages of differentiation.
In summary, this study provides evidence to support that the negative selection magnetic separation procedure to isolate monocytes generates the highest yield of monocytes with no significant differences in monocyte viability when compared with monocytes isolated by adherence method. In turn, after seven days, the monocytes isolated by magnetic separation differentiated into MDDCs with significantly higher proliferative capacity and higher amount of cells expressing double positive (CD11c+/CD14+) phenotype without affecting MDDC viability. When comparing both methods, the findings have demonstrated the ability of both techniques to simultaneously generate monocytes that are capable of proliferating (Figure 3) and differentiating (Figure 4) into CD11c+ MDDCs after seven days in culture since both methods yielded > 70% CD11c+ MDDCs. However, further analysis looking at maturation markers may prove useful to fully characterize the functional MDDC population. In conclusion, both methods are advantageous alternatives for the isolation of CD11c+ MDDCs and provide an adequate yield for research applications and may even be useful for future clinical applications. The lab is currently focusing on the generation of MDDCs to study the effects of alcohol abuse and other substances of abuse on the innate immune system, in particularly the ability of these substances to modify MDDC phenotype and function. Therefore, the current study will provide a baseline of MDDC characterization and enhance the ability to proceed with further experimentation in order to elucidate the molecular mechanisms mediating monocyte-derived dendritic cell function in the context of substance abuse.
Disclosures
All authors disclose no competing financial interests.
Acknowledgments
This research is supported by the National Institute on Alcohol Abuse and Alcoholism, award K99/R00 AA021264. Additional lab support as part of startup package has been received from the Department of Immunology, Institute of NeuroImmune Pharmacology, Herbert Wertheim College of Medicine, and FIU- Office of Research and Economic Development. Gianna Casteleiro was supported by NIH/NIGMS R25 GM061347. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
References
- Cella M, Sallusto F, Lanzavecchia A. Origin maturation and antigen presenting function of dendritic cells. Curr Opin Imunol. 1997;9:10–16. doi: 10.1016/s0952-7915(97)80153-7. [DOI] [PubMed] [Google Scholar]
- Banchereau J, et al. Immunobiology of Dendritic Cells. Ann Rev Immunol. 2000;18:767–811. doi: 10.1146/annurev.immunol.18.1.767. [DOI] [PubMed] [Google Scholar]
- Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998;392:245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- Kaouther M, Ridha O. Dendritic Cell-Based Graft Tolerance. ISRN Pharmacol. 2011. [DOI] [PMC free article] [PubMed]
- Steinman R, Gutchinov B, Witmer M, Nussenzweig M. Dendritic cells are the principal stimulators of the primary mixed leukocyte reaction in mice. J Exp Med. 1983;157:613–627. doi: 10.1084/jem.157.2.613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nair MN, et al. RNAi-directed inhibition of DC-SIGN by dendritic cells: Prospects for HIV-1 therapy. AAPS J. 2005;7:E572–E578. doi: 10.1208/aapsj070358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agudelo M, et al. Dendritic Cells: Types, Life Cycles and Biological Functions. Nova Publishers; 2010. Chapter 10; pp. 167–177. [Google Scholar]
- Kajihara M, Takakura K, Ohkusa T, Koido S. The impact of dendritic cell-tumor fusion cells on cancer vaccines - past progress and future strategies. Immunotherapy. 2015. [DOI] [PubMed]
- Suwandi J, Toes R, Nikolic T, Roep B. Inducing tissue specific tolerance in autoimmune disease with tolerogenic dendritic cells. Clin Exp Rheumatol. 2015;33:0097–0103. [PubMed] [Google Scholar]
- Gorelik M, Frischmeyer-Guerrerio PA. Innate and adaptive dendritic cell responses to immunotherapy. Curr Opin Allergy Immunol. 2015;15:575–580. doi: 10.1097/ACI.0000000000000213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zwolak A, et al. Peripheral blood dendritic cells in alcoholic and autoimmune liver disorders. Hum Exp Toxicol. 2012;31:438–446. doi: 10.1177/0960327111426582. [DOI] [PubMed] [Google Scholar]
- Agudelo M, et al. Differential expression and functional role of cannabinoid genes in alcohol users. Drug Alcohol Depend. 2013;133:789–793. doi: 10.1016/j.drugalcdep.2013.08.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nair MP, Figueroa G, Casteleiro G, Muñoz K, Agudelo M. Alcohol Versus Cannabinoids: A Review of Their Opposite Neuro-Immunomodulatory Effects and Future Therapeutic Potentials. J Alcohol Drug Depend. 2015;3:184. doi: 10.4172/2329-6488.1000184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boukli NM, et al. Implications of ER Stress, the Unfolded Protein Response, and Pro- and Anti-Apoptotic Protein Fingerprints in Human Monocyte-Derived Dendritic Cells Treated With Alcohol. Alcohol Clin Exp Res. 2010;34:2081–2088. doi: 10.1111/j.1530-0277.2010.01304.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nair MN, Mahajan S, Sykes D, Bapardekar M, Reynolds J. Methamphetamine Modulates DC-SIGN Expression by Mature Dendritic Cells. J Neuroimmune Pharmacol. 2006;1:296–304. doi: 10.1007/s11481-006-9027-1. [DOI] [PubMed] [Google Scholar]
- Napuri J, et al. Cocaine Enhances HIV-1 Infectivity in Monocyte Derived Dendritic Cells by Suppressing microRNA-155. PLoS ONE. 2013;8:e83682. doi: 10.1371/journal.pone.0083682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nair MPN, Saiyed ZM. Effect of methamphetamine on expression of HIV coreceptors and CC-chemokines by dendritic cells. Life Sciences. 2011;88:987–994. doi: 10.1016/j.lfs.2010.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nair MPN, et al. Cocaine Modulates Dendritic Cell-Specific C Type Intercellular Adhesion Molecule-3-Grabbing Nonintegrin Expression by Dendritic Cells in HIV-1 Patients. J Immunol. 2005;174:6617–6626. doi: 10.4049/jimmunol.174.11.6617. [DOI] [PubMed] [Google Scholar]
- Reynolds JL, Mahajan SD, Sykes DE, Schwartz SA, Nair MPN. Proteomic analyses of methamphetamine (METH)-induced differential protein expression by immature dendritic cells (IDC) Biochem Biophys Acta. 2007;1774:433–442. doi: 10.1016/j.bbapap.2007.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Voorhis W, Hair L, Steinman R, Kaplan G. Human dendritic cells. Enrichment and characterization from peripheral blood. J Exp Med. 1982;155:1172–1187. doi: 10.1084/jem.155.4.1172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis G. The Mac-1 and p150,95 beta 2 integrins bind denatured proteins to mediate leukocyte cell-substrate adhesion. Exp Cell Res. 1992;200:242–252. doi: 10.1016/0014-4827(92)90170-d. [DOI] [PubMed] [Google Scholar]
- Delirezh N, Shojaeefar E. Phenotypic and functional comparison between flask adherent and magnetic activated cell sorted monocytes derived dendritic cells. Iran J Immunol. 2012;9:98–108. [PubMed] [Google Scholar]
- Lehner M, Holter W. Endotoxin-Free Purification of Monocytes for Dendritic Cell Generation via Discontinuous Density Gradient Centrifugation Based on Diluted Ficoll-Paque Plus<sup>®</sup>. Int Arch Allergy Immunol. 2002;128:73–76. doi: 10.1159/000058006. [DOI] [PubMed] [Google Scholar]
- Van Brussel I, et al. Fluorescent activated cell sorting: An effective approach to study dendritic cell subsets in human atherosclerotic plaques. J. Immunol Methods. 2015;417:76–85. doi: 10.1016/j.jim.2014.12.010. [DOI] [PubMed] [Google Scholar]
- Mucci I, et al. The methodological approach for the generation of humandendritic cells from monocytes affects the maturation state of the resultant dendritic cells. Biologicals. 2009;37:288–296. doi: 10.1016/j.biologicals.2009.05.004. [DOI] [PubMed] [Google Scholar]
- Yuan N, et al. The American Association of Immunologists (AAI) Miami Beach, FL: AAI; 2007. [Google Scholar]
- Romani N, et al. Proliferating dendritic cell progenitors in human blood. J Exp Med. 1994;180:83–93. doi: 10.1084/jem.180.1.83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett S, Breit SN. Variables in the isolation and culture of human monocytes that are of particular relevance to studies of HIV. J Leukoc Biol. 1994;56:236–240. doi: 10.1002/jlb.56.3.236. [DOI] [PubMed] [Google Scholar]
- Grützkau A, Radbruch A. Small but mighty: How the MACS®-technology based on nanosized superparamagnetic particles has helped to analyze the immune system within the last 20 years. Cytometry Part A. 2010;77A:643–647. doi: 10.1002/cyto.a.20918. [DOI] [PubMed] [Google Scholar]
- El-Sahrigy SA, Mohamed NA, Talkhan HA, Rahman AMA. Comparison between magnetic activated cell sorted monocytes and monocyte adherence techniques for in vitro generation of immature dendritic cells: an Egyptian trial. Cent Eur J Immunol. 2015;40:18–24. doi: 10.5114/ceji.2015.50828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Curry CV. Differential Blood Count. 2015. http://emedicine.medscape.com/article/2085133-overview.
- Sallusto F, Lanzavecchia A. Efficient presentation of soluble antigen by cultured human dendritic cells is maintained by granulocyte/macrophage colony-stimulating factor plus interleukin 4 and downregulated by tumor necrosis factor alpha. J Exp Med. 1994;179:1109–1118. doi: 10.1084/jem.179.4.1109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavanagh LL, Saal RJ, Grimmett KL, Thomas R. Proliferation in Monocyte-Derived Dendritic Cell Cultures Is Caused by Progenitor Cells Capable of Myeloid Differentiation. Blood. 1998;92:1598–1607. [PubMed] [Google Scholar]
- Ardeshna SM, et al. Monocyte-derived dendritic cells do not proliferate and are not susceptible to retroviral transduction. Br J Haematol. 2000;108:817–824. doi: 10.1046/j.1365-2141.2000.01956.x. [DOI] [PubMed] [Google Scholar]
- Chapuis F, et al. Differentiation of human dendritic cells from monocytes in vitro. Eur J Immunol. 1997;27:431–441. doi: 10.1002/eji.1830270213. [DOI] [PubMed] [Google Scholar]
- Zhou LJ, Tedder TF. CD14+ blood monocytes can differentiate into functionally mature CD83+ dendritic cells. Proc Natl Acad Sci. 1996;93:2588–2592. doi: 10.1073/pnas.93.6.2588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caux C, et al. B70/B7-2 is identical to CD86 and is the major functional ligand for CD28 expressed on human dendritic cells. J Exp Med. 1994;180:1841–1847. doi: 10.1084/jem.180.5.1841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caux C, et al. Activation of human dendritic cells through CD40 cross-linking. J Exp Med. 1994;180:1263–1272. doi: 10.1084/jem.180.4.1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujii Si, Liu K, Smith C, Bonito AJ, Steinman RM. The Linkage of Innate to Adaptive Immunity via Maturing Dendritic Cells In Vivo Requires CD40 Ligation in Addition to Antigen Presentation and CD80/86 Costimulation. J Exp Med. 2004;199:1607–1618. doi: 10.1084/jem.20040317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohammadi A, Mehrzad J, Mahmoudi M, Schneider M, Haghparast A. Effect of culture and maturation on human monocyte-derived dendritic cell surface markers, necrosis and antigen binding. Biotech Histochem. 2015;90:445–452. doi: 10.3109/10520295.2015.1017536. [DOI] [PubMed] [Google Scholar]


