Abstract
Background:
Subacute subdural hematoma (SASDH) is an entity which is yet to capture the popular imagination among the neurosurgeons. Its management is often equated clinically to that of the chronic subdural hematoma (CSDH). However, their neurological deterioration is usually rapid, which seems to align them with acute subdural hematoma (ASDH). We proceed for their epidemiological evaluation. The advantages of a novel “double barrel technique (DbT)” over the conventional burrhole drainage are also presented.
Methods:
This retrospective study was conducted on all the patients having clinical and radiological evidence of SASDH, admitted to a tertiary care referral institute, during the period August 2013 to December 2015. Postoperatively, patients were followed-up for 3–24 months.
Results:
46.87% of the patients belonged to the 35–54 year age group with a male predominance (3.6:1); 68.7% had a history of alcohol abuse, whereas aspirin users were 25%. 87.5% cases were unilateral, 18.75% were hemispheric, and 46.87% were present on the left side. Altered consciousness (100%) followed by headache (37.5%) were the most common presenting clinical features.
Conclusion:
SASDH is an uncommon neurosurgical entity (0.89% of traumatic brain injury cases in our study) and mimics both CSDH as well as ASDH. The true incidence of SASDH may have been underestimated due to its clinical imitation with CSDH. This study in a South Asian nation also provides the epidemiological data of this rare neurosurgical entity. Outcome of surgery is good; our retrospective study confirms that “DbT” is an adequate and safe treatment. However, a better designed, randomized control trial will be needed to reinforce our findings.
Keywords: Alcohol, double barrel technique, neomembrane, subacute subdural hematoma, trivial trauma
BACKGROUND
Subacute subdural hematoma (SASDH) is an entity which is yet to gain popular foothold among treating neurosurgeons; its management is often equated clinically to that of the chronic subdural hematoma (CSDH). However, their neurological deterioration is usually rapid, which seems to align them with acute subdural hematoma (ASDH).[2] Not only in terms of duration or computed tomography (CT) scan findings but pathologically also neomembrane of SASDH differs from that of CSDH, showing marked inflammatory process with T lymphocytes and neutrophil infiltration.[7] Hence, we proceed for the epidemiological evaluation of all the cases of SASDH, admitted to our institution, in the period August 2013 to December 2015. The advantage of the “double barrel technique (DbT)” over the conventional burrhole drainage is presented, along with their clinical follow up.
MATERIALS AND METHODS
A retrospective study was conducted on all patients admitted to the Department of Neurosurgery, Shriram Chandra Bhanja (SCB) Medical College, Cuttack during the period August 2013 to December 2015. All cases of SASDH admitted to this hospital, both unilateral and bilateral, having clinical and radiological evidence, are included in this study. Postoperatively, patients were followed-up for 3–24 months.
The data were collected from the patient proforma [Table 1], case sheets, operation theatre records, and hospital discharge summaries. Diagnosis was confirmed by cranial CT scan. All the patients were operated under general anesthesia.
Table 1.
Proforma

Inclusion criteria
SASDH at any site, e.g., frontal, parietal, hemispheric, or any cause, e.g., traumatic, associated with alcohol and anticoagulants; patients with any preoperative Glasgow coma score (GCS) and any pupillary size; mass effect on CT scan of the brain.
Exclusion criteria
SASDH not large enough to be operated and cases lost to follow-up after surgery.
Proforma
Investigations
After admission, routine investigations along with coagulation tests such as bleeding time (BT), clotting time (CT), total platelet count (TPC), plasma thromboplastin time (PTT), and international normalized ration (INR); liver function tests (LFT), for alcoholics, was done, and then the patients were posted for surgery.
Surgery
The patients were divided into two groups based on the operative procedure employed; Gr-[1] conventional burr hole drainage. Gr-[2] DbT.
In Gr-[1], a burr hole was placed at the frontal or parietal site depending on the localization of the hematoma, based on the preoperative CT scan. Sometimes, two burrholes were needed to drain the liquid hematoma when
Septae/neomembranes could be visualized on the CT scan
A large hemispheric SASDH was present.
Dura was opened after cauterization, and liquid blood was allowed to drain out. Then, warm normal saline irrigation was done. Amount of normal saline needed for irrigation ranged 1–2 L; we used a 20 ml syringe, attached to the infant feeding tubes (number 6/8), to further drain the hematoma. The septae/neomembranes, when encountered, was dealt with copious irrigation to flush them out. Sometimes, cerebral swelling/compensation ensues as soon as the hematoma is evacuated and reaches the surface in no time. Here, further irrigation with the tubes was avoided, a gelfoam was put over the burr hole, and the residual hematoma was left there under the compensated/swollen brain to become absorbed into the body fluid.
In Gr-[2], similarly, burr holes were placed at the site depending on the localization of the hematoma, based on the preoperative CT scan. The novel strategy was to put two thin infant feeding tubes (5 Fr) in the burrhole [the so called (DbT)]. Both the tubes were put in the same direction, i.e., frontal or parietal, at any given point. The idea is to irrigate the subdural semiliquid hematoma with the 1st tube via a 20 ml syringe and to recapture the effluent through the returning 2nd tube, applying low to moderate suction using another 20 ml syringe [Figure 1a and b]. While taking out the effluents, gravity first removes as much liquid hematoma as possible, and then the “syringing suction” comes into play to evacuate the remaining “lakes.” Thereby, a more thorough and efficient irrigation of the deep corners of the subdural space can be achieved with minimal risk to the cortical bridging venules. When the brain compensates/swells to the surface, the double barrels can be withdrawn, just in time [Figure 1c].
Figure 1.
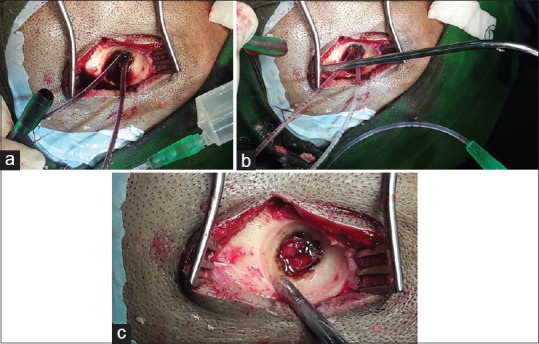
(a) The novel “double barrel technique (DbT).” (b) Appearance after the evacuation. (c) At the completion, with withdrawal of the tubes “just in time” to prevent injuring the cortical veins compressing against the burrhole margin with the tubes in situ
On completion of the irrigation, the head was elevated to 30° and the subdural space was slowly filled with saline so as to eliminate air to the maximum possible extent before securing a single-layered skin closure. A stamp-sized gelfoam piece was used to plug the burrhole opening before closure.
Follow up
Postoperatively, the patient was adequately hydrated, allowed ambulation after 24 h, and discharged after 48 h. Use of anticonvulsants was restricted only to those with a previous seizure history. Clinical status at admission and outcome was assessed in terms of the Glasgow outcome score scale (GOS) at 3 weeks following discharge.
Representational case
HM, a 49-year-old male patient, presented with head injury caused by a fall from trivial trauma (i.e., collision of head with the side walls in his house while walking at night) under the influence of alcohol. He was unconscious when brought to the neurosurgical care. He had a history of intermittent headache with intermittent vomiting for the last 7 days and multiple episodes of such spontaneous falls in the past 15 days.
On examination, he had a GCS of E2V2M5 (9/15), had mild fever, a pulse rate of 58 beats/min, and a blood pressure of 134/86 mmHg. His left pupil was of size 5 mm, sluggishly reacting to light, and the right was 4 mm, normally reacting to the light. There was absence of any ear, nose, and throat bleeding or convulsions. Examination of other systems revealed no abnormality.
Laboratory investigations showed: Coagulation profile (PT = 12 s, INR = 1.05, aPTT = 29.3 s testing being within normal limits, LFT showing mild elevation of liver enzymes, i.e., aspartate transaminase (AST) =55 units/L and alanine transaminase (ALT) =53 units/L, total platelet count (TPC) =2.61 lakhs/mm3.
A plain CT scan of brain [Figure 2] demonstrated a large frontotemporoparietal iso to hyperdense heterogenous crescentic hematoma on the left side, with effacement of the left lateral ventricle and a midline shift of 9 mm. With these findings, SASDH was the final diagnosis.
Figure 2.
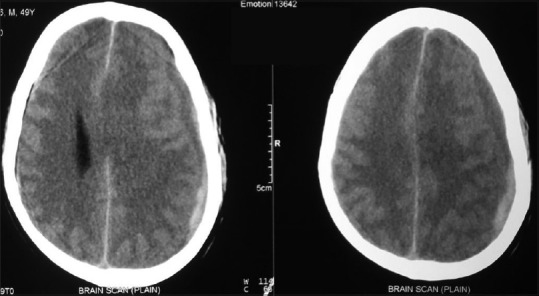
Left frontotemporoparietal iso to hyperdense subacute subdural hematoma on computed tomography scan of the brain
The patient underwent DbT procedure [Figure 1]; around 20 ml of altered blood first came out under pressure, and then, the brain started coming upto the surface. At this juncture, two fine (no. 5) feeding tubes were inserted first on the frontal burr hole side at a time. With the right hand 20 ml syringe, irrigation of the subdural space was started, and the effluents were taken back by the aspirating left hand 20 ml syringe fitted to the 2nd feeding tube. This was continued till the effluents below the surfaced brain became clear. The procedure was repeated on the parietal burrhole. A gelfoam was put at the burrhole site after the conclusion of the irrigation. Wounds were closed without any subgaleal/subdural drain.
RESULTS
The total number of traumatic brain injury cases admitted during the study period is 3613. Out of these cases, 371 presented with clinical and radiological features of ASDH, 32 of SASDH, and 738 of CSDH. Thus, the incidence of SASDH in our study is 0.89%.
46.87% belong to 35–54 year age group [Chart 1] with a male predominance (3.6:1) [Chart 2], 68.7% had a history of alcohol abuse [Chart 3]. Chronic anticoagulant/antiplatelet (Aspirin) uses were risk factors in 25% of cases. 87.5% of cases were unilateral [Chart 4], 18.75% were hemispheric [Chart 5] and 46.87% were present on the left side [Chart 6]. Altered consciousness (100%) followed by headache (37.5%) were m/c presenting clinical features [Chart 7]. Recurrence was seen in 0% [Chart 8]. Complete recovery occurred in 84.37% on postoperative follow-up, with another 12.5% showing partial recovery [Chart 9]. 28.12% of the patients had a midline shift on CT scan of less than 5 mm; the rest had bigger mass effects [Chart 10].
Chart 1.
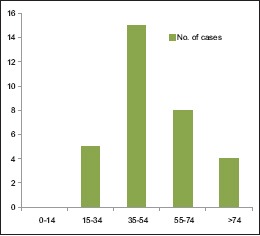
Age distribution of subacute subdural hematoma
Chart 2.
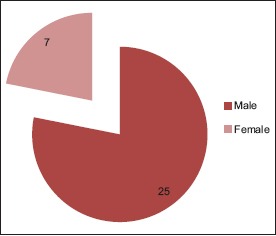
Sex distribution of subacute subdural hematoma
Chart 3.
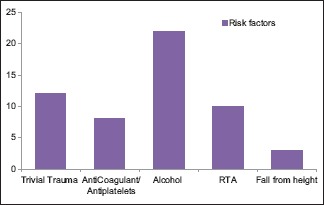
Risk factors of subacute subdural hematoma
Chart 4.
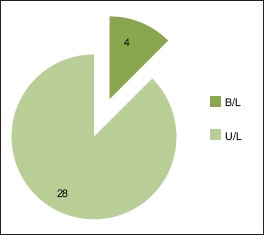
Bilateral versus unilateral occurrence
Chart 5.
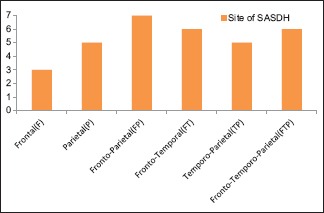
Site of occurrence
Chart 6.
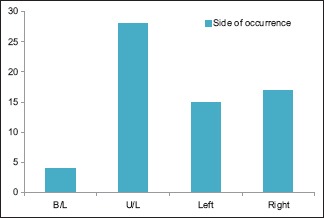
Side of occurrence
Chart 7.
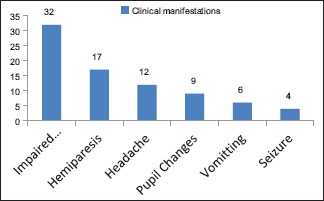
Clinical manifestations of subacute subdural hematoma
Chart 8.
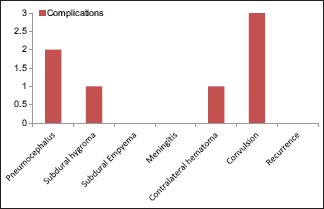
Complications
Chart 9.
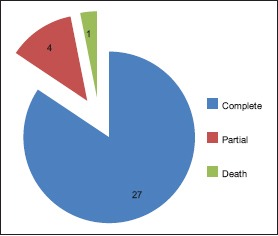
Recovery after surgical treatment
Chart 10.
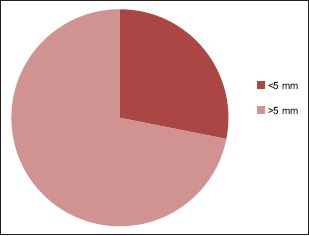
Midline shift (in mm)
Mixed density denotes an acute bleed into a chronic hematoma. Isodense SASDH are the most difficult to diagnose on plain CT scan and may need contrast enhanced CT scan (CECT) or magnetic resonance imaging (MRI) for it.
Seizures were noted in 4 (12.5%), hemiparesis in 17 (53.12%), features of raised intracranial pressure, i.e., headache in 12 (37.5%) of the cases.
Death occurred in only 1 patient (3.12%) who was admitted with low general condition, having electrolyte abnormalities, because of unnecessary treatment for erroneous cerebrovascular accident diagnosis elsewhere. Among the complications, convulsion constitutes the major part (9.37%). Meningitis, subdural empynema (SDE), and recurrence were not reported in our study. Three patients (9.37%) underwent DbT, whereas the remaining underwent the conventional treatment [Chart 11].
Chart 11.
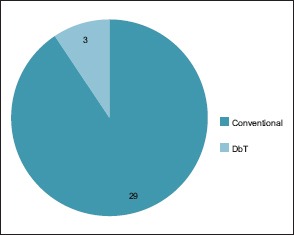
No of cases (Conventional vs. double barrel technique)
All patients had a GOS of 5 at 3 weeks, 3 months, and 6 months following discharge, except the one patient who dies during treatment, after he was operated with the conventional burrhole drainage.
DISCUSSION
A detail account of SASDH is not something that readers come across very often in the published literature; hence, this section is dedicated to elaborate its pathophysiology, histologic evolution, clinical features, imaging characteristics, differential diagnosis, and the surgical procedures.
Subdural hematoma is divided into three types, acute (ASDH), subacute (SASDH), and chronic (CSDH). Duration of onset of <3 days constitute ASDH, 4–21 days SASDH, and >21 days CSDH. These three show separate clincoradiological spectrum. SASDH is similar to CSDH in the clinicoradiological findings, however, preoperatively the findings suggest more of an ASDH picture.
Characteristics of ASDH and SASDH superimpose on each other but with the following glaring differences.[6]
Develops in the elderly population with a history of trivial trauma or minor head injury
There is a relatively long period of clear consciousness; the patients visit the hospital when they are in the subacute stage
The patients have a heavy alcohol abuse history with a high risk of hypertension and diabetes
Brain CT findings, done 5–7 days after the trauma, reveal a mixed density hematoma
Hemorrhage from a cortical artery is occasionally noted as the operative finding; hematoma membrane is absent
The outcome is poor when there are systemic comorbidities.
Pathophysiology
SASDH is the result of subdural effusion in the 1-3 week period. Cerebrospinal fluid (CSF) is considered accountable for the increase in the mass size.[5] In SASDH, there is a collection of partially liquefied clot with resorbing blood products that is surrounded on both sides by a “neomembrane” of organizing granulation tissue. The outermost membrane adheres to the dura and is typically thicker than the inner membrane, which abuts the thin, delicate arachnoid.[1]
In a strict anatomic sense, there is no “true” space called “subdural space;” rather it is a potential space that is created by the rupture of the bridging veins and expansion of the hematoma that gradually separates the layers of the dura and creates the “virtual” subdural space.[4] The space that the SASDH occupies is technically the intradural space. It is a disruption of the dural border cell layer from the deep pachymeninges. Blood in this space provokes an inflammatory reaction, which results in an enveloping membrane surrounding the blood.
With time, SASDH undergoes organization, lysis, and neomembrane formation. Within 2 to 3 days, the initial soft, loosely organized clot of an ASDH becomes organized. Breakdown of blood products and the formation of organizing granulation tissue change the imaging appearance of SASDH.
Brain atrophy may be an important contributing mechanism of SASDH, not due to ageing but due to chronic alcohol abuse.
Histologic evolution
The etiology of SASDH is not fully understood. Traumatic SDE is widely accepted as a preliminary stage in the development of SASDH.[3] Traumatic SDE is a result of arachnoid tearing caused by head injury, and this fluid with or without blood in subdural space facilitates the formation of the so called “outer membrane.” This membrane then forms internal capillaries or sinusoids. These fenestrated blood vessels allow plasma fluid leakage into the subdural space. This phenomenon results in a progressive expansion of the subdural space.[3] Bleeding occurs repeatedly from capillaries with degenerative endothelium, and is accompanied by local hyperfibrinolysis, which is one of the causes for the growth of effusions into SASDH. Fluid ingress driven by osmotic gradient generated by the fibrinolytic products in the hematoma further helps in the genesis of SASDH.
In some cases, repetitive hemorrhages of different ages arising from the friable granulation tissue may be present. In others, liquefaction of the hematoma over time produces serous blood-tinged fluid.
Repeated minor hemorrhages are usually related to bleeding and coagulation disorder (due most commonly to the use of antiplatelet agents such as aspirin and clopidogrel or anticoagulants such as warfarin; or rarely to hemoglobinopathy—sickle cell disorder, hemophilia, protein C, protein S deficiency, factor V Leiden mutation, von willebrand factor deficiency, glucose 6 phosphate dehydrogenase deficiency, complement pathway disorders, leukemia-acute myelogenous leukemia-M3 variety. etc.).
Clinical features
SASDH are common findings at imaging and autopsy. In contrast to ASDH, this shows a distinct bimodal distribution with children and the elderly as the most commonly affected age groups. Clinical symptoms vary from asymptomatic state to loss of consciousness and hemiparesis caused by sudden rehemorrhage into a SASDH. Headache and seizure are other common presentations. Many SASDH resolve spontaneously. In some cases, repeated hemorrhages may cause sudden enlargement with the resultant mass effect. Surgical drainage may be indicated if SASDH is enlarging or it becomes symptomatic.[1]
Imaging
Findings are related to the hematoma age and the presence of encasing membranes. Evolution of an untreated and uncomplicated SASDH follows a very predictable pattern on CT. Density of an extra-axial hematoma decreases approximately 1–2 HU each day.[1] Therefore, an SASDH will become nearly isodense with the underlying cerebral cortex within a few days following trauma.
CT
SASDH are typically crescentic fluid collections that are iso to slightly hyperdense, compared to the underlying cortex on any CT. Medial displacement of the gray–white interface (“buckling”) is often present along with a “dot-like foci of CSF” in the trapped partially effaced sulci underlying the SASDH. Mixed density hemorrhages are common. Bilateral SASDH may be difficult to detect because of their “balanced mass effect.” Sulcal effacement with displaced gray–white matter interface is the typical appearance. CECT scans show that the enhanced cortical veins are displaced medially. The encasing membranes, especially the thicker superficial layer may enhance.[1]
MRI
It can be very helpful in identifying SASDH, especially small lesions that are virtually isodense with underlying brain on CT scan. Signal intensity varies with hematoma age, but is less predictable than on CT, making precise aging of subdural collections more problematic. In general, early SASDH are isointense with cortex on T1 and hypointense on T2, however, gradually become more hyperintense as extracellular methhemoglobin increases. Most late stage SASDH is “T1/T2 bright-bright.” A linear T2 hypointensity representing the encasing membrane that surrounds the SASDH is sometimes present.
FLAIR (Fluid attenuated inversion recovery)
It is the most sensitive sequence for detecting SASDH as the collection is typically hyperintense. Because FLAIR signal varies depending on relative contribution of T1 and T2 effects, early SASDH may initially appear hypointense due to intrinsic T2 shortening. T2 scans are also very sensitive as ASDH show distinct “blooming.”[1]
DWI (Diffusion weighted imaging)
Its signal intensity also varies with hematoma age. It shows a crescentic high intensity area with a low intensity rim closer to the brain surface (“double layer appearance”). The low intensity area corresponds to a “mixture of resolved clots and CSF,” whereas the high intensity area correlates with “solid clot.”[1]
T1 + C (contrast enhanced T1):
These scans demonstrate enhancing, thickened, encasing membranes. The membranes surrounding a SASDH are usually thicker on the dural side of collection. Delayed scans may show gradual filling and increasing hyperintensity of SASDH.
Differential diagnosis
It is most commonly an isodense ASDH; typically seen in extremely anemic and anticoagulated patients
A subdural effusion that follows surgery or meningitis or that occurs as a component of intracranial hypotension can also mimic an SASDH
Subdural hygroma (SDh) is typically isodense/intense with CSF and does not demonstrate enhancing, encapsulating membranes.[1]
Surgical procedures
Craniotomy with membranectomy
Burrhole drainage: Conventional method
Burrhole drainage: “DbT.”
The procedures most commonly resorted to are craniotomy with membranectomy and burrhole drainage, that is, the conventional method.
The “novel strategy” is to put two thin infant feeding tubes (no. 5) in the burrhole [the so called DbT]. Both the tubes are put in the same direction. The idea is to irrigate the subdural semiliquid hematoma with one tube and catch back the effluent through the returning 2nd tube. Thereby, a more thorough and efficient irrigation of the deep corners of the subdural space can be made with minimal risk to the cortical bridging venules. When the brain compensates/swells to the surface, the double barrels can be withdrawn, just in time.
CONCLUSION
SASDH is an uncommon neurosurgical entity (0.89% in our study) which mimics both CSDH as well as ASDH, depending upon the duration, after which the patient comes to neurosurgical attention. Hence, a high degree of suspicion is necessary. Its management is often equated clinically to that of the CSDH
The true incidence of SASDH may have been underestimated for ages due to its clinical imitation by CSDH; the radiological mixed density lesions are frequently confounded with severe anemic patients of ASDH. These two reasons in unison might be the most important factors for the absence of SASDH in neurosurgical textbooks and/or scientific literature at a magnitude it deserves
Outcome of surgery is good; hence, urgent surgical intervention should be contemplated. Our retrospective study confirms that DbT is an adequate and safe treatment. It has definite superiority over the conventional burrhole drainage, where brain surfacing during irrigation is more often expected, leaving deep inaccessible pockets of pathological fluid. A short operating time and ease and simplicity of the procedure are also its advantages
This study in a south Asian nation also provides with the epidemiological data of this rare neurosurgical entity. However a better designed, double blind randomized control trial with more number of participants will be needed to reinforce our findings.
Patient consent
The patient/next of kin/guardian has consented to the submission of the case report for submission to the journal.
Competing interests
We have no financial/nonfinancial (political, personal, religious, ideological, academic, intellectual, commercial, or any other) competing interests.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Footnotes
Contributor Information
Soubhagya R. Tripathy, Email: soubhagya.tripathy@gmail.com.
Pankaj K. Swarnakar, Email: pankaj.swarnakar123@gmail.com.
Sanjib Mishra, Email: drsanjibmishra@gmail.com.
Sudhanshu S. Mishra, Email: drssmishra@gmail.com.
Manmath K. Dhir, Email: drmkmed@gmail.com.
Sanjay K. Behera, Email: drsanjusurgery@gmail.com.
Pratap C. Nath, Email: drpratapnath@rediffmail.com.
Somnath P. Jena, Email: somnath.jena@gmail.com.
Itibrata Mohanta, Email: itibrata@yahoo.com.
Deepak Das, Email: docdpk@gmail.com.
Mani C. Satapathy, Email: drmcsatapathy@gmail.com.
Sitansu K. Rout, Email: sitansurout02@gmail.com.
Bikash R. Behera, Email: drtinku007@gmail.com.
Deepak K. Parida, Email: deepak7077@gmail.com.
Tanushree S. Rath, Email: tanushreesandipta.rath7@gmail.com.
REFERENCES
- 1.Anne G. Osborn’s Brain–Imaging, Pathology and Anatomy. 1st ed. Canada: Amirsys Publishing Inc; 2013. pp. 23–5. [Google Scholar]
- 2.Kpelao E, Beketi KA, Moumouni AK, Doleagbenou A, Ntimon B, Egbohou P, et al. Clinical profile of subdural hematoma: Dangerousness of subacute subdural hematoma. Neurosurg Rev. 2016;39:237–40. doi: 10.1007/s10143-015-0669-4. [DOI] [PubMed] [Google Scholar]
- 3.Kumar P, Kiran U. Management of chronic subdural haematoma: Single burr-hole drainage and irrigation using the technique of “syringing”. Indian J Neurotrauma. 2013;10:105–8. [Google Scholar]
- 4.Marike Z, Muizelaar JP. Clinical pathophysiology of traumatic brain injury. In: Youman’s Neurol Surgery. 5th ed. Philadelphia, PA: Elsevier Saunders; 2004. [Google Scholar]
- 5.Morinaga K1, Matsumoto Y, Hayashi S, Omiya N, Mikami J, Sato H, et al. Subacute subdural hematoma: Findings in CT, MRI and operations & review of onset mechanisms. No Shinkei Geka. 1995;23:213–6. [PubMed] [Google Scholar]
- 6.Morinaga K, Matsumoto Y, Hayashi S, Omiya N, Mikami J, Sato H, et al. Subacute subdural hematoma–Report of 4 cases and a review of literature. No To Shinkei. 1990;42:131–6. [PubMed] [Google Scholar]
- 7.Tao Z, Ding S, Hu M, Li J. Delayed hypertensive process in subacute subdural hematoma. J Craniofac Surg. 2015;26:e443–5. doi: 10.1097/SCS.0000000000001889. [DOI] [PubMed] [Google Scholar]


