Abstract
Background and Objectives
Coronary angiography (CAG) is generally needed in the setting of systolic heart failure (HF) with an unidentified etiology as a part of diagnostic strategy. On the other hand, the clinical value of this invasive strategy is largely unknown. Platelet-lymphocyte ratio (PLR) has recently emerged as a novel inflammatory index that may serve as an important predictor of inflammatory state and overall mortality. The present study aimed to search the predictive value of PLR in determining the extent of coronary atherosclerosis in asymptomatic low ejection fraction (EF) patients.
Subjects and Methods
156 asymptomatic heart failure (HF) subjects (without angina or HF symptoms, mean age: 58 years; to male: 71.2%) were enrolled, and thereafter a CAG was performed. Gensini Score was used to determine the severity of coronary artery disease (CAD) on CAG. According to this scoring system, the overall study group was categorized into three distinct subgroups: control group with the score 0, mild atherosclerosis group with the score 0 to 20 and severe atherosclerosis group with the score of >20. Thereafter, a comparison was made among groups with regard to mean values of PLR.
Results
The severe atherosclerosis group had a substantially higher level of mean PLR in comparison to other groups (p<0.001). Pre-CAG PLR levels as well as a variety of clinical variables including age, low density lipoprotein (LDL)-cholesterol demonstrated an independent correlation with Gensini score through a multivariate analysis.
Conclusion
These findings suggest the potential association of high PLR levels with severe atherosclerosis in the setting of asymptomatic systolic HF. A simple measurement of PLR helps to identify the severity of coronary atherosclerosis prior to conducting coronary angiography.
Keywords: Low ejection fraction, Asymptomatic disease, Coronary artery disease, Gensini score, Platelet-lymphocyte ratio
Introduction
Performing invasive procedures including CAG in the setting of asymptomatic systolic HF is controversial. Angiography should also be considered in low ejection fraction (EF) patients with evidence of reversible myocardial ischemia on non-invasive testing.1) However, coronary angiography is applied for the elucidation of the etiology of low EF. As the prognosis of patients with ischemic etiology already remains worse,2) prediction of atherosclerosis burden may obviate the need for an unnecessary angiography with potential risks and harms to these vulnerable patients.
It is known that inflammation has a substantial role in the initiation and production of the complex atherosclerotic process3) that lies beneath cardiovascular disease (CVD). The role of inflammation in CVD has been studied and the relationship between various inflammatory markers has been established in the past.4),5) Platelets and leukocytes are the important components of these processes associated with the development of atherosclerosis6) Platelet-to-lymphocyte ratio (PLR) is a new prognostic index that integrates the risk prediction of these 2 parameters. It gives an idea about both the aggregation and inflammation pathways. Increased PLR values emerged as a valuable predictor adverse cardiovascular events in certain types of acute coronary syndromes in the long term7) and in predicting critical limb ischemia in peripheral artery disease.8) Moreover , higher PLR values emerged as a significant index that may independently predict the severity of coronary arterosclerotic disease.9) To date, no study has evaluated the role of PLR in Asymptomatic Low EF patients. We hypothesized that PLR levels could predict the severity of coronary atherosclerosis in the setting of asymptomatic systolic HF.
Subjects and Methods
Study population
The study herein is a single-center and retrospectively designed study, consisting of 156 consecutive patients eligible for selective (CAG) between May 2013 and July 2014 in our clinic. The study was approved by the local ethics committee, and was implemented in complete concordance with the Declaration of Helsinki on human research. We selected only subjects with systolic HF that persisted with an unknown etiology after an initial non-invasive clinical assessment. Inclusion criteria were: left ventricular ejection fraction (LVEF) <40% with global left ventricular hypokinesia and age of ≥18 years.
Information including gender, age, smoking status, hyperlipidemia, hypertension (HT), and diabetes mellitus (DM) were gathered. The definition of HT appeared as a systolic blood pressure (BP) value of ≥140 mmHg and/or a diastolic BP value of ≥90 mmHg at least on >2 office BP measurements or being on an antihypertensive therapy. The definition of DM comprised a blood sugar value of ≥126 mg/dL (7.0 mmol/L) in the fasting state or being on an antidiabetic therapy whereas the status of hyperlipidemia was based on the presence of a blood cholesterol level of ≥200 mg/dL or a triglyceride level of ≥150 mg/dL in the fasting state. Smoking was defined as currently smoking or ex-smokers who forwent smoking within the past 6months.
Patients with moderate or advanced cardiac valvulopathy, malignancy, clinically decompensated congestive HF, haematological disorder, severe kidney or liver dysfunction, active infection or systemic inflammatory conditions, or autoimmune disorders and patients using steroids were excluded from the study.
Biochemical and hematological parameters
After an overnight fasting, peripheral venous blood samples were drawn from patients. Dry tubes were used for biochemical analysis, and EDTA tubes were used for hematological tests. White blood cells and erythrocyte counts, hemoglobin along with hematocrit levels were analysed by an automated hematology device Coulter Counter LH Series (Beckman Coulter Inc, Hialeah, FL, USA). The biochemical measurements were determined by an automated analyzer of biochemistry (Abbott Laboratories, Abbott Park, IL, USA).
Coronary angiography and assessment of coronary atherosclerosis severity
All patients enrolled in the study underwent selective CAG by Judkins technique through the femoral artery. The Gensini scoring system was harnessed to identify the severity of coronary artery disease.10) According to this score, 1% to 25% of stenosis in the lumen of coronary arteries is assigned a score of 1 whereas the stenoses of 26% to 50%, 51% to 75%, 76% to 90%, 91% to 99% and 100% (totally occluded) represent the scores of 2, 4, 8, 16 and 32. The score based on the stenosis degree is thereafter multiplied by a constant number in accordance with the location of lesions along the coronary arteries. A multiplication constant of 5 points is generally assigned for left main coronary artery (LMCA) lesions whereas 2.5 points are assigned for left circumflex (LCX) or proximal left anterior descending (LAD) artery lesions. Regarding LCX and LAD, lesions involving mid segments and distal segments are given the points of 1.5 and 1, respectively. The lesions involving the first obtuse marginal (OM) and diagonal branches, intermediate and posterior descending arteries (PDA) are generally multiplied with a constant of 1 point while the lesions in second OM and diagonal branches are assigned the multiplication constant of 0.5. Assessment of Gensini score was made by 2 interventional cardiologists totally blinded to the laboratory and clinical data and the coronary evaluation of the other cardiologist. In the case of disagreement, a third blinded interventionalist assessed the coronary angiograms.
According to their coronary angiograms, subjects were categorized into three groups. The first group comprised 60 patients with a normal CAG (control group). The rest was categorized into two based on their Gensini score: mildly atherosclerotic group (n=64; score of <25 points) and severely atherosclerotic group (n=32; score of ≥25 points).
Echocardiography
All echocardiographic examinations were performed by a certified cardiologist experienced in this field using a Vıvıd-7 (GE Healthcare, Horten, Norway) device in compliance with American Society of Echocardiography (ASE) guidelines.11) LVEF was calculated from apical 4-chamber views, according to a modified Simpson's rule.
Statistics
Continuous variables were defined as means and standard deviation; categorical variables were presented as percentages. The normality of distribution for continuous variables was confirmed with the Kolmogorov-Smirnov test. According to the distribution pattern of continuous variables, Mann-Whitney U test or independent-sample t-test was used for continuous variables, and the chi-square test was used for categorical variables. One-way analysis of variance (ANOVA) or Kruskal-Wallis test was used to compare 3 groups. When there was a significant difference between the three study groups, the comparison of two groups in terms of the relevant parameter was performed with post hoc tests in a one-way ANOVA and with Mann-Whitney U test after Bonferroni correction in Kruskal-Wallis test. Correlations were assessed using either Pearson's correlation test or Spearman's rank test according to the distribution pattern of the variable. Independent associations between Gensini score and independent variables were evaluated through a backward stepwise multiple linear regression analysis by comprehending all parameters showing p value of less than 0.1 on a univariate analysis. Coefficients of standardized β regression along with their significance from the multiple linear regression analysis were also reported. A receiver operating characteristic (ROC) curve analysis was used to determine the optimum cut-off level of preprocedural PLR values to predict severe coronary atherosclerosis. Statistical analyses were performed using SPSS 16.0 (SPSS Inc., Chicago, IL, USA). A two-tailed p value of <0.05 was regarded as statistically significant.
Results
A total of 96 patients with coronary artery disease (men 69%, mean age: 61±11 years) and 60 control subjects (71.2% male, mean age: 54±13 years) with normal coronary arteries were enrolled in the study. Baseline demographic, biochemical, and hematological characteristics of the groups are outlined in Table 1. Baseline echocardiographic properties were similar in three groups and shown in Table 2.
Table 1. Comparison of baseline demographic characteristics and cardiovascular risk factors of the study population.
| Control group (n=60) | Mild atherosclerosis (n=64) | Severe atherosclerosis (n=32) | p | |
|---|---|---|---|---|
| Age | 54.3±13.1 | 61.8±11.6 | 60.8±10.2 | 0.002 |
| Male, n (%) | 43 (72) | 46 (72) | 22 (69) | 0.945 |
| Coronary risk factors, n (%) | ||||
| Smoking | 9 (15) | 8 (13) | 7 (22) | 0.486 |
| Hypertension | 18 (30) | 32 (50) | 15 (47) | 0.063 |
| Diabetes | 10 (17) | 10 (16) | 10 (32) | 0.154 |
| Biochemical parameters | ||||
| LDL-C (mg/dL) | 139 (34–317) | 168.5 (60–296) | 176.4 (72–362) | 0.003 |
| HDL-C (mg/dL) | 42 (19–81) | 46.3 (20–101) | 46.7 (22–81) | 0.108 |
| Triglyceride (mg/dL) | 149 (53–330) | 122.7 (104–366) | 147.3 (61–335) | 0.069 |
| Glucose (mg/dL) | 120.9 (68–323) | 111 (59–354) | 117.2 (63–298) | 0.473 |
| Creatinine (mg/dL) | 1.04 (0.52–1.87) | 1.05 (0.6–2.3) | 1.0 (0.65–2.6) | 0.829 |
| Hematologic parameters | ||||
| Hemoglobin (g/dL) | 13.9±1.87 | 15.5±5.4 | 14.1±1.66 | 0.060 |
| WBC (×103/µL) | 7.86±2.19 | 7.80±2.08 | 7.67±2.03 | 0.907 |
| Platelet (×103/µL) | 213.0±66.1 | 233.7±69.8 | 305.7±80.3 | 0.001 |
| Lymphocyte (×103/µL) | 1.91±0.63 | 1.80±0.76 | 1.78±0.70 | 0.704 |
| PLR | 118.4±40.4 | 145.2±96.5 | 183.5±61.6 | 0.001 |
Values are mean±SD; median (25-75 percentiles), or n (%). Comparison between three groups was performed with one-way ANOVA or Kruskal-Wallis test. For the results of post hoc tests or Mann-Whitney U test with Bonferroni correction, the significance levels are: P1 (control vs. mild atherosclerosis), P2 (control vs. severe atherosclerosis), P3 (mild vs. severe atherosclerosis) aP1=0.450 (N/S), P2=0.001, P3=0.010. P1=0.006, P2<0.001, P3=0.027 (N/S due to Bonferroni correction) cP1=0.044, P2<0.001, P3=0.058 (N/S) dP1=0.317 (N/S), P2<0.001, P3=0.005 eP1=0.797 (N/S), P2<0.001, P3<0.001 fP1=0.509 (N/S), P2=0.004, P3<0.001. ANOVA: analysis of variance, HDL: high-density lipoprotein cholesterol, LDL: low-density lipoprotein cholesterol, MPV: mean platelet volume, N/S: non-significant, PLR: platelet-to-lymphocyte ratio, SD-standard deviation, WBC: white blood cell count
Table 2. Comparison of echocardiographic properties of the study population.
| Control group (n=60) | Mild atherosclerosis (n=64) | Severe atherosclerosis (n=32) | p | |
|---|---|---|---|---|
| Left ventricular end-diastolic diameter (mm) | 58±10 | 56±7.6 | 55±6.3 | 0.305 |
| EF (%) | 33±5.2 | 32±7.0 | 34±6.0 | 0.402 |
| Left ventricular end-diastolic volume (mL) | 191±31.1 | 185±31.7 | 187±34.3 | 0.709 |
| Left ventricular end-systolic volume (mL) | 127±21.4 | 124±24.1 | 125±24.3 | 0.417 |
| Left ventricular mass index (gr/m²) | 153±21.2 | 149±24.1 | 148±22.2 | 0.327 |
| Left atrial diameter (mm) | 41±8.4 | 42±7.5 | 40±7.8 | 0.685 |
EF: ejection fraction
Patients in the severe atherosclerosis group and mild atherosclerosis group were older compared to the control group. (p=0.018 and p=0.01, respectively). Fasting serum glucose ,serum creatinine, triglyceride and HDL level of three groups were similar. Serum LDL was significantly higher in the severe atherosclerosis group than controls (p<0.001); also, it was significantly higher in the mild atherosclerosis group than control group (p=0.03). In severe and mild atherosclerosis groups LDL levels were similar (168.5±6.8 vs. 176.4±10.1, p=0.51, respectively)
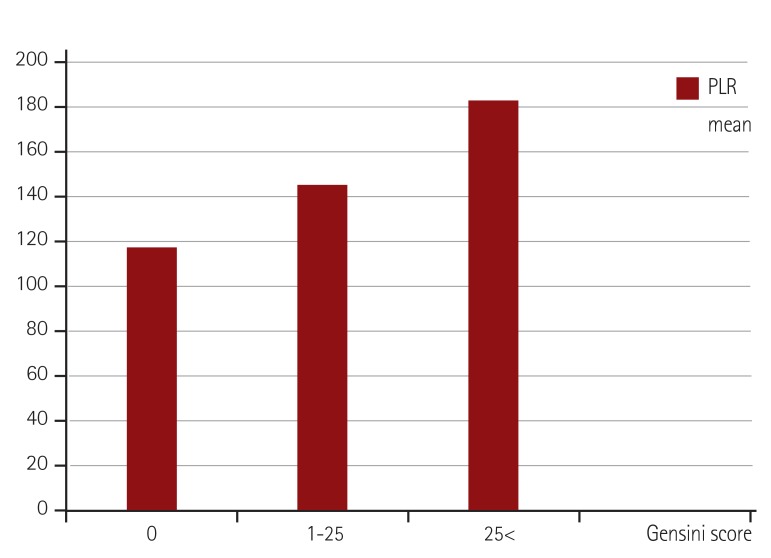
Among hematological parameters, hemoglobin levels and white blood cell count were similar between all three groups. The severe atherosclerosis group had significantly higher platelet counts compared to the mild atherosclerosis group (p<0.001) and controls (p=0.001), though platelet counts of the last two groups were similar (p=0.094). Lymphocyte count was higher in control groups than the others (1.91±0.63 vs. 1.84±0.76 and 1.78±0.70) but this is not statistically important (p=0.704). PLR was significantly higher in the severe atherosclerosis group compared to the mild atherosclerosis (p<0.001) and control groups (p<0.001) (183.5±61.6 vs. 145.2±96.5 vs. 118.4±40.4 respectively, p<0.001 for ANOVA) (Fig. 1).
Fig. 1. Mean PLR of controls and mild and severe atherosclerosis groups. PLR: platelet-to-lymphocyte ratio.
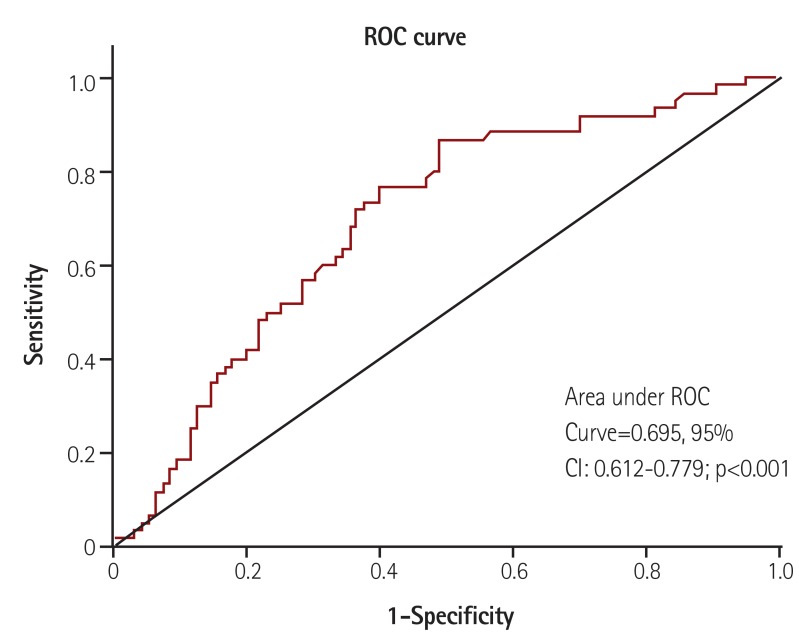
PLR was also significantly higher in patients with coronary artery disease (CAD) (n=96) compared to controls (n=60) (158±88 vs. 118±40, p<0.001). Using a cut-off level of 132, PLR predicted severe atherosclerosis with a sensitivity of 76% and specificity of 60%. The area under ROC curve was 0.695 (95% CI: 0.612-0.779, p<0.001) (Fig. 2).
Fig. 2. The ROC curve analysis of PLR for predicting severe atherosclerosis. ROC: receiver operating characteristic, CI: confidence interval, PLR: platelet-to-lymphocyte ratio.
Independent associations between Gensini score and independent variables were assessed by multivariate linear regression analysis by including all parameters showing a correlation with a p value of less than 0.1 on a univariate analysis (patient age, PLR and LDL). Pre-procedural PLR was independently associated with Gensini score (β=0.347, p=0.001), together with LDL (β=0.259, p=0.001), age (β=-0.180, p=0.014) (Table 3).
Table 3. Multivariate linear regression analysis to determine independent variables significantly associated with Gensini score.
| Dependent variable | Independent variable | B | β | t | p | VIF |
|---|---|---|---|---|---|---|
| Gensini | Age | 0.011 | 0.180 | 2.495 | 0.014 | 1.029 |
| PLR | 0.003 | 0.347 | 4.826 | 0.001 | 1.020 | |
| LDL | 0.003 | 0.259 | 3.486 | 0.001 | 1.082 |
B: unstandardized coefficients, β: standardized regression coefficients, VIF: variance inflation factor, PLR: platelet-to-lymphocyte ratio, LDL: low density lipoprotein
Discussion
The present study demonstrated that a high PLR level was independently related to the severity and extent of atherosclerotic heart disease in asymptomatic low EF patients. Patients with high pre-procedural PLR had significantly higher Gensini scores, and there was a strong link between PLR values and Gensini scores of patients with CAD. Additionally, this study showed that pre-procedural PLR >132 predicted severe atherosclerosis with a sensitivity of 76% and specificity of 60%. PLR had an independent association with Gensini score on top of high LDL levels and age as demonstrated by a multivariate analysis. This might reportedly be the first study demonstrating the link between PLR and severity of CAD in the setting of asymptomatic systolic HF.
According to the recommendations of current guidelines for chronic heart failure, cardiac catheterization should be considered in the etiological investigation of patients with left ventricular dysfunction in the presence of risk factors for coronary artery disease, angina, or refractory symptoms of heart failure.1) However demonstrate the ischemic or non ischemic etiology is important for prognosis in low EF patients. Bart et al.2) showed that ischemic etiology might still be considered as an important factor independently predicting a 5-year mortality. In a previous study for elucidation etiology in patients with left ventricle dysfunction, coronary angiography was performed and ischemic etiology was determined as 9.3%.12) Gensini score was used in our study and the detection of the severe coronary artery disease was 20% of the study group.
In a study, coronary computed tomography angiography (CCTA) was compared with invasive CAG in newly diagnosed heart failure patients in terms of detection of the severity of atherosclerotic heart disease has been determined with CCTA similar to invasive coronary angiography.13)
PLR is a new prognostic marker that gives an idea about both the aggregation and inflammation pathways. The role of leucocytes and platelets in atherosclerosis is well known.6),14) Increased platelet and low lymphocyte counts has been shown with poor cardiovascular outcomes in earlier studies.14),15),16),17) It has been shown that PLR is associated with long-term and shortterm mortality in acute coronary syndrome patients.7),18) Yildiz et al.19) showed that higher PLR was associated with poor prognosis in patients with CAD. Acar et al.20) demonstrated that high PLR is related with coronary collateral development in patients with chronic total occlusions. Sarı et al.21) revealed that pre-procedural PLR is associated with coronary atherosclerosis severity.
Oxidized LDL (atherogenic) may emerge as a trigger of endothelial dysfunction strongly associated with the atherosclerotic process. The association between the serum LDL and atherogenesis has been well established.22),23) In our study, pre procedural LDL levels also served as major factors independently predicting the severity of coronary atherosclerosis as expected.
Our study has important clinical implications; PLR which is a simple, cheap, and readily available marker can be used as a prognostic indicator to determine asymptomatic low EF patients who are at higher coronary atherosclerosis risk. In patients with elevated PLR, even if they are asymptomatic, investigation ischemia and performing coronary angiography may impact on prognosis. In patients with low PLR, noninvasive coronary imaging techniques may be recommended for coronary anatomy viewing.
Study limitations
There are several limitations regarding the present study. Sample size was small, enrolment was retrospective and it was a single center design. The amount of coronary atherosclerosis was evaluated only by a coronary angiography. We know it does not provide sufficient data about the coronary plaque burden, and shows only the lumen of coronary arteries. An intravascular ultrasound and/or coronary CT may provide necessary data about the amount of coronary atherosclerosis. We had no data on inflammation markers including C-reactive protein, tumour necrosis factor-alpha, etc. However, assessment of these substances may strengthen the study. We did not analyze other platelet indices including mean platelet volume, platelet distribution width, and inflammatory indices including neutrophil lymphocyte ratio (NLR) or correlation of these parameters with the PLR. Notwithstanding these limitations, this is reportedly the first study focusing on PLR with regard to its predictive value in asymptomatic low EF subjects, and more comprehensive studies are still warranted to corroborate our findings.
Conclusions
This is reportedly the first trial primarily focusing on the potential association between PLR and the severity of atherosclerotic heart disease in asymptomatic low EF patients. Our results indicate that harnessing the index of PLR as well as a variety of clinical variables including advanced age, DM and high LDL levels may work as valuable predictors of atherosclerotic burden, hence severity of atherosclerosis. However, the current information on this issue is still at its infancy suggesting the need for future clinical studies to validate absolute implications of this index in the setting of atherosclerotic heart disease.
Footnotes
The authors have no financial conflicts of interest.
References
- 1.McMurray JJ, Adamopoulos S, Anker SD, et al. ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: The Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2012;14:803–869. doi: 10.1093/eurjhf/hfs105. [DOI] [PubMed] [Google Scholar]
- 2.Bart BA, Shaw LK, McCants CB, Jr, et al. Clinical determinants of mortality in patients with angiographically diagnosed ischemic or nonischemic cardiomyopathy. J Am Coll Cardiol. 1997;30:1002–1008. doi: 10.1016/s0735-1097(97)00235-0. [DOI] [PubMed] [Google Scholar]
- 3.Hansson GK. Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med. 2005;352:1685–1695. doi: 10.1056/NEJMra043430. [DOI] [PubMed] [Google Scholar]
- 4.Duffy BK, Gurm HS, Rajagopal V, Gupta R, Ellis SG, Bhatt DL. Usefulness of an elevated neutrophil to lymphocyte ratio in predicting long-term mortality after percutaneous coronary intervention. Am J Cardiol. 2006;97:993–996. doi: 10.1016/j.amjcard.2005.10.034. [DOI] [PubMed] [Google Scholar]
- 5.Tamhane UU, Aneja S, Montgomery D, Rogers EK, Eagle KA, Gurm HS. Association between admission neutrophil to lymphocyte ratio and outcomes in patients with acute coronary syndrome. Am J Cardiol. 2008;102:653–657. doi: 10.1016/j.amjcard.2008.05.006. [DOI] [PubMed] [Google Scholar]
- 6.Davi G, Patrono C. Platelet activation and atherothrombosis. N Engl J Med. 2007;357:2482–2494. doi: 10.1056/NEJMra071014. [DOI] [PubMed] [Google Scholar]
- 7.Ozcan Cetin EH, Cetin MS, Aras D, et al. Platelet to lymphocyte ratio as a prognostic marker of in-hospital and long-term major adverse cardiovascular events in ST-segment elevation myocardial infarction. Angiology. 2016;67:336–345. doi: 10.1177/0003319715591751. [DOI] [PubMed] [Google Scholar]
- 8.Gary T, Pichler M, Belaj K, et al. Platelet-to-lymphocyte ratio: a novel marker for critical limb ischemia in peripheral arterial occlusive disease patients. PLoS One. 2013;8:e67688. doi: 10.1371/journal.pone.0067688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yuksel M, Yildiz A, Oylumlu M, et al. The association between platelet/lymphocyte ratio and coronary artery disease severity. Anatol J Cardiol. 2015;15:640–647. doi: 10.5152/akd.2014.5565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gensini GG. A more meaningful scoring system for determining the severity of coronary heart disease. Am J Cardiol. 1983;51:606. doi: 10.1016/s0002-9149(83)80105-2. [DOI] [PubMed] [Google Scholar]
- 11.American College of Cardiology Foundation Appropriate Use Criteria Task Force; American Society of Echocardiography; American Heart Association et al. ACCF/ASE/AHA/ASNC/HFSA/HRS/SCAI/SCCM/SCCT/SCMR 2011 Appropriate use criteria for echocardiography. A report of the American College of Cardiology Foundation Appropriate Use Criteria Task Force, American Society of Echocardiography, American Heart Association, American Society of Nuclear Cardiology, Heart Failure Society of America, Heart Rhythm Society, Society for Cardiovascular Angiography and Interventions, Society of Critical Care Medicine, Society of Cardiovascular Computed Tomography, Society for Cardiovascular Magnetic Resonance American College of Chest Physicians. J Am Soc Echocardiogr. 2011;24:229–267. doi: 10.1016/j.echo.2010.12.008. [DOI] [PubMed] [Google Scholar]
- 12.Melo RM, Melo EF, Biselli B, Souza GE, Bocchi EA. Clinical usefulness of coronary angiography in patients with left ventricular dysfunction. Arq Bras Cardiol. 2012;98:437–441. doi: 10.1590/s0066-782x2012005000035. [DOI] [PubMed] [Google Scholar]
- 13.ten Kate GJ, Caliskan K, Dedic A, et al. Computed tomography coronary imaging as a gatekeeper for invasive coronary angiography in patients with newly diagnosed heart failure of unknown aetiology. Eur J Heart Fail. 2013;15:1028–1034. doi: 10.1093/eurjhf/hft090. [DOI] [PubMed] [Google Scholar]
- 14.Zouridakis EG, Garcia-Moll X, Kaski JC. Usefulness of the blood lymphocyte count in predicting recurrent instability and death in patients with unstable angina pectoris. Am J Cardiol. 2000;86:449–451. doi: 10.1016/s0002-9149(00)00963-2. [DOI] [PubMed] [Google Scholar]
- 15.Iijima R, Ndrepepa G, Mehilli J, et al. Relationship between platelet count and 30-day clinical outcomes after percutaneous coronary interventions. Pooled analysis of four ISAR trials. Thromb Haemost. 2007;98:852–857. [PubMed] [Google Scholar]
- 16.Nikolsky E, Grines CL, Cox DA, et al. Impact of baseline platelet count in patients undergoing primary percutaneous coronary intervention in acute myocardial infarction (from the CADILLAC trial) Am J Cardiol. 2007;99:1055–1061. doi: 10.1016/j.amjcard.2006.11.066. [DOI] [PubMed] [Google Scholar]
- 17.Thaulow E, Erikssen J, Sandvik L, Stormorken H, Cohn PF. Blood platelet count and function are related to total and cardiovascular death in apparently healthy men. Circulation. 1991;84:613–617. doi: 10.1161/01.cir.84.2.613. [DOI] [PubMed] [Google Scholar]
- 18.Azab B, Shah N, Akerman M, McGinn JT., Jr Value of platelet/lymphocyte ratio as a predictor of all-cause mortality after non-ST-elevation myocardial infarction. J Thromb Thrombolysis. 2012;34:326–334. doi: 10.1007/s11239-012-0718-6. [DOI] [PubMed] [Google Scholar]
- 19.Yildiz A, Yuksel M, Oylumlu M, et al. The utility of the platelet-lymphocyte ratio for predicting no reflow in patients with ST-segment elevation myocardial infarction. Clin Appl Thromb Hemost. 2015;21:223–228. doi: 10.1177/1076029613519851. [DOI] [PubMed] [Google Scholar]
- 20.Açar G, Kalkan ME, Avci A, et al. The relation of platelet-lymphocyte ratio and coronary collateral circulation in patients with stable angina pectoris and chronic total occlusion. Clin Appl Thromb Hemost. 2015;21:462–468. doi: 10.1177/1076029613508599. [DOI] [PubMed] [Google Scholar]
- 21.Sari I, Sunbul M, Mammadov C, et al. Relation of neutrophil to lymphocyte and platelet to lymphocyte ratio with coronary artery disease severity in patients undergoing coronary angiography. Kardiol Pol. 2015;73:1310–1316. doi: 10.5603/KP.a2015.0098. [DOI] [PubMed] [Google Scholar]
- 22.Stancel N, Chen CC, Ke LY, et al. Interplay between CRP, Atherogenic LDL, and LOX-1 and Its Potential Role in the Pathogenesis of Atherosclerosis. Clin Chem. 2016;62:320–327. doi: 10.1373/clinchem.2015.243923. [DOI] [PubMed] [Google Scholar]
- 23.Stemme S, Faber B, Holm J, Wiklund O, Witztum JL, Hansson GK. T lymphocytes from human atherosclerotic plaques recognize oxidized low density lipoprotein. Proc Natl Acad Sci USA. 1995;92:3893–3897. doi: 10.1073/pnas.92.9.3893. [DOI] [PMC free article] [PubMed] [Google Scholar]