Abstract
Liver infections with hepatotropic viruses, such as hepatitis B virus and hepatitis C virus are accompanied by viral persistence and immune failure. CD8+ T cells are crucial mediators of the intrahepatic antiviral immune response. Chronic infections of the liver and other organs correlate with T-cell exhaustion. It was previously suggested that high antigen load could result in T-cell exhaustion. We aimed at elucidating the impact of different intrahepatic antigen loads on the quality of CD8+ T-cell-mediated immunity by employing an infection-free transgenic mouse model expressing ovalbumin (Ova) as the target antigen. Adoptive transfer of OT-I cells induced a transient intrahepatic immune response toward both high and low Ova levels. However, antigen clearance was achieved only in mice expressing low antigen levels. In contrast, T cells exposed to high antigen levels underwent exhaustion and became depleted, causing antigen persistence. Moreover, when functional T cells were exposed to high intrahepatic antigen levels, a complete transition toward exhaustion was observed. Thus, this study shows that the antigen expression level in the liver correlates inversely with T-cell immunity in vivo and governs the efficiency of immune responses upon antigen presentation.
Keywords: antigen load, chronicity, immune modulation, T-cell exhaustion
Introduction
Antigen-specific CD8+ T cells are crucial mediators of the antiviral responses to hepatitis B virus (HBV) and hepatitis C virus (HCV). They recognize target cells that present viral epitopes in the context of major histocompatibility complex (MHC) class I molecules and induce their killing. If such an acute response is established, clearance of virus infected cells can be achieved.1,2 However, in patients who are chronically infected with either virus, antigen-specific CD8+ T cells display an exhausted phenotype, as indicated by the expression of inhibitory surface markers and the loss of effector function.3,4 Induction of T-cell exhaustion is not restricted to the liver but is also observed in other organs, as a consequence of bacterial or viral infections.5,6 Such functionally impaired T cells can result in the establishment of a chronic infection state, which is characterized by viral persistence.6,7,8,9 Several mechanisms contribute to the failure of an immune response.10 Some studies suggest that viral load might impact T-cell potency. In clinical studies evaluating chronically HIV-infected patients, less exhausted T cells were found when the antigen load was reduced, i.e., upon successful antiviral therapy.11,12 These observations suggest that CD8+ T-cell exhaustion not only is a consequence of a failure in T-cell priming but also is affected by the antigen level. This is supported by studies showing that high levels of antigen presented by dendritic cells favor chronic progression rather than acute clearance.13,14 However, these studies relied on antigen presentation as a result of a previous infection. Thus, the immunomodulatory effects caused by the infection process cannot be excluded.
T-cell activation inside the liver remains controversial, because both the induction of tolerance that is mediated by T-cell deletion15,16 and the development of full effector function have been observed.17 One reason that tolerance is induced in the liver may be its immunosuppressive environment. This is due to the liver's physiological role and its continuous exposure to a large amount of food-derived neo antigens, which enter the liver by the portal vein and are presented by various cell types.2,18 As a consequence, the liver represents various antigenic stimuli that can potentially activate immune responses. At the same time, the liver is rich in cells that shape immunity.19 High anti-inflammatory cytokine levels drives the liver to create a tolerogenic rather than an immunogenic environment.20,21,22,23,24 Furthermore, fenestration of the endothelial layer allows for direct contact between naïve CD8+ T cells and hepatocytes, leading to full activation of CD8+ T cells, even in the absence of CD4+ T cells.17,25,26,27 These unique immunological properties of the liver require a delicate balance of mechanisms that regulate the onset of immunity. Moreover, this environment also contributes to the impaired immune response to pathogens that exclusively infect hepatocytes, in particular HBV and HCV. Indeed, these pathogens frequently cannot be cleared, leading to chronic infections.
In this study, we employed a recently established Tamoxifen (Tam)-inducible transgenic mouse model28 to investigate whether the number of antigen-presenting hepatocytes influences intrahepatic T-cell activation. In this model, the expression of a synthetic intracellular protein encoding immunogenic ovalbumin (Ova) antigen peptides is restricted to low or high hepatocyte numbers, resulting in low or high liver antigen load, respectively. Thus, we mimicked the conditions of T-cell activation in the early stages of viral infection, in which antigen presentation is predominantly mediated by hepatocytes.2 We demonstrate that only T cells exposed to low antigen levels are capable of eradicating their target cells, whereas a higher antigen load induces severe T-cell exhaustion. The results suggest that elevated antigen expression levels significantly impair the formation of a potent intrahepatic immunity and that the level of antigen in the liver might be the factor that balances successful immunity with tolerance in the liver.
Materials and methods
Ethics statement
All of the animal experiments were performed in accordance with the German Animal Welfare Law and were approved by the local government of Lower Saxony.
Mouse experiments
The Ova mice encoded a synthetic fusion protein comprising an Ova fragment (aa 246-353, including the well-characterized Kb- and Ad/b-binding epitopes, with an isoleucine to valine change at position 258). Expression was induced upon Cre-mediated inversion of the cassette (see Supplementary Figure S1 and Ref. 28). Importantly, the synthetic Ova peptide is not secreted or cross-presented by professional antigen-presenting cells (APCs).28 Mice were bred and maintained in individually ventilated cages under specific pathogen-free conditions in an in-house animal facility. All of the experiments were performed in mice aged 5–16 weeks. The Tam application (50 µg; Ratiopharm, Ulm, Germany), alanine aminotransferase (ALT) measurements and OT-I transfer are described elsewhere.29 For the adoptive transfer experiments, spleen-derived naïve OT-I CD8 T+ cells were isolated from OT-I transgenic donor mice (C57Bl/6 background) using a CD8a+ T-cell isolation kit (Miltenyi Biotec, Bergisch Gladbach, Germany) according to the manufacturer's protocol; however, phosphate-buffered saline (PBS)/1% fetal bovine serum buffer was used instead of MACS buffer. The quality and purity of the isolated OT-I cells was assessed by fluorescence-activated cell sorting analysis (Supplementary Figure S2a). For pre-activation of the OT-I cells, splenic cells from OT-I mice were isolated, as described above and cultured for 48 h in the presence of 3 µg mL−1 SIINFEKL-peptide (Supplementary Figure S2b). Afterwards, OT-I cells were separated using a CD8a+ T-cell isolation kit (Miltenyi). Cell numbers were determined using a guava easy Cyte Flow Cytometer (Merck, Millipore, Darmstadt, Germany).
In vivo CTL
Splenocytes from C57Bl/6 mice were pulsed with 10 µg of Ova peptide for 45 min at 37 °C. Labeling of CFSEhi and CFSElo cells was achieved by incubation with 3.4 × 10−4 mM and 3.4 × 10−5 mM carboxyflouresceinsuccinimidyl ester (CFSE) (Cell Trace Cell Proliferation Kit, Invitrogen, Life Technologies, Darmstadt, Germany), respectively, for 10 min at 37 °C. The cells were washed twice with PBS, and 2 × 107 cells of each cell population were mixed in 100 µL and transferred intravenously to the recipient mice. The cytotoxicity percentage was calculated as described elsewhere.30
RNA isolation and qRT-PCR
RNA isolation and qRT-PCR were performed as described previously.29 Quantification of Ova was performed with primer pairs 1a (5′-CAGGCACTCCTTTCAAGACC-3′) and 4a (5′-GCGGTTGAGGACAAACTCTT-3′) and normalized to albumin expression (Forward-primer: 5′-GACAAGGAAAGCTGCCTGAC-3′/Reverse-primer: 5′-TTCTGCAAAGTCAGCATTGG-3′).
Flow cytometry
To gate the OT-I cells, the isolated immune cells were stained with fluorescently labeled monoclonal anti-mouse antibodies: anti-CD8-PerCPCy5.5 and anti-Thy1.1-PE or APC. The CD8+/Thy1.1 double positive cells were further analyzed by staining with anti-PD-1-PE or FITC; anti-Lag-3-PE; anti-CD44-APC and anti-CD62L-PeCy7 or APC (eBioscience, Frankfurt a.M., Germany). Antibodies were diluted in 2% fetal calf serum (FCS) in PBS. Prior to staining, blocking of the Fc-receptor (CD16/CD32) was performed. To investigate the effector cytokine expression levels, the cells were adjusted to a concentration of 1 × 106 cells mL−1 if they were isolated from the liver or to a concentration of 5 × 106 cells mL−1 if they were isolated from the spleen. Cells were cultured in RPMI (5% FCS, 1% glutamine, and 1% Pen-Strep) at 37 °C in the presence of 2.5 µg mL−1 SIINFEKL peptide for 7 h. Two hours after the initial culture, cytokine secretion was impaired by adding 3 µg mL−1 Brefeldin A to the assay to block secretion of the Golgi apparatus. Following the cell surface staining, the cells were fixed with a Cytofix/Cytoperm kit (BD Biosciences, Heidelberg, Germany) and stained with anti-TNFα-APC and anti-IFNγ-FITC. The effector cytokine expression of the CD8+/Thy1.1+ cells was analyzed and compared with CD8+ single positive cells. Flow cytometry was performed using an LSR II (Becton Dickinson, Heidelberg, Germany), and the analysis was conducted with Flow Jo software (TreeStar Inc., Oregon, USA).
Statistical analysis
The data are represented as the mean of the biological replicates from the mouse groups that are specified in the figure legends. The standard deviations are indicated. The Mann–Whitney U test was used for all comparisons of two data sets. Significant differences between the sets were considered for the following p-values unless indicated otherwise: *p ≤ 0.05; **p ≤ 0.01; ***p ≤ 0.001; and ****p ≤ 0.0001.
Results
CD8+ T-cell-mediated clearance depends on the antigen load in the liver
To study the CD8+ T-cell-mediated immune responses in the liver toward different antigen loads in the absence of infection, we employed a transgenic mouse model, Ova × CreERT2.29 Ova × CreERT2 mice carry a single copy of a loxP-flanked, inversely oriented cassette that encodes an antigenic Ova fragment fused to GFP under control of the ubiquitously active Rosa26 promoter. CreERT2 is expressed from the endogenous albumin promoter.31 A single application of Tam results in transient Cre activation in hepatocytes and reversible inversion of the antigen-expressing cassette. Upon clearance of Tam, a fraction of the cells displays continuous antigen expression, whereas the remaining cells are devoid of antigen presentation (Figure S1 and Ref. 28). This was confirmed with single cell luminescence microscopy that was based on the Luc X CreERT2 mouse model displaying a homologous cassette design (Supplementary Figure S1b). Ova qRT-PCR was used to specifically quantify the functional sense mRNA expression in the livers of the Ova × CreERT2 mice. Cre-deficient Ova single transgenic control mice displayed no ova expression (Supplementary Figure S1a). In the Ova × CreERT2 mice, low expression of ova sense mRNA was observed in the absence of Tam, which corresponded to a small fraction of hepatocytes expressing the antigen and appeared to result from basal Cre activity (Supplementary Figure S1b and Ref. 29). We further determined the level of sense mRNA expression in the Ova × CreERT2 mice. We applied a single dose of Tam and waited 21 days until Tam was cleared, resulting in a fixed state of the antigen encoding cassette. A five-fold increase in the sense mRNA levels were detected compared with the non-treated mice (Supplementary Figure S1c). In the following, we refer to the basal antigen expression in the Ova × CreERT2 mice as a ‘low antigen expression' level and the Tam-induced state as ‘high antigen expression'.
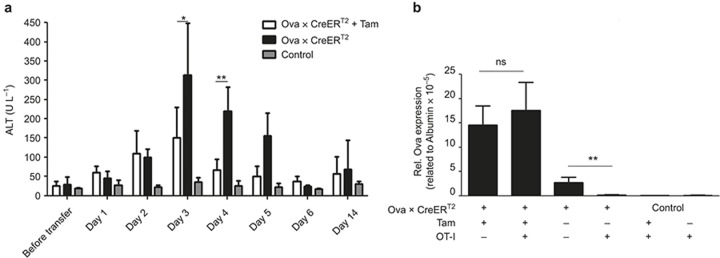
Previous characterization of the model confirmed that the Ova antigen is presented via MHC class I molecules and is recognized by OT-I cells.28,29 Importantly, no OT-I cell activation was observed upon in vitro co-culture with non-parenchymal cells (Supplementary Figure S3a). Furthermore, early T-cell activation was restricted to the liver (Supplementary Figure S3b). Additionally, no change in the CD4+ T-cell numbers was observed upon treatment of the Ova × CreERT2 mice with Tam (data not shown). Together, these data exclude aberrant antigen expression or the accidental release of antigen that might cause (cross-)presentation in other organs before the induction of CD8+ T-cell-mediated immunity. To investigate the CD8+ T-cell-mediated immune response to low and high antigen levels, 5 × 106 naïve antigen-specific OT-I cells were adoptively transferred into untreated and Tam-treated Ova × CreERT2 mice at day 21 upon initial Tam application. The CreERT2-deficient Ova mice, which were devoid of antigen presentation, were used as controls. Serum ALT levels were analyzed to monitor hepatocyte damage over time. After adoptive transfer, the serum ALT levels increased in both the Tam-treated and untreated Ova × CreERT2 mice. The liver damage in both groups peaked at day 3 and decreased to physiological levels by day 6 (Figure 1a). Interestingly, the liver damage was significantly higher in mice-presenting low antigen levels (Figure 1a).
Figure 1.
Adoptive transfer of antigen-specific T cells induces acute hepatitis. (a) Analysis of ALT activity in Ova × CreERT2 mouse plasma on the indicated days upon adoptive transfer of 5 × 106 naïve OT-I cells. ALT activity below 40 U L−1 was considered to be physiological. (b) qRT-PCR-based analysis of Ova expression within the liver of Ova × CreERT2 mice on day 13 upon adoptive transfer of OT-I cells. The means of n ≥ 4 mice/group ± SD are shown. The kinetic analysis was performed twice, analysis of day 3 was done more than 6 times.
We investigated whether the difference in the extent of acute hepatitis (as indicated by the ALT levels) was reflected by a different capacity of cells to clear the Ova antigen. To this end, the antigen expression levels in the liver were determined on day 13 by qRT-PCR. Interestingly, the Ova level within the liver was unaltered in the high antigen-expressing mice upon adoptive transfer. In contrast, adoptive transfer of OT-I cells into Ova × CreERT2 mice with low antigen expression resulted in complete elimination of Ova-expressing hepatocytes (Figure 1b). Notably, clearance of high antigen load was also not achieved upon repeated (5×) transfer of 5 × 106 cells, suggesting that the lack of clearance was not a result of limiting the T-cell numbers (data not shown). We further tested OT-I cells that were pre-activated in vitro prior to adoptive transfer. However, under these conditions, the specific T cells also failed to clear the high antigen load (Supplementary Figure S4), excluding the possibility that the lack of cytotoxic activity was a result of inefficient priming by hepatocytes.
High antigen expression within the liver is accompanied with low CD8+ T-cell numbers and severe exhaustion
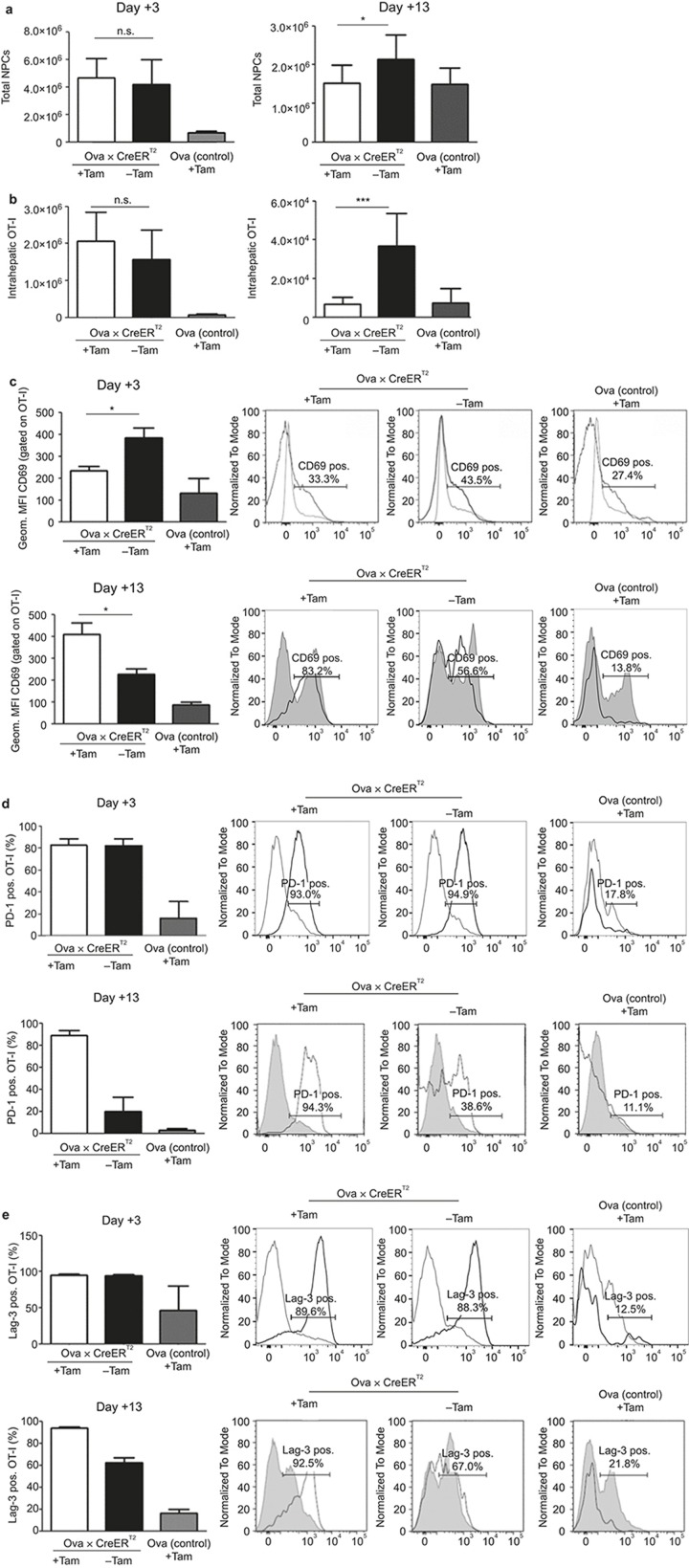
To explore the mechanisms that control the T-cell response outcomes in high and low antigen conditions, we investigated the fate of antigen-specific T cells upon intrahepatic activation. For this purpose, we determined the number of intrahepatic T cells. On day 3, i.e., at a time point when differences in acute hepatitis were observed, the total non-parenchymal cell numbers within the liver and lymphocytes in the spleen were not significantly different between the high and low antigen-expressing mice, whereas the intrahepatic OT-I cells in the high antigen-expressing mice were slightly elevated (Figure 2a and Supplementary Figure S5a). However, analysis of the intrahepatic cell numbers at day 13, i.e., a time point at which clearance of low levels of antigens was observed, revealed significantly increased liver-resident non-parenchymal cell (NPC) numbers in the low but not high antigen-expressing mice (Figure 2a). This was accompanied by increased antigen-specific CD8+ cells, as determined by examination of the Thy1.1+ OT-I cells. These cells were present in the liver of low antigen-expressing mice, whereas the corresponding numbers in the high antigen-expressing mice were significantly reduced (Figure 2b). At the same time, in the spleens of the high antigen-expressing mice, comparable total lymphocyte counts were observed, but significantly lower numbers of OT-I cells were detected (Supplementary Figure S5b).
Figure 2.
The differentiation of antigen-specific T cells depends on the amount of antigen-presenting cells in the liver. 5 × 106 OT-I cells were adoptively transferred into Ova × CreERT2 mice that were Tam-induced or untreated. On days 3 and 13, the total numbers of non-parenchymal liver cells (a) and antigen-specific Thy1.1+ OT-I cells (b) were calculated. (c–e) Phenotypic characterization of Thy1.1+ OT-I cells isolated from recipient mouse livers. The expression of the activation marker CD69, as reflected by the geometric mean fluorescence intensity (MFI), was evaluated by flow cytometry on days 3 and 13, after adoptive transfer (c). Analysis of PD-1 (d) and Lag-3 (e) exhaustion marker expression on days 3 and 13 after adoptive transfer of liver localized Thy1.1+ OT-I cells. The PD-1 and Lag-3 expression levels are depicted as the percentage of positive cells, as determined by flow cytometry. The expression levels were compared with those of non-specific CD8+ T cells. The means of n = 3–8 mice ± SD (bar diagrams) and the representative histograms are depicted from one of the four performed experiments.
To determine whether the observed hepatitis was due to T-cell activation, we investigated the phenotype of the OT-I cells during the acute phase of early activation. The marker CD69 is one of the earliest de novo synthesized molecules upon T-cell activation.32,33 Upon adoptive transfer on day 3, CD69 was up-regulated when T cells were exposed to both high and low antigen levels. In line with the more pronounced liver damage, CD69 was expressed to a greater extent in liver-localized OT-I cells from low antigen-expressing mice, whereas no differences were detected in the spleen (Figure 2c and Supplementary Figure S5c). On day 13, when the low amount of antigen was cleared, CD69 not only was still expressed on the OT-I cells from the low antigen-expressing mice but also was significantly increased on localized liver cells that were exposed to high antigen levels (Figure 2c). This suggested that ongoing triggering of T cells in response to high antigen levels was occuring. Notably, the splenic OT-I cell CD69 expression levels were not different. These results suggest that the fate of antigen-specific CD8+ T cells is determined and fixed at early time points after antigen exposure.
To elucidate why the activated T cells could not overcome a high antigen load, we characterized the cells for exhaustion markers. T-cell exhaustion correlates with the enhanced expression and high abundance of the surface markers PD-1 and Lag-3.34,35,36 Importantly, PD-1 and Lag-3 up-regulation was found on liver-localized OT-I cells in high antigen-expressing mice, whereas the OT-I cells that were primed under low antigen conditions showed that the expression levels of both markers were comparable to the Ova single transgenic, non-expressing control mice (Figure 2d–e). This was in contrast to the day 3 results, when both markers were expressed at comparable levels in the liver and the spleen (Figures 2d and S5d–e). Evaluation of the splenocytes on day 13 revealed that the OT-I cells that were primed in the high antigen level-expressing mice showed an up-regulation of both exhaustion markers, whereas the OT-I cells in the low antigen-expressing and Ova single transgenic control mice were low for PD-1 and Lag-3 expression (Figures 2d–e and S5d–e). These results suggest that a high antigen load in the liver induces systemic exhaustion of antigen-specific CD8+ T cells.
Low antigen load in the liver induces effector T cells
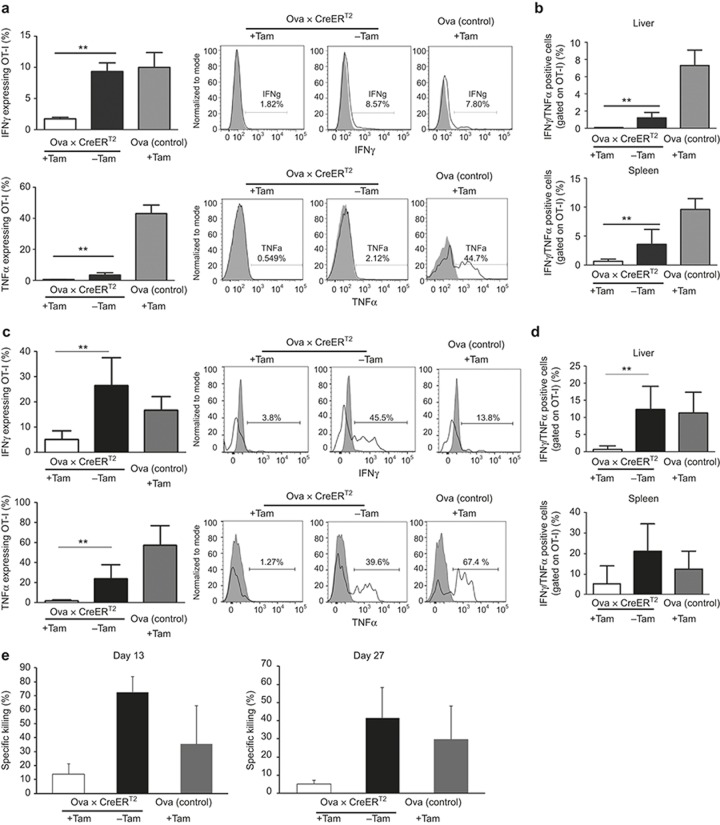
We asked to what extent the activated OT-I cells produced effector cytokines. We re-isolated adoptively transferred OT-I cells from the livers of high and low antigen-expressing mice on days 3 and 13 and stimulated them with Kb/Ova 257-264 peptide to induce cytokine expression. Quantification of effector cytokine release by liver resident OT-I cells at day 3 clearly showed significantly reduced secretion of IFNγ and TNFα from the intrahepatic and splenic OT-I cells when compared with the low antigen-primed cells or even non-activated cells from the Ova single transgenic control mice (Figure 3a and Supplementary Figure S6). This suggested an impairment in the high antigen-primed OT-I cells. Of the cytokine-producing cells, 2% of the intrahepatic and 4% of the splenic OT-I cells were double positive for IFNγ and TNFα at day 3 (Figure 3b). This phenotype was confirmed on day 13, because the OT-I cells from the low antigen-expressing mice had a higher capability of producing IFNγ and TNFα than the OT-I cells of the high antigen-expressing mice (Figure 3c). Moreover, approximately 10% of the liver resident and 20% of the splenic OT-I cells were double positive for TNFα and IFNγ when they were primed under low antigen conditions, indicating a polyfunctional phenotype. In contrast, the cells that were primed with a high antigen load did not show this phenotype (Figure 3d). These results indicate that the fate of intrahepatic CD8+ T cells is determined at early time points by the antigen load, because it not only influences the survival of antigen-specific T cells, but also influences their capability to produce effector cytokines.
Figure 3.
T-cell effector function upon adoptive transfer. (a) On day 3 after adoptive transfer, the OT-I cells were isolated from the liver and stimulated in vitro with Ova peptide to induce effector cytokine expression. Seven hours after stimulation, the cells were stained for the effector cytokines IFNγ (upper panel) and TNFα (lower panel). The expression levels were compared with those of Ova single transgenic control mice. (b) Quantification of IFNγ/TNFα double positive OT-I cells isolated from the livers and spleens of Ova × CreERT2 mice on day 3. (c) Investigation of cytokine expression in OT-I cells on day 13 following adoptive transfer. Cells from the liver were treated and analyzed as described in (a). IFNγ (upper panel) and TNFα (lower panel) release was investigated. (d) Quantification of the IFNγ/TNFα double positive OT-I cells in the livers and spleens of the respective recipient groups is shown. (e) In vivo cytotoxic activity of OT-I cells in the periphery of adoptively transferred recipient mice. OT-I recipients were challenged with Ova-pulsed (CFSEhi) and control (CFSElo) splenocytes on days 13 (left panel) and 27 (right panel). Eighteen hours after transfer, the CFSE-labeled cell populations in the spleen were analyzed by flow cytometry to investigate the specific lysis percentage. Results from n ≥ 5 mice/group ± SD from two independent experiments are shown.
We investigated whether the OT-I cells that were formed upon exposure to low antigen were protective and reactive against Ova-presenting target cells in the periphery, by employing an in vivo cytotoxic T lymphocyte (CTL) assay. After adoptive transfer of the naïve OT-I cells to either the low- or high antigen-expressing Ova × CreERT2 mice, the remaining OT-I cells were challenged by transfer of Kb/Ova 257-264 peptide-pulsed splenocytes. In accordance with the previous data, a significantly enhanced percentage of target cell killing was observed exclusively in mice with low antigen levels, when challenged at days 13 and 27 (Figure 3e and Supplementary Figure S7).
T cells that are activated by low antigen expression are depleted upon subsequent exposure to high antigen levels in the liver
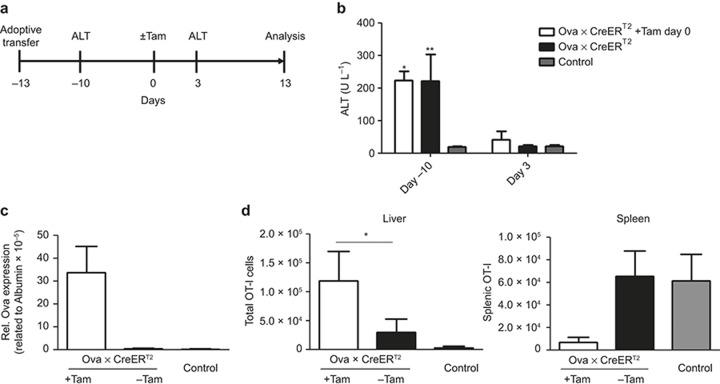
We next asked whether T cells that acquired a functional cytotoxic phenotype by low antigen conditions were also able to act efficiently once exposed to high levels of antigen. Thus, the OT-I cells were adoptively transferred into Ova × CreERT2 mice with low antigen expression (day -13, Figure 4a). When the low antigen expression was cleared (day 0), we induced high antigen expression by Tam administration and followed the liver damage by monitoring the ALT levels (Figure 4a). Importantly, the increased serum ALT level was not detectable 3 days after Tam application (Figure 4b). We evaluated the clearance efficiency by determining the Ova expression level. In agreement with the absence of hepatitis, the antigen expression levels remained unchanged and were comparable to those of the Tam-treated mice in the absence of T cells (Figure 4c). This indicated that OT-I cells that had been primed in low antigen conditions and that had successfully cleared low numbers of antigen presenting hepatocytes were not able to clear elevated numbers of antigen-presenting hepatocytes, although the intrahepatic OT-I cells were present in significantly increased numbers (Figure 4d). Phenotypic characterization of these cells showed a significantly higher expression of the exhaustion marker PD-1, suggesting that the induction of high intrahepatic antigen expression drives T-cell exhaustion in these conditions (Supplementary Figure S8). Moreover, challenging the mice with the Ova peptide-pulsed target cells on day 14 upon Tam administration led to efficient killing in the mice that did not receive Tam (Supplementary Figure S9). Thus, the high antigen load could not be reduced even if the OT-I cells that were successfully primed in vivo.
Figure 4.
The capacity of OT-I cells primed inflow antigen conditions and subsequently exposed to high antigen loads. (a) The experimental scheme. (b) Serum ALT levels upon adoptive transfer of 5 × 106 naïve OT-I cells into low antigen-expressing mice on days -10 and 3 upon induction of a high antigen load. The intrahepatic Ova expression levels were assessed with qRT-PCR 14 days following Tam application (c). At the same time, the numbers of Thy1.1+ OT-I cells in the liver (left panel) and spleen (right panel) were calculated (d). Mean of n ≥ 3 mice/group ± SD is shown for one of two independent experiments.
Together, these results indicate that high antigen levels in the liver downmodulate intrahepatic immunity.
Discussion
It is well accepted that the tolerogenic milieu of the liver has a severe impact on the outcome of T-cell responses. Different reports have described the fate of T cells upon priming in the liver and have demonstrated poor effector functions and the subsequent deletion of effector T cells,15,37,38 a phenomenon that is attributed to the tolerogenic environment of the liver. Once T cells are activated and mediate killing of an antigen-presenting hepatocyte, the antigens are subsequently exposed via cross-presentation by professional APCs, such as Kupffer cells and liver sinusoidal endothelial cells (LSECs), which can efficiently activate T cells.
In this study, we elucidated the impact of an intrahepatic antigen, by employing a recently established mouse model Ova × CreERT2 for mosaic induction of hepatocyte-specific antigen expression, thereby mimicking the early events of infection. In this respect, it is of note that in our mouse model, the level of Ova expression is relatively low; indeed, OVA expression is 80–100-fold less than actin.29
In the non-induced state, only low numbers of hepatocytes expressed the antigen. Tam-mediated activation of CreERT2 activated transgene expression in a higher number of hepatocytes compared with the non-induced state. Thereby, we established a highly controlled setting in which CD8+ T cells were challenged with different antigen concentrations in the absence of innate inflammation. Indeed, these settings mimic aspects of early HBV infection events, which are characterized by the absence of an innate defense.39,40
We observed that the outcome of the CD8+ T-cell response is critically dependent on the antigen expression level in liver. Adoptive transfer of OT-I cells to both the low and high antigen-expressing Ova × CreERT2 mice was accompanied by liver pathology, as demonstrated by increased serum ALT levels. However, the extent of tissue damage inversely correlated with the antigen expression level (Figure 1a). Importantly, efficient clearance was achieved only in mice expressing antigen at low levels. The T cells in the high antigen-expressing mice developed an exhausted phenotype, as indicated by increased PD-1 and Lag-3 expression levels, which are markers that have previously been correlated with T-cell exhaustion during chronic infections.7,34,36,41 Exhaustion of antigen-specific T cells is characterized by the progressive loss of effector cytokine production and, in severe cases, by the deletion of antigen-specific cells.6,8,9 These characteristics were observed only if T cells were exposed to high levels of antigen-presenting hepatocytes (Figures 2 and 3). Importantly, in low antigen conditions, the OT-I cells were activated and functional in the liver milieu, as demonstrated by the capacity of the T cells to release effector cytokines and perform targeted cell killing (Figure 3 and Supplementary Figures S6 and S7). This indicated that the CD8+ T-cell response in the liver of low antigen-expressing mice supports the development of a functional effector T-cell population. However, this capacity was lost when the T cells were subsequently challenged with a high antigen load (Figure 4). The functional T-cell phenotype that developed in low antigen conditions reverted completely upon increasing the amount of antigen-presenting hepatocytes to the high levels (compared with Supplementary Figure S8).
Thus, we conclude that elevated levels of antigen expression impair a functional T-cell response in the liver. Furthermore, adoptive transfer of in vitro pre-activated OT-I cells was accompanied by failure of antigen clearance, indicating severe exhaustion and depletion of antigen-specific T cells (Supplementary Figure S4). It is worth mentioning that this result does not exclude the presence of tolerance mechanisms in vivo; however, it indicates that it is antigen quantity that predominantly governs the outcome of immune responses within the liver. To investigate whether the immunity outcomes depended on the quantity of antigen-specific CD8+ T cells, we increased the number of adoptively transferred OT-I cells five-fold. Notably, even under these conditions, antigen clearance was not efficient when high antigen levels were expressed (data not shown). This suggests that the lack of antigen clearance in the Tam-induced mice was not just a result of limited specific T-cell numbers. Rather, it seems that the T cells were specifically impaired under these conditions. Still, in a less physiological Ova × CreERT2 × OT-I mouse model, which provides endogenous OT-I cells, the induction of intrahepatic immunity and the subsequent clearance of the high antigen levels can be achieved.29 This proves that the clearance of the induced hepatocyte fraction expressing the Ova antigen is indeed possible under certain conditions. However, the Ova × CreERT2 × OT-I mice are characterized by the continuous production of large numbers of naïve OT-I cells, a setting that does not reflect natural conditions and might override regulatory mechanisms. Although our experiments show no cross-presentation of the antigen by other APCs prior to the T-cell-mediated killing, we cannot formally exclude this possibility. Still, this does not alter our conclusions concerning the dose-dependent impairment of T cells.
Upon adenoviral infection, Tay et al. have recently demonstrated that the efficiency of antigen clearance by CD8+ T cells inversely correlates with the amount of infected hepatocytes. They further have demonstrated that this mechanism is also active in the absence of cross-presentation.27 Here, we show that the antigen quantity governs the outcome of T-cell responses in the liver, even in ‘sterile' conditions, i.e., in the absence of infection-mediated pro-inflammatory conditions. This particularly rules out that the described dose effect critically depends on infection-associated effects, such as inflammation.
The induction of T-cell exhaustion upon encountering high concentrations of antigen suggests that the host immune system possesses a self-regulatory mechanism that protects against excessive responses that might be associated with severe and life-threatening liver destruction. This may also contribute to the lack of an efficient immune response in HBV- or HCV-infected patients who cannot clear viral infections. It remains to be elucidated whether the amount of antigen is the only factor that triggers the subsequent fate of antigen-specific CD8+ T cells or whether other components of the immune system contribute to the regulation of the immune response upon initial liver damage and ongoing infection.
Some recent studies have demonstrated that antigen-specific CD8+ T-cell exhaustion can occur as a consequence of elevated antigen-presenting dendritic cell numbers.13,14 Additionally, the contribution of LSECs in T-cell activation remains controversial: on the one hand, LSECs have been demonstrated to perform cross-presentation of hepatocyte-derived antigen. However, on the other hand, they are able to dampen the response of previously activated T cells.42
Our findings might reflect a general concept for the development of chronic infections. Here, we demonstrate a significant impact of the antigen level itself, which might help in the design or improvement of therapeutic and preventive vaccination strategies.
Acknowledgments
The authors wish to acknowledge Dr. Andrea Kröger for discussions and Viola Weichelt for cell isolation support. This work was supported by the HZI Grad School, the Hannover Biomedical Research School (HBRS) and the Center for Infection Biology (ZIB). The authors acknowledge funding from the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) for the Cluster of Excellence REBIRTH (From Regenerative Biology to Reconstructive Therapy) and Wi2648, as well as funding from the Helmholtz Association cross-program activity ‘Metabolic Dysfunction and Human Disease' and the Helmholtz Initiative for Synthetic Biology.
Footnotes
Supplementary information of this article can be found on the Cellular & Molecular Immunology's website (http://www.nature.com/cmi).
Supplementary Information
References
- Thomson AW, Knolle PA. Antigen-presenting cell function in the tolerogenic liver environment. Nat Rev 2010; 10: 753–766. [DOI] [PubMed] [Google Scholar]
- Protzer U, Maini MK, Knolle PA. Living in the liver: hepatic infections. Nat Rev 2012; 12: 201–213. [DOI] [PubMed] [Google Scholar]
- Rehermann B. Hepatitis C virus versus innate and adaptive immune responses: a tale of coevolution and coexistence. J Clin Invest 2009; 119: 1745–1754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rehermann B. Pathogenesis of chronic viral hepatitis: differential roles of T cells and NK cells. Nat Med 2013; 19: 859–868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Day CL, Abrahams DA, Lerumo L, Janse van Rensburg E, Stone L, O'Rie T et al. Functional capacity of Mycobacterium tuberculosis-specific T cell responses in humans is associated with mycobacterial load. J Immunol 2011; 187: 2222–2232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Virgin HW, Wherry EJ, Ahmed R. Redefining chronic viral infection. Cell 2009; 138: 30–50. [DOI] [PubMed] [Google Scholar]
- Wherry EJ, Ha SJ, Kaech SM, Haining WN, Sarkar S, Kalia V et al. Molecular signature of CD8+ T cell exhaustion during chronic viral infection. Immunity 2007; 27: 670–684. [DOI] [PubMed] [Google Scholar]
- Zajac AJ, Blattman JN, Murali-Krishna K, Sourdive DJ, Suresh M, Altman JD et al. Viral immune evasion due to persistence of activated T cells without effector function. J Exp Med 1998; 188: 2205–2213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wherry EJ, Blattman JN, Murali-Krishna K, van der Most R, Ahmed R. Viral persistence alters CD8 T-cell immunodominance and tissue distribution and results in distinct stages of functional impairment. J Virol 2003; 77: 4911–4927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blackburn SD, Shin H, Haining WN, Zou T, Workman CJ, Polley A et al. Coregulation of CD8+ T cell exhaustion by multiple inhibitory receptors during chronic viral infection. Nat Immunol 2009; 10: 29–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Streeck H, Brumme ZL, Anastario M, Cohen KW, Jolin JS, Meier A et al. Antigen load and viral sequence diversification determine the functional profile of HIV-1-specific CD8+ T cells. PLoS Med 2008; 5: e100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Day CL, Kiepiela P, Leslie AJ, van der Stok M, Nair K, Ismail N et al. Proliferative capacity of epitope-specific CD8 T-cell responses is inversely related to viral load in chronic human immunodeficiency virus type 1 infection. J Virol 2007; 81: 434–438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richter K, Brocker T, Oxenius A. Antigen amount dictates CD8+ T-cell exhaustion during chronic viral infection irrespective of the type of antigen presenting cell. Eur J Immunol 2012; 42: 2290–2304. [DOI] [PubMed] [Google Scholar]
- Mueller SN, Ahmed R. High antigen levels are the cause of T cell exhaustion during chronic viral infection. Proc Natl Acad Sci U S A 2009; 106: 8623–8628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bertolino P, Heath WR, Hardy CL, Morahan G, Miller JF. Peripheral deletion of autoreactive CD8+ T cells in transgenic mice expressing H-2Kb in the liver. Eur J Immunol 1995; 25: 1932–1942. [DOI] [PubMed] [Google Scholar]
- Bowen DG, Zen M, Holz L, Davis T, McCaughan GW, Bertolino P. The site of primary T cell activation is a determinant of the balance between intrahepatic tolerance and immunity. J Clin Invest 2004; 114: 701–712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wuensch SA, Pierce RH, Crispe IN. Local intrahepatic CD8+ T cell activation by a non-self-antigen results in full functional differentiation. J Immunol 2006; 177: 1689–1697. [DOI] [PubMed] [Google Scholar]
- Bogdanos DP, Gao B, Gershwin ME. Liver immunology. Compr Physiol 2013; 3: 567–598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ebrahimkhani MR, Mohar I, Crispe IN. Cross-presentation of antigen by diverse subsets of murine liver cells. Hepatology 2011; 54: 1379–1387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calne RY, Sells RA, Pena JR, Davis DR, Millard PR, Herbertson BM et al. Induction of immunological tolerance by porcine liver allografts. Nature 1969; 223: 472–476. [DOI] [PubMed] [Google Scholar]
- Pillarisetty VG, Shah AB, Miller G, Bleier JI, DeMatteo RP. Liver dendritic cells are less immunogenic than spleen dendritic cells because of differences in subtype composition. J Immunol 2004; 172: 1009–1017. [DOI] [PubMed] [Google Scholar]
- Chen Y, Jiang G, Yang HR, Gu X, Wang L, Hsieh CC et al. Distinct response of liver myeloid dendritic cells to endotoxin is mediated by IL-27. J Hepatol 2009; 51: 510–519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Creus A, Abe M, Lau AH, Hackstein H, Raimondi G, Thomson AW. Low TLR4 expression by liver dendritic cells correlates with reduced capacity to activate allogeneic T cells in response to endotoxin. J Immunol 2005; 174: 2037–2045. [DOI] [PubMed] [Google Scholar]
- Luth S, Huber S, Schramm C, Buch T, Zander S, Stadelmann C et al. Ectopic expression of neural autoantigen in mouse liver suppresses experimental autoimmune neuroinflammation by inducing antigen-specific Tregs. J Clin Invest 2008; 118: 3403–3410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bertolino P, Trescol-Biemont MC, Rabourdin-Combe C. Hepatocytes induce functional activation of naive CD8+ T lymphocytes but fail to promote survival. Eur J Immunol 1998; 28: 221–236. [DOI] [PubMed] [Google Scholar]
- Derkow K, Muller A, Eickmeier I, Seidel D, Rust Moreira MV, Kruse N et al. Failure of CD4 T-cells to respond to liver-derived antigen and to provide help to CD8 T-cells. PLoS One 2011; 6: e21847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tay SS, Wong YC, McDonald DM, Wood NA, Roediger B, Sierro F et al. Antigen expression level threshold tunes the fate of CD8 T cells during primary hepatic immune responses. Proc Natl Acad Sci USA 2014; 111: E2540–E2549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandhu U, Cebula M, Behme S, Riemer P, Wodarczyk C, Metzger D et al. Strict control of transgene expression in a mouse model for sensitive biological applications based on RMCE compatible ES cells. Nucleic Acids Res 2011; 39: e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cebula M, Ochel A, Hillebrand U, Pils MC, Schirmbeck R, Hauser H et al. An inducible transgenic mouse model for immune mediated hepatitis showing clearance of antigen expressing hepatocytes by CD8+ T cells. PLoS One 2013; 8: e68720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheehy ME, McDermott AB, Furlan SN, Klenerman P, Nixon DF. A novel technique for the fluorometric assessment of T lymphocyte antigen specific lysis. J Immunol Methods 2001; 249: 99–110. [DOI] [PubMed] [Google Scholar]
- Schuler M, Dierich A, Chambon P, Metzger D. Efficient temporally controlled targeted somatic mutagenesis in hepatocytes of the mouse. Genesis 2004; 39: 167–172. [DOI] [PubMed] [Google Scholar]
- Cebrian M, Yague E, Rincon M, Lopez-Botet M, de Landazuri MO, Sanchez-Madrid F. Triggering of T cell proliferation through AIM, an activation inducer molecule expressed on activated human lymphocytes. J Exp Med 1988; 168: 1621–1637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castellanos Mdel C, Lopez-Giral S, Lopez-Cabrera M, de Landazuri MO. Multiple cis-acting elements regulate the expression of the early T cell activation antigen CD69. Eur J Immunol 2002; 32: 3108–3117. [DOI] [PubMed] [Google Scholar]
- Isogawa M, Chung J, Murata Y, Kakimi K, Chisari FV. CD40 activation rescues antiviral CD8 (+) T cells from PD-1-mediated exhaustion. PLoS Pathog 2013; 9: e1003490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackson SR, Berrien-Elliott MM, Meyer JM, Wherry EJ, Teague RM. CD8+ T cell exhaustion during persistent viral infection is regulated independently of the virus-specific T cell receptor. Immunol Invest 2013; 42: 204–220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paley MA, Kroy DC, Odorizzi PM, Johnnidis JB, Dolfi DV, Barnett BE et al. Progenitor and terminal subsets of CD8+ T cells cooperate to contain chronic viral infection. Science 2012; 338: 1220–1225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klugewitz K, Blumenthal-Barby F, Schrage A, Knolle PA, Hamann A, Crispe IN. Immunomodulatory effects of the liver: deletion of activated CD4+ effector cells and suppression of IFN-gamma-producing cells after intravenous protein immunization. J Immunol 2002; 169: 2407–2413. [DOI] [PubMed] [Google Scholar]
- Holz LE, Benseler V, Bowen DG, Bouillet P, Strasser A, O'Reilly L et al. Intrahepatic murine CD8 T-cell activation associates with a distinct phenotype leading to Bim-dependent death. Gastroenterology 2008; 135: 989–997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guidotti LG, Chisari FV. Immunobiology and pathogenesis of viral hepatitis. Annu Rev Pathol 2006; 1: 23–61. [DOI] [PubMed] [Google Scholar]
- Wieland SF, Chisari FV. Stealth and cunning: hepatitis B and hepatitis C viruses. J Virol 2005; 79: 9369–9380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barber DL, Wherry EJ, Masopust D, Zhu B, Allison JP, Sharpe AH et al. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature 2006; 439: 682–687. [DOI] [PubMed] [Google Scholar]
- Wohlleber D, Kashkar H, Gartner K, Frings MK, Odenthal M, Hegenbarth S et al. TNF-induced target cell killing by CTL activated through cross-presentation. Cell Rep 2012; 2: 478–487. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.