Abstract
Background
In a large number of patients with multiple gastrointestinal adenomatous polyps, no causal germline mutation can be found. Non-genetic factors may contribute to the development of adenomatous polyps in these unexplained polyposis patients.
In the development of gastrointestinal cancer, prior exposure to abdominal radiotherapy has been identified as such a factor, as it increases the gastrointestinal cancer risk in cancer survivors. A relationship of radiotherapy with intestinal polyposis, however, has not yet been described. Despite the increased cancer risk, these cancer survivors do not receive gastrointestinal screening recommendations. This case series describes three patients with adenomatous polyposis after abdominal radiotherapy.
Case presentation
Patient 1 was diagnosed with testicular cancer at the age of 31 and was treated with hemicastration, radiotherapy and chemotherapy. Thirty-nine years later, he was diagnosed with more than 30 colonic adenomas. Additionally, gastroduodenoscopy revealed a well-differentiated adenocarcinoma in the antrum of the stomach.
Patient 2 was diagnosed with a nephroblastoma at the age of 10, which was resected and treated with radiotherapy and chemotherapy. At age 36, a rectal adenocarcinoma was diagnosed and treated by radiotherapy and a total mesorectal excision. During 11 years of surveillance endoscopies, 21 colonic adenomas and three duodenal adenomas were detected.
Patient 3 was diagnosed with Hodgkin lymphoma at the age of 20 and treated with radiotherapy, followed by chemotherapy for a recurrence 3 years later. At age 62, a subtotal colectomy was performed because of colonic polyposis: 36 adenomas were detected. During screening gastro-duodenoscopy, three duodenal adenomas were detected.
In all three patients, germline analysis did not reveal a mutation in the APC and MYH genes. The gastric and rectal cancer were both microsatellite stable.
Conclusion
This report describes three patients with adenomatous polyposis, of which two developed a gastrointestinal cancer. The polyposis was not explained by a germline mutation in APC or MYH and all patients received abdominal radiotherapy. Although an etiologic role has not been established, an association between radiotherapy and intestinal adenomatous polyposis and the subsequent development of cancer seems very likely in our patients.
Keywords: Cancer survivor, Intestinal polyposis, Gastrointestinal malignancies, Radiotherapy
Background
Patients with classical familial adenomatous polyposis (FAP) present with over 100 adenomatous colonic polyps, whereas patients with attenuated FAP (AFAP) have approximately 10−100 adenomatous polyps. These syndromes are often explained by a genetic mutation in the adenomatous polyposis coli (APC) gene. A small number of cases of polyposis are caused by other germline mutations, in genes such as MutY homolog (MYH), DNA polymerase ɛ (POLE) and δ (POLD1) [1–3]. However, in a significant number of cases no mutation can be detected: 10−30 % for patients with over 100 adenomas and up to 90 % for patients with approximately 10−90 adenomas [4–6]. These adenomatous polyposis syndromes without a confirmed germline mutation can be referred to as ‘clinical FAP’ and ‘adenomatous polyposis of unknown origin’ respectively [7].
Several non-genetic factors have been associated with an increased risk of developing multiple intestinal polyps and cancer, such as high age, smoking and high body mass index [8, 9]. There is evidence in the literature that prior cancer treatment, e.g. abdominal radiotherapy and several chemotherapeutics (especially alkylating agents), also increases the risk for the development of gastrointestinal cancer [10–16]. Many cancer survivors have an increased risk of developing gastrointestinal malignancies, including survivors of Hodgkin lymphoma, testicular cancer and nephroblastoma [12, 14–22]. The reported risks of these cancer survivors range from 2- to 13-fold for gastric cancer, 11-fold for cancer in the small bowel and 2- to 7-fold for colorectal cancer.
Although it is likely that these cancers arise from precancerous lesions, an association between radiotherapy and intestinal polyposis has not yet been established and the prevalence of intestinal polyps and the pattern of radiation-induced carcinogenesis is unknown. Because of the lack of this knowledge, these cancer survivors do not receive screening or surveillance recommendations. This case series describes three Caucasian patients with intestinal adenomatous polyposis after abdominal radiotherapy for a prior malignancy.
Case presentation
Case 1
A 70-year-old asymptomatic man underwent a colonoscopy after a positive fecal immunochemical test that was performed within the national colorectal cancer (CRC) screening program. The patients’ medical history consisted of a malignant teratoma with intermediate differentiation of the right testis and a positive lymphography at the age of 31 years. He was treated with hemicastration and external beam radiotherapy (8 megavolt (MV) photons, opposed fields, one field per day) on both lungs (20 Gray (Gy)), all retroperitoneal lymph nodes (40 Gy in 24 fractions) and a surdosage on para-aortic lymph nodes (10 Gy in 5 fractions). Nine months after diagnosis, jugular lymph node and lung metastases were detected and treated with additional chemotherapy (vincristine and actinomycin, followed by vinblastine injections, which were continued for 2 years). In addition, the patient suffered from two myocardial infarctions at the age of 51. The patient was a non-smoker, drank 5 units of alcohol per week and his body mass index (BMI) was 26.1.
At colonoscopy, more than 30 adenomatous polyps with a maximum size up to 20 mm were detected, mostly located in the right colon (Fig. 1). Several of these polyps were removed and histopathology demonstrated tubular and tubulovillous adenomas with low-grade and high-grade dysplasia. As these large flat colonic polyps were numerous and difficult to detect and delineate, surveillance was considered suboptimal and a subtotal colectomy with ileosigmoidal anastomosis was advised. However, as the patient declined surgery, continuous colonoscopy surveillance is scheduled.
Fig. 1.
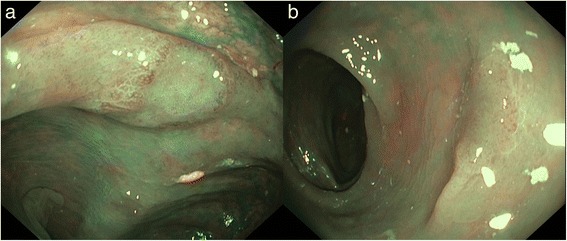
Endoscopic images of patient 1. a: Flat adenomatous lesion, non-granular type (Kudo 3 s), approximately 10 mm, descending colon (with narrow-band imaging). b: Flat adenomatous lesion, non-granular type (Kudo 3 s), approximately 10 mm, transverse colon (with narrow-band imaging)
As polyposis patients also have an increased risk for duodenal polyps and cancer, a screening gastro-duodenoscopy was performed. No duodenal polyps were identified. However, an erosive pangastritis was seen with a poorly circumscribed, slightly elevated erythematous region of approximately 4 cm, located in the greater curvature of the antrum. This region was biopsied and histopathology demonstrated adenomatous tissue with high-grade dysplasia. Endoscopic submucosal dissection of the gastric lesion was performed, and histopathology demonstrated a well-differentiated adenocarcinoma with invasion into the muscularis mucosae (pT1m3NxMx) in the background of low- and high-grade dysplasia. No additional oncologic treatment was indicated as this T1 lesion was radically resected. During this procedure, a second irregularly shaped and slightly elevated lesion of 3 mm was detected in the stomach and removed, which appeared an adenoma with low-grade dysplasia.
For further analysis of the intestinal polyposis, the patient was referred to a clinical geneticist. His father and father’s brother were both diagnosed with colorectal cancer at the age of 65 and 86 years, respectively. No germline mutation was detected in the CDH1, APC or MYH genes and the gastric adenocarcinoma was microsatellite stable with normal expression of the mismatch repair genes (MLH1, MSH2, MSH6, PMS2). A multigene custom panel containing polyposis-associated genes did not reveal any additional germline mutations in ACVRL1, AXIN2, BMPR1A, ENG, EPCAM, GREM1, MLH1, MSH2, MSH6, NTHL1, PMS2, POLD1, POLE, PTEN, SMAD4, STK11, TSC1 and TSC2 (SeqCap Ez choice, Nimblegen).
Case 2
This case describes a woman who was diagnosed with a nephroblastoma of the right kidney at the age of 10. The tumor was treated with neoadjuvant radiotherapy (20 Gy on the abdomen and a surdosage of 10 Gy on the tumor, in 19 fractions, external beam radiotherapy, ≤9 MV photons), nephrectomy, and six courses of dactinomycin and vincristine. At 18 years, an osteosarcoma of the sixth rib was resected.
At age 36, she presented with bloody diarrhea. Colonoscopy revealed a rectal adenocarcinoma, which was treated by 5x5 Gy radiotherapy and a total mesorectal excision. The moderately differentiated adenocarcinoma, ypT2N0M0, was radically resected. She was a non-smoker, who did not consume alcohol, with a BMI of 25.6.
During 11 years of follow up, surveillance colonoscopies revealed 21 adenomas, which were removed (tubular adenomas with low-grade dysplasia and one tubular adenoma with high-grade dysplasia). A screening gastroduodenoscopy was performed, resulting in the detection of one 3 mm tubular adenoma with low-grade dysplasia in the descending duodenum. Random sampling of the gastric mucosa did not reveal any metaplasia or dysplasia. During annual gastroduodenal surveillance, two additional tubulovillous adenomas with low-grade dysplasia (max. diameter 15 mm) were detected and resected in the duodenum (Fig. 2).
Fig. 2.
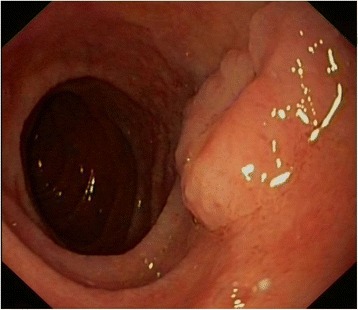
Endoscopic images of patient 2. Sessile adenomatous lesion, approximately 20 mm, duodenum
Because of the polyposis and the young onset of CRC, the patient was referred to a clinical geneticist. The only family member with cancer was her father’s brother who was diagnosed with renal cancer at unknown age. The patient was tested for germline mutations in the APC, MYH and TP53 genes, revealing no mutation. In addition, the rectal adenocarcinoma appeared microsatellite stable with normal expression of the mismatch repair genes (MLH1, MSH2, MSH6, PMS2).
Case 3
Our third patient was diagnosed with stage IIA Hodgkin lymphoma at the age 20 years. He was treated with irradiation of the neck, axillary and mediastinal nodes (35 Gy). After 3 years, he developed a recurrence and was treated with additional para-aortic and iliac radiotherapy (40 Gy in 22 fractions, external beam radiotherapy, 8 MV photons, opposed fields, one field per day). A second recurrence was treated with radiotherapy on the left axilla (40 Gy) and 12 courses of MOPP (mechlorethamine, vincristine, procarbazine, prednisone). At the age of 62, he presented with epigastric pain. The patient consumed alcohol socially and smoked 8–10 cigarettes per day. His BMI was 19.0.
A gastro-duodenoscopy was performed, which demonstrated no gastric abnormalities. In the duodenum, however, a sessile polyp of 8 mm was detected and endoscopically removed. Histopathology demonstrated a tubular adenoma with low-grade dysplasia. During follow-up, two small tubular adenomas with low-grade dysplasia of the duodenum were detected, of which one was removed. Surveillance gastroduodenoscopies are scheduled.
Because of the known association between duodenal and colonic adenomas, a colonoscopy was performed. Approximately 30 adenomas were detected throughout the colon (Fig. 3) for which a prophylactic subtotal colectomy with ileorectal anastomosis was performed. In total, this patient had a total of 36 adenomas with low-grade and high-grade dysplasia and several hyperplastic polyps (including all endoscopically resected polyps and polyps in the surgical specimen).
Fig. 3.

Endoscopic images of patient 3. a: Lateral spreading flat adenomatous lesion on top of a colonic fold, non-granular type (Kudo 3 s), approximately 20−25 mm, descending colon. b: Flat elevated adenomatous lesion, non-granular type (Kudo 3 s), approximately 15−20 mm, descending colon
The family history revealed that one second-degree relative (his mother’s brother) was diagnosed with CRC at the age of 87 years. The clinical geneticist did not detect a germline mutation in the APC or MYH gene.
Discussion
This report described three patients with intestinal adenomatous polyposis that could not be explained by a germline mutation in the APC or MYH gene, the most common mutations for this condition [23]. All three patients developed intestinal polyposis after receiving abdominal radiotherapy for different malignancies, with a latency of 26–42 years. The radiotherapy fields of these cases largely overlapped with the presence of adenomas. The development of intestinal cancer most likely occurs within an irradiated segment [10]. Abdominal radiotherapy fields deliver the highest exposure to the transverse colon (para-aortic field) and the cecum and rectosigmoid (iliac fields) [24]. In the stomach, the antrum is the site that is exposed most extensively (para-aortic field) [14].
As radiotherapy is known to accelerate gastrointestinal carcinogenesis in mouse models and increases gastrointestinal cancer risk in humans, a causal role for radiotherapy in the development of intestinal adenomatous polyposis seems plausible [10, 11, 25, 26]. These two arguments also apply for the potential causal role for alkylating agents [10, 11, 25, 27, 28]. One of our patients also received alkylating agents, including procarbazine, which may have attributed to the development of adenomatous polyposis.
When assessing the cause of adenomatous polyposis, multiple factors should be addressed. Firstly, in a large proportion of patients with less than 100 adenomas, genetic analysis of the germline mutations APC and MYH is not conclusive, which results in a diagnosis of adenomatous polyposis of unknown origin [6, 7]. The development of adenomatous polyposis could theoretically also be explained by unclassified or unrecognized mutations in polyposis-associated genes, mutations in unidentified polyposis-associated genes or mosaicism [23, 29]. In addition, the recently discovered mutations in genes such as POLE and POLD1 have not (yet) been evaluated in two of three cases [3]. Although acquired genetic aberrations have never been discovered in the etiology of polyposis, epidemiologic studies have also described non-inherited risk factors for polyposis, such as age, smoking and body mass index [8, 9]. We believe that the contribution of these factors, however, is very limited.
Only a few case reports on radiation-induced CRC have been published. They confirm that the development of normal mucosa into radiation-induced gastrointestinal adenocarcinomas is poorly investigated and hypothesize that cancer may arise from flat adenomas and/or an inflammatory background [30–32]. A recent report showed a cumulative risk of 16.4 % for developing a subsequent primary cancer beyond 30 years after treatment for Wilms tumor; 41 % of these excess cancers, all treated with abdominal radiotherapy, were attributable to digestive cancer [16]. Up to date, only one case report on radiotherapy- and chemotherapy-associated intestinal adenomatous polyposis has been published [33]. This report is limited to five childhood cancer survivors who developed adenomatous polyposis, but did not develop malignancies at follow up. This may be explained by the relative low mean age of endoscopy at 24.8 years (range 13−35) which is lower than the age of colonoscopy in our cases. This raises the possibility that the development of cancer was prevented by early detection and removal of polyps in those patients. The authors conclude that endoscopic surveillance may be indicated [33]. In our patients, earlier surveillance potentially may have prevented both the rectal and gastric cancer.
Although the significance of our case report is limited by the number and heterogeneity of cases, it does raise the question whether endoscopic surveillance is indicated to prevent cancer in patients who received abdominal radiotherapy. The increased risk of gastrointestinal malignancies seems to rise at 10 years after the primary cancer treatment [10, 13], and the risk remains increased after a follow-up period of over 30 years [12, 16–18, 34, 35], When considering these risks, one should take into account the fact that in more recent years a lowering of therapeutic radiation exposure has resulted in a decline in late-mortality among childhood cancer survivors over the last decades [35].
In our opinion, the long-term increased risk for gastrointestinal cancer in cancer survivors underscores the need for a personalized surveillance recommendation in those patients. For childhood cancer survivors who received 30 Gy or more abdominal irradiation, the American Children’s Oncology Group recommends colonoscopy surveillance every 5 years beginning at 35 years [36]. The Dutch cancer survivor guidelines do not include surveillance for gastrointestinal cancer, because important criteria for surveillance are not met, including information on the pathogenesis of radiation- and chemotherapy-induced malignancies and on risk stratification in subgroups of cancer survivors. Currently, a prospective cohort study of Hodgkin lymphoma survivors who received abdominal radiotherapy and/or chemotherapy at increased CRC risk is ongoing in the Netherlands, evaluating the efficacy of a CRC screening program in these patients. (Dutch Trial Registry NTR4961).
Conclusions
In conclusion, these three patients with intestinal adenomatous polyposis of which two developed cancer, demonstrate the potential causal role of radiotherapy. For cancer survivors who were treated with abdominal radiotherapy, a delay in the diagnosis of gastrointestinal cancer may be prevented by awareness of the increased risk. Further research on the etiology of adenomatous polyposis and gastrointestinal cancer in those cancer survivors and on optimal surveillance and prevention methods is urgently needed.
Acknowledgements
We would like to thank the patients for their collaboration.
Funding
No funding was obtained for this study.
Availability of data and materials
All the data supporting our findings are contained within the manuscript. We adhered to CARE guidelines.
Authors’ contributions
LR and FK prepared the manuscript. BB, MvL and ED were involved in clinical evaluation of the patients. All authors revised the manuscript. All authors read and approved the final manuscript.
Competing interests
The authors declare that they have no competing interests.
Consent for publication
Written informed consent was obtained from all patients for publication of this case series and any accompanying images.
Ethics approval and consent to participate
All clinical reports used for this case series had been used for patient care, and according to Dutch law, no additional consent is needed for these anonymized descriptions of cases. All patients had provided written informed consent. The current study was carried out in accordance with the Helsinki Declaration.
Authors’ information
Corresponding author: prof. dr. Dekker, gastroenterologist, Academic Medical Center, Amsterdam, the Netherlands (e.dekker@amc.uva.nl).
Abbreviations
- AFAP
Attenuated familial adenomatous polyposis
- APC
Adenomatous polyposis coli
- BMI
Body mass index
- CRC
Colorectal cancer
- FAP
Familial adenomatous polyposis
- Gy
Gray
- MV
Megavolt
- MOPP
Mechlorethamine, vincristine, procarbazine, prednisone
- MYH
MutY homolog
- POLD1
DNA polymerase δ
- POLE
DNA polymerase ɛ
Contributor Information
Lisanne Sara Rigter, Email: l.rigter@nki.nl.
Frank G. J. Kallenberg, Email: f.g.kallenberg@amc.uva.nl
Barbara Bastiaansen, Email: b.a.bastiaansen@amc.uva.nl.
Theo A. M. van Os, Email: t.a.vanos@amc.uva.nl
Floor E. van Leeuwen, Email: f.v.leeuwen@nki.nl
Monique Esther van Leerdam, Email: m.v.leerdam@nki.nl.
Evelien Dekker, Email: e.dekker@amc.uva.nl.
References
- 1.Russell AM, Zhang J, Luz J, Hutter P, Chappuis PO, Berthod CR, Maillet P, Mueller H, Heinimann K. Prevalence of MYH germline mutations in Swiss APC mutation-negative polyposis patients. Int J Cancer. 2006;118(8):1937–1940. doi: 10.1002/ijc.21470. [DOI] [PubMed] [Google Scholar]
- 2.Syngal S, Brand RE, Church JM, Giardiello FM, Hampel HL, Burt RW, American College of G ACG clinical guideline: Genetic testing and management of hereditary gastrointestinal cancer syndromes. Am J Gastroenterology. 2015;110(2):223–262. doi: 10.1038/ajg.2014.435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Valle L, Hernandez-Illan E, Bellido F, Aiza G, Castillejo A, Castillejo MI, Navarro M, Segui N, Vargas G, Guarinos C, et al. New insights into POLE and POLD1 germline mutations in familial colorectal cancer and polyposis. Hum Mol Genet. 2014;23(13):3506–3512. doi: 10.1093/hmg/ddu058. [DOI] [PubMed] [Google Scholar]
- 4.Cruz-Correa M, Giardiello FM. Familial adenomatous polyposis. Gastrointest Endosc. 2003;58(6):885–894. doi: 10.1016/S0016-5107(03)02336-8. [DOI] [PubMed] [Google Scholar]
- 5.Galiatsatos P, Foulkes WD. Familial adenomatous polyposis. Am J Gastroenterol. 2006;101(2):385–398. doi: 10.1111/j.1572-0241.2006.00375.x. [DOI] [PubMed] [Google Scholar]
- 6.Grover S, Kastrinos F, Steyerberg EW, Cook EF, Dewanwala A, Burbidge LA, Wenstrup RJ, Syngal S. Prevalence and phenotypes of APC and MUTYH mutations in patients with multiple colorectal adenomas. JAMA. 2012;308(5):485–492. doi: 10.1001/jama.2012.8780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.CBO. Heriditary colorectal cancer, National Guideline. In.; 2008.
- 8.Ben Q, An W, Jiang Y, Zhan X, Du Y, Cai QC, Gao J, Li Z. Body mass index increases risk for colorectal adenomas based on meta-analysis. Gastroenterology. 2012;142(4):762–772. doi: 10.1053/j.gastro.2011.12.050. [DOI] [PubMed] [Google Scholar]
- 9.Ji BT, Weissfeld JL, Chow WH, Huang WY, Schoen RE, Hayes RB. Tobacco smoking and colorectal hyperplastic and adenomatous polyps. Cancer Epidemiol Biomarkers Prev. 2006;15(5):897–901. doi: 10.1158/1055-9965.EPI-05-0883. [DOI] [PubMed] [Google Scholar]
- 10.Nottage K, McFarlane J, Krasin MJ, Li C, Srivastava D, Robison LL, Hudson MM. Secondary colorectal carcinoma after childhood cancer. J Clin Oncol. 2012;30(20):2552–2558. doi: 10.1200/JCO.2011.37.8760. [DOI] [PubMed] [Google Scholar]
- 11.Tukenova M, Diallo I, Anderson H, Hawkins M, Garwicz S, Sankila R, El Fayech C, Winter D, Rubino C, Adjadj E, et al. Second malignant neoplasms in digestive organs after childhood cancer: a cohort-nested case-control study. Int J Radiat Oncol Biol Phys. 2012;82(3):e383–e390. doi: 10.1016/j.ijrobp.2011.05.069. [DOI] [PubMed] [Google Scholar]
- 12.Hauptmann M, Fossa SD, Stovall M, van Leeuwen FE, Johannesen TB, Rajaraman P, Gilbert ES, Smith SA, Weathers RE, Aleman BM, et al. Increased stomach cancer risk following radiotherapy for testicular cancer. Br J Cancer. 2015;112(1):44–51. doi: 10.1038/bjc.2014.552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.van den Belt-Dusebout AW, Aleman BM, Besseling G, de Bruin ML, Hauptmann M, Veer V t MB, de Wit R, Ribot JG, Noordijk EM, Kerst JM, et al. Roles of radiation dose and chemotherapy in the etiology of stomach cancer as a second malignancy. Int J Radiat Oncol Biol Physics. 2009;75(5):1420–1429. doi: 10.1016/j.ijrobp.2009.01.073. [DOI] [PubMed] [Google Scholar]
- 14.Morton LM, Dores GM, Curtis RE, Lynch CF, Stovall M, Hall P, Gilbert ES, Hodgson DC, Storm HH, Johannesen TB, et al. Stomach cancer risk after treatment for hodgkin lymphoma. J Clin Oncol. 2013;31(27):3369–3377. doi: 10.1200/JCO.2013.50.6832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Henderson TO, Oeffinger KC, Whitton J, Leisenring W, Neglia J, Meadows A, Crotty C, Rubin DT, Diller L, Inskip P, et al. Secondary gastrointestinal cancer in childhood cancer survivors: a cohort study. Ann Intern Med. 2012;156(11):757–766. doi: 10.7326/0003-4819-156-11-201206050-00002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wong KF, Reulen RC, Winter DL, Guha J, Fidler MM, Kelly J, Lancashire ER, Pritchard-Jones K, Jenkinson HC, Sugden E, et al. Risk of adverse health and social outcomes up to 50 years after wilms tumor: the British childhood cancer survivor study. J Clin Oncol. 2016;34(15):1772–1779. doi: 10.1200/JCO.2015.64.4344. [DOI] [PubMed] [Google Scholar]
- 17.Birdwell SH, Hancock SL, Varghese A, Cox RS, Hoppe RT. Gastrointestinal cancer after treatment of Hodgkin's disease. Int J Radiat Oncol Biol Phys. 1997;37(1):67–73. doi: 10.1016/S0360-3016(96)00489-0. [DOI] [PubMed] [Google Scholar]
- 18.Travis LB, Fossa SD, Schonfeld SJ, McMaster ML, Lynch CF, Storm H, Hall P, Holowaty E, Andersen A, Pukkala E, et al. Second cancers among 40,576 testicular cancer patients: focus on long-term survivors. J Natl Cancer Inst. 2005;97(18):1354–1365. doi: 10.1093/jnci/dji278. [DOI] [PubMed] [Google Scholar]
- 19.Hodgson DC, Gilbert ES, Dores GM, Schonfeld SJ, Lynch CF, Storm H, Hall P, Langmark F, Pukkala E, Andersson M, et al. Long-term solid cancer risk among 5-year survivors of Hodgkin's lymphoma. J Clin Oncol. 2007;25(12):1489–1497. doi: 10.1200/JCO.2006.09.0936. [DOI] [PubMed] [Google Scholar]
- 20.van den Belt-Dusebout AW, de Wit R, Gietema JA, Horenblas S, Louwman MW, Ribot JG, Hoekstra HJ, Ouwens GM, Aleman BM, van Leeuwen FE. Treatment-specific risks of second malignancies and cardiovascular disease in 5-year survivors of testicular cancer. J Clin Oncol. 2007;25(28):4370–4378. doi: 10.1200/JCO.2006.10.5296. [DOI] [PubMed] [Google Scholar]
- 21.Kollmannsberger C, Hartmann JT, Kanz L, Bokemeyer C. Therapy-related malignancies following treatment of germ cell cancer. Int J Cancer. 1999;83(6):860–863. doi: 10.1002/(SICI)1097-0215(19991210)83:6<860::AID-IJC32>3.0.CO;2-L. [DOI] [PubMed] [Google Scholar]
- 22.van Leeuwen FE, Stiggelbout AM, van den Belt-Dusebout AW, Noyon R, Eliel MR, van Kerkhoff EH, Delemarre JF, Somers R. Second cancer risk following testicular cancer: a follow-up study of 1,909 patients. J Clin Oncol. 1993;11(3):415–424. doi: 10.1200/JCO.1993.11.3.415. [DOI] [PubMed] [Google Scholar]
- 23.Jung I, Gurzu S, Turdean GS. Current status of familial gastrointestinal polyposis syndromes. World J Gastrointestinal Oncol. 2015;7(11):347–355. doi: 10.4251/wjgo.v7.i11.347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Youn P, Li H, Milano MT, Stovall M, Constine LS, Travis LB. Long-term survival among Hodgkin's lymphoma patients with gastrointestinal cancer: a population-based study. Ann Oncol. 2013;24(1):202–208. doi: 10.1093/annonc/mds218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yu S, Yang M, Nam KT. Mouse models of gastric carcinogenesis. J Gastric Cancer. 2014;14(2):67–86. doi: 10.5230/jgc.2014.14.2.67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Haines J, Bacher J, Coster M, Huiskamp R, Meijne E, Mancuso M, Pazzaglia S, Bouffler S. Microsatellite instability in radiation-induced murine tumours; influence of tumour type and radiation quality. Int J Radiat Biol. 2010;86(7):555–568. doi: 10.3109/09553001003734600. [DOI] [PubMed] [Google Scholar]
- 27.Wojciechowicz K, Cantelli E, Van Gerwen B, Plug M, Van Der Wal A, Delzenne-Goette E, Song JY, De Vries S, Dekker M, Riele HT. Temozolomide increases the number of mismatch repair-deficient intestinal crypts and accelerates tumorigenesis in a mouse model of lynch syndrome. Gastroenterology. 2014;147(5):1064–1072. doi: 10.1053/j.gastro.2014.07.052. [DOI] [PubMed] [Google Scholar]
- 28.Bugni JM, Meira LB, Samson LD. Alkylation-induced colon tumorigenesis in mice deficient in the Mgmt and Msh6 proteins. Oncogene. 2009;28(5):734–741. doi: 10.1038/onc.2008.426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mongin C, Coulet F, Lefevre JH, Colas C, Svrcek M, Eyries M, Lahely Y, Flejou JF, Soubrier F, Parc Y. Unexplained polyposis: a challenge for geneticists, pathologists and gastroenterologists. Clin Genet. 2012;81(1):38–46. doi: 10.1111/j.1399-0004.2011.01676.x. [DOI] [PubMed] [Google Scholar]
- 30.Morita H, Koyama N, Tamura Y. Development of flat adenoma and superficial rectal cancer after pelvic radiation. J Clin Gastroenterology. 1998;26(3):171–174. doi: 10.1097/00004836-199804000-00004. [DOI] [PubMed] [Google Scholar]
- 31.Asayama N, Ikehara H, Yano H, Saito Y. Endoscopic submucosal dissection of multiple flat adenomas in the radiated rectum. World J Gastrointest Endosc. 2013;5(3):128–131. doi: 10.4253/wjge.v5.i3.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hugen N, van Beek JJ, de Wilt JH, Nagtegaal ID. Insight into mucinous colorectal carcinoma: clues from etiology. Ann Surgical Oncol. 2014;21(9):2963–2970. doi: 10.1245/s10434-014-3706-6. [DOI] [PubMed] [Google Scholar]
- 33.Yurgelun MB, Hornick JL, Curry VK, Ukaegbu CI, Brown EK, Hiller E, Chittenden A, Goldberg JE, Syngal S. Therapy-associated polyposis as a late sequela of cancer treatment. Clin Gastroenterol Hepatol. 2014;12(6):1046–1050. doi: 10.1016/j.cgh.2013.11.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Schaapveld M, Aleman BM, van Eggermond AM, Janus CP, Krol AD, van der Maazen RW, Roesink J, Raemaekers JM, de Boer JP, Zijlstra JM, et al. Second cancer risk up to 40 years after treatment for hodgkin's lymphoma. N Engl J Med. 2015;373(26):2499–2511. doi: 10.1056/NEJMoa1505949. [DOI] [PubMed] [Google Scholar]
- 35.Armstrong GT, Chen Y, Yasui Y, Leisenring W, Gibson TM, Mertens AC, Stovall M, Oeffinger KC, Bhatia S, Krull KR, et al. Reduction in late mortality among 5-year survivors of childhood cancer. New England J Med. 2016;374(9):833–842. doi: 10.1056/NEJMoa1510795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Landier W, Bhatia S, Eshelman DA, Forte KJ, Sweeney T, Hester AL, Darling J, Armstrong FD, Blatt J, Constine LS, et al. Development of risk-based guidelines for pediatric cancer survivors: the children’s oncology group long-term follow-up guidelines from the children’s oncology group late effects committee and nursing discipline. J Clin Oncol. 2004;22(24):4979–4990. doi: 10.1200/JCO.2004.11.032. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All the data supporting our findings are contained within the manuscript. We adhered to CARE guidelines.


