Abstract
There are few studies on the obstetrical consequences of dengue fever during pregnancy. To determine whether dengue epidemics were associated with an increase in adverse obstetrical outcomes, a semi-ecological study combining individual data on obstetrical events from the perinatal registry and aggregated exposure data from the epidemiologic surveillance of dengue was conducted in Cayenne French Guiana between 2004 and 2007. After adjustment for individual risk factors, this semi-ecological analysis showed that an epidemic level of dengue transmission during the first trimester was associated with an increased risk of postpartum hemorrhage and preterm birth. The associated risks seemed to depend on the epidemic level. Despite its limitations, the present study suggests that dengue in the first trimester may be related to preterm birth and to postpartum bleeding, thus leading to specific hypotheses that should be tested in prospective studies.
Keywords: Dengue; Dengue Virus; Premature Birth; Prevalence; Epidemics; Female; French Guiana; Humans; Postpartum Hemorrhage; Pregnancy; Pregnancy Outcome; Pregnancy Trimester, First
INTRODUCTION
Dengue is an increasing problem in the tropical world. Every year there are between 50 and 100 million cases of dengue fever and 500,000 cases of dengue hemorrhagic fever worldwide 1. Given the incidence during dengue epidemics, a significant number of pregnant women must also get infected with dengue. However, there are few studies on the obstetrical consequences of dengue during pregnancy. There have been reports of severe complications for both the mother and foetus of symptomatic dengue in case series in French Guiana 2,3, but, a recent systematic review of the published studies was inconclusive for the adverse pregnancy outcomes 4. Most of the available studies are case reports or case series with only few comparative studies. Given the lack of data on this question and its potential public health importance, it is crucial to conduct further studies. We aimed to test the hypothesis that dengue has adverse consequences using a combination of two sources: aggregated exposure data from the epidemiologic surveillance of confirmed dengue from the Institut Pasteur, and individual obstetrical data from the Perinatal Registry in French Guiana, which contains exhaustive individual data on obstetrical events 5.
MATERIALS AND METHODS
Ethics statement
Adverse maternal outcomes in French Guiana are reported in the Perinatal Registry. This registry is completely anonymous, is approved by the Commission Nationale de l’Informatique et des Libertés (CNIL) and is managed by the maternal and child care department of the Conseil Général de la Guyane. The data access was based on special permissions given by the Conseil Général de la Guyane.
Individual obstetrical data
The Perinatal Registry includes all births (living and dead) in public and private hospitals in French Guiana. Data were available for a 4-year period (January 2004 to December 2007). Inclusion criteria for births are a gestational age >22 weeks or a weight >500 grams. Each birth is registered by an obstetrician or a midwife. The anonymous data is gathered from the mother’s interview, medical files, and maternity follow up booklets, which allowed us to compile 28 different items relating to the pregnancy and delivery of the infant. Various adverse obstetrical outcomes were studied: preterm birth, low birth weight, malformations, preeclampsia, postpartum hemorrhage, and caesarean section.
Postpartum hemorrhage was defined as bleeding > 500ml for a vaginal delivery and >1000 ml for caesarean section. Preeclampsia was defined as blood pressure >160/90 mm Hg and proteinuria >0.5 g per 24h after 20 weeks since last menstrual period. A range of malformations (cardiovascular, orthopedic, neurological, multiple, and unknown) were pooled into a single variable representing all malformations. Down syndrome was excluded from this list. All adverse obstetrical outcomes were determined independently of the mother’s dengue fever status at delivery. The analysis was restricted to pregnancies for which pregnant women were living in the urban area of Cayenne consisting of the city of Cayenne and its neighboring communes (Matoury, Macouria and Rémire Montjoly). The analysis was restricted to this circumscribed area of Cayenne so that dengue cases occurred in the area where the women lived, thus genuinely reflecting an increased risk of getting dengue during the pregnancy. For each studied outcome, only pregnancies with no missing data on maternal age, gestational time, parity, gravidity and birth weight were used in the analysis.
Aggregated dengue surveillance data
For each pregnancy, aggregated data from the epidemiologic surveillance of dengue confirmed by the National Reference Center for arboviruses, (the Institut Pasteur de la Guyane (IPG)), were used. A confirmed dengue diagnosis was defined (i) during the acute phase of the disease as positive virus isolation, and/or positive viral RNA detection by reverse transcriptase-polymerase chain reaction (RT-PCR), and/or positive NS1 antigen detection, and/or positive DENV IgM detection; (ii) the diagnosis of early convalescent dengue cases was based on DENV IgM detection6. Weekly totals of confirmed cases are transmitted to the Cellule de l’InVS en Region Antilles Guyane (CIRE) for epidemiologic surveillance, alert and intervention. We used cases from a single reference laboratory so the number of confirmed cases would not be artificially inflated by the increase in the number of labs able to perform tests for dengue during the time period of the study.
Statistical methods
Univariate and multivariate logistic regression models were used to test if dengue epidemic levels during the three defined time periods were significant risk factors for various adverse maternal outcomes. To calculate the total monthly number of confirmed dengue fever cases, the month of delivery and the term at delivery (established using the last menstrual period) were used to determine the month of conception. To investigate potential biologic mechanisms of dengue fever on pregnancy outcomes, we constructed three periods of exposure for dengue, the first one beginning from the estimated month of conception and covering the next two months (first trimester). The second one included the three months following the first trimester (second trimester). The third one included the last three months before delivery, regardless of the trimester of pregnancy. Since preterm births have an incomplete or absent third trimester, the associated total number of dengue cases in the third trimester would be artificially low for preterm births compared to term births. The use of the last three months of pregnancy instead of the traditional third trimester was used in order to avoid an artificial protective association between dengue in the third trimester and adverse outcomes related to gestational duration. Therefore, the last three months of pregnancy cannot be interpreted in this study to represent the third trimester, as it may well be a combination of data from the second and third trimester. The total number of dengue cases per period were categorized (<20, 20–60 and >60) and transformed into indicator variables. This categorization roughly corresponded to the level of cases observed during a non epidemic period, a moderate epidemic period or a high epidemic period in French Guiana during this time period. To control for potential confounding, adjustment variables were also included. The adjustment variables were the woman’s age, ethnic origin, gravidity, parity, twin pregnancy, having had a medical follow up, the number of ultrasound scans, and having had various pathologies during pregnancy, including chronic health conditions of the mother, as well as pregnancy specific medical conditions. Low birth weight (<2,500 g) and preterm birth (gestation < 37 weeks) were also included in the model depending on the outcome under investigation. Statistical significance was set at p<0.05. All statistical analyses were conducted using R 2.12.0 and the package Epicalc 7.
RESULTS
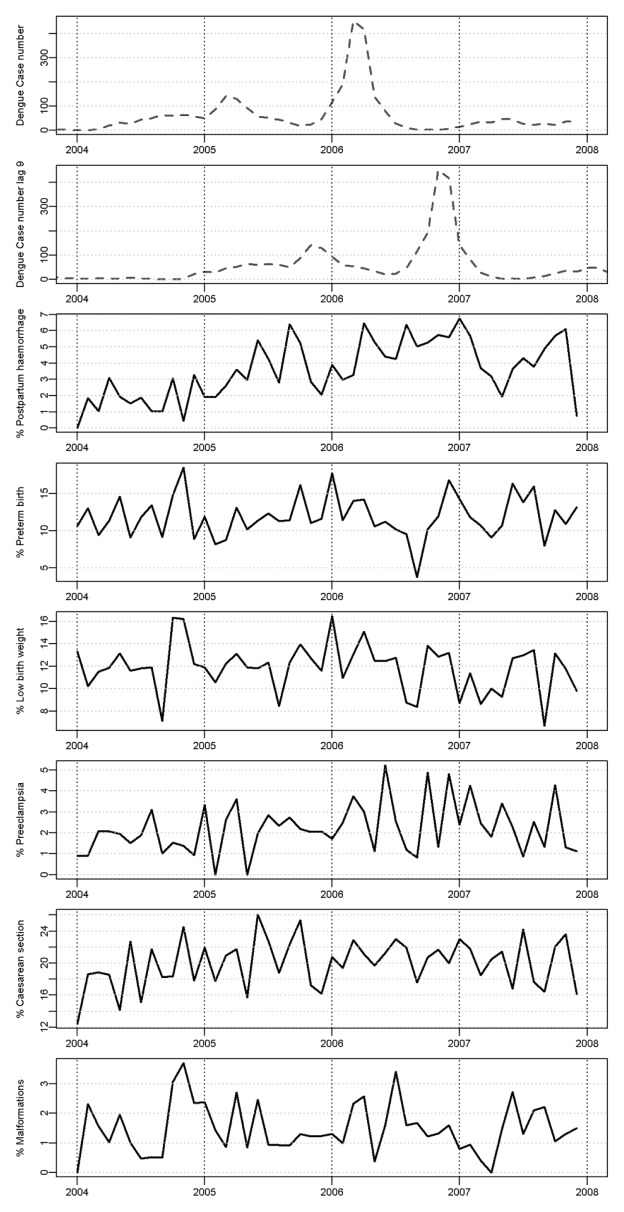
During the 2004–2007 period, 24,624 pregnancies occurred throughout French Guiana of which 11,004 (44.7 %) were among women living in the urban area of Cayenne. Among these pregnancies, 10,771 (97.9 %) had no missing data for maternal age, gestational time, parity, gravidity and birth weight. Preterm birth, low birth weight, malformations, preeclampsia, postpartum hemorrhage, and caesarean section were observed in 1,284 (11.9%), 1,277 (11.9%), 160 (1.4%), 240 (2.2 %), 393 (3.6 %) and 2,154 (20.0 %) pregnancies, respectively. A vast majority of the preterm births that occurred during the study period were born between 34–36 weeks of gestation (63.6 %). Only 4.1 % were classified as extreme preterm births (gestation < 25 weeks). Figure 1 shows the monthly numbers of confirmed dengue cases and the monthly incidences of studied outcomes in the Cayenne area during 2004–2007.
Figure 1.
Monthly confirmed dengue cases (lag 0 and 9 months) and incidence of the 6 studied adverse obstetrical outcomes in the urban area of Cayenne between 2004 and 2007.
Table 1 shows that after adjusting for potential confounders, dengue epidemic levels were statistically significant risk factors during the first trimester of pregnancy for both postpartum hemorrhage and preterm birth. For postpartum hemorrhage, a number of confirmed cases of dengue ≥20 during the first trimester of pregnancy corresponded to an increased adjusted odds ratio ranging from 2.39 [1.46, 3.93] to 3.06 [2.04, 4.59], depending on the level of the dengue epidemic. Similarly confirmed dengue cases > 60 during the first trimester was significantly associated with preterm birth, adjusted odds ratio of 1.31 [1.06, 1.63]. Furthermore, for postpartum hemorrhage, a number of confirmed cases of dengue between 20 and 60 during the second trimester of pregnancy was also significantly associated with an increased adjusted odds ratio, adjusted odds ratio of 1.65 [1.01, 2.68], but not with a number of confirmed cases of dengue >60, adjusted odds ratio of 0.91 [0.57, 1.46]. No statistically significant association of dengue epidemic levels was found for the other studied adverse obstetrical outcomes (low birth weight, malformations, preeclampsia, and caesarean section).
Table 1.
Univariate and multivariate $ logistic regressions predicting impact of dengue epidemics on 6 adverse obstetrical outcomes for 107776 pregnant women residing in the Cayenne area, French Guiana. 2004–2007
| Outcomes | Pregnancy period | N confirmed dengue cases | N events (%) | Crude OR [95%CI] | Adj. OR [95%CI] | P(Wald’s test) |
|---|---|---|---|---|---|---|
|
| ||||||
| Preterm birth | First trimester | <20 | 246 (10.5) | 1 | 1 | - |
| 20–60 | 112 (10.6) | 1.00 [0.79, 1.27] | 1.17 [0.89, 1.54] | 0.268 | ||
| >60 | 926 (12.6) | 1.22 [1.05, 1.41] | 1.31 [1.06, 1.63] | 0.014* | ||
|
| ||||||
| Second trimester | <20 | 198 (11.0) | 1 | 1 | - | |
| 20–60 | 133 (12.2) | 1.12 [0.89, 1.42] | 1.12 [0.84, 1.5] | 0.423 | ||
| >60 | 953 (12.1) | 1.11 [0.95, 1.31] | 0.96 [0.71, 1.3] | 0.796 | ||
|
| ||||||
| Last 3 months | <20 | 164 (12.1) | 1 | 1 | - | |
| 20–60 | 122 (11.1) | 0.90 [0.70, 1.16] | 0.90 [0.69, 1.17] | 0.442 | ||
| >60 | 998 (12.0) | 0.99 [0.83, 1.18] | 0.95 [0.75, 1.22] | 0.702 | ||
|
| ||||||
| Postpartum hemorrhage | First trimester | <20 | 45 (1.9) | 1 | 1 | |
| 20–60 | 36 (3.4) | 1.78 [1.14, 2.78] | 2.39 [1.46, 3.93] | < 0.001* | ||
| >60 | 312 (4.2) | 2.25 [1.64, 3.08] | 3.06 [2.04, 4.59] | < 0.001* | ||
|
| ||||||
| Second trimester | <20 | 58 (3.2) | 1 | 1 | ||
| 20–60 | 37 (3.4) | 1.05 [0.69, 1.60] | 1.65 [1.01, 2.68] | 0.044* | ||
| >60 | 298 (3.8) | 1.18 [0.89, 1.57] | 0.91 [0.57, 1.46] | 0.689 | ||
|
| ||||||
| Last 3 months | <20 | 46 (3.4) | 1 | 1 | - | |
| 20–60 | 51 (4.6) | 1.38 [0.92, 2.07] | 1.47 [0.95, 2.28] | 0.086 | ||
| >60 | 296 (3.6) | 1.05 [0.76, 1.44] | 0.97 [0.65, 1.45] | 0.890 | ||
|
| ||||||
| Low birth weight | First trimester | <20 | 243 (10,4) | 1 | 1 | - |
| 20–60 | 104 (9,8) | 0.93 [0.73, 1.19] | 1.02 [0.74, 1.39] | 0.907 | ||
| >60 | 930 (12,6) | 1.24 [1.07, 1.44] | 1.19 [0.97, 1.47] | 0.099 | ||
|
| ||||||
| Second trimester | <20 | 94 (12.6) | 1 | 1 | - | |
| 20–60 | 76 (10.6) | 0.82 [0.60, 1.13] | 0.89 [0.59, 1.35] | 0.596 | ||
| >60 | 1107 (11.9) | 0.94 [0.75, 1.17] | 0.84 [0.62, 1.14] | 0.270 | ||
|
| ||||||
| Last 3 months | <20 | 170 (12,6) | 1 | 1 | - | |
| 20–60 | 116 (10,5) | 0.82 [0.64, 1.05] | 0.84 [0.6, 1.17] | 0.297 | ||
| >60 | 991 (11,9) | 0.94 [0.79, 1.12] | 0.9 [0.71, 1.14] | 0.395 | ||
|
| ||||||
| Preeclampsia | First trimester | <20 | 47 (2.0) | 1 | 1 | - |
| 20–60 | 14 (1.3) | 0.65 [0.36, 1.18] | 0.74 [0.39, 1.41] | 0.367 | ||
| >60 | 179 (2.4) | 1.21 [0.88, 1.68] | 1.22 [0.83, 1.80] | 0.304 | ||
|
| ||||||
| Second trimester | <20 | 22 (1.9) | 1 | 1 | - | |
| 20–60 | 12 (1.7) | 0.56 [0.28, 1.14] | 0.57 [0.27, 1.22] | 0.148 | ||
| >60 | 206 (2.2) | 0.75 [0.48, 1.17] | 0.79 [0.48, 1.29] | 0.349 | ||
|
| ||||||
| Last 3 months | <20 | 35 (2.6) | 1 | 1 | - | |
| 20–60 | 25 (2.3) | 0.87 [0.52, 1.47] | 1.06 [0.6, 1.86] | 0.842 | ||
| >60 | 180 (2.2) | 0.83 [0.58, 1.20] | 0.80 [0.53, 1.20] | 0.284 | ||
|
| ||||||
| Caesarean section | First trimester | <20 | 431 (18.5) | 1 | 1 | |
| 20–60 | 198 (18.6) | 0.65 [0.36, 1.18] | 0.74 [0.39, 1.41] | 0.367 | ||
| >60 | 1525 (20.7) | 1.21 [0.88, 1.68] | 1.22 [0.83, 1.80] | 0.304 | ||
|
| ||||||
| Second trimester | <20 | 159 (21.3) | 1 | 1 | ||
| 20–60 | 144 (20.1) | 0.56 [0.28, 1.14] | 0.57 [0.27, 1.22] | 0.148 | ||
| >60 | 1851 (19.9) | 0.75 [0.48, 1.17] | 0.79 [0.48, 1.29] | 0.349 | ||
|
| ||||||
| Last 3 months | <20 | 254 (18.8) | 1 | 1 | ||
| 20–60 | 211 (19,2) | 0.87 [0.52, 1.47] | 1.06 [0.60, 1.86] | 0.842 | ||
| >60 | 1689 (20.3) | 0.83 [0.58, 1.20] | 0.80 [0.53, 1.20] | 0.284 | ||
|
| ||||||
| Malformations | First trimester | <20 | 24 (1.0) | 1 | 1 | |
| 20–60 | 18 (1.6) | 1.66 [0.90, 3.07] | 1.66 [0.88, 3.15] | 0.118 | ||
| >60 | 118 (1.7) | 1.57 [1.01, 2.44] | 1.42 [0.88, 2.28] | 0.152 | ||
|
| ||||||
| Second trimester | <20 | 13 (1.7) | 1 | 1 | ||
| 20–60 | 14 (1.9) | 1.12 [0.52, 2.40] | 1.15 [0.52, 2.53] | 0.729 | ||
| >60 | 133 (1.4) | 0.82 [0.46, 1.45] | 0.89 [0.49, 1.62] | 0.701 | ||
|
| ||||||
| Last 3 months | <20 | 18 (1.3) | 1 | 1 | ||
| 20–60 | 14 (1.3) | 0.95 [0.47, 1.93] | 0.90 [0.43, 1.86] | 0.767 | ||
| >60 | 128 (1.5) | 1.16 [0.70, 1.90] | 1.03 [0.61, 1.75] | 0.900 | ||
P-value < 0.05
The adjustment variables in the multivariate models were the woman’s age, ethnic origin, gravidity, parity, twin pregnancy, having had a medical follow up, the number of ultrasound scans, and having had various pathologies during pregnancy, including chronic health conditions of the mother, as well as pregnancy specific medical conditions. Low birth weight (<2500 g) and preterm birth (gestation < 37 weeks) were also used depending the outcome under investigation.
DISCUSSION
Despite its limitations, the present study is the first to examine the relation between the magnitude of dengue epidemics and a range of obstetrical outcomes using a combination of individual and ecological data. After adjusting for potential confounders, there was a statistically significant association between dengue epidemics and postpartum hemorrhage, and between dengue epidemics and preterm birth. The measure of association seemed to be in proportion with the level of the epidemic.
It is important to note that the pregnancy registry data and the confirmed dengue totals have been calculated per month. Not having exact dates for either dengue cases or delivery information results in an imperfect fit between the variables we used to represent the first and second trimesters and the last three months of pregnancy and real associated pregnancy periods. Using the total number of confirmed cases in Cayenne for 3 months as a reflection of an individual woman’s risk of getting dengue is a very coarse reflection of actual dengue cases in pregnant women. It rests on the plausible assumption that the number of pregnant women having dengue will be proportional to the overall magnitude of the dengue epidemic in the population. Since there were many deliveries (n=11,004), during these four years we feel we have adequate statistical power to detect associations. Although it is possible that the observed associations could have resulted from an information bias in the perinatal records, local experience has indicated that there is an under-reporting of adverse outcomes in the Perinatal Registry in French Guiana due to the priority given to care relative to data collection. For the purpose of this study we assumed that this under-reporting was fairly constant during the study period and did not affect the results. It could also be argued that a possible synchronous seasonal pattern of both dengue epidemics and studied adverse outcomes could also explain the observed relations 8. However, including in the final model a variable representing the month of birth did not affect results. Another explanation to the observed associations could also be that being exposed to a dengue epidemic implies having a greater probability of exposure to vector control pesticides. Thus the “toxic” environment during a dengue epidemic is also a potential hypothesis for the observed associations. Furthermore, underlying trends in the data, such as the unexpected decrease of preterm births which was observed in 2007, followed by an increase in preterm births, as well as the increased frequency of postpartum hemorrhages during most of our study period, makes the interpretation of the significant associations more complicated.
Using aggregated data leads to potential ecological biases. However, except for the “toxic” hypothesis, we were not able to identify any other potential explanation that could account for the association between dengue epidemics and postpartum hemorrhage and preterm birth. In addition, adjustment for a range of potential confounders available in the individual registry data did not affect the results. It was counter-intuitive to observe that dengue during the first trimester, and less clearly during the second trimester, was linked to postpartum hemorrhage and preterm birth. Direct placental damage in the first trimester could lead to placental anatomical and functional abnormalities leading to subsequent complications. This could suggest that the pathophysiology of postpartum bleeding and preterm birth are not solely linked to acute thrombopenia or endothelial dysfunction during dengue, but also to other mechanisms that have effects after the acute episode. It has been shown that dengue infection leads to the synthesis of anti-platelet antibodies 9,10 and anti endothelial cell antibodies 10, which can persist months after the initial dengue episode 11. It has been speculated that these autoantibodies are linked to shared epitopes between NS1, platelet and endothelium surface molecules 10. It is possible that these autoantibodies are also associated with postpartum hemorrhage and preterm birth.
In this study, no significant association (p<0.05) was seen between dengue epidemic levels and other adverse obstetrical outcomes. Previous studies have shown an association between dengue infection and low birth weight 4. We have found a trend for an association between dengue during the first semester and low birth weight, but not during the second trimester and the last 3 months of pregnancy (Table 1). However, the ecological design may have not been optimal here to detect a significant difference. Similarly, although no significant association was seen, a trend during the first trimester seemed to suggest a possible association between dengue epidemic levels and malformations. There again, the ecological design combined with the limited number of events for this adverse outcome may have not been able to detect a significant association.
Overall, the present study identified a possible relation between potential exposure to dengue epidemics in the first trimester of pregnancy and preterm birth and postpartum hemorrhage. Prospective studies on the obstetrical consequences of dengue and their pathophysiology are needed to confirm these results and improve our insufficient knowledge of this potentially important public health problem.
Acknowledgments
FUNDING: This work was supported by INSERM (Institut National de la Santé et de la Recherche Médicale) and the Cayenne General Hospital. Funders had no role in the study design; in the collection, analysis, and interpretation of data; in the writing of the report; and in the decision to submit the paper for publication.
Footnotes
CONFLICTS OF INTEREST: MH, EF, CB, AR, PB, PD, CF, GC, PB, GB, BC and MN: no conflict
ETHICAL APPROVAL: Yes
CONTRIBUTORS: MH participated in the research design, performed data analysis and interpretation, and prepared the manuscript. EF, CB, AR, PB, PD, CF, GC, PB, GB and BC helped in interpretation and participated in manuscript revision as well. MN initiated the study, participated in the research design, provided guidance on data analyses and was involved in the interpretation of data and manuscript revision. All authors read and approved the final manuscript.
References
- 1.Halstead SB. Dengue. Lancet. 2007;370:1644–1652. doi: 10.1016/S0140-6736(07)61687-0. [DOI] [PubMed] [Google Scholar]
- 2.Basurko C, Carles G, Youssef M, Guindi WEL. Maternal and fetal consequences of dengue fever during pregnancy. Eur J Obstet Gynecol Reprod Biol. 2009;147:29–32. doi: 10.1016/j.ejogrb.2009.06.028. [DOI] [PubMed] [Google Scholar]
- 3.Carles G, Peiffer H, Talarmin A. Effects of dengue fever during pregnancy in French Guiana. Clin Infect Dis. 1999;28:637–640. doi: 10.1086/515144. [DOI] [PubMed] [Google Scholar]
- 4.Pouliot SH, Xiong X, Harville E, Paz-Soldan V, Tomashek KM, et al. Maternal dengue and pregnancy outcomes: a systematic review. Obstet Gynecol Surv. 2010;65:107–118. doi: 10.1097/OGX.0b013e3181cb8fbc. [DOI] [PubMed] [Google Scholar]
- 5.Cardoso T, Carles G, Patient G, Clayette P, Tescher G, et al. Perinatal care and mortality in French Guyana. From 1992–1999. J Gynecol Obstet Biol Reprod (Paris) 2003;32:345–355. [PubMed] [Google Scholar]
- 6.Dussart P, Petit L, Labeau B, Bremand L, Leduc A, et al. Evaluation of Two New Commercial Tests for the Diagnosis of Acute Dengue Virus Infection Using NS1 Antigen Detection in Human Serum. PLoS Neglected Tropical Diseases. 2008;2:e280. doi: 10.1371/journal.pntd.0000280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.R Development Core Team. R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing; 2011. [Google Scholar]
- 8.Strand LB, Barnett AG, Tong S. The influence of season and ambient temperature on birth outcomes: a review of the epidemiological literature. Environ Res. 2011;111:451–462. doi: 10.1016/j.envres.2011.01.023. [DOI] [PubMed] [Google Scholar]
- 9.Sun D-S, King C-C, Huang H-S, Shih Y-L, Lee C-C, et al. Antiplatelet autoantibodies elicited by dengue virus non-structural protein 1 cause thrombocytopenia and mortality in mice. J Thromb Haemost. 2007;5:2291–2299. doi: 10.1111/j.1538-7836.2007.02754.x. [DOI] [PubMed] [Google Scholar]
- 10.Hung NT, Lan NT, Lin Y-S, Lin C-F, Lien LB, et al. Anti-Platelet and Anti-Endothelial Cell Autoantibodies in Vietnamese Infants and Children with Dengue Hemorrhagic Fever. American Journal of Infectious Diseases. 2008;4:41–49. doi: 10.3844/ajidsp.2008.41.49. [DOI] [Google Scholar]
- 11.Lin CF, Lei HY, Liu CC, Liu HS, Yeh TM, et al. Generation of IgM anti-platelet autoantibody in dengue patients. J Med Virol. 2001;63:143–149. [PubMed] [Google Scholar]