The vascular smooth muscle cell (VSMC) is a major component of the blood vessel wall and plays a critical role in the pathogenesis of atherosclerosis and vascular restenosis [1]. VSMCs undergo significant phenotype changes in response to inflammatory stimuli and mechanical injury associated with these disorders, which leads to aberrant proliferation and migration, apoptosis, and induction of matrix metalloproteinases (MMPs) [2]. Phenotypic switching of VSMCs has also been reported in the context of aortic aneurysms [3]. Although atherosclerosis and aortic aneurysms share several common risk factors (e.g. age, hypertension, smoking and obesity), aortic aneurysms are distinguished from atherosclerosis by distinct mechanisms of disease progression (e.g. prominent VSMC apoptosis, media degradation and elastin destruction) [4]. The role of VSMCs in aortic aneurysms is controversial, as they can contribute to inflammation and matrix metalloproteinase (MMP) production [5], yet they are also capable of synthesizing matrix proteins. Yamanouchi et al. reported that treatment with a caspase inhibitor suppressed aneurysm formation in a mouse model, most likely by preventing VSMC apoptosis [6], suggesting that VSMCs can play a salutary role in aneurysm pathogenesis.
Oxidative stress is pronounced in aortic aneurysm tissue and has been implicated in VSMC apoptosis and aneurysm pathogenesis [7]. VSMCs express various NADPH oxidases (NOX) isoforms which can regulate VSMC apoptosis, proliferation, migration, etc. [8]. VSMC-specific overexpression of catalase, a peroxisomal enzyme which scavenges hydrogen peroxide formed through dismutation of NOX-derived superoxide, protected against abdominal aortic aneurysm formation [9]. The transgenic mice with VSMC-specific overexpression of catalase exhibited less apoptosis, vascular inflammation, and MMP activity as compared to controls. This suggests that specifically protecting VSMC against oxidative stress has a salubrious effect on aortic aneurysm formation via several potential mechanisms.
Hypoxic conditions in aneurysmal tissues can also alter vascular function, potentially leading to apoptosis, inflammation and weakening of the arterial wall [10]. Hypoxia in aortic aneurysm tissues has been reported to stimulate inflammation, resulting in local aortic wall weakening [11]. HIF-1α, a master transcription factor induced by hypoxia, was reported to be markedly upregulated in human aneurysmal arteries, localized primarily in VSMCs and adventitia of aneurysmal tissues [12]. In mice, HIF-1α expression was also increased in tissues from elastase-induced abdominal aortic aneurysms [13]. Interestingly, increased HIF-1α in abdominal aortic aneurysm was reported to induce MMP expression in human VSMCs [14]. Furthermore, pharmacological inhibition or genetic silencing of HIF-1α was recently reported to attenuate abdominal aortic aneurysm formation by downregulating MMP-2 and MMP-9 expression and activity [15, 16], suggesting that HIF-1α may positively regulate aortic aneurysm formation.
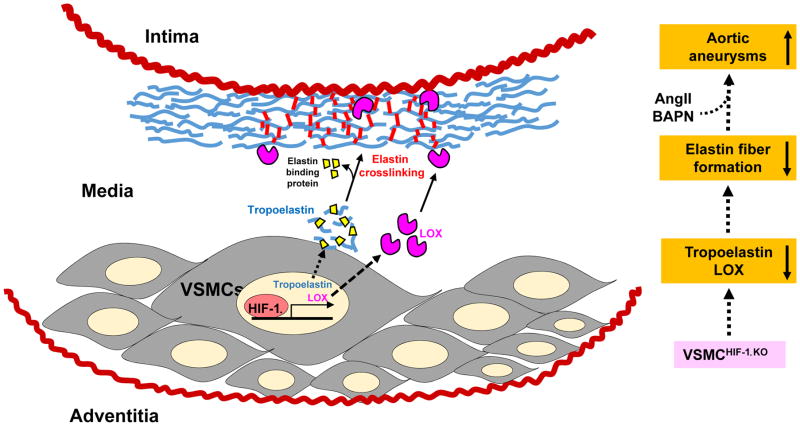
In this issue of ATVB, Imanish et al. shed new light on the protective role of VSMC-specific HIF-1α for the maintenance of vascular wall integrity in the context of aortic aneurysms. Previously, they showed that VSMC-specific HIF-1α deficiency (VSMCHIF-1αKO) inhibited angiotensin II (AngII)-induced medial thickening via modulating VSMC hypertrophy and collagen I expression/fibrosis [17]. In the current study, using the VSMCHIF-1αKO mouse infused with AngII plus β-aminopropionitrile (BAPN), they report that deficiency of HIF-1α in VSMC exacerbates aortic aneurysm formation by suppressing elastin expression and cross-linking rather than by modulating MMP activity. Mechanistically, VSMCHIF-1αKO mice exhibited reduced expression of tropoelastin, a major source of elastin fiber, as well as lysyl oxidase (LOX), an elastin cross-linking enzyme in the vascular media. These findings suggest that endogenous HIF-1α in VSMC augments elastin fiber formation, thereby opposing matrix degradation and aortic wall weakening that leads to aneurysm formation (Figure). The authors also reported excessive loss of VSMC in the VSMCHIF-1αKO mice; although apoptosis was not specifically quantified, they imply that it was mild. In vitro studies in VSMC from VSMCHIF-1αKO mice demonstrated reduced tropoelastin mRNA expression, suggesting a transcriptional regulatory mechanism linked to HIF-1α.
Figure 1.
Proposed role of HIF-1α expression in VSMC in aortic elastin fiber formation. HIF-1α deficiency in VSMC suppresses tropoelastin and LOX expression in the vascular media, thereby diminishing mature cross-linked elastin fiber formation. VSMCHIF-1αKO mice exhibited augmented aortic aneurysm formation when infused with AngII + BAPN.
The findings of the present study are contrary to recent reports in AngII-infused hyperlipidemic mice that HIF-1α induction exacerbated AngII-induced abdominal aortic aneurysms by upregulating MMPs, while HIF-1α inhibition or gene silencing attenuated aneurysm formation or progression [15, 16]. These results likely reflect the divergent effects of HIF-1α in the various cell types that contribute to the pathogenesis of aortic aneurysms. In other words, the protective effect that HIF-1α plays in VSMC may be overshadowed by detrimental effects in other cell types that foster aneurysm disease. Other factors, such as the specific aneurysm model employed, may also be important. In particular, the use of BAPN, which inhibits LOX activity, in the study by Imanish et al. may have compounded the effect of VSMCHIF-1αKO to reduce LOX expression in the blood vessel wall.
With advances in vascular biology research, the molecular and cellular mechanisms of aortic aneurysm formation have begun to be revealed. The role of aortic VSMC in the pathogenesis of aneurysms has received little attention to date, especially compared with the plethora of studies focused on inflammatory cells. The study by Imanish et al. adds to the growing body of data that VSMCs can protect against aortic aneurysm formation. Importantly, the authors provide proof-of-principle that HIF-1α-driven synthesis of matrix proteins by VSMC can actively oppose protease-mediated degradation and defend the integrity of the aortic wall.
Acknowledgments
Sources of Funding
Dr. Weintraub is supported by grants HL126949, HL112640, AR070029, and HL134354 from the National Institutes of Health.
Footnotes
Disclosures
None.
References
- 1.Lacolley P, Regnault V, Nicoletti A, Li Z, Michel JB. The vascular smooth muscle cell in arterial pathology: a cell that can take on multiple roles. Cardiovasc Res. 2012;95(2):194–204. doi: 10.1093/cvr/cvs135. [DOI] [PubMed] [Google Scholar]
- 2.Gomez D, Owens GK. Smooth muscle cell phenotypic switching in atherosclerosis. Cardiovasc Res. 2012;95(2):156–164. doi: 10.1093/cvr/cvs115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mao N, Gu T, Shi E, Zhang G, Yu L, Wang C. Phenotypic switching of vascular smooth muscle cells in animal model of rat thoracic aortic aneurysm. Interact Cardiovasc Thorac Surg. 2015;21(1):62–70. doi: 10.1093/icvts/ivv074. [DOI] [PubMed] [Google Scholar]
- 4.Peshkova IO, Schaefer G, Koltsova EK. Atherosclerosis and aortic aneurysm - is inflammation a common denominator? FEBS J. 2016;283(9):1636–1652. doi: 10.1111/febs.13634. [DOI] [PubMed] [Google Scholar]
- 5.Airhart N, Brownstein BH, Cobb JP, Schierding W, Arif B, Ennis TL, Thompson RW, Curci JA. Smooth muscle cells from abdominal aortic aneurysms are unique and can independently and synergistically degrade insoluble elastin. J Vasc Surg. 2014;60(4):1033–1041. doi: 10.1016/j.jvs.2013.07.097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yamanouchi D, Morgan S, Kato K, Lengfeld J, Zhang F, Liu B. Effects of caspase inhibitor on angiotensin II-induced abdominal aortic aneurysm in apolipoprotein E-deficient mice. Arterioscler Thromb Vasc Biol. 2010;30(4):702–7. doi: 10.1161/ATVBAHA.109.200527. [DOI] [PubMed] [Google Scholar]
- 7.McCormick ML, Gavrila D, Weintraub NL. Role of oxidative stress in the pathogenesis of abdominal aortic aneurysms. Arterioscler Thromb Vasc Biol. 2007;27:461–469. doi: 10.1161/01.ATV.0000257552.94483.14. [DOI] [PubMed] [Google Scholar]
- 8.Clempus RE, Griendling KK. Reactive oxygen species signaling in vascular smooth muscle cells. Cardiovasc Res. 2006;71(2):216–225. doi: 10.1016/j.cardiores.2006.02.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Parastatidis I, Weiss D, Joseph G, Taylor WR. Overexpression of catalase in vascular smooth muscle cells prevents the formation of abdominal aortic aneurysms. Arterioscler Thromb Vasc Biol. 2013;33(10):2389–2396. doi: 10.1161/ATVBAHA.113.302175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vorp DA, Federspiel WJ, Webster MW. Does laminated intraluminal thrombus within abdominal aortic aneurysm cause anoxia of the aortic wall? J Vasc Surg. 1996;23:540–541. doi: 10.1016/s0741-5214(96)80027-9. [DOI] [PubMed] [Google Scholar]
- 11.Vorp DA, Lee PC, Wang DH, Makaroun MS, Nemoto EM, Ogawa S, Webster MW. Association of intraluminal thrombus in abdominal aortic aneurysm with local hypoxia and wall weakening. J Vasc Surg. 2001;34(2):291–9. doi: 10.1067/mva.2001.114813. [DOI] [PubMed] [Google Scholar]
- 12.Hu XH, Yang J, Liu CW, Zhang ZS, Zhang Q. The expression and significance of hypoxia-inducible factor-1 alpha and related genes in abdominal aorta aneurysm. Zhonghua Wai Ke Za Zhi. 2004;42:1509–1012. [PubMed] [Google Scholar]
- 13.Van Vickle-Chavez SJ, Tung WS, Absi TS, Ennis TL, Mao D, Cobb JP, Thompson RW. Temporal changes in mouse aortic wall gene expression during the development of elastase-induced abdominal aortic aneurysms. J Vasc Surg. 2006;43:1010–1020. doi: 10.1016/j.jvs.2006.01.004. [DOI] [PubMed] [Google Scholar]
- 14.Erdozain OJ, Pegrum S, Winrow VR, Horrocks M, Stevens CR. Hypoxia in abdominal aortic aneurysm supports a role for HIF-1α and Ets-1 as drivers of matrix metalloproteinase upregulation in human aortic smooth muscle cells. J Vasc Res. 2011;48(2):163–170. doi: 10.1159/000318806. [DOI] [PubMed] [Google Scholar]
- 15.Tsai SH, Huang PH, Hsu YJ, Peng YJ, Lee CH, Wang JC, Chen JW, Lin SJ. Inhibition of hypoxia inducible factor-1α attenuates abdominal aortic aneurysm progression through the down-regulation of matrix metalloproteinases. Sci Rep. 2016;6:28612. doi: 10.1038/srep28612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yang L, Shen L, Li G, Yuan H, Jin X, Wu X. Silencing of hypoxia inducible factor-1α gene attenuated angiotensin II-induced abdominal aortic aneurysm in apolipoprotein E-deficient mice. Atherosclerosis. 2016;252:40–49. doi: 10.1016/j.atherosclerosis.2016.07.010. [DOI] [PubMed] [Google Scholar]
- 17.Imanishi M, Tomita S, Ishizawa K, Kihira Y, Ueno M, Izawa-Ishizawa Y, Ikeda Y, Yamano N, Tsuchiya K, Tamaki T. Smooth muscle cell-specific Hif-1alpha deficiency suppresses angiotensin II-induced vascular remodelling in mice. Cardiovasc Res. 2014;102:460–468. doi: 10.1093/cvr/cvu061. [DOI] [PubMed] [Google Scholar]