Abstract
Due to concerns about global warming, there is interest in 2,3,3,3-Tetrafluoroprop-1-ene (R1234yf) and trans-1,3,3,3-Tetrafluoropropene (R1234ze(E)) as potential replacements for refrigerants with high global warming potential (GWP). In this manuscript we survey available data and provide viscosity correlations that cover the entire fluid range including vapor, liquid, and supercritical regions. The correlation for R1234yf is valid from the triple point (220 K) to 410 K at pressures up to 30 MPa, and the correlation for R1234ze(E) is valid from the triple point (169 K) to 420 K at pressures up to 100 MPa. The estimated uncertainty for both correlations at a 95 % confidence level is 2 % for the liquid phase over the temperature range 243 K to 363 K at pressures to 30 MPa, and 3 % for the gas phase at atmospheric pressure.
Keywords: 2,3,3,3-Tetrafluoroprop-1-ene; trans-1,3,3,3-Tetrafluoropropene; correlation; R1234yf; R1234ze(E); viscosity
1. Introduction
Concerns about climate change and global warming have led to interest in searching for replacement refrigerants with low global warming potential (GWP). R134a (1,1,1,2-tetrafluoroethane) has been widely used in automotive air conditioning systems, but has a high GWP. In response to this, European directive 2006/40/EC requires all new car platforms for sale in Europe to use refrigerants in their air conditioning systems with a GWP below 150. Other common refrigerants with high GWP’s will need to be considered for phase out as well. The present search for alternatives considers the possibility of using hydrofluorethers (HFE’s), and also unsaturated hydrofluorocarbons such as hydrofluoroolefins (HFO’s). Systematic searches have been done to identify if there may be other classes of compounds that may be suitable as well (Kazakov et al., 2012; McLinden et al., 2016). Viable candidate fluids must have other properties in addition to low GWP; they must have zero or very low ozone depletion potential, chemical stability, low toxicity, appropriate thermophysical properties, and must satisfy safety concerns regarding flammability. McLinden et al. (McLinden et al., 2016) identified 28 low-GWP fluids as potential pure-fluid replacements for unitary AC systems. These fluids included halogenated alkanes and oxygenates, hydrocarbons, halogenated nitrogen and sulfur compounds, inorganic compounds, and HFO’s. HFO’s were the largest group, with 10 compounds. Two promising HFO’s identified were 2,3,3,3-Tetrafluoroprop-1-ene (R1234yf, CAS 754-12-1) and trans-1,3,3,3-Tetrafluoropropene (R1234ze(E), CAS 29118-24-9).Kedzierski et al. (Kedzierski et al., 2015) evaluated cycle performance in a typical air conditioner for 39 refrigerants identified as low-GWP alternatives, and found R1234yf as one of the top three candidates. Mota-Babiloni and coworkers (Mota-Babiloni et al., 2015a) examined mixtures of R1234ze(E) and R134a as a drop-in replacement for R134a in a vapor compression plant. They also (Mota-Babiloni et al., 2015b) analyzed mixtures with R1234yf and R1234ze(E) as alternatives for refrigeration and HVAC systems and presented a review of R1234ze(E) systems for HVACR applications (Mota-Babiloni et al., 2015c). Other studies have focused on evaluating low-GWP fluids for power generation in organic Rankine cycles (ORC). Luo et al. (Luo et al., 2015) found mixtures with R1234yf and the pure fluids R1234ze(E) as promising candidates. Liu et al. (Liu et al., 2014) investigated the performance of eight different HFO’s including R1234yf and R1234ze(E) for application as working fluids in ORC’s for geothermal power applications.
In order to efficiently design refrigeration systems and power cycles, accurate models for thermophysical properties of the working fluids are needed. For R1234yf there currently are two high-accuracy equation of state formulations (Richter et al., 2011); Akasaka, 2011) available, and three for R1234ze(E) (Thol and Lemmon, 2016; McLinden et al., 2010; Akasaka, 2011). Wide-ranging correlations for the thermal conductivity of both fluids have been published (Perkins and Huber, 2011), and the surface tension of R1234ze(E) has been investigated (Tanaka and Higashi, 2013). Mulero et al. (Mulero et al., 2012) have developed a correlation for the surface tension R1234yf based on available literature data. However, there currently are few wide-ranging correlations for the viscosity of R1234yf or R1234ze(E). There is one extended corresponding states (ECS) model for the viscosity of R1234yf (Akasaka, 2010) and unpublished ECS models for both R1234yf and R1234ze(E) in REFPROP (Lemmon et al., 2013), and most recently a free-volume based model (He et al., 2015). In this work we develop correlations for the viscosity as a function of temperature and density that are valid over a wide range of fluid states including gas, liquid, and supercritical regions for R1234yf and R1234ze(E) and compare with known experimental data.
2. Viscosity Correlation
The viscosity η can be expressed (Dymond et al., 1996) as the sum of four independent contributions, as
| (1) |
where ρ is the molar density and T is the absolute temperature. There is theoretical guidance for the first two terms, η0(T) and η1(T), that will be discussed below. The critical enhancement term, Δηc(ρ,T), is confined to a small region, becoming relevant only at temperatures and densities very close to the critical point, and will not be included in this work. Finally, the term Δη(ρ,T), the residual term, must be handled empirically by fitting experimental data.
Table 1 summarizes, to the best of our knowledge, the experimental measurements of the viscosity of R1234yf and R1234ze(E), including sample purities, experimental method, and the uncertainties ascribed by the original authors. The sets marked in bold face were used in determining the coefficients of the correlations; other sets are included only for comparison purposes. The distribution of the experimental data and the phase boundary in the T,p plane are shown in Figures 1 and 2. For R1234yf, the densities and the phase boundary are calculated from the equation of state of Richter et al.(Richter et al., 2011) This equation is valid from 220 K (the triple-point temperature) to 410 K at pressures up to 30 MPa. We also incorporate the critical parameters from this equation of state, given in Table 2. For R1234ze(E), we calculate densities and the phase boundary with the equation of state of Thol and Lemmon (Thol and Lemmon, 2016). This equation is valid from 169 K (the triple-point temperature) to 420 K at pressures up to 100 MPa. The critical temperature and critical density are given in Table 2.
Table 1.
Experimental Measurements of the Viscosity of R1234yf and R1234ze(E)
| 1st author | Year Publ. |
Technique employeda |
Purity (%) |
Uncertainty (%) |
No. of data |
Temperature range (K) |
Pressure range (MPa) |
|---|---|---|---|---|---|---|---|
| R1234yf | |||||||
| Dang | 2015a | MovP | 99.9 | 1 | 25 | 283–321 | 0.6–1.3 |
| Dang,b | 2015b | FB | 99 | 1.5 | 8 | 274–338 | 0.1 |
| Zhaoc | 2014 | LScat | 99.9 | 2–6 | 10 | 293–365 | 0.6–3.2 |
| Meng | 2013 | VibW | 99.9 | 2 | 110 | 243–363 | 0.1–30 |
| Cousins | 2012 | SGCap | 99.96 | 2.5–5 | 20 | 247–340 | 0.1–1.9 |
| Hulse | 2009 | OscP | na | 7–14 | 39 | 257–307 | 0.2–2.1 |
| Yamaguchi | 2009 | RB | na | 1.5 | 94 | 263–323 | 0.1–19.6 |
| R1234ze(E) | |||||||
| Zhaoc | 2014 | LScat | 99.9 | 2–6 | 9 | 295–373 | 0.5–3.1 |
| Meng | 2013 | VibW | 99.9 | 2 | 119 | 243–373 | 0.1–30 |
| Cousins | 2012 | SGCap | 99.96 | 2.5–5 | 20 | 247–340 | 0.1–1.5 |
| Grebenkovd | 2009 | Cap | na | 2.5 | 53 | 257–369 | 0.1–6.1 |
Cap, Capillary; FB, Falling Ball; LScat, Light Scattering; MovP, Moving Piston; OscP, Oscillating Piston; RB, Rolling Ball; SGCap, Sealed Gravitational Capillary; VibW, Vibrating Wire.
Also presented in conference paper (Arakawa et al., 2010)
Kinematic viscosities, converted to absolute viscosity with the equation of state of Richter et al (R1234yf) and Thol and Lemmon (R1234ze(E))
Gas phase data of Grebenkov were not used in regression.
Figure 1.
Distribution of experimental viscosity data for R1234yf in the T,p plane
Figure 2.
Distribution of the experimental viscosity data for R1234ze(E) in the T,p plane
Table 2.
| R1234yf | R1234ze(E) | |
|---|---|---|
| Scaling parameters | ε/kB = 275 Κ σ = 0.531 nm |
ε/kB = 340 Κ σ = 0.500 nm |
| Mw (g mol−1) | 114.0416 | 114.0416 |
| Tc(K) | 367.85 | 382.513 |
| ρc(mol L−1) | 4.17 | 4.29 |
| coefficients for Eq(6) | ||
| a0 | −836950 | −963382 |
| a1 | 6336.28 | 9614.09 |
| a2 | −2.3547 | −13.233 |
| a3 | 0.039 556 3 | 0.0360562 |
| a4 | 39509.1 | 122059 |
| a5 | 121.018 | −224.741 |
| Coefficients bi for Eq. (9) from (Vogel et al., 1998) for both R1234yf, R1234ze(E) | ||
| b0 = −19.572 881 | b1 = 219.73999 | b2 = −1015.322 6 |
| b3 = 2471.012 5 | b4 = −3375.171 7 | b5 = 2491.659 7 |
| b6 = −787.260 86 | b7 = 14.085 455 | b8 = −0.346 641 58 |
| Coefficients ci for Eq. (10) | ||
| c0 | −0.194 259 10 | 8.61691913 |
| c1 | −2.079 577 245 | 0 |
| c2 | 0 | 20.83024738 |
| c3 | −43.470 272 88 | 0 |
| c4 | 0 | 0.542 43690 |
| c5 | 0 | −10.496 848 41 |
| c6 | 0 | −1.38137689 |
| c7 | −3.536 827 91 | 1 |
| c8 | 1 | 0 |
2.1 The dilute-gas limit and the initial-density dependence terms
The dilute-gas limit viscosity, η0(T), can be analyzed independently of all other contributions in Eq. (1) and is only a function of temperature. If there are accurate values for the gas thermal conductivity λ0 and the ideal gas heat capacity at constant volume Cv,0, one can estimate the dilute-gas viscosity using the modified Eucken correlation (Reid, et al. 1987)
| (2) |
where R is the molar gas constant, (Mohr et al., 2015) 8.314 4598 J mol−1 K−1.For both R1234yf and R1234ze(E), there are correlations for the thermal conductivity of the dilute gas in the limit of zero density (Perkins and Huber, 2011), and the heat capacity at constant volume can be obtained from the equations of state for R1234yf (Richter et al., 2011) and R1234ze(E) (Thol and Lemmon, 2016). Assuming that the Lennard-Jones 12-6 potential is applicable, one can use Chapman-Enskog theory (Reid et al., 1987) to express the dilute-gas viscosity as
| (3) |
where Ω(2,2) is the Lennard-Jones collision integral that can be calculated by the empirical correlation developed by (Neufeld et al., 1972),
| (4) |
with T*=kBT/ε, ε is the Lennard Jones energy parameter, and σ is the Lennard Jones collision diameter. In engineering units this may be expressed as
| (5) |
where in Eq. (5) η0 is in μPa s, Mw is in g mol−1, T is in K and σ is in nm. We used Eqs. (2)–(5) to obtain the Lennard Jones parameters ε/kB and σ, given in Table 2. For ease of use in calculations, η0 was fit to a rational polynomial form:
| (6) |
where the units for η0 are μPa s, T is in K and the ai coefficients are in Table 2.
To represent the temperature dependence of the term η1(T) in Eq. (1), we use a model presented by Vogel et al.(Vogel et al., 1986) expressed by means of the second viscosity virial coefficient Bη(T) as
| (7) |
The second viscosity virial coefficient can be obtained according to the theory of Rainwater and Friend (Friend and Rainwater, 1984; Rainwater and Friend, 1987) as a function of a reduced second viscosity virial coefficient, , as
| (8) |
where (Vogel et al., 1998)
| (9) |
In the above equations, NA is Avogadro’s constant (Mohr et al., 2015), 6.022 140 857×1023 mol−1. If the units for molar density in Eq. (1) are mol m−3 and the viscosity is in μPa s, then the size scaling parameter σ should be expressed in meters and Bη is in m3 mol−1. The coefficients bi from Vogel et al., 1998 are given in Table 2. Note that T* is a dimensionless temperature defined as T*=T/(ε/kB), where ε/kB and σ are energy and size scaling parameters for the Lennard Jones potential, given in Table 2.
2.2 The residual term
Theory is not available for the residual viscosity term Δη(ρ,T), and its evaluation is based entirely on experimentally obtained data. We used symbolic regression software (Nutonian, 2016) to fit the experimental data to determine the function and coefficients to represent the residual viscosity. This approach was used successfully for the viscosity surface of hydrogen (Muzny et al., 2013), benzene (Avgeri et al., 2014), toluene (Avgeri et al., 2015), hexane (Michailidou et al., 2013), and heptane (Michailidou et al., 2014). We follow those works and incorporate a form suggested by the hard-sphere model employed by (Assael et al., 1992), , where the symbolic regression method was used to determine the functional form for F(ρr,Tr). The dilute-gas limit η0(T) and the initial density dependence terms η1(T)ρ were calculated for each experimental point (employing Eqs. (6) – (9)) and subtracted from the experimental viscosity to obtain the residual term. A simple expression was found:
| (10) |
Coefficients ci are given in Table 2 for both fluids. Tr is the reduced temperature Tr=T/Tc and ρr is the reduced density ρr=ρ/ρc, with Δη in μPa s.
2.3 Comparisons with Experimental Data
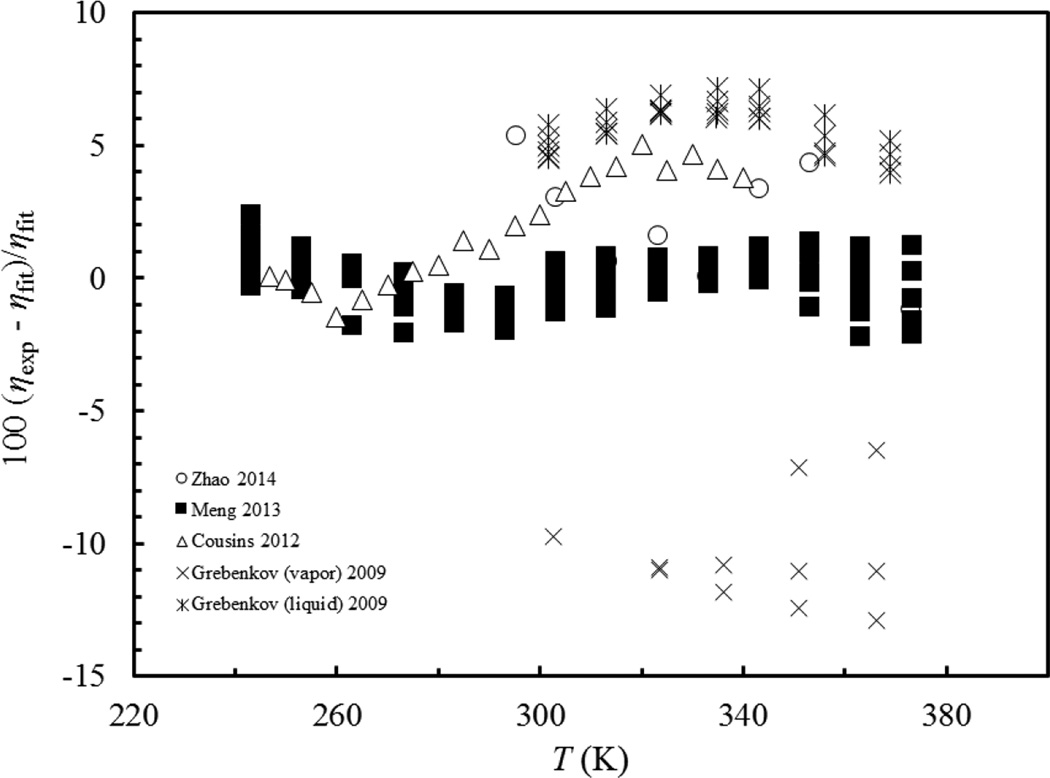
Table 3 summarizes comparisons of all experimental data with the correlations. In the Table we use percent deviation, PCTDEV = 100*(ηexp−ηfit)/ηfit, average absolute percent deviation (AAD), AAD = (∑|PCTDEV|)/n, and BIAS = (∑PCTDEV)/n, where ηexp is the experimental value of the viscosity, ηfit is the value calculated from the correlation, and the summation is over all n points. Figures 3 and 4 show the deviations of all data sets from the correlations for R1234yf and R1234ze(E) respectively. In both figures, the most comprehensive viscosity measurements are the measurements of Meng and coworkers (Meng et al., 2013) performed in an absolute vibrating-wire instrument with an uncertainty of 2 %. This set is backed by a full theory and is the most wide-ranging data set available, and the correlation represents those data sets to within experimental uncertainty. The other data sets generally have more scatter and larger deviations. To obtain an estimate of the uncertainty of the correlation, comparisons with the highest- quality data sets (those with the lowest uncertainty) are used. We estimated the uncertainty of the R1234yf correlation in the liquid phase at pressures up to 30 MPa to be 2 % at a 95 % confidence level over the temperature range 243 K to 363 K, based on the data of Meng et al. (Meng et al., 2013). In the gas phase, the best measurements are those of Dang et al. (Dang et al., 2015b) The correlation represents this set to within 3 %; there is much more scatter in the Yamaguchi data (Yamaguchi et al., 2009). Additional comparisons are available in Supplemental Information. Although the available experimental data only cover a limited temperature range, 243 K- 365 K, and pressures only to 30 MPa, the correlation extrapolates smoothly and in a physically reasonable manner, and can be used over the full range of the equation of state (Richter et al., 2011), from the triple point to 410 K and pressures up to 30 MPa; however uncertainties will be larger especially as the triple point is approached and at high pressures. Similarly we estimate the uncertainty of the liquid phase for R1234ze(E) to be 2 % at pressures up to 30 MPa (based on the data of Meng et al. (Meng et al., 2013)) and 3 % in the gas phase, with the correlation behaving in a physically reasonable manner up to the limits of the equation of state (420 K and 100 MPa), although the uncertainty may be larger in regions where no data were available.
Table 3.
Comparisons of the viscosity correlations with all experimental data.
Figure 3.
Comparisons of all experimental data with the correlation for R1234yf
Figure 4.
Comparisons of all experimental data with the correlation for R1234ze(E)
2.4 Computer-Program Verification
To assist the user in computer-program verification, we provide the following numbers for checking calculations: For R1234yf at 300 K and 0 mol L−1, the viscosity is 11.579 μPa s; at 300 K and 0.044 mol L−1 it is 11.549 μPa s; and at 300 K and 10.522 mol L−1 it is 217.97 μPa s. For R1234ze(E) at 300 K and 0 mol L-1, the viscosity is 11.777 μPa s; at 300 K and 0.044 mol L−1 it is 12.041 μPa s; and at 300 K and 10.522 mol L−1 it is 217.89 μPa s.
3. Conclusions
We present correlations for the viscosity of R1234yf and R1234ze(E) based upon currently available experimental data. These correlations are valid over the entire fluid surface from the triple point to the upper temperature limit of the equation of state, at pressures up to the upper limit of the equation of state. The estimated uncertainty for both correlations is 2 % in the liquid phase for 243 K to 363 K at pressures up to 30 MPa, and 3 % in the gas phase. Both correlations behave in a physically reasonable manner up to the limits of the equation of state although the uncertainty may be larger in regions where no data were available for comparisons.
Supplementary Material
Acknowledgments
We thank Prof. Ryo Akasaka for providing help with Japanese-language data sources.
Footnotes
Contribution of the National Institute of Standards and Technology, not subject to copyright in the U.S.
Contributor Information
Marcia L. Huber, Applied Chemical and Materials Division, National Institute of Standards and Technology Boulder, Colorado 80305-3328, U.S.A.
Marc J. Assael, Laboratory of Thermophysical Properties and Environmental Processes, Chemical Engineering Department, Aristotle University, Thessaloniki 54636, Greece
REFERENCES
- Akasaka R. Viscosity Correlation for 2,3,3,3-Tetrafluoropropene (HFO-1234yf) Based on the Extended Corresponding States Model. J. Therm. Sci. Tech. 2010;5:200–205. [Google Scholar]
- Akasaka R. New Fundamental Equations of State with a Common Functional Form for 2,3,3,3-Tetrafluoropropene (R-1234yf) and Trans-1,2,2,2-Tetrafluoropropene (R-1234ze(E) Int. J. Thermophys. 2011;32:1125–1147. [Google Scholar]
- Arakawa Y, Kim H-S, Kamiaka T, Dong C, Hihara E. Thermophysical Property Measurement of HFO-1234yf+HFC-32 Mixtures. 2010 International Symposium on Next-Generation Air Conditioning and Refrigeration Technology; Tokyo Japan. 2010. [Google Scholar]
- Assael MJ, Dymond JH, Papadaki M, Patterson PM. Correlation and Prediction of Dense Fluid Transport Coefficients - I. n-Alkanes. Int. J. Thermophys. 1992;13:269–281. [Google Scholar]
- Avgeri S, Assael MJ, Huber ML, Perkins RA. Reference Correlation of the Viscosity of Benzene from the Triple Point to 675 K and up to 300 MPa. J. Phys. Chem. Ref. Data. 2014;43:033103. [Google Scholar]
- Avgeri S, Assael MJ, Huber ML, Perkins RA. Reference Correlation of the Viscosity of Toluene from the Triple Point to 675 K and up to 500 MPa. J. Phys. Chem. Ref. Data. 2015;44:033101. [Google Scholar]
- Cousins DS, Laesecke A. Sealed Gravitational Capillary Viscometry of Dimethyl Ether and Two Next-Generation Alternative Refrigerants. J. Res. Nat. Inst. Stand. Tech. 2012;117:231–256. doi: 10.6028/jres.117.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dang YG, Kamiaka T, Dang CB, Hihara E. Liquid viscosity of low-GWP refrigerant mixtures (R32+R1234yf) and (R125+R1234yf) J. Chem. Thermodyn. 2015a;89:183–188. [Google Scholar]
- Dang YG, Kim HS, Dang CB, Hihara E. Measurement of vapor viscosity of R1234yf and its binary mixtures with R32, R125. Int. J. Refrigeration. 2015b;58:131–136. [Google Scholar]
- Dymond JH, Bich E, Vogel E, Wakeham WA, Vesovic V, Assael MJ. In: Transport Properties of Fluids. Millat J, Dymond JH, Nieto de Castro CA, editors. Their Correlation, Prediction and Estimation, Cambridge University Press; 1996. p. 66. [Google Scholar]
- Friend DG, Rainwater JC. Transport properties of a moderately dense gas. Chem. Phys. Lett. 1984;107:590–594. [Google Scholar]
- Grebenkov AJ, Hulse R, Pham H, Singh R. Physical properties and equation of state for trans-1,2,2,2-tetrafluoropropene. Paper 191, 3rd IIR Conference on Thermophysical Properties and Transfer Processes of Refrigerants; Boulder, CO. 2009. [Google Scholar]
- He M, Qi X, Liu X, Lv N. Estimating the viscosity of pure refrigerants and their mixtures by free-volume theory. Int. J. Refrig. 2015;54:55–66. [Google Scholar]
- Hulse R, Singh R, Pham H. Physical properties of HFO-1234yf. paper 178, 3rd IIR Conference on Thermophysical Properties and Transfer Processes of Refrigerants; Boulder, CO. 2009. [Google Scholar]
- Kazakov A, McLinden MO, Frenkel M. Computational Design of New Refrigerants Fluids Based on Environmental, Safety, and Thermodynamic Characteristics. Ind. Eng. Chem. Res. 2012;51:12537–12548. [Google Scholar]
- Kedzierski MA, Brown JS, Koo J. Performance ranking of refrigerants with low global warming potential. Science and Technology for the Built Environment. 2015;21:207–219. [Google Scholar]
- Lemmon EW, Huber ML, McLinden MO. NIST Reference Fluid Thermodynamic and Transport Properties Database, NIST 23, (REFPROP), v9.1. Gaithersburg, MD: National Institute of Standards and Technology; 2013. [Google Scholar]
- Liu W, Meinel D, Wieland C, Spliethoff H. Investigation of hydrofluoroolefins as potential working fluids in organic Rankine cycle for geothermal power generation. Energy. 2014;67:106–116. [Google Scholar]
- Luo D, Mahmoud A, Cogswell F. Evaluation of Low_GWP fluids for power generation with Organic Rankine Cycle. Energy. 2015;85:481–488. [Google Scholar]
- McLinden MO, Brown JS, Brignoli R, Kazakov AF, Domanski PA. Limited options for low-global-warming-potential refrigerants, submitted to Nature Communications. 2016 doi: 10.1038/ncomms14476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLinden MO, Thol M, Lemmon EW. Thermodynamci Properties of trans-1,3,3,3-tetrafluoropropene [R1234ze(E)]: Measurements of Density and Vapor Pressure and a Comprehensive Equation of State. Paper 2189, International Refrigeration and Air Conditioning Conference at Purdue; July 12–15, 2010.2010. [Google Scholar]
- Meng XY, Qiu GS, Wu JT, Abdulagatov IM. Viscosity measurements for 2,3,3,3-tetrafluoroprop-1-ene (R1234yf) and trans-1,3,3,3-tetrafluoropropene (R1234ze(E)) J. Chem. Thermodyn. 2013;63:24–30. [Google Scholar]
- Michailidou EK, Assael MJ, Huber ML, Perkins RA. Reference Correlation of the Viscosity of n-Hexane from the Triple Point to 600 K and up to 100 MPa. J. Phys. Chem. Ref. Data. 2013;42:033104. [Google Scholar]
- Michailidou EK, Assael MJ, Huber ML, Perkins RA. Reference Correlation of the Viscosity of n-Heptane from the Triple Point to 600 K and up to 248 MPa. J. Phys. Chem. Ref. Data. 2014;43:023103. [Google Scholar]
- Mohr PJ, Newell DB, Taylor BN. CODATA Recommended Values of the Fundamental Physical Constants: 2014. 2015 doi: 10.1063/5.0064853. http://arxiv.org/abs/1507.07956v1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mota-Babiloni A, Navarro-Esbri J, Barragan-Cervera A, Moles F, Peris B. Experimental study of an R1234ze(E)/R134a mixture (R450A) as R134a replacement. Int. J. Refrigeration. 2015a;51:52–58. [Google Scholar]
- Mota-Babiloni A, Navarro-Esbri J, Barragan-Cervera A, Moles F, Peris B. Analysis based on EU Regulation No 517/2014 of new HFC/HFO mixtures as alternatives of high GWP refrigerants in refrigeration and HVAC systems. Int. J. Refrigeration. 2015b;52:21–31. [Google Scholar]
- Mota-Babiloni A, Navarro-Esbri J, Moles F, Barragan-Cervera A, Peris B, Verdu G. A review of refrigerant R1234ze(E) recent investigations. Applied Thermal Engineering. 2015c;95:211–222. [Google Scholar]
- Mulero A, Cachadiña I, Parra MI. Recommended Correlations for the Surface Tension of Common Fluids. J. Phys. Chem. Ref. Data. 2012;41:043105. [Google Scholar]
- Muzny CD, Huber ML, Kazakov AF. Correlation for the Viscosity of Normal Hydrogen Obtained from Symbolic Regression. J. Chem. Eng. Data. 2013;58:969–979. [Google Scholar]
- Neufeld PD, Janzen AR, Aziz RA. Empirical equations to calculate 16 of the transport collision integrals Ω(l.s)* for the Lennard-Jones (12-6) Potential. J. Chem. Phys. 1972;57:1100–1102. [Google Scholar]
- Nutonian, Inc. EUREQA Formulize v 1.24.0. Cambridge MA, USA: Nutonian Inc.; 2016. Commercial equipment, instruments, or materials are identified only in order to adequately specify certain procedures. In no case does such identification imply recommendation or endorsement by the National Institute of Standards and Technology, nor does it imply that the products identified are necessarily the best available for the purpose.
- Perkins RA, Huber ML. Measurement and Correlation of the Thermal Conductivity of 2,3,3,3-Tetrafluoroprop-1-ene (R1234yf) and trans-1,3,3,3-Tetrafluoropropene (R1234ze(E)) J. Chem. Eng. Data. 2011;56:4868–4874. [Google Scholar]
- Rainwater JC, Friend DG. Second viscosity and thermal-conductivity virial coefficients of gases: Extension to low reduced temperature. Phys. Rev. A. 1987;36:4062–4066. doi: 10.1103/physreva.36.4062. [DOI] [PubMed] [Google Scholar]
- Reid RC, Prausnitz JM, Poling BE. The Properties of Gases and Liquids. Fourth. New York: McGraw-Hill; 1987. [Google Scholar]
- Richter M, McLinden MO, Lemmon EW. Thermodynamic Properties of 2,3,3,3-Tetrafluoroprop-1-ene (R1234yf): Vapor Pressure and p-ρ-T Measurements and an Equation of State. J. Chem. Eng. Data. 2011;56:3254–3264. [Google Scholar]
- Tanaka K, Higashi Y. Surface tensions of trans-1,3,3,3-Tetrafluoropropene and trans-1,3,3,3-Tetrafluoropropene plus Difluoromethane Mixture. J. Chem. Eng. Japan. 2013;46:371–375. [Google Scholar]
- Thol M, Lemmon EW. Equation of State for the Thermodynamic Properties of trans-1,3,3,3-Tetrafluoropropene [R-1234ze(E)] Int. J. Thermophys. 2016;37:28. [Google Scholar]
- Vogel E, Bich E, Nimz R. The initial density dependence of the viscosity of organic vapours: benzene and methanol. Physica A. 1986;139:188–207. [Google Scholar]
- Vogel E, Küchenmeister C, Bich E, Laesecke A. Reference Correlation of the Viscosity of Propane. J. Phys. Chem. Ref. Data. 1998;27:947–970. [Google Scholar]
- Yamaguchi S, Matsuo S, Sotani T. Viscosity measurement of HFO1234yf by means of rolling-ball method. 30th Japan Symposium on Thermophysical Properties; Yonezawa, Japan. 2009. [Google Scholar]
- Zhao GJ, Bi SS, Froba AP, Wu JT. Liquid Viscosity and Surface Tension of R1234yf and R1234ze Under Saturation Conditions by Surface Light Scattering. J. Chem. Eng. Data. 2014;59:1366–1371. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.