Abstract
Influential research by Warburg and Cori in the 1920s ignited interest in how cancer cells' energy generation is different from that of normal cells. They observed high glucose consumption and large amounts of lactate excretion from cancer cells compared with normal cells, which oxidised glucose using mitochondria. It was therefore assumed that cancer cells were generating energy using glycolysis rather than mitochondrial oxidative phosphorylation, and that the mitochondria were dysfunctional. Advances in research techniques since then have shown the mitochondria in cancer cells to be functional across a range of tumour types. However, different tumour populations have different bioenergetic alterations in order to meet their high energy requirement; the Warburg effect is not consistent across all cancer types. This review will discuss the metabolic reprogramming of cancer, possible explanations for the high glucose consumption in cancer cells observed by Warburg, and suggest key experimental practices we should consider when studying the metabolism of cancer.
Keywords: cancer, glycolysis, mitochondrial respiration, Warburg effect
The Warburg effect
Despite decades of research and countless financial investments, cancer continues to elude our complete understanding and more importantly our therapies. Pivotal research in the 1920s by Warburg and Cori demonstrated that cancer avidly consumes glucose and excretes lactate [1,2]. When oxygen is present, normal cells use mitochondria to oxidise glucose, but in the absence of oxygen, glucose is converted into lactate. Otto Warburg first described in the 1920s that cancer cells utilised higher levels of glucose in the presence of oxygen with an associated increase in lactate production. The phenomenon of aerobic glycolysis, termed the Warburg effect, has been observed in a variety of other tumour types, including colorectal cancer [3], breast [4], lung [5] and glioblastoma [6,7]. From his observations, Warburg concluded that the mitochondria were dysfunctional [8,9]. The Warburg effect has been confirmed in previous studies including those of DeBerardinis et al. [10], where cells were incubated under oxygenated conditions in 10 mM C-13-labelled glucose. Cells were then perfused using 4 mM glucose prior to metabolomics analysis and even in the presence of oxygen, high levels of glycolytic metabolites were observed supporting Warburg's hypothesis. In addition, Fantin et al. [11] made the observation that inhibiting lactate dehydrogenase preventing the conversion of pyruvate to lactate reduced tumourigenicity. These data were interpreted as tumourigenicity being dependent on high levels of energy derived from glycolysis. Another study by Schulz et al. [12] showed that when mitochondrial oxidative phosphorylation is up-regulated by overexpression of frataxin, malignant growth and tumourigenic capacity are decreased. The authors suggest that rather than an increase in glycolysis being the main cause of malignant tumour growth, it is the efficiency of mitochondrial energy conversion that is the key metabolic factor.
Over the past couple of decades, advances in technology have allowed mitochondrial function to be studied in a far greater detail, and it is now realised that cancer cells have active and functional mitochondria, contrary to Warburg's theory [13,14]. In the last decade, research has shown that different tumour types (and indeed subpopulations within a tumour) have different bioenergetic alterations. This was shown as early as 1967, when Weinhouse reported that slow-growing rat hepatoma cells were oxidative, whereas the more proliferative hepatomas were glycolytic [15]. The Warburg effect is not consistent across all tumours, and the phenomenon of aerobic glycolysis has now been challenged by several groups with many cell lines reported as having mitochondrial function [16–18]. In a tumour, it is likely that a dynamic interplay exists between oxidative metabolism and glycolysis. Metabolic flexibility has now been observed in a range of cancers, including cervical, breast and pancreatic cancer (see ref. [16] and reviewed in ref. [19]). In 2004, Zu and Guppy reported that adenosine triphosphate (ATP) derived through glycolysis in various cancers and cell lines accounts for only 17% of the total ATP. In fact, the ATP generated through glycolysis was highly dependent on the cell type and could be as low as 0.31% (fibrosarcoma) or as high as 64% (hepatoma), with the remaining ATP being derived from mitochondrial oxidative phosphorylation [20]. In addition to metabolic flexibility linked to environmental conditions, there is also the influence of various cancer-associated mutations, many of which have an impact on metabolism. Mutations in the mitochondrial tricarboxylic acid cycle and respiratory chain component succinate dehydrogenase, for example, can cause phenochromocytoma and paraganglioma, where neuroendocrine tumours arise in the adrenal medulla and paraganglia in the autonomic nervous system [21,22]. Mutations in isocitrate dehydrogenase 1 are associated with adult cases of glioblastoma and appear to have a major role in the development of the tumour by a gain-of-function effect [23,24]. Understanding how cancer cell environment and mutations affect metabolism will be of fundamental importance in selecting appropriate metabolic drug combinations to impact on patients' cancer cell growth.
Cancer hallmarks and metabolic reprogramming
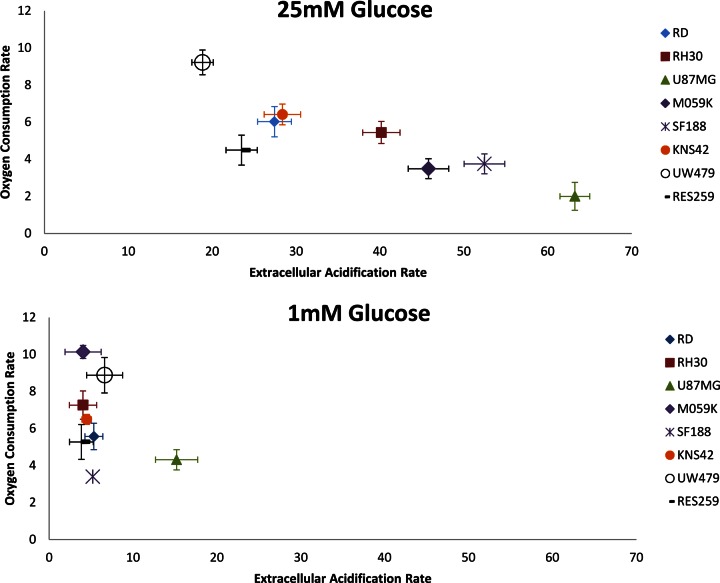
Cancer cells show complex, dynamic behaviour allowing survival even in the most unfavourable conditions of substrate and oxygen stress. Advances in technology have helped in furthering our knowledge of the underlying molecular processes underpinning cancer, but there are still many unanswered questions. In 2000, Hanahan and Weinberg published a highly cited review article identifying six cancer hallmarks [25]. These included uncontrolled proliferative signalling, resistance to apoptosis, initiating angiogenesis, acquiring replicative immortality, activating invasion and metastasis and evading growth suppressors. Over the last decade, research has increased our knowledge of cancer, and in 2011, Hanahan and Weinberg extended the list of cancer hallmarks to include metabolic reprogramming/deregulated cellular energetics as an emerging hallmark and potential cancer target [26]. Uncontrolled proliferation is one of the essential characteristics of cancer. It has been proposed that reprogramming energy metabolism is essential to fuel and maintain such behaviour [26]. The exact reasons behind the metabolic switch are not known, but likely reasons include: (i) sustaining high proliferative rates in hypoxia [27] and (ii) evading apoptosis as a result of reduced mitochondrial function [28]. Increases in glycolysis have been linked to invasiveness, with changes in glycolysis identified in several studies [29,30]. However, in all studies listed above, cancer cells were grown on cell culture media containing high levels of glucose between 10 and 25 mM. This is considerably higher than plasma glucose, which lies between 4 and 6 mM. Levels in a rapidly dividing tumour with poor vasculature are considerably lower. The same is true of studies investigating the role of hypoxia in down-regulating mitochondrial respiration and increasing glycolysis, where 25 mM glucose is used in the culture media of key publications [31–34]. The impact of high levels of glucose on the above findings is a key consideration for future studies, where it is crucial to test new drugs targeting cancer cell metabolism under physiologically relevant conditions. Metformin, for example, a drug currently being investigated as an anticancer agent in a wide range of cancers [35], has recently been shown to be more effective in enhancing chemotherapy sensitivity of oesophageal squamous cancer cells under reduced glucose conditions [36]. Although its mode of action on cancer cells in vivo is not entirely clear, mitochondrial studies suggest that metformin can directly impair complex I of the respiratory chain [37,38]. The effect observed by Yu et al. is probably due to a greater reliance of the cancer cells on mitochondrial respiration for energy production when cultured on reduced glucose conditions. Previous studies have shown that high levels of glucose in the culture media can significantly reduce levels of mitochondrial respiration, with reduced glucose conditions showing much higher rates of mitochondrial respiration, as cells use other substrates for cellular ATP production [39,40]. Similar results are shown in Figure 1, where the oxygen consumption rates (OCRs; mitochondrial respiration) and extracellular acidification rates (ECAR; glycolysis) of a range of cancer cell lines are compared under high (25 mM) and low (1 mM) glucose conditions. Under high glucose (25 mM) conditions, cancer cell lines either show the Warburg effect (low OCR and high ECAR), high rates of OCR and low ECAR or something in between high/moderate OCR with high/moderate ECAR. A finding highly relevant to the situation in vivo is that when cultured under low glucose (1 mM) conditions, all cancer lines tested show high–moderate OCR with very little ECAR (glycolysis).
Figure 1. Bioenergetic profiles of cancer cell lines RD, RH30, U87MG, M059K, SF188, KNS42, UW479 and Res259.
OCR and ECAR are plotted to quantify mitochondrial respiration and to give an indication of glycolysis rates. OCR and ECAR are expressed as changes in fluorescence life time/h/75 000 cells (n = 3). Assays were set up in black 96-well plates with pre-incubation in 1 mM and 25 mM glucose media carried out for 16 h. Oxygen and glycolysis sensing probes: MitoXpress xtra and pH xtra from Luxcel Biosciences were used to determine OCR and ECAR (http://luxcel.com/).
Positron emission tomography imaging and the reverse Warburg effect
Positron emission tomography (PET) imaging uses a radioisotope-labelled glucose tracer, 18F-fluorodeoxyglucose (18F-FDG), to identify areas of high glucose uptake/metabolism in the body. 18F-FDG is transported into cells by glucose transporters (GLUTs) and phosphorylated by hexokinase (HK) to 18F-FDG-6-phosphate (18F-FDG-6-P). Once inside the cell, 18F-FDG-6-P cannot be further metabolised through the glycolytic pathway and due to its high polar nature becomes trapped. Tumours above a certain size label strongly with this approach, and it is used to identify the presence of solid tumours and the effectiveness of treatments. Other highly metabolically active tissues, such as the brain and heart, also label strongly. It is believed that PET scans show an increased uptake of glucose in tumours due to overexpression of GLUTs [41–43]. Historically, increased glucose uptake has been associated with supporting the Warburg effect [20]. However, high glucose uptake does not automatically equate to increased glycolysis and reduced mitochondrial metabolism. An increased PET signal could be due to a general increase in glucose oxidation with increased glycolysis and mitochondrial respiration or a high demand for lipids derived from glucose. In addition, overexpression of GLUTs cannot be assumed to correlate with increased metabolic flux. A high PET signal would be obtained if glucose entered the cancer cell and was not metabolised. Many tumours are characterised by increased levels of GLUTs (particularly GLUT 1 and GLUT 3) and high expression of HK (I and II), which is associated with the increases in 18F-FDG signal compared with surrounding tissues as reviewed in ref. [44]. However, not all cancers are easily detected by PET imaging, including renal clear cell carcinoma, which is a prototypical Warburg cancer [45]. Conversely, not all 18F-FDG avid tissue is malignant; inflammation can also lead to a positive PET signal. Interestingly, Hodgkin's lymphoma responds well to PET imaging [46]. This tumour is less than 10% cancer cells, and the remaining cells are stromal/inflammation cells [47].
Although PET imaging is undeniably an extremely useful and an important clinical technique, there can be issues with the interpretation of the image. It is a common problem that the PET images can often overestimate the actual size of the tumour. A possible reason for this is that the microenvironment of the tumour is glycolytic in a phenomenon that has been called the reverse Warburg effect. First postulated by Pavlides et al. in 2009 [48], the reverse Warburg effect describes how oxidative stress in the cancer-associated fibroblasts (CAF) induces mitophagy and autophagy. The hypothesis is that hydrogen peroxide secreted from the cancer cells leads to oxidative stress in the CAF. The fibroblasts then undergo cellular catabolism, which results in a loss of mitochondrial function and ultimately a switch from aerobic metabolism to glycolysis [49]. This glycolytic switch results in increased lactate production by CAF, which is then exported into the extracellular space by monocarboxylate transporter 4 (MCT4). The lactate is ultimately taken up by the cancer cells via MCT1 and used to fuel oxidative metabolism [50,51]. In ref. [50], the authors demonstrate support for the reverse Warburg effect by culturing human breast cancer cell lines with human fibroblasts. Both MCF-7 and MDA-MB-231, when co-cultured with fibroblasts, show reduced mitochondrial function in fibroblasts with increased activity in the cancer cell lines. As in the studies of the Warburg effect and the impact of hypoxia described earlier, studies to date on the reverse Warburg effect use high concentrations of glucose 25 mM. If glucose is rapidly removed from the media by the cancer cells, this would tend to drive the cancer cells to use mitochondrial respiration (see Figure 1). Under high glucose conditions, this property of cancer cell lines will be held back by glucose inhibition of mitochondrial respiration.
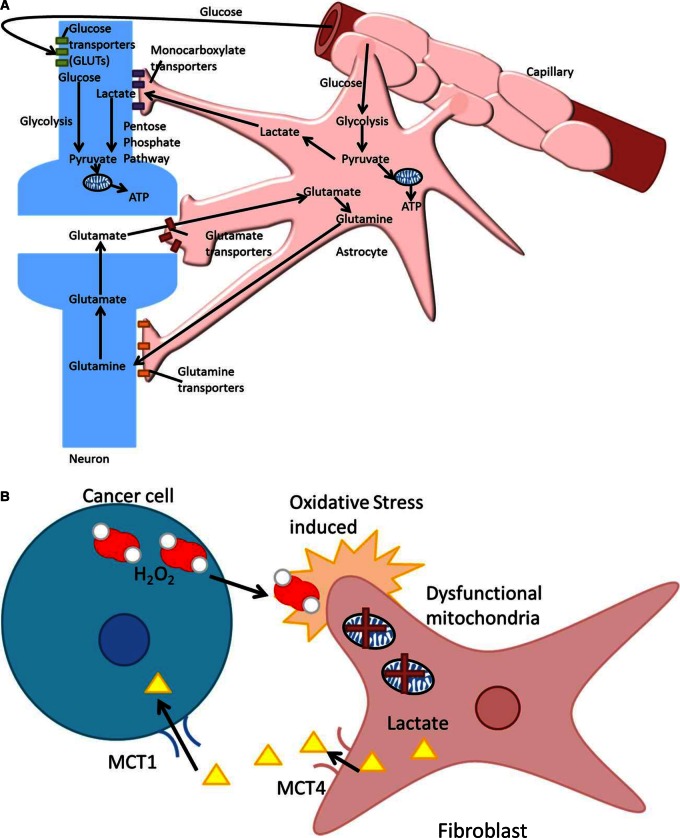
Lactate was discovered in the late 1700s and was traditionally thought of as a waste product of glycolysis. In reality, lactate is an extremely efficient fuel and also an important signalling molecule [52,53]. It is constantly turned around in our cells, regardless of oxygenation state. Lactate is a key metabolite in the body, capable of replacing glucose as an energy source. Lactate is also capable of stabilising hypoxia-inducing factor and increasing vascular endothelial growth factor expression [53]. The fibroblast–tumour metabolic coupling proposed to exist in the reverse Warburg effect is analogous to the metabolic symbiosis seen in the brain (Figure 2). The brain is a metabolically demanding organ that gives high PET signals. Its avidity for glucose was historically attributed to the neurons; however, it is the astrocytes that are glucose hungry and glycolytic. The lactate secreted by the astrocytes then fuels the neurons, which use oxidative phosphorylation to generate ATP [54]. The Warburg effect is not a universal feature of cancer, and similarly the reverse Warburg is not universal in all tumours. Yoshida, in 2015, showed that tumours expressing high levels of MCT4 do no exhibit the reverse Warburg effect [55]. The microenvironment of cancer is ever changing, and cancer cells can and do vary in their metabolic phenotype even within the same tumour mass [56]. Although hard to generalise in solid tumours with a hypoxic core, perhaps the Warburg effect most probably predominates with reduced oxygen levels driving the cells to make the most of all available glucose. The more actively proliferating cells in the periphery may perhaps use the lactate excreted in the hypoxic region and oxidise it [57], leading to a symbiotic relationship between the hypoxic and aerobic cell populations. In vitro metabolism studies are useful tools, but it only serves to hinder therapeutic translation when non-physiological glucose concentrations up to and including 25 mM glucose are used instead of a physiological concentration of 5 mM (plasma) or <5 mM (tissue).
Figure 2. The astrocyte–neuron shuttle (A) and the reverse Warburg effect (B).
(A) Glutamate is released from activated synapses and taken up by astrocytes triggering an increase in glycolysis and lactate production. The lactate can be oxidised by the neurons in response to their increased energy requirement to produce ATP. (B) nIn the proposed reverse Warburg effect, hydrogen peroxide is secreted by cancer cells leading to oxidative stress in the associated fibroblasts. The resulting loss of mitochondrial function acts as a switch from aerobic metabolism to glycolysis.
In summary, we have sought here to revisit the Warburg effect and review its significance in cancer based on recent advances in our knowledge and understanding of the complex biology underlying this disease. It is indisputable that certain features of cancer are indeed hallmarks that are essential for most types of cancer. However, looking to the future, the role of aerobic glycolysis needs further elucidation, as it is not a consistent feature in all cancers. Recent research has shown there to be a broad spectrum of bioenergetic phenotypes displayed by cancer both in vivo and in vitro, with many cancer types displaying a surprising degree of mitochondrial activity. When investigating the role of aerobic glycolysis in vitro, it is pertinent to use physiologically relevant concentrations of nutrients, in particular glucose. The excessive glucose often found in cell culture media can decrease mitochondrial respiration, allowing aerobic glycolysis to predominate. Reducing the glucose in the media to physiological levels will give a truer picture of the complex metabolic processes at work.
Abbreviations
18F-FDG, 18F-fluorodeoxyglucose; 18F-FDG-6-P, 18F-FDG-6-phosphate; ATP, adenosine triphosphate; CAF, cancer-associated fibroblasts; ECAR, extracellular acidification rate; GLUT, glucose transporter; HK, hexokinase; MCT4, monocarboxylate transporter 4; OCR, oxygen consumption rate; PET, positron emission tomography.
Funding
Michelle Potter and Emma Newport were supported by Williams fund (http://www.williamsfund.co.uk/).
Competing Interests
The Authors declare that there are no competing interests associated with the manuscript.
References
- 1.Warburg O., Wind F. and Negelein E. (1927) The metabolism of tumors in the body. J. Gen. Physiol. 8, 519–530 doi: 10.1085/jgp.8.6.519 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cori C.A. and Cori G.T. (1925) The carbohydrate metabolism of tumours. J. Biol. Chem. 65, 397–405 [Google Scholar]
- 3.Sakashita M., Aoyama, N., Minami R., Maekawa S., Kuroda K., Shirasaka D. et al. (2001) Glut1 expression in T1 and T2 stage colorectal carcinomas: its relationship to clinicopathological features. Eur. J. Cancer 37, 204–209 doi: 10.1016/S0959-8049(00)00371-3 [DOI] [PubMed] [Google Scholar]
- 4.Grover-McKay M., Walsh S.A., Seftor E.A., Thomas P.A. and Hendrix M.J.C. (1998) Role for glucose transporter 1 protein in human breast cancer. Pathol. Oncol. Res. 4, 115–120 doi: 10.1007/BF02904704 [DOI] [PubMed] [Google Scholar]
- 5.Wu M., Neilson A., Swift A.L., Moran R., Tamagnine J., Parslow D. et al. (2007) Multiparameter metabolic analysis reveals a close link between attenuated mitochondrial bioenergetic function and enhanced glycolysis dependency in human tumor cells. Am. J. Physiol. Cell Physiol. 292, C125–C136 doi: 10.1152/ajpcell.00247.2006 [DOI] [PubMed] [Google Scholar]
- 6.Lai J.-H., Jan H.-J., Liu L.-W., Lee C.-C., Wang S.-G., Hueng D.-Y. et al. (2013) Nodal regulates energy metabolism in glioma cells by inducing expression of hypoxia-inducible factor 1α. Neuro-Oncology 15, 1330–1341 doi: 10.1093/neuonc/not086 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Michelakis E.D., Sutendra G., Dromparis P., Webster L., Haromy A., Niven E. et al. (2010) Metabolic modulation of glioblastoma with dichloroacetate. Sci. Transl. Med. 2, 31ra34 doi: 10.1126/scitranslmed.3000677 [DOI] [PubMed] [Google Scholar]
- 8.Warburg O. (1956) On the origin of cancer cells. Science 123, 309–314 doi: 10.1126/science.123.3191.309 [DOI] [PubMed] [Google Scholar]
- 9.Warburg O. (1956) On respiratory impairment in cancer cells. Science 124, 269–270 PMID: [PubMed] [Google Scholar]
- 10.DeBerardinis R.J., Mancuso A., Daikhin E., Nissim I., Yudkoff M., Wehrli S. et al. (2007) Beyond aerobic glycolysis: transformed cells can engage in glutamine metabolism that exceeds the requirement for protein and nucleotide synthesis. Proc. Natl Acad. Sci. USA 104, 19345–19350 doi: 10.1073/pnas.0709747104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fantin V.R., St-Pierre J. and Leder P. (2006) Attenuation of LDH-A expression uncovers a link between glycolysis, mitochondrial physiology, and tumor maintenance. Cancer Cell 9, 425–434 doi: 10.1016/j.ccr.2006.04.023 [DOI] [PubMed] [Google Scholar]
- 12.Schulz T.J., Thierbach R., Voigt A., Drewes G., Mietzner B., Steinberg P. et al. (2006) Induction of oxidative metabolism by mitochondrial frataxin inhibits cancer growth: OTTO WARBURG REVISITED. J. Biol. Chem. 281, 977–981 doi: 10.1074/jbc.M511064200 [DOI] [PubMed] [Google Scholar]
- 13.Ju Y.S., Alexandrov L.B., Gerstung M., Martincorena I., Nik-Zainal S., Ramakrishna M. et al. (2014) Origins and functional consequences of somatic mitochondrial DNA mutations in human cancer. eLife 3, e02935 doi: 10.7554/eLife.02935 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Xu X.D., Shao S.X., Jiang H.P., Cao Y.W., Wang Y.H., Yang X.C. et al. (2015) Warburg effect or reverse Warburg effect? A review of cancer metabolism. Oncol. Res. Treat. 38, 117–122 doi: 10.1159/000375435 [DOI] [PubMed] [Google Scholar]
- 15.Weinhouse S. (1967) Hepatomas. Science 158, 542–545 doi: 10.1126/science.158.3800.542 [DOI] [PubMed] [Google Scholar]
- 16.Jose C., Bellance N. and Rossignol R. (2011) Choosing between glycolysis and oxidative phosphorylation: a tumor's dilemma? Biochim. Biophys. Acta, Bioenerg. 1807, 552–561 doi: 10.1016/j.bbabio.2010.10.012 [DOI] [PubMed] [Google Scholar]
- 17.Moreno-Sánchez R., Rodríguez-Enríquez S., Marín-Hernández A. and Saavedra E. (2007) Energy metabolism in tumor cells. FEBS J. 274, 1393–1418 doi: 10.1111/j.1742-4658.2007.05686.x [DOI] [PubMed] [Google Scholar]
- 18.Martin M., Beauvoit B., Voisin P.J., Canioni P., Guérin B. and Rigoulet M. (1998) Energetic and morphological plasticity of C6 glioma cells grown on 3-D support; effect of transient glutamine deprivation. J. Bioenerg. Biomembr. 30, 565–578 doi: 10.1023/A:1020584517588 [DOI] [PubMed] [Google Scholar]
- 19.Obre E. and Rossignol R. (2015) Emerging concepts in bioenergetics and cancer research: metabolic flexibility, coupling, symbiosis, switch, oxidative tumors, metabolic remodeling, signaling and bioenergetic therapy. Int. J. Biochem. Cell Biol. 59, 167–181 doi: 10.1016/j.biocel.2014.12.008 [DOI] [PubMed] [Google Scholar]
- 20.Zu X.L. and Guppy M. (2004) Cancer metabolism: facts, fantasy, and fiction. Biochem. Biophys. Res. Commun. 313, 459–465 doi: 10.1016/j.bbrc.2003.11.136 [DOI] [PubMed] [Google Scholar]
- 21.Astuti D., Latif F., Dallol A., Dahia P.L.M., Douglas F., George E. et al. (2001) Gene mutations in the succinate dehydrogenase subunit SDHB cause susceptibility to familial pheochromocytoma and to familial paraganglioma. Am. J. Hum. Genet. 69, 49–54 doi: 10.1086/321282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Baysal B.E., Ferrell R.E., Willett-Brozick J.E., Lawrence E.C., Myssiorek D., Bosch A. et al. (2000) Mutations in SDHD, a mitochondrial complex II gene, in hereditary paraganglioma. Science 287, 848–851 doi: 10.1126/science.287.5454.848 [DOI] [PubMed] [Google Scholar]
- 23.Dang L., White D.W., Gross S., Bennett B.D., Bittinger M.A., Driggers E.M. et al. (2009) Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature 462, 739–744 doi: 10.1038/nature08617 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yan H., Bigner D.D., Velculescu V. and Parsons D.W. (2009) Mutant metabolic enzymes are at the origin of gliomas. Cancer Res. 69, 9157–9159 doi: 10.1158/0008-5472.CAN-09-2650 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hanahan D. and Weinberg R.A. (2000) The hallmarks of cancer. Cell 100, 57–70 doi: 10.1016/S0092-8674(00)81683-9 [DOI] [PubMed] [Google Scholar]
- 26.Hanahan D. and Weinberg R.A. (2011) Hallmarks of cancer: the next generation. Cell 144, 646–674 doi: 10.1016/j.cell.2011.02.013 [DOI] [PubMed] [Google Scholar]
- 27.Gatenby R.A. and Gillies R.J. (2004) Why do cancers have high aerobic glycolysis? Nat. Rev. Cancer 4, 891–899 doi: 10.1038/nrc1478 [DOI] [PubMed] [Google Scholar]
- 28.Gogvadze V., Orrenius S. and Zhivotovsky B. (2008) Mitochondria in cancer cells: what is so special about them? Trends Cell Biol. 18, 165–173 doi: 10.1016/j.tcb.2008.01.006 [DOI] [PubMed] [Google Scholar]
- 29.Weinhouse S., Shatton J.B., Criss W.E. and Morris H.P. (1972) Molecular forms of enzymes in cancer. Biochimie 54, 685–693 doi: 10.1016/S0300-9084(72)80167-6 [DOI] [PubMed] [Google Scholar]
- 30.Bettum I.J., Gorad S.S., Barkovskaya A., Pettersen S., Moestue S.A., Vasiliauskaite K. et al. (2015) Metabolic reprogramming supports the invasive phenotype in malignant melanoma. Cancer Lett. 366, 71–83 doi: 10.1016/j.canlet.2015.06.006 [DOI] [PubMed] [Google Scholar]
- 31.Iyer N.V., Kotch L.E., Agani F., Leung S.W., Laughner E., Wenger R.H. et al. (1998) Cellular and developmental control of O2 homeostasis by hypoxia-inducible factor 1α. Genes Dev. 12, 149–162 doi: 10.1101/gad.12.2.149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kim J.-w., Tchernyshyov I., Semenza G.L. and Dang C.V. (2006) HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell Metab. 3, 177–185 doi: 10.1016/j.cmet.2006.02.002 [DOI] [PubMed] [Google Scholar]
- 33.Papandreou I., Cairns R.A., Fontana L., Lim A.L. and Denko N.C. (2006) HIF-1 mediates adaptation to hypoxia by actively downregulating mitochondrial oxygen consumption. Cell Metab. 3, 187–197 doi: 10.1016/j.cmet.2006.01.012 [DOI] [PubMed] [Google Scholar]
- 34.Seagroves T.N., Ryan H.E., Lu H., Wouters B.G., Knapp M., Thibault P. et al. (2001) Transcription factor HIF-1 is a necessary mediator of the Pasteur effect in mammalian cells. Mol. Cell. Biol. 21, 3436–3444 doi: 10.1128/MCB.21.10.3436-3444.2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Quinn B.J., Kitagawa H., Memmott R.M., Gills J.J. and Dennis P.A. (2013) Repositioning metformin for cancer prevention and treatment. Trends Endocrinol. Metab. 24, 469–480 doi: 10.1016/j.tem.2013.05.004 [DOI] [PubMed] [Google Scholar]
- 36.Yu H., Bian X., Gu D. and He X. (2016) Metformin synergistically enhances cisplatin-induced cytotoxicity in esophageal squamous cancer cells under glucose-deprivation conditions. Biomed. Res. Int. 2016, 8 doi: 10.1155/2016/8678634 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.El-Mir M.-Y., Nogueira V., Fontaine E., Averet N., Rigoulet M. and Leverve X. (2000) Dimethylbiguanide inhibits cell respiration via an indirect effect targeted on the respiratory chain complex I. J. Biol. Chem. 275, 223–228 doi: 10.1074/jbc.275.1.223 [DOI] [PubMed] [Google Scholar]
- 38.Owen M.R., Doran E. and Halestrap A.P. (2000) Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain. Biochem. J. 348, 607–614 doi: 10.1042/bj3480607 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Gohil V.M., Sheth S.A., Nilsson R., Wojtovich A.P., Lee J.H., Perocchi F. et al. (2010) Nutrient-sensitized screening for drugs that shift energy metabolism from mitochondrial respiration to glycolysis. Nat. Biotechnol. 28, 249–255 doi: 10.1038/nbt.1606 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Marroquin L.D., Hynes J., Dykens J.A., Jamieson J.D. and Will Y. (2007) Circumventing the Crabtree effect: replacing media glucose with galactose increases susceptibility of HepG2 cells to mitochondrial toxicants. Toxicol. Sci. 97, 539–547 doi: 10.1093/toxsci/kfm052 [DOI] [PubMed] [Google Scholar]
- 41.Park S.G., Lee J.H., Lee W.A. and Han K.M. (2012) Biologic correlation between glucose transporters, hexokinase-II, Ki-67 and FDG uptake in malignant melanoma. Nucl. Med. Biol. 39, 1167–1172 doi: 10.1016/j.nucmedbio.2012.07.003 [DOI] [PubMed] [Google Scholar]
- 42.Riedl C.C., Brader P., Zanzonico P., Reid V., Woo Y., Wen B. et al. (2008) Tumor hypoxia imaging in orthotopic liver tumors and peritoneal metastasis: a comparative study featuring dynamic 18F-MISO and 124I-IAZG PET in the same study cohort. Eur. J. Nucl. Med. Mol. Imaging 35, 39–46 doi: 10.1007/s00259-007-0522-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Yen T.C., See L.C., Lai C.H., Yah-Huei C.W., Ng K.K., Ma S.Y., et al. (2004) 18F-FDG uptake in squamous cell carcinoma of the cervix is correlated with glucose transporter 1 expression. J. Nucl. Med. 45, 22–29 PMID: [PubMed] [Google Scholar]
- 44.Barron C.C., Bilan P.J., Tsakiridis T. and Tsiani E. (2016) Facilitative glucose transporters: implications for cancer detection, prognosis and treatment. Metabolism 65, 124–139 doi: 10.1016/j.metabol.2015.10.007 [DOI] [PubMed] [Google Scholar]
- 45.Sotgia F., Martinez-Outschoorn U.E., Pavlides S., Howell A., Pestell R.G. and Lisanti M.P. (2011) Understanding the Warburg effect and the prognostic value of stromal caveolin-1 as a marker of a lethal tumor microenvironment. Breast Cancer Res. 13, 213 doi: 10.1186/bcr2892 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Weiler-Sagie M., Bushelev O., Epelbaum R., Dann E.J., Haim N., Avivi I. et al. (2010) 18F-FDG avidity in lymphoma readdressed: a study of 766 patients. J. Nucl. Med. 51, 25–30 doi: 10.2967/jnumed.109.067892 [DOI] [PubMed] [Google Scholar]
- 47.Aldinucci D., Gloghini A., Pinto A., De Filippi R. and Carbone A. (2010) The classical Hodgkin's lymphoma microenvironment and its role in promoting tumour growth and immune escape. J. Pathol. 221, 248–263 doi: 10.1002/path.2711 [DOI] [PubMed] [Google Scholar]
- 48.Pavlides S., Whitaker-Menezes D., Castello-Cros R., Flomenberg N., Witkiewicz A.K., Frank P.G. et al. (2009) The reverse Warburg effect: aerobic glycolysis in cancer associated fibroblasts and the tumor stroma. Cell Cycle 8, 3984–4001 doi: 10.4161/cc.8.23.10238 [DOI] [PubMed] [Google Scholar]
- 49.Wallace D.C. (2012) Mitochondria and cancer. Nat. Rev. Cancer 12, 685–698 doi: 10.1038/nrc3365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Martinez-Outschoorn U.E., Lin Z., Trimmer C., Flomenberg N., Wang C., Pavlides S. et al. (2011) Cancer cells metabolically ‘fertilize’ the tumor microenvironment with hydrogen peroxide, driving the Warburg effect: implications for PET imaging of human tumors. Cell Cycle 10, 2504–2520 doi: 10.4161/cc.10.15.16585 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Whitaker-Menezes D., Martinez-Outschoorn U.E., Lin Z., Ertel A., Flomenberg N., Witkiewicz A.K. et al. (2011) Evidence for a stromal-epithelial ‘lactate shuttle’ in human tumors. Cell Cycle 10, 1772–1783 doi: 10.4161/cc.10.11.15659 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gladden L.B. (2004) Lactate metabolism: a new paradigm for the third millennium. J. Physiol. 558, 5–30 doi: 10.1113/jphysiol.2003.058701 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Goodwin M.L., Gladden L.B., Nijsten M.W.N. and Jones K.B. (2015) Lactate and cancer: revisiting the Warburg effect in an era of lactate shuttling. Front. Nutr. 1, 27 doi: 10.3389/fnut.2014.00027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Itoh Y., Esaki T., Shimoji K., Cook M., Law M. J., Kaufman E. et al. (2003) Dichloroacetate effects on glucose and lactate oxidation by neurons and astroglia in vitro and on glucose utilization by brain in vivo. Proc. Natl Acad. Sci. USA 100, 4879–4884 doi: 10.1073/pnas.0831078100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Yoshida G.J. (2015) Metabolic reprogramming: the emerging concept and associated therapeutic strategies. J. Exp. Clin. Cancer Res. 34, 111 doi: 10.1186/s13046-015-0221-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lee M. and Yoon J.-H. (2015) Metabolic interplay between glycolysis and mitochondrial oxidation: the reverse Warburg effect and its therapeutic implication. World J. Biol. Chem. 6, 148–161 doi: 10.4331/wjbc.v6.i3.148 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Sonveaux P., Végran F., Schroeder T., Wergin M.C., Verrax J., Rabbani Z.N. et al. (2008) Targeting lactate-fueled respiration selectively kills hypoxic tumor cells in mice. J. Clin. Invest. 118, 3930–3942 doi: 10.1172/JCI36843 [DOI] [PMC free article] [PubMed] [Google Scholar]