Abstract
Eukaryotic cells display an asymmetric distribution of cellular compartments relying on their adhesion and the underlying anisotropy of the actin and microtubule cytoskeleton. Studies using a minimal cell culture system based on confined adhesion on micropatterns have illustrated that trafficking compartments are well organized at the single cell level in response to the geometry of cellular adhesion cues. Expanding our analysis on cellular uptake processes, we have found that cellular adhesion additionally defines the topology of endocytosis and signaling. During endocytosis, transferrin (Tfn) and epidermal growth factor (EGF) concentrate at distinct cellular sites in micropatterned cells. Tfn is enriched in adhesive sites during uptake, whereas EGF endocytosis is restricted to the dorsal cellular surface. This unexpected dorsal/ventral asymmetry is regulated by uptake mechanisms and actin dynamics. Interestingly, restricted EGF uptake leads to asymmetry of EGF receptor activation that is required to sustain downstream signaling. Based on our results, we propose that differential sorting begins at the plasma membrane leading to spatially distinct intracellular trafficking routes that are well defined in space. We speculate that the intracellular positioning of trafficking compartments sustains an important coupling between the endocytic and signaling systems that allows cells to sense their environment.
Keywords: spatial distribution, clathrin-dependent endocytosis, clathrin-independent endocytosis, intracellular trafficking fate, micropatterned cells, internal cell organization, myosin 2, actin
Introduction
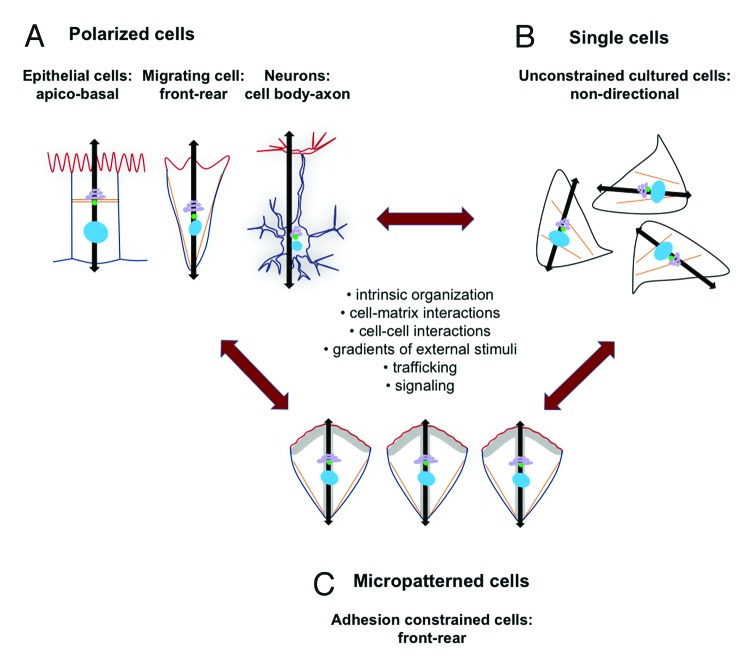
Cellular polarity is instrumental for normal cell function and tissue homeostasis. It is defined by an asymmetrical distribution of cellular constituents along a polarity axis. Many mechanisms have been implicated in the establishment of cell polarity.1,2 Important roles are played by cell-matrix as well as cell-cell interactions, gradients of external stimuli and underlying signal transduction cascades. Additionally, intracellular trafficking events have been implicated in cell polarity establishment and maintenance. The polarized localization of receptors and downstream signaling components arise from the targeted delivery and recycling of vesicles toward spatial clues. For instance, it has been shown that the small GTPase Rab5 is required for apico-basal polarity maintenance in Drosophila.3 Another example is the preferential delivery of secretory vesicles toward the leading edge of migrating fibroblasts.4 Cells in epithelium layers, migrating cells and antigen-presenting cells as well as neurons are mammalian cellular polarity models, since the repetitive presence of spatial cues allows one to define a polarity axis and a conserved cell organization within a given cell type (Fig. 1A). However, because polarity establishment and maintenance is complex, the assignment of the role that different factors play in this process is challenging.
Figure 1. Model of cell polarity and internal cell organization. Cell polarity may be regarded as a gradual change in cell appearance that is regulated by a complex interplay between intrinsic cell organization, cell-matrix interactions, cell-cell interactions, gradients of external stimuli, trafficking pathways and signal transduction cascades. (A) Polarity in cellular organization is observed in epithelial cells (apico-basal polarity), in migrating cells (front-rear polarity) and neurons (cell body- axon polarity). The corresponding polarity axes are represented with black flashes. The nucleus, centrosome and Golgi apparatus are often oriented along this axis. Further, different membrane domains with distinct and mutual exclusive lipid compositions are characteristic for cell polarization (represented schematically with a red and blue line). Nuclei are blue, the Golgi apparatus is purple, the centrosome is green and stress fibers are in orange. (B) Single cells in unconstrained culture condition show an intrinsic cell organization that is however non-directional. Since the internal polarity axes of single cells are not aligned, cells are considered non-polarized. No distinct membrane domains have been observed in unconstrained cultured cells. (C) Micropatterned cells reveal a constant shape due to restrained adhesion that mimic, in vitro, a simplified restriction in space typical for cells in body tissues. On micropatterns, the cell internal polarity axis is aligned along a front-rear orientation.
Then again, generic cultured animal cells display an intrinsic internal organization. Although they are considered non-polarized, many cell types reveal a perinuclear positioning of the Golgi apparatus and the endosomal recycling compartment (ERC). Moreover, secretory vesicles have been observed to concentrate at filopodia. Therefore, it seems that the presence of intrinsic cell organization is basic to all cells (Fig. 1B). How intrinsic cell organization and cell polarity are related is not clear. Importantly, it is not known whether the mechanisms that sustain single cell organization and polarity within a population of cells are similar.
To dissect the mechanisms involved in single cell organization or polarity, we used a minimal cell culture system based on micropatterning (Fig. 1C). Micropatterns reveal a given geometry and impose cells to spread on adhesive and non-adhesive areas. In body tissues, all cells are restrained in their adhesion geometry and space,5 thus cultures on micropatterns mimic restrained cellular adhesion in vitro,6 yet, reduce the complexity. Because generic cell lines are employed that are considered non-polarized, this system allows us to study the mechanisms involved in intrinsic cell organization that might be important for the establishment of cell polarity. Using a population-based mathematical method termed “density estimation,” we have shown previously that trafficking compartments are well organized at the single cell level7 and that cell internal polarity in micropatterned cells is highly reproducible and responds to the geometry of cellular adhesion cues.7 Our recent work reveals that uptake of different cargos is asymmetric.8 We find an unexpected dorsal/ventral asymmetry in both clathrin-dependent and clathrin-independent endocytosis that predefines uptake of transferrin (Tfn) and epidermal growth factor (EGF) at distinct cellular sites.
Single micropatterned cells show asymmetric uptake of different ligands
Endocytosis is the fundamental process for eukaryotic cells to internalize cell surface receptors, parts of the plasma membrane, and soluble molecules from the extracellular medium. Several endocytic mechanisms exist for a molecule to enter the cell.9 Despite the large body of scientific publications on proteins involved in endocytosis, only few studies have addressed whether endocytic events occur randomly distributed around the cell or whether the cellular microenvironment, such as cell-matrix interactions, determines the sites of uptake. It has been observed previously that clathrin-dependent endocytosis is polarized at the cell leading edge in migrating cells.10 Interestingly, accumulations of endocytic sites or “hot spots” of endocytosis are found in a variety of specialized cells such as neurons and hepatocytes11,12
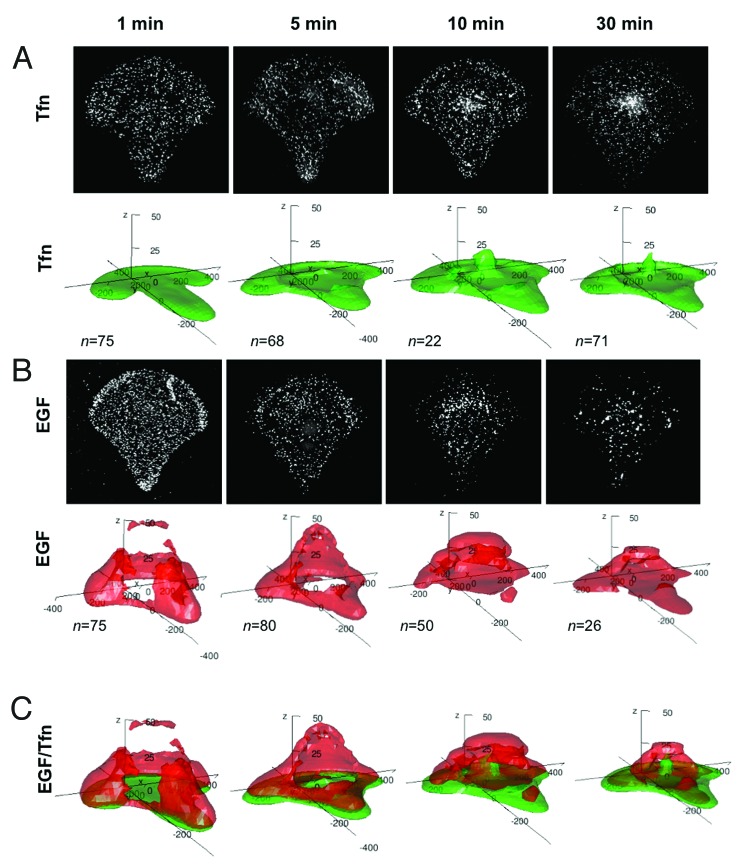
We studied the distribution of Tfn and EGF, two ligands of receptor-mediated endocytosis, one-minute after their addition to cells using “density estimation” that allows quantification of spatial organization. We made the unexpected observation that ligands were not distributed equally all around the cell but concentrated at different sites of the cell during uptake. Tfn was enriched in adhesive areas on the top of micropatterns. In contrast, EGF (at high concentrations) was strongly concentrated on the dorsal surface of the cell and no EGF was located at the cell bottom (Fig. 2). Surprisingly, the corresponding receptors were uniformly distributed over the entire cell surface, thus not explaining the polarization of ligand uptake. This indicated that the presence of receptors at the plasma membrane was not sufficient to initiate endocytosis after cargo binding. We found that asymmetric Tfn uptake depended on specific recruitment of adaptor protein AP2 and clathrin to adhesive areas. Thus, Tfn, and by extension, clathrin-dependent endocytosis was enriched at sites of cell-matrix interactions. Because the actin cytoskeleton is intimately linked to the adhesion geometry of a cell,13 actin dynamics was altered using drugs which disrupt actin organization or gene silencing of actin nucleation regulatory proteins such as Arp2/3. Under these conditions, ligand uptake was redistributed throughout the entire surface. Additionally, we found that myosin contractility regulated asymmetry in endocytosis, since inhibition of myosin 2 by blebbistatin led to similar results.
Figure 2. Uptake of transferrin (Tfn) and epidermal growth factor (EGF) in micropatterned cells. (A) Maximum intensity projection (MIP) of fluorescently marked Tfn in single crossbow-shaped micropatterned cells at 1 min, 5 min, 10 min and 30 min after ligand addition (upper panel) and corresponding 3D density maps of the 50% probability contour for n cells (lower panel). Density maps represent the smallest regions in which 50% of fluorescent structures are found. (B) MIP of fluorescently marked EGF in single crossbow-shaped micropatterned cells at 1 min, 5 min, 10 min and 30 min after ligand addition (upper panel) and corresponding 3D density maps of the 50% probability contour for n cells (lower panel). Density maps represent the smallest regions in which 50% of fluorescent structures are found. (C) Merged 3D density maps of fluorescently marked Tfn (green) and EGF (red) at different time points. For ease of visualization, the height (Z axis) has been stretched 5-fold.
Together these results showed that single micropatterned cells reveal a dorsal/ventral asymmetry in both clathrin-dependent and clathrin-independent endocytosis and that the topology of endocytosis was regulated by actin organization. We concluded that actin integrates the cell with its extracellular environment using a delicate balance between restriction and propagation of endocytosis.
Is there a functional coupling between entry sites, uptake mechanisms and intracellular trafficking pathways ?
One questing that arises from the observation of asymmetric uptake is whether endocytosis at different sites predisposes the downstream intracellular fate of trafficking. The ligands used, Tfn and EGF, are taken up by constitutive and induced endocytosis, respectively, that follow different intracellular trafficking routes: Tfn is mainly recycled back to the plasma membrane14 and EGF is mainly degraded in lysosomes.15 It has been noted previously, that these two cargos are not found in the same populations of early endosomes.16,17 In agreement, we found that Tfn and EGF pass through different cell volumes during intracellular trafficking as visualized by probabilistic density maps8 (Fig. 2). This indicates that different trafficking routes are spatially distinct within the cellular space and that ligand sorting begins at the plasma membrane. Interestingly, different mechanisms of endocytosis seem to be employed to sort ligands at the plasma membrane: clathrin-dependent uptake preferentially at adhesive surfaces and clathrin-independent at the dorsal cellular side. A functional coupling between different uptake mechanisms and intracellular trafficking pathways has been proposed about ten years ago.18 An emerging picture is that although different ligands can probably be sorted from all given compartments, the majority of ligands will not completely be mixed in the early endocytic compartment but will be separated before. Similarly, it is important to point out that although different ligands are separated during endocytosis, a minority of them will enter into the same endocytic carrier and will reach the same intracellular compartment. For instance, clathrin-dependent endocytosis was enriched above the micropattern but was not restricted to these sites and found on the dorsal surface of the cell, where some clathrin-coated pits co-localized with EGF. Taken together, we propose that different ligands will enter the cell at different sites by distinct “site-specific” mechanisms that will predispose ligands for defined trafficking routes. A minority of ligands will enter at random sites by unspecific mechanisms and will be further sorted during intracellular trafficking. This exemplifies the observed specificity described for several ligands on the one hand, and the adaptability of cellular trafficking pathways on the other hand. To test a functional coupling between entry sites, uptake mechanisms and intracellular trafficking pathways, it will be critical to study the sites of endocytosis of different ligands (such as other growth factors, cytokines, lectins, toxins) and their receptors.
Asymmetric endocytosis of EGF is accompanied by spatial restriction of EGF receptor activation
Different from prokaryotes, for which most chemical messengers traverse the cell membrane and bind directly to receptors in the cytoplasm,19 complex eukaryotes acquire and integrate information during endocytosis that involves membrane-bound structures.20 After signal initiation at the plasma membrane, the signal is maintained, propagated, amplified or modified during the successive steps of intracellular trafficking.21 The concept of the 'signaling endosome' demonstrates this important interconnection.22 Therefore, signal transduction is inseparably linked to trafficking pathways in eukaryotic cells. Despite tight connection and a large body of scientific publications, little is known about how signaling cascades and intracellular trafficking are integrated and coordinated within the topological relationships between the numerous intracellular compartments.
EGF-initiated signal transduction is probably the best-understood intracellular signaling cascade regulating cell proliferation, differentiation, motility and cell death.23 Upon binding to its receptor, EGF is rapidly and efficiently internalized.24 Simultaneously, EGF binding results in phosphorylation of EGF receptor (EGFR) that leads to the assembly and subsequent phosphorylation of distinct downstream scaffold complexes and effectors, activating signaling via the extensively studied mitogen-activated protein kinase (MAPK) pathways25 whose effectors, are found at the plasma membrane as well as on endosomes.26
We asked whether asymmetry in EGF uptake is translated into asymmetry in signal transduction. For this, EGF-induced signal initiation was directly monitored in living cells using a fluorescence resonance energy transfer (FRET)-based probe that monitors phosphorylation of the EGFR due to changes in FRET ratio.27 We found that addition of EGF decreased FRET ratio at the cell periphery at the upper and middle part of the cells but did not change FRET ratio at the central bottom region of the cells. This demonstrated that asymmetry in EGF endocytosis leads to asymmetric EGFR phosphorylation in micropatterned cells. Similar analyses were performed in cells whose actin organization was altered using cytochalasin D. Under this condition that redistributes EGF uptake throughout the entire surface, EGFR stimulation was detected in all parts of the cell. These results directly demonstrated that actin dependent asymmetry in EGF uptake leads to asymmetry in EGFR activation. Additionally, we found that redistribution of EGF uptake throughout the cell reduced downstream signaling, because phosphorylation of downstream MAPK and AKT was decreased when either actin organization was disturbed or myosin 2 was inhibited. Our results reveal that EGF-dependent signal propagation is spatially regulated and suggest that asymmetry in endocytosis / signaling is required to sustain appropriate signal transduction (Fig. 3) by regulating the spatial and temporal distribution of activated receptors along intracellular trafficking. We conclude that the cellular cytoskeleton integrates the cell with its extracellular environment, restricting and propagating endocytosis and signaling. We proposed that the observed heterogeneity of endocytic mechanisms allows cells to sense their environment and respond to stimulations in a polarized fashion.
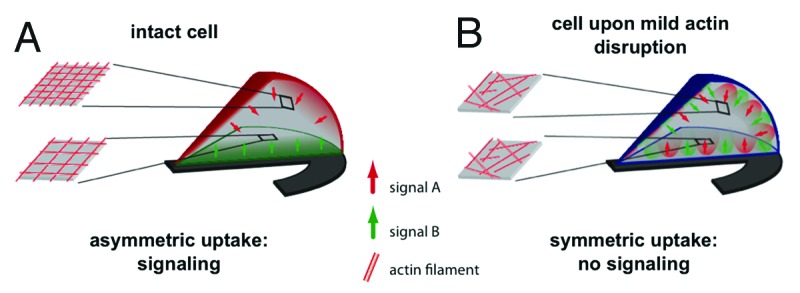
Figure 3. Cellular model of how actin based asymmetry in endocytosis may determine polarized downstream signaling. (A) The “fence and picket” model36 predicts that the actin cortex partitions the plasma membrane into domains with potentially variable molecular properties. As a consequence, differences in the actin cytoskeleton may be present at the dorsal and ventral side of the cell due to the presence or absence of spatial cues (as adhesion surfaces). The actin network defines uptake mechanisms that will be preferentially employed at distinct sites. Different ligands (at different concentrations) show a preference for different uptake mechanisms and thus will be endocytosed asymmetrically. Ligand internalization initiates signaling cascades that will only be sustained when downstream effectors are locally higher concentrated than the total average cell concentration (are polarized). As a consequence, polarized endocytosis of ligands concentrates distribution of downstream signaling gradients, thus propagating signals from the extracellular into the intracellular space and allowing cells to sense their environment. (B) Upon disruption of the actin cytoskeleton effectors downstream of membrane receptors are recruited to the entire surface of the plasma membrane. This recruitment to a larger surface leads to a decrease in the local concentration of downstream effectors, not allowing signal propagation.
Cell organization/polarity as integrator of trafficking and signaling pathways?
Evidence is accumulating that signal transduction cascades are regulated by the cell’s internal organization.20,28-30 Several studies indicate that changes in the steady-state positioning of intracellular compartments impact signaling. For instance, it has been shown that clustering of late endosomes at the cell periphery decreased epidermal growth factor (EGF) receptor degradation and prolonged signaling whereas lysosomal clustering in the cell center accelerated EGF receptor degradation and decreased signaling.31,32 Additionally, cellular nutrient responses have been shown to be regulated by the positioning of lysosomes.33 Thus, a tempting hypothesis is that internal cell organization allows integration of trafficking pathways with signal transduction cascades to resolve the tremendous amount of information that cells need to interpret in a precise spatial and temporal code.
We propose that cell internal organization determines the spatial distribution of signaling pathways, thus propagating signals from the extracellular into the intracellular space. This may provide a cellular mechanism for the maintenance of spatial memory that will allow cells to sense their environment. An important task in the future will be to resolve whether and how the topological relationships between the numerous intracellular compartments connect trafficking pathways and signal transduction.
Cell internal organization seems to be instrumental for normal cell function and tissue homeostasis, because the loss of normal cell organization is a common feature of pathologic conditions, including advanced malignant cells and pathogen-infected cells.34,35 It is not well understood how alterations in inner cell organization alter cell function. Thus, increasing the understanding of molecular mechanisms regulating these processes will be critical for the full understanding of cellular physiology and pathogenesis as well as current approaches of tissues engineering.
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
Acknowledgments
We thank Bruno Latgé and Jean-Philippe Grossier for help with illustrations and discussion and Lena Oesterlin for critical reading of the manuscript. K.S. received funding from the Fondation pour la Recherche Médicale en France and Association pour la Recherche sur le Cancer. This project was further supported by grants from Agence Nationale de la Recherche (#2010 BLAN 122902), the Centre National de la Recherche Scientifique and Institut Curie.
References
- 1.Dow LE, Humbert PO. . Polarity regulators and the control of epithelial architecture, cell migration, and tumorigenesis. Int Rev Cytol 2007; 262:253 - 302; http://dx.doi.org/ 10.1016/S0074-7696(07)62006-3; PMID: 17631191 [DOI] [PubMed] [Google Scholar]
- 2.Nelson WJ. . Adaptation of core mechanisms to generate cell polarity. Nature 2003; 422:766 - 74; http://dx.doi.org/ 10.1038/nature01602; PMID: 12700771 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lu H, Bilder D. . Endocytic control of epithelial polarity and proliferation in Drosophila. Nat Cell Biol 2005; 7:1232 - 9; http://dx.doi.org/ 10.1038/ncb1324; PMID: 16258546 [DOI] [PubMed] [Google Scholar]
- 4.Schmoranzer J, Kreitzer G, Simon SM. . Migrating fibroblasts perform polarized, microtubule-dependent exocytosis towards the leading edge. J Cell Sci 2003; 116:4513 - 9; http://dx.doi.org/ 10.1242/jcs.00748; PMID: 14576345 [DOI] [PubMed] [Google Scholar]
- 5.Gumbiner BM. . Cell adhesion: the molecular basis of tissue architecture and morphogenesis. Cell 1996; 84:345 - 57; http://dx.doi.org/ 10.1016/S0092-8674(00)81279-9; PMID: 8608588 [DOI] [PubMed] [Google Scholar]
- 6.Théry M, Racine V, Piel M, Pépin A, Dimitrov A, Chen Y, Sibarita JB, Bornens M. . Anisotropy of cell adhesive microenvironment governs cell internal organization and orientation of polarity. Proc Natl Acad Sci U S A 2006; 103:19771 - 6; http://dx.doi.org/ 10.1073/pnas.0609267103; PMID: 17179050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schauer K, Duong T, Bleakley K, Bardin S, Bornens M, Goud B. . Probabilistic density maps to study global endomembrane organization. Nat Methods 2010; 7:560 - 6; http://dx.doi.org/ 10.1038/nmeth.1462; PMID: 20512144 [DOI] [PubMed] [Google Scholar]
- 8.Grossier JP, Xouri G, Goud B, Schauer K. . Cell adhesion defines the topology of endocytosis and signaling. EMBO J 2014; 33:35 - 45; http://dx.doi.org/ 10.1002/embj.201385284; PMID: 24366944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Doherty GJ, McMahon HT. . Mechanisms of endocytosis. Annu Rev Biochem 2009; 78:857 - 902; http://dx.doi.org/ 10.1146/annurev.biochem.78.081307.110540; PMID: 19317650 [DOI] [PubMed] [Google Scholar]
- 10.Rappoport JZ, Simon SM. . Real-time analysis of clathrin-mediated endocytosis during cell migration. J Cell Sci 2003; 116:847 - 55; http://dx.doi.org/ 10.1242/jcs.00289; PMID: 12571282 [DOI] [PubMed] [Google Scholar]
- 11.Cao H, Krueger EW, McNiven MA. . Hepatocytes internalize trophic receptors at large endocytic “Hot Spots”. Hepatology 2011; 54:1819 - 29; http://dx.doi.org/ 10.1002/hep.24572; PMID: 21793030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gaffield MA, Tabares L, Betz WJ. . Preferred sites of exocytosis and endocytosis colocalize during high- but not lower-frequency stimulation in mouse motor nerve terminals. J Neurosci 2009; 29:15308 - 16; http://dx.doi.org/ 10.1523/JNEUROSCI.4646-09.2009; PMID: 19955383 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Théry M, Pépin A, Dressaire E, Chen Y, Bornens M. . Cell distribution of stress fibres in response to the geometry of the adhesive environment. Cell Motil Cytoskeleton 2006; 63:341 - 55; http://dx.doi.org/ 10.1002/cm.20126; PMID: 16550544 [DOI] [PubMed] [Google Scholar]
- 14.Watts C. . Rapid endocytosis of the transferrin receptor in the absence of bound transferrin. J Cell Biol 1985; 100:633 - 7; http://dx.doi.org/ 10.1083/jcb.100.2.633; PMID: 2857182 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Beguinot L, Lyall RM, Willingham MC, Pastan I. . Down-regulation of the epidermal growth factor receptor in KB cells is due to receptor internalization and subsequent degradation in lysosomes. Proc Natl Acad Sci U S A 1984; 81:2384 - 8; http://dx.doi.org/ 10.1073/pnas.81.8.2384; PMID: 6326124 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lakadamyali M, Rust MJ, Zhuang X. . Ligands for clathrin-mediated endocytosis are differentially sorted into distinct populations of early endosomes. Cell 2006; 124:997 - 1009; http://dx.doi.org/ 10.1016/j.cell.2005.12.038; PMID: 16530046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Leonard D, Hayakawa A, Lawe D, Lambright D, Bellve KD, Standley C, Lifshitz LM, Fogarty KE, Corvera S. . Sorting of EGF and transferrin at the plasma membrane and by cargo-specific signaling to EEA1-enriched endosomes. J Cell Sci 2008; 121:3445 - 58; http://dx.doi.org/ 10.1242/jcs.031484; PMID: 18827013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Johannes L, Lamaze C. . Clathrin-dependent or not: is it still the question?. Traffic 2002; 3:443 - 51; http://dx.doi.org/ 10.1034/j.1600-0854.2002.30701.x; PMID: 12047552 [DOI] [PubMed] [Google Scholar]
- 19.Schauer K, Gouget B, Carrière M, Labigne A, de Reuse H. . Novel nickel transport mechanism across the bacterial outer membrane energized by the TonB/ExbB/ExbD machinery. Mol Microbiol 2007; 63:1054 - 68; http://dx.doi.org/ 10.1111/j.1365-2958.2006.05578.x; PMID: 17238922 [DOI] [PubMed] [Google Scholar]
- 20.Di Fiore PP, De Camilli P. . Endocytosis and signaling. an inseparable partnership. Cell 2001; 106:1 - 4; http://dx.doi.org/ 10.1016/S0092-8674(01)00428-7; PMID: 11461694 [DOI] [PubMed] [Google Scholar]
- 21.Miaczynska M, Pelkmans L, Zerial M. . Not just a sink: endosomes in control of signal transduction. Curr Opin Cell Biol 2004; 16:400 - 6; http://dx.doi.org/ 10.1016/j.ceb.2004.06.005; PMID: 15261672 [DOI] [PubMed] [Google Scholar]
- 22.Scita G, Di Fiore PP. . The endocytic matrix. Nature 2010; 463:464 - 73; http://dx.doi.org/ 10.1038/nature08910; PMID: 20110990 [DOI] [PubMed] [Google Scholar]
- 23.Roberts PJ, Der CJ. . Targeting the Raf-MEK-ERK mitogen-activated protein kinase cascade for the treatment of cancer. Oncogene 2007; 26:3291 - 310; http://dx.doi.org/ 10.1038/sj.onc.1210422; PMID: 17496923 [DOI] [PubMed] [Google Scholar]
- 24.Haigler H, Ash JF, Singer SJ, Cohen S. . Visualization by fluorescence of the binding and internalization of epidermal growth factor in human carcinoma cells A-431. Proc Natl Acad Sci U S A 1978; 75:3317 - 21; http://dx.doi.org/ 10.1073/pnas.75.7.3317; PMID: 356052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Seger R, Krebs EG. . The MAPK signaling cascade. FASEB J 1995; 9:726 - 35; PMID: 7601337 [PubMed] [Google Scholar]
- 26.Burke P, Schooler K, Wiley HS. . Regulation of epidermal growth factor receptor signaling by endocytosis and intracellular trafficking. Mol Biol Cell 2001; 12:1897 - 910; http://dx.doi.org/ 10.1091/mbc.12.6.1897; PMID: 11408594 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Offterdinger M, Georget V, Girod A, Bastiaens PI. . Imaging phosphorylation dynamics of the epidermal growth factor receptor. J Biol Chem 2004; 279:36972 - 81; http://dx.doi.org/ 10.1074/jbc.M405830200; PMID: 15215236 [DOI] [PubMed] [Google Scholar]
- 28.Cavalli V, Corti M, Gruenberg J. . Endocytosis and signaling cascades: a close encounter. FEBS Lett 2001; 498:190 - 6; http://dx.doi.org/ 10.1016/S0014-5793(01)02484-X; PMID: 11412855 [DOI] [PubMed] [Google Scholar]
- 29.Seto ES, Bellen HJ, Lloyd TE. . When cell biology meets development: endocytic regulation of signaling pathways. Genes Dev 2002; 16:1314 - 36; http://dx.doi.org/ 10.1101/gad.989602; PMID: 12050111 [DOI] [PubMed] [Google Scholar]
- 30.Teis D, Huber LA. . The odd couple: signal transduction and endocytosis. Cell Mol Life Sci 2003; 60:2020 - 33; http://dx.doi.org/ 10.1007/s00018-003-3010-2; PMID: 14618253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hoepfner S, Severin F, Cabezas A, Habermann B, Runge A, Gillooly D, Stenmark H, Zerial M. . Modulation of receptor recycling and degradation by the endosomal kinesin KIF16B. Cell 2005; 121:437 - 50; http://dx.doi.org/ 10.1016/j.cell.2005.02.017; PMID: 15882625 [DOI] [PubMed] [Google Scholar]
- 32.Taub N, Teis D, Ebner HL, Hess MW, Huber LA. . Late endosomal traffic of the epidermal growth factor receptor ensures spatial and temporal fidelity of mitogen-activated protein kinase signaling. Mol Biol Cell 2007; 18:4698 - 710; http://dx.doi.org/ 10.1091/mbc.E07-02-0098; PMID: 17881733 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Korolchuk VI, Saiki S, Lichtenberg M, Siddiqi FH, Roberts EA, Imarisio S, Jahreiss L, Sarkar S, Futter M, Menzies FM, et al. . Lysosomal positioning coordinates cellular nutrient responses. Nat Cell Biol 2011; 13:453 - 60; http://dx.doi.org/ 10.1038/ncb2204; PMID: 21394080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ham H, Sreelatha A, Orth K. . Manipulation of host membranes by bacterial effectors. Nat Rev Microbiol 2011; 9:635 - 46; http://dx.doi.org/ 10.1038/nrmicro2602; PMID: 21765451 [DOI] [PubMed] [Google Scholar]
- 35.Wodarz A, Näthke I. . Cell polarity in development and cancer. Nat Cell Biol 2007; 9:1016 - 24; http://dx.doi.org/ 10.1038/ncb433; PMID: 17762893 [DOI] [PubMed] [Google Scholar]
- 36.Kusumi A, Sako Y. . Cell surface organization by the membrane skeleton. Curr Opin Cell Biol 1996; 8:566 - 74; http://dx.doi.org/ 10.1016/S0955-0674(96)80036-6; PMID: 8791449 [DOI] [PubMed] [Google Scholar]