Abstract
Head and neck squamous cell carcinoma (HNSCC) is the sixth leading cause of cancer worldwide. The treatment of choice in case of head and neck cancer is surgery, followed by chemo- or/and radiotherapy. A potentially effective instrument to improve the outcome of numerous diseases, including viral infections, diabetes and cancer, is RNA interference (RNAi). It has been demonstrated that small interfering RNA and microRNA molecules are strongly involved in the regulation of various different pathological processes in cancer development. RNAi has become a valuable research tool allowing a better understanding of the mechanisms regulating cancer pathogenesis. Considering those advantages over other current therapeutics (including specificity and high efficacy), RNAi appears to be a potentially useful tool in cancer treatment. The present review discusses the current knowledge about the possibility of using RNAi in HNSCC therapy.
Keywords: head and neck cancer, RNA interference, gene therapy, siRNA, miRNA
1. Introduction
Head and neck squamous cell carcinoma (HNSCC), the sixth most common cancer worldwide, represents >500,000 cases of all the cancer cases diagnosed every year (1,2). Previous epidemiological studies have demonstrated that exposure to carcinogens such as tobacco and alcohol, as well as infection by oncogenic human papillomavirus 16 and 18, results in increased risk of HNSCC development (2–4). Currently, the treatment of choice for head and neck cancer is surgery, followed by postoperative chemo- and/or radiotherapy (1,5,6). Despite advances in conventional methods, the 5-year mortality rate of patients with HNSCC has not improved (2). With progress in technologies and molecular genetics, there is a growing potential of gene therapy as a powerful tool for HNSCC treatment.
RNA interference (RNAi) is a natural physiological process, which is supposed to protect the genome from viruses and transposons (7). RNAi technology is based on the inhibition of target transcript expression or degradation by exogenous or endogenous homologous double-stranded (ds)RNA, which has the ability to specifically and more effectively silencing genes than single-stranded RNA individually does (8,9). The first phase of the RNAi mechanism is the hydrolysis of exogenous dsRNAs to small interfering (si)RNAs by Dicer endoribonuclease (10,11). Each strand of siRNA has 5′-phosphate and 3′-hydroxyl termini, in addition to 2–3-nucleotides 3′ overhangs (11,12). In the next step, Dicer enzyme, in cooperation with cofactors, mediates the binding of siRNAs to RNA-induced silencing complex (RISC) (8–13). The endogenous substrate of RNAi are small non-coding RNA molecules of micro (mi)RNA (14). The long miRNA precursor, termed primary (pri)-miRNA, is composed of several hundred nucleotides and is cleaved by the Microprocessor complex (15,16). The core of this complex are Drosha (a class 2 ribonuclease III enzyme) and DiGeorge syndrome critical region 8 protein (16). As a result of the hydrolysis, precursor (pre)-miRNAs are formed, which are hairpin-loop structures composed of ~70 nucleotides that are subsequently transported from the nucleus to the cytosol, where they undergo a maturation process mediated by Dicer nuclease (16–22). Mature miRNAs are targeted to the RISC effector complex (10,18–24).
RNAi is a potentially effective instrument in gene therapy. However, it is required to establish efficient systems of its in vitro and in vivo applications. These systems should work specifically in a defined cell type or tissue, and should also eliminate the risk of potential immunological response (25). Currently, there are two main systems of introduction of RNA molecules into organisms: Viral (retroviruses, including lentiviruses), adenoviruses and adeno-associated viruses) (26–33) and non-viral (34,35). The aim of the present study is to review the usefulness of the RNAi mechanism in head and neck oncology.
2. Nanoparticle delivery of HIF1α siRNA combined with PDT as a potential treatment strategy for head and neck cancer
Hypoxia inducible factor 1 (HIF1) is a master transcriptional regulator of the cellular and systemic hypoxia response (36). HIF1 is a heterodimer, and consists of two subunits (HIF1β and HIF1α) (37). It belongs to the family of basic helix-loop-helix transcription factors (37). Under normoxic conditions, HIF1α is degraded rapidly with the participation of a proline hydroxylase, which performs an oxygen-hydroxylation of proline residues 402 and 564 (37). Hydroxylated HIF1α is subsequently recognized by Von Hippel-Lindau protein (pVHL), a component of an E3 ubiquitin-protein ligase, and degraded in the proteasome (37). Under low concentration of oxygen, pVHL does not bind to HIF1α, and it is translocated to the nucleus instead, where it forms a heterodimer with the HIF1β subunit (37,38). This subunit (also known as aryl hydrocarbon receptor nuclear translocator) specifically binds to hypoxia-responsive elements of oxygen-regulated genes promoters (37,38). The formation of HIF1 heterodimers results in the transcriptional activation of several genes, including vascular endothelial growth factor (VEGF), glucose transporter 1 and carbonic anhydrase IX, which are involved in self-renewal, survival and induction of angiogenesis and metastases, which in turn contributes to increased cancer progression and therapy resistance (39). Therefore, HIF1 plays a pivotal role in tumorigenesis by determining the ability of self-renewal and multipotency of cancer stem cells in a hypoxic environment (36–40).
Chen et al (36) investigated the potential of silencing HIF1α combined with Photosan-based photodynamic therapy (PDT) in human oral (O)SCC. Anisamide-targeted lipid-calcium-phosphate (LCP-AA) nanoparticles were used to deliver HIF1α siRNA to the cytosol of SCC4 and SAS cell lines (derived from a squamous carcinoma of human tongue with expression of sigma receptors) (36). Cells were also subjected to PDT. To investigate the efficiency of LCP delivery, double-stranded HIF1α oligonucleotides (DNA) labeled with Texas Red dye were used. The study revealed that LCP-AA was able to successfully and efficiently deliver siRNA in a sigma receptor-mediated process (36). To confirm these results, SCC4 tumor bearing nude mice were intravenously injected with AA-targeted Texas Red labeled LCP-AA. After 4 h, the fluorescence intensity in the tumor and organs was measured. The tumor region exhibited the strongest signal, confirming the efficient delivery of LCP-AA to SCC4 cells in vivo (36). The effect of HIF1α knockdown on the viability of SCC4 cells, LCP toxicity and in vivo therapeutic outcomes of the combined treatment were also evaluated. HIF1α depletion by siRNA inhibited the proliferation of OSCC cells and induced their apoptosis (36). Immune response or toxicity of LCP were not observed (36). These studies demonstrate that systemic administration of HIF1α siRNA by targeted LCP appears to enable the stable and effective inhibition of OSCC proliferation (36). These results were also confirmed by Ahn et al and Liang et al, who suppressed tumor growth with HIF1α depletion via regulation of VEGF (5,6).
3. Suppression of ABCG2 inhibits the process of LSCC tumor growth
ATP-binding cassette (ABC), subfamily G, member 2 (ABCG2, also known as breast cancer resistance protein) is a 655-amino acid protein of 72 kDa, which is a member of the ABC transporter family (41–46). It was first cloned from doxorubicin-resistant human MCF-7 breast cancer cells (41). Overexpression of ABCG2 is observed in multiple tumor types, including leukemias and certain SCC (41). Increased expression of ABCG2 leads to drug resistance by promoting the proliferation of tumor cells and suppressing apoptosis (41–46).
Xie et al (41) investigated the role of ABCG2 in laryngeal (L)SCC tumor growth and its influence on the accumulation of mitoxantrone (MX) in cancer cells. ABCG2 siRNA was introduced into two LSCC cell lines: Hep2 and Hep2T (Taxol-resistant). To evaluate the efficiency and effect of silencing the ABCG2 gene in LSCC cells, a number of analyses, including flow cytometry, apoptosis assay, reverse transcription-quantitative polymerase chain reaction (RT-qPCR) and western blotting, were performed. The results revealed that the suppression of ABCG2 inhibits LSCC tumor growth by regulating cell proliferation and apoptosis. It was also confirmed that ABCG2 knockdown increases MX accumulation in both Hep2 and Hep2T cell lines, thus enhancing its anti-tumor effect (41). These findings suggest that ABCG2 is a potential therapeutic target for LSCC.
4. Suppression of ITG3A increases radiosensitivity and induces apoptosis in HNSCC cells
Integrins are heterodimeric surface receptors that are responsible for cell adhesion to extracellular matrix (ECM) proteins and are important in cell-cell adhesions, transmembrane connections to the cytoskeleton and intracellular signaling pathways (47–49). There are ≥24 distinct integrin heterodimers formed by the combination of 18 α-subunits and 8 β-subunits, and each of them has a different specificity to ECM proteins (38). A wide variety of integrins contribute to tumor progression, including tumors derived from epithelial cells (50,51). In addition, integrins may contribute to migration, proliferation and survival of tumor cells (51). Furthermore, depending on the environmental conditions, integrins have the ability to either enhance cell survival or initiate apoptosis (51). Considering their features, integrins are regarded as attractive targets for cancer therapy (47–51).
Steglich et al (48) investigated the influence of α3 integrin (ITG3A) depletion on the progression and radiosensitivity of HNSCC cells. The expression of ITG3A in these cells was evaluated by western blot analysis, which revealed that ITG3A is overexpressed in HNSCC. Subsequently, the HNSCC cell lines UTSCC5, UTSCC14, UTSCC15, Cal33 and HSC4 were transfected with siRNA against ITG3A. It was demonstrated that ITG3A knockdown results in reduced clonogenic cell survival, induced apoptosis and enhanced radiosensitivity (48). Therefore, ITG3A may be considered as a potential target in HNSCC treatment.
5. Genetic silencing and pharmacological inhibition of Bmi1 as an instrument of TSCC gene therapy
Polycomb group proteins (PcG) are critical transcriptional repressors that epigenetically modify chromatin and contribute to cell fate, stem cell self-renewal and cancer development (52,53). B lymphoma Mo-MLV insertion region 1 homolog (Bmi1) is the core member of the polycomb repressive complex 1 (PRC1), one of two PRCs in PcG (53). Bmi1 is an epigenetic silencer of multiple target genes, including p14Arf, p19Arf and p16Ink4a (52,53).
Bmi1-mediated chromatin modifications are involved in crucial cellular processes, including stem cell maintenance, proliferation, senescence, apoptosis and epithelial-mesenchymal transition (EMT) (52,53). Overexpression of Bmi1 is associated with multiple human malignancies, including myeloid leukemia as well as lung, breast and head and neck cancer (53). In addition, overexpression of Bmi1 is associated with the therapeutic resistance, aggressive clinicopathological behavior and poor prognosis of the above cancers (52,53). Furthermore, it was demonstrated that Bmi1 overexpression promotes malignant transformation, cancer cell proliferation, EMT and metastatic spreading, while its suppression inhibits cancer progression, thus inducing senescence and cell apoptosis (52,53).
Li et al (53) investigated the potential of Bmi1 as a target in the genetic and pharmacological treatment of tongue (T)SCC. siRNAs against different regions of Bmi1 were delivered to TSCC cell lines (HN4, HN6, HN12, Tca8113, Cal27, SCC9 and SCC25) using Lipofectamine 2000. To evaluate the efficacy of Bmi1 suppression, cells were cultured for 24 h and subsequently analyzed by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, flow cytometry, RT-qPCR, cell migration and wound healing assay. Simultaneously, an experiment on a mouse TSCC xenograft model was conducted to determine the anticancer effects of histone deacetylase inhibitors (HDACis). It was demonstrated that Bmi1 knockdown inhibits cell migration and proliferation and induces cell senescence and apoptosis in TSCC. Furthermore, it was revealed that HDACis such as sodium butyrate and trichostatin A inhibit Bmi1 expression in tongue cancer cells and exert an anticancer effect (53). Therefore, the pharmacological and genetic disruption of Bmi1 may represent a novel therapeutic strategy against tongue cancers.
6. AURKA inhibition and paclitaxel as targeted combination therapy for HNSCC
Aurora kinase A (AURKA) is a member of the conserved serine/threonine protein kinase family, and a cell cycle-regulated kinase involved in spindle formation and chromosome segregation (54–60). It was observed that AURKA overexpression induces oncogenic transformation accompanied by centrosome amplification and aneuploidy in rodent cells (55,56). Furthermore, the AURKA gene is amplified and overexpressed in numerous human cancers, including breast, bladder, colorectal, ovarian gastric and pancreatic cancer, causing resistance to paclitaxel (Taxol) (54). A correlation between AURKA mRNA overexpression, tumor progression and shortened survival in patients with HNSCC was determined (54–60).
Mazumdar et al (54) investigated the potential of AURKA as a therapeutic target in HNSCC. AURKA-targeted siRNA was introduced into HNSCC cell lines (Tu138, UMSCC1, Tu167, OSC19, Tu177 and JMAR). Cells were cultured for 72 h and then harvested. AURKA knockdown was confirmed by western blot analysis. MTT assay and flow cytometry analysis were also performed to evaluate cell proliferation and cell cycle disruption, respectively. It was demonstrated that AURKA knockdown inhibited HNSCC cells proliferation, significantly reducing the proportion of G1-phase cells. Furthermore, AURKA depletion significantly increased the cytotoxicity of paclitaxel (54). The suppressive effect of AURKA depletion on tumor growth was also confirmed by Tanaka et al (60), who demonstrated that AURKA plays a pivotal role in the growth of human OSCC cells, and that AURKA silencing appears to be a potentially useful therapeutic approach for patients with OSCC.
7. RNAi in clinical trials
The first clinical trial involving siRNA was conducted in 2004 by Acuity Pharmaceuticals, Inc. (OPKO Health, Inc., Miami, FL, USA) for the treatment of age-related macular degeneration (AMD) (61). Naked siRNA targeted to VEGF and VEGF receptor genes displayed therapeutic potential in the inhibition of the excessive vascularization of the eye that leads to AMD. The completed trials reported a good tolerability of all siRNA doses and improved eyesight in patients (61). The detailed data about current RNAi trials are recorded in the ClinicalTrials.gov website (https://clinicaltrials.gov/), which is maintained by the National Library of Medicine of the National Institutes of Health (Bethesda, MD, USA), and provides access to information on clinical studies on a wide range of diseases and conditions (62). According to this database, the Comprehensive Cancer Center of Wake Forest University (Elkin, NC, USA) is currently conducting a phase I trial on the side effects and best dose of siRNA-transfected peripheral blood mononuclear cells (termed APN401) in treating patients with melanoma, renal cancer, pancreatic cancer or other solid tumors that cannot be removed by surgery (63). It has been hypothesized that these modified immune cells will have a high anticancer activity (63). The University of Texas MD Anderson Cancer Center (Houston, TX, USA) is currently conducting a phase I study on the safety and the highest tolerable dose of siRNA-ephrin type-A receptor 2 (EphA2)-1,2-dioleoyl-sn-glycero-3-phosphocholine, which is designed to shut down the activity of the genetic biomarker EphA2 to treat patients with advanced solid tumors (64). Phase I trials are also being conducted by Calando Pharmaceuticals, Inc. (Pasadena, CA, USA) to determine the safety, toxicity and maximum tolerated dose of CALAA-01, which is administered intravenously to patients with relapsed or refractory cancer (65). The active ingredient in CALAA-01 is a siRNA that inhibits tumor growth by reducing the expression of the M2 subunit of ribonucleotide reductase R2 (65). A phase I study was conducted by Silenseed Ltd. (Modi'in, Israel) to investigate the safety of siG12D Local Drug EluteR (LODER) in patients diagnosed with pancreas adenocarcinoma (66). siG12D LODER is a miniature biodegradable polymeric matrix that contains siRNA designed to shut down the activity of the Kirsten rat sarcoma viral oncogene homolog (KRAS) gene. It is considered that silencing KRAS will lead to the apoptosis of cancer cells, and thereby slow down or even halt tumor growth (66). Silenseed Ltd. is actually conducting a phase II study on siG12D LODER to assess the efficacy and local distribution of this drug in patients with non-operable pancreas adenocarcinoma (67).
8. Summary
The discovery of RNAi has had a significant influence on research and development. There is large evidence that siRNA and miRNA molecules are strongly involved in the regulation of different pathological processes, including cancer development. Therefore, RNAi technology has become a valuable research tool for better understanding the mechanisms of cancer pathogenesis. Considering its ability to specific eliminate defective genes products, siRNA is a theoretically perfect therapeutic instrument for certain diseases, including cancer, viral infections and diabetes (Fig. 1) (35). Its application in gene therapy has a variety of advantages such as high efficiency and specificity, which allow to reduce side effects, including immunological response. Therefore, RNAi could be successfully used in the treatment of numerous diseases whose molecular mechanisms of pathogenesis are known.
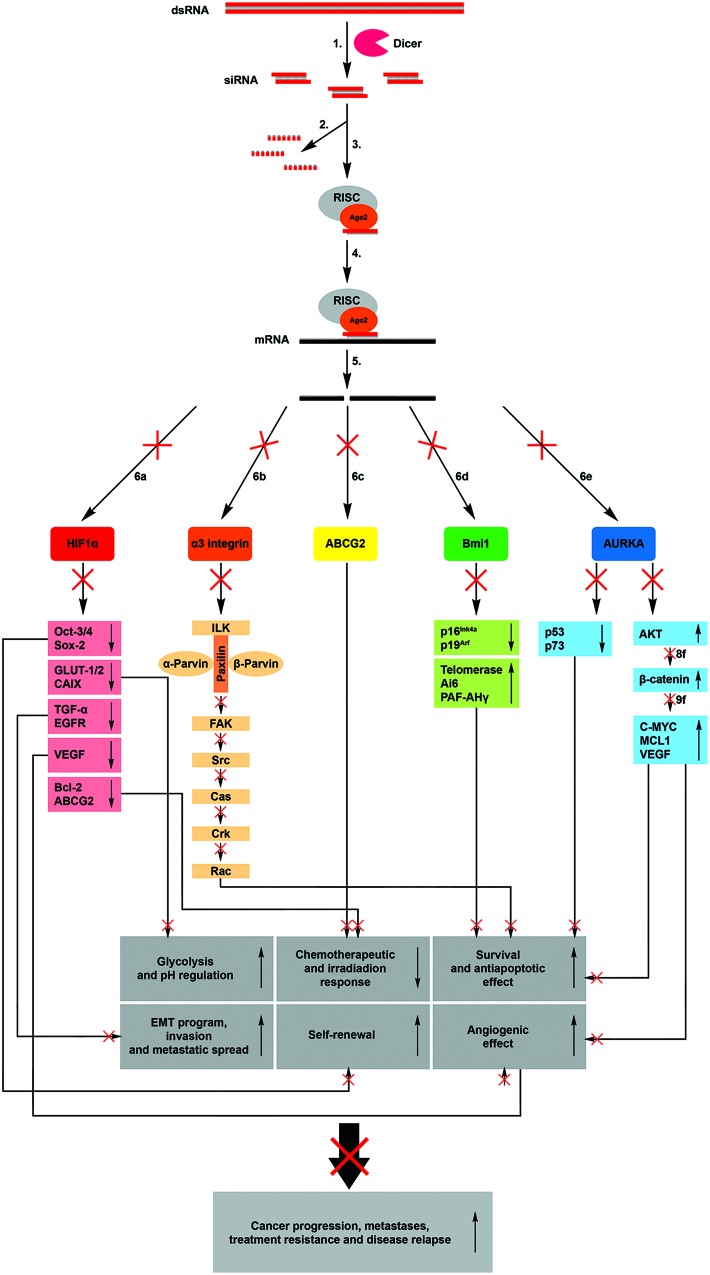
Figure 1.
Mechanisms of RNA interference. Introducing exogenous dsRNA into a cell activates the ribonuclease protein Dicer, which binds and cleaves dsRNA into short (21–24 bp) fragments termed siRNAs (1). The sense strand of siRNA is degraded (2), while the anti-sense strand binds to active RISC (3). The RISC-siRNA complex identifies the homologous target messenger RNA (4) and induces its cleavage (5), thus preventing the translation of the target proteins hypoxia inducible factor 1α (6a), α3 integrin (6b), ATP-binding cassette, subfamily G, member 2 (6c), B lymphoma Mo-MLV insertion region 1 homolog (6d) and aurora kinase A (6e). This process results in the transcriptional inhibition of numerous genes involved in anaerobic glycolysis, pH regulation, survival, antiapoptotic and angiogenic effects, metastases, self-renewal and treatment resistance of cancer cells. ds, double-stranded; si, small interfering; m, messenger; RISC, RNA-induced silencing complex; HIF, hypoxia inducible factor; Ago, argonaute; ABCG2, ATP-binding cassette, subfamily G, member 2; Bmi1, B lymphoma Mo-MLV insertion region 1 homolog; AURKA, aurora kinase A; Oct, organic cation transporter; GLUT, glucose transporter; TGF, transforming growth factor; VEGF, vascular endothelial growth factor; Bcl, B-cell lymphoma; ILK, integrin-linked kinase; FAK, focal adhesion kinase; Cas, CRISPR-associated; Rac, Ras-related C3 botulinum toxin substrate; PAF-AH, platelet-activating factor-acetylhydrolase; MCL1, myeloid cell keukemia; EMT, epithelial-mesenchymal transition.
Acknowledgements
The present work was supported by the National Science Centre (grant no., 2015/17/N/NZ5/00686) and the Greater Poland Cancer Centre research grant (grant no., 9/11/2015/PRB/WCO/21).
References
- 1.Suh Y, Amelio I, Urbano T Guerrero, Tavassoli M. Clinical update on cancer: Molecular oncology of head and neck cancer. Cell Death Dis. 2014;5:e1018. doi: 10.1038/cddis.2013.548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Thomas SM, Grandis JR. The current state of head and neck cancer gene therapy. Hum Gene Ther. 2009;20:1565–1575. doi: 10.1089/hum.2009.163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Maiti GP, Mondal P, Mukherjee N, Ghosh A, Ghosh S, Dey S, Chakrabarty J, Roy A, Biswas J, Roychoudhury S, Panda CK. Overexpression of EGFR in head and neck squamous cell carcinoma is associated with inactivation of SH3GL2 and CDC25A genes. PLoS One. 2013;8:e63440. doi: 10.1371/journal.pone.0063440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Martinez-Useros J, Garcia-Foncillas J. The challenge of blocking a wider family members of EGFR against head and neck squamous cell carcinomas. Oral Oncol. 2015;51:423–430. doi: 10.1016/j.oraloncology.2015.02.092. [DOI] [PubMed] [Google Scholar]
- 5.Ahn SH, Choi JY, Kim DW, Lee DY, Jeon EH, Jeong WJ, Paik JH. Targeting HIF1α peri-operatively increased post-surgery survival in a tongue cancer animal model. Ann Surg Oncol. 2015;22:3041–3048. doi: 10.1245/s10434-014-4323-0. [DOI] [PubMed] [Google Scholar]
- 6.Liang J, Zhang Z, Liang L, Shen Y, Ouyang K. HIF-1α regulated tongue squamous cell carcinoma cell growth via regulating VEGF expression in a xenograft model. Ann Transl Med. 2014;2:92. doi: 10.3978/j.issn.2305-5839.2014.08.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hamilton AJ, Baulcombe D. A species of small antisense RNA in posttranscriptional gene silencing in plants. Science. 1999;286:950–952. doi: 10.1126/science.286.5441.950. [DOI] [PubMed] [Google Scholar]
- 8.Napoli C, Lemieux C, Jorgensen R. Introduction of a chimeric chalcone synthase gene into petunia results in reversible co-suppression of homologous genes in trans. Plant Cell. 1990;2:279–289. doi: 10.1105/tpc.2.4.279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature. 1998;391:806–811. doi: 10.1038/35888. [DOI] [PubMed] [Google Scholar]
- 10.Collins RE, Cheng X. Structural and biochemical advances in mammalian RNAi. J Cell Biochem. 2006;99:1251–1266. doi: 10.1002/jcb.21069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Agrawal N, Dasaradhi PVN, Mohmmed A, Malhotra P, Bhatnagar RK, Mukherjee SK. RNA interference: Biology, mechanism, and applications. Microbiol Mol Biol Rev. 2003;67:657–685. doi: 10.1128/MMBR.67.4.657-685.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lee Y, Ahn C, Han J, Choi H, Kim J, Yim J, Lee J, Provost P, Rådmark O, Kim S, Kim VN. The nuclear RNase III drosha initiates microRNA processing. Nature. 2003;425:415–419. doi: 10.1038/nature01957. [DOI] [PubMed] [Google Scholar]
- 13.Wilson RC, Doudna JA. Molecular mechanisms of RNA interference. Annu Rev Biophys. 2003;42:217–239. doi: 10.1146/annurev-biophys-083012-130404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Filipowicz W, Jaskiewicz L, Kolb FA, Pillai RS. Post-transcriptional gene silencing by siRNAs and miRNAs. Curr Opin Struct Biol. 2005;15:331–341. doi: 10.1016/j.sbi.2005.05.006. [DOI] [PubMed] [Google Scholar]
- 15.Liu Q, Paroo Z. Biochemical principles of small RNA pathways. Annu Rev Biochem. 2010;79:295–319. doi: 10.1146/annurev.biochem.052208.151733. [DOI] [PubMed] [Google Scholar]
- 16.Ouellet DL, Perron MP, Gobeil LA, Plante P, Provost P. MicroRNAs in gene regulation: When the smallest governs it all. J Biomed Biotechnol. 2006;2006:69616. doi: 10.1155/JBB/2006/69616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yeom KH, Lee Y, Han J, Suh MR, Kim VN. Characterization of DGCR8/Pasha, the essential cofactor for Drosha in primary miRNA processing. Nucleic Acids Res. 2006;34:4622–4629. doi: 10.1093/nar/gkl458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Khvorova A, Reynolds A, Jayasena SD. Functional siRNAs and miRNAs exhibit strand bias. Cell. 2003;115:209–216. doi: 10.1016/S0092-8674(03)00801-8. [DOI] [PubMed] [Google Scholar]
- 19.Schwarz DS, Hutvágner G, Du T, Xu Z, Aronin N, Zamore PD. Asymmetry in the assembly of the RNAi enzyme complex. Cell. 2003;115:199–208. doi: 10.1016/S0092-8674(03)00759-1. [DOI] [PubMed] [Google Scholar]
- 20.Meister G, Landthaler M, Patkaniowska A, Dorsett Y, Teng G, Tuschl T. Human Argonaute2 mediates RNA cleavage targeted by miRNAs and siRNAs. Mol Cell. 2004;15:185–197. doi: 10.1016/j.molcel.2004.07.007. [DOI] [PubMed] [Google Scholar]
- 21.Elbashir SM, Martinez J, Patkaniowska A, Lendeckel W, Tuschl T. Functional anatomy of siRNAs for mediating efficient RNAi in Drosophila melanogaster embryo lysate. EMBO J. 2001;20:6877–6888. doi: 10.1093/emboj/20.23.6877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Eulalio A, Huntzinger E, Izaurralde E. Getting to the root of miRNA-mediated gene silencing. Cell. 2008;132:9–14. doi: 10.1016/j.cell.2007.12.024. [DOI] [PubMed] [Google Scholar]
- 23.Gregory RI, Chendrimada TP, Cooch N, Shiekhattar R. Human RISC couples microRNA biogenesis and posttranscriptional gene silencing. Cell. 2005;123:631–640. doi: 10.1016/j.cell.2005.10.022. [DOI] [PubMed] [Google Scholar]
- 24.Meister G. miRNAs get an early start on translational silencing. Cell. 2007;131:25–28. doi: 10.1016/j.cell.2007.09.021. [DOI] [PubMed] [Google Scholar]
- 25.Pecot CV, Calin GA, Coleman RL, Lopez-Berestein G, Sood AK. RNA interference in the clinic: Challenges and future directions. Nat Rev Cancer. 2011;11:59–67. doi: 10.1038/nrc2966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bouard D, Alazard-Dany D, Cosset FL. Viral vectors: From virology to transgene expression. Br J Pharmacol. 2009;157:153–165. doi: 10.1038/bjp.2008.349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kantor B, Bailey RM, Wimberly K, Kalburgi SN, Gray SJ. Methods for gene transfer to the central nervous system. Adv Genet. 2014;87:125–197. doi: 10.1016/B978-0-12-800149-3.00003-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Galanis E, Vile R, Russell SJ. Delivery systems intended for in vivo gene therapy of cancer: Targeting and replication competent viral vectors. Crit Rev Oncol Hemato. 2001;38:177–192. doi: 10.1016/S1040-8428(01)00103-2. [DOI] [PubMed] [Google Scholar]
- 29.Morris KV, Rossi JJ. Lentiviral-mediated delivery of siRNAs for antiviral therapy. Gene Ther. 2006;13:553–558. doi: 10.1038/sj.gt.3302688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wold WS, Toth K. Adenovirus vectors for gene therapy, vaccination and cancer gene therapy. Curr Gene Ther. 2013;13:421–433. doi: 10.2174/1566523213666131125095046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hermens WT, Verhaagen J. Viral vectors, tools for gene transfer in the nervous system. Prog Neurobiol. 1998;55:399–432. doi: 10.1016/S0301-0082(98)00007-0. [DOI] [PubMed] [Google Scholar]
- 32.Tomar RS, Matta H, Chaudhary PM. Use of adeno-associated viral vector for delivery of small interfering RNA. Oncogene. 2003;22:5712–5715. doi: 10.1038/sj.onc.1206733. [DOI] [PubMed] [Google Scholar]
- 33.Lentz TB, Gray SJ, Samulski RJ. Viral vectors for gene delivery to the central nervous system. Neurobiol Dis. 2012;48:179–188. doi: 10.1016/j.nbd.2011.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ramamoorth M, Narvekar A. Non viral vectors in gene therapy-an overview. J Clin Diagn Res. 2015;9:GE01–GE06. doi: 10.7860/JCDR/2015/10443.5394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Uprichard SL. The therapeutic potential of RNA interference. FEBS Lett. 2005;579:5996–6007. doi: 10.1016/j.febslet.2005.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chen WH, Lecaros RL, Tseng YC, Huang L, Hsu YC. Nanoparticle delivery of HIF1α siRNA combined with photodynamic therapy as a potential treatment strategy for head-and-neck cancer. Cancer Lett. 2015;359:65–74. doi: 10.1016/j.canlet.2014.12.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sasabe E, Tatemoto Y, Li D, Yamamoto T, Osaki T. Mechanism of HIF-1alpha-dependent suppression of hypoxia-induced apoptosis in squamous cell carcinoma cells. Cancer Sci. 2005;96:394–402. doi: 10.1111/j.1349-7006.2005.00065.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sasabe E, Zhou X, Li D, Oku N, Yamamoto T, Osaki T. The involvement of hypoxia-inducible factor-1alpha in the susceptibility to gamma-rays and chemotherapeutic drugs of oral squamous cell carcinoma cells. Int J Cancer. 2007;120:268–277. doi: 10.1002/ijc.22294. [DOI] [PubMed] [Google Scholar]
- 39.Mimeault M, Batra SK. Hypoxia-inducing factors as master regulators of stemness properties and altered metabolism of cancer- and metastasis-initiating cells. J Cell Mol Med. 2013;17:30–54. doi: 10.1111/jcmm.12004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jing S, Wang J, Liu Q, Cheng Y, Yang C, Wang Y, Cao F, Wen B, Jiao W, Guo Y. Relationship between hypoxia inducible factor-1α and esophageal squamous cell carcinoma: A meta analysis. Zhonghua Bing Li Xue Za Zhi. 2014;43:593–599. (In Chinese) [PubMed] [Google Scholar]
- 41.Xie J, Jin B, Li DW, Shen B, Cong N, Zhang TZ, Dong P. ABCG2 regulated by MAPK pathways is associated with cancer progression in laryngeal squamous cell carcinoma. Am J Cancer Res. 2014;4:698–709. [PMC free article] [PubMed] [Google Scholar]
- 42.Doyle LA, Yang W, Abruzzo LV, Krogmann T, Gao Y, Rishi AK, Ross DD. A multidrug resistance transporter from human MCF-7 breast cancer cells. Proc Natl Acad Sci USA. 1998;95:15665–15670. doi: 10.1073/pnas.95.26.15665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chen Z, Liu F, Ren Q, Zhao Q, Ren H, Lu S, Zhang L, Han Z. Suppression of ABCG2 inhibits cancer cell proliferation. Int J Cancer. 2010;126:841–851. doi: 10.1002/ijc.24796. [DOI] [PubMed] [Google Scholar]
- 44.Noguchi K, Katayama K, Sugimoto Y. Human ABC transporter ABCG2/BCRP expression in chemoresistance: Basic and clinical perspectives for molecular cancer therapeutics. Pharmgenomics Pers Med. 2014;5:53–64. doi: 10.2147/PGPM.S38295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Natarajan K, Xie Y, Baer MR, Ross DD. Role of breast cancer resistance protein (BCRP/ABCG2) in cancer drug resistance. Biochem Pharmacol. 2012;15:1084–1103. doi: 10.1016/j.bcp.2012.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Nakanishi T, Ross DD. Breast cancer resistance protein (BCRP/ABCG2): Its role in multidrug resistance and regulation of its gene expression. Chin J Cancer. 2012;31:73–99. doi: 10.5732/cjc.011.10320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hynes RO. Integrins: Bidirectional, allosteric signaling machines. Cell. 2002;110:673–687. doi: 10.1016/S0092-8674(02)00971-6. [DOI] [PubMed] [Google Scholar]
- 48.Steglich A, Vehlow A, Eke I, Cordes N. α integrin targeting for radiosensitization of three-dimensionally grown human head and neck squamous cell carcinoma cells. Cancer Lett. 2015;357:542–548. doi: 10.1016/j.canlet.2014.12.009. [DOI] [PubMed] [Google Scholar]
- 49.Hehlgans S, Haase M, Cordes N. Signalling via integrins: Implications for cell survival and anticancer strategies. Biochim Biophys Acta. 2007;1775:163–180. doi: 10.1016/j.bbcan.2006.09.001. [DOI] [PubMed] [Google Scholar]
- 50.Onodera Y, Nam JM, Sabe H. Intracellular trafficking of integrins in cancer cells. Pharmacol Ther. 2013;140:1–9. doi: 10.1016/j.pharmthera.2013.05.007. [DOI] [PubMed] [Google Scholar]
- 51.Desgrosellier JS, Cheresh DA. Integrins in cancer: Biological implications and therapeutic opportunities. Nat Rev Cancer. 2010;10:9–22. doi: 10.1038/nrc2748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lukacs RU, Memarzadeh S, Wu H, Witte ON. Bmi-1 is a crucial regulator of prostate stem cell self-renewal and malignant transformation. Cell Stem Cell. 2010;7:682–693. doi: 10.1016/j.stem.2010.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Li Z, Wang Y, Yuan C, Zhu Y, Qiu J, Zhang W, Qi B, Wu H, Ye J, Jiang H, et al. Oncogenic roles of Bmi1 and its therapeutic inhibition by histone deacetylase inhibitor in tongue cancer. Lab Invest. 2014;94:1431–1445. doi: 10.1038/labinvest.2014.123. [DOI] [PubMed] [Google Scholar]
- 54.Mazumdar A, Henderson YC, El-Naggar AK, Sen S, Clayman GL. Aurora kinase A inhibition and paclitaxel as targeted combination therapy for head and neck squamous cell carcinoma. Head Neck. 2009;31:625–634. doi: 10.1002/hed.21007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bischoff JR, Anderson L, Zhu Y, Mossie K, Ng L, Souza B, Schryver B, Flanagan P, Clairvoyant F, Ginther C, et al. A homologue of Drosophila aurora kinase is oncogenic and amplified in human colorectal cancers. EMBO J. 1998;17:3052–3065. doi: 10.1093/emboj/17.11.3052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Goepfert TM, Adigun YE, Zhong L, Gay J, Medina D, Brinkley WR. Centrosome amplification and overexpression of aurora A are early events in rat mammary carcinogenesis. Cancer Res. 2002;62:4115–4122. [PubMed] [Google Scholar]
- 57.Kovarikova V, Burkus J, Rehak P, Brzakova A, Solc P, Baran V. Aurora kinase A is essential for correct chromosome segregation in mouse zygote. Zygote. 2016;15:326–337. doi: 10.1017/S0967199415000222. [DOI] [PubMed] [Google Scholar]
- 58.Katsha A, Belkhiri A, Goff L, El-Rifai W. Aurora kinase A in gastrointestinal cancers: Time to target. Mol Cancer. 2015;20:106. doi: 10.1186/s12943-015-0375-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sun JM, Yang LN, Xu H, Chang B, Wang HY, Yang G. Inhibition of Aurora A promotes chemosensitivity via inducing cell cycle arrest and apoptosis in cervical cancer cells. Am J Cancer Res. 2015;5:1133–1145. [PMC free article] [PubMed] [Google Scholar]
- 60.Tanaka H, Nakashiro K, Iwamoto K, Tokuzen N, Fujita Y, Shirakawa R, Oka R, Goda H, Hamakawa H. Targeting Aurora kinase A suppresses the growth of human oral squamous cell carcinoma cells in vitro and in vivo. Oral Oncol. 2013;49:551–559. doi: 10.1016/j.oraloncology.2013.02.002. [DOI] [PubMed] [Google Scholar]
- 61.Whitehead KA, Langer R, Anderson DG. Knocking down barriers: Advances in siRNA delivery. Nat Rev Drug Discov. 2009;8:129–138. doi: 10.1038/nrd2742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.U.S. National Institutes of Health, corp. https://clinicaltrials.gov/ct2/about-site/background. ClinicalTrials.gov: ClinicalTrials.gov Background. 2015 Jun 29; Available from. Accessed.
- 63.U.S. National Institutes of Health, corp. http://clinicaltrials.gov/show/NCT02166255. ClinicalTrials.gov: APN401 in Treating Patients With Melanoma, Kidney Cancer, Pancreatic Cancer, or Other Solid Tumors That Are Metastatic or Cannot Be Removed By Surgery. 2015 Jun 29; Available from. Accessed.
- 64.U.S. National Institutes of Health, corp. http://clinicaltrials.gov/show/NCT01591356. ClinicalTrials.gov: EphA2 Gene Targeting Using Neutral Liposomal Small Interfering RNA Delivery. 2015 Jun 29; Available from. Accessed.
- 65.U.S. National Institutes of Health, corp. http://clinicaltrials.gov/show/NCT00689065. ClinicalTrials.gov: Safety Study of CALAA-01 to Treat Solid Tumor Cancers. 2015 Jun 29; Available from. Accessed. [Google Scholar]
- 66.U.S. National Institutes of Health, corp. http://clinicaltrials.gov/show/NCT01188785. ClinicalTrials.gov: Phase I-Escalating Dose Study of siG12D LODER (Local Drug EluteR) in Patients With Locally Advanced Adenocarcinoma of the Pancreas and a Single Dose Study of siG12D LODER (Local Drug EluteR) in Patients With Non-operable Adenocarcinoma of the Pancreas. 2015 Jun 29; Available from. Accessed. [Google Scholar]
- 67.U.S. National Institutes of Health, corp. http://clinicaltrials.gov/show/NCT01676259. ClinicalTrials.gov: A Phase II Study of siG12D LODER in Combination With Chemotherapy in Patients With Unresectable Locally Advanced Pancreatic Cancer. 2015 Jun 29; Available from. Accessed.