Abstract
Temporomandibular disorders (TMD) are among the most common maxillofacial complaints and a major cause of orofacial pain. Although, current treatments provide short- and long-term relief, alternative tissue engineering solutions are in great demand. Particularly, the development of strategies, providing long-term resolution of TMD to help patients regain normal function is a high priority. An absolute prerequisite of tissue engineering is to understand normal structure and function. The current knowledge of anatomical, mechanical, and biochemical characteristics of the temporomandibular joint (TMJ) and associated tissues will be discussed, followed by a brief description of current TMD treatments. The main focus is on recent tissue engineering developments for regenerating TMJ tissue components, with or without a scaffold. The expectation for effectively managing TMD is that tissue engineering will produce biomimetic TMJ tissues that recapitulate the normal structure and function of the TMJ.
Keywords: Temporomandibular joint disorder, Condylar fibrocartilage, Temporomandibular joint disc, Scaffold-based tissue engineering, Scaffold-free tissue engineering, TMD treatment methods
Introduction
TMJ is a synovial joint that provides for mandibular motion relative to the cranial base, and distributes the normal stresses of function(chewing and speaking), and parafunction (clenching and bruxism). It is often referred to as ginglymoarthrodial joint due to its hinging and sliding function. The TMJ connects the mandibular condyle (lower jaw) to the temporal bone of the skull. The fibrocartilaginous disc, sandwiched between mandibular condyle and the glenoid fossa-articular eminence of the temporal bone, divides the joint space into superior and inferior compartments, both of which are filled with synovial fluid (Fig. 1A).
Figure 1.
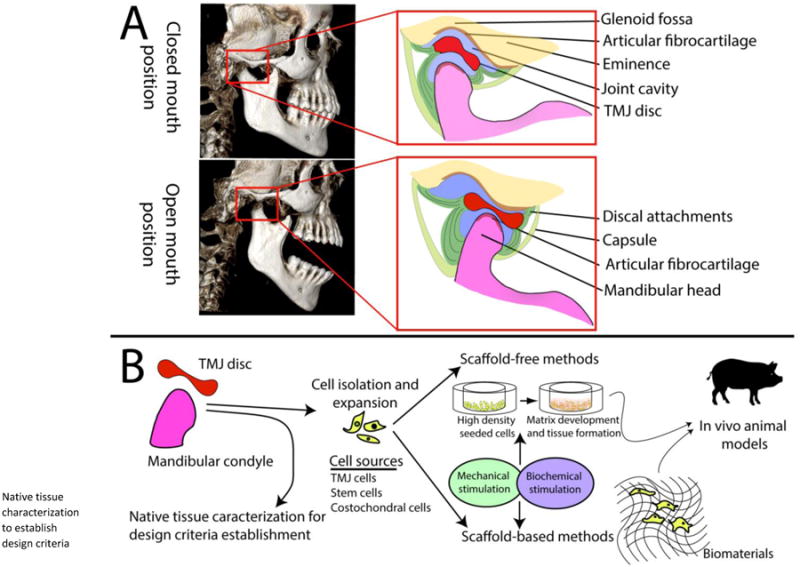
A) Cone beam CT images of the human skull with focus on the TMJ. The soft tissue of the disc is not visible on CT scans of the skull, but it is shown in the schematic. During the closed mouth position, the disc is lodged between the anterior aspect of the mandibular condyle and glenoid fossa. As the mouth opens, the disc glides anteriorly onthe articular eminence as the mandibular condyle rotates anteriorly. B) Summary of TMJ tissue engineering approaches is based on characterization of the target tissues, and identification of the cell sources. Methods of tissue engineering include mechanical and biochemical stimuli to improve the properties of neotissues (new tissues produced). The feasibility of these methods is explored in a large animal model such as a pig.
The TMJ is affected by various disorders, that are collectively referred to as temporomandibular disorder (TMD). TMD is a heterogeneous group of clinically important dysfunctions and pathologies involving the TMJ, the associated jaw muscles, or both. Symptoms of TMD include painful or pain-free joint sounds, reduced range of motion, and joint and/or muscular pain. Interestingly, these symptoms occur disproportionally between sexes, with much higher incidence in females than in males [1, 2]. Importantly, up to 70% of TMD patients suffer from an abnormal position of the TMJ disc during function, which is often referred to as internal derangement (ID) [2]. Although the pathogenesis and progression of TMD are not completely understood, degenerative osteoarthropathy is the most common outcome. TMJ degeneration is manifest by: 1. displacement, thickening, and/or perforation of the disc, 2. attenuation to complete loss of the articular fibrocartilage, 3. sclerosis and remodeling of supporting bone, and 4. periarticular osteophyte formation [3]. Studies show that 25%-60% of the US population have experienced at least one symptom of TMD, but only a small percentage of these subjects seek treatment. Importantly, treatment of TMD costs about $4 billion per annum in the US [1, 2]. The patients' ages range from 20 to 50 years old, which represents a relatively early age range for onset of osteoarthritis [4, 5].
The management options for TMD vary according to the severity of the disease. Non-invasive and minimally invasive treatments are preferred for patients in early stages of TMD while the more invasive approaches are reserved for advanced stages of the disease. Unfortunately, no treatment consistently offers permanent recovery, and many patents require repeat therapy or follow-up surgeries [6]. The lack of consistently successful treatment approaches necessitates development of novel solutions such as those based in tissue engineering. This review discusses the tissue structure and function of the TMJ relative to current TMD treatment methods. Special focus then turns to important advances in the field of tissue engineering for producing the TMJ tissue components. Specifically, scaffold-free and scaffold-based methods using cell-based approaches are discussed.
TMJ structure and function
Because of the complex loading patterns that engineered tissues will experience in the TMJ, acquisition of complete design parameters from the native tissue is critical. Particularly, TMJ disc, condyle, and condylar fibrocartilage replacements are in great demand due to these tissues' poor regenerative capacity and high rate of involvement in TMD. In response, several studies characterizing the properties of these components have been performed [7, 8]. Although glenoid fossa and articular eminence are also involved in TMD, they have not been fully characterized. In the following section, structural characteristics of the TMJ tissues are summarized.
Unlike most synovial joints, the articular surface of which is covered with hyaline cartilage, the TMJ articulation is covered by a layer of fibrous tissue resembling periosteum, also referred to as a fibrous zone [9]. Continuous and underlying this layer there is fibrocartilage layer that can be in turn schematically subdivided into proliferative and hypertrophic zones [9]. Although the biochemical composition of ECM in each of the articulating layer zones has been characterized in multiple species and man, there is an ongoing debate on the embryonic origin of mandibular articulating surface. One theory is supporting periosteal origin [10], while other claims blastemal origin [11]. The fibrocartilage thickness is highly dependent on the location, age, and loading conditions. The maximum thickness of condylar fibrocartilage in humans reaches 0.48 mm [12, 13].
The biochemical properties of the condylar fibrocartilage vary with respect to zones. It was shown that in pigs the primary extracellular matrix (ECM) component is collagen mixed with a small amount of proteoglycans [14]. The superficial fibrous zone is rich in type I collagen while collagen type II is minimally present, unlike in hyaline articular cartilage. Inferior to the fibrous zone is the proliferative zone [15] that functions as a cell reservoir [16]. Similar to the fibrous zone, collagen type I is more abundant in this layer [16]. The collagen fiber orientation in these two zones was determined to be anisotropic, with primarily anteroposterior alignment of the fibers [15, 17]. The predominate proteoglycan type in the fibrous and proliferative zones was found to be a versican-like chondroitin-sulfate [14, 18]. The mature and hypertrophic zones are populated by chondrocytes, and the ECM here is primarily comprised of collagen type II, but collagen types I and X are also present [18]. The predominant proteoglycan type in these two layers is aggrecan. Compared to hyaline articular cartilage, mandibular condylar fibrocartilage contains much less glycosaminoglycan (GAG) by weight. In the rat, for example, GAG per wet weight of the mandibular condyle was significantly lower than in the cartilage of the femoral head [19]. GAG and collagen contents play important roles in compressive and tensile mechanical properties [20]. This is an important concept to mimic the properties of the native tissue, with respect to tissue engineering.
The mechanical properties of condylar fibrocartilage have been characterized by a variety of methods for various species [18]. Assays include creep indentation, atomic force microscopy, and uniaxial tension. In most studies, the highest compressive properties (1.4-2.3 MPa) were detected in the anterior or anteromedial regions of the condyle as compared to other regions. In tension, the Young's modulus values were higher in the anteroposterior (9.0-21.4 MPa) than in mediolateraldirection (6.6-11.3 MPa) [15].
The TMJ disc is attached along its periphery to the condyleand temporal bone via fibrous connective tissue. The anterior portion of the disc is attached to the anterior aspect of the condylar process inferiorly and to the articular eminence superiorly by blending with the joint capsule [21]. The posterior portion of the disc transitions into fibrous tissue referred to as the “bilaminar zone,” which, as the name suggests, splits and attaches superiorly to the temporal bone and inferiorly to the posterior aspect of the condylar neck (Fig. 1A). Laterally and medially, the disc attachments blend with the joint capsule near its attachment to the condylar process [22]. The biconcave, elliptical shape of the disc and its motion reflect the function of this structure, which is to compensate for the incongruence of the articulating surfaces and to transmit forces between them. The dimensions of the disc in humans are approximately 23 mm mediolaterally and 14 mm anteroposteriorly [8]. The periphery of the disc is thicker than the center. The cells of the TMJ disc are distinctly different from hyaline cartilage and composed of heterogeneously distributed populations of fibroblasts, fibrocytes, and fibrochondrocytes [23]. The central portion of the disc is avascular, but its periphery and attachments are vascularized [24].
The TMJ disc is composed primarily of type I collagen with small percentages of collagen II, III, VI, IX, and XII [25]. There is a significant degree of anisotropy present in the disc [26]. In the central portion, the fibers are predominantly oriented anteroposteriorly, while a more circumferential arrangement is observed in the band region. Vertical and intersectional patterns are also present occasionally [26]. Importantly, in both humans and animals the collagen fibers of the disc are crimped, which contributes to the mechanical properties of the tissue, especially under tension [27]. GAG comprises a much smaller fraction of the TMJ disc ECM. The GAG fraction ranges from 1 to 10% by dry weight, and dermatan sulfate and chondroitin sulfate are its dominant components [28]. This amount of GAG is lower than what is reported of hyaline articular cartilage [29].
The biomechanical properties of the TMJ disc are consistent with models for viscoelastic materials, meaning that its response to loading depends on its strain history. TMJ disc mechanical properties vary with respect to species and region. Several studies have focused on the disc's mechanical properties and behavior under tension and compression using experimental and modeling methods [30-32]. Correlations between disc mechanical and biochemical properties have been reported. For instance, human disc tensile properties in anteroposterior and mediolateral directions reflect the anisotropy of the collagen fiber arrangement; the tensile strength of the central region is 13.8MPa and 4.2MPa in anteroposterior and mediolateral directions, respectively [8]. Mimicking this unique structure is one of the challenges for engineering the TMJ disc.
TMJ disease, current treatments, and limitations
The etiology of TMD is multi-factorial and complex [33], and the pathogenesis of the disease is still poorly understood. The factors that contribute to TMD development and progression are best categorized as predisposing, initiating, and perpetuating [34, 35]. These factors may include trauma [36], malocclusion [37], estrogen influence [38], bruxism [39], genetic variations [40], and even psychological aspects [41]. Whatever the combination of factors contributing to the disease, they all lead to mechanical overload of the joint components, which eventually results in development of degenerative changes and osteoarthrosis [42, 43•]. The best treatment strategy, therefore, would be identifying and eliminating the primary factor of TMD. Given that this option is seldom available, other treatment options have been devised. In the following sections, the most common therapeutic options for TMD are discussed.
The current management options of TMD include non-invasive, minimally invasive, and invasive techniques (Table 1). More often than not, a combination of these treatments is applied in an attempt to address both the potential cause and the resulting symptoms of TMD. Examples of non-invasive treatments are occlusal splints (orthotics) [44], medications, physical therapy, and acupuncture. Although the use of occlusal orthotics has been practiced since the 18th century, and they are still widely used, the effectiveness of this method remains questionable [45, 46]. Depending on the severity of TMD, various classes of oral or topical medications can be prescribed for TMD patients to relieve pain and discomfort [47] (Table 1). While oral NSAIDs (nonsteroidal anti-inflammatory drugs) are the most common, alternative options such as muscle relaxants, anxiolytics [48], opioids, and even topical medications are available [49]. Despite the ease of use and popularity of these medications, the numbers of studies that support or refute the long-term effectiveness of pharmacologic interventions are similar [50].
Table 1.
Available clinical treatments for TMD patients. Advantages and disadvantages of these treatments need to be studied and elucidated further.
| Category | Method | Main advantage/disadvantage |
|---|---|---|
| Non-invasive | Occlusal splints | Stabilizes the joint and minimizes pain; not a long-term solution |
| Medication | Reduces inflammation and pain; negative side effects such as nausea, sedation, and psychomotor impairment | |
| Physical therapy | Increases mobility and reduces pain; should be applied with other treatments | |
| Minimally invasive | Intra-articular injection | Lasts longer than non-invasive methods, increases mobility, and decreases pain; needs to be performed in early stages of degeneration |
| Arthrocentesis and arthroscopy | Deemed to be safe and quick; more research is needed to identify long-term effects | |
| Invasive | Discectomy | Improves mandibular range of motion and reduces orofacial pain; does not prevent joint degradation |
| Arthroplasty | Successful results, especially in patients with unaffected condyle; surgical complications such as nerve injury | |
| Total joint replacement | Only treatment method for severely damaged joints with end-stage disease; secondary surgery is often necessary |
Minimally invasive treatment options include intra-articular injections, arthrocentesis, and arthroscopy. Though these options can be quite invasive, especially arthroscopy, they are still classified as minimally invasive. Via intra-articular injections, medications such as corticosteroids alone or in combination with high molecular weight sodium hyaluronate can be administered directly into the joint in either or both joint compartments [49, 51]. The advantage of these techniques is in the ability to get direct access to the joint space. Although significant improvement in TMD symptoms was reported by several studies, repeated injections and or arthroscopies are not recommended, and thus, long term efficacy of these treatments remains questionable [51]. In arthrocentesis, usually two needles are inserted into the superior TMJ space followed by irrigation of the space with saline. The objective of arthrocentesis is to wash out the inflammatory mediators responsible for inflammation in the joint. It is also thought that irrigation and lavage under high pressures can potentially remove adhesions to improve joint mobility. In some reports morphine was injected into joint space following saline irrigation [52]. Arthrocentesis was reported to be highly successful for the treatment of TMD over both the long and short terms [52, 53] with an 83.5% treatment success rate [54]. Arthroscopy is similar but with the ability to visualize and manipulate intra-articular space. It can now be performed with extremely small-diameter endoscopes, and permits direct removal and treatment of pathologic intra-articular tissues [55]. With proper patient selection, the success rate for arthroscopy can be 85–90% [55, 56].
Invasive treatments include open joint surgery (arthrotomy), represented by either modification of joint components or complete replacement of the entire joint with autogenous or alloplastic prostheses (arthroplasty). Examples of cases where surgery may be the only option include ankylosis, neoplasia, chronic or recurrent dislocation, and developmental disorders [57, 58]. Comparison of benefits among surgical procedures is difficult, because, unlike pharmacological treatments, placebo control is not possible, due to ethical reasons [59]. Disc repair and repositioning is not favored because of the short-lived success of this procedure [60]. Discectomy (complete removal of the TMJ disc) on the contrary, is still widely used and can have a significant positive long-term effect on the management of patients with advanced TMD who fail to respond to non-invasive treatments [61]. However, this procedure cannot prevent osteoarthrosis, which eventually is manifest as regressive remodeling of the mandibular condyles, destruction of condylar articular surface, and osteophyte formation [61]. Implantation of synthetic disc alternatives such as Teflon-Proplast or silicone implants resulted in disastrous outcomes [62, 63]. Substitution of the TMJ disc with autologous dermal grafts [61], did not provide significant advantage over discectomy either. The only benefit these grafts had over discectomy was in minimizing crepitus in the TMJs with no disc [61]. No overwhelming success was observed with other types of implants such as ear cartilage, full thickness skin graft, or use of pedicled temporalis muscle [58, 64, 65].
Total joint replacement and reconstruction approaches, using autologous tissues or metallic prostheses, have also been explored [66]. In greatly simplified terms, the autologous reconstruction is favored in children, due to the ability of autologous implants to grow and remodel, while alloplasty is preferred for adults [67]. Although erosion and heterotopic bone formation are described in cases where alloplastic devices are used [68], newer, custom-made prostheses seem to offer favorable outcomes up to 15 years [67].
Currently, the repair and replacement of TMJ tissue components constitutes an unmet need, highlighting the importance of developing novel approaches toward treating patients with TMD. Tissue engineering approaches have emerged as promising treatment options for musculoskeletal disorders. Since introducing these methods, significant effort was expended to design and produce TMJ components that have the ability to replicate the mechanical and biochemical properties of the native tissues. In the following section exciting advancements in tissue engineering of TMJ components are reviewed.
Tissue engineering approaches for TMJ tissues
Tissue engineering is emerging as a promising option to repair or, potentially replace the diseased tissues of the mechanically demanding, and biologically complex TMJ. Traditionally, the principal elements of tissue engineering arecells, stimuli, and scaffolds (Fig. 1B). Recently, a cell-based, scaffold-free method has also emerged [69]. This section discusses engineering of TMJ disc and condylar fibrocartilaginous tissues, as organized by scaffold-based and scaffold-free methodologies; cells and stimuli used for each tissue are also presented with respect to the discussed technique.
Scaffold-based approaches
Scaffolds serve as a supportive structure to render shape and volume to the engineered tissues, allowing for well-defined geometries and ease of handling [70]. The scaffold diffusional characteristics can be modulated by changing pore size and porosity. The mechanical properties and surface characteristics can also be tailored to the tissue being engineered. Ideally, scaffold selection should match scaffold degradation with matrix synthesis. Scaffold degradation rates depend on scaffold chemistry and can be altered by manipulating, for example, the molecular weight of synthetic polymers. To induce more rapid matrix synthesis in vivo, scaffolds can be impregnated with growth factors. Both synthetic and naturally-derived scaffolding materials have been examined for engineering the TMJ disc and condyle, but there is little progress with similar methods for the mandibular fossa [6].
TMJ disc
Scaffolds used in TMJ disc tissue engineering include alginate hydrogels [71], polylactic acid (PLA) [72], polyglycolic acid (PGA) [71, 73, 74, 20], poly–L–lacticacid (PLLA) [75], decellularized native ECM materials [76], polytetrafluoroethylene (ePTFE) monofilaments [77], poly (glycerol sebacate) (PGS) [78], and, recently, polycaprolactone (PCL) polyester [79••]. As noted above, scaffold characteristics must be tailored to the cell types used to engineer the tissue. For example, porcine TMJ disc cells seeded on PGA scaffolds result in constructs that contract severely because the PGA degrades much faster than the matrix is produced. Although the addition of insulin-like growth factor-1 (IGF-1), basic fibroblast growth factor (bFGF), and transforming growth factor-β1 (TGF-β1) improved matrix synthesis [73, 74], the scaffold still degradesfast. As a result, a different scaffold material, PLLA, was examined for its much slower degradation rate [75]. Overall, PLLA scaffolds seeded with porcine TMJ cells and treated with TGF-β1 demonstrated higher collagen and GAG contents and improved mechanical properties (1.4 MPa Young's modulus) when compared to constructs seeded on PGA [75]. Maintaining the desired shape and size during tissue engineering can be challenging not only for the disc [20], but other tissues as well [80], and scaffold optimization toward this parameter remains an ongoing endeavor.
The advent of new manufacturing techniques may allow for the production of scaffolds that more closely mimic the unique structures of TMJ components, including tissue anisotropy. Toward this end, additive manufacturing was used to 3D print PCL scaffolds with an anisotropic internal structure [79••]. Seeded with mesenchymal stem cells (MSCs), anisotropic properties were observed. However, given the short time points examined in this study, it is unclear if the anisotropic properties would be retained long-term, since the observed anisotropy was likely due to the scaffold as opposed to the matrix produced. The goal of using scaffolds, to engineer anisotropic TMJ tissues composed of only cell-generated matrices, is yet to be realized.
Central to the efforts of tissue engineering are identifying a suitable source of cells and seeding density. TMJ disc cells, as well as articular chondrocytes derived from the condyle, fossa-eminence and shoulder, in addition to dermal fibroblasts have all been examined for engineering the TMJ disc [81]. For example, dermal fibroblasts showed chondrogenic potential when treated with IGF-1. These cells are particularly promising since they are clinically relevant for autologous therapy without significant donor site morbidity. Cell seeding density is another important factor that influences the composition of the resultant construct [82]. Increasing cell seeding density does not always improve functional or biomechanical properties, so seeding density must be carefully controlled. For example, TMJ disc cells seeded from 15-120M cells/ml of scaffold volume onto PGA scaffolds show variable results. Increasing the cell number up to 120M cells/ml of scaffold volume increased GAG and collagen content without significant improvement in the compressive properties of the engineered tissue. For each new cell source, identified for engineering the TMJ disc, the lowest seeding density that yields desirable functional properties must be determined.
Mechanical stimuli approximating physiological loading profiles have been used to condition engineered TMJ discs to improve their mechanical and biochemical properties. In situ, the native TMJ disc is exposed to tension, compression, shear, and hydrostatic pressure during joint movement. When applying mechanical stimuli, a potential disadvantage in using scaffolds is stress-shielding. For example, applying tension and compression onto cell-seeded scaffolds can result in the scaffold bearing the load as opposed to having the load propagate down to the cellular level [83]. Spinner flask and orbital shaker cultures have both been used to apply fluid-induced shear [71]. When cultured in spinner flasks, TMJ disc cells seeded on PGA showed higher ECM production 4 weeks after seeding compared to constructs in static culture. However, no significant improvement was observed in mechanical properties in comparison with static conditions [84]. Hydrostatic pressure at 10 MPa, applied either constantly or intermittently for 4 hours a day, using a duty cycle of 2 days on, 1 day off for 1 week, showed that the constantly applied load led to the highest amount of collagen and number of cells per construct [85]. The use of these bioreactors results in enhanced GAG and collagen synthesis, but corresponding improvements in mechanical properties must be demonstrated.
Bioactive signals have been used to promote collagen and GAG synthesis in engineered TMJ discs, with the expectation that this approach would lead to improved mechanical properties of the engineered tissues. Effects elicited by bioactive signals depend greatly on conditions of the experiment. For example, in a study that compared platelet derived growth factor-AB (PDGF), bFGF, and IGF-1 at various concentrations in monolayer, bFGF was shown to result in the greatest improvements in GAG and collagen production (∼2 and 4.5 fold increases, respectively, compared to control) [86]. However, when examining IGF-1, bFGF, and TGF-β1 on cell-seeded PGA scaffolds in spinner flasks, the results suggested that IGF-1 elicited the greatest collagen production [73]. Both studies used porcine TMJ disc cells, but the conditions varied, the former was a two-dimensional, static culture, while the latter was a three-dimensional culture subjected to fluid-induced shear. Due to the limited number of studies on the efficacy of growth factors for scaffold-based engineering of TMJ discs, additional work is needed to optimize relevant growth factors, doses and regimens. However, a comprehensive picture will not emerge until this characterization is performed for each scaffold and culture condition.
TMJ condylar fibrocartilage
Numerous types of scaffolds for engineering the TMJ condylar fibrocartilage have been examined and reviewed elsewhere [6]; the most common of these include PLGA, PGA, PCL, and poly(ethylene glycol) (PEG). Rat bone-marrow MSCs, induced to differentiate into chondrogenic and osteogenic lineages, were seeded into a condyle-shaped PEG hydrogel [87]. Unfortunately, the functional properties of the engineered tissue were not confirmed by mechanical testing, but there was histological and immunohistochemical evidence for both cartilage and bone. In general, engineering the condyle may require generating both cartilage and osseous tissue, and studies that attempt to do so should characterize the mechanical properties of each component.
Various cell types, from chondrocytes to stem cells, have been compared to TMJ condylar cells with respect to engineering condyle fibrocartilage using scaffolds [88]. Chondrocytes from the ankle were compared to fibrochondrocytes from the mandibular condyle seeded on PGA and treated with IGF-1; the anklechondrocytes outperformed condylar fibrochondrocytes and produced 10- and 6-fold more GAG and collagen, respectively [16]. When human umbilical cord matrix stem cells were compared to TMJ condyle fibrochondrocytes, PGA constructs seeded with the stem cells contained significantly higher GAG and cell number, but there was no increase in collagen content [89]. Notably, collagen types I and II were observed for the stem cell-seeded constructs, but not for the cultures seeded with condylar fibrochondrocytes. From these studies, it is clear that more work is needed to optimize the culture of condylar fibrochondrocytes. As presently reviewed, various cell types have demonstrated greater promise than TMJ condylar cells for the tissue engineering of condylar fibrocartilage.
Bioreactors for engineering condylar fibrocartilage have been reviewed elsewhere [88]. A few studies have also looked at the effects of biomechanical stimuli on other cell types, but not all the results are positive. For example, applying sinusoidal, dynamic loading at 0.3 Hz and a 15% amplitude strain to condyle fibrochondrocytes in PEG hydrogels resulted in inhibition of cell proliferation and proteoglycan synthesis [90]. In another study, applying hydrostatic pressure up to 90kPa for 720 minutes to rabbit mandibular fibrochondrocytes in monolayer increased cell proliferation; however, alkaline phosphatase activity was also elevated, which might induce an undesirable mineralization effect when engineering fibrocartilage [91]. Bioreactors have also been used for engineering bone in a condylar shape [92]. In this respect, rotational [93] and perfusion [94] bioreactors were used to culture porcine MSCs in PLGA and human MSCs in a cellular bone, respectively. The rotational bioreactor experiment was a pilot study that did not compare the tissues generated with other bioreactors or static controls, so the utility of a rotational bioreactor remains unclear for engineering bone constructs for the condyle. A perfusion bioreactor was shown to improve cell attachment by 2-fold compared to static culture [94]. Bioreactors that have shown efficacy in cartilage and bone tissue engineering may also be beneficial for condyle fibrocartilage tissue engineering [88], though this has yet to be confirmed. Due to the limited studies on engineering the condyle, a robust conclusion on the effectiveness of bioreactors is premature.
Growth factors have been studied for increasing cell proliferation and biosynthesis in condylar fibrochondrocytes, both in a monolayer and seeded in scaffolds. In monolayer, 10 ng/mL basic fibroblast growth factor (bFGF) resulted in the highest increase (65%) in proliferation of human mandibular condylar fibrochondrocytes, compared to increases due to 10 ng/mL TGF-β1 (13%) and 10 ng/mL IGF-1 (24%) [95]. When comparing hyaline chondrocytes to condylar fibrochondrocytes, only the chondrocytes were responsive to IGF-1, forming a fibrocartilage-like tissue with types I and II collagen, with about an 8-fold increase in both GAG and collagen contents over unstimulated controls [16]. Although the data on the use of growth factors in engineering the condyle are scarce, bFGF and IGF-1 have emerged as effective stimuli for cell proliferation and biosynthesis, respectively.
Scaffold-free approach
As reviewed elsewhere [96], limitations of scaffold-based methods include: 1. potential mismatches between the rates of scaffold degradation and matrix production, 2. induction of foreign body responses, 3. limited mechanical strength after scaffold degradation, 4. phenotypic alterations due to the scaffold, and 5. stress shielding. In response, scaffold-free techniques have emerged that produce new tissues by mimicking the developmental process. Thermodynamically driven modalities, known as self-organization and self-assembling process, have both been used to engineer the TMJ disc, but not yet the condyle [97•].
TMJ disc
TMJ disc cells, dermal fibroblasts, and costal chondrocytes, among other cell types, have been used to engineer the TMJ disc in a scaffold-free manner. A study comparing TMJ disc cells and costal chondrocytes at passage numbers 0, 1, 3, and 5 showed that, costal chondrocytes outperform TMJ disc cells [98]. More GAG was observed in constructs formed from passaged, versus primary, costal chondrocytes, but collagen content remained unaffected. Another study compared constructs derived from costal chondrocytes, dermal fibroblasts, and a mixture of the two to constructs formed by TMJ disc cells [99]. It was found that constructs derived either from costal chondrocytes or from the mixture of costal chondrocytes and dermal fibroblasts had superior morphological and biochemical qualities compared to constructs formed using TMJ cells. Compared to constructs formed using TMJ disc cells, the diameter of costal chondrocyte-derived constructs was 3 times larger, and the collagen content was 40 times greater [99]. When compared with passaged articular chondrocytes, passaged costal chondrocytes resulted in constructs with greater GAG content, but functional properties and collagen content did not show significant improvement [100]. Costal cartilage can be harvested relatively easily, and it is currently used for mandibular reconstructions. By extension, costal chondrocytes may serve as an autologous cell source that eliminates complications such as rejection. To date, passaged costal chondrocytes appear to be the most promising cell source for scaffold-free tissue engineering of the TMJ disc.
Compression loading of TMJ discs engineered with the scaffold-free method, has yielded not only increased mechanical properties, but also induced anisotropy. The application of a static load on shape-specific TMJ disc constructs, formed using a co-culture of bovine articular chondrocytes and meniscal fibrochondrocytes, improved both functional properties and biosynthesis [101]. With a 0.1N load application, significant increases were reported in the compression relaxation modulus and tensile Young's modulus by 96% and 255%, respectively, compared to controls. In addition, results found the 0.1N load to significantly increase both collagen and GAG synthesis by 27% and 67%, respectively [101]. Interestingly, the 0.1N load induced anisotropy and increased fiber alignment by 202% as compared to controls. Due to the shape of the engineered construct, static uniaxial load result in both compressive and tensile stresses. The scaffold-based approach does not usually yield anisotropic matrices, but scaffold-free constructs, under the influence of mechanical forces, do produce anisotropic properties akin to those found in the native TMJ.
The use of soluble factorsis emerging as a central player in scaffold-free tissue engineering of the TMJ disc. Scaffold-free constructs formed using a co-culture of bovine articular chondrocytes and fibrochondrocytes showed positive responses to continuous application of TGF-β1. Furthermore, collagen synthesis, compressive stiffness, and Young's modulus increased by 20, 130, and 170%, respectively, compared to unstimulated controls [102]. In contrast, scaffold-free TMJ disc constructs derived from goat costal chondrocyte cultures showed no significant response to TGF-β1, IGF-1, EGF, PDGF, or bFGF at the concentrations and dosing regimens examined [103]. In contrast, soluble factors whose mechanisms involve direct action on synthesized matrices appear to have broad effects across multiple culture platforms. For example, the catabolic enzyme chondroitinase-ABC (C-ABC), which removes GAGs to collapse the collagen network, has been applied both to co-cultures of articular chondrocytes and fibrochondrocytes, as well as to costal chondrocytes to elicit improved tensile properties [104,105•,106]. Lysyl oxidase-like protein 2 (LOXL2) is an enzyme that crosslinks collagen. Applied either to co-cultures of chondrocytes and fibrochondrocytes or to costal chondrocytes, LOXL2 improved integration of scaffold-free constructs to native TMJ discs [106, 107]. Combining the soluble factors discussed above has yielded synergistic effects; TGF-β1, C-ABC, and LOXL2 treatment synergistically enhanced Young's modulus and ultimate tensile strength of scaffold-free constructs by 245% and 185%, respectively [108••]. Thus far, the soluble factors discussed above that act directly on the extracellular matrix have only been applied to scaffold-free cultures. Those interesting results warrant examination of the same factors in scaffold-based systems.
TMJ Condylar fibrocartilage
For engineering condylar fibrocartilage, scaffold-based studies greatly outnumber those employing scaffold-free techniques. The few attempts at generating scaffold-free condylar tissue are limited to forming only one phase (e.g., cartilage) using scaffold-free techniques. For example, an osteochondral, condyle-shaped construct was formed by adhering scaffold-free cartilage on top of MSC-seeded alginate in the shape of a condyle using fibrin glue. After being subcutaneously implanted into nude mice for 8 weeks, endochondral ossification was observed with vascularized bone formation in the alginate scaffold, while the cartilage phase retained its phenotype [109]. The mechanical integrity of the interface was not tested, though histological analysis showed that the integration between the two phases was retained. It is expected that, for a condylar implant to survive the mechanically stressful environment of the TMJ, a mechanically robust interface between the engineered cartilage and bone must first be achieved, and future condyle tissue engineering attempts should take integration into consideration.
Conclusion
Engineering of tissue replacements for TMD patients is emerging as a promising approach toward providing biological solutions to these as-of-yet intractable problems. Although there is no commercial, tissue engineered product currently on the market for treating TMD, several studies have been performed toward the development of appropriate tools for engineering TMJ tissues. Ideally, the goal is to employ a biomimetic approach to produce new tissues de novo (neotissues) with qualities similar to the native TMJ.
In addition to the tissue engineering issues reviewed, an important future challenge is the engineering of shape-specific constructs that are of similar dimensions to the specific TMJ tissues to be replaced. Most tissue engineering studies examine flat and relatively small neotissues. However, since both TMJ disc and condyle have complex geometries, from a translational perspective, a scale-up approach is needed that also considers shape fidelity.
Even if large biomimetic constructs are engineered, a looming challenge is that of surgically accessing the ailing TMJ tissues and integrating the neotissues within the native milieu. Indeed, it is important to develop appropriate surgical approaches for the TMJ, especially with regards to implantation of tissue engineered constructs. Concomitant to surgical approaches, achieving neotissue integration into the TMJ is of paramount importance. As discussed above, LOXL2 appears to exhibit integrative efficacy in vitro, but its effects have not been examined in vivo. Attempts should be made to identify factors that can enhance interfacial strength between neotissues and native tissues.
As new tissue engineering approaches for the TMJ evolve, establishment of suitable animal model is needed to standardize the translational pathway, similar to what has been achieved in other joints, such as the knee. Although the minipig or the domestic pig are emerging as suitable models, appropriate studies are needed to confirm their application to the TMJ.
As the tissue engineering field is poised to move from the bench to the bedside treatment of TMD, it is important that appropriate FDA guidelines be developed to allow for the efficacious development of suitable clinical solutions. Despite the many challenges, there is a great opportunity to use tissue engineering as the vehicle to provide cogent solutions for TMD, one of the most notoriously complex musculoskeletal problems.
Acknowledgments
This work was supported by grant R01DE19666 from the National institute of Health. The authors would like to acknowledge Dr. David Hatcher for provision of cone beam CT images. Kyriacos Athanasiou reports grants from the NIH during the conduct of the study.
Footnotes
Compliance with Ethics Guidelines: Ashkan Aryaei, Natalia Vapniarsky, and Jerry Hu report no conflicts of interest.
Human and Animal Rights and Informed Consent: This article does not contain any studies with human or animal subjects performed by any of the authors.
Contributor Information
Ashkan Aryaei, Email: aryaei@ucdavis.edu.
Natalia Vapniarsky, Email: vapniarsky@ucdavis.edu.
Jerry C Hu, Email: jcyhu@ucdavis.edu.
References
Papers of particular interest, published recently, have been highlighted as:
• Of importance
•• Of major importance
- 1.Gatchel RJ, Stowell AW, Wildenstein L, Riggs R, Ellis E. Efficacy of an early intervention for patients with acute temporomandibular disorder–related pain: a one-year outcome study. J Am Dent Assoc. 2006;137:339–347. doi: 10.14219/jada.archive.2006.0183. [DOI] [PubMed] [Google Scholar]
- 2.Solberg WK, Woo MW, Houston JB. Prevalence of mandibular dysfunction in young adults. J Am Dent Assoc. 1979;98:25–34. doi: 10.14219/jada.archive.1979.0008. [DOI] [PubMed] [Google Scholar]
- 3.Zarb GA, Carlsson GE. Temporomandibular disorders: osteoarthritis. J Orofac Pain. 1999;13 [PubMed] [Google Scholar]
- 4.Van Loon JP, De Bont L, Stegenga B, Spijkervet F, Verkerke G. Groningen temporomandibular joint prosthesis. Development and first clinical application. Int J Oral Maxillofac Surg. 2002;31:44–52. doi: 10.1054/ijom.2001.0175. [DOI] [PubMed] [Google Scholar]
- 5.Chisnoiu AM, Picos AM, Popa S, Chisnoiu PD, Lascu L, Picos A, Chisnoiu R. Factors involved in the etiology of temporomandibular disorders-a literature review. Clujul Medical. 2015;88:473. doi: 10.15386/cjmed-485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Murphy MK, MacBarb RF, Wong ME, Athanasiou KA. Temporomandibular Joint Disorders: A Review of Etiology, Clinical Management, and Tissue Engineering Strategies. Int J Oral Maxillofac Implants. 2013;28:e393. doi: 10.11607/jomi.te20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Athanasiou KA, Almarza AJ, Detamore MS, Kalpakci KN. Tissue engineering of temporomandibular joint cartilage. Synthesis Lectures on Tissue Engineering. 2009;1:1–122. [Google Scholar]
- 8.Kalpakci K, Willard V, Wong M, Athanasiou K. An interspecies comparison of the temporomandibular joint disc. J Dent Res. 2011;90:193–198. doi: 10.1177/0022034510381501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ngan PW, Deguchi T, Roberts EW. Orthodontic Treatment of Class III Malocclusion. 2014 [Google Scholar]
- 10.Symons NB. The development of the human mandibular joint. J Anat. 1952;86:326–333. [PMC free article] [PubMed] [Google Scholar]
- 11.Baume LJ. Ontogenesis of the human temporomandibular joint. 1. Development of the condyles. J Dent Res. 1962;41:1327–1339. doi: 10.1177/00220345620410060901. [DOI] [PubMed] [Google Scholar]
- 12.Hansson T, Öberg T, Carlsson GE, Kopp S. Thickness of the soft tissue layers and the articular disk in the temporomandibular joint. Acta Odontologica Scandinavica. 1977;35:77–83. doi: 10.3109/00016357709055993. [DOI] [PubMed] [Google Scholar]
- 13.Bibb C, Pullinger A, Baldioceda F. Serial variation in histological character of articular soft tissue in young human adult temporomandibular joint condyles. Arch Oral Biol. 1993;38:343–352. doi: 10.1016/0003-9969(93)90142-9. [DOI] [PubMed] [Google Scholar]
- 14.Roth S, Müller K, Fischer DC, Dannhauer KH. Specific properties of the extracellular chondroitin sulphate proteoglycans in the mandibular condylar growth centre in pigs. Arch Oral Biol. 1997;42:63–76. doi: 10.1016/s0003-9969(97)83718-1. [DOI] [PubMed] [Google Scholar]
- 15.Singh M, Detamore M. Biomechanical properties of the mandibular condylar cartilage and their relevance to the TMJ disc. J Biomech. 2009;42:405–417. doi: 10.1016/j.jbiomech.2008.12.012. [DOI] [PubMed] [Google Scholar]
- 16.Wang L, Lazebnik M, Detamore M. Hyaline cartilage cells outperform mandibular condylar cartilage cells in a TMJ fibrocartilage tissue engineering application. Osteoarthritis and Cartilage. 2009;17:346–353. doi: 10.1016/j.joca.2008.07.004. [DOI] [PubMed] [Google Scholar]
- 17.Detamore MS, Athanasiou KA. Tensile properties of the porcine temporomandibular joint disc. J Biomech Eng. 2003;125:558–565. doi: 10.1115/1.1589778. [DOI] [PubMed] [Google Scholar]
- 18.Kuroda S, Tanimoto K, Izawa T, Fujihara S, Koolstra J, Tanaka E. Biomechanical and biochemical characteristics of the mandibular condylar cartilage. Osteoarthritis and Cartilage. 2009;17:1408–1415. doi: 10.1016/j.joca.2009.04.025. [DOI] [PubMed] [Google Scholar]
- 19.Delatte M, Von den Hoff JW, Van Rheden RE, Kuijpers-Jagtman AM. Primary and secondary cartilages of the neonatal rat: the femoral head and the mandibular condyle. Eur J Oral Sci. 2004;112:156–162. doi: 10.1111/j.0909-8836.2004.00108.x. [DOI] [PubMed] [Google Scholar]
- 20.Detamore MS, Athanasiou KA. Structure and function of the temporomandibular joint disc: implications for tissue engineering. J Oral Maxillofac Surg. 2003;61:494–506. doi: 10.1053/joms.2003.50096. [DOI] [PubMed] [Google Scholar]
- 21.Murphy M, Arzi B, Hu J, Athanasiou K. Tensile characterization of porcine temporomandibular joint disc attachments. J Dent Res. 2013;92:753–758. doi: 10.1177/0022034513494817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Christo J, Bennett S, Wilkinson T, Townsend G. Discal attachments of the human temporomandibular joint. Aust Dent J. 2005;50:152–160. doi: 10.1111/j.1834-7819.2005.tb00354.x. [DOI] [PubMed] [Google Scholar]
- 23.Detamore MS, Hegde JN, Wagle RR, Almarza AJ, Montufar-Solis D, Duke PJ, Athanasiou KA. Cell type and distribution in the porcine temporomandibular joint disc. J Oral Maxillofac Surg. 2006;64:243–248. doi: 10.1016/j.joms.2005.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wong GB, Weinberg S, Symington JM. Morphology of the developing articular disc of the human temporomandibular joint. J Oral Maxillofac Surg. 1985;43:565–569. doi: 10.1016/0278-2391(85)90121-1. [DOI] [PubMed] [Google Scholar]
- 25.Detamore MS, Orfanos JG, Almarza AJ, French MM, Wong ME, Athanasiou KA. Quantitative analysis and comparative regional investigation of the extracellular matrix of the porcine temporomandibular joint disc. Matrix Biol. 2005;24:45–57. doi: 10.1016/j.matbio.2004.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Scapino RP, Obrez A, Greising D. Organization and function of the collagen fiber system in the human temporomandibular joint disk and its attachments. Cells Tissues Organs. 2006;182:201–225. doi: 10.1159/000093969. [DOI] [PubMed] [Google Scholar]
- 27.Berkovitz B. Collagen crimping in the intra-articular disc and articular surfaces of the human temporomandibular joint. Arch Oral Biol. 2000;45:749–756. doi: 10.1016/s0003-9969(00)00045-5. [DOI] [PubMed] [Google Scholar]
- 28.Almarza A, Bean A, Baggett L, Athanasiou K. Biochemical analysis of the porcine temporomandibular joint disc. Br J Oral Maxillofac Surg. 2006;44:124–128. doi: 10.1016/j.bjoms.2005.05.002. [DOI] [PubMed] [Google Scholar]
- 29.Plumb M, Aspden R. The response of elderly human articular cartilage to mechanical stimuli in vitro. Osteoarthritis and cartilage. 2005;13:1084–1091. doi: 10.1016/j.joca.2005.07.002. [DOI] [PubMed] [Google Scholar]
- 30.Aoun M, Mesnard M, Monède-Hocquard L, Ramos A. Stress analysis of temporomandibular joint disc during maintained clenching using a viscohyperelastic finite element model. J Oral Maxillofac Surg. 2014;72:1070–1077. doi: 10.1016/j.joms.2013.11.031. [DOI] [PubMed] [Google Scholar]
- 31.Sun M, Yang J, Zhou R, Li N, Xia J, Gu F. Mechanical analysis on individualized finite element of temporal-mandibular joint under overlarge jaw opening status. Int J Clin Exp Med. 2015;8:9046. [PMC free article] [PubMed] [Google Scholar]
- 32.Tanaka E, van Eijden T. Biomechanical behavior of the temporomandibular joint disc. Crit Rev Oral Biol. 2003;14:138–150. doi: 10.1177/154411130301400207. [DOI] [PubMed] [Google Scholar]
- 33.Pullinger AG, Seligman DA. Multifactorial analysis of differences in temporomandibular joint hard tissue anatomic relationships between disk displacement with and without reduction in women. J Prosthet Dent. 2001;86:407–419. doi: 10.1067/mpr.2001.118563. [DOI] [PubMed] [Google Scholar]
- 34.Berger M, Szalewski L, Bakalczuk M, Bakalczuk G, Bakalczuk S, Szkutnik J. Association between estrogen levels and temporomandibular disorders: a systematic literature review. Przeglad menopauzalny= Menopause review. 2015;14:260–270. doi: 10.5114/pm.2015.56538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bi RY, Ding Y, Gan YH. A new hypothesis of sex-differences in temporomandibular disorders: Estrogen enhances hyperalgesia of inflamed TMJ through modulating voltage-gated sodium channel 1.7 in trigeminal ganglion? Med Hypotheses. 2015;84:100–103. doi: 10.1016/j.mehy.2014.12.010. [DOI] [PubMed] [Google Scholar]
- 36.Fischer DJ, Mueller BA, Critchlow CW, LeResche L. The association of temporomandibular disorder pain with history of head and neck injury in adolescents. J Orofac Pain. 2006;20 [PubMed] [Google Scholar]
- 37.Barrera-Mora JM, Escalona EE, Labruzzi CA, Carrera JML, Ballesteros EJC, Reina ES, Rocabado M. The relationship between malocclusion, benign joint hypermobility syndrome, condylar position and TMD symptoms. CRANIO®. 2012;30:121–130. doi: 10.1179/crn.2012.018. [DOI] [PubMed] [Google Scholar]
- 38.Stemig M, Myers SL, Kaimal S, Islam MS. Estrogen receptor-alpha polymorphism in patients with and without degenerative disease of the temporomandibular joint. CRANIO®. 2015;33:129–133. doi: 10.1179/2151090314Y.0000000013. [DOI] [PubMed] [Google Scholar]
- 39.Dias GM, Bonato LL, Guimarães JP, Silva JNN, Ferreira LA, Grossmann E, Carvalho ACP. A Study of the Association Between Sleep Bruxism, Low Quality of Sleep, and Degenerative Changes of the Temporomandibular Joint. J Craniofac Surg. 2015;26:2347–2350. doi: 10.1097/SCS.0000000000002084. [DOI] [PubMed] [Google Scholar]
- 40.Meloto CB, Serrano PO, Ribeiro-DaSilva MC, Rizzatti-Barbosa CM. Genomics and the new perspectives for temporomandibular disorders. Arch Oral Biol. 2011;56:1181–1191. doi: 10.1016/j.archoralbio.2011.03.012. [DOI] [PubMed] [Google Scholar]
- 41.Suvinen TI, Reade PC. Temporomandibular Disorders: A Critical Review of the Nature of Pain and Its Assessment. J Orofac Pain. 1995;9 [PubMed] [Google Scholar]
- 42.Jerjes W, Upile T, Abbas S, Kafas P, Vourvachis M, Rob J, Mc Carthy E, Angouridakis N, Hopper C. Muscle disorders and dentition-related aspects in temporomandibular disorders: controversies in the most commonly used treatment modalities. Int Arch Med. 2008;1:23. doi: 10.1186/1755-7682-1-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43•.Wang X, Zhang J, Gan Y, Zhou Y. Current understanding of pathogenesis and treatment of TMJ osteoarthritis. J Dent Res. 2015;94:666–673. doi: 10.1177/0022034515574770. This short review paper gives a clear and comprehensive understanding on the pathogenesis and current treatment options of TMJ disorders. [DOI] [PubMed] [Google Scholar]
- 44.Klasser GD, Greene CS. Oral appliances in the management of temporomandibular disorders. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;107:212–223. doi: 10.1016/j.tripleo.2008.10.007. [DOI] [PubMed] [Google Scholar]
- 45.Ingawalé S, Goswami T. Temporomandibular joint: disorders, treatments, and biomechanics. Ann Biomed Eng. 2009;37:976–996. doi: 10.1007/s10439-009-9659-4. [DOI] [PubMed] [Google Scholar]
- 46.Al-Ani MZ, Davies SJ, Gray RJM, Sloan P, Glenny AM. Stabilisation splint therapy for temporomandibular pain dysfunction syndrome. Cochrane Database Syst Rev. 2004:CD002778. doi: 10.1002/14651858.CD002778.pub2. [DOI] [PubMed] [Google Scholar]
- 47.Hersh EV, Balasubramaniam R, Pinto A. Pharmacologic management of temporomandibular disorders. Oral Maxillofac Surg Clin North Am. 2008;20:197–210. doi: 10.1016/j.coms.2007.12.005. [DOI] [PubMed] [Google Scholar]
- 48.Liu F, Steinkeler A. Epidemiology, diagnosis, and treatment of temporomandibular disorders. Dent Clin North Am. 2013;57:465–479. doi: 10.1016/j.cden.2013.04.006. [DOI] [PubMed] [Google Scholar]
- 49.Dym H, Bowler D, Zeidan J. Pharmacologic Treatment for Temporomandibular Disorders. Dent Clin North Am. 2016;60:367–379. doi: 10.1016/j.cden.2015.11.012. [DOI] [PubMed] [Google Scholar]
- 50.Mujakperuo HR, Watson M, Morrison R, Macfarlane TV. Pharmacological interventions for pain in patients with temporomandibular disorders. Cochrane Database Syst Rev. 2010;(10) doi: 10.1002/14651858.CD004715.pub2. [DOI] [PubMed] [Google Scholar]
- 51.Machado E, Bonotto D, Cunali PA. Intra-articular injections with corticosteroids and sodium hyaluronate for treating temporomandibular joint disorders: a systematic review. Dental Press J Orthod. 2013;18:128–133. doi: 10.1590/s2176-94512013000500021. [DOI] [PubMed] [Google Scholar]
- 52.Brennan PA, Ilankovan V. Arthrocentesis for temporomandibular joint pain dysfunction syndrome. J Oral Maxillofac Surg. 2006;64:949–951. doi: 10.1016/j.joms.2006.02.010. [DOI] [PubMed] [Google Scholar]
- 53.Carvajal WA, Laskin DM. Long-term evaluation of arthrocentesis for the treatment of internal derangements of the temporomandibular joint. J Oral Maxillofac Surg. 2000;58:852–855. doi: 10.1053/joms.2000.8201. [DOI] [PubMed] [Google Scholar]
- 54.Monje-Gil F, Nitzan D, González-Garcia R. Temporomandibular joint arthrocentesis. Review of the literature. Med Oral Patol Oral Cir Bucal. 2012;17:575–81. doi: 10.4317/medoral.17670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Dym H, Israel H. Diagnosis and treatment of temporomandibular disorders. Dent Clin North Am. 2012;56:149–161. doi: 10.1016/j.cden.2011.08.002. [DOI] [PubMed] [Google Scholar]
- 56.Rigon M, Pereira LM, Bortoluzzi MC, Loguercio AD, Ramos AL, Cardoso JR. Arthroscopy for temporomandibular disorders. Cochrane Database Syst Rev. 2011;(5) doi: 10.1002/14651858.CD006385.pub2. [DOI] [PubMed] [Google Scholar]
- 57.Elgazzar R, Abdelhady A, Saad K, Elshaal M, Hussain M, Abdelal S, Sadakah A. Treatment modalities of TMJ ankylosis: experience in Delta Nile, Egypt. Int J Oral Maxillofac Surg. 2010;39:333–342. doi: 10.1016/j.ijom.2010.01.005. [DOI] [PubMed] [Google Scholar]
- 58.Dimitroulis G. Temporomandibular joint surgery: what does it mean to the dental practitioner? Aust Dent J. 2011;56:257–264. doi: 10.1111/j.1834-7819.2011.01351.x. [DOI] [PubMed] [Google Scholar]
- 59.Reston JT, Turkelson CM. Meta-analysis of surgical treatments for temporomandibular articular disorders. J Oral Maxillofac Surg. 2003;61:3–10. doi: 10.1053/joms.2003.50000. [DOI] [PubMed] [Google Scholar]
- 60.Dolwick MF. Disc preservation surgery for the treatment of internal derangements of the temporomandibular joint. J Oral Maxillofac Surg. 2001;59:1047–1050. doi: 10.1053/joms.2001.26681. [DOI] [PubMed] [Google Scholar]
- 61.Dimitroulis G. Condylar morphology after temporomandibular joint discectomy with interpositional abdominal dermis-fat graft. J Oral Maxillofac Surg. 2011;69:439–446. doi: 10.1016/j.joms.2010.07.021. [DOI] [PubMed] [Google Scholar]
- 62.Kaplan P, Ruskin J, Tu H, Knibbe M. Erosive arthritis of the temporomandibular joint caused by Teflon-Proplast implants: plain film features. AJR Am J Roentgenol. 1988;151:337–339. doi: 10.2214/ajr.151.2.337. [DOI] [PubMed] [Google Scholar]
- 63.Westesson PL, Eriksson L, Lindström C. Destructive lesions of the mandibular condyle following diskectomy with temporary silicone implant. Oral Surg Oral Med Oral Pathol. 1987;63:143–150. doi: 10.1016/0030-4220(87)90302-1. [DOI] [PubMed] [Google Scholar]
- 64.Dimitroulis G, Slavin J. Histological evaluation of full thickness skin as an interpositional graft in the rabbit craniomandibular joint. J Oral Maxillofac Surg. 2006;64:1075–1080. doi: 10.1016/j.joms.2006.03.011. [DOI] [PubMed] [Google Scholar]
- 65.Bradley P, Brockbank J. The temporalis muscle flap in oral reconstruction: a cadaveric, animal and clinical study. J Maxillofac Surg. 1981;9:139–145. doi: 10.1016/s0301-0503(81)80034-3. [DOI] [PubMed] [Google Scholar]
- 66.Vega LG, González-García R, Louis PJ. Reconstruction of acquired temporomandibular joint defects. Oral Maxillofac Surg Clin North Am. 2013;25:251–269. doi: 10.1016/j.coms.2013.02.008. [DOI] [PubMed] [Google Scholar]
- 67.Sidebottom AJ. Alloplastic or autogenous reconstruction of the TMJ. Journal of Oral Biology and Craniofacial Research. 2013;3:135–139. doi: 10.1016/j.jobcr.2013.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Mercuri L. Alloplastic temporomandibular joint replacement: rationale for the use of custom devices. Int J Oral Maxillofac Surg. 2012;41:1033–1040. doi: 10.1016/j.ijom.2012.05.032. [DOI] [PubMed] [Google Scholar]
- 69.Hu JC, Athanasiou KA. A self-assembling process in articular cartilage tissue engineering. Tissue Eng. 2006;12:969–979. doi: 10.1089/ten.2006.12.969. [DOI] [PubMed] [Google Scholar]
- 70.Vacanti JP, Morse MA, Saltzman WM, Domb AJ, Perez-Atayde A, Langer R. Selective cell transplantation using bioabsorbable artificial polymers as matrices. Journal of pediatric surgery. 1988;23:3–9. doi: 10.1016/s0022-3468(88)80529-3. [DOI] [PubMed] [Google Scholar]
- 71.Almarza AJ, Athanasiou KA. Seeding techniques and scaffolding choice for tissue engineering of the temporomandibular joint disk. Tissue Eng. 2004;10:1787–1795. doi: 10.1089/ten.2004.10.1787. [DOI] [PubMed] [Google Scholar]
- 72.Ahtiainen K, Mauno J, Ella V, Hagstrom J, Lindqvist C, Miettinen S, Ylikomi T, Kellomaki M, Seppanen R. Autologous adipose stem cells and polylactide discs in the replacement of the rabbit temporomandibular joint disc. J R Soc Interface. 2013;10:20130287. doi: 10.1098/rsif.2013.0287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Detamore MS, Athanasiou KA. Evaluation of three growth factors for TMJ disc tissue engineering. Ann Biomed Eng. 2005;33:383–390. doi: 10.1007/s10439-005-1741-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Almarza A, Athanasiou K. Evaluation of three growth factors in combinations of two for temporomandibular joint disc tissue engineering. Arch Oral Biol. 2006;51:215–221. doi: 10.1016/j.archoralbio.2005.07.002. [DOI] [PubMed] [Google Scholar]
- 75.Allen K, Athanasiou K. Scaffold and growth factor selection in temporomandibular joint disc engineering. J Dent Res. 2008;87:180–185. doi: 10.1177/154405910808700205. [DOI] [PubMed] [Google Scholar]
- 76.Brown BN, Badylak SF. Extracellular matrix as an inductive scaffold for functional tissue reconstruction. Transl Res. 2014;163:268–285. doi: 10.1016/j.trsl.2013.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Springer IN, Fleiner B, Jepsen S, Açil Y. Culture of cells gained from temporomandibular joint cartilage on non-absorbable scaffolds. Biomater. 2001;22:2569–2577. doi: 10.1016/s0142-9612(01)00148-x. [DOI] [PubMed] [Google Scholar]
- 78.Hagandora CK, Gao J, Wang Y, Almarza AJ. Poly (glycerol sebacate): a novel scaffold material for temporomandibular joint disc engineering. Tissue Eng Part A. 2012;19:729–737. doi: 10.1089/ten.tea.2012.0304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79••.Legemate K, Tarafder S, Jun Y, Lee C. Engineering Human TMJ Discs with Protein-Releasing 3D-Printed Scaffolds. J Dent Res. 2016 doi: 10.1177/0022034516642404. Paper mimics the structure of the native TMJ disc using 3D printing method. Appropriate growth factors were delivered to MSC cells by encapsulating them in microparticles. The results showed anisotropic scaffold microstructures can be used to fabricate heterogeneous fibrocartilaginous matrix and region-dependent viscoelastic properties similar to the native tissue. [DOI] [PubMed] [Google Scholar]
- 80.Moutos FT, Freed LE, Guilak F. A biomimetic three-dimensional woven composite scaffold for functional tissue engineering of cartilage. Nature materials. 2007;6:162–167. doi: 10.1038/nmat1822. [DOI] [PubMed] [Google Scholar]
- 81.Allen KD, Athanasiou KA. Tissue engineering of the TMJ disc: a review. Tissue Eng. 2006;12:1183–1196. doi: 10.1089/ten.2006.12.1183. [DOI] [PubMed] [Google Scholar]
- 82.Almarza AJ, Athanasiou KA. Effects of initial cell seeding density for the tissue engineering of the temporomandibular joint disc. Ann Biomed Eng. 2005;33:943–950. doi: 10.1007/s10439-005-3311-8. [DOI] [PubMed] [Google Scholar]
- 83.Makris EA, Gomoll AH, Malizos KN, Hu JC, Athanasiou KA. Repair and tissue engineering techniques for articular cartilage. Nature Reviews Rheumatology. 2015;11:21–34. doi: 10.1038/nrrheum.2014.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Detamore MS, Athanasiou KA. Use of a rotating bioreactor toward tissue engineering the temporomandibular joint disc. Tissue Eng. 2005;11:1188–1197. doi: 10.1089/ten.2005.11.1188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Almarza AJ, Athanasiou KA. Effects of hydrostatic pressure on TMJ disc cells. Tissue Eng. 2006;12:1285–1294. doi: 10.1089/ten.2006.12.1285. [DOI] [PubMed] [Google Scholar]
- 86.Detamore MS, Athanasiou KA. Effects of growth factors on temporomandibular joint disc cells. Arch Oral Biol. 2004;49:577–583. doi: 10.1016/j.archoralbio.2004.01.015. [DOI] [PubMed] [Google Scholar]
- 87.Alhadlaq A, Elisseeff JH, Hong L, Williams CG, Caplan AI, Sharma B, Kopher RA, Tomkoria S, Lennon DP, Lopez A. Adult stem cell driven genesis of human-shaped articular condyle. Ann Biomed Eng. 2004;32:911–923. doi: 10.1023/b:abme.0000032454.53116.ee. [DOI] [PubMed] [Google Scholar]
- 88.Wang L, Detamore MS. Tissue engineering the mandibular condyle. Tissue Eng. 2007;13:1955–1971. doi: 10.1089/ten.2006.0152. [DOI] [PubMed] [Google Scholar]
- 89.Bailey MM, Wang L, Bode CJ, Mitchell KE, Detamore MS. A comparison of human umbilical cord matrix stem cells and temporomandibular joint condylar chondrocytes for tissue engineering temporomandibular joint condylar cartilage. Tissue Eng. 2007;13:2003–2010. doi: 10.1089/ten.2006.0150. [DOI] [PubMed] [Google Scholar]
- 90.Nicodemus GD, Villanueva I, Bryant SJ. Mechanical stimulation of TMJ condylar chondrocytes encapsulated in PEG hydrogels. J Biomed Mater Res A. 2007;83:323–331. doi: 10.1002/jbm.a.31251. [DOI] [PubMed] [Google Scholar]
- 91.Chen Y, Zhang M, Wang J. Study on the effects of mechanical pressure to the ultrastructure and secretion ability of mandibular condylar chondrocytes. Arch Oral Biol. 2007;52:173–181. doi: 10.1016/j.archoralbio.2006.08.016. [DOI] [PubMed] [Google Scholar]
- 92.Gadjanski I, Vunjak-Novakovic G. Challenges in engineering osteochondral tissue grafts with hierarchical structures. Expert Opin Biol Ther. 2015;15:1583–1599. doi: 10.1517/14712598.2015.1070825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Abukawa H, Terai H, Hannouche D, Vacanti JP, Kaban LB, Troulis MJ. Formation of a mandibular condyle in vitro by tissue engineering. J Oral Maxillofac Surg. 2003;61:94–100. doi: 10.1053/joms.2003.50015. [DOI] [PubMed] [Google Scholar]
- 94.Grayson WL, Fröhlich M, Yeager K, Bhumiratana S, Chan ME, Cannizzaro C, Wan LQ, Liu XS, Guo XE, Vunjak-Novakovic G. Engineering anatomically shaped human bone grafts. Proc Natl Acad Sci. 2010;107:3299–3304. doi: 10.1073/pnas.0905439106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Jiao Y, Wang D, Han WL. Effects of various growth factors on human mandibular condylar cartilage cell proliferation. Zhonghua kou qiang yi xue za zhi= Zhonghua kouqiang yixue zazhi= Chinese journal of stomatology. 2000;35:346–349. [PubMed] [Google Scholar]
- 96.Hadidi P, Eswaramoorthy R, Hu JC, Athanasiou KA. The Self-Assembling Process of Articular Cartilage and Self-Organization in Tissue Engineering. Tissue and Organ Regeneration: Advances in Micro-and Nanotechnology. 2014:369. [Google Scholar]
- 97•.Athanasiou KA, Eswaramoorthy R, Hadidi P, Hu JC. Self-organization and the self-assembling process in tissue engineering. Annu Rev Biomed Eng. 2013;15:115. doi: 10.1146/annurev-bioeng-071812-152423. This review paper explains principles of different scaffold-free approaches that are used in tissue engineering. Self-assembling process as one of the approaches in scaffold-free method has been discussed in detail. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Anderson DE, Athanasiou KA. Passaged goat costal chondrocytes provide a feasible cell source for temporomandibular joint tissue engineering. Ann Biomed Eng. 2008;36:1992–2001. doi: 10.1007/s10439-008-9572-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Johns D, Wong M, Athanasiou K. Clinically relevant cell sources for TMJ disc engineering. J Dent Res. 2008;87:548–552. doi: 10.1177/154405910808700609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Anderson D, Athanasiou K. A comparison of primary and passaged chondrocytes for use in engineering the temporomandibular joint. Arch Oral Biol. 2009;54:138–145. doi: 10.1016/j.archoralbio.2008.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.MacBarb RF, Paschos NK, Abeug R, Makris EA, Hu JC, Athanasiou KA. Passive strain-induced matrix synthesis and organization in shape-specific, cartilaginous neotissues. Tissue Eng Part A. 2014;20:3290–3302. doi: 10.1089/ten.tea.2013.0694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Kalpakci KN, Kim EJ, Athanasiou KA. Assessment of growth factor treatment on fibrochondrocyte and chondrocyte co-cultures for TMJ fibrocartilage engineering. Acta Biomater. 2011;7:1710–1718. doi: 10.1016/j.actbio.2010.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Johns D, Athanasiou K. Growth factor effects on costal chondrocytes for tissue engineering fibrocartilage. Cell Tissue Res. 2008;333:439–447. doi: 10.1007/s00441-008-0652-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Makris EA, Responte DJ, Paschos NK, Hu JC, Athanasiou KA. Developing functional musculoskeletal tissues through hypoxia and lysyl oxidase-induced collagen cross-linking. Proc Natl Acad Sci. 2014;111:E4832–E4841. doi: 10.1073/pnas.1414271111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105•.MacBarb RF, Makris EA, Hu JC, Athanasiou KA. A chondroitinase-ABC and TGF-β1 treatment regimen for enhancing the mechanical properties of tissue-engineered fibrocartilage. Acta Biomater. 2013;9:4626–4634. doi: 10.1016/j.actbio.2012.09.037. Paper presents the effects of bioactive stimulation and cell ratio on mechanical properties improvement of tissue engineered fibrocartilage. It has been shown the application of both Chondroitinase-ABC and TGF-β1 increased the collagen content of treated constructs compared to controls. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.MacBarb RF, Chen AL, Hu JC, Athanasiou KA. Engineering functional anisotropy in fibrocartilage neotissues. Biomater. 2013;34:9980–9989. doi: 10.1016/j.biomaterials.2013.09.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Murphy MK, Arzi B, Prouty SM, Hu JC, Athanasiou KA. Neocartilage integration in temporomandibular joint discs: physical and enzymatic methods. J R Soc Interface. 12:201541075. doi: 10.1098/rsif.2014.1075. 201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108••.Makris EA, MacBarb RF, Paschos NK, Hu JC, Athanasiou KA. Combined use of chondroitinase-ABC, TGF-β1, and collagen crosslinking agent lysyl oxidase to engineer functional neotissues for fibrocartilage repair. Biomater. 2014;35:6787–6796. doi: 10.1016/j.biomaterials.2014.04.083. Paper demonstrates the combined usage of Chondroitinase-ABC, TGF-β1, and collagen crosslinking agent lysyl oxidase improves mechanical and biochmeical properties at higher timepoints. Followed by in vivo study, authors showed robust integration between the engineered and native tissues using mechanical testing and histology. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Sheehy EJ, Mesallati T, Kelly L, Vinardell T, Buckley CT, Kelly DJ. Tissue Engineering Whole Bones Through Endochondral Ossification: Regenerating the Distal Phalanx. Biores Open Access. 2015;4:229–241. doi: 10.1089/biores.2015.0014. [DOI] [PMC free article] [PubMed] [Google Scholar]


