Abstract
Renal and pancreatic cysts and tumors are the most common visceral manifestations of von Hippel-Lindau (VHL) disease, a heritable multisystem cancer syndrome characterized by development of a variety of malignant and benign tumors. We report a case of a VHL patient with multiple renal cystic and complex cystic/solid lesions. The patient underwent 68Ga-DOTA-TATE-PET/CT showing intensely increased activity by a solid lesion which demonstrated enhancement on both CT and MRI scans, raising high suspicion for malignancy. The presented case indicates the application of SSTR-imaging using 68Ga-DOTA-conjugated-peptides in VHL patients, and emphasizes the need for cautious interpretation of renal parenchyma 68Ga-DOTA-TATE activity.
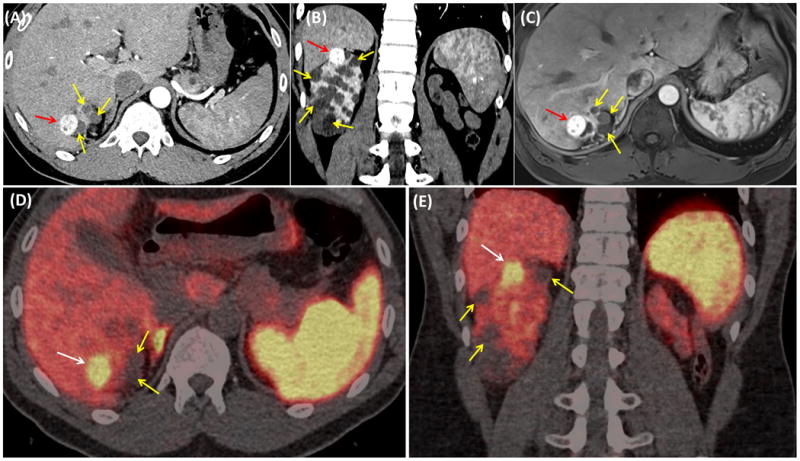
We report on a 45-year old male with known history of von Hippel-Lindau (VHL) disease affecting several organs including cerebellar and spinal hemangioblastomas, pancreatic neuroendocrine tumors (NETs) and kidney lesions leading to total left nephrectomy. On annual follow up the patient underwent i.v.-contrast CT of the abdomen which showed multiple cystic and complex solid/cystic lesions throughout the right kidney (Fig.1A, 1B: axial and coronal post-contrast CT images, yellow arrows) with a solid enhancing (average H.U.:158) mass seen on the superior pole and measuring 2,4cm×2cm (Fig1A, 1B: red arrow). Subsequently MRI scan confirmed the right kidney lesions (Fig1C: yellow arrows) including the enhancing solid mass in the upper pole (Fig1C: axial post-contrast T1 image red arrow). The patient underwent whole-body PET/CT scan using 68Ga-DOTA-TATE which showed multiple photopenic areas in the right kidney (Fig 1D, 1E: axial and coronal fused PET/CT images, yellow arrows), corresponding to cystic lesions. However, the upper pole solid mass which was enhancing on both CT and MRI scans, demonstrated intensely increased (SUVmax: 26,3) 68Ga-DOTA-TATE activity (Fig 1D, 1E: white arrow), suggesting cell-surface over-expression of somatostatin receptors (SSTRs) and raising high suspicion of malignancy. Unfortunately, the patient refused to pursue with surgical treatment and nephron-sparing resection of this mass aiming to preserve function of the solitary right kidney was not performed. VHL is an autosomal dominantly inherited familial cancer syndrome characterized by development of a variety of benign and malignant tumors, which is associated with a germline mutation of the VHL tumor suppressor gene on the short arm of chromosome [1, 2]. VHL's spectrum of manifestations is broad with 40 different lesions in 14 different organs [3]. Of the most common visceral VHL manifestations are renal cysts and renal cell carcinoma (RCC), which account for the most common causes of death in VHL patients [3,4]. Kidney malignancies often remain asymptomatic for long intervals while macroscopically normal-appearing renal tissue can contain microscopic tumoral foci [5]. Thus, serial imaging with different modalities is critical for early detection of any malignant transformation of seemingly benign cysts, enabling less morbid operations such as nephron-sparing resection. Since many VHL manifestations such as hemangioblastomas or pancreatic NETs are known to over-express SSTRs they can be effectively targeted and monitored with radiolabeled SST-analogues [6, 7]. The introduction of 68Ga-DOTA-conjugated-peptides (SST-analogues) into clinical practice enabled SSTR-imaging with PET, and evolves as the imaging standard of reference for the detection and characterization of SSTR-positive tumors, with high potential in VHL-disease [8, 9]. In the current case 68Ga-DOTA-TATE-PET/CT confirmed SSTR-overexpression by the enhancing solid renal lesion, raising high suspicion of RCC or pancreatic NET metastases. The differential diagnosis should also include primary renal carcinoid tumors, which are extremely rare kidney tumors exhibiting SSTRs cell-surface over-expression [10, 11]. Considering the wide spectrum of VHL manifestations, the presented data strongly supports the application of SSTR-imaging with 68Ga-DOTA-conjugated-peptides in early diagnosis of various SSTR-positive lesions, as well as surveillance of asymptomatic gene carriers and highlights the need for meticulous interpretation of renal parenchyma 68Ga-DOTATATE activity, where normal tracer uptake may obscure a suspicious lesion.
Figure 1.
Footnotes
Disclosure: All authors have nothing to disclose
References
- 1.Lonser RR, Glenn GM, Walther M, et al. Von Hippel-Lindau disease. Lancet. 2003;361:2059–2067. doi: 10.1016/S0140-6736(03)13643-4. [DOI] [PubMed] [Google Scholar]
- 2.Choyke PL, Glenn GM, Walther MM, et al. Von Hippel Lindau disease: genetic, clinical, and imaging features. Radiology. 1995;194:629–642. doi: 10.1148/radiology.194.3.7862955. [DOI] [PubMed] [Google Scholar]
- 3.Leung RS, Biswas SV, Duncan M, et al. Imaging features of von Hippel-Lindau disease. Radiographics. 2008;28:65–67. doi: 10.1148/rg.281075052. [DOI] [PubMed] [Google Scholar]
- 4.Wood CG, Stromberg LJ, Harmath CB, et al. CT and MR imaging for evaluation of cystic renal lesions and diseases. Radiographics. 2015;35:125–141. doi: 10.1148/rg.351130016. [DOI] [PubMed] [Google Scholar]
- 5.Meister M, Choyke P, Anderson C, et al. Radiological evaluation, management, and surveillance of renal masses in Von Hippel–Lindau disease. Clin Radiol. 2009;64:589–600. doi: 10.1016/j.crad.2008.10.010. [DOI] [PubMed] [Google Scholar]
- 6.Ambrosini V, Campana D, Allegri V, et al. 68Ga-DOTANOC PET/CT detects somatostatin receptors expression in von Hippel-Lindau cerebellar disease. Clin. Nucl. Med. 2011;36:64–65. doi: 10.1097/RLU.0b013e3181fef14a. [DOI] [PubMed] [Google Scholar]
- 7.Oh JR, Kulkarni H, Carreras C, et al. 68Ga Somatostatin Receptor PET/CT in von Hippel-Lindau Disease. Nucl Med Mol Imaging. 2012;46:129–133. doi: 10.1007/s13139-012-0133-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hofman MS, Lau WF, Hicks RJ. Somatostatin Receptor Imaging with 68Ga-DOTATATE PET/CT: clinical utility, normal patterns, pearls, and pitfalls in interpretation. Radiographics. 2015;35:500–516. doi: 10.1148/rg.352140164. [DOI] [PubMed] [Google Scholar]
- 9.Papadakis GZ, Bagci U, Sadowski SM, et al. Ectopic ACTH and CRH co-secreting tumor localized by 68Ga-DOTA-TATE PET/CT. Clin Nucl Med. 2015;40:576–578. doi: 10.1097/RLU.0000000000000806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Romero FR, Rais-Bahrami S, Permpongkosol S, et al. Primary carcinoid tumors of the kidney. J Urol. 2006;176:2359–2366. doi: 10.1016/j.juro.2006.07.129. [DOI] [PubMed] [Google Scholar]
- 11.Yoon JH. Primary renal carcinoid tumor: a rare cystic renal neoplasm. World J Radiol. 2013;5:328–333. doi: 10.4329/wjr.v5.i8.328. [DOI] [PMC free article] [PubMed] [Google Scholar]


