Abstract
This review article describes fundamental aspects of cell membrane-inspired phospholipid polymers and their usefulness in the development of medical devices. Since the early 1990s, polymers composed of 2-methacryloyloxyethyl phosphorylcholine (MPC) units have been considered in the preparation of biomaterials. MPC polymers can provide an artificial cell membrane structure at the surface and serve as excellent biointerfaces between artificial and biological systems. They have also been applied in the surface modification of some medical devices including long-term implantable artificial organs. An MPC polymer biointerface can suppress unfavorable biological reactions such as protein adsorption and cell adhesion – in other words, specific biomolecules immobilized on an MPC polymer surface retain their original functions. MPC polymers are also being increasingly used for creating biointerfaces with artificial cell membrane structures.
Keywords: Phospholipid polymers, Biocompatibility, Biointerface, Surface modification, Medical devices
1. Development of MPC polymer science
The cell membrane is considered the best surface for smooth interaction with biological components such as proteins and cells. A model of the structure of a cell membrane, which is well known as the fluid-mosaic model, was proposed by Singer and Nicolson [1]. According to this model, amphiphilic phospholipids are arranged in a bilayer structure and proteins are located in or on it. The distribution of these components is asymmetric, that is, negatively charged phospholipids such as phosphatidylserine are predominantly found on the inner, cytoplasmic side of the membrane, whereas neutral, zwitterionic phosphorylcholine lipids such as phosphatidylcholines are more commonly located in the outer leaflet [2]. Phosphatidylcholine provides an inert surface for the biological reactions of proteins and glycoproteins to occur easily on the membrane. This fact is extremely important for the design of a biointerface that can function between artificial and biological systems.
A new concept was proposed for designing blood-compatible polymeric materials with cell membrane-like surfaces; this concept uses 2-methacryloyloxyethyl phosphorylcholine (MPC, figure 1), which is a methacrylate monomer with a phosphorylcholine (PC) group. Although MPC polymers were studied back in 1978, their properties were not understood [3]. The synthesis of MPCs was rather difficult and complex, and therefore sufficient amounts of pure MPC could not be obtained. Later, Ishihara et al established a refined and complete process for the synthesis and purification of MPC [4]. This achievement brought about considerable progress in the development of MPC polymers as biomaterials.
Figure 1.
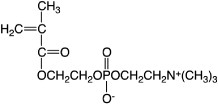
Chemical structure of MPC.
In 1999, with the cooperation of Ishihara and Nakabayashi and the support of the Japan Science and Technology Agency, the Japanese chemical company NOF Corporation built the world's first commercial plant to produce MPC and its polymers on an industrial scale.
Because MPC is easily polymerized, numerous MPC polymers having a wide variety of molecular architectures, that is, random copolymers [5], block-type copolymers [6–9], graft-type copolymers [10–13], and terminal-functioned polymers [10, 14], have been prepared, as shown in figure 2(a). Polymerization of MPC and its derivatives are summarized in this review article [15]. MPC polymers are soluble in water, and the solubility can be easily adjusted by changing the structure and fraction of the comonomers. Free-radical polymerization is one of the most preferable processes to make MPC polymers and typical initiators including redox types [16].
Figure 2.
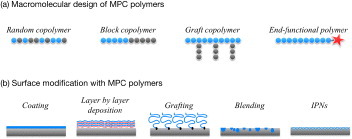
Schematic of molecular designs (a) and surface modification (b) performed with MPC polymer science.
In recent years, much effort has been focused on the development of a ‘living’ free-radical polymerization process, which would be useful in the syntheses of homopolymers and block copolymers with controlled molecular weight and narrow molecular weight distribution. Armes et al developed the atom transfer radical polymerization (ATRP) of MPC, and many studies on this topic have been reported [17–21]. This method of polymerization can be used to introduce a specific functional group in the terminal of the polymer chain and produce a variety of multi-branched polymers [17, 22–24].
Reverse addition fragmentation transfer (RAFT) polymerization is another process with a living polymerization approach. It is useful for making block and end-functional polymers and various types of amphiphilic MPC copolymers. RAFT polymerization of MPC was first performed by Yusa et al [25] and elaborated in further reports [26–28]. This method of polymerization was also performed in protic media; in contrast with the ATRP process, it does not require the removal of metal catalysts from the polymers [29–31].
2. Surface design with MPC polymers
2.1. Coating of artificial materials with MPC polymers
Figure 2(b) shows the available surface modification processes with MPC polymers on substrates [32]. Among them, the coating process is most appropriate and suitable for immobilization of MPC polymers. Considering the molecular interactions expected between the polymers and the surface, a hydrophobic group should be introduced in the MPC polymers. For this purpose, random copolymerization of MPC with an alkyl methacrylate is commonly used for preparation of the MPC polymers [5]. Not only the solubility of the wetting solvent for the substrate but also the molecular weight of the MPC polymer is important for achieving an adequate and stable coating [33, 34]. The most common MPC polymer for a coating on the substrate is poly(MPC-co-n-butyl methacrylate) (that is, poly(MPC-co-n-BMA) or PMB, figure 3) with 30 mol% of the MPC unit in the polymer (PMB37) [4, 5, 32]. The MPC polymer coating surface shows a higher contact angle for water because the hydrophobic alkyl methacrylates enriched at the air–material interface reduce surface free energy. The surface is dynamic; thus, surface equilibrium with aqueous media is necessary to orient the polar PC groups in the MPC units at the surface [35]. To shorten the equilibrium period, the mobility of the PC groups has been studied as a function of the chemical structure of monomer [35, 36], solvent composition [37], and types of comonomers [38]. For example, polydimethylsiloxane (PDMS) is one of the most commonly used materials. However, coating of PDMS with another polymer is difficult because of its extreme hydrophobicity and high mobility of the polymer chains. Fukazawa et al developed a system of MPC polymer and solvent for coating [39], that is, poly(MPC-co-2-ethylhexyl methacrylate-co-N,N-diethylaminoethyl methacrylate) (abbreviated as poly(MPC-co-2-EHMA-co-N,N-DEAEMA) or PMED) in a 20 vol% solution of ethanol in water. PMED stably covers the PDMS surface owing to complex molecular interactions such as hydrophobic interaction induced by the EHMA units and electrostatic force generated by the DEAEMA units. The solvent composition regulates the PMED conformation in the solution by spreading the polymer chains.
Figure 3.
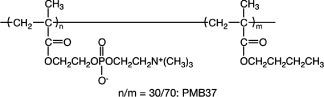
Chemical structure of BMB37.
By comparison with random polymers, a block-type polymer with a poly(MPC) segment has much higher mobility and enrichment of PC groups at the surface. Other segments in such a block-type polymer play an important role in attaching the poly(MPC) segment to the substrate [31, 40]. Layer-by-layer molecular integration is also applicable for modifying the substrate with MPC polymers [41–43].
2.2. Hybridization of MPC polymers with conventional materials
For the surface modification of materials, the formation of blending [44–49] and interpenetrating networks (IPNs) [50–54] is effective in reducing the elution of MPC polymers from the substrate. Segmented polyurethane (SPU) and polysulfone (PSf) are engineered polymers that are suitable for this task because of their good mechanical properties [44–46]. Blends of SPU and PSf with MPC polymers can be prepared by solvent evaporation. The MPC content can be optimized to improve the surface properties of the blend while preserving the mechanical properties of SPU and PSf.
2.3. Grafting of MPC polymers on materials
Graft polymerization of MPC is a familiar method used to modify surface properties; the modified surface is relatively stable because the graft polymers are connected to the substrate by covalent bonding. Graft polymerization can be achieved by chemical reactions [55, 56] as well as by physical treatments such as plasma treatment [57], corona discharge [58], and photoirradiation [59, 60]. Comb-shaped MPC graft polymers at the interface do not need reorientation with aqueous media to exhibit nonfouling properties. As an alternative method, surface-initiated ATRP (SI-ATRP) of MPC was also applied for surface modification of a solid surface [61–63]. This procedure is called ‘grafting from’ and can be used to prepare dense polymer brushes—cf with the adsorption of functionalized polymers to solid/liquid interfaces, which is called ‘grafting to’ because of steric hindrance of the polymers [64]. Surface patterning of polymer brushes is not only of interest for many applications ranging from microelectronics to biomaterials, but is also useful for scientific studies of surface-tethered polymer films. The poly(MPC) brush region and its dimensions can be well controlled via selective decomposition of surface-bound ATRP initiators by UV irradiation, resulting in micropatterns of poly(MPC) brushes as shown in figure 4.
Figure 4.
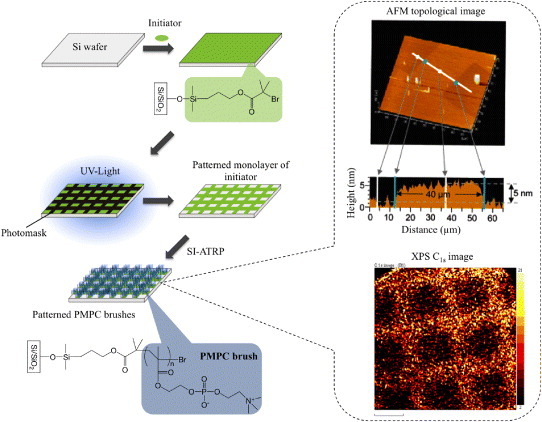
Preparation of a micropatterned poly(MPC) brush surface.
2.4. Functionalization of MPC polymers with other biomolecules
Immobilization of biomolecules, including proteins, polysaccharides, and DNA, is important in the preparation of sensors, monitors and diagnostic devices based on bioaffinity. To that end, a simple method is necessary for immobilization under gentle conditions. Additionally, the activity of biomolecules after immobilization should be maintained as that in the native state. MPC units in the polymer surface effectively reduced the denaturation of proteins immobilized with the polymers [65]. Then, a novel MPC polymer based on the chemical structure of PMB: poly(MPC-co-BMA-co-p-nitrophenyloxycarbonyl poly(ethylene glycol) methacrylate (MEONP)) (PMBN) was synthesized [66]. One of the monomer units, the MEONP unit, has an active ester group in the side chain, and can react with a specific biomolecule via condensation with its amino group. After that, the substrate is coated with the water-insoluble PMBN via solvent evaporation, and the biomolecules are immobilized. The PMBN is coated on glass, cyclic polyolefin, and gold substrates. For example, a PMBN surface is useful for DNA immobilization, and is applied to produce DNA chips such as Prime Surface® by Sumitomo Bakelite Co. Ltd. Kinoshita et al focused on the hybridization properties with regard to a suitable surface chemistry for a cyclic olefin copolymer (COC) surface with PMBN. They also discussed new approaches for the application of an on-chip DNA detection method through multiple primer extension (MPEX) by DNA polymerase [67]. Besides the nitrophenyloxycarbonyl group, other functional groups were also proposed for the conjugation of proteins with MPC polymers [20, 21, 68]. Iwata and coworkers utilized well-defined block copolymer brushes consisting of poly(MPC) and poly(glycidyl methacrylate) (poly(GMA)) on silicon wafers to immobilize antibody Fab′ fragments in a defined orientation [68]. The orientation of the antibody fragments was defined by derivatizing the GMA units with pyridyl disulfide and immobilizing the antibodies via thiol–disulfide interchange reactions.Very recently, instead of MEONP units, other active ester units, N-succinimidyloxycarbonyl di(ethylene glycol) methacrylate units, were introduced into an MPC polymer (PMBS) [69]. Both PMBN and PMBS were fabricated as nanofibers deposited by electrospinning, and antibodies were immobilized to capture a specific antigen. Alternatively, a water-soluble amphiphilic PMBN was synthesized with controlled monomer fractions in PMBN, and monodispersed nanoparticles covered with the PMBN were prepared (figure 5(a)) [70]. On the surface of the nanoparticles, several kinds of enzymes were immobilized, and sequential enzymatic reactions on the nanoparticles were conducted. The enzyme-immobilized nanoparticles were applied as biosensing devices coupled with a microdialysis system [66, 71]. Using these devices, acetylcholine and choline can be detected continuously. Ishihara et al immobilized arginine octapeptide (R8) on PMBN-covered nanoparticles (figure 5(b)) [72–74]. The nanoparticles were well penetrated into HeLa cells, whereas PMBN-covered nanoparticles without immobilization of R8 were not delivered to the cells (figure 5(c)). This result indicates that the interaction between cells and PMBN nanoparticles can be controlled by a ligand immobilized on the nanoparticles.
Figure 5.
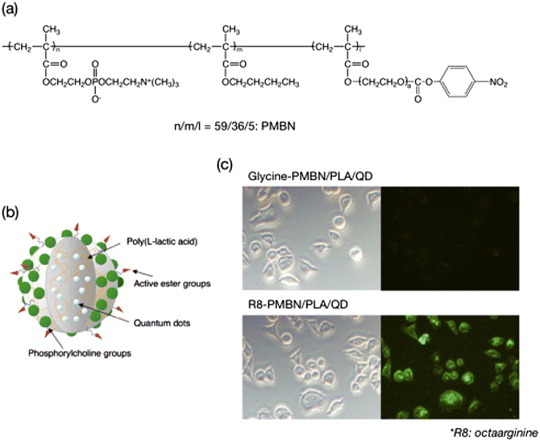
(a) Chemical structure of PMBN. (b) Schematic of one polymer nanoparticle embedding quantum dots within an artificial cell membrane surface. (c) Uptake of polymer nanoparticles covered with artificial cell membrane without and with R8 immobilization.
3. Biointerfaces of MPC polymers
3.1. Nonfouling surfaces
MPC polymers have great potential in the design of biointerfaces as shown in figure 6. Particularly, the nonfouling nature of MPC polymers is essential for biomedical materials. Protein adsorption is the first phenomenon that occurs when synthetic materials come in contact with a living organism. The uncontrolled protein adsorption works as a trigger for foreign body reactions with materials from a host. Therefore, protein adsorption is important in the preparation of synthetic materials for biomedical applications.
Figure 6.
Biointerfacial aspects of MPC polymers.
PMB37 is one of the MPC polymers used for the surface modification of implantable medical devices. The blood compatibility of PMB is differentiated for each blood component, e.g., cells, plasma protein, phospholipids, and water [32, 75–80].
Figure 7 shows experimental columns containing polymer-coated poly(methyl methacrylate) beads, and micrographs of the bead surface after a contact with human blood without any anticoagulant. On poly(BMA), which does not have any MPC units, many adherent blood cells are observed and a clot is generated. In contrast, PMB37 effectively suppresses cell adhesion. While adherent platelets may sometimes easily detach from the surface, they are strongly activated by contact with the polymer surface and induce embolization. Therefore, to understand the blood compatibility it is necessary to evaluate the activation of platelets that barely contact the polymer surface and those that adhere to it. The activation of platelets after contact with PMB37 was evaluated by measuring the concentration of cytoplasmic calcium ions in the platelets (figure 8) [81]. The results clearly indicated a lower degree of activation for the platelets that contacted the PMB37 surface than those on other polymer substrates or glass. That is, PMB37 is an excellent antithrombogenic polymer for the suppression of cell activation and adhesion.
Figure 7.
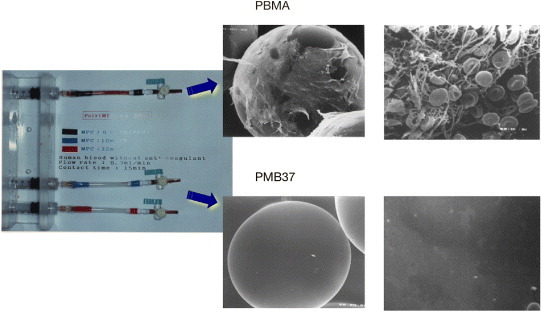
Microsphere columns after passage of whole blood for 15 min (left) and scanning electron microscopy images of polymer-coated beads packed in the columns (right).
Figure 8.
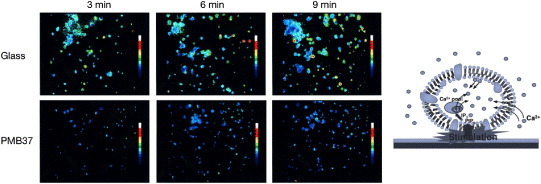
Change in cytoplasmic calcium ion concentration of platelets after contact with glass and PMB37 surfaces.
As mentioned above, SI-ATRP results in a well-controlled molecular architecture [82]. Previous papers described the effectiveness of ATRP in preparing a well-defined graft polymer on a solid surface [83–85]. ATRP has also been applied for the fabrication of polymer brush micropatterns on solid surfaces [62, 86–89]. On well-defined poly(MPC) brushes, protein adsorption was effectively reduced. Furthermore, protein and cell manipulations were easily performed when the thickness of the polymer brushes was just above 5 nm [62, 63, 90]. Preparation of poly(MPC) brushes by ATRP has been reported by Feng and Brash [61, 63, 91]. They prepared PMPC brushes with various graft densities from 0.06 to 0.39 chains nm–2 and chain lengths from 5 to 200 MPC units, and characterized their effects on protein adsorption [91]. A marked reduction in fibrinogen adsorption was observed on surfaces with high graft densities and long poly(MPC) chains, as shown in figure 9. These experiments demonstrated the ability of poly(MPC) brushes to considerably reduce protein adsorption and cell adhesion. Yoshimoto and coworkers compared the nonfouling characteristics of a poly(MPC)-modified gold surface and a poly(ethylene glycol) (PEG)-modified gold surface [92]. They clarified that the number of MPC units is an important factor in the excellent protein resistance offered by poly(MPC)-modified gold surfaces fabricated by the ‘grafting to’ method, which is in sharp contrast to that of PEG-tethered chains.
Figure 9.
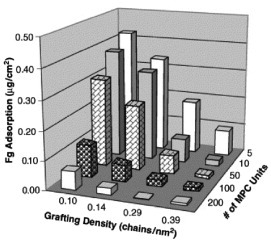
Profile of fibrinogen adsorption on poly(MPC)-grafted silicon surfaces with varying graft density and chain length in a tris-buffered saline buffer for 2 h at room temperature. Fibrinogen concentration: 1.0 mg ml−1. (Reprinted from [91] with permission from Elsevier.)
Polymer brush surfaces have unique friction properties. In particular, the presence of hydrophilic polymer or polyelectrolyte chains is related to the state of water molecules at the interface. The mechanism for high lubrication of poly(MPC) brushes generated on a solid was elaborated by Takahara and co-workers [93, 94]. They prepared poly(MPC) brushes on a silicon wafer via ATRP and characterized the surface properties using a water lubrication system. Poly(MPC) brushes showed an extremely low coefficient of friction even under humid air conditions when probed by a brush-tethered glass ball, probably owing to the adsorption of moisture forming a water-lubrication layer on the surface.
3.2. Biologically specific surfaces based on poly(MPC) brushes
Poly(MPC) brush surfaces can also significantly inhibit lipid vesicle adsorption. Vesicle fusion on patterned PMPC brushes resulted in the preparation of phospholipid bilayer microarrays (PLBMAs), as illustrated in figure 10(a) [80]. A glycolipid containing an oligosaccharide unit for specific binding by cholera toxin B (CTB) subunits was incorporated into phospholipid bilayers containing 2 mol% of ganglioside GM1. In addition, phospholipid bilayers containing 2 mol% of biotin-cap-DOPE (DOPE stands for 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine) were prepared for specific binding to NeutrAvidin. The binding assay (figure 10(b)) was performed using human plasma as a medium. The fluorescence microscopy images in the left column in figure 10(b) show pure 1,2-dioleoyl-sn-glycero-3-phosphocholine (DOPC) PLBMAs for controls (i and iv). The middle column in figure 10(b) shows PLBMAs with GM1 (ii and v). The right column shows PLBMAs with biotin-capped lipids (iii and vi). The top row in figure 10(b) shows contact with each PLBMA and the Alexa Fluor 488-labeled 50 nM CTB, whereas the bottom row shows contact with the Alexa Fluor 488-labeled 50 nM NeutrAvidin. As expected, the CTB binds specifically to GM1-containing PLBMAs (ii). In addition, NeutrAvidin binds specifically to biotin-containing PLBMAs (vi). More importantly, the authors observed neither cross reaction nor non-specific protein binding to lipid bilayer elements that did not contain a ligand other than the one required by the protein–ligand pair of interest (i, iii, iv, v). Similar results were obtained when using phosphate buffered saline instead of human plasma as a medium. These data only demonstrate molecular recognition on PLBMAs using Alexa Fluor 488-labeled proteins. The relevance of PLBMAs as substrates for detection of non-labeled proteins can be clarified using other methods, e.g. enzyme-linked immunosorbent assay.
Figure 10.
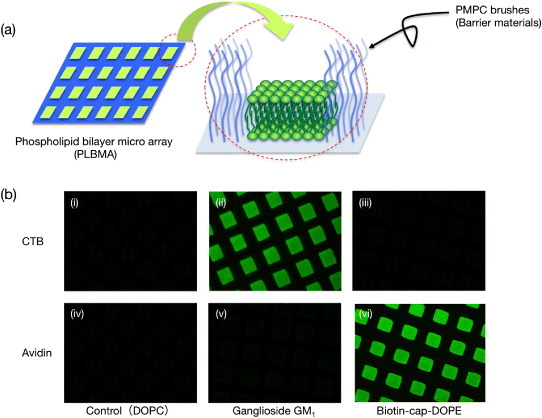
(a) Schematic of phospholipid bilayer microarray (PLBMA) decorated by well-defined poly(MPC) brushes. (b) Fluorescence micrographs of PLBMA containing: (i, iv) DOPC; (ii, v) 2.0 mol% GM1 in DOPC; (iii, vi) 2.0 mol% biotin-cap-DOPE in DOPC. The top row shows contact with each PLBMA and the Alexa 488-labeled 50 nM CTB (i, ii, iii); the bottom row shows contact with the Alexa 488-labeled 50 nM NeutrAvidin (iv, v, vi) in human plasma.
3.3. Cell-function controllable surfaces
Cell compatibility of MPC polymers has also been determined by measuring the amount of secreted IL-1β mRNA from adherent macrophage-like cells on polymer surfaces [95, 96]. The mRNA expression from the adherent cells on MPC polymer surfaces was significantly lower than that on conventional polymeric biomaterials. This property was the basis for studies of MPC polymers as materials for cell cultivation.
Carbohydrates on a cell surface contribute to most forms of communication between living cells and their environment; therefore, they can also act as surface-immobilized ligands [97]. In particular, galactose residues are preferred conjugated polymers to interact with hepatocytes, which are asialoglycoprotein receptors (ASGPRs). ASGPR is a lectin for receptor-mediated endocytosis found at the hepatocyte cell surface, which is bound to galactose/N-acetylgalactosamine (GalNAc)-terminated ligands in a calcium-dependent manner [98, 99]. Although the ASGPR does not function physiologically as an adhesion receptor, galactose-containing polymers have been used to induce adhesion in primary hepatocytes [100–102]. Various polymers bearing galactose residues as ligands were prepared for drug delivery [103] and cellular matrices [104]. While these approaches have been quite successful, limitations remain in terms of selective recognition [105]. Indeed, most of the earlier reports did not focus on the reduction of non-specific interactions. Iwasaki et al immobilized carbohydrate side chains on PMB37 for inducing specific interactions to cells [106]. They prepared poly(MPC-co-BMA-co-2-lactobionamidoethyl methacrylate) (poly(MPC-co-BMA-co-2-LAMA) or PMBL, figure 11(a)) and coated it on substrates by solvent evaporation. Figure 11(b) shows the density of adherence of the human hepatocellular liver carcinoma cell line (HepG2) having asialoglycoprotein receptors (ASGPRs) and mouse fibroblasts (NIH-3T3) on the polymer surfaces. On poly(BMA), many adherent cells were observed and were well spread with monolayer adhesion, but the cell adhesion was reduced on PMB37. HepG2 adhesion was observed on PMBL. Because the cell has ASGPRs, the number of cells adhering to the PMBL polymer surfaces increased with the density of the galactose residues on the surface. By contrast, adhesion of NIH 3T3 cells to PMBL was reduced in a manner similar to that on PMB because the NIH 3T3 cells did not have ASGPRs. Cell adhesion to the PMBL surface was well regulated by ligand–receptor interactions. Furthermore, some of the cells adhering to the PMBL surface had spheroidal shapes (figure 11(c)), and similar spheroids were scattered on the surface. Although poly(BMA-co-LAMA) (PBL) has galactose residues, the adherent cells were spread as on poly(BMA). The MPC units in PMBL contribute to the spheroidal shape of HepG2 cells. The amount of albumin secreted from a cell was compared with the chemical structure of the substrate. The spheroidal cells cultured on the PMBL surface secreted much more albumin than did the spreading cells that adhered to the poly(BMA). Iwasaki et al concluded that the carbohydrate-immobilized MPC polymers produced a suitable interface for biorecognition and preservation of cell functions.
Figure 11.
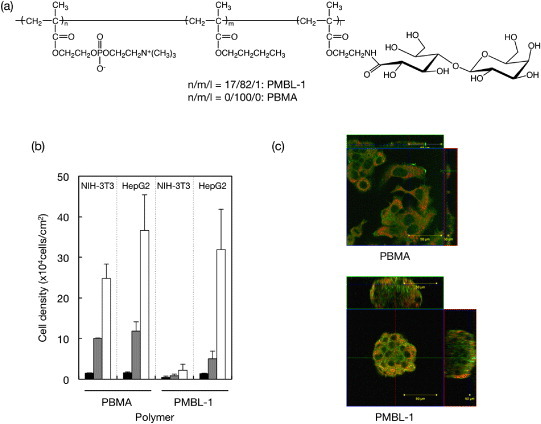
(a) Chemical structure of PMBL. (b) Density of adherent HepG2 and NIH-3T3 on PBMA and PMBL. (c) Confocal micrographs of adherent cells: (green) F-actin and (orange) phospholipid double layer.
Recent cell engineering has progressed toward regenerative medicine. Soon, cells with superior functions will be obtained and treated by the novel nano- and biotechnologies. Polymeric matrices for cell cultures are very important for achieving these goals. Aliphatic polyesters, such as poly(lactic acid) (PLA), poly(glycolic acid), and their copolymers have been commonly used as matrices for cell cultures [107–109]. These conventional biodegradable polymers are usually highly hydrophobic that hinders penetration of the matrixes by the cell culture medium [110]. Furthermore, significant and non-specific protein adsorption on conventional polymer surfaces makes it difficult to regulate the functions of adherent cells. To overcome these disadvantages of PLA, the MPC polymer is hybridized with PLA by blending [111] and grafting [112]. By controlling the concentration of MPC polymer in the MPC polymer-blended PLA, the inflammatory reaction of adherent cells could be effectively reduced with no decrease in the adherent cell number and proliferation [111].
Saito et al recently proposed an artificial stem cell niche using MPC polymers having a p-vinylboronic acid (VPBA) unit (figure 12(a)) [113]. Figure 12(b) shows phase-contrast microscopy images of adhered cells on poly(MPC-co-BMA-co-VPBA) (PMBV352; MPC:BMA:VPBA = 30:50:20 mol%), PMB37, and conventional tissue culture polystyrene (TCPS). PMB37 without the VPBA unit completely inhibited non-specific cell adhesion. On the other hand, PMBV352, having the VPBA unit, effectively induced cell adhesion despite the surface was fully covered by bioinert phosphorylcholine groups. This result indicated that the VPBA unit induced specific cell adhesion without non-specific protein interactions.
Figure 12.
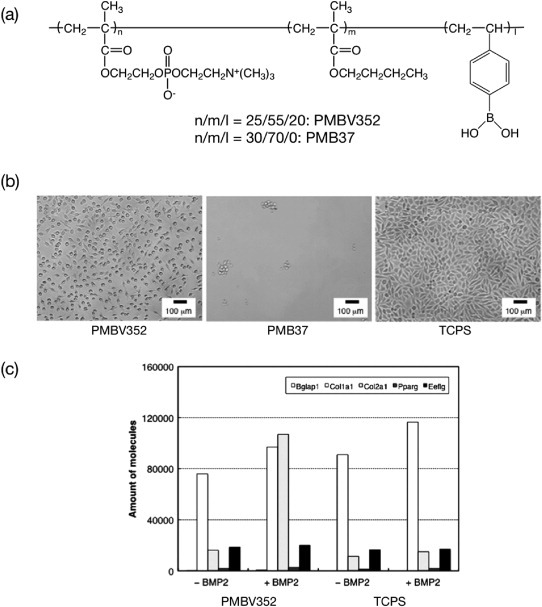
(a) Chemical structure of phospholipid PMBV. (b) Phase-contrast micrographs of adherent L929 cells after 24 h of cultivation on polymer surfaces. The bar corresponds to 100 μm. (c) RT-PCR results showing gene expression in C3H10T1/2 cells grown on polymer surfaces.
Mesenchymal stem cells, C3H10T1/2, on PMBV352 were differentiated to chondrocyte cells in the presence of a stimulation factor, BMP-2. The real-time polymerase chain reaction (RT-PCR) results are shown in figure 12(c). A chondrocyte marker, Col2a1, was highly expressed from the adhered cells onto PMBV352 with BMP2 signal rather than conventional materials. This result indicates that PMBV352 is a useful artificial stem cell niche to guide the desired differentiation property [114–116].
To extend PMBV from 2D to 3D cell cultivation, water-soluble PMBV was synthesized and mixed with poly(vinyl alcohol) (PVA) for preparing hydrogel matrices. Polymer gelation occurs spontaneously and rapidly when mixing these polymer solutions. This process can be used to encapsulate cells and proteins, and the cell activity can be maintained for more than 7 days [114]. The reaction between boronic acid and multivalent alcoholic compounds was used as the crosslinking mechanism between MPC polymers in an aqueous medium. In a tetrahedral anionic structure, boronic acid produces stable complexes with alcohol compounds including PVA, glucose, etc [117–119]. The hydrogel is reversibly dissociated by the addition of low-molecular-weight compounds such as D-glucose. A new cell maintenance system called a ‘cell-container’ based on this hydrogel was proposed. By coupling the gelation system and microfluidic technique, encapsulation of a single cell into microgel beads was achieved by Aikawa et al [120].
4. Medical devices modified with MPC polymers
4.1. Cardiovascular devices
Figure 13 outlines current applications of MPC polymers in biomedical and other fields. MPC is biocompatible due to the similarities with cell walls. Its polymerizable unit is an ideal new material for contact lenses, eye care products such as contact lens cleanser, cosmetics, toiletries, and medical devices. This biocompatibility will enable the creation of a wide range of applications as discussed in previous review articles [121–123].
Figure 13.
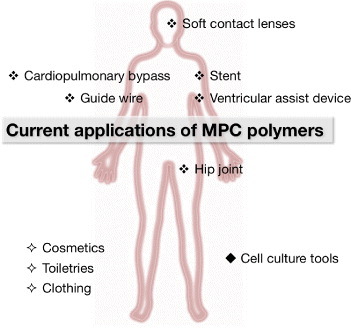
Biomedical and life science applications of MPC polymers.
As mentioned above, it has been widely shown that surface modifications with MPC polymers are effective in improving blood compatibility by suppressing protein adsorption, platelet adhesion, and platelet activation at blood-contacting surfaces. MPC polymers have recently been applied to improve blood compatibility of blood-contacting and cardiovascular devices such as medical membranes for oxygenators and hemodialysis membranes [124–129], vascular prostheses [130–133], stents [134, 135], cardiopulmonary bypasses [136–138], and ventricular assist devices (VADs) [139, 140].
Since the first intracoronary stent placement in patients in 1986, mainly for treating acute closure, coronary stenting indications have expanded remarkably. In particular, a drug-eluting stent (DES) is more efficient for reducing in-stent restenosis (ISR) and target lesion revascularization compared with bare metal stents. Because MPC polymers composed of alkyl methacrylates exhibit not only antithrombogenicity but also solute permeability, the polymer is suitable as a coating material for DES. Habara et al recently followed up long-term implantation of MPC copolymer-coated stents [141]. Seventy-five patients were treated with MPC copolymer-coated stents for de novo lesions. The data showed that the cumulative major adverse cardiac events (MACE)-free survival rate after 6 months, 18 months and 8 years was 86.3, 83.6 and 78.6%, respectively, without occurrence of thrombosis. The safely of MPC polymer-coated, small-diameter (<2 mm) stents was also evaluated by Grenadier and coworkers [142].
Some articles described the advantages of MPC polymer coating on cardiopulmonary bypass (CPB) devices for improving the preservation of platelet count and reducing the expression of proinflammatory cytokines in cardiac surgery [137, 138]. The enhanced biocompatibility of MPC polymer-coated circuits can also be combined with other anti-inflammatory strategies.
Titanium alloys are the most common base materials in the latest VADs. VADs provide circulatory support to patients in late-stage heart failures via pulsatile or rotary blood pumps. Unfortunately, VAD implantation is associated with a number of complications including infection, bleeding and thromboembolism. The concerns over biocompatibility may be a major reason why VADs are underutilized in patients with heart failure [143]. In particular, platelet adhesion and thrombus formation occur on titanium surfaces, resulting in the risk of thromboembolism or increased bleeding due to the use of anticoagulants in these patients. To improve the blood compatibility of titanium alloy surfaces, they have been coated with MPC polymers [144]. Snyder and coworkers assessed the blood compatibility of the MPC polymer (PMB37)-coated EVAHEART® VAD (Sun Medical Technology Research Co., Suwa, Japan), through animal testing (figure 14(a)) [145, 146]. Circulating platelet CD62P expression and annexin V binding were significantly lower in animals receiving MPC polymer-coated components compared with components coated with diamond-like carbon (DLC), starting from day 21 and 24 after the operation, respectively, as shown in figure 14(b). Figure 14(c) shows an EVAHEART® VAD explant, coated with an MPC polymer, 34 days after operation. The pump had no anticoagulation treatment, yet no significant thrombus formation was observed.
Figure 14.
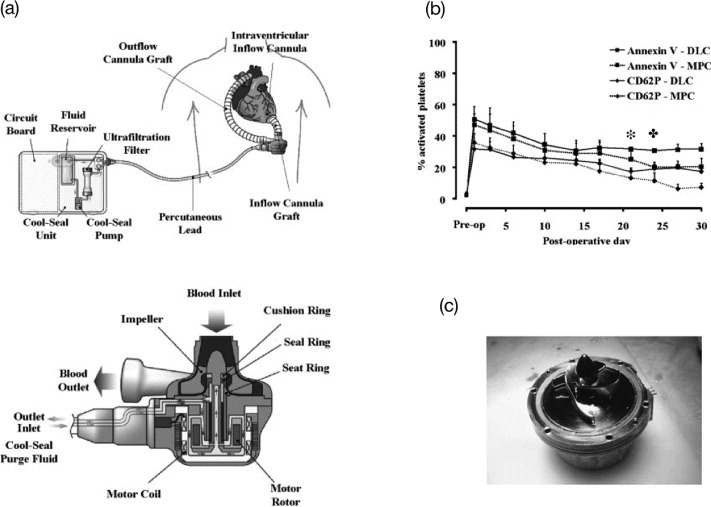
(a) EVAHEART VAD in the preferred anatomic placement for left ventricular support. The system consists of the inflow cannula, pump, outflow cannula, and lubricating purge system. The Cool-Seal purge system consists of a pump, ultrafiltration filter, and reservoir contained in a small case that can be carried by the patient. Water is recirculated through the system by the Cool-Seal pump, the ultrafiltration unit ensures sterility of fluid egressing the unit to the blood pump, and the reservoir can be exchanged or refilled as needed. (b) The concentrations of circulating activated platelets as quantified by annexin V binding or CD62P expression are shown for DLC-coated (N = 4) and MPC polymer-coated (N = 16) components. The days when the MPC values become significantly lower than the DLC values for annexin V ( ) and CD62P (
) and CD62P ( ) are marked above the curves. (c) Pump (MPC polymer coating) after 34 days of support without anticoagulation treatment. (Reprinted from [146, 147] with permission from Wiley.)
) are marked above the curves. (c) Pump (MPC polymer coating) after 34 days of support without anticoagulation treatment. (Reprinted from [146, 147] with permission from Wiley.)
In May 2005, the first clinical implantation of an EVAHEART® was successfully performed in a pilot study. EVAHEART® is an implantable centrifugal blood pump made from pure titanium; it is sized 55 × 64 mm, and weighs 420 g [147]. In Japan, the cost of the EVAHEART® has been covered by the National Health Insurance since March 2011. A clinical trial of the EVAHEART® has been started in the United States.
4.2. Orthopedic devices
Synovia (or synovial fluid) is a clear, thixotropic, lubricating fluid secreted by membranes in joint cavities, tendon sheaths and bursae, which maintains lubricity in body joints. It contains sugar, proteins and lipids. It is important for reconstruction of joint functions to secure the same lubricous state as in a living body joint. In this section, we examine the method of developing and making the neutral phosphatide of artificial synovia and achieving lubricity; the artificial synovia contains the same ingredients as living body synovia and is adsorbed on the surface of an artificial joint.
Foy et al reported that MPC polymers were useful for improving the surface lubrication of polymeric materials under wet conditions [148]. As a result of surface-initiated graft polymerization of MPC, surfaces exhibiting ultralow friction have been obtained. Ishihara et al explored a new methodology, namely photoinduced polymerization of MPC on a polyethylene surface [59]. A water-insoluble photoinitiator, benzophenone, was coated on a polyethylene substrate from an acetone solution. The substrate was then exposed to UV light in an MPC aqueous solution. MPC polymerization was initiated on the polyethylene surface, and the molecular weight of the poly(MPC) has been monitored during the irradiation. Moro et al applied this technique to the surface modification of a crosslinked ultrahigh molecular weight polyethylene (CLPE) cup of an artificial hip joint [149]. The surface showed high lubricity, which was similar to fluidic friction. The wear of the poly(MPC)-grafted CLPE was significantly reduced compared with that of bare CLPE and of CLPE grafted with other hydrophilic polymers, as shown in figure 15 [150, 151]. Furthermore, the PMPC-modified polyethylene particles did not stimulate osteoclast cells, whereas non-modified polyethylene particles generated from friction and wear of artificial joints did induce osteolysis in artificial hip joints [149]. Very recently, an innovative hip joint system with poly(MPC)-grafted CLPE cup Aquala® (Kyocera Medical Co. Ltd, Osaka, Japan) was approved in Japan; the hip joint is expected to last longer than conventional artificial hip joints.
Figure 15.
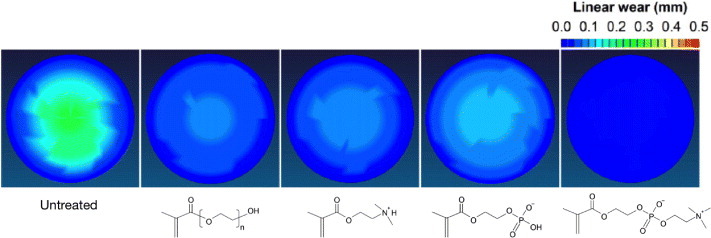
Spatial maps of wear in CLPE cups grafted with poly(MPC) or other polyelectrolytes after a hip simulator test.
Kyomoto et al reported self-initiated surface grafting with poly(MPC) on poly(ether-ether-ketone) (PEEK) [60, 152], which is a group of polymeric biomaterials with excellent mechanical properties and chemical stability. Benzophenone units were included in the PEEK molecules and then radicals were generated by UV irradiation. The PEEK specimens were soaked in MPC aqueous solution, after which photoinduced graft polymerization was performed at 60 °C for 90 min. The PMPC graft chains were generated on the PEEK surface without reducing mechanical properties of the specimens. Lately, PEEK has emerged as the leading high-performance thermoplastic candidate for replacing metal implant components, especially in the fields of orthopedics and trauma surgery. The improved nonfouling, hydrophilic, and lubricant properties of PEEK with MPC may widen the biomedical applications of PEEK.
4.3. Ophthalmology devices
The soft contact lens (SCL) is one of the most representative products of hydrogel application; it was first introduced in 1961 using poly(2-hydroxyethyl methacrylate). Since then, various poly(2-hydroxyethyl methacrylate)-based hydrogels were developed to improve lens properties. Both oxygen permeability and wettability are required for an SCL, and MPC is a suitable monomer. Biocompatible Co. Ltd (UK) has produced MPC polymer-based SCLs containing 20% of MPC (Proclear®, omafilcon A), which are now commercially available from CooperVision [153]. Recently, silicone hydrogels have been used as base materials for SLC because of their high oxygen permeability. Silicone hydrogels are normally prepared by copolymerization of macromonomers bearing silicone and hydrophilic monomers. The wettability of the silicone hydrogels is sometimes not adequate to provide the wearer with a comfortable feeling. MPC polymers have been developed for coatings to improve the surface wettability of silicone hydrogels; the surfaces also show good antifouling properties. Through the formation of interpenetrating poly(MPC) networks, Ishihara and co-workers have succeeded in preparing superhydrophilic silicone hydrogels with a high degree of transparency [52].
5. Conclusions
In this review article, the molecular design and fundamental properties of cell membrane-inspired phospholipid polymers, MPC polymers, are summarized. In addition, surface modification processes using MPC polymers to regulate biological responses are described. MPC polymers are suitable for preparing not only nonfouling surfaces but also platforms of biomolecules and living cells. Through the combination of nanotechnology and biotechnology, MPC polymer science has made considerable progress with devices such as biosensors, biochips and bioimaging tools [154]. MPC and various kinds of MPC polymers are now available commercially worldwide, and many medical devices treated with MPC polymers are used in clinics. Therefore, we expect that the novel biomedical technology based on MPC polymer science will be further explored.
References
- Singer S J. and Nicolson G L. Science. 1972;175:723. doi: 10.1126/science.175.4023.720. [DOI] [PubMed] [Google Scholar]
- Virtanen J A, Cheng K H. and Somerharju P. Proc. Natl Acad. Sci. USA. 1998;95:4964. doi: 10.1073/pnas.95.9.4964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kadoma Y, Nakabayashi N, Masuhara E. and Yamauchi J. Koubunshi Ronbunshu. 1978;35:423. doi: 10.1295/koron.35.423. (in Japanese) [DOI] [Google Scholar]
- Ishihara K, Ueda T. and Nakabayashi N. Polym. J. 1990;22:355. doi: 10.1295/polymj.22.355. [DOI] [Google Scholar]
- Ueda T, Oshida H, Kurita K, Ishihara K. and Nakabayashi N. Polym. J. 1992;24:1259. doi: 10.1295/polymj.24.1259. [DOI] [Google Scholar]
- Inoue Y, Watanabe J, Takai M. and Ishihara K. J. Biomater. Sci. Polym. Edn. 2004;15:1153. doi: 10.1163/1568562041753034. [DOI] [PubMed] [Google Scholar]
- Ma Y, Tang Y, Billingham N C, Armes S P, Lewis A L, Lloyd A W. and Salvage J P. Macromolecules. 2003;36:3475. doi: 10.1021/ma021762c. [DOI] [Google Scholar]
- Ma Y, Tang Y, Billingham N C, Armes S P. and Lewis A L. Biomacromolecules. 2003;4:864. doi: 10.1021/bm034118u. [DOI] [PubMed] [Google Scholar]
- Li Y, Armes S P, Jin X. and Zhu S. Macromolecules. 2003;36:8268. doi: 10.1021/ma035160k. [DOI] [Google Scholar]
- Ishihara K, Tsuji T, Kurosaki K. and Nakabayashi N. J. Biomed. Mater. Res. 1994;28:225. doi: 10.1002/jbm.820280213. [DOI] [PubMed] [Google Scholar]
- Iwasaki Y. and Akiyoshi K. Macromolecules. 2004;37:7637. doi: 10.1021/ma049043g. [DOI] [Google Scholar]
- Watanabe J, Eriguchi T. and Ishihara K. Biomacromolecules. 2002;3:1109. doi: 10.1021/bm025586r. [DOI] [PubMed] [Google Scholar]
- Watanabe J, Eriguchi T. and Ishihara K. Biomacromolecules. 2002;3:1375. doi: 10.1021/bm025652p. [DOI] [PubMed] [Google Scholar]
- Samanta D, McRae S, Cooper B, Hu Y, Emrick T, Pratt J. and Charles S A. Biomacromolecules. 2008;9:2891. doi: 10.1021/bm8006715. [DOI] [PubMed] [Google Scholar]
- Ishihara K. Encyclopedia Polym. Sci. Tech. 2012 at press. [Google Scholar]
- Kiritoshi Y. and Ishihara K. Polymer. 2004;45:7499. doi: 10.1016/j.polymer.2004.09.014. [DOI] [Google Scholar]
- Lobb E J, Ma I, Billingham N C, Armes S P. and Lewis A L. J. Am. Chem. Soc. 2001;123:791. doi: 10.1021/ja003906d. [DOI] [PubMed] [Google Scholar]
- Ma I, Lobb E J, Billingham N C, Armes S P, Lewis A L, Lloyd A W. and Salvage J. Macromolecules. 2002;35:9306. doi: 10.1021/ma0210325. [DOI] [Google Scholar]
- Madsen J, Warren N J, Armes S P. and Lewis A L. Biomacromolecules. 2011;12:2225. doi: 10.1021/bm200311s. [DOI] [PubMed] [Google Scholar]
- Seo J H, Matsuno R, Lee Y, Takai M. and Ishihara K. Biomaterials. 2009;30:4859. doi: 10.1016/j.biomaterials.2009.05.080. [DOI] [PubMed] [Google Scholar]
- Chen X, McRae S, Samanta D. and Emrick T. Macromolecules. 2010;43:6261. doi: 10.1021/ma101156e. [DOI] [Google Scholar]
- Xu J P, Ji J, Chen W D. and Shen J C. Macromol. Biosci. 2005;5:164. doi: 10.1002/mabi.200400139. [DOI] [PubMed] [Google Scholar]
- Iwasaki Y. and Akiyoshi K. Biomacromolecules. 2006;7:1433. doi: 10.1021/bm050917w. [DOI] [PubMed] [Google Scholar]
- Li Y, Tang Y, Narain R, Lewis A L. and Armes S P. Langmuir. 2005;21:9946. doi: 10.1021/la050356u. [DOI] [PubMed] [Google Scholar]
- Yusa S, Fukuda K, Yamamoto T, Ishihara K. and Morishima Y. Biomacromolecules. 2005;6:663. doi: 10.1021/bm0495553. [DOI] [PubMed] [Google Scholar]
- Bhuchar N, Thundat T, Sunasee R, Ishihara K. and Narain R. Bioconjug. Chem. 2012;23:75. doi: 10.1021/bc2003814. [DOI] [PubMed] [Google Scholar]
- Bhuchar N, Deng Z, Ishihara K. and Narain R. Polym. Chem. 2011;2:623. [Google Scholar]
- Yu B, Lowe A B. and Ishihara K. Biomacromolecules. 2009;10:950. doi: 10.1021/bm8014945. [DOI] [PubMed] [Google Scholar]
- Yuan J J, Schmid A, Armes S P. and Lewis A L. Langmuir. 2006;22:11022. doi: 10.1021/la0616350. [DOI] [PubMed] [Google Scholar]
- Akkahat P, Kiatkamjornwong S, Yusa S, Hoven V P. and Iwasaki Y. Langmuir. 2012;28:5872. doi: 10.1021/la204229t. [DOI] [PubMed] [Google Scholar]
- Iwasaki Y, Matsumoto A. and Yusa S. ACS Appl. Mater. Interfaces. 2012;4:3254. doi: 10.1021/am3006065. [DOI] [PubMed] [Google Scholar]
- Iwasaki Y. and Ishihara K. Anal. Bioanal. Chem. 2005;381:534. doi: 10.1007/s00216-004-2805-9. [DOI] [PubMed] [Google Scholar]
- Ishihara K, Oshida H, Endo Y, Ueda T, Watanabe A. and Nakabayashi N. J. Biomed. Mater. Res. 1992;26:1543. doi: 10.1002/jbm.820261202. [DOI] [PubMed] [Google Scholar]
- Ishihara K, Iwasaki Y. and Nakabayashi N. Polym. J. 1999;31:1231. doi: 10.1295/polymj.31.1231. [DOI] [Google Scholar]
- Yamasaki A, Imamura Y, Kurita K, Iwasaki Y, Nakabayashi N. and Ishihara K. Colloid. Surf. B. 2003;28:53. doi: 10.1016/S0927-7765(02)00130-3. [DOI] [Google Scholar]
- Iwasaki Y, Yamasaki A. and Ishihara K. Biomaterials. 2003;24:3599. doi: 10.1016/S0142-9612(03)00212-6. [DOI] [PubMed] [Google Scholar]
- Lewis A L, Hughes P D, Kirkwood L C, Leppard S W, Redman R P, Tolhurst L A. and Stratford P W. Biomaterials. 2000;21:1847. doi: 10.1016/S0142-9612(00)00055-7. [DOI] [PubMed] [Google Scholar]
- Futamura K, Matsuno R, Konno T, Takai M. and Ishihara K. Langmuir. 2008;24:10340. doi: 10.1021/la801017h. [DOI] [PubMed] [Google Scholar]
- Fukazawa K. and Ishihara K. Colloid. Surf. B. 2012;97:70. doi: 10.1016/j.colsurfb.2012.04.008. [DOI] [PubMed] [Google Scholar]
- Iwasaki Y, Takamiya M, Iwata R, Yusa S. and Akiyoshi K. Colloid. Surf. B. 2007;57:226. doi: 10.1016/j.colsurfb.2007.02.007. [DOI] [PubMed] [Google Scholar]
- Choi J, Konno T, Takai M. and Ishihara K. Biomaterials. 2012;33:954. doi: 10.1016/j.biomaterials.2011.10.006. [DOI] [PubMed] [Google Scholar]
- Choi J, Konno T, Matsuno R, Takai M. and Ishihara K. Colloid. Surf. B. 2008;67:216. doi: 10.1016/j.colsurfb.2008.08.025. [DOI] [PubMed] [Google Scholar]
- Pang S, Zhu C, Xu F, Chen C. and Ji J. Colloid. Surf. B. 2012;94:22. doi: 10.1016/j.colsurfb.2012.01.006. [DOI] [PubMed] [Google Scholar]
- Ishihara K, Hanyuda H. and Nakabayashi N. Biomaterials. 1995;16:873. doi: 10.1016/0142-9612(95)94150-J. [DOI] [PubMed] [Google Scholar]
- Ishihara K, Shibata N, Tanaka S, Iwasaki Y, Kurosaki T. and Nakabayashi N. J. Biomed. Mater. Res. 1996;32:401. doi: 10.1002/(SICI)1097-4636(199611)32:3%3C401::AID-JBM13%3E3.0.CO;2-J. [DOI] [PubMed] [Google Scholar]
- Yoneyama T, Ishihara K, Nakabayashi N, Ito M. and Mishima Y. J. Biomed. Mater. Res. 1998;43:15. doi: 10.1002/(SICI)1097-4636(199821)43:1%3C15::AID-JBM2%3E3.0.CO;2-P. [DOI] [PubMed] [Google Scholar]
- Ishihara K, Fukumoto K, Iwasaki Y. and Nakabayashi N. Biomaterials. 1999;20:1545. doi: 10.1016/S0142-9612(99)00052-6. [DOI] [PubMed] [Google Scholar]
- Hasegawa T, Iwasaki Y. and Ishihara K. Biomaterials. 2001;22:243. doi: 10.1016/S0142-9612(00)00180-0. [DOI] [PubMed] [Google Scholar]
- Ishihara K, Nishiuchi D, Watanabe J. and Iwasaki Y. Biomaterials. 2004;25:71. doi: 10.1016/S0142-9612(03)00474-5. [DOI] [PubMed] [Google Scholar]
- Iwasaki Y, Aiba Y, Morimoto N, Nakabayashi N. and Ishihara K. J. Biomed. Mater. Res. 2000;52:701. doi: 10.1002/1097-4636(20001215)52:4%3C701::AID-JBM15%3E3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- Morimoto N, Iwasaki Y, Nakabayashi N. and Ishihara K. Biomaterials. 2002;23:4881. doi: 10.1016/S0142-9612(02)00246-6. [DOI] [PubMed] [Google Scholar]
- Shimizu T, Goda T, Minoura N, Takai M. and Ishihara K. Biomaterials. 2010;31:3274. doi: 10.1016/j.biomaterials.2010.01.026. [DOI] [PubMed] [Google Scholar]
- Hoshi T, Sawaguchi T, Matsuno R, Konno T, Takai M. and Ishihara K. J. Mater. Chem. 2012;20:4897. doi: 10.1039/b925522b. [DOI] [Google Scholar]
- Hoshi T, Sawaguchi T, Matsuno R, Konno T, Takai M. and Ishihara K. J. Supercritical Fluids. 2008;44:391. doi: 10.1016/j.supflu.2007.09.020. [DOI] [Google Scholar]
- Ishihara K, Fukumoto K, Aoki J. and Nakabayashi N. Biomaterials. 1992;13:145. doi: 10.1016/0142-9612(92)90062-S. [DOI] [PubMed] [Google Scholar]
- Ishihara K, Miyazaki H, Kurosaki T. and Nakabayashi N. J. Biomed. Mater. Res. 1995;29:181. doi: 10.1002/jbm.820290207. [DOI] [PubMed] [Google Scholar]
- Hsiue G H, Lee S D, Chang P C T. and Kao C Y. J. Biomed. Mater. Res. 1998;42:134. doi: 10.1002/(SICI)1097-4636(199810)42:1%3C134::AID-JBM17%3E3.0.CO;2-L. [DOI] [PubMed] [Google Scholar]
- Iwasaki Y, Sawada S, Nakabayashi N, Khang G, Lee H B. and Ishihara K. Biomaterials. 1999;20:2185. doi: 10.1016/S0142-9612(99)00123-4. [DOI] [PubMed] [Google Scholar]
- Ishihara K, Iwasaki Y, Ebihara S, Shindo Y. and Nakabayashi N. Colloid. Surf. B. 2000;18:325. doi: 10.1016/S0927-7765(99)00158-7. [DOI] [PubMed] [Google Scholar]
- Kyomoto M. and Ishihara K. ACS Appl. Mater. Interf. 2009;1:537. doi: 10.1021/am800260t. [DOI] [PubMed] [Google Scholar]
- Feng W, Brash J. and Zhu S P. J. Polym. Sci. A: Polym. Chem. 2004;42:2931. doi: 10.1002/pola.20095. [DOI] [Google Scholar]
- Iwata R, Suk-In P, Hoven V P, Takahara A, Akiyoshi K. and Iwasaki Y. Biomacromolecules. 2004;5:2308. doi: 10.1021/bm049613k. [DOI] [PubMed] [Google Scholar]
- Feng W, Zhu S, Ishihara K. and Brash J L. Langmuir. 2005;21:5980. doi: 10.1021/la050277i. [DOI] [PubMed] [Google Scholar]
- Tsujii Y, Ohno K, Yamamoto S Goto A. and Fukuda T. Adv. Polym. Sci. 2006;197:1. [Google Scholar]
- Ishihara K, Nomura H, Mihara T, Kurita K, Iwasaki Y. and Nakabayashi N. J. Biomed. Mater. Res. 1998;39:323. doi: 10.1002/(SICI)1097-4636(199802)39:2%3C323::AID-JBM21%3E3.0.CO;2-C. [DOI] [PubMed] [Google Scholar]
- Konno K, Watanabe J. and Ishihara K. Biomacromolecules. 2004;5:342. doi: 10.1021/bm034356p. [DOI] [PubMed] [Google Scholar]
- Kinoshita K. Nucleic Acids Res. 2006;35:e3. doi: 10.1093/nar/gkl939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwata R, Sato R, Iwasaki Y. and Akiyoshi K. Colloid. Surf. B. 2008;62:288. doi: 10.1016/j.colsurfb.2007.10.018. [DOI] [PubMed] [Google Scholar]
- Chantasirichot S. and Ishihara K. Biosens. Bioelectron. 2012;38:209. doi: 10.1016/j.bios.2012.05.029. [DOI] [PubMed] [Google Scholar]
- Watanabe J. and Ishihara K. Biomacromolecules. 2006;7:171. doi: 10.1021/bm050544z. [DOI] [PubMed] [Google Scholar]
- Watanabe J. and Ishihara K. Nanobiotechnology. 2007;3:76. doi: 10.1007/s12030-008-9005-1. [DOI] [Google Scholar]
- Goto Y, Matsuno R, Konno T, Takai M. and Ishihara K. Biomacromolecules. 2008;9:3252. doi: 10.1021/bm800819r. [DOI] [PubMed] [Google Scholar]
- Ishihara K, Goto Y, Matsuno R, Inoue Y. and Konno T. Biochim. Biophys. Acta. 2012;1810:268. doi: 10.1016/j.bbagen.2010.04.008. [DOI] [PubMed] [Google Scholar]
- Matsuno R. and Ishihara K. Nano Today. 2011;6:61. doi: 10.1016/j.nantod.2010.12.009. [DOI] [Google Scholar]
- Ishihara K, Aragaki R, Ueda T, Watanabe A. and Nakabayashi N. J. Biomed. Mater. Res. 1990;24:1069. doi: 10.1002/jbm.820240809. [DOI] [PubMed] [Google Scholar]
- Ishihara K, Ziats N P, Tierney B P, Nakabayashi N. and Anderson J M. J. Biomed. Mater. Res. 1991;25:1397. doi: 10.1002/jbm.820251107. [DOI] [PubMed] [Google Scholar]
- Ishihara K, Oshida H, Endo Y, Watanabe A, Ueda T. and Nakabayashi N. J. Biomed. Mater. Res. 1993;27:1309. doi: 10.1002/jbm.820271011. [DOI] [PubMed] [Google Scholar]
- Zhang S, Benmakroha B, Rolf P, Tanaka S. and Ishihara K. Biosens. Bioelectron. 1996;11:1019. doi: 10.1016/0956-5663(96)87661-5. [DOI] [PubMed] [Google Scholar]
- Fujii K, Matsumoto H N, Koyama Y, Iwasaki Y, Ishihara K. and Takakuda K. J. Vet. Med. Sci. 2008;70:167. doi: 10.1292/jvms.70.167. [DOI] [PubMed] [Google Scholar]
- Nakai K, Morigaki K. and Iwasaki Y. Soft Matter. 2010;6:5937. doi: 10.1039/c0sm00086h. [DOI] [Google Scholar]
- Iwasaki Y, Mikami A, Kurita K, Ishihara K. and Nakabayashi N. J. Biomed. Mater. Res. 1997;36:508. doi: 10.1002/(SICI)1097-4636(19970915)36:4%3C508::AID-JBM8%3E3.0.CO;2-I. [DOI] [PubMed] [Google Scholar]
- Matyjaszewski K. and Xia J. Chem. Rev. 2001;101:2921. doi: 10.1021/cr940534g. [DOI] [PubMed] [Google Scholar]
- Matyjaszewski K. Macromolecules. 1999;32:8716. doi: 10.1021/ma991146p. [DOI] [Google Scholar]
- Kong X, Kawai T, Abe J. and Iyoda T. Macromolecules. 2001;34:1837. doi: 10.1021/ma001152h. [DOI] [Google Scholar]
- Huang W, Kim J B, Bruening M L. and Baker G L. Macromolecules. 2002;35:1175. doi: 10.1021/ma011159e. [DOI] [Google Scholar]
- Shah R R, Merreceyes D, Husemann M, Rees I, Abbott N L, Hawker C J. and Hedrick J L. Macromolecules. 2000;33:597. doi: 10.1021/ma991264c. [DOI] [Google Scholar]
- Jones D M. and Huck W T S. Adv. Mater. 2001;13:1256. doi: 10.1002/1521-4095(200108)13:16%3C1256::AID-ADMA1256%3E3.0.CO;2-B. [DOI] [Google Scholar]
- Tsujii Y, Ejaz M, Yamamoto S, Fukuda T, Shigeto K, Mibu K. and Shinjo T. Polymer. 2002;43:3837. doi: 10.1016/S0032-3861(02)00124-6. [DOI] [Google Scholar]
- Maeng I S. and Park J W. Langmuir. 2003;19:9973. doi: 10.1021/la034898s. [DOI] [Google Scholar]
- Inoue Y, Nakanishi T. and Ishihara K. React. Funct. Polym. 2011;71:350. doi: 10.1016/j.reactfunctpolym.2010.11.017. [DOI] [Google Scholar]
- Feng W, Brash J L. and Zhu S. Biomaterials. 2006;27:847. doi: 10.1016/j.biomaterials.2005.07.006. [DOI] [PubMed] [Google Scholar]
- Yoshimoto K, Hirase T, Madsen J, Armes S P. and Nagasaki Y. Macromol. Rapid. Commun. 2009;30:2136. doi: 10.1002/marc.200900484. [DOI] [PubMed] [Google Scholar]
- Kobayashi M, Yamaguchi H, Terayama Y, Wang Z, Ishihara K, Hino M. and Takahara A. Macromol. Symp. 2009;279:79. doi: 10.1002/masy.200950513. [DOI] [Google Scholar]
- Kobayashi M. and Takahara A. Chem. Rec. 2010;10:208. doi: 10.1002/tcr.201000001. [DOI] [PubMed] [Google Scholar]
- Iwasaki Y, Sawada S, Ishihara K, Khang G. and Lee H B. Biomaterials. 2002;23:3897. doi: 10.1016/S0142-9612(02)00135-7. [DOI] [PubMed] [Google Scholar]
- Sawada S, Sakaki S, Iwasaki Y, Nakabayashi N. and Ishihara K. J. Biomed. Mater. Res. A. 2003;64:411. doi: 10.1002/jbm.a.10433. [DOI] [PubMed] [Google Scholar]
- Pricer W E, Hudgin R L, Ashwell G, Stockert R J. and Morell A G. Methods Enzymol. 1974;34:688. doi: 10.1016/S0076-6879(74)34090-6. [DOI] [PubMed] [Google Scholar]
- Stockert R J. Physiol. Rev. 1995;75:591. doi: 10.1152/physrev.1995.75.3.591. [DOI] [PubMed] [Google Scholar]
- Weigel P H, Scnnaar R L, Kuhlenschmidt M S, Schmell E, Lee R T, Lee Y C. and Roseman S. J. Biol. Chem. 1979;254:10830. [PubMed] [Google Scholar]
- Kobayashi A, Akaike T, Kobayashi K. and Sumitomo H. Makromol. Chem., Rapid Commun. 1986;7:645. doi: 10.1002/marc.1986.030071005. [DOI] [Google Scholar]
- Gutsche A T, Parsons-Wingerter P, Chand D, Saltzman W M. and Leong K W. Biotechnol. Bioeng. 1994;43:801. doi: 10.1002/bit.260430815. [DOI] [PubMed] [Google Scholar]
- Sagara K. and Kim S W. J. Control. Release. 2002;79:271. doi: 10.1016/S0168-3659(01)00555-7. [DOI] [PubMed] [Google Scholar]
- Kim S H, Hoshiba T. and Akaike T. Biomaterials. 2004;25:1813. doi: 10.1016/j.biomaterials.2003.08.035. [DOI] [PubMed] [Google Scholar]
- Sato K, Miura Y, Saito N, Kobayashi K. and Takai O. Biomacromolecules. 2007;8:753. doi: 10.1021/bm061095l. [DOI] [PubMed] [Google Scholar]
- Yarema K J, Mahal L K, Bruehl R E, Rodriguez E C. and Bertozzi C R. J. Biol. Chem. 1998;273:31168. doi: 10.1074/jbc.273.47.31168. [DOI] [PubMed] [Google Scholar]
- Iwasaki Y, Takami U, Shinohara U, Kurita K. and Akiyoshi K. Biomacromolecules. 2007;8:2788. doi: 10.1021/bm700478d. [DOI] [PubMed] [Google Scholar]
- Barrera D A, Zylstra E, Lansbury P. and Langer R. Macromolecules. 1995;28:425. doi: 10.1021/ma00106a004. [DOI] [Google Scholar]
- Vunjak-Novakovic G, Obradovic B, Martin I, Bursac P M, Langer R. and Freed L E. Biotechnol. Prog. 1998;14:193. doi: 10.1021/bp970120j. [DOI] [PubMed] [Google Scholar]
- Ishaug-Riley S L, Crane-Kruger G M, Yaszemski M J. and Mikos A G. Biomaterials. 1998;19:1405. doi: 10.1016/S0142-9612(98)00021-0. [DOI] [PubMed] [Google Scholar]
- Wald L, Sarakinos A G, Lyman M D, Mikos A G, Vacanti J P. and Langer R. Biomaterials. 1993;14:270. doi: 10.1016/0142-9612(93)90117-K. [DOI] [PubMed] [Google Scholar]
- Iwasaki Y, Sawada S, Ishihara K, Khang G. and Lee H B. Biomaterials. 2002;23:3897. doi: 10.1016/S0142-9612(02)00135-7. [DOI] [PubMed] [Google Scholar]
- Liu C, Long L, Li Z, He B, Wang L, Wang J, Yuan X. and Sheng J. J. Microencapsul. 2012;29:242. doi: 10.3109/02652048.2011.646328. [DOI] [PubMed] [Google Scholar]
- Saito A, Konno T, Ikake H, Kurita K. and Ishihara K. Biomed. Mater. 2010;5:064101. doi: 10.1088/1748-6041/5/5/054101. [DOI] [PubMed] [Google Scholar]
- Konno T. and Ishihara K. Biomaterials. 2007;28:1770. doi: 10.1016/j.biomaterials.2006.12.017. [DOI] [PubMed] [Google Scholar]
- Xu Y, Jang K, Yamashita T, Tanaka Y, Mawatari K. and Kitamori T. Anal. Bioanal. Chem. 2012;402:99. doi: 10.1007/s00216-011-5296-5. [DOI] [PubMed] [Google Scholar]
- Xu Y, Sato K, Mawatari K, Konno T, Ishihara K. and Kitamori T. Adv. Mater. 2010;22:3017. doi: 10.1002/adma.201000006. [DOI] [PubMed] [Google Scholar]
- Kitano S, Koyama Y, Kataoka K, Okano T. and Sakurai Y. J. Control. Release. 1992;19:161. doi: 10.1016/0168-3659(92)90073-Z. [DOI] [Google Scholar]
- James T D, Sandanayake K R A S. and Shinkai S. Angew. Chem., Int. Ed. 1994;33:2207. doi: 10.1002/anie.199422071. [DOI] [Google Scholar]
- Miyata T, Uragami T. and Nakamae K. Adv. Drug Deliv. Rev. 2002;54:79. doi: 10.1016/S0169-409X(01)00241-1. [DOI] [PubMed] [Google Scholar]
- Aikawa T, Konno T, Takai M. and Ishihara K. Langmuir. 2012;28:2145. doi: 10.1021/la2037586. [DOI] [PubMed] [Google Scholar]
- Ishihara K. Sci. Technol. Adv. Mater. 2000;1:131. doi: 10.1016/S1468-6996(00)00012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwasaki Y, Ishihara K. and Nakabayashi N. Recent Res. Dev. Polym. Sci. 1997;1:37. [Google Scholar]
- Ishihara K. TRIP. 1997;5:401. [Google Scholar]
- Hasegawa T, Iwasaki Y. and Ishihara K. J. Biomed. Mater. Res. 2002;63:333. doi: 10.1002/jbm.10210. [DOI] [PubMed] [Google Scholar]
- Iwasaki Y, Nakabayashi N. and Ishihara K. J. Artif. Organs. 2003;6:260. doi: 10.1007/s10047-003-0234-8. [DOI] [PubMed] [Google Scholar]
- Ye S H, Watanabe J, Takai M, Iwasaki Y. and Ishihara K. Biomaterials. 2005;26:5032. doi: 10.1016/j.biomaterials.2005.01.049. [DOI] [PubMed] [Google Scholar]
- Dahe G J, Kadam S S, Sabale S S, Kadam D P, Sarkate L B. and Bellare J R. PLoS One. 2011;6:064101. doi: 10.1371/journal.pone.0025236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ueda H, Watanabe J, Konno T, Takai M, Saito A. and Ishihara K. J. Biomed. Mater. Res. A. 2006;77:19. doi: 10.1002/jbm.a.30606. [DOI] [PubMed] [Google Scholar]
- Myers G J, Johnstone D R, Swyer W J, McTeer S, Maxwell S L. and Squires C. J. Extra Corpor. Technol. 2003;35:6. [PubMed] [Google Scholar]
- Yoneyama T, Ishihara K, Nakabayashi N, Ito M. and Mishima Y. J. Biomed. Mater. Res. 1998;43:15. doi: 10.1002/(SICI)1097-4636(199821)43:1%3C15::AID-JBM2%3E3.0.CO;2-P. [DOI] [PubMed] [Google Scholar]
- Yoneyama T, Ito M, Sugihara K, Ishihara K. and Nakabayashi N. Artif. Organs. 2000;24:23. doi: 10.1046/j.1525-1594.2000.06433.x. [DOI] [PubMed] [Google Scholar]
- Yoneyama T, Sugihara K, Ishihara K, Iwasaki Y. and Nakabayashi N. Biomaterials. 2002;23:1455. doi: 10.1016/S0142-9612(01)00268-X. [DOI] [PubMed] [Google Scholar]
- Soletti L, Nieponice A, Hong Y, Ye S H, Stankus J J, Wagner W R. and Vorp D A. J. Biomed. Mater. Res. A. 2011;96:436. doi: 10.1002/jbm.a.32997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis A L, Furze J D, Small S, Robertson J D, Higgins B J, Taylor S. and Ricci D R. J. Biomed. Mater. Res. 2002;63:699. doi: 10.1002/jbm.10387. [DOI] [PubMed] [Google Scholar]
- Lewis A L, Willis S L, Small S A, Hunt S R, O'Byrne V. and Stratford P W. Biomed. Mater. Eng. 2004;14:355. [PubMed] [Google Scholar]
- Iwasaki Y, Uchiyama S, Kurita K, Morimoto N. and Nakabayashi N. Biomaterials. 2002;23:3421. doi: 10.1016/S0142-9612(02)00044-3. [DOI] [PubMed] [Google Scholar]
- De S F, Van B Y, Caes F, François K, Arnout J, Bossuyt X, Taeyans Y. and Van N G. Perfusion. 2002;17:39. doi: 10.1191/0267659102pf526oa. [DOI] [PubMed] [Google Scholar]
- Schulze C J, Han L, Ghorpade N, Etches W S, Stang L, Koshal A. and Wang S H. J. Card. Surg. 2009;24:363. doi: 10.1111/j.1540-8191.2009.00895.x. [DOI] [PubMed] [Google Scholar]
- Yamazaki K, Kihara S, Akimoto T, Tagusari O, Kawai A, Umezu M, Tomioka J, Kormos R L, Griffith B P. and Kurosawa H. Japan J. Thorac. Cardiovasc. Surg. 2002;50:461. doi: 10.1007/BF02919636. [DOI] [PubMed] [Google Scholar]
- Kitao T. Artif. Organs. 2011;35:543. doi: 10.1111/j.1525-1594.2010.01192.x. [DOI] [PubMed] [Google Scholar]
- Habara S. Int. Heart J. 2011;52:88. doi: 10.1536/ihj.52.88. [DOI] [PubMed] [Google Scholar]
- Grenadier E, Roguin A, Hertz I, Peled B, Boulos M, Nikolsky E, Amikam S, Kerner A, Cohen S. and Beyar R. Catheter Cardiovasc. Interv. 2002;55:303. doi: 10.1002/ccd.10130. [DOI] [PubMed] [Google Scholar]
- Deng M C, Edwards L B, Hertz M I, Rowe A W, Keck B M, Kormos R L, Naftel D C, Kirklin J K. and Taylor D O. J. Heart Lung Transplant. 2005;24:1182. doi: 10.1016/j.healun.2005.07.002. [DOI] [PubMed] [Google Scholar]
- Ye S H, Johnson C A, Jr, Woolley J R, Snyder T A, Gamble L J. and Wagner W R. J. Biomed. Mater. Res. A. 2009;91:18. doi: 10.1002/jbm.a.32184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kihara S. Artif. Organs. 2003;27:188. doi: 10.1046/j.1525-1594.2003.t01-2-06993.x. [DOI] [PubMed] [Google Scholar]
- Snyder T A, Tsukui H, Kihara S, Akimoto T, Litwak K N, Kameneva M V, Yamazaki K. and Wagner W R. J. Biomed. Mater. Res. A. 2007;81:85. doi: 10.1002/jbm.a.31006. [DOI] [PubMed] [Google Scholar]
- Yamazaki K, Saito S, Kihara S, Tagusari O. and Kurosawa H. Gen. Thorac. Cardiovasc. Surg . 2007;55:158. doi: 10.1007/s11748-007-0098-6. [DOI] [PubMed] [Google Scholar]
- Foy J R, Williams P F III, Powell G L, Ishihara K, Nakabayashi N. and LaBerge M. Proc. Inst. Mech. Eng. H. 1999;213:5. doi: 10.1243/0954411991534762. [DOI] [PubMed] [Google Scholar]
- Moro T, Takatori Y, Ishihara K, Konno T, Takigawa Y, Matsushita T, Chung U I, Nakamura K. and Kawaguchi H. Nat. Mater. 2004;3:829. doi: 10.1038/nmat1233. [DOI] [PubMed] [Google Scholar]
- Kyomoto M, Moro T, Konno T, Takadama H, Yamawaki N, Kawaguchi H, Takatori Y, Nakamura K. and Ishihara K. J. Biomed. Mater. Res. A. 2007;82:10. doi: 10.1002/jbm.a.31134. [DOI] [PubMed] [Google Scholar]
- Kyomoto M, Moro T, Saiga K, Hashimoto M, Ito H, Kawaguchi H, Takatori Y. and Ishihara K. Biomaterials. 2012;33:4451. doi: 10.1016/j.biomaterials.2012.03.028. [DOI] [PubMed] [Google Scholar]
- Kyomoto M, Moro T, Takatori Y, Kawaguchi H, Nakamura K. and Ishihara K. Biomaterials. 2010;31:1017. doi: 10.1016/j.biomaterials.2009.10.055. [DOI] [PubMed] [Google Scholar]
- Goda T. and Ishihara K. Expert Rev. Med. Devices. 2006;3:167. doi: 10.1586/17434440.3.2.167. [DOI] [PubMed] [Google Scholar]
- Lewis A L. and Lloyd A W. In: Biomimetic, Bioresponsive, Bioactive Materials: An Introduction to Integrating Materials with Tissues. Santin M, , Phillips G J, , editors. Hoboken, NJ Wiley; 2012. p. p 95. [Google Scholar]


