Abstract
A frailty index (FI) of deficit accumulation could quantify and predict the risk of fractures based on the degree of frailty in the elderly. We aimed to compare the predictive powers between the FI and the fracture risk assessment tool (FRAX) in predicting risk of major osteoporotic fracture (hip, upper arm or shoulder, spine, or wrist) and hip fracture, using the data from the Global Longitudinal Study of Osteoporosis in Women (GLOW) 3-year Hamilton cohort. There were 3985 women included in the study, with the mean age of 69.4 years (standard deviation [SD] = 8.89). During the follow-up, there were 149 (3.98%) incident major osteoporotic fractures and 18 (0.48%) hip fractures reported. The FRAX and FI were significantly related to each other. Both FRAX and FI significantly predicted risk of major osteoporotic fracture, with a hazard ratio (HR) of 1.03 (95% confidence interval [CI]: 1.02–1.05) and 1.02 (95% CI: 1.01–1.04) for per-0.01 increment for the FRAX and FI respectively. The HRs were 1.37 (95% CI: 1.19–1.58) and 1.26 (95% CI: 1.12–1.42) for an increase of per-0.10 (approximately one SD) in the FRAX and FI respectively. Similar discriminative ability of the models was found: c-index = 0.62 for the FRAX and c-index = 0.61 for the FI. When cut-points were chosen to trichotomize participants into low-risk, medium-risk and high-risk groups, a significant increase in fracture risk was found in the high-risk group (HR = 2.04, 95% CI: 1.36–3.07) but not in the medium-risk group (HR = 1.23, 95% CI: 0.82–1.84) compared with the low-risk women for the FI, while for FRAX the medium-risk (HR = 2.00, 95% CI: 1.09–3.68) and high-risk groups (HR = 2.61, 95% CI: 1.48–4.58) predicted risk of major osteoporotic fracture significantly only when survival time exceeded 18 months (550 days). Similar findings were observed for hip fracture and in sensitivity analyses. In conclusion, the FI is comparable with FRAX in the prediction of risk of future fractures, indicating that measures of frailty status may aid in fracture risk assessment and fracture prevention in the elderly. Further evidence from randomized controlled trials of osteoporosis medication interventions is needed to support the FI and FRAX as validated measures of fracture risk.
Keywords: FRAX, Frailty index, Hip fracture, Major osteoporotic fracture, Predictive power, GLOW
Introduction
As the population ages worldwide, increasing fractures in the elderly become a greater public health issue [1]. Approximately 340,000 hospitalizations due to hip fractures occurred among persons older than 65 years in USA in 1996 [2], and it is estimated that this annual number of hip fractures will double by the year 2040 [3,4]. Fractures in the elderly are usually followed by impaired quality of life, long-term care, permanent disability and high mortality [5,6].
The fracture risk assessment tool (FRAX) is a recognized computer-based algorithm that uses clinical risk factors to predict absolute 10-year fracture probability in men and women [7,8]. Currently FRAX is being used worldwide with or without femoral neck bone mineral density (BMD) to enhance the prediction of fracture risk [7,8]. Nevertheless, concerns have been expressed about FRAX including that it is difficult to interpret in clinical practice, does not capture many other risk factors (e.g., falls, physical activity, or dose of alcohol, smoking and glucocorticoid use), and has a wide variation and limited discriminative ability in different populations in the prediction of fractures [9–14].
Frailty is defined as a dynamic state of vulnerability which affects an individual in his/her physical, psychological and social functioning and increases the risks of adverse health outcomes [15]. A frailty index (FI) of deficit accumulation is widely used to measure frailty in terms of using a range of health deficits to quantify the cumulative effect [16, 17]. In the elderly, a higher FI was related with higher risk of adverse health outcomes at all ages, despite the deficit accumulation with chronological age across the age spectrum [18,19]. A validated FI was especially useful in evaluating the preventive or therapeutic performance of health and social services provision and policies as a population indication with precise estimates of risks of adverse health outcomes [20–22].
The FI could grade and predict the risk of fractures based on the degree of frailty in the elderly, which had been explored in previous studies [23–26]. However, limited evidence comparing the predictive accuracy of the FI and FRAX in the risk of fractures is available. Therefore, in this study, we aimed to compare the predictive powers between the FI of deficit accumulation and FRAX in predicting risk of fractures, based on the data from the Global Longitudinal Study of Osteoporosis in Women (GLOW) 3-year Hamilton cohort. The primary objective was to compare the predictive accuracy between the FRAX and FI in 3-year risk of major osteoporotic fracture (hip, upper arm or shoulder, spine, or wrist), and the secondary objective aimed at 3-year risk of hip fracture.
Methods
Participants and setting
GLOW is an international longitudinal study which involves 17 sites in 10 countries (Canada, US, Australia, Belgium, France, Germany, Italy, Netherlands, Spain and UK). It has been described previously in detail [27]. GLOW was performed to assess the health consequences of and risk factors for fragility fractures in 60,393 women no less than 55 years old who had consulted their physician in the past 24 months. The participants were stratified according to age strata such that approximately two-thirds of women aged ≥65 years. Women were eligible to enter the cohort if they had no cognitive impairment or language barriers, and were not institutionalized or too ill to finish the study survey [27].
Our study was a longitudinal analysis of the 3-year GLOW Hamilton, Canada cohort. A sample of about 4000 women were enrolled between May 2008 and March 2009 and followed up every year to complete the survey. Participants were surveyed by mailing questionnaires annually, in which the questionnaires collected data on participant characteristics and risk factors, health care access and use, co-morbidities, perception about fracture risk and osteoporosis, physical activity, medication use, physical function and quality of life [27]. Telephone interviews were conducted if the participant did not return the mailed survey or needed assistance in completing the questionnaire. The study was approved by the Western Institutional Review Board. All participants provided written informed consent before the survey.
The FRAX and the FI of deficit accumulation
The FRAX algorithm aimed to provide an assessment for the prediction of 10-year risks of hip fracture and major osteoporotic fracture (hip, clinical spine, wrist, or proximal humerus fracture) in women and men, using clinical risk factors with or without femoral neck BMD [7,8]. The clinical risk factors included age, gender, weight, height, a history of fragility fracture, parental hip fracture, current smoking, alcohol intake of ≥3 units daily, use of oral glucocorticoids, diagnosis of rheumatoid arthritis, and secondary osteoporosis [7,28]. Since data on BMD testing was not collected in the GLOW protocol [27], BMD was not included in the FRAX algorithm to predict risks of fractures. We used the Canadian FRAX algorithm [29] to calculate the scores of 10-year fracture risk in this study.
The FI of deficit accumulation was calculated by 34 baseline health deficits including activity of daily living (n = 12), signs and symptoms (n = 6), comorbidities (n = 15) and healthcare utilization (n = 1). The construction of the FI has been described previously based on the GLOW Hamilton cohort [30]. Each deficit was coded by dichotomization or polychotomization to map the interval 0–1 so that the coding could represent the frequency or severity of the deficits. For instance, as regards the question of ‘self-rating of health’, the response of ‘poor’ was coded as 1, ‘fair’ as 0.75, ‘good’ as 0.5, ‘very good’ as 0.25 and ‘Excellent’ as 0. The deficits included in the FI and their individual coding were presented in Supplementary Table 1. To calculate a FI for an individual participant, her values of deficits were summed up and divided by the total number of variables (n = 34) in the FI, with the FI ranging from 0 to 1 [30]. For example, if a women had 4 deficits with each score of 1 point, 2 deficits with each score of 0.5 and the other 28 deficits with each score of 0, then her FI of deficit accumulation would hence be 5 divided by 34 giving a FI = 0.15.
Outcomes
Participants reported fragility fractures that had occurred from age 45 years in baseline questionnaires including spine, clavicle, rib, upper arm or shoulder, wrist, pelvis, hip, upper leg, lower leg or ankle fracture. During the annual follow-up survey, participants documented their incident fractures and the dates in the questionnaires. To directly compare the FI with FRAX, the primary outcome was survival time to first incident major osteoporotic fracture (hip, upper arm or shoulder, spine, or wrist) during the follow-up, while the secondary outcome was time to first incident hip fracture.
Statistical analyses
Data were presented as mean and standard deviation (SD) for continuous variables and frequency and proportion for categorized variables, respectively. Comparison at baseline by the 3-year incident major osteoporotic fracture status was examined using Chi-square tests for the categorical variables and Student’s t-test for continuous variables.
To analyze the associations between FRAX or FI and incident fractures during follow-up, Cox proportional hazards models were performed and hazard ratios (HRs) were used to quantify the relationship. Unadjusted HR for FRAX was presented in univariable analyses, whereas results were reported in the age-adjusted model for the FI given that health deficits were all age-associated and age was not included in the FI in the elderly [22,31,32].
Results were presented according to an increase of 0.01 (or 1%) on the FI and FRAX [33]. We also reported the HRs measured by per-0.10 (approximately one SD) increment of the FI and FRAX [26,30]. Moreover, findings from the categorized groups were also displayed using the cut-points of FRAX and FI. For major osteoporotic fracture, participants were categorized into low-risk, medium-risk and high-risk group based on the 10-year fracture risk probability calculated from FRAX, with the cut-points of 10% and 20% for FRAX [34,35]. To directly compare the predictive accuracy between FRAX and FI, we adopted Rockwood’s methodology [36] to choose the cut-points of the FI and trichotomize the participants. The cut-points of 0.20 and 0.35 on the FI were identified by overlapping the FI’s density distributions of the robust (or low-risk), pre-frail (or medium-risk) and frail (high-risk) women, in which the robust, pre-frail and frail group were defined by the widely used and validated phenotypic frailty model in geriatrics [30,36,37]. The rationale for this methodology was that the frailer a woman was, the greater the likelihood that she would fracture in the future [22,26,38]. On the other hand, for hip fracture, we dichotomized the women into low-risk and high-risk group using the cut-point of 3% for FRAX as recommended [35]. With regard to the FI, we chose its median of 0.22 to categorize participants into low-risk and high-risk group, for the purpose of direct comparison with FRAX in predicting risk of hip fracture.
Survival curves for major osteoporotic fracture were graphed using the Kaplan–Meier (KM) method and compared by the log-rank test between the three groups categorized by the aforementioned cut-points of FRAX and FI, respectively. To assess the proportional hazards (PH) assumption, both a statistical test and a graphical examination using Schoenfeld residuals were performed. Because evidence was found that the PH assumption was not satisfied for the grouped FRAX (low-risk, medium-risk and high-risk), an extended Cox model was used by splitting the survival time. The goodness of fit of the models was assessed based on the Cox–Snell residuals and the Gronnesby and Borgan test. Model discrimination was assessed with the use of Harrell’s c-index.
We performed two sensitivity analyses to assess the robustness of the findings. First, a sensitivity analysis was conducted using an accelerated failure time model with a Weibull distribution to compare the findings from Cox proportional hazards model analyses. Another sensitivity analysis was performed to assess the impact of missing data. We used multiple imputation approach [39] to impute missing data if the percentage of missing data was more than 10%. If data of <10% on a variable were missing, the median or mean of the variable was used for imputation. Furthermore, we conducted an analysis stratified by the women who were taking and not-taking anti-osteoporotic treatment, calcium and/or vitamin D supplementation at baseline, to investigate the impact of the treatment and/or supplementation on the prediction of the FRAX and FI in fractures in this study.
All tests were two-sided according to a significance level of 0.05. All analyses were performed using STATA Version 12 (Stata Corp., College Station, TX, USA).
Results
There were 3985 women included in the study at baseline (Table 1). The mean age was 69.4 years (SD = 8.89), and the mean body mass index (BMI) was 27.7 (SD = 5.77) kg/m2. There were 11% and 49% of participants smoking and drinking respectively. The proportions of the other components include in FRAX by self-report were low: 22% for history of fragility fracture, 16% for parental hip fracture, 12% for diagnosis of rheumatoid arthritis, 4% for use of glucocorticoids and 24% for diagnosis of secondary osteoporosis.
Table 1.
Baseline characteristics of participants and comparison by the 3-year incident major osteoporotic fracture status*.
| Characteristics | Total participants (n = 3985) | Major osteoporotic fracturea
|
||
|---|---|---|---|---|
| No (n = 3594) | Yes (n = 149) | p-Value | ||
| Age: mean (SD), years | 69.4 (8.89) | 69.1 (8.71) | 71.3 (9.43) | 0.002b |
| BMI: mean (SD), kg/m2 | 27.7 (5.77) | 27.7 (5.73) | 27.5 (5.74) | 0.53b |
| Smoker, n (%) | ||||
| Yes | 447 (11.30) | 393 (11.01) | 15 (10.20) | 0.76c |
| No | 3510 (88.70) | 3178 (88.99) | 132 (89.80) | |
| Drinking (drinks/week), n (%) | ||||
| 0 | 2027 (51.21) | 1809 (50.63) | 84 (57.14) | 0.58d |
| 7 | 1414 (35.73) | 1299 (36.36) | 42 (28.57) | |
| 7–13 | 428 (10.81) | 389 (10.89) | 16 (10.88) | |
| 14–20 | 76 (1.92) | 66 (1.85) | 4 (2.72) | |
| >20 | 13 (0.33) | 10 (0.28) | 1 (0.68) | |
| Race, n (%) | ||||
| White | 3717 (93.27) | 3374 (93.88) | 138 (92.62) | 0.53c |
| Non-white | 268 (6.73) | 220 (6.12) | 11 (7.38) | |
| Education, n (%) | ||||
| High school or less | 2509 (64.10) | 2239 (63.36) | 97 (66.44) | 0.45c |
| More than high school | 1405 (35.90) | 1295 (36.64) | 49 (33.56) | |
| Prior fractures since 45 years old, n (%) | ||||
| Yes | 862 (22.31) | 737 (21.11) | 52 (35.86) | <0.001c |
| No | 3001 (77.69) | 2754 (78.89) | 93 (64.14) | |
| Parental hip fracture, n (%) | ||||
| Yes | 599 (15.88) | 534 (15.67) | 33 (23.40) | 0.014c |
| No | 3173 (84.12) | 2874 (84.33) | 108 (76.60) | |
| Rheumatoid arthritis, n (%) | ||||
| Yes | 447 (11.57) | 383 (10.97) | 26 (18.06) | 0.008c |
| No | 3418 (88.43) | 3107 (89.03) | 118 (81.94) | |
| Use of glucocorticoids, n (%) | ||||
| Yes | 141 (3.63) | 121 (3.45) | 10 (6.90) | 0.029c |
| No | 3748 (96.37) | 3387 (96.55) | 135 (93.10) | |
| Secondary osteoporosis, n (%) | ||||
| Yes | 931 (24.21) | 827 (23.83) | 43 (30.07) | 0.087c |
| No | 2914 (75.79) | 2644 (76.17) | 100 (69.93) | |
| FRAXe: mean (SD) | ||||
| All participants (n = 2847) | 0.16 (0.099) | 0.16 (0.098) | 0.20 (0.11) | <0.001b |
| Low-risk (n = 929) | 0.075 (0.015) | 0.075 (0.015) | 0.079 (0.015) | – |
| Medium-risk (n = 1159) | 0.15 (0.029) | 0.14 (0.029) | 0.16 (0.028) | – |
| High-risk (n = 759) | 0.29 (0.089) | 0.29 (0.089) | 0.31 (0.091) | – |
| FI: mean (SD) | ||||
| All participants (n = 3985) | 0.24 (0.13) | 0.24 (0.13) | 0.28 (0.15) | <0.001b |
| Low-risk (n = 1749) | 0.12 (0.047) | 0.12 (0.047) | 0.13 (0.049) | – |
| Medium-risk (n = 1357) | 0.27 (0.043) | 0.27 (0.043) | 0.27 (0.044) | – |
| High-risk (n = 879) | 0.44 (0.073) | 0.44 (0.072) | 0.46 (0.086) | – |
SD: standard deviation; BMI: body mass index; FRAX: fracture risk assessment tool; FI: frailty index.
Mean follow-up = 3.01 years.
Hip, upper arm or shoulder, spine, or wrist.
Student’s t-test.
Chi-square test.
Mantel–Haenszel Chi-square test.
Baseline FRAX scores of 10-year risk of major osteoporotic fractures.
FRAX and FI related to each other significantly, with correlation coefficients of 0.20 for both the continuous values and categorized groups (p < 0.001). The baseline FRAX scores of 10-year risk of major osteoporotic fractures had a mean of 0.16 (SD = 0.099), ranging from 0.03 to 0.79. The mean of the FI was 0.24 (SD = 0.13), with a range from 0 to 0.72. Based on the cut-points, there were 41% (n = 1159) and 27% (n = 759) of the women categorized into medium-risk and high-risk group by FRAX respectively, whereas the proportions were 34% (n = 1357) and 22% (n = 879) for medium-risk and high-risk group defined by the FI respectively (Table 1).
During the 3-year follow-up, there were 149 (3.98%) incident major osteoporotic fractures (hip, upper arm or shoulder, spine, or wrist) reported. Women with incident fractures were older, had significantly more prior fractures, parental hip fractures, rheumatoid arthritis and the use of glucocorticoids than the participants without incident major osteoporotic fractures (all p-values < 0.05, Table 1). Women with incident major osteoporotic fractures (n = 149) had significantly higher baseline FRAX scores (0.20) and FI (0.28) than those without fractures (FRAX = 0.16; FI = 0.24; n = 3594). The 3-year incidence rates for the low-risk, medium-risk and high-risk group defined by the FRAX were 2.48% (n = 23), 3.81% (n = 44) and 5.80% (n = 44) respectively, while the proportions were 2.86% (n = 50), 3.62% (n = 49) and 5.69% (n = 50) for the FI groups respectively.
Table 2 shows the comparison of prediction and model performances between FRAX and FI in risk of 3-year major osteoporotic fracture. Both the FRAX and FI significantly predicted risk of major osteoporotic fracture, with a HR of 1.03 (95% CI: 1.02–1.05, p < 0.001) and 1.02 (95% CI: 1.01–1.04, p < 0.001) for per-0.01 increment of the FRAX and FI respectively. The HRs were 1.37 (95% CI: 1.19–1.58) and 1.26 (95% CI: 1.12–1.42) for an increase of per-0.10 (approximately one SD) in the FRAX and FI respectively. No PH assumption violation was observed for the incremental FRAX or FI. The models were both of good fit (p > 0.05). The c-indices implied similar discriminative values of the models for major fracture: c-index = 0.62 for the FRAX and c-index = 0.61 for the FI respectively.
Table 2.
Comparison of prediction and model performances between FI and FRAX in 3-year risk of major osteoporotic fracture.
| Model | Major osteoporotic fracturea (n = 149)
|
||
|---|---|---|---|
| HR (95% CI), p-value | C-index | p-Value for goodness of fit test | |
| Models with per-0.01 increment | |||
| FRAX | 1.03 (1.02–1.05), <0.001 | 0.62 | 0.15 |
| FIb | 1.02 (1.01–1.04), <0.001 | 0.61 | 0.85 |
| Models with categorized groups | |||
| FRAXc | |||
| Medium-risk | N/Ae | N/Ae | |
| <365 days | 1.33 (0.75–2.38), 0.34 | ||
| 365–550 days | 1.28 (0.62–2.65), 0.51 | ||
| >550 days | 2.00 (1.09–3.68), 0.026 | ||
| High-risk | |||
| <365 days | 1.73 (0.78–3.87), 0.18 | ||
| 365–550 days | 1.66 (0.80–3.44), 0.17 | ||
| >550 days | 2.61 (1.48–4.58), 0.001 | ||
| FIb,d | |||
| Medium-risk | 1.23 (0.82–1.84), 0.31 | 0.61 | 0.22 |
| <365 days | 1.10 (0.69–1.75), 0.70 | ||
| 365–550 days | 1.47 (0.81–2.67), 0.21 | ||
| >550 days | 1.84 (1.16–2.93), 0.009 | ||
| High-risk | 2.04 (1.36–3.07), 0.001 | ||
| <365 days | 1.62 (0.85–3.09), 0.15 | ||
| 365–550 days | 1.27 (0.81–2.01), 0.30 | ||
| >550 days | 2.14 (1.20–3.82), 0.006 | ||
HR: hazard ratio; CI: confidence interval; FRAX: fracture risk assessment tool; FI: frailty index.
Hip, upper arm or shoulder, spine, or wrist.
Adjusted for age.
Low-risk group taken as reference group; the cut-points were 10% and 20% for major osteoporotic fracture; comparisons of high-risk versus low-risk and medium-risk versus low-risk group were within the same follow-up duration strata.
Low-risk group taken as reference group; the cut-points were 0.20 and 0.35 for major osteoporotic fracture; comparisons of high-risk versus low-risk and medium-risk versus low-risk group were within the same follow-up duration strata.
No estimation of c index or goodness of fit test conducted due to time-varying variable included in the model.
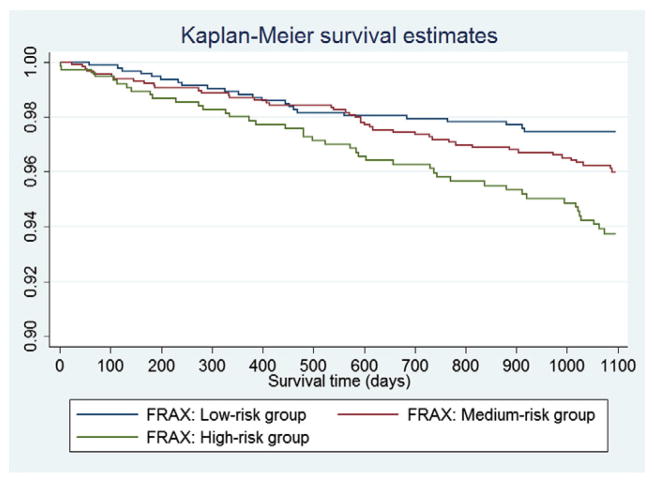
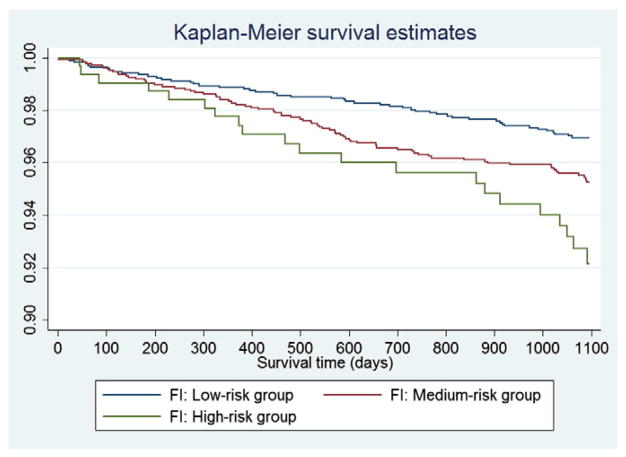
Figs. 1 and 2 show the KM survival curves for the participants trichotomized by the FRAX and FI respectively. As shown in Fig. 1, the KM curves crossed approximately at 12 months (365 days) and 18 months (550 days). The statistical test and graphical examination based on Schoenfeld residuals also indicated that the PH assumption was violated for the categorized groups by FRAX. Therefore, an extended Cox model was conducted by splitting survival time into <365 days, 365–550 days and >550 days (Table 2). The relationship between risk of major osteoporotic fracture and the FRAX was significant only when survival time exceeded 550 days, with the HRs of 2.00 (95% CI: 1.09–3.68, p = 0.026) and 2.61 (95% CI: 1.48–4.58, p = 0.001) for the medium-risk and high-risk group compared with the low-risk group respectively (Table 2). On the other hand, no PH assumption violation was found for the three groups defined by the FI. Log-rank test indicated the significant difference of survival curves between the three groups (p < 0.001). Compared with the low-risk group, only the high-risk group presented a significant effect on the major osteoporotic fracture (HR = 2.04, 95% CI: 1.36–3.07, p = 0.001). The model for the categorized FI was of good fit (p = 0.22) and it had a c-index of 0.61 (Table 2). Moreover, to show the direct comparison between FRAX and FI, we also conducted an extended Cox analysis for the FI by splitting the same survival time spans (<365 days, 365–550 days and >550 days). Similarly, significant associations between risk of major osteoporotic fracture and the FI were only detected when survival time was larger than 550 days: HR = 1.84 (95% CI: 1.16–2.93, p = 0.009) for medium-risk group and HR = 2.14 (95% CI: 1.20–3.82, p = 0.006) for high-risk group, compared with low-risk women.
Fig. 1.
Kaplan–Meier survival curves in the three groups defined by the FRAX.
Fig. 2.
Kaplan–Meier survival curves in the three groups defined by the FI.
There were 18 (0.48%) incident hip fractures reported during the 3-year follow-up (Table 3). Significant associations were found between the per-0.01 increment for FRAX and FI and risk of hip fracture, with a HR of 1.05 (95% CI: 1.02–1.09) and 1.03 (95% CI: 1.00–1.07) for the FRAX and FI respectively. Per-0.10 increase in the FRAX and FI yielded a HR of 1.64 (95% CI: 1.16–2.32) and 1.39 (95% CI: 1.00–1.94) respectively. When the participants were dichotomized into low-risk and high-risk group, nevertheless, only the high-risk group defined by FRAX predicted the risk of hip fracture significantly (HR = 4.05, 95% CI: 1.12–14.72) (Table 3). PH assumption was satisfied for all the models for hip fracture.
Table 3.
Comparison of prediction and model performances between FI and FRAX in 3-year risk of hip fracture.
| Model | Hip fracture (n = 18)
|
||
|---|---|---|---|
| HR (95% CI), p-value | C-index | p-Value for goodness of fit test | |
| Models with per-0.01 increment | |||
| FRAX | 1.05 (1.02–1.09), 0.004 | 0.70 | 0.22 |
| FIa | 1.03 (1.00–1.07), 0.050 | 0.72 | 0.78 |
| Models with categorized groups | |||
| FRAXb | |||
| High-risk | 4.05 (1.12–14.72), 0.034 | 0.66 | N/Ad |
| FIc | |||
| High-risk | 2.09 (0.72–6.04), 0.18 | 0.72 | N/Ad |
HR: hazard ratio; CI: confidence interval; FRAX: fracture risk assessment tool; FI: frailty index.
Adjusted for age.
Low-risk group taken as reference group; the cut-point was 3% for the FRAX; comparison of high-risk versus low-risk group was within the same follow-up duration strata.
Low-risk group taken as reference group; the cut-point was 0.22 for the FI; comparison of high-risk versus low-risk group was within the same follow-up duration strata.
No goodness of fit test conducted due to small sample size.
A sensitivity analysis using accelerated failure time models with a Weibull distribution yielded similar results to the Cox models for the relationship between the FRAX and FI and risk of major osteoporotic fracture and hip fracture (Table 4). Because approximately 29% (n = 1138) of the women had missing data on the FRAX scores at baseline, models were performed by using multiple imputations to investigate the relationship between FRAX and risk of major osteoporotic fracture and hip fracture, without altering the findings (Supplementary Table 2). Furthermore, to directly compare the prediction of the FRAX and FI in fractures, we restricted the analyses in the same participants whose both baseline FRAX and FI were available (n = 2847). The results remained unchanged from the main analyses (Supplementary Table 3).
Table 4.
Results from sensitivity analyses using accelerated failure time models for major osteoporotic fracture and hip fracture.
| Model | Major osteoporotic fracturea (n = 149)
|
Hip fracture (n = 18)
|
||
|---|---|---|---|---|
| HR (95% CI)e | p-Value | HR (95% CI)e | p-Value | |
| Models with per-0.01 increment | ||||
| FRAX | 1.03 (1.02–1.05) | <0.001 | 1.05 (1.02–1.09) | 0.003 |
| FIb | 1.03 (1.01–1.04) | <0.001 | 1.03 (1.00–1.08) | 0.049 |
| Models with categorized groups | ||||
| FRAXc | ||||
| Medium-risk | N/Af | N/Af | ||
| <365 days | 1.38 (0.80–2.40) | 0.25 | ||
| 365–550 days | 1.45 (0.80–2.64) | 0.23 | ||
| >550 days | 1.76 (1.03–3.02) | 0.039 | ||
| High-risk | 4.05 (1.12–14.73) | 0.034 | ||
| <365 days | 1.93 (0.97–3.85) | 0.062 | ||
| 365–550 days | 2.02 (1.10–3.72) | 0.024 | ||
| >550 days | 2.45 (1.46–4.14) | 0.001 | ||
| FIb,d | ||||
| Medium-risk | 1.23 (0.82–1.85) | 0.30 | N/Af | N/Af |
| High-risk | 2.04 (1.36–3.07) | 0.001 | 2.09 (0.72–6.05) | 0.17 |
HR: hazard ratio; CI: confidence interval; FRAX: fracture risk assessment tool; FI: frailty index.
Hip, upper arm or shoulder, spine, or wrist.
Adjusted for age.
Low-risk group taken as reference group; the cut-points were 10% and 20% for major osteoporotic fracture, while the cut-point was 3% for hip fracture; comparisons of high-risk versus low-risk and medium-risk versus low-risk group were within the same follow-up duration strata.
Low-risk group taken as reference group; the cut-points were 0.20 and 0.35 for major osteoporotic fracture, while the cut-point was 0.22 for hip fracture; comparisons of high-risk versus low-risk and medium-risk versus low-risk group were within the same follow-up duration strata.
Converted from accelerated factor.
Not applicable for hip fracture due to its dichotomization.
Table 5 shows results of the relationship between the 3-year risk of major osteoporotic fracture and FI and FRAX stratified by participants’ use of anti-osteoporotic treatment and/or supplementation. During follow-up, there were 108 and 41 incident major osteoporotic fractures reported in the participants receiving anti-osteoporotic treatment, calcium and/or vitamin D supplementation (n = 2912) and the women taking none of these drugs (n = 1073) at baseline, respectively. Similar findings were observed in the women with and without treatment and/or supplementation (Table 5), indicating no significant effect of the treatment and/or supplementation on the prediction of FI and FRAX in major osteoporotic fracture in this population.
Table 5.
Relationship between risk of major osteoporotic fracture and FI and FRAX stratified by participants’ use of antiosteoporotic treatment, calcium and/or vitamin D supplementation.
| Model | Major osteoporotic fracturea
|
|||
|---|---|---|---|---|
| Participants with antiosteoporotic treatment and/or supplementation (n = 2912)b
|
Participants without antiosteoporotic treatment or supplementation (n = 1073)c
|
|||
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | |
| Models with per-0.01 increment | ||||
| FRAX | 1.03 (1.01–1.05) | <0.001 | 1.04 (1.01–1.08) | 0.019 |
| FId | 1.02 (1.01–1.04) | 0.001 | 1.02 (1.00–1.05) | 0.033 |
| Models with categorized groups | ||||
| FRAXe | ||||
| Medium-risk | ||||
| <365 days | 1.54 (0.78–3.08) | 0.22 | 0.92 (0.30–2.78) | 0.87 |
| 365–550 days | 1.52 (0.66–3.53) | 0.33 | 0.62 (0.12–3.29) | 0.58 |
| >550 days | 1.89 (0.98–3.65) | 0.051 | 2.35 (0.64–8.64) | 0.20 |
| High-risk | ||||
| <365 days | 1.97 (0.76–5.11) | 0.17 | 1.26 (0.26–6.03) | 0.77 |
| 365–550 days | 1.94 (0.84–4.48) | 0.12 | 0.86 (0.16–4.83) | 0.87 |
| >550 days | 2.42 (1.25–4.71) | 0.009 | 3.26 (1.10–9.67) | 0.034 |
| FId,f | ||||
| Medium-risk | 1.21 (0.76–1.93) | 0.43 | 1.29 (0.58–2.88) | 0.54 |
| High-risk | 2.00 (1.24–3.23) | 0.004 | 2.14 (1.03–4.45) | 0.041 |
HR: hazard ratio; CI: confidence interval; FRAX: fracture risk assessment tool; FI: frailty index.
Hip, upper arm or shoulder, spine, or wrist.
There were 108 incident major osteoporotic fracture reported.
There were 41 incident major osteoporotic fracture reported.
Adjusted for age.
Low-risk group taken as reference group; the cut-points were 10% and 20% for major osteoporotic fracture, while the cut-point was 3% for hip fracture; comparisons of high-risk versus low-risk and medium-risk versus low-risk group were within the same follow-up duration strata.
Low-risk group taken as reference group; the cut-points were 0.20 and 0.35 for major osteoporotic fracture, while the cut-point was 0.22 for hip fracture; comparisons of high-risk versus low-risk and medium-risk versus low-risk group were within the same follow-up duration strata.
Discussion
Main findings
Using the 3-year GLOW Hamilton cohort, the predictive accuracy between the FI of deficit accumulation and FRAX was compared in risk of major osteoporotic fracture and hip fracture. The FRAX and FI related to each other significantly. Similar results were found for the per-0.01 increment of the FRAX and FI in predicting risk of fractures. The high-risk group defined by FRAX predicted 3-year risk of major osteoporotic fracture and hip fracture significantly, while the high-risk group categorized by the FI only showed a significant association with major osteoporotic fracture. The models for FRAX and FI were of good fit and with similar discriminative values. Results from accelerated failure time models and multiple imputations corroborated the robustness of the findings.
In this study, we reported similar results from the continuous FRAX and FI, given that the continuous results could be used as a population indication in evaluating the preventive or therapeutic performance of health [20]. On the other hand, the categorical FRAX was adopted widely in clinical practice to aid in the decisions of further assessment and pharmacologic treatment [12,35]. This was also consistent with the categorical FI, in which the therapeutic or preventive interventions should be specifically directed to the frail or high-risk population [36,40]. The widely-used FRAX consists of clinical risk factors identified from meta-analyses and it has been externally validated and calibrated in specific populations [14]. For the elderly, frailty was a nonspecific state of vulnerability that could be measured and quantified using a FI of deficit accumulation, in which the FI covered a comprehensive domains of deficits related with aging including co-morbidities, signs and symptoms, activities of daily living, healthcare utilization, etc [30,40]. Compared with the clinical risk factors of FRAX, the FI of deficit accumulation approach predicted the risk of future fractures by quantifying the frailty status. Because participants with more deficits were more likely to be frail and more likely to experience adverse health events [22], measuring their cumulative deficits could contribute to predicting their increased risk of fractures [26]. In this study, comparable findings from continuous and categorical FRAX and FI supported that the frailty approach was not inferior to the recognized FRAX in prediction of fractures (Tables 2, 3).
In this study, comparable results were found in the women who were taking and not-taking anti-osteoporotic treatment and/or supplementation at baseline (Table 5). These findings were also consistent with the main results using the whole population (Table 2), implying no significant effect of the baseline anti-osteoporotic treatment and/or supplementation on the prediction of FRAX and FI in major osteoporotic fracture. Some studies argued that previous falls should be included into FRAX, since previous falls were an independent and significant risk factor for future fractures [12,28]. Nevertheless, evidence showed that models incorporating falls as a risk factor may not be superior to FRAX alone in predicting future risk of fractures [41]. In our study, a post-hoc analysis incorporating baseline falls into the models yielded similar results for both FRAX and FI to the parsimonious models (Table 2), e.g., for the per-0.01 increment the HR was 1.03 and 1.02 and the c-index was 0.61 and 0.61 for FRAX and FI respectively.
Comparison with other studies
Previous studies investigating the relationship between a FI and fractures reported significant results [23–26], which was in line with our findings. For example, the Canadian Multicentre Osteoporosis Study (CaMos) using a 30-item FI to predict 10-year fracture risk reported significant associations between the FI and fractures in adults, with a HR of 1.18 for hip fracture and 1.30 for clinical vertebral fractures (p-values < 0.05) for per-0.10 increase in FI [26]. Nevertheless, no evidence directly comparing the prediction of the FRAX and FI approaches in fractures was available. Besides, there were some suggestions incorporating frailty into FRAX, especially in the previous fallers, to improve the precision of FRAX’s predictive power [42]. However, in this study we did not combine the FI and FRAX into the same model, because the FI shared the same three components of FRAX (i.e., use of oral glucocorticoids, rheumatoid arthritis, and secondary osteoporosis) and both prediction tools were significantly associated with each other. Nonetheless, the suggestions did shed some light in that the measures of frailty contributed to the prediction of fractures and the management of frailty could assist in reducing the risk of future fractures [42–44].
Clinical implication
This study yielded similar and comparable relationship between major osteoporotic and hip fracture and the FRAX and FI, indicating that the frailty measure may be as important as FRAX based on clinical risk factors to predict risk of fracture in the elderly. However, compared with the wide-spread application of FRAX in clinical settings, the FI approach may be helpful only as a research tool in population settings, especially given the large amount of information required.
Limitations and strengths
Our study has several limitations. Originally FRAX was designed to predict 10-year risk of fractures, while this study only provided 3-year data. As shown in Fig. 1 and Table 2, the grouped FRAX predicted the risk of major osteoporotic fracture significantly with longer follow-up (i.e., > 550 days), when the medium-risk or high-risk group was compared with the low-risk group. Therefore the short-term observation may compromise the predictive power of both FRAX and FI. Furthermore, since the dates of death were not available in GLOW, the FI could not take into account the competing risk of death for prediction of future fractures, whereas the competing mortality risk was included in the FRAX algorithm. Failing to account for the competing risk of death may bias the prediction of risk of fractures using the FI. Another limitation was due to the inclusion criteria of no cognitive impairment for the participants in GLOW, because cognitive impairment was a significant risk factor for fractures [45]. Moreover, all the data collected in GLOW were only from self-report of participants and could not be validated by medical records. Besides, recall bias was inevitably induced when participants were completing questionnaires. Furthermore, the measures of BMD may assist FRAX with improvement of predictive accuracy in risk of fractures [46], but BMD was not available in GLOW. Even though emerging evidence reported that FRAX with BMD was in good agreement with FRAX alone, and FRAX alone provided the same prediction as FRAX with BMD [47–50], it was unknown whether the FRAX with BMD outperformed the FRAX alone in the elderly in this study. Lastly, the small number of hip fracture (n = 18) resulted in imprecise estimates and wide confidence intervals (Tables 3 and 4). Similarly, due to the small proportions of the older participants and small number of facture events, no analysis was conducted for the very old women. For example, there were only 5 incident major osteoporotic fractures and 4 hip fractures reported in the women aged >85 years (n = 139, 3%). Likewise, there were only 13 major osteoporotic fractures and 5 hip fractures found in the participants aged >80 years (n = 494, 12%).
This study yielded comparable findings from the FI and FRAX in the prediction of risk of major osteoporotic and hip fractures in the elderly, which may provide some insights into the fracture prevention and risk assessment using the measures of frailty status. The FI approach assisted with the investigation of aging and the prediction of adverse health events including fractures, by quantifying the degree of frailty. This may aid in the decision of intervention plans and evaluations for geriatricians, bone specialists, health researchers and policy-makers. Furthermore, different strategies and sensitivity analyses yielded similar results, which underpinned the robustness of our findings. A third strength was the representative sample because of the unique sampling method in GLOW [27]. Women were enrolled according to the lists provided by their physician practices with few exclusion criteria, which therefore led to the overall participants being representative of the practices [51].
Conclusion
To conclude, both the FI and FRAX significantly predict risk of major osteoporotic and hip fracture significantly using the GLOW 3-year Hamilton cohort. The FI is comparable with FRAX in prediction of risk of future fractures, indicating that measures of frailty status may aid in the fracture risk assessment and fracture prevention in the elderly. Further evidence from randomized controlled trials of osteoporosis medication interventions is needed to support the FI and FRAX as validated measures of fracture risk.
Supplementary Material
Acknowledgments
Funding
Financial support for the GLOW was provided by Warner Chilcott Company, LLC and Sanofi-Aventis to the Center for Outcomes Research, University of Massachusetts Medical School. GL received a Father Sean O’Sullivan Research Award, the Research Institute of St. Joe’s Hamilton, and a doctoral award from the China Scholarship Council. The sponsors did not get involved in the study design or conduct; data collection, analyses or interpretation; and writing, review or approval of this manuscript.
The authors acknowledged the whole GLOW research group investigators herein.
Abbreviations
- FI
frailty index
- FRAX
fracture risk assessment tool
- GLOW
Global Longitudinal Study of Osteoporosis in Women
- CI
confidence interval
- SD
standard deviation
- HR
hazard ratio
- BMD
bone mineral density
- KM
Kaplan–Meier
- PH
proportional hazards
- BMI
body mass index
Appendix A. Supplementary data
Supplementary data to this article can be found online at http://dx.doi.org/10.1016/j.bone.2015.04.028.
Footnotes
Conflict of interest
The authors declare that they have no conflicts of interest.
References
- 1.Cummings SR, Melton LJ. Epidemiology and outcomes of osteoporotic fractures. Lancet. 2002;359:1761–7. doi: 10.1016/S0140-6736(02)08657-9. [DOI] [PubMed] [Google Scholar]
- 2.Stevens JA, Olson S. Reducing falls and resulting hip fractures among older women. MMWR Recomm Rep. 2000;49:3–12. [PubMed] [Google Scholar]
- 3.Antapur P, Mahomed N, Gandhi R. Fractures in the elderly: when is hip replacement a necessity? Clin Interv Aging. 2011;6:1–7. doi: 10.2147/CIA.S10204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cummings SR, Rubin SM, Black D. The future of hip fractures in the United States. Numbers, costs, and potential effects of postmenopausal estrogen. Clin Orthop Relat Res. 1990:163–6. [PubMed] [Google Scholar]
- 5.Beaupre LA, Jones CA, Saunders LD, Johnston DW, Buckingham J, Majumdar SR. Best practices for elderly hip fracture patients. A systematic overview of the evidence. J Gen Intern Med. 2005;20:1019–25. doi: 10.1111/j.1525-1497.2005.00219.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Court-Brown CM, Caesar B. Epidemiology of adult fractures: a review. Injury. 2006;37:691–7. doi: 10.1016/j.injury.2006.04.130. [DOI] [PubMed] [Google Scholar]
- 7.Kanis JA, Johnell O, Oden A, Johansson H, McCloskey E. FRAX and the assessment of fracture probability in men and women from the UK. Osteoporos Int. 2008;19:385–97. doi: 10.1007/s00198-007-0543-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kanis JA, McCloskey EV, Johansson H, Oden A, Strom O, Borgstrom F. Development and use of FRAX in osteoporosis. Osteoporos Int. 2010;21(Suppl 2):S407–13. doi: 10.1007/s00198-010-1253-y. [DOI] [PubMed] [Google Scholar]
- 9.Tremollieres FA, Pouilles JM, Drewniak N, Laparra J, Ribot CA, Dargent-Molina P. Fracture risk prediction using BMD and clinical risk factors in early postmenopausal women: sensitivity of the WHO FRAX tool. J Bone Miner Res. 2010;25:1002–9. doi: 10.1002/jbmr.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Azagra R, Roca G, Encabo G, Aguye A, Zwart M, Guell S, et al. FRAX(R) tool, the WHO algorithm to predict osteoporotic fractures: the first analysis of its discriminative and predictive ability in the Spanish FRIDEX cohort. BMC Musculoskelet Disord. 2012;13:204. doi: 10.1186/1471-2474-13-204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yu R, Leung J, Woo J. Sarcopenia combined with FRAX probabilities improves fracture risk prediction in older Chinese men. J Am Med Dir Assoc. 2014;15:918–23. doi: 10.1016/j.jamda.2014.07.011. [DOI] [PubMed] [Google Scholar]
- 12.Unnanuntana A, Gladnick BP, Donnelly E, Lane JM. The assessment of fracture risk. J Bone Joint Surg. 2010;92:743–53. doi: 10.2106/JBJS.I.00919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kanis JA, Oden A, Johansson H, Borgstrom F, Strom O, McCloskey E. FRAX and its applications to clinical practice. Bone. 2009;44:734–43. doi: 10.1016/j.bone.2009.01.373. [DOI] [PubMed] [Google Scholar]
- 14.Leslie W, Lix L. Comparison between various fracture risk assessment tools. Osteoporos Int. 2014;25:1–21. doi: 10.1007/s00198-013-2409-3. [DOI] [PubMed] [Google Scholar]
- 15.Gobbens RJ, Luijkx KG, Wijnen-Sponselee MT, Schols JM. Toward a conceptual definition of frail community dwelling older people. Nurs Outlook. 2010;58:76–86. doi: 10.1016/j.outlook.2009.09.005. [DOI] [PubMed] [Google Scholar]
- 16.Rockwood K, Song X, MacKnight C, Bergman H, Hogan DB, McDowell I, et al. A global clinical measure of fitness and frailty in elderly people. CMAJ. 2005;173:489–95. doi: 10.1503/cmaj.050051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Li G, Thabane L, Ioannidis G, Kennedy C, Papaioannou A, Adachi JD. Comparison between frailty index of deficit accumulation and phenotypic model to predict risk of falls: data from the global longitudinal study of osteoporosis in women (GLOW) Hamilton cohort. PLoS One. 2015;10:e0120144. doi: 10.1371/journal.pone.0120144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rockwood K, Mitnitski A. Frailty in relation to the accumulation of deficits. J Gerontol A Biol Sci Med Sci. 2007;62:722–7. doi: 10.1093/gerona/62.7.722. [DOI] [PubMed] [Google Scholar]
- 19.Rockwood K, Song X, Mitnitski A. Changes in relative fitness and frailty across the adult lifespan: evidence from the Canadian National Population Health Survey. CMAJ. 2011;183:E487–94. doi: 10.1503/cmaj.101271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rockwood K, Song X, MacKnight C, Bergman H, Hogan DB, McDowell I, et al. A global clinical measure of fitness and frailty in elderly people. Can Med Assoc J. 2005;173:489–95. doi: 10.1503/cmaj.050051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cesari M, Gambassi G, van Kan GA, Vellas B. The frailty phenotype and the frailty index: different instruments for different purposes. Age Ageing. 2014;43:10–2. doi: 10.1093/ageing/aft160. [DOI] [PubMed] [Google Scholar]
- 22.Rockwood K, Mitnitski A. Frailty in relation to the accumulation of deficits. J Gerontol A: Biol Med Sci. 2007;62:722–7. doi: 10.1093/gerona/62.7.722. [DOI] [PubMed] [Google Scholar]
- 23.Fang X, Shi J, Song X, Mitnitski A, Tang Z, Wang C, et al. Frailty in relation to the risk of falls, fractures, and mortality in older Chinese adults: results from the Beijing longitudinal study of aging. J Nutr Health Aging. 2012;16:903–7. doi: 10.1007/s12603-012-0368-6. [DOI] [PubMed] [Google Scholar]
- 24.Krishnan M, Beck S, Havelock W, Eeles E, Hubbard RE, Johansen A. Predicting outcome after hip fracture: using a frailty index to integrate comprehensive geriatric assessment results. Age Ageing. 2014;43:122–6. doi: 10.1093/ageing/aft084. [DOI] [PubMed] [Google Scholar]
- 25.Albaba M, Cha SS, Takahashi PY. The Elders Risk Assessment Index, an electronic administrative database-derived frailty index, can identify risk of hip fracture in a cohort of community-dwelling adults. Mayo Clin Proc. 2012;87:652–8. doi: 10.1016/j.mayocp.2012.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kennedy C, Ioannidis G, Rockwood K, Thabane L, Adachi J, Kirkland S, et al. A Frailty Index predicts 10-year fracture risk in adults age 25 years and older: results from the Canadian Multicentre Osteoporosis Study (CaMos) Osteoporos Int. 2014;25:2825–32. doi: 10.1007/s00198-014-2828-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hooven FH, Adachi JD, Adami S, Boonen S, Compston J, Cooper C, et al. The Global Longitudinal Study of Osteoporosis in Women (GLOW): rationale and study design. Osteoporos Int. 2009;20:1107–16. doi: 10.1007/s00198-009-0958-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kanis JA, Oden A, Johansson H, Borgström F, Ström O, McCloskey E. FRAX® and its applications to clinical practice. Bone. 2009;44:734–43. doi: 10.1016/j.bone.2009.01.373. [DOI] [PubMed] [Google Scholar]
- 29.Leslie WD, Lix LM, Langsetmo L, Berger C, Goltzman D, Hanley DA, et al. Construction of a FRAX(R) model for the assessment of fracture probability in Canada and implications for treatment. Osteoporos Int. 2011;22:817–27. doi: 10.1007/s00198-010-1464-2. [DOI] [PubMed] [Google Scholar]
- 30.Li G, Ioannidis G, Pickard L, Kennedy C, Papaioannou A, Thabane L, et al. Frailty index of deficit accumulation and falls: data from the Global Longitudinal Study of Osteoporosis in Women (GLOW) Hamilton cohort. BMC Musculoskelet Disord. 2014;15:185. doi: 10.1186/1471-2474-15-185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rockwood K, Mitnitski A. Frailty defined by deficit accumulation and geriatric medicine defined by frailty. Clin Geriatr Med. 2011;27:17–26. doi: 10.1016/j.cger.2010.08.008. [DOI] [PubMed] [Google Scholar]
- 32.Searle SD, Mitnitski A, Gahbauer EA, Gill TM, Rockwood K. A standard procedure for creating a frailty index. BMC Geriatr. 2008;8:24. doi: 10.1186/1471-2318-8-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kulminski AM, Ukraintseva SV, Kulminskaya IV, Arbeev KG, Land KK, Yashin AI. Cumulative deficits better characterize susceptibility to death in elderly people than phenotypic frailty: lessons from the Cardiovascular Health Study. J Am Geriatr Soc. 2008;56:898–903. doi: 10.1111/j.1532-5415.2008.01656.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Siminoski K, Leslie WD, Frame H, Hodsman A, Josse RG, Khan A, et al. Recommendations for bone mineral density reporting in Canada. J Can Assoc Radiol. 2005;56:178. [PubMed] [Google Scholar]
- 35.Cosman F, de Beur S, LeBoff M, Lewiecki E, Tanner B, Randall S, et al. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25:2359–81. doi: 10.1007/s00198-014-2794-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rockwood K, Andrew M, Mitnitski A. A comparison of two approaches to measuring frailty in elderly people. J Gerontol A: Biol Med Sci. 2007;62:738–43. doi: 10.1093/gerona/62.7.738. [DOI] [PubMed] [Google Scholar]
- 37.Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, et al. Frailty in older adults evidence for a phenotype. J Gerontol A: Biol Med Sci. 2001;56:M146–57. doi: 10.1093/gerona/56.3.m146. [DOI] [PubMed] [Google Scholar]
- 38.Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet. 2013;381:752–62. doi: 10.1016/S0140-6736(12)62167-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Graham JW. Missing data analysis: making it work in the real world. Annu Rev Psychol. 2009;60:549–76. doi: 10.1146/annurev.psych.58.110405.085530. [DOI] [PubMed] [Google Scholar]
- 40.De Vries N, Staal J, Van Ravensberg C, Hobbelen J, Olde Rikkert M, Nijhuis-Van der Sanden M. Outcome instruments to measure frailty: a systematic review. Ageing Res Rev. 2011;10:104–14. doi: 10.1016/j.arr.2010.09.001. [DOI] [PubMed] [Google Scholar]
- 41.Sambrook PN, Flahive J, Hooven FH, Boonen S, Chapurlat R, Lindsay R, et al. Predicting fractures in an international cohort using risk factor algorithms without BMD. J Bone Miner Res. 2011;26:2770–7. doi: 10.1002/jbmr.503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Blain H, Rolland Y, Beauchet O, Annweiler C, Benhamou CL, Benetos A, et al. Usefulness of bone density measurement in fallers. Joint Bone Spine. 2014;81:403–8. doi: 10.1016/j.jbspin.2014.01.020. [DOI] [PubMed] [Google Scholar]
- 43.Rolland Y, Abellan van Kan G, Benetos A, Blain H, Bonnefoy M, Chassagne P, et al. Frailty, osteoporosis and hip fracture: causes, consequences and therapeutic perspectives. J Nutr Health Aging. 2008;12:335–46. doi: 10.1007/BF02982665. [DOI] [PubMed] [Google Scholar]
- 44.Masud T, Binkley N, Boonen S, Hannan MT. Official positions for FRAX® clinical regarding falls and frailty: can falls and frailty be used in FRAX®?: from Joint Official Positions Development Conference of the International Society for Clinical Densitometry and International Osteoporosis Foundation on FRAX®. J Clin Densitom. 2011;14:194–204. doi: 10.1016/j.jocd.2011.05.010. [DOI] [PubMed] [Google Scholar]
- 45.Guo Z, Wills P, Viitanen M, Fastbom J, Winblad B. Cognitive impairment, drug use, and the risk of hip fracture in persons over 75 years old: a community-based prospective study. Am J Epidemiol. 1998;148:887–92. doi: 10.1093/oxfordjournals.aje.a009714. [DOI] [PubMed] [Google Scholar]
- 46.Johansson H, Kanis J, Odén A, Johnell O, McCloskey E. BMD, clinical risk factors and their combination for hip fracture prevention. Osteoporos Int. 2009;20:1675–82. doi: 10.1007/s00198-009-0845-x. [DOI] [PubMed] [Google Scholar]
- 47.Gadam RK, Schlauch K, Izuora KE. Frax prediction without BMD for assessment of osteoporotic fracture risk. Endocr Pract. 2013;19:780–4. doi: 10.4158/EP12416.OR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Olmez Sarikaya N, Kapar Yavasi S, Tan G, Satiroglu S, Yildiz AH, Oz B, et al. Agreement between FRAX scores calculated with and without bone mineral density in women with osteopenia in Turkey. Clin Rheumatol. 2014;33:1785–9. doi: 10.1007/s10067-014-2491-8. [DOI] [PubMed] [Google Scholar]
- 49.Hamdy RC, Kiebzak GM. Variance in 10-year fracture risk calculated with and without T-scores in select subgroups of normal and osteoporotic patients. J Clin Densitom. 2009;12:158–61. doi: 10.1016/j.jocd.2008.12.003. [DOI] [PubMed] [Google Scholar]
- 50.Leslie WD, Morin S, Lix LM, Johansson H, Oden A, McCloskey E, et al. Fracture risk assessment without bone density measurement in routine clinical practice. Osteoporos Int. 2012;23:75–85. doi: 10.1007/s00198-011-1747-2. [DOI] [PubMed] [Google Scholar]
- 51.Díez-Pérez A, Adachi JD, Adami S, Anderson FA, Boonen S, Chapurlat R, et al. Risk factors for treatment failure with antiosteoporosis medication: the Global Longitudinal Study of Osteoporosis in Women (GLOW) J Bone Miner Res. 2014;29:260–7. doi: 10.1002/jbmr.2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.