Abstract
Neurons innervating the airways contribute to airway hyperreactivity (AHR), a hallmark feature of asthma. Several observations suggested that acid-sensing ion channels (ASICs), neuronal cation channels activated by protons, might contribute to AHR. For example, ASICs are found in vagal sensory neurons that innervate airways, and asthmatic airways can become acidic. Moreover, airway acidification activates ASIC currents and depolarizes neurons innervating airways. We found ASIC1a protein in vagal ganglia neurons, but not airway epithelium or smooth muscle. We induced AHR by sensitizing mice to ovalbumin and found that ASIC1a-/- mice failed to exhibit AHR despite a robust inflammatory response. Loss of ASIC1a also decreased bronchoalveolar lavage fluid levels of substance P, a sensory neuropeptide secreted from vagal sensory neurons that contributes to AHR. These findings suggest that ASIC1a is an important mediator of AHR and raise the possibility that inhibiting ASIC channels might be beneficial in asthma.
Introduction
Studies over the last few decades indicate that the nervous system is a critical mediator of hallmark features of asthma, including cough, mucus secretion and airway hyperreactivity (AHR) [1–6]. Several lines of evidence support this conclusion. First, greater sensory nerve innervation [7] and increased levels of sensory neuropeptides have been observed in asthmatic airways [8, 9]. Second, persistent AHR in the absence of inflammation is associated with a doubling of airway smooth muscle innervation [10]. Third, ablation of airway sensory neurons that express the transient receptor potential vanilloid 1 (TRPV1) gene reduces AHR in inflamed airways [11]. Fourth, inactivation of sensory neurons expressing Nav1.8 or blockade of Nav1.8 reduces AHR and asthmatic symptoms [12, 13]. Fifth, elimination of the transient receptor potential cation channel, subfamily A, member 1 (TRPA1), a sensory neuron receptor, decreases AHR [14, 15]. Sixth, acute vagotomy just prior to methacholine challenge prevents AHR in mice [16]. Seventh, anticholinergics and sympathomimetics are neurotransmitter antagonists and agonists that mitigate bronchoconstriction [17, 18] and AHR [19, 20].
Both nociceptors and non-nociceptors innervating the airway express Nav1.8 [21], and approximately 50% of Nav1.8-positive neurons express TRPV1 [12]. Many TRPV1-positive neurons innervating the airway also express TRPA1 [22]. Eliminating neurons that expressed TRPV1 prevented AHR in a murine model of asthma [11], without decreasing inflammation. In contrast, silencing neurons that express Nav1.8 [12] or elimination of TRPA1 prevented AHR [14] and decreased inflammation. These findings suggest that a unique subset of vagal ganglia neurons modifies airway inflammation and AHR, whereas another subset, characterized by the expression of TRPV1, modifies only the manifestation of AHR. Since loss of the TRPV1 gene itself did not protect against AHR [14], then it is unlikely that TRPV1 is the key sensor that mediates AHR. It also seems unlikely that TRPA1 is key receptor in TRPV1-expressing neurons since eliminating TRPA1 decreases inflammation [14], yet inflammation remained unchanged in mice with selective ablation of TRPV1-expressing neurons [11]. Thus, the sensor mediating AHR in TRPV1-expressing neurons remains uncertain.
In addition to expressing TRPA1[22], vagal airway sensory neurons that express TRPV1 also express acid-sensing ion channels (ASICs) [23–28]. ASICs are voltage-insensitive cation channels in the epithelial Na+ channel/degenerin superfamily that are activated by extracellular protons [29, 30]. Several studies indicate that the airway becomes acidic in asthma [31–34]. The proposed mechanisms inducing acidification are immune cell infiltration, inflammation and oxidative stress [34]. Of note, aspiration can also acidify the airways and elicit asthmatic symptoms [35, 36]. In addition, acid inhalation and airway acidification induce airway constriction [37–40]. The airway acidification elicits airway constriction through activation of TRPV1 and ASICs, and the subsequent release of sensory neuropeptides such as tachykinins [24, 27, 37–41]. Therefore, ASICs might play a key role in mediating AHR.
Rat vagal airway sensory neurons express ASIC1a, -1b, -2 and -3 mRNA [24]. Approximately 45% of rat vagal airway sensory neurons display H+-gated currents with the features of both TRPV1 and ASIC channels [23]. The transient component of those H+-gated currents has properties characteristic of ASIC currents and is blocked by the ASIC blocker amiloride [23]. The sustained component has properties of TRPV1 currents and is blocked by the TRPV1 antagonist capsazepine. The onset of acid-evoked action potentials in airway vagal sensory neurons coincides with ASIC-mediated depolarization, but not TRPV1-mediated depolarization [23]. From those studies, the authors concluded that ASIC1, -2, and -3 are responsible for the ASIC currents in rat airway vagal sensory neurons. A separate study supported that conclusion and found that ASIC currents in rat airway vagal sensory neurons were likely due to heteromers consisting of some combination of ASIC1, -2 and -3 [26]. Application of acid to guinea pig vagal nerve fibers innervating the airway also elicited currents with characteristic properties of ASIC channels [28]. Of note, some airway vagal sensory neurons in the guinea pig demonstrated H+-gated currents consistent with expression of only ASICs, and not both ASICs and TRPV1 [28]. A similar finding has been found in vagal neurons innervating the esophagus, where mRNA expression of ASIC1, 2, and 3 is found in TRPV1-negative neurons [42]. Collectively, the location and function of ASICs suggests that they might be important mediators of AHR.
To study whether ASICs channels modified AHR, we studied ASIC1a-/- mice. Previous work showed that disrupting the ASIC1a gene modifies the physiological properties of H+-gated currents in neurons and behavioral responses to acid [43, 44]. We did not study ASIC2-/- mice because they exhibit an impaired baroreceptor reflex [45], which could affect tracheal dilation [46]. We also did not study ASIC3-/- mice because they have diminished sympathetic tone [47], which could confound airway resistance measurements. We did not use a pharmacological approach because the mixed pharmacology and state-dependent activity of many ASIC channel blockers, such as amiloride [48], PcTx1[49, 50], APETx2 [51, 52], and Diclofenac [53] would make interpretation of results less clear.
Materials and Methods
Animals
Adult (8–9 weeks old) ASIC1a-/- [54] and wild-type male mice were maintained on a congenic C57BL/6J background. These studies were approved by the University of Iowa Animal Care and Use Committee.
OVA sensitization
Mice were sensitized as previously described [55, 56]. Briefly, 8–9 week-old mice were sensitized by intraperitoneal injection of 10 μg of OVA (Sigma) mixed with 1 mg of alum in 0.9% saline on days 0 and 7. Control mice received saline with 1 mg of alum on days 0 and 7. On days 14–16, mice received 1% OVA or 0.9% saline for 40 min by nebulization.
Bronchoalveolar lavage and analyses
All mice that completed FlexiVent procedures were subjected to a bronchoalveolar lavage. Lungs received three sequential 1 ml lavages of 0.9% sterile saline delivered into the airways through a cannula secured in the euthanized mouse trachea. All collected material from one mouse was pooled, spun at 500 X g, and the supernatant was removed and frozen at -80°C.
Cell count analysis and percent granulocytes was calculated once as previously described [57]. IL4, IL5, and IL13 were assayed by DuoSet ELISA kits (R&D Systems). Each ELISA was run once; duplicates of the lavage fluid per each animal were run. Substance P was assayed by ELISA (Enzo Life Sciences) and performed after cytokines were assessed. Because of variations in the amount of retrieved bronchoalveolar lavage fluid, adequate amounts of bronchoalveolar lavage fluid were only available from 6 wild-type non-sensitized mice, 6 wild-type OVA-sensitized mice, 6 ASIC1a-/- non-sensitized mice, and 7 ASIC1a-/- OVA-sensitized mice. Duplicates of the lavage fluid per each animal were run. All ELISAs were performed according to the manufacturer’s instructions.
Vagal ganglia isolation
Mice were euthanized by overdose of isofluorane inhalation. The vagal ganglia were exposed by gently pulling on the vagus nerve and then delicately cutting. They were immediately placed in RIPA buffer and stored at -80°C until protein isolation.
Western blot
Total protein from mouse brain, vagal ganglia, trachea, and lung were isolated using RIPA buffer (Sigma). Samples (40 μg) were denatured and run on a 4–15% polyacrylamide gel. Whole brain lysate from a wild-type mouse served as a control (10 μg loaded). A rabbit polyclonal antisera directed against mouse ASIC1a was provided as a kind gift from Dr. John Wemmie for western blot analysis. The western blotting of vagal ganglia was performed on two separate occasions using pooled tissues from the same three wild-type mice; similar results were observed. The airway was assessed by western blot on three separate occasions using pooled tissues from three wild-type mice; similar results were obtained.
Immunocytochemistry
Whole vagal ganglia were dissected from wild-type and ASIC1a-/- mice and fixed in 2% PFA for 15 min. Samples were then washed and permeabilized as previously described [58]. Samples were incubated in anti-ASIC1a polyclonal goat antibody (Sigma) at a ratio of 1:250 overnight at room temperature with gentle shaking. An alexa 488 secondary antibody (Life Technologies) at 1:500 was used for detection. Sections were mounted with vectashield and viewed with an Olympus Fluoview confocal microscope. Images were taken with identical settings. Post-collection adjustments were made identically. Two vagal ganglia from one wild-type and one ASIC1a-/- mouse were assessed by immunocytochemistry; similar results were achieved. The airway was assessed by immunocytochemistry on four separate occasions using independent wild-type and ASIC1a-/- mice; similar results were achieved.
Quantitative RT-PCR
RNA from total mouse airways and vagal ganglia was isolated using Qiagen Lipid Kit and treated with DNAse. RNA integrity was assessed by an Agilent Bioanalyzer. RNA was then reverse transcribed using VILO mastermix. Primers were designed for murine muc5AC as previously described [55]. Transcript abundance was assessed once. RNA was isolated from the airways of 8 wild-type non-sensitized mice, 7 wild-type OVA-sensitized mice, 7 ASIC1a-/- non-sensitized mice, and 7 ASIC1a-/- OVA-sensitized mice.
Mouse cultures
Mouse tracheal epithelial cells were cultured as previously described [59].
FlexiVent
Flexivent experiments were carried out on two separate cohorts of mice. Ketamine and xylazine sedation were used to preserve vagal reflexes [60, 61]. For each cohort, one mouse from each genotype and treatment was run on a single day. Data were collected over a period of 4 days for each cohort. FlexiVent procedures were performed as previously described [55]. Increasing doses of methacholine were aerosolized using an ultrasonic nebulizer. The aerosols were delivered for 10 sec into the inspiratory line of the ventilator. Measurements for each methacholine dose were taken at 10 sec intervals over the course of 5 mins. Two wild-type OVA-sensitized mice died during FlexiVent procedures (one from each cohort) and their FlexiVent data were not used. One wild-type non-sensitized mouse died during tracheostomy and was not included in the study.
Chemicals
Acetyl-beta-methacholine-chloride (Sigma) was dissolved in 0.9% saline for flexiVent studies.
Histopathology
Following euthanasia, the left lung was removed and placed in 10% normal buffered formalin. Lungs were removed from all animals that underwent OVA-sensitization and their respective non-sensitized controls. A single wild-type OVA-sensitized mouse lung was not collected due to user error. Samples were sectioned and stained as previously described [62]. A pathologist masked to groups performed scoring on H&E stained mouse lung sections [63]. The following scores were assigned for bronchovascular inflammation severity: 1, within normal limits; 2, focal solitary cells with uncommon aggregates; 3, multifocal nominal to moderate sized aggregates; 4, moderate to high cellularity, multifocal large cellular aggregates that may be expansive into adjacent tissues. The following scores were assigned for bronchovascular inflammation distribution: 1, within normal limits; 2, minor to localized aggregates, <33% of lung; 3, multifocal aggregates, 33–66% of lung; 4, coalescing to widespread, >66% of lung. Scoring occurred once.
Lung fixation and micro-CT scanning
Mice were euthanized with an intraperitoneal injection (Euthasol; Vibrac, Fort Worth, TX), and their lungs were surgically excised. The lungs were fixed via airway instillation at a pressure of 25 cmH2O as previously described [64]. The fixative was composed (by volume) of 55% distilled water, 25% polyethylene glycol, 10% ethyl alcohol (190 proof), and 10% formaldehyde. The lungs were removed from the fixative after 24 hr and placed in an oven at 60°C for 24 hr. While in the oven, an airway pressure of 25 cmH2O was maintained.
Lungs were imaged by micro computed tomography (micro-CT) on a Siemen’s Inveon PET/CT/SPECT scanner. Scanner settings were: 50 kVp voltage, 500 μA current, 2150 ms exposure time, 360 degrees of rotation, and 720 projections. The resulting voxels were cuboidal with 40 μm sides. Airway measurements were obtained from the micro-CT scans with Pulmonary Workstation 2.0 (VIDA Diagnostics Inc., Coralville, IA) as previously described [64]. Measurements were made perpendicular to the airway centerline and were obtained for the 35 airways highlighted by Thiesse et al. [65]. Airway measurements occurred over a period of several days, with the operator blinded to genotype.
Statistical analysis
We designed our study based upon an anticipated effect size of 1.6–1.8. These values were obtained from previous data generated from published literature [55]. Using a g-power analysis for a two-tailed “Difference between two independent means (two groups)”, the calculated number of animals required for an alpha value of 0.05 and a beta value of 0.2 was 6–8 animals per group. A two-way ANOVA was performed for studies with two or more groups and two or more conditions. When two or more groups were present, but only one condition was being tested, a one-way ANOVA was performed. Post-hoc comparisons were performed using a LSD test. For micro-CT studies, a test of normality was performed indicating data was not normally distributed. Therefore, a Mann Whitney test was used to assess differences on the combined total airway lumen area (35 segmented branches combined for each genotype). For histopathological scoring, a non-parametric ANOVA was used; when significance was found, non-parametric a Mann Whitney test between two individual groups was performed. Significance for all tests was assessed as p<0.05. Exact p values are shown in figure legends.
Results
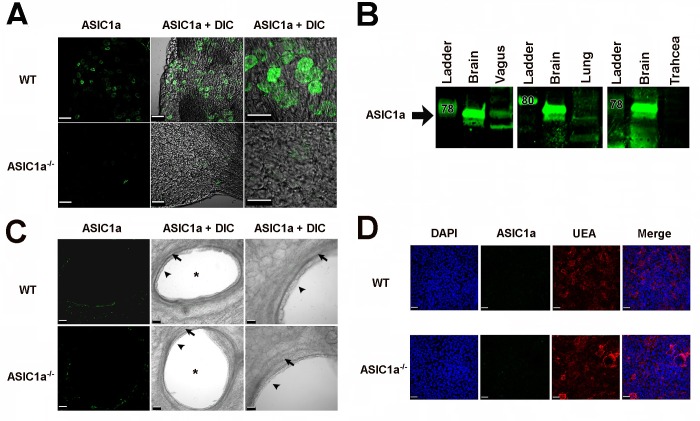
Immunofluorescence and western blotting revealed ASIC1a expression in the vagal ganglia, consistent with earlier work [24] (Fig 1A and 1B). We found negligible protein expression in the lung (Fig 1B). Compared to ASIC1a-/- tissue, tissue from wild-type mice showed no specific immunostaining in airway smooth muscle (Fig 1C) or airway epithelia (Fig 1C and 1D), even though immunostaining procedures occurred at the same time and under the same conditions as the vagal ganglia immunostaining. We made numerous attempts to identify ASIC1a immunostaining in nerve endings innervating the airway, but no specific staining was observed.
Fig 1. ASIC1a is present in vagal ganglia and expression in the airway is non-specific or negligible.
A) Images of wild-type (WT) and ASIC1a-/- mouse vagal ganglia. ASIC1a immunostaining is in green, and DIC indicates differential interference contrast images. Scale bar in left and middle panels is 60 μm; scale bar in the right panel is 40 μm. B) Western blot of ASIC1a in the vagal ganglia. Brain is a positive control. For vagal ganglia, trachea, and lung, 40 μg of protein from tissues of 3 WT mice were pooled. For brain, 10 μg of protein was loaded. C) Images of wild-type (WT) and ASIC1a-/- mouse lung cross-sections. ASIC1a immunostaining is shown in green, and DIC indicates differential interference contrast images. Scale bar in left and middle panels is 50 μm; scale bar in the right panel is 30 μm. Asterisks indicate airways; arrowheads show epithelia; arrows identify smooth muscle. D) Images of wild-type (WT) and ASIC1a-/- mouse cultured airway epithelia. DAPI staining is blue (nuclei), ASIC1a immunostaining is in green, ulex europaeus agglutinin (UEA) staining is red (mucin-producing cells), and DIC indicates differential interference contrast images. Scale bar is 30 μm. Abbreviations: WT, wild-type; ASIC, acid-sensing ion channel; DIC, differential interference contrast. UEA, ulex europaeus agglutinin; DAPI, 4',6-diamidino-2-phenylindole. Staining of airways and cultures occurred using same procedures and same conditions as the vagal ganglia.
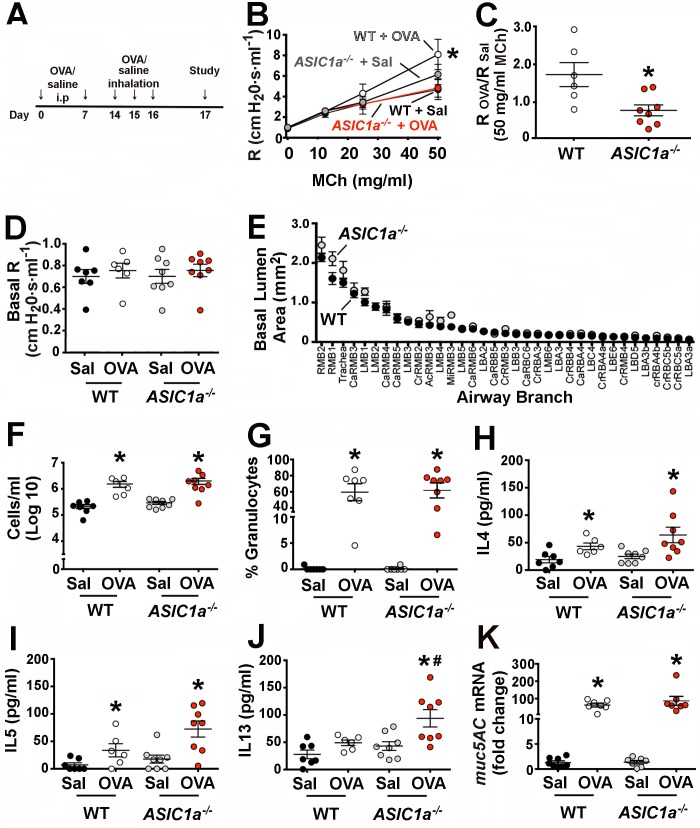
We induced AHR by using a common sensitization protocol that elicits an allergic reaction to ovalbumin (OVA) [55, 56]. Briefly, adult mice received OVA intraperitoneally on days 0 and 7 (Fig 2A). On days 14–16, mice inhaled a 1% OVA/saline solution to elicit an airway-specific reaction. On day 17, we assessed AHR by measuring airway resistance in response to nebulized methacholine; in mice that exhibit AHR, the degree of airway narrowing in response to methacholine is exaggerated and reflected as a higher airway resistance. As the concentration of methacholine increases, the degree of airway narrowing increases and thus airway resistance increases. Both non-sensitized wild-type and ASIC1a-/- mice showed similar airway resistance in response to increasing concentrations of methacholine (Fig 2B). As expected, OVA-sensitization induced AHR in wild-type mice (Fig 2B and 2C). The degree of AHR was consistent with previous reports in C57Bl/6 mice [55, 66]. In contrast, OVA-sensitization failed to elicit AHR in ASIC1a-/- mice.
Fig 2. Loss of ASIC1a prevents airway hyperreactivity.
A) Male mice (8–9 weeks-old) were sensitized by intraperitoneal injection of 10 μg of OVA (Sigma) mixed with 1 mg of alum in 0.9% saline on days 0 and 7. Control mice received saline with 1 mg of alum at day 0 and 7. On days 14–16, mice were nebulized with either 1% OVA or 0.9% saline for 40 min in a chamber. B) Airway resistance (R) was measured by flexiVent in OVA-sensitized wild-type and ASIC1a-/- mice before and following administration of increasing doses of methacholine. Data are mean±SEM. WT + Sal, n = 7 mice; WT + OVA, n = 6 mice; ASIC1a-/- + Sal, n = 8 mice; ASIC1a-/- + OVA, n = 8 mice. * indicates p = 0.043. C) Ratio of airway resistance after administration of 50 mg/ml methacholine in OVA-sensitized mice compared to non-sensitized mice. A ratio of 1 indicates that airway resistance of OVA-sensitized and non-sensitized mice was the same. * indicates p = 0.012. Ratios for ASIC1a-/- mice were not statistically different from one (p = 0.18). D) Baseline airway resistance (R) prior to administering methacholine. p = 0.89. E) Airway measurements obtained from micro-CT scans. Data are mean±SEM area for 35 different airways. Airways are shown according to size. References to abbreviations and methods are in the Methods section. WT, n = 4 mice; ASIC1a-/-, n = 5 mice. p = 0.35. F) Number of cells in bronchoalveolar lavage fluid from non-sensitized and sensitized mice. For WT + Sal vs. WT + OVA, * indicates p = 0.004; for ASIC1a-/- + Sal vs. ASIC1a-/- + OVA, * indicates p = 0.006. G) The percentage of granulocytes in bronchoalveolar lavage fluid. For WT + Sal vs. WT + OVA, * indicates p<0.0001; for ASIC1a-/- + Sal vs. ASIC1a-/- + OVA, * indicates p<0.0001. H) Levels of IL4 in bronchoalveolar lavage fluid. For WT + Sal vs. WT + OVA, * indicates p = 0.03; for ASIC1a-/- + Sal vs. ASIC1a-/- + OVA, * indicates p = 0.018. I) Levels of IL5 in bronchoalveolar lavage fluid. For WT + Sal vs. WT + OVA, * indicates p = 0.049; for ASIC1a-/- + Sal vs. ASIC1a-/- + OVA, * indicates p = 0.005. A Pearson’s normality test showed that IL5 values in the ASIC1a-/- OVA-sensitized mice do not differ from a normal distribution. (J) Levels of IL13 in bronchoalveolar lavage fluid. For WT + Sal vs. WT + OVA, p = 0.054; for ASIC1a-/- + Sal vs. ASIC1a-/- + OVA, * indicates p = 0.013; for WT + OVA vs. ASIC1a-/- + OVA, # indicates p = 0.036. A Pearson’s normality test showed that IL13 values in the ASIC1a-/- OVA-sensitized mice do not differ from a normal distribution. K) muc5AC mRNA in mouse airways. For WT + Sal vs. WT + OVA, * indicates p<0.0001; for ASIC1a-/- + Sal vs. ASIC1a-/- + OVA, * indicates p = 0.018. For all panels, individual points represent data collected from a single mouse. Bars and whiskers indicate mean±SEM. Abbreviations: OVA, ovalbumin; Sal, saline; WT, wild-type; ASIC, acid-sensing ion channel; MCh, methacholine.
One possible explanation for the lack of AHR in ASIC1a-/- mice might be that ASIC1a-/- mice contained larger airways. However, baseline airway resistance was not reduced in the ASIC1a-/- mice (Fig 2D). In addition, micro-CT studies and airway segmentation analysis revealed no differences in the airway lumen diameters between genotypes (Fig 2E). Thus, a larger airway diameter did not explain the lack of AHR in ASIC1a-/- mice.
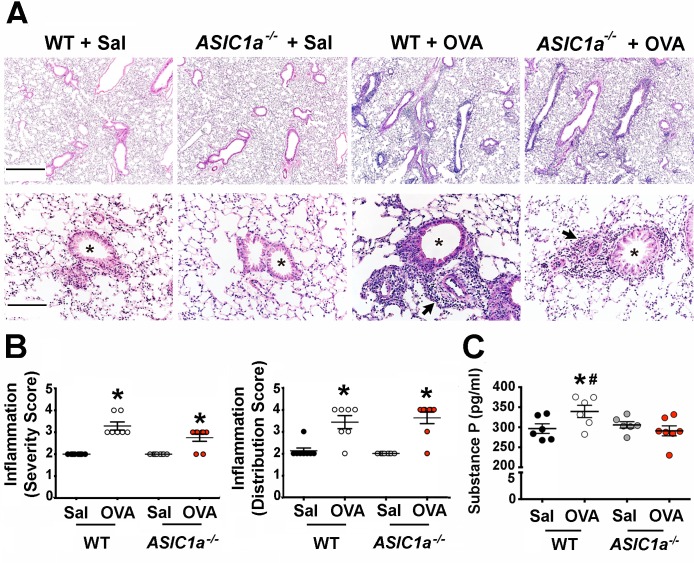
Airway inflammation is a key component of asthma and of the OVA-sensitization model [67, 68] and is characterized by the presence of granulocytes and increased levels of Th2 cytokines such as IL13, IL4 and IL5 [69–71]. Accordingly, we asked whether ASIC1a gene disruption reduced the inflammatory response to OVA. As previously reported [55], OVA-sensitization induced inflammatory cells in the bronchoalveolar lavage fluid of wild-type mice. OVA-sensitization ASIC1a-/- mice also showed increased inflammatory cells in the bronchoalveolar lavage fluid (Fig 2F and 2G). Both genotypes of mice showed a similar induction of key inflammatory cytokines in response to OVA-sensitization (Fig 2H–2J), although IL13 levels were unexpectedly statistically greater in OVA-sensitized ASIC1a-/- mice than in controls. Inflammation increases mucus production, and as such, increased mucus is a manifestation of asthma and inflammation [72]. We found that OVA-sensitization increased transcripts for muc5AC, the major murine airway mucin glycoprotein in mucus, in both wild-type and ASIC1a-/- mice (Fig 2K). This result was consistent with the presence of robust inflammation in both genotypes. We also assessed inflammation using quantitative histopathology. As expected and consistent with our other findings, OVA-sensitization increased bronchovascular inflammation in both wild-type and ASIC1a-/- mice (Fig 3A). The severity and distribution of bronchovascular inflammation did not differ between genotypes (Fig 3B). Collectively, these findings suggest that loss of ASIC1a decreases AHR without reducing the inflammatory response.
Fig 3. OVA-sensitization induces similar bronchovascular inflammation in wild-type and ASIC1a-/- mice.
A) Representative hematoxylin and eosin staining of mouse lung sections. Asterisks indicate airways; arrows indicate examples of bronchovascular inflammation. Scale bar for top panels indicates 700 μm; for lower panels bar indicates 140 μm. B) Bronchovascular inflammation score. Bronchovascular inflammation severity was scored as follows: 1, within normal limits; 2, focal solitary cells with uncommon aggregates; 3, multifocal nominal to moderate sized aggregates; 4, moderate to high cellularity, multifocal large cellular aggregates that may be expansive into adjacent tissues. The following scores were assigned for bronchovascular inflammation distribution: 1, within normal limits; 2, minor to localized aggregates, <33% of lung; 3, multifocal aggregates, 33–66% of lung; 4, aggregates coalescing to widespread, >66% of lung. For severity score: WT + Sal vs. WT + OVA, * indicates p = 0.0002; for ASIC1a-/- + Sal vs. ASIC1a-/- + OVA, * indicates p = 0.007. For distribution score: WT + Sal vs. WT + OVA, * indicates p = 0.005; for ASIC1a-/- + Sal vs. ASIC1a-/- + OVA, * indicates p = 0.001. C) Substance P measured by ELISA in the bronchoalveolar lavage fluid as a test of sensory nerve activity. For WT + Sal vs. WT + OVA, * indicates p = 0.05; for WT + OVA vs. ASIC1a-/- + OVA, # indicates p = 0.03. For panels B and C, each symbol indicates data from an individual mouse. Bars and whiskers indicate mean±SEM. Abbreviations: OVA, ovalbumin; Sal, saline; WT, wild-type; ASIC, acid-sensing ion channel.
Substance P is a tachykinin mediator of airway smooth muscle contraction in many species, including mice [73, 74] and contributes to AHR [15, 75–77]. Substance P is enriched in c-fibers and its release is thought to reflect c-fiber activity [78, 79]. Consistent with this, disrupting sensory nerve function by eliminating the TRPA1 chemosensory receptor decreases substance P and other sensory neuropeptides in the bronchoalveolar lavage fluid of mice [14]. Therefore, we used substance P as an indicator of sensory nerve function. We measured the concentration of substance P in the bronchoalveolar lavage fluid and found that loss of ASIC1a prevented the OVA-induced increase in substance P (Fig 3C). This result suggests that part of the protection against AHR might involve reduced sensory nerve function and/or decreased release of pro-contractile neuropeptides, such as substance P.
Discussion
Our data show that disrupting the ASIC1a gene prevented AHR in an OVA-sensitization model. They also emphasize the importance of the nervous system in the manifestation of AHR.
Although inflammation is a prerequisite for the development of AHR in allergic asthma [68], we found that loss of ASIC1a decreased AHR without reducing inflammation. That dissociation has also been reported by others. For example, Trankner and colleagues ablated a population of vagal sensory neurons in mice and found that it prevented AHR following OVA-sensitization, but did not reduce inflammation [11]. Crimi and colleagues found no correlation between numbers of inflammatory cells and the degree of AHR in humans [80]. Similarly, Wilder reported a dissociation of AHR from immune responses in mice [81]. OVA-sensitization in neonatal mice doubled airway smooth muscle innervation and induced persistent AHR even after inflammation had subsided [10]. Ablated TRPV1-expressing vagal sensory neurons in mice prevented AHR following OVA-sensitization without reducing airway inflammation [11]. However, another study found that ablating sensory neurons both prevented AHR and reduced inflammation [12]. In that study, ~80% of airway nociceptors were silenced, and the authors concluded that inactivating a large population of nocieptor sensory neurons might be required to dampen inflammation. Similarly, mice lacking TRPA1 also had reduced AHR and decreased inflammation [14]. These studies collectively suggest that the degree of AHR does not necessarily correlate with the degree of inflammation, and that many factors, including the initiating event and/or responsible ligands, the type of sensory receptor, and the specific neurons and other cell types expressing the receptor, ultimately determine the relationship between AHR and inflammation.
Previous studies have shown that tachykinin antagonists decrease airway inflammation [76, 77, 82]. Therefore, it is interesting to note the paradoxical increase in IL-13 in the bronchoalveolar lavage fluid of OVA-sensitized ASIC1a-/- mice despite decreased levels of substance P. A somewhat similar paradox occurs with ASIC3-/- mice in a murine model of arthritis; ASIC3-/- mice display a lack of pain despite having greater IL-6 levels [83]. While the mechanisms underlying the elevated IL-13 in the OVA-sensitized ASIC1a-/- mice are uncertain, it is possible that loss of ASIC1a prevents proton-mediated repression of IL-13 release and/or transcription. It is also possible that ASIC1a expression in dendritic cells [84] or T cells [85] contributes. However, T-cell-mediated cytokine production is not affected by loss of ASIC1a [85]. Therefore, the cell type and mechanisms underlying increased IL-13 levels in OVA-sensitized ASIC1a-/- mice remain unknown.
These findings suggest that ASICs play a key role in the bronchoconstriction associated with the OVA sensitization model and perhaps with asthma. When combined with previous studies, our results suggest that the acidosis associated with asthma [31–34] may activate ASICs on vagal sensory neurons. Consistent with that suggestion, acid depolarizes vagal nociceptive and mechanosensory airway afferents [28]. The pH reductions induce activity in these afferents, and the currents exhibit kinetics of ASIC channels independent of TRPV1. The activation of vagal neurons may initiate reflex efferent nerve activity and/or may release sensory neuropeptides, including substance P [39] and CGRP [14]. Congruent with that prediction, loss of ASIC1a reduced the substance P concentration in bronchoalveolar lavage liquid; this finding mirrors the finding that loss of the TRPA1 chemosensory receptor decreases bronchoalveolar lavage fluid levels of substance P, CGRP, and neurokinin A [14]. Multiple studies indicate that substance P mediates airway smooth muscle contraction and contributes to AHR [73, 74, 76, 77, 86], although some studies suggest that substance P can relax pre-contracted smooth muscle [87, 88]. Thus, the reduced substance P might, in part, contribute to the reduced AHR in ASIC1a-/- mice.
Even though loss of ASIC1a reduces acid-induced transient currents, we are not aware of any study suggesting that loss of ASIC1a causes a universal elimination of neural activity or universal loss of function. For example, ASIC1a-/- mice develop secondary paw hyperalgesia in response to carrageenan-induced muscle inflammation [89], and in ASIC1a-/- mice, paw withdrawal responses to heat are not affected [90]. Thus, it is predicted that the loss of ASIC1a prevents airway hyperreactivity only when the initiating stimulus involves an acidic pH or a ligand that activates ASIC1a.
Our study also has limitations. Although ASICs are present in neurons innervating airways [24, 91], we do not know the identity of the specific neuronal afferents. In addition, we cannot determine the contributions to AHR of ASIC1a in peripheral vs. central neurons. It is also possible that ASIC1a might contribute to AHR by modifying mechanosensation [92], and although we could not detect ASIC expression in airway smooth muscle, loss of ASIC1a in non-neuronal cells might also be important [93–95]. Finally, given species differences in innervation of the airway, we are uncertain about whether our observations in ASIC1a-/- mice will apply to other species.
In summary, our data identify ASIC1a as an important mediator for AHR in OVA-sensitized mice, and suggest that ASICs may play a novel role in the coupling/decoupling of airway inflammation and AHR. In addition, we report for the first time a role for ASIC1a in diminishing the release and/or induction of substance P in inflamed airways. Whether loss of ASIC1a affects concentrations of other sensory neuropeptides remains to be determined. Finally, we speculate that ASIC channel inhibitors might be beneficial in asthma and other airway diseases.
Acknowledgments
We thank Theresa Mayhew and Sarah Horgen for help in preparing the manuscript. We thank the University of Iowa Small Animal Imaging Core, Christopher Benson, Luis Vargas Buonfiglio, Anthony Fischer, Dan Grigsby, Paul Naumann, Lynda Ostedgaard, Peter Snyder, Austin Stark, Matthew Strub, and Rebecca Taugher.
Data Availability
All relevant data are within the paper.
Funding Statement
This work was in part supported by the National Institutes of Health 1K99HL119560-01A1 (LRR) and 1P01HL091842 (MJW), the America Asthma Foundation (DAS), and by the Roy J Carver Charitable Trust. DAS was funded by the Gilead Sciences Research Scholars Program in Cystic Fibrosis. MJW is an Investigator of the Howard Hughes Medical Institute.
References
- 1.Canning BJ, Chang AB, Bolser DC, Smith JA, Mazzone SB, McGarvey L, et al. Anatomy and neurophysiology of cough: CHEST Guideline and Expert Panel report. Chest. 2014;146(6):1633–48. 10.1378/chest.14-1481 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Coleridge JC, Coleridge HM. Afferent vagal C fibre innervation of the lungs and airways and its functional significance. Rev Physiol Biochem Pharmacol. 1984;99:1–110. . [DOI] [PubMed] [Google Scholar]
- 3.Ho CY, Gu Q, Lin YS, Lee LY. Sensitivity of vagal afferent endings to chemical irritants in the rat lung. Respir Physiol. 2001;127(2–3):113–24. . [DOI] [PubMed] [Google Scholar]
- 4.Lee LY, Yu J. Sensory nerves in lung and airways. Compr Physiol. 2014;4(1):287–324. 10.1002/cphy.c130020 . [DOI] [PubMed] [Google Scholar]
- 5.Barnes PJ. Asthma as an axon reflex. Lancet. 1986;1(8475):242–5. . [DOI] [PubMed] [Google Scholar]
- 6.Canning BJ. Reflex regulation of airway smooth muscle tone. J Appl Physiol (1985). 2006;101(3):971–85. 10.1152/japplphysiol.00313.2006 . [DOI] [PubMed] [Google Scholar]
- 7.Ollerenshaw SL, Jarvis D, Sullivan CE, Woolcock AJ. Substance P immunoreactive nerves in airways from asthmatics and nonasthmatics. Eur Respir J. 1991;4(6):673–82. . [PubMed] [Google Scholar]
- 8.Nieber K, Baumgarten CR, Rathsack R, Furkert J, Oehme P, Kunkel G. Substance P and beta-endorphin-like immunoreactivity in lavage fluids of subjects with and without allergic asthma. J Allergy Clin Immunol. 1992;90(4 Pt 1):646–52. . [DOI] [PubMed] [Google Scholar]
- 9.Tomaki M, Ichinose M, Miura M, Hirayama Y, Yamauchi H, Nakajima N, et al. Elevated substance P content in induced sputum from patients with asthma and patients with chronic bronchitis. Am J Respir Crit Care Med. 1995;151(3 Pt 1):613–7. 10.1164/ajrccm.151.3.7533601 . [DOI] [PubMed] [Google Scholar]
- 10.Aven L, Paez-Cortez J, Achey R, Krishnan R, Ram-Mohan S, Cruikshank WW, et al. An NT4/TrkB-dependent increase in innervation links early-life allergen exposure to persistent airway hyperreactivity. FASEB J. 2014;28(2):897–907. 10.1096/fj.13-238212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Trankner D, Hahne N, Sugino K, Hoon MA, Zuker C. Population of sensory neurons essential for asthmatic hyperreactivity of inflamed airways. Proc Natl Acad Sci U S A. 2014;111(31):11515–20. 10.1073/pnas.1411032111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Talbot S, Abdulnour RE, Burkett PR, Lee S, Cronin SJ, Pascal MA, et al. Silencing Nociceptor Neurons Reduces Allergic Airway Inflammation. Neuron. 2015;87(2):341–54. 10.1016/j.neuron.2015.06.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kocmalova M, Joskova M, Franova S, Banovcin P, Sutovska M. Airway Defense Control Mediated via Voltage-Gated Sodium Channels. Adv Exp Med Biol. 2016. 10.1007/5584_2016_244 . [DOI] [PubMed] [Google Scholar]
- 14.Caceres AI, Brackmann M, Elia MD, Bessac BF, del Camino D, D'Amours M, et al. A sensory neuronal ion channel essential for airway inflammation and hyperreactivity in asthma. Proc Natl Acad Sci U S A. 2009;106(22):9099–104. 10.1073/pnas.0900591106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Devos FC, Boonen B, Alpizar YA, Maes T, Hox V, Seys S, et al. Neuro-immune interactions in chemical-induced airway hyperreactivity. Eur Respir J. 2016. 10.1183/13993003.01778-2015 . [DOI] [PubMed] [Google Scholar]
- 16.McAlexander MA, Gavett SH, Kollarik M, Undem BJ. Vagotomy reverses established allergen-induced airway hyperreactivity to methacholine in the mouse. Respir Physiol Neurobiol. 2015;212–214:20–4. 10.1016/j.resp.2015.03.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Aviado DM. Antiasthmatic action of sympathomimetics: a review of the literature on their bronchopulmonary effects. J Clin Pharmacol J New Drugs. 1970;10(4):217–21. . [PubMed] [Google Scholar]
- 18.Prakash YS. Airway smooth muscle in airway reactivity and remodeling: what have we learned? Am J Physiol Lung Cell Mol Physiol. 2013;305(12):L912–33. 10.1152/ajplung.00259.2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Magnussen H, Nowak D, Wiebicke W. Effect of inhaled ipratropium bromide on the airway response to methacholine, histamine, and exercise in patients with mild bronchial asthma. Respiration. 1992;59(1):42–7. . [DOI] [PubMed] [Google Scholar]
- 20.Terzano C, Petroianni A, Ricci A, D'Antoni L, Allegra L. Early protective effects of tiotropium bromide in patients with airways hyperresponsiveness. Eur Rev Med Pharmacol Sci. 2004;8(6):259–64. . [PubMed] [Google Scholar]
- 21.Kwong K, Carr MJ, Gibbard A, Savage TJ, Singh K, Jing J, et al. Voltage-gated sodium channels in nociceptive versus non-nociceptive nodose vagal sensory neurons innervating guinea pig lungs. J Physiol. 2008;586(5):1321–36. 10.1113/jphysiol.2007.146365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nassenstein C, Kwong K, Taylor-Clark T, Kollarik M, Macglashan DM, Braun A, et al. Expression and function of the ion channel TRPA1 in vagal afferent nerves innervating mouse lungs. J Physiol. 2008;586(6):1595–604. 10.1113/jphysiol.2007.148379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gu Q, Lee LY. Characterization of acid signaling in rat vagal pulmonary sensory neurons. Am J Physiol Lung Cell Mol Physiol. 2006;291(1):L58–65. 10.1152/ajplung.00517.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lee LY, Gu Q, Xu F, Hong JL. Acid-sensing by airway afferent nerves. Pulm Pharmacol Ther. 2013;26(5):491–7. 10.1016/j.pupt.2013.03.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kollarik M, Ru F, Undem BJ. Acid-sensitive vagal sensory pathways and cough. Pulm Pharmacol Ther. 2007;20(4):402–11. 10.1016/j.pupt.2006.11.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Vysotskaya ZV, Moss CR 2nd, Gu Q. Differential regulation of ASICs and TRPV1 by zinc in rat bronchopulmonary sensory neurons. Lung. 2014;192(6):927–34. 10.1007/s00408-014-9634-1 . [DOI] [PubMed] [Google Scholar]
- 27.Gu Q, Lee LY. Airway irritation and cough evoked by acid: from human to ion channel. Curr Opin Pharmacol. 2011;11(3):238–47. 10.1016/j.coph.2011.04.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kollarik M, Undem BJ. Mechanisms of acid-induced activation of airway afferent nerve fibres in guinea-pig. J Physiol. 2002;543(Pt 2):591–600. 10.1113/jphysiol.2002.022848 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wemmie JA, Taugher RJ, Kreple CJ. Acid-sensing ion channels in pain and disease. Nat Rev Neurosci. 2013;14(7):461–71. 10.1038/nrn3529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Du J, Reznikov LR, Price MP, Zha XM, Lu Y, Moninger TO, et al. Protons are a neurotransmitter that regulates synaptic plasticity in the lateral amygdala. Proc Natl Acad Sci U S A. 2014;111(24):8961–6. 10.1073/pnas.1407018111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Brunetti L, Francavilla R, Tesse R, Fiermonte P, Fiore FP, Lore M, et al. Exhaled breath condensate cytokines and pH in pediatric asthma and atopic dermatitis. Allergy Asthma Proc. 2008;29(5):461–7. 10.2500/aap.2008.29.3152 . [DOI] [PubMed] [Google Scholar]
- 32.Hunt JF, Fang K, Malik R, Snyder A, Malhotra N, Platts-Mills TA, et al. Endogenous airway acidification. Implications for asthma pathophysiology. Am J Respir Crit Care Med. 2000;161(3 Pt 1):694–9. 10.1164/ajrccm.161.3.9911005 . [DOI] [PubMed] [Google Scholar]
- 33.Kodric M, Shah AN, Fabbri LM, Confalonieri M. An investigation of airway acidification in asthma using induced sputum: a study of feasibility and correlation. Am J Respir Crit Care Med. 2007;175(9):905–10. 10.1164/rccm.200607-940OC . [DOI] [PubMed] [Google Scholar]
- 34.Kostikas K, Papatheodorou G, Ganas K, Psathakis K, Panagou P, Loukides S. pH in expired breath condensate of patients with inflammatory airway diseases. Am J Respir Crit Care Med. 2002;165(10):1364–70. 10.1164/rccm.200111-068OC . [DOI] [PubMed] [Google Scholar]
- 35.Allen GB, Leclair TR, von Reyn J, Larrabee YC, Cloutier ME, Irvin CG, et al. Acid aspiration-induced airways hyperresponsiveness in mice. J Appl Physiol (1985). 2009;107(6):1763–70. 10.1152/japplphysiol.00572.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gustafsson PM, Kjellman NI, Tibbling L. Bronchial asthma and acid reflux into the distal and proximal oesophagus. Arch Dis Child. 1990;65(11):1255–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.El-Hashim AZ, Amine SA. The role of substance P and bradykinin in the cough reflex and bronchoconstriction in guinea-pigs. Eur J Pharmacol. 2005;513(1–2):125–33. 10.1016/j.ejphar.2005.02.007 . [DOI] [PubMed] [Google Scholar]
- 38.Yasumitsu R, Hirayama Y, Imai T, Miyayasu K, Hiroi J. Effects of specific tachykinin receptor antagonists on citric acid-induced cough and bronchoconstriction in unanesthetized guinea pigs. Eur J Pharmacol. 1996;300(3):215–9. . [DOI] [PubMed] [Google Scholar]
- 39.Ricciardolo FL. Mechanisms of citric acid-induced bronchoconstriction. Am J Med. 2001;111 Suppl 8A:18S–24S. . [DOI] [PubMed] [Google Scholar]
- 40.Lopes FD, Alvarenga GS, Quiles R, Dorna MB, Vieira JE, Dolhnikoff M, et al. Pulmonary responses to tracheal or esophageal acidification in guinea pigs with airway inflammation. J Appl Physiol (1985). 2002;93(3):842–7. 10.1152/japplphysiol.00013.2002 . [DOI] [PubMed] [Google Scholar]
- 41.Jia Y, Lee LY. Role of TRPV receptors in respiratory diseases. Biochim Biophys Acta. 2007;1772(8):915–27. 10.1016/j.bbadis.2007.01.013 . [DOI] [PubMed] [Google Scholar]
- 42.Dusenkova S, Ru F, Surdenikova L, Nassenstein C, Hatok J, Dusenka R, et al. The expression profile of acid-sensing ion channel (ASIC) subunits ASIC1a, ASIC1b, ASIC2a, ASIC2b, and ASIC3 in the esophageal vagal afferent nerve subtypes. Am J Physiol Gastrointest Liver Physiol. 2014;307(9):G922–30. 10.1152/ajpgi.00129.2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ziemann AE, Allen JE, Dahdaleh NS, Drebot II, Coryell MW, Wunsch AM, et al. The amygdala is a chemosensor that detects carbon dioxide and acidosis to elicit fear behavior. Cell. 2009;139(5):1012–21. 10.1016/j.cell.2009.10.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ziemann AE, Schnizler MK, Albert GW, Severson MA, Howard MA 3rd, Welsh MJ, et al. Seizure termination by acidosis depends on ASIC1a. Nat Neurosci. 2008;11(7):816–22. 10.1038/nn.2132 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lu Y, Ma X, Sabharwal R, Snitsarev V, Morgan D, Rahmouni K, et al. The ion channel ASIC2 is required for baroreceptor and autonomic control of the circulation. Neuron. 2009;64(6):885–97. 10.1016/j.neuron.2009.11.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Schultz HD, Pisarri TE, Coleridge HM, Coleridge JC. Carotid sinus baroreceptors modulate tracheal smooth muscle tension in dogs. Circ Res. 1987;60(3):337–45. . [DOI] [PubMed] [Google Scholar]
- 47.Cheng CF, Kuo TB, Chen WN, Lin CC, Chen CC. Abnormal cardiac autonomic regulation in mice lacking ASIC3. Biomed Res Int. 2014;2014:709159 10.1155/2014/709159 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Carr MJ, Gover TD, Weinreich D, Undem BJ. Inhibition of mechanical activation of guinea-pig airway afferent neurons by amiloride analogues. Br J Pharmacol. 2001;133(8):1255–62. 10.1038/sj.bjp.0704197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Chen X, Kalbacher H, Grunder S. Interaction of acid-sensing ion channel (ASIC) 1 with the tarantula toxin psalmotoxin 1 is state dependent. J Gen Physiol. 2006;127(3):267–76. 10.1085/jgp.200509409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Escoubas P, Bernard C, Lambeau G, Lazdunski M, Darbon H. Recombinant production and solution structure of PcTx1, the specific peptide inhibitor of ASIC1a proton-gated cation channels. Protein Sci. 2003;12(7):1332–43. 10.1110/ps.0307003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Diochot S, Baron A, Rash LD, Deval E, Escoubas P, Scarzello S, et al. A new sea anemone peptide, APETx2, inhibits ASIC3, a major acid-sensitive channel in sensory neurons. EMBO J. 2004;23(7):1516–25. 10.1038/sj.emboj.7600177 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Diochot S, Salinas M, Baron A, Escoubas P, Lazdunski M. Peptides inhibitors of acid-sensing ion channels. Toxicon. 2007;49(2):271–84. 10.1016/j.toxicon.2006.09.026 . [DOI] [PubMed] [Google Scholar]
- 53.Voilley N, de Weille J, Mamet J, Lazdunski M. Nonsteroid anti-inflammatory drugs inhibit both the activity and the inflammation-induced expression of acid-sensing ion channels in nociceptors. J Neurosci. 2001;21(20):8026–33. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Price MP, Lewin GR, McIlwrath SL, Cheng C, Xie J, Heppenstall PA, et al. The mammalian sodium channel BNC1 is required for normal touch sensation. Nature. 2000;407(6807):1007–11. 10.1038/35039512 . [DOI] [PubMed] [Google Scholar]
- 55.Sanders PN, Koval OM, Jaffer OA, Prasad AM, Businga TR, Scott JA, et al. CaMKII is essential for the proasthmatic effects of oxidation. Sci Transl Med. 2013;5(195):195ra97 10.1126/scitranslmed.3006135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Padrid PA, Mathur M, Li X, Herrmann K, Qin Y, Cattamanchi A, et al. CTLA4Ig inhibits airway eosinophilia and hyperresponsiveness by regulating the development of Th1/Th2 subsets in a murine model of asthma. Am J Respir Cell Mol Biol. 1998;18(4):453–62. 10.1165/ajrcmb.18.4.3055 . [DOI] [PubMed] [Google Scholar]
- 57.Stoltz DA, Meyerholz DK, Pezzulo AA, Ramachandran S, Rogan MP, Davis GJ, et al. Cystic fibrosis pigs develop lung disease and exhibit defective bacterial eradication at birth. Sci Transl Med. 2010;2(29):29ra31 10.1126/scitranslmed.3000928 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Reznikov LR, Dong Q, Chen JH, Moninger TO, Park JM, Zhang Y, et al. CFTR-deficient pigs display peripheral nervous system defects at birth. Proc Natl Acad Sci U S A. 2013;110(8):3083–8. 10.1073/pnas.1222729110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Shah VS, Meyerholz DK, Tang XX, Reznikov L, Abou Alaiwa M, Ernst SE, et al. Airway acidification initiates host defense abnormalities in cystic fibrosis mice. Science. 2016;351(6272):503–7. 10.1126/science.aad5589 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Antonaccio MJ, Robson RD, Kerwin L. Evidence for increased vagal tone and enhancement of baroreceptor reflex activity after xylazine (2-(2,6-dimethylphenylamino)-4-H-5,6-dihydro-1,3-thiazine) in anesthestized dogs. Eur J Pharmacol. 1973;23(3):311–6. . [DOI] [PubMed] [Google Scholar]
- 61.Hoka S, Takeshita A, Sasaki T, Yoshitake J. Preservation of baroreflex control of vascular resistance under ketamine anesthesia in rats. J Anesth. 1988;2(2):207–12. 10.1007/s0054080020207 . [DOI] [PubMed] [Google Scholar]
- 62.Meyerholz DK, Stoltz DA, Pezzulo AA, Welsh MJ. Pathology of gastrointestinal organs in a porcine model of cystic fibrosis. Am J Pathol. 2010;176(3):1377–89. 10.2353/ajpath.2010.090849 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Gibson-Corley KN, Olivier AK, Meyerholz DK. Principles for valid histopathologic scoring in research. Vet Pathol. 2013;50(6):1007–15. 10.1177/0300985813485099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Adam RJ, Michalski AS, Bauer C, Abou Alaiwa MH, Gross TJ, Awadalla MS, et al. Air trapping and airflow obstruction in newborn cystic fibrosis piglets. Am J Respir Crit Care Med. 2013;188(12):1434–41. 10.1164/rccm.201307-1268OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Thiesse J, Namati E, Sieren JC, Smith AR, Reinhardt JM, Hoffman EA, et al. Lung structure phenotype variation in inbred mouse strains revealed through in vivo micro-CT imaging. J Appl Physiol (1985). 2010;109(6):1960–8. 10.1152/japplphysiol.01322.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Johnson JR, Folestad E, Rowley JE, Noll EM, Walker SA, Lloyd CM, et al. Pericytes contribute to airway remodeling in a mouse model of chronic allergic asthma. Am J Physiol Lung Cell Mol Physiol. 2015;308(7):L658–71. 10.1152/ajplung.00286.2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Featherstone RL, Hutson PA, Holgate ST, Church MK. Active sensitization of guinea-pig airways in vivo enhances in vivo and in vitro responsiveness. Eur Respir J. 1988;1(9):839–45. . [PubMed] [Google Scholar]
- 68.Wills-Karp M, Luyimbazi J, Xu X, Schofield B, Neben TY, Karp CL, et al. Interleukin-13: central mediator of allergic asthma. Science. 1998;282(5397):2258–61. . [DOI] [PubMed] [Google Scholar]
- 69.Kearley J, McMillan SJ, Lloyd CM. Th2-driven, allergen-induced airway inflammation is reduced after treatment with anti-Tim-3 antibody in vivo. J Exp Med. 2007;204(6):1289–94. 10.1084/jem.20062093 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Robinson DS, Hamid Q, Ying S, Tsicopoulos A, Barkans J, Bentley AM, et al. Predominant TH2-like bronchoalveolar T-lymphocyte population in atopic asthma. N Engl J Med. 1992;326(5):298–304. 10.1056/NEJM199201303260504 . [DOI] [PubMed] [Google Scholar]
- 71.Cohn L, Homer RJ, Niu N, Bottomly K. T helper 1 cells and interferon gamma regulate allergic airway inflammation and mucus production. J Exp Med. 1999;190(9):1309–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Erle DJ, Sheppard D. The cell biology of asthma. J Cell Biol. 2014;205(5):621–31. 10.1083/jcb.201401050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Xiao L, Wu ZX. Substance p regulates environmental tobacco smoke-enhanced tracheal smooth muscle responsiveness in mice. J Allergy (Cairo). 2012;2012:423612 10.1155/2012/423612 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Tournoy KG, De Swert KO, Leclere PG, Lefebvre RA, Pauwels RA, Joos GF. Modulatory role of tachykinin NK1 receptor in cholinergic contraction of mouse trachea. Eur Respir J. 2003;21(1):3–10. . [DOI] [PubMed] [Google Scholar]
- 75.Murlas CG, Lang Z, Chodimella V. Dexamethasone reduces tachykinin but not ACh airway hyperreactivity after O3. Lung. 1993;171(2):109–21. . [DOI] [PubMed] [Google Scholar]
- 76.Schuiling M, Zuidhof AB, Zaagsma J, Meurs H. Role of tachykinin NK1 and NK2 receptors in allergen-induced early and late asthmatic reactions, airway hyperresponsiveness, and airway inflammation in conscious, unrestrained guinea pigs. Clin Exp Allergy. 1999;29 Suppl 2:48–52. . [DOI] [PubMed] [Google Scholar]
- 77.Schuiling M, Zuidhof AB, Zaagsma J, Meurs H. Involvement of tachykinin NK1 receptor in the development of allergen-induced airway hyperreactivity and airway inflammation in conscious, unrestrained guinea pigs. Am J Respir Crit Care Med. 1999;159(2):423–30. 10.1164/ajrccm.159.2.9804125 . [DOI] [PubMed] [Google Scholar]
- 78.Mazzone SB. An overview of the sensory receptors regulating cough. Cough. 2005;1:2 10.1186/1745-9974-1-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Bessac BF, Jordt SE. Breathtaking TRP channels: TRPA1 and TRPV1 in airway chemosensation and reflex control. Physiology (Bethesda). 2008;23:360–70. 10.1152/physiol.00026.2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Crimi E, Spanevello A, Neri M, Ind PW, Rossi GA, Brusasco V. Dissociation between airway inflammation and airway hyperresponsiveness in allergic asthma. Am J Respir Crit Care Med. 1998;157(1):4–9. 10.1164/ajrccm.157.1.9703002 . [DOI] [PubMed] [Google Scholar]
- 81.Wilder JA, Collie DD, Wilson BS, Bice DE, Lyons CR, Lipscomb MF. Dissociation of airway hyperresponsiveness from immunoglobulin E and airway eosinophilia in a murine model of allergic asthma. Am J Respir Cell Mol Biol. 1999;20(6):1326–34. 10.1165/ajrcmb.20.6.3561 . [DOI] [PubMed] [Google Scholar]
- 82.Nenan S, Germain N, Lagente V, Emonds-Alt X, Advenier C, Boichot E. Inhibition of inflammatory cell recruitment by the tachykinin NK(3)-receptor antagonist, SR 142801, in a murine model of asthma. Eur J Pharmacol. 2001;421(3):201–5. . [DOI] [PubMed] [Google Scholar]
- 83.Sluka KA, Rasmussen LA, Edgar MM, O'Donnell JM, Walder RY, Kolker SJ, et al. Acid-sensing ion channel 3 deficiency increases inflammation but decreases pain behavior in murine arthritis. Arthritis Rheum. 2013;65(5):1194–202. 10.1002/art.37862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Tong J, Wu WN, Kong X, Wu PF, Tian L, Du W, et al. Acid-sensing ion channels contribute to the effect of acidosis on the function of dendritic cells. J Immunol. 2011;186(6):3686–92. 10.4049/jimmunol.1001346 . [DOI] [PubMed] [Google Scholar]
- 85.Friese MA, Craner MJ, Etzensperger R, Vergo S, Wemmie JA, Welsh MJ, et al. Acid-sensing ion channel-1 contributes to axonal degeneration in autoimmune inflammation of the central nervous system. Nat Med. 2007;13(12):1483–9. 10.1038/nm1668 . [DOI] [PubMed] [Google Scholar]
- 86.Lundberg JM, Martling CR, Saria A. Substance P and capsaicin-induced contraction of human bronchi. Acta Physiol Scand. 1983;119(1):49–53. 10.1111/j.1748-1716.1983.tb07304.x . [DOI] [PubMed] [Google Scholar]
- 87.Mhanna MJ, Dreshaj IA, Haxhiu MA, Martin RJ. Mechanism for substance P-induced relaxation of precontracted airway smooth muscle during development. Am J Physiol. 1999;276(1 Pt 1):L51–6. . [DOI] [PubMed] [Google Scholar]
- 88.Manzini S. Bronchodilatation by tachykinins and capsaicin in the mouse main bronchus. Br J Pharmacol. 1992;105(4):968–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Walder RY, Rasmussen LA, Rainier JD, Light AR, Wemmie JA, Sluka KA. ASIC1 and ASIC3 play different roles in the development of Hyperalgesia after inflammatory muscle injury. J Pain. 2010;11(3):210–8. 10.1016/j.jpain.2009.07.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Page AJ, Brierley SM, Martin CM, Martinez-Salgado C, Wemmie JA, Brennan TJ, et al. The ion channel ASIC1 contributes to visceral but not cutaneous mechanoreceptor function. Gastroenterology. 2004;127(6):1739–47. . [DOI] [PubMed] [Google Scholar]
- 91.Canning B KM, Meeker S, Nanako M, Undem BJ. Acid evoked coughing in anesthetized guinea pigs depends upon Acid-Sensing Ion Channel (ASIC)-dependent activation of capsaicin-insensitive vagal afferent neurons. FASEB: The FASEB Journal; 2013. [Google Scholar]
- 92.Kang S, Jang JH, Price MP, Gautam M, Benson CJ, Gong H, et al. Simultaneous disruption of mouse ASIC1a, ASIC2 and ASIC3 genes enhances cutaneous mechanosensitivity. PLoS One. 2012;7(4):e35225 10.1371/journal.pone.0035225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Nitta CH, Osmond DA, Herbert LM, Beasley BF, Resta TC, Walker BR, et al. Role of ASIC1 in the development of chronic hypoxia-induced pulmonary hypertension. Am J Physiol Heart Circ Physiol. 2014;306(1):H41–52. 10.1152/ajpheart.00269.2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Jernigan NL, Paffett ML, Walker BR, Resta TC. ASIC1 contributes to pulmonary vascular smooth muscle store-operated Ca(2+) entry. Am J Physiol Lung Cell Mol Physiol. 2009;297(2):L271–85. 10.1152/ajplung.00020.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Faisy C, Planquette B, Naline E, Risse PA, Frossard N, Fagon JY, et al. Acid-induced modulation of airway basal tone and contractility: role of acid-sensing ion channels (ASICs) and TRPV1 receptor. Life Sci. 2007;81(13):1094–102. 10.1016/j.lfs.2007.08.026 . [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All relevant data are within the paper.