Abstract
Background
Early life rhinovirus (RV) wheezing illnesses and aeroallergen sensitization increase the risk of asthma at school age. Whether these remain risk factors for the persistence of asthma out to adolescence is not established.
Objective
We sought to define the relationships among specific viral illnesses and the type and timing of aeroallergen sensitization with the persistence of asthma into adolescence.
Methods
A total of 217 children were followed prospectively from birth to age 13 years. The etiology and timing of viral wheezing illnesses during the first 3 years of life were assessed along with patterns of allergen sensitization. The associations between viral wheezing illnesses, presence and pattern of aeroallergen sensitization, and asthma diagnosis at age 13 years were evaluated.
Results
When adjusted for all viral etiologies, wheezing with RV (odds ratio = 3.3; 95% CI, 1.5-7.1), but not respiratory syncytial virus (odds ratio = 1.0; 95% CI, 0.4-2.3), was associated with asthma at age 13 years. Age of aeroallergen sensitization also influenced asthma risk; 65% of children sensitized by age 1 year had asthma at age 13 years, compared with 40% of children not sensitized at age 1 year but sensitized by age 5 years, and 17% of children not sensitized at age 5 years. Early life aeroallergen sensitization and RV wheezing had additive effects on asthma risk at adolescence.
Conclusions
In a high-risk birth cohort, the persistence of asthma at age 13 years was most strongly associated with outpatient wheezing illnesses with RV and aeroallergen sensitization in early life.
Key words: Rhinovirus, respiratory syncytial virus, allergic sensitization, asthma
Abbreviations used: COAST, Childhood Origins of Asthma; OR, Odds ratio; RSV, Respiratory syncytial virus; RV, Rhinovirus
The inception of asthma has been the focus of research by multiple groups over the past several decades.1, 2, 3, 4, 5, 6 A clear link has been established between both early life viral wheezing illnesses and allergic sensitization and the risk of developing childhood asthma. The etiologic role of specific viruses in the development of asthma has been investigated by our group7 and others.8, 9 Initial investigations in this area focused on the role of respiratory syncytial virus (RSV) infections and the risk for asthma and allergic disease.8, 10 The role of human rhinovirus (RV) infections in early life wheezing was underappreciated because of poor growth of RV using standard culture techniques. However, with the advent of molecule-based techniques,11 RV has now been well established to play an important role in early life viral wheezing. In the Childhood Origins of ASThma (COAST) birth cohort, we have reported that wheezing illnesses with RV in the first 3 years of life are the most significant predictor of asthma by age 6 years.7, 12 Moreover, the combined influence of early life viral wheezing and aeroallergen sensitization has been clearly linked to an even greater risk for the development of asthma by our group and others.7, 13 We have reported an important sequential relationship whereby allergic sensitization precedes viral wheezing, most notably with RV infections, in this developmental pathway.14 Furthermore, the link between RV wheezing and subsequent asthma can be modified by other factors, such as genetic susceptibility at 17q21.15
Although the role of viruses and allergic sensitization has been clearly implicated in the onset of asthma, their role in the persistence of asthma into later childhood and early adolescence has not been as well studied. The Tucson Children's Respiratory Study group previously reported that early life wheezing with RSV had a pronounced impact on early school-age asthma, but that this association dissipated over time and was no longer significant by age 13 years.8 With regard to allergic sensitization, the question of type and magnitude of aeroallergen sensitization and the resultant impact on childhood asthma have been evaluated,16 and longitudinal observations have shown that a differential effect on asthma inception based on the timing and type of sensitization exists.16 Specifically, early sensitization to multiple allergens confers the greatest risk of subsequent asthma inception and morbidity.16, 17 However, the natural history of these relationships out to adolescence has not been characterized.
In this study, we examined these complex interactions and their associations with the initial expression and persistence of asthma into adolescence. In particular, we aimed to assess the natural history of these factors in our high-risk birth cohort by prospectively evaluating the relationship among early life viral wheezing, the timing and pattern of aeroallergen sensitization, and the persistence of childhood asthma out to age 13 years.
Methods
Study subjects
A total of 289 newborns were enrolled from November 1998 through May 2000 in the COAST study as previously described.18, 19, 20 Of these children, 259 were followed prospectively to age 6 years and 217 to age 13 years. To qualify, at least 1 parent was required to have respiratory allergies (defined as 1 or more positive aeroallergen skin test results) and/or a history of physician-diagnosed asthma. The Human Subjects Committee of the University of Wisconsin approved the study, and informed consent was obtained from the parents and age-appropriate written assent was obtained from the children. There were no differences in demographic characteristics between those children followed to at least age 1 year compared with those followed up to age 13 years.
Nasal lavage samples
Nasopharyngeal mucus samples were collected during scheduled clinic visits (2, 4, 6, 9, 12 months, and annually afterward) and during times of acute respiratory illnesses as previously described.12 Briefly, parents notified a study coordinator when their child developed a respiratory tract illness and a symptom scorecard was completed.19 If the symptom score was 5 or greater, identifying a moderate to severe respiratory illness, a nasal lavage was performed and processed as described.19
Viral diagnostics
Nasal specimens were analyzed for respiratory viruses including RSV, RV, influenza types A and B, parainfluenza virus types 1 to 4, adenovirus, and enteroviruses using standard techniques.21 In addition, samples were evaluated for RV by seminested RT-PCR,21 and for the viruses listed above plus coronaviruses (OC143, NL63, and 0229) and metapneumoviruses by multiplex PCR (Respiratory MultiCode PLx Assay; EraGen Biosciences, Madison, Wis).22
Allergen-specific IgE
Allergen-specific IgE value was determined at years 1, 2, 3 for Dermatophagoides pteronyssinus, Dermatophagoides farinae, Alternaria alternata, dog, and cat allergens. Allergens tested at years 5, 6, 9, 11, and 13 were D pteronyssinus, D farinae, A alternata, dog, cat, ragweed, silver birch, timothy grass, and cockroach. Allergen-specific IgE values of 0.35 kU/L (class I) or greater were considered positive. The presence of allergic sensitization was defined by having 1 or more positive values for allergen-specific IgE.
Clinical definitions
Daycare attendance23 and atopic dermatitis20 were defined as previously described. A wheezing respiratory illness during the first 3 years of life was defined as meeting 1 or more of the following criteria as described previously7: (1) physician- diagnosed wheezing at an office visit; (2) an illness for which the child was prescribed short- or long-acting beta-agonists and/or controller medications; or (3) an illness given the following specific diagnoses: bronchiolitis, wheezing illness, reactive airway disease, asthma, or asthma exacerbation. Current asthma was diagnosed, as previously described,7 at ages 6, 8, 11, and 13 years on the basis of the documented presence of 1 or more of the following characteristics in the previous year: (1) physician diagnosis of asthma, (2) use of albuterol for coughing or wheezing episodes (prescribed by physician), (3) use of a daily controller medication, (4) step-up plan including the use of albuterol or short-term use of inhaled corticosteroids during illness, and (5) use of prednisone for asthma exacerbation.
Statistical methods
The relationships between early life risk factors and subsequent development of asthma at age 6 or 13 years were examined using logistic regression models of asthma, including univariate models for each risk factor, multivariate models including all risk factors, and multivariate models including a subset of risk factors chosen by stepwise backward variable selection using the Akaike information criterion. Relationships between the occurrence of 1 or more viral wheezing illnesses in the first 3 years of life and asthma at ages 6, 8, 11, and 13 years were examined using logistic regression models of asthma, both univariate models for each virus, and multivariate models including all viruses. Similarly, logistic regression models were also used to assess the relationships between asthma and aeroallergen sensitization, and between asthma and RV wheeze and sensitization. Results are reported as asthma rates and odds ratios (ORs) with 95% CIs. A 2-sided P value of less than .05 was regarded as statistically significant.
To investigate the relationship between patterns and number of aeroallergen sensitizations over time and subsequent asthma diagnosis, we conducted the following analysis. First, to address missing values for the number of sensitizations, 15 imputed data sets were created, using the aregImpute function in the R package Hmisc24 (http://CRAN.R-project.org/package=Hmisc), under an imputation model that included the number of sensitizations at each year (1, 2, 3, 5, 6, 9, 11, and 13); total IgE level at each year; asthma diagnoses at years 6, 8, 11, and 13; sex; wheezing history with RV and RSV during the first 3 years of life; fraction of exhaled nitric oxide measurements obtained from years 6 through 11; and atopic eczema patterns over the first 8 years of life. Next, classification trees were fit with asthma at year 13 as the response and the number of sensitizations at each year as potential predictors, using the R package rpart (http://CRAN.R-project.org/package=rpart); separate trees were fit using each imputed data set, and also a data set that averaged (median) the imputed number of sensitizations across all imputations. To avoid overfitting, cost-complexity pruning using cross-validation was performed on each tree. The consensus tree model was a 3-node tree formed by first splitting the population by the number of sensitizations by year 5 (0 vs 1+), then splitting the 1+ node according to the number of sensitizations by year 1 (0 vs 1+). Means and 95% CIs for asthma rates in each group were calculated by combining the estimates from each imputed data set using Rubin's method.25
Results
Viral detection
As previously reported, a total of 454 wheezing respiratory illnesses were documented during the first 3 years of life: 153 illnesses in 76 children aged 0 to 1 year, 155 illnesses in 72 children aged 1 to 2 years, and 146 illnesses in 63 children in the third year of life. Nasopharyngeal wash specimens were obtained during 442 (97%) of these wheezing illnesses. A viral etiology was identified in 398 (90%) of these specimens. The types of viruses detected during the first 3 years of life included RV (212 [48%]), RSV (93 [21%]), parainfluenza virus types 1 to 4 (51 [12%]), metapneumoviruses (33 [7%]), coronaviruses (20 [5%]), adenovirus (17 [4%]), influenza types A and B (16 [4%]), and enteroviruses (10 [2%]).7, 18
Risk factors and asthma
The associations between environmental factors and the development of asthma at age 6 and 13 years were analyzed. In stepwise multivariate analyses, allergic sensitization to aeroallergen or food at age 1 year (aeroallergen: OR, 2.7; 95% CI, 1.2-6.2; food: OR, 2.0; 95% CI, 1.0-3.9) and older siblings in the home during infancy (OR, 1.9; 95% CI, 1.0-3.5) were associated with increased risk for asthma at age 6 years (Table I ). At 13 years, only aeroallergen sensitization by age 1 year was associated with increased asthma risk (OR, 6.1; 95% CI, 2.5-14.4) (Table I). Regarding protective factors, the presence of a dog in the home at the time of birth was associated with reduced asthma risk by univariate analyses (OR, 0.5; 95% CI, 0.3-0.9), but this did not reach statistical significance in a stepwise multivariate model (OR, 0.6; 95% CI, 0.3-1.1) (Table I). At age 13 years, the presence of a dog in the household at the time of birth was no longer significantly protective, but having a cat at birth was associated with decreased asthma risk at age 13 years (OR, 0.5; 95% CI, 0.2-1.0) (Table I).
Table I.
Early life risk factors for asthma development
| Risk factors for asthma at age 13 y | With risk factor | Univariate |
Multivariate |
Multivariate (stepwise) |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| OR | 95% CI | P value | OR | 95% CI | P value | OR | 95% CI | P value | |||
| Maternal asthma∗ | 92/216 | 43% | 1.0 | (0.6-1.9) | .92 | 1.0 | (0.5-2.1) | .97 | — | — | — |
| Paternal asthma∗ | 68/215 | 32% | 1.4 | (0.8-2.6) | .27 | 1.3 | (0.6-2.8) | .58 | — | — | — |
| Birth month | — | — | — | — | .92 | — | — | .52 | — | — | — |
| Birth weight (lb) | — | — | 0.9 | (0.7-1.1) | .23 | 0.8 | (0.6-1.1) | .13 | — | — | — |
| Cat in household at birth | 66/217 | 30% | 0.6 | (0.3-1.2) | .13 | 0.5 | (0.2-1.1) | .08 | 0.5 | (0.2-1.0) | .05 |
| Dog in household at birth | 76/217 | 35% | 0.9 | (0.5-1.7) | .81 | 1.5 | (0.7-3.2) | .30 | — | — | — |
| Older siblings | 119/217 | 55% | 1.2 | (0.7-2.2) | .48 | 1.3 | (0.6-2.7) | .53 | — | — | — |
| Passive smoke exposure (first year) | 55/217 | 25% | 1.7 | (0.9-3.2) | .13 | 1.6 | (0.7-3.6) | .29 | — | — | — |
| Daycare (first year) | 105/217 | 48% | 1.1 | (0.6-2.0) | .65 | 1.2 | (0.6-2.6) | .61 | — | — | — |
| Exclusive breast-feeding (first 6 mo) | 75/217 | 35% | 1.2 | (0.6-2.1) | .63 | 1.6 | (0.7-3.5) | .27 | — | — | — |
| Atopic dermatitis (first year) | 58/211 | 27% | 1.8 | (0.9-3.4) | .07 | 2.2 | (1.0-4.7) | .06 | — | — | — |
| Aeroallergen sensitization (first year) | 27/214 | 13% | 5.1 | (2.2-12) | .0002 | 6.6 | (2.1-20.8) | .001 | 6.0 | (2.5-14.4) | <.0001 |
| Food sensitization (first year) | 54/214 | 25% | 2.7 | (1.4-5.1) | .003 | 1.5 | (0.6-3.5) | .39 | — | — | — |
| Risk factors for asthma at age 6 y | With risk factor | Univariate |
Multivariate |
Multivariate (stepwise) |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| OR | 95% CI | P value | OR | 95% CI | P value | OR | 95% CI | P value | |||
| Maternal asthma∗ | 111/258 | 43% | 1.3 | (0.8-2.3) | .32 | 1.8 | (0.9-3.4) | .09 | — | — | — |
| Paternal asthma∗ | 77/255 | 30% | 1.3 | (0.7-2.3) | .44 | 1.1 | (0.5-2.3) | .79 | — | — | — |
| Birth month | — | — | — | — | .30 | — | — | .39 | — | — | — |
| Birth weight (lb) | — | — | 1.1 | (0.8-1.3) | .67 | 0.9 | (0.7-1.2) | .53 | — | — | — |
| Cat in household at birth | 76/259 | 29% | 1.0 | (0.5-1.7) | .90 | 0.9 | (0.4-1.9) | .82 | — | — | — |
| Dog in household at birth | 92/259 | 36% | 0.5 | (0.3-0.9) | .02 | 0.5 | (0.3-1.1) | .09 | 0.6 | (0.3-1.1) | .08 |
| Older siblings | 145/259 | 56% | 1.8 | (1.0-3.1) | .05 | 1.8 | (0.9-3.6) | .08 | 1.9 | (1.0-3.5) | .03 |
| Passive smoke exposure (first year) | 64/259 | 25% | 1.1 | (0.6-2.1) | .76 | 0.7 | (0.3-1.5) | .37 | — | — | — |
| Daycare (first year) | 123/259 | 47% | 0.7 | (0.4-1.2) | .20 | 0.6 | (0.3-1.3) | .21 | — | — | — |
| Exclusive breast-feeding (first 6 mo) | 84/259 | 32% | 1.1 | (0.6-2.0) | .70 | 0.9 | (0.4-1.8) | .74 | — | — | — |
| Atopic dermatitis (first year) | 65/253 | 26% | 1.7 | (0.9-3.1) | .08 | 1.1 | (0.5-2.2) | .86 | — | — | — |
| Aeroallergen sensitization (first year) | 34/255 | 13% | 3.6 | (1.7-7.5) | .0008 | 4.3 | (1.6-11.8) | .005 | 2.7 | (1.2-6.2) | .02 |
| Food sensitization (first year) | 65/255 | 25% | 2.8 | (1.6-5.2) | .0006 | 1.7 | (0.8-3.7) | .21 | 2.0 | (1.0-3.9) | .04 |
Risk factors included in the multivariate stepwise model were chosen using backward elimination on the basis of Akaike's information criterion.
Maternal and paternal asthma data have been updated (fewer missing values) since Jackson et al.7
Longitudinal evaluation of viral illnesses in early childhood and asthma
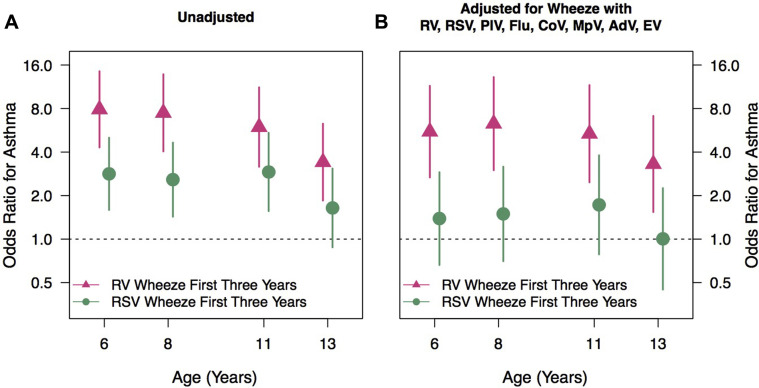
A wheezing history during the first 3 years of life was variably associated with a persistent increase in asthma risk at 13 years depending on viral etiology. Wheezing with RSV in the first 3 years of life was associated with an increased risk of asthma at ages 6, 8, and 11 years, but this was no longer significant at 13 years (OR, 2.8, 2.6, 2.9, and 1.6; 95% CI, 1.6-5, 1.4-4.6, 1.6-5.4, and 0.9-3.1) (Fig 1 , A). In contrast, wheezing with RV during the first 3 years of life was associated with a greater increased risk of asthma at age 6, 8, and 11 years that, unlike RSV, persisted out to the 13th year of age (OR, 7.9, 7.5, 6, and 3.4; 95% CI, 4.3-14, 4-14, 3.2-11, and 1.8-6.3) (Fig 1, A). In a multivariate model adjusted for wheezing with other viruses, RSV wheezing in the first 3 years of life was not associated with an increased asthma risk at any age (Fig 1, B). In contrast, RV wheezing was associated with a persistent increase in the risk of asthma at 6, 8, 11, and 13 years (OR, 5.5, 6.3, 5.4, and 3.3; 95% CI, 3.7-11, 3-13, 2.5-12, and 1.5-7.1) (Fig 1, B).
Fig 1.
A and B, Impact of viral etiology on asthma risk at age 6, 8, 11, and 13 years. AdV, Adenovirus; CoV, coronaviruses; EV, enteroviruses; Flu, influenza types A and B; MpV, metapneumoviruses; PIV, parainfluenza virus types 1 to 4.
Associations between developmental allergen sensitization patterns and asthma
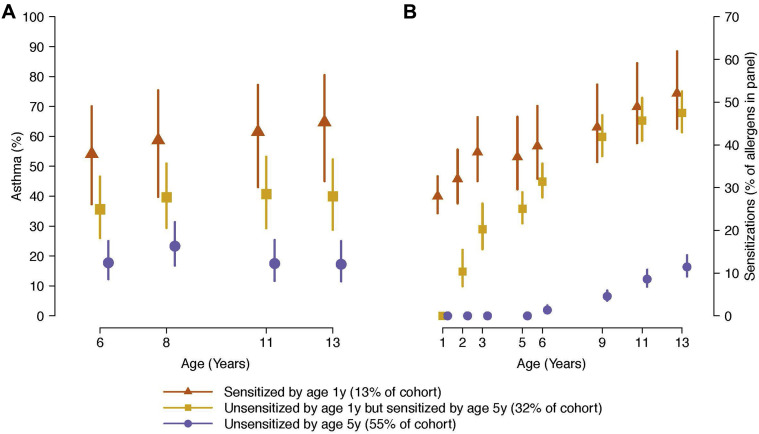
Developmental patterns of allergic sensitization and their relationship to subsequent asthma risk were examined using a decision tree learning model. Children who were sensitized to 1 or more aeroallergens by age 1 year (13% of the cohort) had the highest rate of asthma at year 13 (65%) (Fig 2 , A). Children who were not sensitized at year 1 but sensitized to 1 or more aeroallergens by age 5 years (32% of the cohort) had a 40% rate of asthma at age 13 years (Fig 2, A). The remaining children (55% of the cohort) were not sensitized by age 5 years and had an asthma rate of 17% at age 13 years (Fig 3 , A). Notably, among this last group, nearly half (46%) of the children later became sensitized between ages 6 and 13 years to at least 1 aeroallergen, but their asthma risk at age 13 years was not different from that of children who were not sensitized through year 13 (16% and 18%, respectively).
Fig 2.
A, Identification of the role of timing of aeroallergen in asthma risk. B, Children sensitized by age 1 or 5 years had similar numbers of positive allergen sensitizations at age 13 years, but significantly more than children not sensitized by age 5 years.
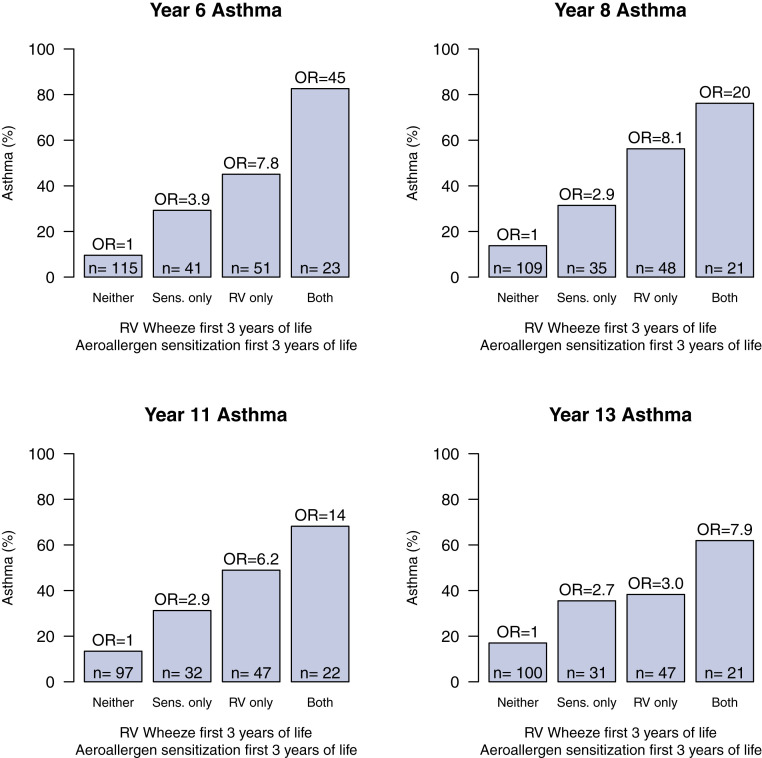
Fig 3.
Both aeroallergen sensitization and RV wheezing illnesses in the first 3 years of life increased asthma risk. The effects are additive and those children with both risk factors had the highest risk between age 6 and 13 years.
The average number of sensitizations at each year in each group is shown in Fig 3, B. Interestingly, in contrast to their lower asthma risk at age 13 years, children who were sensitized by year 5 but not by year 1 have similar numbers of sensitizations at ages 9, 11, and 13 years compared with children who were sensitized by age 1 year.
Allergic sensitization, RV wheezing, and asthma
Given the importance of early life allergen sensitization and RV wheezing in subsequent asthma risk at school age,7 we next examined the persistence of these relationships out to 13 years. Aeroallergen sensitization at year 3 without RV wheezing in the first 3 years of life and RV wheezing without concomitant aeroallergen sensitization were each associated with increased risk of asthma at age 6, 8, 11, and 13 years (OR, 3.9 and 7.8, 2.9 and 8.1, 2.9 and 6.2, 2.7 and 3, respectively) (Fig 3). Children with both RV wheezing and aeroallergen sensitization by age 3 years had the highest incidence of subsequent asthma at year 6, 8, 11, and 13 years (OR, 45, 20, 14, and 7.9) (Fig 3).
Discussion
In this prospective birth cohort study, we sought to extend our previous findings regarding the roles of viral infections, overall aeroallergen sensitization, as well as type and timing of sensitization, and their associations with asthma at 13 years. The most striking findings in this study are that the timing of sensitization to aeroallergens in early life has a strong influence on asthma risk to adolescence, and RV, but not RSV, outpatient wheezing illnesses during the first 3 years of life increase the risk of asthma to at least adolescence.
We previously reported that wheezing illnesses in the first 3 years of life with RSV were associated with an OR of 2.6 for year 6 asthma.7 The Tucson Children's Respiratory Study group evaluated the association between RSV during childhood out to age 13 years and found that although antecedent RSV lower respiratory tract illnesses were significantly associated with frequent wheezing at age 6 years, this effect dissipated over time and was no longer significant at age 13 years.8 In contrast, the prospective data reported by Sigurs et al9, 10, 26 demonstrated that RSV bronchiolitis in early life, severe enough to require hospitalization, was a risk factor for asthma not only at age 7 years but at age 13 and 18 years as well. Because these data would suggest that the impact of RSV lower respiratory tract illnesses in early life on future asthma risk is greatest during early school age, we sought to determine whether similar developmental relationships were also true for RV wheezing illnesses in early life. Using a model to adjust for early life viral wheezing illnesses with RV, RSV, parainfluenza virus types 1 to 4, influenza types A and B, coronaviruses, metapneumoviruses, adenovirus, and enteroviruses, we noted a persistent significant association with RV wheezing illnesses in early life and asthma at all ages evaluated out to 13 years. The OR for a diagnosis of asthma at 13 years following a wheezing viral illness with RV in the first 3 years of life was 3.3. In contrast, we confirmed the Tucson findings that the impact of RSV on subsequent asthma risk lessened with time.
There are design differences between the COAST cohort and those enrolled in the Tucson Respiratory Study and by Sigurs et al that may account for the observed differences in results. First, similar to COAST, the RSV lower respiratory tract illnesses in the Tucson cohort resulted primarily in outpatient-managed illnesses. In contrast, the children experiencing RSV-related illnesses in the Sigurs et al study were diagnosed as having bronchiolitis that was clinically severe enough to require hospitalization. In addition, the presence or absence of RV illnesses in both studies was not evaluated because of technological limitations present at that time.
The influence of various demographic and environmental exposures differed during the first 13 years of life for the COAST participants. At age 6 years, having a dog in the house in early life tended to reduce asthma risk but was no longer a factor at age 13 years. In contrast, the opposite trends were seen when a cat was present in the home during that same time period. Having an older sibling in the home increased asthma risk at age 6 years but not at age 13 years.
Developmental patterns of allergic sensitization and their relationship to subsequent asthma risk were also assessed. Using cutoff points of ages 1 and 5 years revealed that children who were sensitized to 1 or more aeroallergens before age 1 year had the highest rate (65%) of asthma at age 13 years; if aeroallergen sensitization developed between ages 1 and 5 years, asthma risk was also increased at 40%. Interestingly, if sensitization developed in children after age 5 years (46% of the cohort), their asthma risk was no different from that of children who never developed sensitization. These patterns indicate that the timing of sensitization contributes significantly to future asthma risk during early childhood.
The strengths of this study are the prospective design and longitudinal follow-up with high-retention rates. In addition, the children are carefully characterized with respect to allergy and asthma phenotypes. One limitation to this study is that an enrollment criterion included only patients at higher risk for the development of asthma based on parental history of atopy and/or asthma. Furthermore, the COAST cohort is comprised primarily of nonminority, suburban children, and the role of viral infections and sensitization may differ in other populations.
It is of interest that both allergic sensitization and RV-induced wheezing illnesses in early life increase asthma risk, and the presence of both is additive. Although the precise mechanisms responsible are still under investigation, laboratory observations have shed some light on possible explanations.27, 28 First, the number of high-affinity receptors for IgE on the surface of antigen-presenting cells is directly proportional to circulating levels of IgE antibody. Second, increased numbers of these receptors is associated with a decreased IFN response following the exposure of these cells to viruses such as influenza and rhinovirus. Third, when these receptors are cross-linked as occurs in vivo following allergen exposure, the release of IFNs is reduced even further. Thus, the development of allergic sensitization could result in a decreased innate immune response to respiratory viruses that could generate greater degrees of airway inflammation and the potential for airway remodeling and loss of lung function over time. Indeed, the number and severity of virus-associated wheezing exacerbations in early childhood has been associated with reduced lung function during childhood.29 These data clearly link early life wheezing illnesses (predominantly with RV) and aeroallergen sensitization and raise the question of whether early life RV wheezing illnesses cause asthma in childhood or do they identify a cohort of children predisposed to asthma. In addition, it could be asked whether allergic sensitization is causing asthma or, as listed above for RV, is identifying a group of children at risk for asthma. Regardless, the current findings extend our previous observations by demonstrating that these events are not only important in early childhood, but increase asthma risk at least until the onset of adolescence. If primary prevention for asthma is possible, treatments aimed at attenuating or abrogating specific IgE antibody production and viral illnesses appear to be prime targets.
Clinical Implications.
Outpatient viral wheezing illnesses with RV and aeroallergen sensitization during the first 3 years of life increase asthma risk not only at school age but during adolescence as well.
Acknowledgments
We thank the COAST participants, their families, and the COAST staff for their ongoing contributions to the study of childhood asthma.
Footnotes
This study was supported by the National Institutes of Health, National Heart, Lung, and Blood Institute (grant no. PO1 HL70381) and the National Center for Advancing Translational Sciences (grant no. UL1TR000427).
Disclosure of potential conflict of interest: F. J. Rubner has received grants from the National Institutes of Health ([NIH] National Heart, Lung, and Blood Institute grant no. PO1 HL70381 and National Center for Advancing Translational Sciences grant no. ULI TR000427). D. J. Jackson has received a grant from the National Heart, Lung, and Blood Institute and has consultant arrangements with Vectura. M. D. Evans and R. E. Gangnon have received a grant from the NIH. J. E. Gern has received grants from the NIH and GlaxoSmithKline, has consultant arrangements with Genentech, Amgen, Novartis, PREP Biopharm Inc, Janssen, Regeneron, and GlaxoSmithKline; has received payment for development of educational presentations from Boehringer Ingelheim; and has received travel support from Boehringer Ingelheim. R. F. Lemanske, Jr has received travel support from the American Academy of Allergy, Asthma & Immunology; is employed by the University of Wisconsin; has received grants from the National Heart, Lung, and Blood Institute and Pharmaxis; has received personal fees from Merck, Sepracor, SA Boney and Associates, GlaxoSmithKline, American Institute of Research, Genentech, Double Helix Development, Boerhinger Ingelheim, Michigan Public Health, Allegheny General Hospital, AAP, West Allegheny Health, California Chapter 4, Colorado Allergy Society, Pennsylvania Allergy Society, Howard Pilgrim Health, California Society of Allergy, NY Allergy Society, World Allergy Organization, Asia Pacific Association of Pediatric Allergy, Respirology and Immunology, Western Society of Allergy, Asthma, and Immunology, the American Academy of Allergy, Asthma & Immunology, Elsevier, UpToDate, Kuwait Allergy Society, Lurie Children's Hospital, Boston Children's Hospital, Health Star Communications, LA Children's Hospital, Northwestern University, and the Asthma and Allergy Foundation of America - Alaska Chapter. The rest of the authors declare that they have no relevant conflicts of interest.
References
- 1.Jackson D.J., Lemanske R.F., Jr. The role of respiratory infections in childhood asthma inception. Immunol Allergy Clin North Am. 2010;30:513–522. doi: 10.1016/j.iac.2010.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gern J.E., Rosenthal L.A., Sorkness R.L., Lemanske R.F., Jr. Effects of viral respiratory infections on lung development and childhood asthma. J Allergy Clin Immunol. 2005;115:668–674. doi: 10.1016/j.jaci.2005.01.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lemanske R.F., Jr. Viral infections and asthma inception. J Allergy Clin Immunol. 2004;114:1023–1026. doi: 10.1016/j.jaci.2004.08.031. [DOI] [PubMed] [Google Scholar]
- 4.Sly P.D., Boner A.L., Bjorksten B., Bush A., Custovic A., Eigenmann P.A. Early identification of atopy in the prediction of persistent asthma in children. Lancet. 2008;372:1100–1106. doi: 10.1016/S0140-6736(08)61451-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Taussig L.M., Wright A.L., Holberg C.J., Halonen M., Morgan W.J., Martinez F.D. Tucson children's respiratory study: 1980 to present. J Allergy Clin Immunol. 2003;111:661–675. doi: 10.1067/mai.2003.162. [DOI] [PubMed] [Google Scholar]
- 6.Bisgaard H., Bonnelykke K. Long-term studies of the natural history of asthma in childhood. J Allergy Clin Immunol. 2010;126:187–197. doi: 10.1016/j.jaci.2010.07.011. [DOI] [PubMed] [Google Scholar]
- 7.Jackson D.J., Gangnon R.E., Evans M.D., Roberg K.A., Anderson E.L., Pappas T.E. Wheezing rhinovirus illnesses in early life predict asthma development in high-risk children. Am J Respir Crit Care Med. 2008;178:667–672. doi: 10.1164/rccm.200802-309OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Stein R.T., Sherrill D., Morgan W.J., Holberg C.J., Halonen M., Taussig L.M. Respiratory syncytial virus in early life and risk of wheeze and allergy by age 13 years. Lancet. 1999;354:541–545. doi: 10.1016/S0140-6736(98)10321-5. [DOI] [PubMed] [Google Scholar]
- 9.Sigurs N., Aljassim F., Kjellman B., Robinson P.D., Sigurbergsson F., Bjarnason R. Asthma and allergy patterns over 18 years after severe RSV bronchiolitis in the first year of life. Thorax. 2010;65:1045–1052. doi: 10.1136/thx.2009.121582. [DOI] [PubMed] [Google Scholar]
- 10.Sigurs N., Bjarnason R., Sigurbergsson F., Kjellman B. Respiratory syncytial virus bronchiolitis in infancy is an important risk factor for asthma and allergy at age 7. Am J Respir Crit Care Med. 2000;161:1501–1507. doi: 10.1164/ajrccm.161.5.9906076. [DOI] [PubMed] [Google Scholar]
- 11.Ireland D.C., Kent J., Nicholson K.G. Improved detection of rhinoviruses in nasal and throat swabs by seminested RT-PCR. J Med Virol. 1993;40:96–101. doi: 10.1002/jmv.1890400204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lemanske R.F., Jr., Jackson D.J., Gangnon R.E., Evans M.D., Li Z., Shult P.A. Rhinovirus illnesses during infancy predict subsequent childhood wheezing. J Allergy Clin Immunol. 2005;116:571–577. doi: 10.1016/j.jaci.2005.06.024. [DOI] [PubMed] [Google Scholar]
- 13.Kusel M.M., de Klerk N.H., Kebadze T., Vohma V., Holt P.G., Johnston S.L. Early-life respiratory viral infections, atopic sensitization, and risk of subsequent development of persistent asthma. J Allergy Clin Immunol. 2007;119:1105–1110. doi: 10.1016/j.jaci.2006.12.669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jackson D.J., Evans M.D., Gangnon R.E., Tisler C.J., Pappas T.E., Lee W.M. Evidence for a causal relationship between allergic sensitization and rhinovirus wheezing in early life. Am J Respir Crit Care Med. 2012;185:281–285. doi: 10.1164/rccm.201104-0660OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Caliskan M., Bochkov Y.A., Kreiner-Moller E., Bonnelykke K., Stein M.M., Du G. Rhinovirus wheezing illness and genetic risk of childhood onset asthma. N Engl J Med. 2013;368:1398–1407. doi: 10.1056/NEJMoa1211592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Stoltz D.J., Jackson D.J., Evans M.D., Gangnon R.E., Tisler C.J., Gern J.E. Specific patterns of allergic sensitization in early childhood and asthma & rhinitis risk. Clin Exp Allergy. 2013;43:233–241. doi: 10.1111/cea.12050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Simpson A., Tan V.Y., Winn J., Svensen M., Bishop C.M., Heckerman D.E. Beyond atopy: multiple patterns of sensitization in relation to asthma in a birth cohort study. Am J Respir Crit Care Med. 2010;181:1200–1206. doi: 10.1164/rccm.200907-1101OC. [DOI] [PubMed] [Google Scholar]
- 18.Lemanske R.F., Jr. The childhood origins of asthma (COAST) study. Pediatr Allergy Immunol. 2002;13:38–43. doi: 10.1034/j.1399-3038.13.s.15.8.x. [DOI] [PubMed] [Google Scholar]
- 19.Gern J.E., Martin M.S., Anklam K.A., Shen K., Roberg K.A., Carlson-Dakes K.T. Relationships among specific viral pathogens, virus-induced interleukin-8, and respiratory symptoms in infancy. Pediatr Allergy Immunol. 2002;13:386–393. doi: 10.1034/j.1399-3038.2002.01093.x. [DOI] [PubMed] [Google Scholar]
- 20.Neaville W.A., Tisler C., Bhattacharya A., Anklam K., Gilbertson-White S., Hamilton R. Developmental cytokine response profiles and the clinical and immunologic expression of atopy during the first year of life. J Allergy Clin Immunol. 2003;112:740–746. doi: 10.1016/s0091-6749(03)01868-2. [DOI] [PubMed] [Google Scholar]
- 21.Jartti T., Lee W.M., Pappas T., Evans M., Lemanske R.F., Jr., Gern J.E. Serial viral infections in infants with recurrent respiratory illnesses. Eur Respir J. 2008;32:314–320. doi: 10.1183/09031936.00161907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lee W.M., Grindle K., Pappas T., Marshall D.J., Moser M.J., Beaty E.L. High-throughput, sensitive, and accurate multiplex PCR-microsphere flow cytometry system for large-scale comprehensive detection of respiratory viruses. J Clin Microbiol. 2007;45:2626–2634. doi: 10.1128/JCM.02501-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Copenhaver C.C., Gern J.E., Li Z., Shult P.A., Rosenthal L.A., Mikus L.D. Cytokine response patterns, exposure to viruses, and respiratory infections in the first year of life. Am J Respir Crit Care Med. 2004;170:175–180. doi: 10.1164/rccm.200312-1647OC. [DOI] [PubMed] [Google Scholar]
- 24.van Buuren S. Chapman & Hall/CRC; Boca Raton, FL: 2012. Flexible imputation of missing data. [Google Scholar]
- 25.Rubin D.B. Wiley & Sons; New York: 1987. Multiple imputation for nonresponse in surveys. [Google Scholar]
- 26.Sigurs N., Gustafsson P.M., Bjarnason R., Lundberg F., Schmidt S., Sigurbergsson F. Severe respiratory syncytial virus bronchiolitis in infancy and asthma and allergy at age 13. Am J Respir Crit Care Med. 2005;171:137–141. doi: 10.1164/rccm.200406-730OC. [DOI] [PubMed] [Google Scholar]
- 27.Durrani S.R., Montville D.J., Pratt A.S., Sahu S., Devries M.K., Rajamanickam V. Innate immune responses to rhinovirus are reduced by the high-affinity IgE receptor in allergic asthmatic children. J Allergy Clin Immunol. 2012;130:489–495. doi: 10.1016/j.jaci.2012.05.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gill M.A., Bajwa G., George T.A., Dong C.C., Dougherty I.I., Jiang N. Counterregulation between the FcepsilonRI pathway and antiviral responses in human plasmacytoid dendritic cells. J Immunol. 2010;184:5999–6006. doi: 10.4049/jimmunol.0901194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.O’Brian A.L., Lemanske R.F., Jr., Evans M.D., Gangnon R.E., Gern J.E., Jackson D.J. Recurrent severe exacerbations in early life and reduced lung function at school age. J Allergy Clin Immunol. 2012;129:1162–1164. doi: 10.1016/j.jaci.2011.11.046. [DOI] [PMC free article] [PubMed] [Google Scholar]