Abstract
Aim
To evaluate the effect of lung cancer surgery on cardiorespiratory fitness (CRF), and to assess the agreement between the predicted postoperative (ppo) V̇O2peak and actually measured postoperative peak oxygen uptake (V̇O2peak).
Methods
Before and 4–6 weeks after lung cancer surgery, 70 patients (35 women) underwent measurements of pulmonary function and CRF via a cardiopulmonary exercise test. In addition, the 23 non-exercising patients underwent measurements after 6 months. The ppo V̇O2peak calculated from the number of functional segments removed was compared with the actually measured postoperative values of V̇O2peak for accuracy and precision.
Results
After surgery, the V̇O2peak decreased from 23.9±5.8 to 19.2±5.5 mL/kg/min (−19.6±15.7%) (p<0.001). The breathing reserve increased by 5% (p=0.001); the oxygen saturation remained unchanged (p=0.30); the oxygen pulse decreased by −1.9 mL/beat (p<0.001); the haemoglobin concentration decreased by 0.7 g/dL (p=0.001). The oxygen pulse was the strongest predictor for change in V̇O2peak; adjusted linear squared: r2=0.77. Six months after surgery, the V̇O2peak remained unchanged (−3±15%, p=0.27). The ppo V̇O2peak (mL/kg/min) was 18.6±5.4, and the actually measured V̇O2peak was 19.2±5.5 (p=0.24). However, the limits of agreement were large (CI −7.4 to 8.2). The segment method miscalculated the ppo V̇O2peak by more than ±10 and ±20% in 54% and 25% of the patients, respectively.
Conclusions
The reduction in V̇O2peak and lack of improvement 6 months after lung cancer surgery cannot be explained by the loss of functional lung tissue. Predicting postoperative V̇O2peak based on the amount of lung tissue removed is not recommendable due to poor precision.
Trial registration number
Keywords: Lungs, Cancer, Aerobic fitness, Surgery, Exercise physiology
Strengths and limitations of this study.
Following lung cancer surgery, there was a marked decrease in V̇O2peak and lack of improvement 6 months after the surgery.
The decrease was not explained by reduced lung mechanics or lower diffusion capacity in the lungs.
A poor precision was observed between the predicted postoperative and actually measured V̇O2peak based on the number of lung segments resected.
Introduction
Globally, 1.61 million people are diagnosed with lung cancer each year, and the incidence is increasing.1 The complications and mortality rate after surgery for this type of cancer are relatively high compared with other major surgical procedures and depend on the patient's health prior to surgery, and on the extent of the resection.2 Therefore, preoperative risk assessment and the ability to predict postoperative outcomes are of major clinical importance. Cardiorespiratory fitness (CRF) measured as peak oxygen uptake (V̇O2peak) has been reported as being a better predictor of postoperative complications and mortality than the traditionally used pulmonary function variables: forced expiratory volume of air in the 1 s (FEV1) and diffusing capacity of the lung for carbon monoxide (DLCO).3 4 Consequently, current guidelines have recommended exercise testing and defined V̇O2peak cut-off values for risk assessment.5–7 Moreover, the European Respiratory Society/European Society for Thoracic Surgery (ERS/ESTS) guidelines include a modified version of the preoperative Bolliger and Perruchoud8 algorithm, in which V̇O2peak is one of the pivotal measures. This algorithm has been validated and was recently adjusted to lower thresholds,9 thus allowing more patients to undergo surgery. In addition to predicted postoperative (ppo) FEV1 and DLCO, ppo V̇O2peak is included in the algorithm. The ppo V̇O2peak is based on the principle that the amount of resected functional lung tissue corresponds with the drop in V̇O2peak, regardless of whether a pulmonary limitation is present or not. However, V̇O2peak is generally limited by cardiac output and less by pulmonary factors,10 which may question the validity of this measurement by ppo segment method.
In the few studies that have investigated the relationship between ppo V̇O2peak and actually measured postoperative V̇O2peak, the sample size has been small11 12 and the results have been conflicting.12–15 Furthermore, the V̇O2peak cut-off values, and the agreement between ppo and actually measured V̇O2peak values are based on exercise testing using a cycle ergometer instead of a treadmill. Leg discomfort during cycling is an important contribution to exercise termination in patients with lung cancer, rather than cardiopulmonary limitation.12 13 16 17 This may explain the unexpectedly low peak heart rates16 18–20 and high breathing reserves (>40%) reported in previous studies.4 12 13 Thus, when determining the degree of cardiopulmonary reserve and ppo V̇O2peak, additional knowledge is warranted.
The objective of this study was to evaluate the effect of lung cancer surgery on CRF, measured on a treadmill, and to assess the agreement between ppo V̇O2peak and actually measured postoperativeV̇O2peak.
Methods
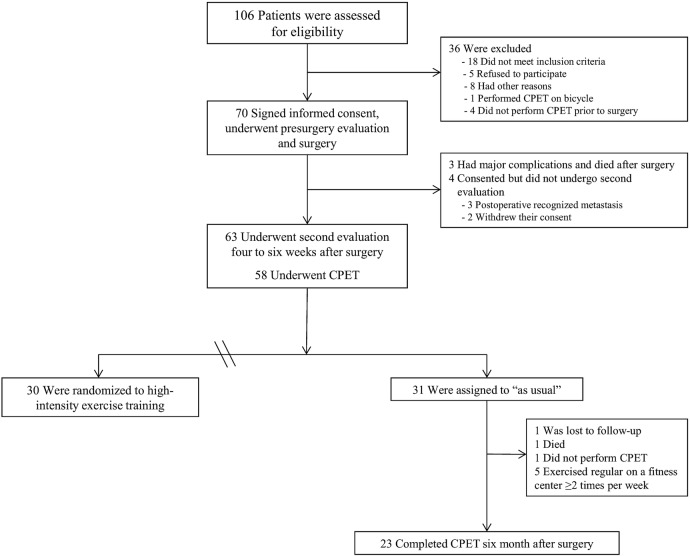
This longitudinal prospective cohort study investigated 70 patients with non-small cell lung cancer (NSCLC) who underwent lung cancer surgery at the Oslo University Hospital or Akershus University Hospital in Norway from November 2010 to September 2012. Eligible patients were aged ≤80 years, had newly diagnosed or suspected NSCLC, and had been accepted for surgery. Patients were not eligible if they were unable to perform a maximal exercise test on a treadmill. The majority of the included patients (n=61) were participants in a randomised controlled trial studying the effect of 20 weeks of exercise training starting 4–6 weeks after surgery.21 The results of the pre-surgery to postsurgery lung function and the cardiopulmonary exercise test (CPET) variables of that trial are included in this paper. In addition, the 6 months results of 23 patients who acted as non-exercising sedentary controls are also included in the current study (figure 1).
Figure 1.
Flow of participants through the study; measurements after exercise training are not included in the data analysis.
The criteria used to determine operability were in accordance with the guidelines of the ERS/ESTS.5 After signing an informed consent form, the patients were enrolled in the study and underwent lung cancer surgery through a muscle-sparing lateral thoracotomy or by video-assisted thoracoscopic surgery (VATS). The study protocol was approved by the Regional Committee for Medical Ethics (REK Sør-Øst B, 2010/2008a) and registered in the ClinicalTrials.gov (NCT01748981).
Measurements
All patients received salbutamol and ipratropium bromide 30 min before the measurements. Among the patients who underwent measurements after 6 months, 33% received four cycles of adjuvant chemotherapy between the second and third measurement. None of the patients underwent organised exercise rehabilitation during the testing period.
Height and body mass were measured to an accuracy of 0.5 cm and 0.1 kg, respectively, with participants wearing light clothes and no shoes; body mass index (BMI) was calculated as body mass/height2 (kg/m2).
Spirometry and DLCO were conducted according to guidelines (Vmax SensorMedics, Yorba Linda, California, USA).22 Maximal voluntary ventilation (MVV) was measured directly by breathing as deep and frequently as possible for 12 s in the standing position.
CPET was performed by uphill walking on a treadmill (Woodway, Würzburg, Germany) until exhaustion. All patients were familiar with treadmill walking before starting the test. Three minutes of warm-up and steady-state measurements were conducted with the treadmill speed individually set between 1.8 and 3.8 km/h, and inclination set at 4% based on the predicted fitness level of the patients. The inclination was then increased every 60 s by 2%, up to 20%. If the participant was still able to continue, the speed was increased by 0.5 km/h until patient reached exhaustion. The test was terminated when the individual could no longer continue, even with encouragement. Gas exchange and ventilatory variables (VE) were measured continuously, breath-by-breath, while breathing into a Hans Rudolph two-way breathing mask (2700 series; Hans Rudolph Inc, Kansas City, Kansas, USA). The mask was connected to a metabolic cart (Vmax SensorMedics, Yorba Linda, California, USA) to assess the oxygen and carbon dioxide content in the expired air to calculate oxygen uptake. HR (heart rate) was recorded each minute using a 12-lead ECG (Cardiosoft, GE Marquette Medical Systems, Milwaukee, Wisconsin, USA).
A capillary blood sample was taken 60 s after test termination (ABL 700 series, Radiometer, Copenhagen, Denmark) for the measurement of haemoglobin (Hb) and blood lactate concentration (La−).
Data handling
The predicted values for FEV1 and DLCO were taken from the European Community for Steel and Coal.23 The exercise variables were reported as a 30 s average and the V̇O2peak was expressed as a percentage of predicted based on the equations of Edvardsen and colleagues.24 The breathing reserve (%) was calculated using the following equation: ([MVV–VEpeak]/MVV)×100. The oxygen pulse was calculated by dividing V̇O2peak (in mL) by the peak heart rate (HRpeak). The actual extent of the operation (ie, wedge resection, lobectomy or pneumonectomy) and number of lung segments removed were recorded after surgery, and the ppo V̇O2peak was calculated using the remaining functional segment technique estimated by bronchoscopy, lung perfusion scan or CT:
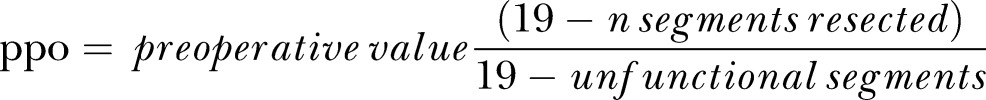 |
For patients who underwent wedge resection, a value of one was used per functional segment.
Statistical analysis
Data were analysed using IBM SPSS Statistical Data Editor, V.21.0. Results are presented as the mean±SD. Differences between pre-surgery and postsurgery variables were analysed using student's paired t test.
Simple linear regression analyses were used to determine the relationship between the change from presurgery to post-surgery values of different CPET variables (independent variables) and change in V̇O2peak (dependent variable), and multiple linear regression analyses were used to study the contribution to the adjusted squared multiple correlation coefficient by including different sets of independent variables. Linear correlations (r2) were reported between actually measured and ppo variables. In addition, linear regression was used to study the adjusted squared linear correlation between the number of functional segments removed and per cent change in V̇O2peak.
The accuracy and precision of ppo versus actually measured values of pulmonary function and V̇O2peak were determined, and the limits of agreement were calculated using a Bland–Altman plot with 95% CIs. p Values ≤0.05 were considered statistically significant.
Results
This study examined 35 women and 35 men undergoing lung cancer surgery (table 1 and figure 1). The majority had adenocarcinoma (44%) and squamous cell carcinoma (39%), and 13 patients (19%) had stage IIIA disease.
Table 1.
Baseline characteristics of the participants
| Characteristics | Participants (n=70) |
|---|---|
| Age, years | |
| Mean | 66.1±9.0 |
| Range | 35–80 |
| BMI, kg/m2 | 24.8±4.8 |
| Health condition | |
| COPD, N (%) | 26 (37) |
| Heart disease, N (%) | 20 (29) |
| Surgery procedure | |
| Wedge/lobectomy/pneumonectomy, n/n/n | 2/56/12 |
| Thoracotomy/VATS, n/n | 59/11 |
| Pulmonary function and physical characteristics | |
| FEV1, % of predicted | 88.4±22.4 |
| DLCO, % of predicted | 80.6±20.9 |
| V̇O2peak, % of predicted | 80.6±16.4 |
| V̇O2peak, mL/kg/min | 23.9±5.8 |
Data are presented as mean±SD or No. (%)
BMI, body mass index, calculated as body mass in kilogram divided by height in metres squared; COPD, chronic obstructive pulmonary disease; Def COPD, FEV1/FVC<70% and FEV1<80% of predicted25; DLCO, carbon monoxide lung diffusion capacity; FEV1, forced expiratory volume after 1 s; VATS, video assisted thoracic surgery; V̇O2peak, peak oxygen uptake.
Physical characteristics before surgery
Pulmonary function and CPET variables before surgery are presented in tables 1 and 2. The breathing reserve was 35.0±14.1%. The CRF of six patients (8%) was limited by their ventilatory capacity, defined as a breathing reserve <15%. At maximal effort during the CPET, the respiratory exchange ratio and blood lactate concentration (La−) were 1.13±0.11 and 5.7±2.3 mmol/L, respectively. Dyspnoea was the most frequent reason for stopping the exercise test (42%), followed by general exhaustion (23%) and leg exhaustion (23%).
Table 2.
Pulmonary function, cardiorespiratory fitness variables and haemoglobin concentration before and 4–6 weeks after surgery
| Before surgery |
After surgery |
Change from baseline (95% CI) |
||||
|---|---|---|---|---|---|---|
| Lobectomy n=58 |
Pulmonectomy n=12 |
Lobectomy n=51 |
Pulmonectomy n=8 |
Lobectomy n=51 |
Pulmonectomy n=8 |
|
| FEV1, L | 2.4±0.8 | 2.6±0.7 | 2.0±0.6 | 1.7±0.3 | −0.4 (−0.5 to −0.3) | −0.9 (−1.3 to −0.5) |
| DLCO, mmol/min/kPa | 6.7±2.1 | 7.2±2.3 | 5.5±1.6 | 4.8±1.2 | −1.3 (−1.6 to −1.0) | −2.7 (−4.1 to −1.3) |
| V̇O2peak, mL/kg/min | 23.4±5.5 | 26.2±6.9 | 19.5±5.4 | 17.8±6.6 | −4.3 (−5.4 to −3.3)* | −9.2 (−14.3 to −4.2)* |
| VEpeak, L/min | 59.8±18.0 | 67.3±16.0 | 47.4±14.0 | 44.4±16.6 | −11.3 (−14.8 to −7.8)* | −21.6 (−32.8 to −10.3)* |
| Breathing reserve, % | 35.3±14.9 | 34.8±11.0 | 41.8±13.0 | 41.0±15.3 | 5.8 (3.0 to 8.6)* | 2.5 (−12.0 to 16.9) |
| SpO2peak, % | 93.4±4.1 | 90.3±6.7 | 93.3±3.3 | 92.9±3.4 | −0.6 (−0.2 to 1.5) | 0.9 (−3.3 to 1.5) |
| Oxygen pulse, mL/beat | 11.8±3.5 | 13.1±3.9 | 10.0±2.8 | 9.9±4.6 | −1.7 (−2.4 to −1.0)* | −2.7 (−4.3 to −1.0)* |
| HRpeak,beat/min | 146.9±21.1 | 152.4±21.3 | 137.4±18.0 | 127.5±18.6 | −9.5 (−12.8 to −6.1)* | −24.9 (−40.2 to −9.5)* |
| Haemoglobin, g/dL | 14.7±1.6 | 14.2±1.8 | 13.9±1.3 | 13.6±2.4 | −0.7 (−1.1 to −0.2)* | −1.1 (−2.4 to −0.3) |
Data are presented as mean±SD; for pulmonary function, n=63 4–6 weeks after surgery.
*Change from baseline is significant at the 0.01 level
DLCO, carbon monoxide lung diffusion capacity; FEV1, forced expiratory volume after 1 s; HRpeak, peak heart rate; SpO2, oxygen saturation; VEpeak, peak minute ventilation; V̇O2peak, peak oxygen uptake.
Changes after surgery
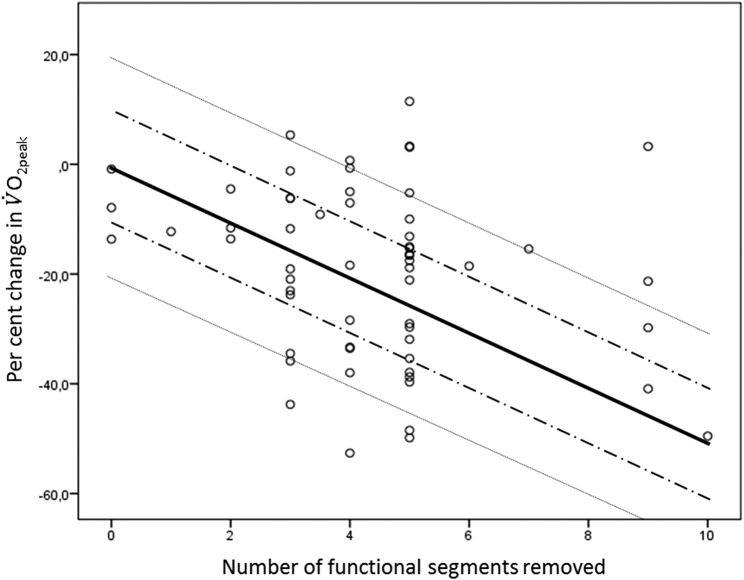
After surgery, 11 patients did not undergo further investigation due to complications, metastases or comorbidities. For the remaining patients (n=59), change in pulmonary function and exercise variables following surgery are presented in table 2. The V̇O2peak decreased by −5.0±4.5 mL/kg/min (−19.6%) (p<0.001). However, nine patients did not exhibit a decrease in V̇O2peak (figure 2). Furthermore, the breathing reserve increased by 5.3±11.1% (p=0.001); the oxygen saturation (SpO2) remained unchanged (p=0.30); the oxygen pulse and Hb concentration decreased by −1.9 mL/beat (p<0.001) and −0.7 g/dL (p=0.001), respectively.
Figure 2.
Per cent change in peak oxygen uptake (V̇O2peak) from before to after surgery for each patient relative to the number of functional lung segments removed. The solid line indicates the per cent change in the calculated postoperative V̇O2peakusing the segment method, ±10% (dashed line) and ±20% (dotted line).
The oxygen pulse was the strongest predictor for change in V̇O2peak; adjusted linear squared r2=0.77. Adding change in FEV1, MVV, breathing reserve, DLCO, peak SpO2, and (Hb) in a multiple regression model resulted in only a modest increase in the predicting value of an adjusted squared r2=0.83, with DLCO as the second contributor.
In the patients who underwent measurements 6 months after surgery (n=23), the FEV1 increased by 7±11% (p=0.002), whereas the DLCO and V̇O2peak remained unchanged compared with the measurement performed 4–6 weeks after surgery, 4±16% (p=0.36) and −3±15% (p=0.27), respectively.
Ppo versus actually measured variables
The ppo V̇O2peak was compared with the actually measured V̇O2peak obtained 4–6 weeks after surgery (table 3). There were no significant differences between the ppo and actually measured values (satisfactory accuracy); however, the limits of agreement were large (poor precision) (figure 3). The linear correlation between ppo and measured V̇O2peak (in mL/kg/min) was r2=0.50 (p<0.001) (for lobecotomy, r2=0.56, p<0.001; and for pneumonectomy, r2=0.15, p=0.187).
Table 3.
Predicted postoperative (ppo) values and actually measured values 4–6 weeks after surgery for pulmonary function (n=63) and peak oxygen uptake (n=59), with limits of agreement and linear correlation (r2)
| Ppo value | Actually measured after surgery | Difference ppo-measured | p Value | Limits of agreement | Linear correlation | |
|---|---|---|---|---|---|---|
| FEV1, % of predicted | 69.5±19.9 | 72.9±17.5 | −3.4±−13.7 | 0.06 | −23.5 to 30.2 | 0.55 |
| DLCO, % of predicted | 63.6±18.9 | 65.4±18.1 | −1.7±−12.3 | 0.27 | −22.3 to 25.8 | 0.61 |
| V̇O2peak, % of predicted | 63.1±16.5 | 65.4±16.9 | −2.3±−13.3 | 0.20 | −23.8 to 28.3 | 0.46 |
| V̇O2peak, mL/kg/min | 18.6±5.4 | 19.2±5.5 | −0.6±−4.1 | 0.24 | −7.4 to 8.7 | 0.50 |
Data are presented as mean±SD.
DLCO, carbon monoxide lung diffusion capacity; FEV1, forced expiratory volume after 1 s; V̇O2peak, peak oxygen uptake.
Figure 3.
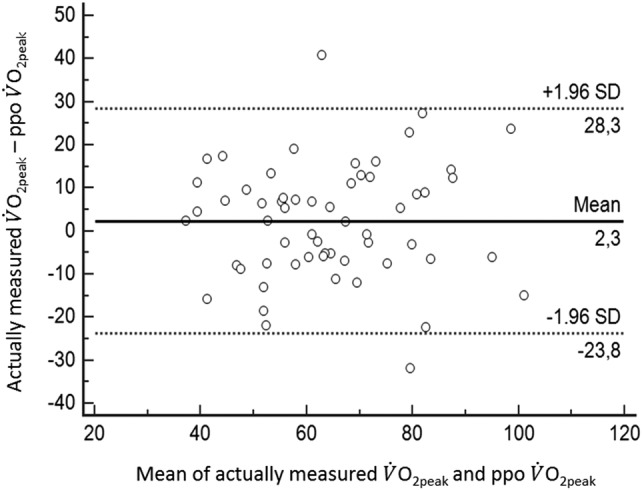
Relationship between the mean of actually measured and predicted postoperative oxygen uptake (ppo V̇O2peak as % of predicted), and the difference between the actually measured V̇O2peakand ppo V̇O2peak with 95% CI (1.96 SD).
Figure 3 demonstrates the poor relationship between the per cent change in actually measured V̇O2peak from before to after surgery, and the number of functional lung segments removed (r2=0.06). The solid black line shows the calculated ppo V̇O2peak using the recommended segment method, and demonstrates the large variance between calculated ppo V̇O2peakand the actually measured values of V̇O2peak. By use of the segment method for predicting postoperative V̇O2peak, 32 patients (54%) were mis-predicted by ≥±10%, and 15 patients (25%) were mispredicted by >±20% (figure 3).
Discussion
The purpose of this study was to evaluate the effect of lung cancer surgery on V̇O2peak as measured on a treadmill, and to assess the agreement between predicted and actually measured V̇O2peak values. There was a clinically important26 and significant reduction in V̇O2peak after surgery, which lasted for more than 6 months. The reduction in V̇O2peak cannot be explained by the number of lung segments removed. Even though the calculation of ppo V̇O2peak was accurate, the precision of the ppo V̇O2peak calculation was poor. Thus, the prediction of postoperative V̇O2peak from the number of lung segments removed should be questioned.
Cardiorespiratory fitness
In the present study, the V̇O2peak measured before surgery was 23.9±5.8 mL/kg/min. Despite the inclusion of a high number of female patients (50%), this is, to our knowledge, the highest reported V̇O2peak in a NSCLC population. In other studies reporting V̇O2peak prior to surgery, the average value has varied between 11 and 20 mL/kg/min.3 11–15 17 18 27 We do not have any indications of the Norwegian NSCLC population as being more fit than other populations. The age, body mass, level of pulmonary function and proportion of comorbidities are comparable with those of other NSCLC populations. Thus, the high V̇O2peak in our population might be explained by the test method. All studies mentioned above used exercise testing on a cycle ergometer, in contrast to our study using a treadmill. When cycling, quadriceps fatigue rather than cardiopulmonary limitation is an important contributor to exercise limitation during CPET in patients with lung cancer.12 13 16 17 Furthermore, the heart rate reserve has been reported to be high when using cycle ergometer4 and consequently, the cardiopulmonary response to the increasing work rate may not have been fully challenged because of leg discomfort. These arguments are reflected in the significantly higher peak heart rate observed in the present study compared with other studies.12 16 18–20 In addition, leg fatigue was only reported in 23% of the patients, which is considerably lower than that reported previously during cycling.12 13 16 17 Uphill walking is a more functional and dynamic exercise mode compared with cycling and generates more muscle mass activation, as recruitment of the quadriceps muscle is increased.28 Simultaneously, it generates lower blood pressure and blood lactate accumulation, as well as higher cardiac output, giving a 10–20% higher V̇O2peak.28–30 We, therefore, recommend treadmill testing for preoperative measurement of physical fitness, enabling the patient to walk slowly up an increasingly steep incline until exhaustion. Preoperative measurement of V̇O2peak is recommended as a tool for risk stratification in all guidelines: ERS/ESTS,5 British Thoracic Society,7 and the American College of Chest Physicians.31
Effect of surgery
Four to 6 weeks after surgery, the V̇O2peak decreased by 17% and 34% in patients who underwent lobectomy and pneumonectomy, respectively. Our results are fairly consistent with those of Nezu et al.17 In contrast, Brunelli et al32 found a minimal loss in V̇O2peak (5%) measured 4 weeks after surgery, despite significantly larger decreases in FEV1 and DLCO. However, the V̇O2peak in that study was estimated from a symptom-limited stair-climbing protocol, using a non-validated equation, thus rendering comparison with the present study difficult.
The reduction in V̇O2peak after surgery could not be explained by loss of lung tissue. This was demonstrated by the lacking relationship between the preoperative to postoperative change in FEV1 and MVV, and the change in V̇O2peak and in addition, by a rather unexpected increase in breathing reserve, defined as a difference between MVV and peak ventilation of less than 15%.33 In fact, only two patients had their postoperative exercise capacity limited by lung mechanics. Furthermore, there was no change in SpO2 during maximum exercise, even though DLCO at rest decreased significantly after surgery. These results are consistent with those of Hsia et al34, who found only a mild decline in arterial O2 saturation during exercise after pneumonectomy, indicating high functional reserves for diffusion capacity in the lungs during exercise. As cardiac output rises during incremental exercise in healthy participants, a twofold increase in diffusion capacity in the lungs is observed in order to maintain oxygenation,35 indicating a higher diffusion capacity reserve compared with cardiac output. This may explain why the majority of patients undergoing lung resection are able to maintain their SpO2 after surgery, even during maximal exercise.
Unfortunately, we did not measure stroke volume during exercise; however, the oxygen pulse, which yields information on the maximal cardiac stroke volume,36–38 was significantly reduced after surgery. In fact, the oxygen pulse was the strongest predictor for the decrease in V̇O2peak. Normally, a low oxygen pulse reflects cardiac limitation if the patient does not desaturate,39 40 indicating a negative effect of surgery on the cardiac function. To confirm the impact on cardiac limitation, we in retrospect calculated the change in the patients’ stroke volume by estimating the arteriovenous oxygen difference,39 41 and found a 10% reduction in the stroke volume (p<0.001) from before to after surgery (data not shown).
Anaemia is a factor that decreases the oxygen-carrying capacity of blood, thereby affecting V̇O2peak negatively.42 According to the multiple regression analysis, the observed decrease in Hb following surgery was not an important contributor to the decrease in V̇O2peak following surgery. Loss of muscle mass may also reduce V̇O2peak. A previously reported dual energy X-ray absorptiometry scanning revealed a significant postoperative loss of muscle mass in our patients.21 Thus, the negative effect of surgery on cardiac function and muscle mass may have contributed to the postoperative decrease in V̇O2peak while, according to the lacking correlation with lung mechanics and the increase in breathing reserve, the loss of lung tissue seems to be of less importance.
Prolonged sedentariness leads to a reduction in cardiac output, as well as muscle wasting.21 43 This may explain the lack of increase in V̇O2peak 6 months after surgery in our group of non-exercising patients.44 Regular high-intensity exercise training following lung cancer surgery has, on the other hand, recently been shown to reverse these conditions,21 highlighting the importance of exercise rehabilitation in this group of patients.
Predicted postoperative V̇O2peak
The second aim of the current study was to evaluate the agreement between ppo V̇O2peak and actually measured postoperative V̇O2peak values during a maximal treadmill test in patients undergoing lung cancer surgery. Estimation of ppo V̇O2peak from the number of lung segments removed is included in the ERS/ESTS guidelines for lung cancer surgery in order to predict surgical risk and functional outcome.5 A ppo V̇O2peak value <10 mL/kg/min or <35% of predicted is used as cut-off values for ‘high-risk patient’,5 thus stressing the importance of applying an accurate formula. Despite satisfying accuracy in ppo V̇O2peak compared with actually measured V̇O2peak after surgery, we found that the variance was large, indicating poor precision. This is in accordance with the lacking correlation between change in V̇O2peak and the number of resected lung segments. In fact, the ppo V̇O2peak value was miscalculated by more than ±20% in as many as 25% of the patients. The results regarding the agreement between ppo V̇O2peak and actually measured values of V̇O2peak in the present study are consistent with those of Brunelli et al.15 They concluded that the ppo V̇O2peak was largely inaccurate and its use should be cautioned against,15 a statement which is supported by our results.
Conclusions
The significant reduction in V̇O2peak and lack of improvement 6 months after lung cancer surgery cannot be explained by the loss of functional lung tissue, but appears to reflect a decrease in the patients’ cardiac function. Predicting postoperative V̇O2peak based on the amount of lung tissue removed is not recommendable due to poor precision.
Acknowledgments
The authors thank Christian Gartman, Vidar Søyseth (Akershus University Hospital), and Ahmed Rizwan Sadiq (Asker and Bærum Hospital Trust) for patient recruitment; Maria Arnesen, Marte Berget, Birgitte Birkeland and Silje Rustad for their expert lab assistance; and Ingar M Holme for excellent statistical guidance. Personnel at the Departments of Thoracic Surgery at Oslo University Hospital and Akershus University Hospital are acknowledged for their kind cooperation.
Footnotes
Contributors: EE, SAA, FB and OHS were responsible for the design and conduct of the study. EE, the guarantor of the paper, was responsible for patient recruitment, data collection, test-procedures, statistical analysis and drafting of the manuscript. All authors contributed to the interpretation of data, and critically reviewed and approved the final manuscript.
Funding: This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: All authors have completed the Unified Competing Interests form at http://www.icmje.org/coi_disclosure.pdf and declare that none of the authors have any non-financial interests that may be relevant to the submitted work.
Ethics approval: Norwegian Committee for Medical Ethics (REK Sør-Øst B, 2010/2008a).
Provenance and peer review: Not commissioned; internally peer reviewed.
Data sharing statement: No additional data are available.
References
- 1.Ferlay J, Shin HR, Bray F et al. . Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 2010;127:2893–917. 10.1002/ijc.25516 [DOI] [PubMed] [Google Scholar]
- 2.Rostad H, Strand TE, Naalsund A et al. . Lung cancer surgery: the first 60 days. A population-based study. Eur J Cardiothorac Surg 2006;29:824–8. 10.1016/j.ejcts.2006.01.055 [DOI] [PubMed] [Google Scholar]
- 3.Benzo R, Kelley GA, Recchi L et al. . Complications of lung resection and exercise capacity: a meta-analysis. Respir Med 2007;101:1790–7. 10.1016/j.rmed.2007.02.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brunelli A, Belardinelli R, Refai M et al. . Peak oxygen consumption during cardiopulmonary exercise test improves risk stratification in candidates to major lung resection. Chest 2009;135:1260–7. 10.1378/chest.08-2059 [DOI] [PubMed] [Google Scholar]
- 5.Brunelli A, Charloux A, Bolliger CT et al. . ERS/ESTS clinical guidelines on fitness for radical therapy in lung cancer patients (surgery and chemo-radiotherapy). Eur Respir J 2009;34:17–41. 10.1183/09031936.00184308 [DOI] [PubMed] [Google Scholar]
- 6.Colice GL, Shafazand S, Griffin JP et al. . Physiologic evaluation of the patient with lung cancer being considered for resectional surgery: ACCP evidenced-based clinical practice guidelines (2nd edition). Chest 2007;132:161S–77S. 10.1378/chest.07-1359 [DOI] [PubMed] [Google Scholar]
- 7.Lim E, Baldwin D, Beckles M et al. . Guidelines on the radical management of patients with lung cancer. Thorax 2010;65(Suppl 3):iii1–27. 10.1136/thx.2010.145938 [DOI] [PubMed] [Google Scholar]
- 8.Bolliger CT, Perruchoud AP. Functional evaluation of the lung resection candidate. Eur Respir J 1998;11:198–212. 10.1183/09031936.98.11010198 [DOI] [PubMed] [Google Scholar]
- 9.Brunelli A. Algorithm for functional evaluation of lung resection candidates: time for reappraisal? Respiration 2009;78:117–18. 10.1159/000210257 [DOI] [PubMed] [Google Scholar]
- 10.di Prampero PE. Metabolic and circulatory limitations to VO2 max at the whole animal level. J Exp Biol 1985;115:319–31. [DOI] [PubMed] [Google Scholar]
- 11.Bolliger CT, Wyser C, Roser H et al. . Lung scanning and exercise testing for the prediction of postoperative performance in lung resection candidates at increased risk for complications. Chest 1995;108:341–8. 10.1378/chest.108.2.341 [DOI] [PubMed] [Google Scholar]
- 12.Wang JS, Abboud RT, Wang LM. Effect of lung resection on exercise capacity and on carbon monoxide diffusing capacity during exercise. Chest 2006;129:863–72. 10.1378/chest.129.4.863 [DOI] [PubMed] [Google Scholar]
- 13.Bolliger CT, Jordan P, Solèr M et al. . Pulmonary function and exercise capacity after lung resection. Eur Respir J 1996;9:415–21. 10.1183/09031936.96.09030415 [DOI] [PubMed] [Google Scholar]
- 14.Bolliger CT, Gückel C, Engel H et al. . Prediction of functional reserves after lung resection: comparison between quantitative computed tomography, scintigraphy, and anatomy. Respiration 2002;69:482–9. doi:66474 [DOI] [PubMed] [Google Scholar]
- 15.Brunelli A, Pompili C, Refai M et al. . Predicted versus observed peak oxygen consumption after major pulmonary resection. Ann Thorac Surg 2012;94:222–5. 10.1016/j.athoracsur.2012.03.034 [DOI] [PubMed] [Google Scholar]
- 16.Pelletier C, Lapointe L, LeBlanc P. Effects of lung resection on pulmonary function and exercise capacity. Thorax 1990;45:497–502. 10.1136/thx.45.7.497 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nezu K, Kushibe K, Tojo T et al. . Recovery and limitation of exercise capacity after lung resection for lung cancer. Chest 1998;113:1511–16. 10.1378/chest.113.6.1511 [DOI] [PubMed] [Google Scholar]
- 18.Campione A, Terzi A, Bobbio M et al. . Oxygen pulse as a predictor of cardiopulmonary events in lung resection. Asian Cardiovasc Thorac Ann 2010;18:147–52. 10.1177/0218492310361792 [DOI] [PubMed] [Google Scholar]
- 19.Jones LW, Eves ND, Peterson BL et al. . Safety and feasibility of aerobic training on cardiopulmonary function and quality of life in postsurgical nonsmall cell lung cancer patients: a pilot study. Cancer 2008;113:3430–9. 10.1002/cncr.23967 [DOI] [PubMed] [Google Scholar]
- 20.Jones LW, Watson D, Herndon JE et al. . Peak oxygen consumption and long-term all-cause mortality in nonsmall cell lung cancer. Cancer 2010;116:4825–32. 10.1002/cncr.25396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Edvardsen E, Skjønsberg OH, Holme I et al. . High-intensity training following lung cancer surgery: a randomised controlled trial. Thorax 2015;70:244–50. 10.1136/thoraxjnl-2014-205944 [DOI] [PubMed] [Google Scholar]
- 22.Brusasco V, Crapo R, Viegi G. Coming together: the ATS/ERS consensus on clinical pulmonary function testing. Eur Respir J 2005;26:1–2. 10.1183/09031936.05.00034205 [DOI] [PubMed] [Google Scholar]
- 23.Quanjer PH, Tammeling GJ, Cotes JE et al. . Symbols, abbreviations and units. Working Party Standardization of Lung Function Tests, European Community for Steel and Coal. Eur Respir J Suppl 1993;16:85–100. 10.1183/09041950.085s1693 [DOI] [PubMed] [Google Scholar]
- 24.Edvardsen E, Hansen BH, Holme IM et al. . Reference values for cardiorespiratory response and fitness on the treadmill in a 20–85-year-old population. Chest 2013;144:241–8. 10.1378/chest.12-1458 [DOI] [PubMed] [Google Scholar]
- 25.Pauwels RA, Buist AS, Calverley PM et al. . Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. NHLBI/WHO Global Initiative for Chronic Obstructive Lung Disease (GOLD) Workshop summary. Am J Respir Crit Care Med 2001;163:1256–76. 10.1164/ajrccm.163.5.2101039 [DOI] [PubMed] [Google Scholar]
- 26.Kaminsky LA, Arena R, Beckie TM et al. . The importance of cardiorespiratory fitness in the United States: the need for a national registry: a policy statement from the American Heart Association. Circulation 2013;127:652–62. 10.1161/CIR.0b013e31827ee100 [DOI] [PubMed] [Google Scholar]
- 27.Win T, Jackson A, Sharples L et al. . Cardiopulmonary exercise tests and lung cancer surgical outcome. Chest 2005;127:1159–65. 10.1378/chest.127.4.1159 [DOI] [PubMed] [Google Scholar]
- 28.Millet GP, Vleck VE, Bentley DJ. Physiological differences between cycling and running: lessons from triathletes. Sports Med 2009;39:179–206. 10.2165/00007256-200939030-00002 [DOI] [PubMed] [Google Scholar]
- 29.Miyamura M, Honda Y. Oxygen intake and cardiac output during maximal treadmill and bicycle exercise. J Appl Physiol 1972;32:185–8. [PubMed] [Google Scholar]
- 30.Christensen CC, Ryg MS, Edvardsen A et al. . Effect of exercise mode on oxygen uptake and blood gases in COPD patients. Respir Med 2004;98:656–60. 10.1016/j.rmed.2003.12.002 [DOI] [PubMed] [Google Scholar]
- 31.Brunelli A, Kim AW, Berger KI et al. . Physiologic evaluation of the patient with lung cancer being considered for resectional surgery: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013;143:e166S–90S. 10.1378/chest.12-2395 [DOI] [PubMed] [Google Scholar]
- 32.Brunelli A, Xiumé F, Refai M et al. . Evaluation of expiratory volume, diffusion capacity, and exercise tolerance following major lung resection: a prospective follow-up analysis. Chest 2007;131:141–7. 10.1378/chest.06-1345 [DOI] [PubMed] [Google Scholar]
- 33.Palange P, Ward SA, Carlsen KH et al. . Recommendations on the use of exercise testing in clinical practice. Eur Respir J 2007;29:185–209. 10.1183/09031936.00046906 [DOI] [PubMed] [Google Scholar]
- 34.Hsia CC, Ramanathan M, Estrera AS. Recruitment of diffusing capacity with exercise in patients after pneumonectomy. Am Rev Respir Dis 1992;145:811–16. 10.1164/ajrccm/145.4_Pt_1.811 [DOI] [PubMed] [Google Scholar]
- 35.Hsia CC. Recruitment of lung diffusing capacity: update of concept and application. Chest 2002;122:1774–83. 10.1378/chest.122.5.1774 [DOI] [PubMed] [Google Scholar]
- 36.Ridgway ZA, Howell SJ. Cardiopulmonary exercise testing: a review of methods and applications in surgical patients. Eur J Anaesthesiol 2010;27:858–65. 10.1097/EJA.0b013e32833c5b05 [DOI] [PubMed] [Google Scholar]
- 37.Crisafulli A, Piras F, Chiappori P et al. . Estimating stroke volume from oxygen pulse during exercise. Physiol Meas 2007;28:1201–12. 10.1088/0967-3334/28/10/006 [DOI] [PubMed] [Google Scholar]
- 38.Bhambhani Y, Norris S, Bell G. Prediction of stroke volume from oxygen pulse measurements in untrained and trained men. Can J Appl Physiol 1994;19:49–59. 10.1139/h94-003 [DOI] [PubMed] [Google Scholar]
- 39.Wasserman K, Hansen JE, Sue DY et al. . Principles of Exercise Testing and Interpretation including Pathophysiology and Clinical Applications. 5th edn. Philadelphia: Lippincott Williams & Wilkins, 2012. [Google Scholar]
- 40.American Thoracic Society / American College of Chest Physicians. ATS/ACCP Statement on cardiopulmonary exercise testing. Am J Respir Crit Care Med 2003;167:211–77. 10.1164/rccm.167.2.211 [DOI] [PubMed] [Google Scholar]
- 41.Stringer WW, Hansen JE, Wasserman K. Cardiac output estimated noninvasively from oxygen uptake during exercise. J Appl Physiol (1985) 1997;82:908–12. [DOI] [PubMed] [Google Scholar]
- 42.Ekblom B, Goldbarg AN, Gullbring B. Response to exercise after blood loss and reinfusion. J Appl Physiol 1972;33:175–80. [DOI] [PubMed] [Google Scholar]
- 43.Krasnoff J, Painter P. The physiological consequences of bed rest and inactivity. Adv Ren Replace Ther 1999;6:124–32. [DOI] [PubMed] [Google Scholar]
- 44.Handy JR Jr, Asaph JW, Skokan L et al. . What happens to patients undergoing lung cancer surgery? Outcomes and quality of life before and after surgery. Chest 2002;122:21–30. 10.1378/chest.122.1.21 [DOI] [PubMed] [Google Scholar]