Abstract
Stem cells reside in niches that provide signals to maintain self-renewal, and differentiation is viewed as a passive process that depends on loss of access to these signals. Here, we demonstrate that the differentiation of somatic cyst stem cells (CySCs) in the Drosophila testis is actively promoted by PI3K/Tor signaling, as CySCs lacking PI3K/Tor activity cannot differentiate properly. We find that an insulin peptide produced by somatic cells immediately outside of the stem cell niche acts locally to promote somatic differentiation through Insulin-like receptor (InR) activation. These results indicate that there is a local ‘differentiation' niche that upregulates PI3K/Tor signaling in the early daughters of CySCs. Finally, we demonstrate that CySCs secrete the Dilp-binding protein ImpL2, the Drosophila homolog of IGFBP7, into the stem cell niche, which blocks InR activation in CySCs. Thus, we show that somatic cell differentiation is controlled by PI3K/Tor signaling downstream of InR and that the local production of positive and negative InR signals regulates the differentiation niche. These results support a model in which leaving the stem cell niche and initiating differentiation are actively induced by signaling.
KEY WORDS: PI3K, Tor, Insulin, Stem cell differentiation, Drosophila, Testis
Summary: Proper differentiation of somatic cyst stem cells as they move away from their niche requires an increase in PI3K/Tor signaling, downstream of Insulin receptor activation.
INTRODUCTION
Stem cells maintain tissue homeostasis in adult organisms by balancing self-renewal and differentiation. Adult stem cells reside in niches that provide signals and space for self-renewal to occur (Losick et al., 2011). Thus, excess self-renewal is prevented by controlling the amount of self-renewal signals produced by the niche, as well as by limiting the available space at the niche. This results in a competition among stem cells to remain in the niche and access the self-renewal signals that it provides (Klein and Simons, 2011). Much work has focused on the signals and the autonomous requirements for stem cell self-renewal. By contrast, little is known about how the rate of differentiation is regulated.
An underlying assumption in many studies is that differentiation is a ‘default' fate that stem cells adopt when deprived of maintenance cues or space at the niche. This view is informed by studies of germline stem cell (GSC) differentiation in the Drosophila ovary, in which the stem cell self-renewal factor Dpp is required to repress transcription of the differentiation gene bag of marbles (bam) (Losick et al., 2011). Loss of this signal leads to derepression of bam and subsequent differentiation, leading to the idea that differentiation needs to be actively repressed in the stem cells but occurs by default once repression is lost. An alternative view is suggested by work in embryonic stem cells (ESCs), where self-renewal can be maintained by removing differentiation-inducing signals (Ying et al., 2008). However, ESCs represent a transitory and singular state of development that is distinct from adult stem cells, where signaling from the niche maintains self-renewal in the long term. This model is supported by recent work in the Drosophila ovary suggesting that somatic support cells, called escort cells, act as a differentiation niche to promote the timely progression of germ cells through differentiation (Kirilly et al., 2011; Luo et al., 2015; Upadhyay et al., 2016; Wang et al., 2015).
We study differentiation in the Drosophila testis stem cell niche, a tissue that shares many characteristics with the ovary (Losick et al., 2011). In the testis, a physical niche called the hub supports two stem cell populations: GSCs and somatic cyst stem cells (CySCs). GSCs divide with oriented mitosis to give rise to gonialblasts, which ultimately differentiate into spermatids. The CySCs divide to produce postmitotic cyst cells. Each gonialblast is ensheathed by two cyst cells that are essential for the proper progression of the germline to meiosis (Fabrizio et al., 2003; Fairchild et al., 2015; Hardy et al., 1979; Kiger et al., 2000; Schulz et al., 2002; Shields et al., 2014; Tran et al., 2000). CySCs require JAK/STAT signaling for self-renewal, and the hub produces the JAK/STAT pathway ligand Unpaired (Upd) to maintain CySCs (Kiger et al., 2001; Leatherman and Dinardo, 2008; Tulina and Matunis, 2001). Additionally, CySCs require Hedgehog, Hippo, Slit/Robo and MAPK signals in order to remain at the niche and compete for space (Amoyel et al., 2013, 2014, 2016; Issigonis et al., 2009; Michel et al., 2012; Stine et al., 2014). In addition to intercellular signaling, many autonomous factors maintain CySCs, particularly the transcription factor Zfh1, which marks the CySC population (Leatherman and Dinardo, 2008). During cyst cell differentiation Zfh1 expression is lost, while the differentiation marker Eyes absent (Eya) is induced (Fabrizio et al., 2003; Leatherman and Dinardo, 2008). It is not known whether cyst cell differentiation is a regulated process, but it is thought to occur by default in cells that are displaced from the niche and can no longer receive self-renewal signals.
We previously showed that CySC clones in which the PI3K/Tor pathway is hyperactivated differentiate rapidly, leading to loss of these mutant stem cells from the niche (Amoyel et al., 2014). However, it was not known whether this reflected a requirement for PI3K/Tor activity during differentiation. The PI3K/Tor pathway is a major regulator of cellular growth, conserved across evolution (Grewal, 2009; Laplante and Sabatini, 2012; Loewith and Hall, 2011). PI3K is activated by receptor tyrosine kinases and phosphorylates phosphatidylinositol (4,5)-bisphosphate (PIP2) lipids to create phosphatidylinositol (3,4,5)-trisphosphate (PIP3) (Fig. 1A). PIP3 activates the kinase Akt1, leading to increased cellular growth through multiple effectors. One major effector and a separate growth regulator in its own right is Tor; Akt1 inactivates the Tor inhibitor Tsc1/2. Tor in turn acts in two major complexes –Tor complex 1 (TORC1) and TORC2 – to regulate multiple targets that affect all aspects of cellular metabolism. TORC1 and TORC2 are distinguished by having different component subunits and differential sensitivity to the inhibitor rapamycin (Laplante and Sabatini, 2012; Loewith and Hall, 2011). Here, we explore the physiological requirement for PI3K/Tor signaling in CySC differentiation and identify this pathway as a crucial mediator of differentiation in stem cells.
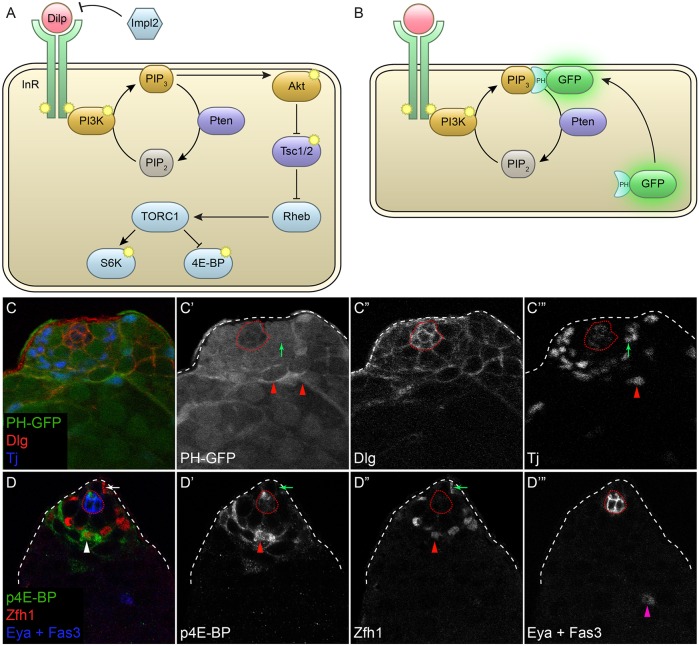
Fig. 1.
PI3K and Tor activity are observed during CySC differentiation. (A) Simplified model of the PI3K/Tor pathway. Here, PI3K is activated by the insulin receptor (InR) tyrosine kinase following binding of a Drosophila insulin-like peptide (Dilp). InR phosphorylates PI3K, which in turn phosphorylates PIP2 lipids to create PIP3. Pten is a lipid phosphatase that catalyzes the reverse reaction. Subsequently, PIP3 activates Akt, which inactivates the Tor inhibitor Tsc1/2. The TORC1 complex is then activated and leads to the phosphorylation of S6K and 4E-BP. The secreted Dilp antagonist ImpL2 blocks Dilps from binding to InR. Phosphorylation events are indicated by the yellow symbols. (B) The PH-GFP reporter of PIP3 consists of GFP fused to the pleckstrin homology (PH) domain of Grp1, which binds PIP3. This GFP fusion is cytoplasmic, but when PIP3 levels are high it translocates to the plasma membrane. (C) The PI3K reporter PH-GFP (green) was predominantly cytoplasmic in CySCs around the hub, indicating low PI3K activity. Arrow in C′,C‴ marks a CySC with low membrane PH-GFP. Membrane-associated PH-GFP was observed as cyst cells begin to differentiate, one further cell diameter away from the hub. Arrowheads in C′,C‴ mark differentiating cyst cells that have upregulated membrane PH-GFP. Tj (blue and C‴) labels CySCs and early cyst cells, Dlg (red and C″) marks somatic cell membranes. (D) The Tor activity reporter p4E-BP (green) was detected at high levels in somatic cells adjacent to stem cells. Arrow in D-D″ marks a Zfh1-positive CySC next to the niche that has low p4E-BP. Arrowhead in D-D″ labels a Zfh1-positive early CySC daughter cell primed for differentiation that is one cell diameter away from the CySCs and that has high levels of p4E-BP. Arrowhead in D‴ marks an Eya-positive differentiating cyst cell. Fas3 (blue and D‴) marks the hub; CySCs are the Zfh1-positive cells (red and D″) that are closest to the hub. Dotted red line outlines the hub.
RESULTS
PI3K and Tor activity are upregulated during somatic differentiation
To determine whether PI3K and Tor are active during cyst cell differentiation, we took advantage of a reporter of PIP3 levels that reflects PI3K activity (Fig. 1B) (Britton et al., 2002) and an antibody against p4E-BP, the phosphorylated form of eIF4-binding protein (Thor – FlyBase), which is a direct target of Tor phosphorylation (Gingras et al., 1999; Miron et al., 2003). We labeled CySCs with Zfh1, a transcription factor that is expressed in ∼40 cells, including the ∼13 CySCs, that contact the hub and their daughters that initiate differentiation (Fig. 1D″, arrow and arrowhead, respectively) (Amoyel et al., 2014; Leatherman and Dinardo, 2008). CySCs are identified as the first row of labeled nuclei around the hub (Fig. 1D-D″, arrow), whereas differentiating daughters are further away and express Eya (Fig. 1D‴, arrowhead). Traffic jam (Tj), a large Maf protein, labels the nuclei of CySCs and early differentiating cells (Li et al., 2003).
The PIP3 reporter consists of GFP fused to the pleckstrin homology (PH) domain of Grp1 (Step – FlyBase), which binds PIP3 (Britton et al., 2002). This GFP fusion is cytoplasmic, but when PIP3 levels are high it translocates to the plasma membrane (Fig. 1B). In control animals, we found that PH-GFP was predominantly cytoplasmic in CySCs surrounding the hub (Fig. 1C′, arrow). However, we observed membrane-associated GFP in the cells immediately distal to the CySCs (Fig. 1C′, arrowheads), suggesting that differentiating cyst cells have higher levels of PIP3 and therefore higher PI3K activity, than CySCs. We confirmed that this reporter was indeed responsive to PI3K activity in the testis by overexpressing the activating subunit of PI3K, Dp110 (Pi3K92E – FlyBase), or an RNAi against the PIP3 phosphatase Pten in somatic cells. In these testes, we observed membrane localization of PH-GFP in all somatic cells, even CySCs (Fig. S1B′,C′, arrow).
Similarly, we show that the Tor activity reporter p4E-BP is detected at low levels in the Zfh1-positive cells closest to the niche (i.e. in CySCs) but is present at high levels in Zfh1-positive cells that are located two cell diameters from the niche (Fig. 1D-D″, arrow and arrowhead, respectively). This upregulation of p4E-BP is not maintained in mature cyst cells. Thus, high PI3K and Tor activity are detected in somatic cells immediately adjacent to the stem cells.
PI3K and Tor activity are required autonomously for cyst cell differentiation
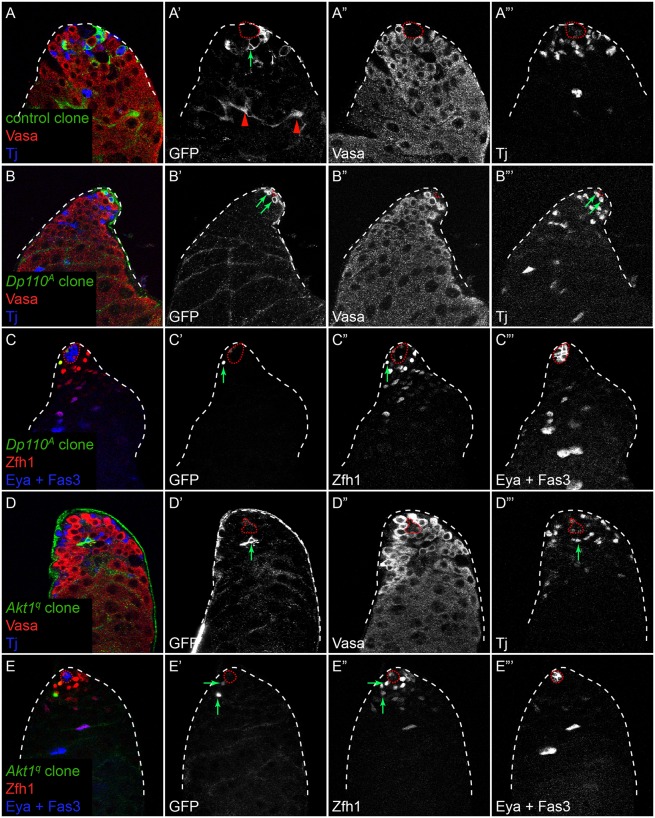
As PI3K/Tor activity is observed early during cyst cell differentiation, we tested whether it is required for CySCs to differentiate. We generated somatic clones mutant for the Drosophila PI3K Dp110 using the null allele Dp110A and for Akt1 using the null allele Akt1q. As these clones are generated by mitotic recombination, they are only produced upon CySC division. Control CySC clones with membrane-targeted GFP were recovered 7 days post clone induction (dpci) and could be identified as stem cells as they extended membrane processes that contacted the hub (Fig. 2A′, arrow). These control clones also gave rise to differentiating offspring that moved away from the niche and ensheathed spermatogonia (n=30 clones in 75 testes examined; Fig. 2A′, arrowheads). Like control clones, Akt1 or Dp110 mutant CySCs that expressed Zfh1 and were close to the niche were recovered at 7 dpci (Fig. 2B′,C′,D′,E′, arrows). However, unlike control clones, we could not find Akt1 or Dp110 mutant cyst cells that ensheathed differentiating germ cells (n=12 Akt1 clones in 64 testes examined and n=8 Dp110 clones in 63 testes examined; Fig. 2B′,D′), and no Akt1 or Dp110 mutant cells were positive for the differentiation marker Eya (Fig. 2C‴,E‴), suggesting that proper somatic cell differentiation was blocked.
Fig. 2.
Dp110 and Akt1 are required autonomously for CySC differentiation. MARCM clones labeled by GFP expression. (A,B,D) Control (A) and Dp110 (B) or Akt1 (D) mutant clones marked with membrane-targeted GFP (green and A′,B′,D′). Vasa (red and A″,B″,D″) labels germ cells and Tj (blue and A‴,B‴,D‴) marks somatic cells. At 7 dpci, control clones (A) contained labeled CySCs contacting the hub (A′, arrow) and differentiated cyst cells ensheathing large germline cysts (A′, arrowheads). By contrast, Dp110 (B′,B‴, arrows) and Akt1 (D′,D‴, arrow) mutant clones contained labeled CySCs contacting the hub but lacked labeled differentiated daughter cells. Note that in B only part of the hub is in the plane. (C,E) Dp110 (C) or Akt1 (E) mutant clones marked with nuclear GFP (green and C′,E′, arrows). Mutant cells expressed the CySC marker Zfh1 (red and C″,E″, arrows) and lacked expression of the differentiation marker Eya (blue and C‴,E‴). The hub is outlined by a red dotted line.
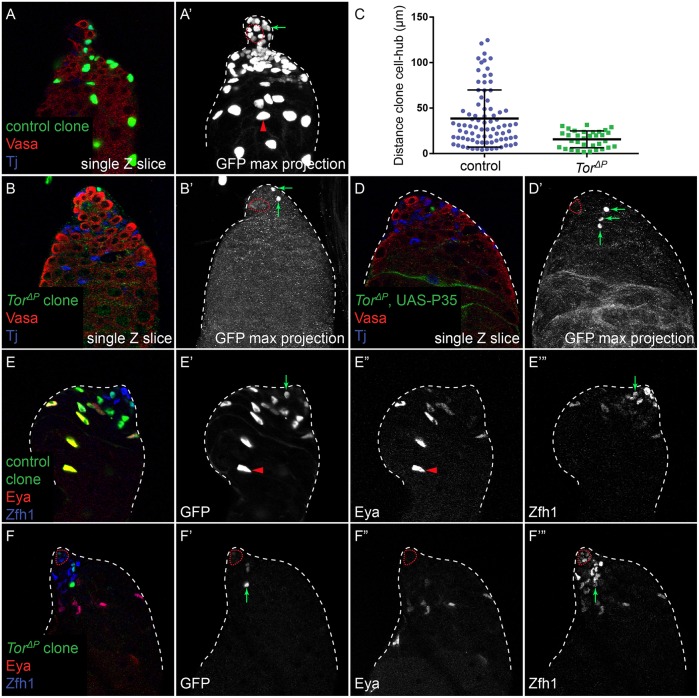
We next examined whether CySCs mutant for Tor exhibited a similar phenotype. We generated control or TorΔP null MARCM clones that expressed nuclear GFP and analyzed these testes at 7 dpci. In testes with control clones, we observed labeled CySCs adjacent to the hub and differentiated cyst cells that have moved away from the hub (Fig. 3A,A′, arrow and arrowhead, respectively). By contrast, Tor mutant clones were only found adjacent to the hub at 7 dpci (Fig. 3B,B′, arrows; quantified in Fig. 3C). We labeled testes carrying marked control or TorΔP mutant clones with the stem cell marker Zfh1 and the differentiation marker Eya to determine whether Tor mutant cells become mature cyst cells. At 7 dpci, we always observed control clones composed of both Zfh1-expressing CySCs and Eya-expressing cyst cells (n=15 clones; Fig. 3E′, arrow and arrowhead, respectively). By contrast, all TorΔP mutant clones examined contained only cells expressing Zfh1 and no cells positive for Eya (n=8 clones, P<0.0001 by Fisher's exact test compared with control clones; Fig. 3F′, arrow), which is similar to what we observed for Akt1 and Dp110 mutant clones labeled with nuclear GFP (Fig. 2C,E).
Fig. 3.
Tor is required autonomously for CySC differentiation. (A) Control nuclear GFP-labeled clone at 7 dpci showing labeled CySCs adjacent to the hub (A′, arrow) and differentiated cyst cells that have moved away from the hub (A′, arrowhead). (A′) A maximum projection of the confocal stack to display all the labeled GFP-positive cells in this sample. (B) TorΔP mutant clone at 7 dpci. All the mutant cells (B′, arrows) in the clone remained close to the hub, as seen in the maximum projection in B′. (C) Quantification of the distance between marked cells in clones and the hub. TorΔP clones remained significantly closer to the hub than control clones (P<1×10−4, Mann–Whitney). (D) Preventing cell death in TorΔP clones at 7 dpci by expressing the caspase inhibitor P35 does not lead to the recovery of differentiated cells (D′, arrows). (E) Control clones contained both Zfh1-expressing CySCs (E′,E‴, arrow) and Eya-expressing differentiated cyst cells (E′,E″, arrowhead). (F) By contrast, TorΔP mutant clones contained only Zfh1-expressing cells (F′,F‴, arrow) and no Eya-expressing cells (F″). The hub is outlined with a red dotted line.
To determine whether Tor mutant cyst cells were differentiating but were not subsequently recovered because they were dying, we prevented apoptosis within the clones by expressing the baculovirus caspase inhibitor P35 (Fig. 3D). These clones were recovered at higher rates than Tor mutant clones alone (45% versus 14%, n=38 and 131 testes, respectively), indicating that Tor mutant cells are eventually lost by apoptosis. However, these clones still did not contain any differentiated cyst cells (Fig. 3D′, arrows). Additionally, as Tor and Akt1 null mutant CySCs had apparent proliferation defects, we tested whether CySCs mutant for an Akt1 (Akt13) or a Tor (TorA948V) hypomorphic allele could differentiate. These mutant CySCs proliferated better, generating more labeled cells within the clone, which could persist up to 7 or 14 days without dying but did not differentiate (Fig. S2A, compare with Fig. 2D; Fig. S2B,C, compare with Fig. 3B). Thus, our data suggest that PI3K/Tor activity is autonomously required for CySCs to differentiate into cyst cells, independently of proliferation or survival.
We note that CySC clones with a proliferation defect can differentiate, as has been shown for CySC clones lacking string (stg), the Drosophila Cdc25 homolog. In fact, CySCs with impaired proliferation cannot be maintained in the niche (Inaba et al., 2011). Therefore, a proliferation defect cannot account for the lack of differentiation of Tor mutant clones.
PI3K acts through TORC1 to regulate the differentiation of somatic cells
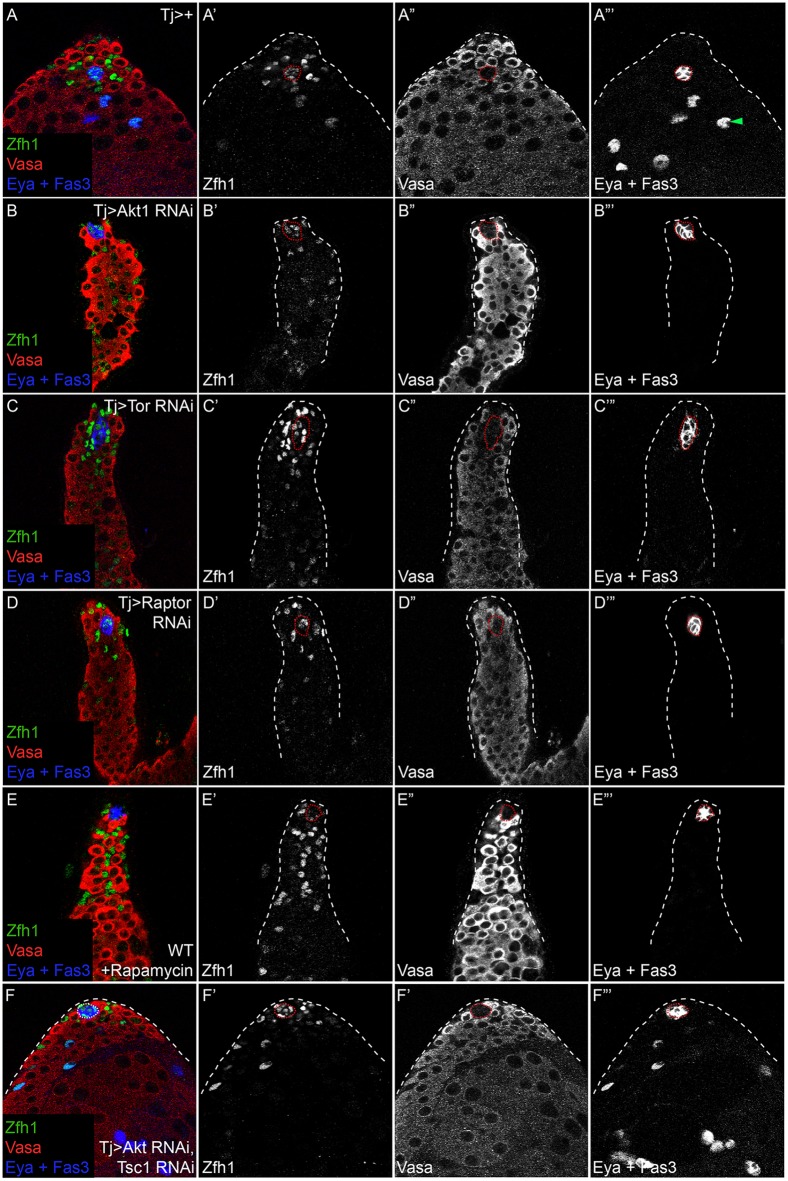
Next, we asked whether decreased PI3K/Tor in the entire somatic lineage affects its progression. We knocked down the PI3K effector Akt1 to determine whether the PI3K pathway is required for normal differentiation. In control testes, Zfh1 is expressed in CySCs and their immediate offspring, concentrated around the hub (n=46 testes; Fig. 4A,A′). As they differentiate, cyst cells lose Zfh1 expression and begin to express Eya (Fig. 4A‴, arrowhead). In testes with somatic depletion of Akt1, we observed somatic cells expressing Zfh1 located many cell diameters from the hub (n=21/21 testes examined; Fig. 4B,B′). Moreover, we detected no or low-level expression of Eya in these samples (Fig. 4B‴).
Fig. 4.
PI3K or TORC1 pathway knockdown in the somatic lineage prevents CySC differentiation. (A) A Control (Tj>+) testis showing Zfh1 (green and A′) in CySCs and early cyst cells near the hub and Eya (blue) in differentiated cyst cells (A‴, arrowhead). Vasa (red and A″) labels the germ line. Germ cells grow larger as they differentiate and move away from the hub. (B) Akt1 knockdown in the somatic lineage led to expansion of the Zfh1-expression domain (green and B′) away from the hub, loss of Eya expression (blue and B‴) and prevented normal germ cell development, as only small germ cells were seen (red and B″). (C) Tor knockdown in somatic cells by RNAi. Many Zfh1-expressing cells (green and C′) were seen far from the hub, whereas Eya expression was absent (blue and C‴). Germ cell differentiation was also disrupted (C″). (D) Knockdown of the TORC1 component Raptor caused a similar phenotype to Akt1 and Tor knockdown. Zfh1 expression (D′) was maintained in cells distant from the hub, whereas Eya was absent (D‴) and germ cell differentiation was blocked (D″). (E) Feeding flies the TORC1 inhibitor rapamycin prevented differentiation, as Eya expression was absent (E‴) whereas Zfh1 (E′) expanded and germ cells (E″) did not differentiate properly. (F) Hyperactivating Tor by knockdown of Tsc1 rescued the loss of differentiation observed when Akt1 is knocked down by RNAi. Note that in contrast to Akt1 knockdown alone (B), concurrent knockdown of Tsc1 leads to robust Eya expression (F‴) and rescue of germ cell differentiation. The hub is outlined with a red dotted line.
We obtained a similar phenotype of impaired somatic differentiation when we knocked down Tor in somatic cells (n=15/16 testes examined; Fig. 4C). Tor acts in two complexes, namely TORC1 and TORC2, that are distinguished by different subunits, although Tor itself is common to both (Laplante and Sabatini, 2012). Moreover, the two complexes differ in their sensitivity to rapamycin (Jacinto et al., 2004). We tested which complex was required in cyst cell differentiation by knocking down the TORC1 member Raptor, and observed a similar phenotype to Tor knockdown alone (n=17/17 testes examined; Fig. 4D). Consistent with this observation, testes from adult males fed the TORC1 inhibitor rapamycin closely resembled testes in which Tor was depleted from the somatic lineage (n=18/18 testes examined; Fig. 4E). We observed ectopic Zfh1-expressing cells away from the hub and a lack of Eya-positive cells (Fig. 4E′,E‴, respectively). Conversely, somatic depletion of Rictor, a TORC2-specific component, did not impact cyst cell differentiation as Eya-positive cyst cells were readily observed (n=13/13 testes appeared similar to control; Fig. S3). These results indicate that Tor acts in the TORC1 complex to promote cyst cell differentiation. Consistent with this, germ cells from testes somatically depleted of Tor or from testes of rapamycin-fed males were maintained in an early, undifferentiated state (Fig. S4), presumably due to the role of CySCs in maintaining GSCs or to the requirement for mature cyst cells to promote germline differentiation (Kiger et al., 2000; Leatherman and Dinardo, 2010; Schulz et al., 2002; Tran et al., 2000).
We ruled out the possibility that the maintenance of Zfh1 expression in somatic cells depleted of Tor signaling results from ectopic activation of Stat92E, a key self-renewal pathway in CySCs. Stabilized Stat92E is a well-established marker of its activation (Flaherty et al., 2010). In wild-type testes, stabilized Stat92E is observed only in GSCs and CySCs in contact with the niche (Fig. S5A), and this pattern is not perturbed in somatic cells depleted of Tor (Fig. S5B).
Finally, we tested whether PI3K activity is linearly upstream of Tor activity in somatic cells of the testis. In testes in which Akt1 was knocked down in somatic cells, we observed a marked decrease in p4E-BP levels, indicating that Tor activity is downstream of PI3K in these cells (Fig. S6B). Moreover, we rescued the defective differentiation of cells with reduced Akt1 by concomitantly knocking down the Tor inhibitor Tsc1 (Fig. 4F). Although Akt1 knockdown alone prevented cyst cell differentiation and blocked Eya expression (n=1/32 testes contained Eya-positive cyst cells; Fig. 4B), Tsc1 knockdown together with Akt1 RNAi led to robust Eya expression and apparently normal progression of both cyst cells and germ cells through differentiation (n=22/23 testes contained Eya-positive cyst cells; Fig. 4F). These results demonstrate that PI3K signaling through TORC1 is essential for differentiation of the cyst lineage in the Drosophila testis.
Somatic cyst cells lacking PI3K and Tor activity remain proliferative
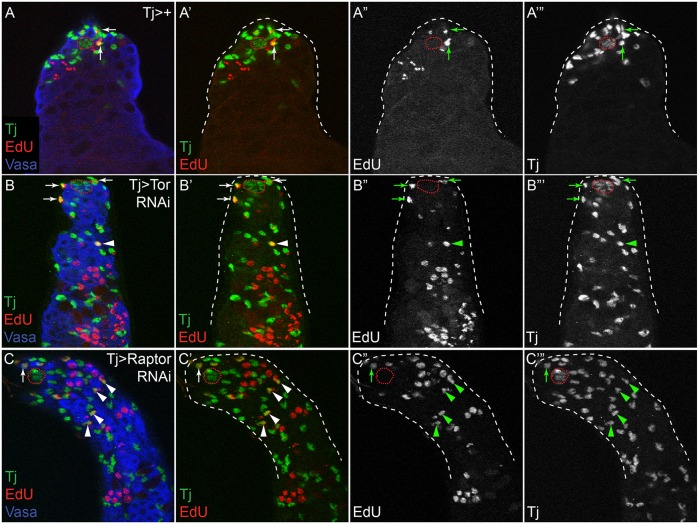
CySCs are the only dividing somatic cells in wild-type testes, and differentiating somatic cells exit the cell cycle and become quiescent. Thus, the ability to divide is a marker of the CySC state. Although CySCs lacking PI3K/Tor proliferate poorly, we showed that clones of hypomorphic mutations in Akt1 and Tor do expand but yet do not differentiate (Fig. 2, Fig. S2). Therefore, we asked whether CySC fate is maintained in lineage-wide knockdowns and assessed proliferative potential as a marker of the CySC state. We examined incorporation of 5-ethynyl-2′-deoxyuridine (EdU), an S-phase marker, in control testes compared with Tor and Raptor knockdown testes. In controls, only somatic cells near the hub incorporated EdU (n=0/34 testes contained any EdU-positive cyst cells located away from the hub; Fig. 5A-A‴, arrows), while germ cells away from the hub replicated synchronously in 2, 4, 8 or 16-cell cysts. In testes with somatic depletion of Tor or the TORC1 component Raptor, we observed EdU-positive somatic stem cells next to the niche (Fig. 5B-C‴, arrows). Importantly, we also detected EdU-positive somatic cells located many cell diameters away from the hub, suggesting that these maintained a stem cell-like fate (n=10/17 and 10/16 testes contained EdU-positive cyst cells located away from the hub, for Tor and Raptor knockdown, respectively; Fig. 5B-C‴, arrowheads). Similarly, EdU-positive somatic cells located many cell diameters from the hub were observed when we knocked down Akt1 in somatic cells (data not shown). These results indicate that somatic cells lacking PI3K/Tor can maintain stem cell-like features.
Fig. 5.
PI3K/Tor pathway knockdown in the somatic lineage blocks quiescence. (A) In a control (Tj>+) testis, only CySCs near the hub underwent S-phase, as revealed by EdU incorporation (A-A‴, arrows). (B,C) Like control testes, EdU incorporation was observed in CySCs next to the niche in testes with somatic knockdown of Tor or Raptor (B-C‴, arrows), but unlike control testes EdU incorporation was also observed in somatic cells far from the hub (B-C‴, arrowheads). Tj is green, EdU is red and Vasa is blue in A-C. The hub is outlined with a red dotted line.
Signaling controls the localization of differentiation
Our results indicate that differentiation is induced by active signaling, suggesting that, as with stem cell self-renewal, there is a ‘niche' for stem cell differentiation (i.e. an environment that promotes a certain cell fate, in this case differentiation) (Kirilly et al., 2011). We sought to identify such a niche by determining how the pattern of p4E-BP is established. Indeed, it is striking that p4E-BP is low in CySCs, but high in the cells immediately adjacent to them, suggesting that the spatial patterning of Tor activity is highly regulated. Since PI3K is upstream of Tor activity (Fig. 4F, Fig. S6B), activation of PI3K is limiting in this context. Removing the Insulin-like receptor (InR) in all somatic cells mimicked PI3K pathway loss of function (Fig. S7A, compare with control in Fig. 4A), which supports the model that PI3K is downstream of the sole Drosophila insulin receptor (InR) (Grewal, 2009). Similar to Dp110 and Akt1 clones, CySC clones mutant for InR were unable to differentiate and remained adjacent to the niche without encysting germ cell cysts (Fig. S7B, arrows, compare with control in Fig. 2A). Moreover, adult males carrying hypomorphic mutations in InR have strongly reduced fertility (Tatar et al., 2001; Ueishi et al., 2009). Therefore, a Drosophila insulin-like peptide (Dilp) is likely to signal to induce CySC differentiation.
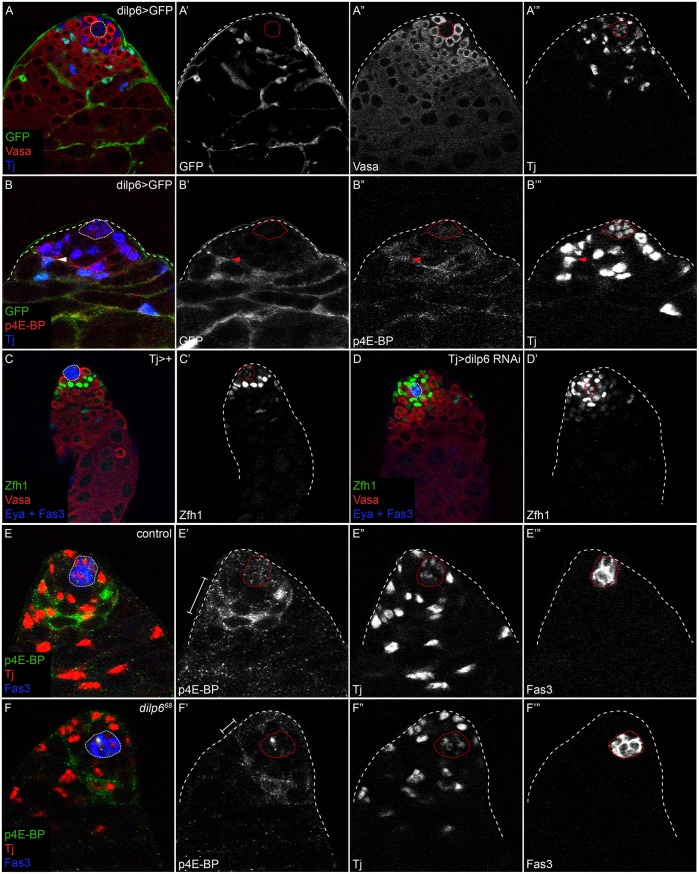
The Drosophila genome encodes seven Dilps, but one of these, Dilp6 (Ilp6 – FlyBase), is functionally and molecularly homologous to insulin-like growth factor (IGF), which acts to promote tissue growth during development, and is therefore a good candidate to be a local inducer of PI3K activity (Brogiolo et al., 2001; Okamoto et al., 2009; Slaidina et al., 2009). We observed that dilp6-gal4 was expressed in differentiating cyst cells (Fig. 6A) and overlapped partly with the domain of p4E-BP (Fig. 6B-B‴, arrowhead), suggesting that it might act as a juxtacrine or paracrine factor. dilp6 mutants displayed a modest but significant accumulation in the number of Zfh1-positive, Eya-negative cells compared with controls [51.9 for dilp641 (n=27) and 51.4 for dilp668 (n=36), compared with 40.1 for control (n=20); P<0.0001 for both versus control, Dunn's multiple comparison test]. This is due to autonomous action within the cyst cell lineage, as knockdown of Dilp6 with Tj-Gal4 led to the same phenotype [compare control in Fig. 6C with Fig. 6D; 51.9 Zfh1-positive, Eya-negative cells in Tj>dilp6 RNAi (n=20) compared with 45 cells in Tj>+ (n=19); P=0.0007, Mann–Whitney test]. Moreover, p4E-BP staining was reduced but not absent in dilp6 mutants (Fig. 6E′,F′, brackets), suggesting that Dilp6 acts partially redundantly with other Dilps to promote differentiation in the testis. Since no other Dilps are expressed near the niche (Wang et al., 2011), it is likely that systemic Dilps are also involved in activating PI3K during CySC differentiation. Indeed, loss of brain-derived Dilps causes defects in germ cell differentiation (Ueishi et al., 2009) and systemic Dilps can affect GSC behavior in both male and female gonads (LaFever and Drummond-Barbosa, 2005; McLeod et al., 2010), indicating that systemic signals reach the gonadal stem cell niches. In turn, if systemic signaling activates PI3K, the pattern of PI3K activation implies that signaling is inhibited in CySCs.
Fig. 6.
Localized Dilp6 expression promotes CySC differentiation. (A) Dilp6-Gal4 driving expression of UAS-GFP in a control testis. GFP (green and A′) is expressed in differentiating cyst cells. Vasa (red and A″) marks germ cells and Tj (blue and A‴) labels somatic cells. (B) Dilp6>GFP (green and B′) expression is partially overlapping with p4E-BP (red and B″), suggesting that Dilp6 could act in a juxtacrine or autocrine manner. Tj (blue and B‴) labels somatic cells. Arrowhead in B-B‴ indicates a Tj-positive somatic cell that expresses the Dilp6 driver and exhibits p4E-BP staining. (C) A control (Tj>+) testis stained with Zfh1 (green and C′). (D) Knockdown of Dilp6 in somatic cells results in an accumulation of Zfh1-expressing cells (green and D′). In C,D, Vasa is red and Eya and Fas3 are blue. More Zfh1-positive cells are seen in a Tj>Dilp6 RNAi testis (D) than in a control (C). (E,F) p4E-BP staining (green and E′,F′) in control (E) or dilp6 mutant (F) testes. Note the reduction in both the width of the p4E-BP domain and the levels of staining (brackets in E′,F′). Tj (red and E″,F″) labels somatic cells and Fas3 (blue and E‴,F‴) marks the hub. The hub is outlined with a dotted line.
ImpL2 inhibits PI3K activity in CySCs to spatially separate self-renewal and differentiation
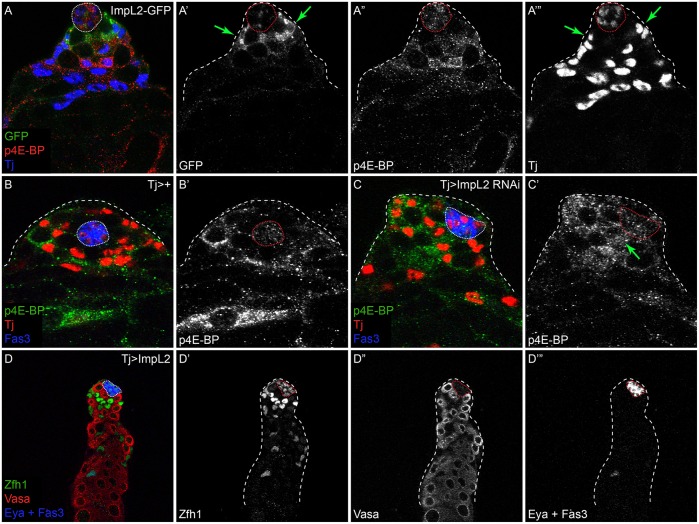
To explore the above possibility, we examined the expression and role of Imaginal morphogenesis protein-late 2 (ImpL2), which is the Drosophila homolog of IGFBP7 and a secreted antagonist of Dilp signaling (Alic et al., 2011; Arquier et al., 2008; Honegger et al., 2008). ImpL2 is upregulated by JAK/STAT signaling and was previously shown to be expressed in cells adjacent to the hub (Terry et al., 2006), suggestive of a role in repressing Dilp activity near the stem cell niche. We confirmed using an enhancer trap that ImpL2 expression is detected specifically in CySCs around the hub (Fig. 7A′, arrows), and an antibody against ImpL2 revealed punctate staining around the hub (Fig. S8A). This staining was lost when ImpL2 was knocked down using Tj-Gal4 and increased when ImpL2 was misexpressed (Fig. S8B,C), indicating that ImpL2 is produced by the somatic lineage.
Fig. 7.
CySCs produce ImpL2 to antagonize local Dilp signaling in stem cells. (A) ImpL2-GFP expression (green) is detected in CySCs contacting the hub (A′,A‴, arrows), in a pattern complementary to p4E-BP (red and A″). Tj (blue and A‴) marks somatic cells. (B,C) p4E-BP levels (green and B′,C′) are altered in testes with somatic depletion of ImpL2. Tj (red) marks somatic cells and Fas3 (blue) labels the hub. p4E-BP is detected in differentiating cyst cells, two cell diameters away from the hub in a control testis (B). However, high levels of p4E-BP are detected in CySCs immediately adjacent to the hub when ImpL2 is knocked down (C′, arrow). (D) Somatic overexpression of ImpL2 results in an accumulation of Zfh1-expressing cells (green and D′) and a delay in the onset of Eya expression (blue and D‴). Vasa labels germ cells (red). The hub is outlined with a dotted line.
Since ImpL2 has been identified as a secreted Dilp antagonist, we tested whether ImpL2 represses PI3K activity in CySCs by analyzing p4E-BP levels as a readout for PI3K/Tor pathway activity in these cells. In testes in which ImpL2 was knocked down, we observed many p4E-BP-positive cells in contact with the hub [compare control in Fig. 7B with Fig. 7C′, arrow; 4.4 p4E-BP-positive cells contacting the hub for Tj>ImpL2 RNAi (n=22) as compared with 1.6 in Tj>+ control (n=15) testes; P<0.0001, Mann–Whitney test]. Conversely, overexpressing ImpL2 led to excess Zfh1-positive cells and a delayed onset of Eya expression, indicating that CySC differentiation was hindered (n=23/25 testes examined; Fig. 7D). These results are consistent with ImpL2 blocking InR activation in somatic stem cells and preventing their differentiation by inhibiting PI3K/Tor pathway activity.
Together, our results suggest that both activating and inhibitory ligands localize the differentiation niche and spatially separate differentiation from self-renewal.
DISCUSSION
We show that PI3K/Tor activity is required for the differentiation of somatic stem cells in the Drosophila testis. Additionally, we identify a ‘differentiation' niche immediately adjacent to the stem cell niche that, through the local production of Dilps, leads to the upregulation of PI3K/Tor activity in early CySC daughters and to their commitment to differentiation. The secretion of ImpL2 by CySCs antagonizes the initiation of differentiation in CySCs by blocking available Dilps in the stem cell niche. As a result, CySCs receive little free Dilp ligand. However, as their daughters move away from the hub, they encounter increasing levels of Dilps and decreasing levels of ImpL2, which leads to the upregulation of PI3K/Tor signaling and proper somatic cell differentiation. The fact that ImpL2 is upregulated by the main self-renewal signal [i.e. JAK/STAT (Terry et al., 2006)] in CySCs leads to a model that accounts for the spatial separation of the stem cell niche and the differentiation niche.
A differentiation niche in the testis
Our results are consistent with a model in which autocrine or paracrine production of Dilp6 by early cyst cells serves as a differentiation niche in the testis, defining where in the tissue upregulation of PI3K/Tor signaling – a prerequisite for differentiation – occurs. This differentiation niche is crucial for somatic development because stem cell markers such as Zfh1 are maintained in the absence of signals such as PI3K/Tor. Notably, JAK/STAT activity is not expanded outside of the niche upon somatic loss of PI3K/Tor signaling, suggesting that differentiation signals play a key role in downregulating stem cell factors. Intriguingly, recent studies in the Drosophila ovary have identified a differentiation niche in this tissue: autocrine Wnt ligands produced by somatic support escort cells regulate escort cell function, proliferation and viability (Luo et al., 2015; Upadhyay et al., 2016; Wang et al., 2015). Taken together, these studies reveal that, at least in Drosophila gonads, there is a defined region immediately adjacent to the stem cell niche where the autocrine production of secreted factors induces the differentiation of somatic cells, which in turn promotes the development of the germ line.
Role of insulin in stem cell niches
Several studies have examined the role of insulin signaling in gonadal stem cells. In both testes and ovaries, systemic Dilps have been shown to affect stem cell behavior (Hsu and Drummond-Barbosa, 2009; Hsu et al., 2008; LaFever and Drummond-Barbosa, 2005; McLeod et al., 2010; Ueishi et al., 2009). In both tissues, nutrition through the regulation of systemic insulin controls the proliferation rate of GSCs (Hsu et al., 2008; McLeod et al., 2010). Our data showing that Akt1, Dp110 or Tor mutant CySC clones proliferate poorly are consistent with these findings and indicate that basal levels of insulin signaling are required for the proliferation and/or survival of both stem cell pools in the testis. Our work also demonstrates that production of the secreted insulin-binding protein ImpL2 by CySCs reduces available Dilps in the stem cell niche, and ImpL2 in the niche milieu should reduce insulin signaling in GSCs and CySCs. Although these data seemingly contradict reports that insulin is required for GSC maintenance (McLeod et al., 2010), we suggest a model in which low constitutive levels of insulin signaling are required for stem cell proliferation and that higher levels are required to induce stem cell differentiation.
PI3K/Tor signaling and the differentiation of stem cells
Prior reports have found that both male and female flies with reduced insulin or Tor activity are sterile (Tatar et al., 2001; Ueishi et al., 2009; Zhang et al., 2006), and the results presented here suggest that this is due at least in part to a lack of somatic cell differentiation. Our results indicate that the IGF homolog Dilp6 plays a local role in CySC differentiation, but acts redundantly with other – presumably systemic – factors, suggesting that both constitutive and nutrient-responsive inputs control CySC differentiation. Indeed, we show that in addition to controlling the proliferation of stem cells, systemic insulin is required for their differentiation, as the poorly proliferative Akt1, Dp110 or Tor mutant CySC clones do not differentiate and eventually die by apoptosis. This combination of reduced proliferation and increased apoptosis might explain why other studies suggest that Tor is required for self-renewal in GSCs; indeed, prior reports indicate that while Tor mutant GSCs are lost, hyperactivation of Tor leads to faster loss of GSCs through differentiation (LaFever et al., 2010; Sun et al., 2010), and recent work indicates that lineage-wide Tor loss blocks the differentiation of GSCs (Sanchez et al., 2016). Our use of hypomorphic alleles enabled a genetic separation of the proliferative effects and differentiation requirements of PI3K and Tor in CySCs. Finally, there is evidence that PI3K/Tor activity promotes the differentiation of stem cells in gonads in mammals, suggesting that our findings might reflect a conserved role of Tor activity in promoting germ cell differentiation, both through autonomous and non-autonomous mechanisms involving somatic support cells (Adhikari et al., 2010; Busada et al., 2015; Hobbs et al., 2010; Reddy et al., 2008).
Moreover, it seems likely that Tor activity may be a more general requirement for the differentiation of many stem cell types, as increased PI3K or Tor has been shown to induce differentiation in many instances. In particular, mouse long-term hematopoietic stem cells are lost to differentiation when the PI3K inhibitor Pten is mutated (Signer et al., 2014; Yilmaz et al., 2006), whereas Drosophila intestinal stem cells differentiate when Tor is hyperactive due to Tsc1/2 complex inactivation (Kapuria et al., 2012; Quan et al., 2013). Moreover, inhibition of Tor activity by rapamycin promotes cellular reprogramming to pluripotency, while cells with increased Tor activity cannot be reprogrammed (Chen et al., 2011; He et al., 2012), suggesting a conserved role for Tor signaling in promoting differentiated states.
MATERIALS AND METHODS
Fly stocks and husbandry
We used the following fly stocks: Oregon R; TorΔP, FRT40A; TorA948V11, FRT40A; FRT82B, Akt1q; FRT82B, Akt13; FRT82B, InRE19; FRT82B, Dp110A; Tj-Gal4; UAS-Akt1 RNAi (TRiP HMS00007); UAS-Tor RNAi (TRiP GL00156); UAS-Tsc1 RNAi (TRiP HMJ21477); UAS-Raptor RNAi (TRiP HMS00124); UAS-Pten RNAi (TRiP JF01859); UAS-Dp110; UAS-P35; UAS-ImpL2 (gift of E. Hafen); UAS-ImpL2-RNAi (VDRC #30931); UAS-Dilp6 RNAi (TRiP JF01348); Dilp6-Gal4 (NP1079-GAL4, DGRC Stock Center, Kyoto, Japan); ImpL2-GFP (ImpL2MI01638-GFSTF.0) (Nagarkar-Jaiswal et al., 2015); and tGPH (PH-GFP) (Britton et al., 2002). Tj-Gal4 is expressed in CySCs and in early differentiating cyst cells. To determine the Zfh1-expressing population in the testis, we counted all Zfh1-positive and Eya-negative cells in an entire confocal z-stack.
Positively marked clones were generated by the MARCM technique after a single 1 h heat shock at 37°C (Lee and Luo, 1999). Lineage-wide misexpression or depletion was achieved using the Gal4/UAS system (Brand and Perrimon, 1993). A gal80ts transgene was used when depleting Akt1 (McGuire et al., 2004).
Rapamycin was obtained from LC Laboratories. Rapamycin-containing fly food was prepared by adding 100 µl of a 4 mM stock solution in ethanol to each food vial and letting it air dry. Flies were transferred to fresh rapamycin-containing food every 2 days.
Flies were raised at 25°C, except Tj-Gal4 crosses which were raised at 18°C and adult flies of the correct genotype were maintained at 29°C for 10 days to achieve maximum Gal4 activity.
For a list of full genotypes by figure, see Table S1.
Immunohistochemistry
The following primary antibodies were used: rabbit anti-Zfh1 (gift of R. Lehmann, NYU School of Medicine, NY, USA), 1:5000; guinea pig anti-Zfh1 (gift of J. Skeath, Washington University, St Louis, MO, USA), 1:500; guinea pig anti-Traffic jam (gift of D. Godt, University of Toronto, Ontario, Canada), 1:3000; rabbit anti-phospho-4E-BP1 (Thr37/46, Cell Signaling, 2855), 1:100; goat anti-Vasa (Santa Cruz, 26877), 1:100; mouse anti-Fas3 (DSHB), 1:20; mouse anti-Eya (DSHB), 1:20; mouse anti-Dlg (DSHB), 1:20; rat anti-ImpL2 (gift of J. Natzle, University of California, Davis, CA, USA), 1:500; rabbit anti-GFP (Life Technologies, A6455), 1:500; and chicken anti-GFP (Immunology Consultants, GFP-1020), 1:500.
EdU incorporation and detection were carried out as previously described (Amoyel et al., 2014).
Statistics
Non-parametric data were subjected to two-tailed Mann–Whitney tests and Dunn's multiple comparison tests. Chi-squared was determined by Fisher's exact test. All data and graphs were analyzed in GraphPad Prism 6 software.
Acknowledgements
We thank E. Hafen, L. Jones, J. Bateman, D. Drummond-Barbosa, D. Greenstein, T. Neufeld, D. Godt, J. Natzle, R. Lehmann, BDSC, Kyoto Stock Center, VDRC and DHSB for flies and antibodies. We thank V. Fernandes, J. Treisman, H.-D. Ryoo, C. Desplan, N. Konstantinides, B. Wells, M. Burel and members of the E.A.B. lab for critical discussions and readings of the manuscript.
Footnotes
Competing interests
The authors declare no competing or financial interests.
Author contributions
M.A. designed the experiments with contributions from E.A.B.; M.A., K.-H.H., S.R.M. and E.A.B. performed the experiments and analyzed the data; M.A. and E.A.B. wrote the manuscript.
Funding
This work was supported by the National Institutes of Health [R01 GM085075 to E.A.B.]; and the New York State Department of Health NYSTEM [C028132 to E.A.B.]. M.A. was funded by a Helen L. and Martin S. Kimmel Center for Stem Cell Biology Senior Postdoc Fellowship. Deposited in PMC for release after 12 months.
Supplementary information
Supplementary information available online at http://dev.biologists.org/lookup/doi/10.1242/dev.139782.supplemental
References
- Adhikari D., Zheng W., Shen Y., Gorre N., Hamalainen T., Cooney A. J., Huhtaniemi I., Lan Z.-J. and Liu K. (2010). Tsc/mTORC1 signaling in oocytes governs the quiescence and activation of primordial follicles. Hum. Mol. Genet. 19, 397-410. 10.1093/hmg/ddp483 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alic N., Hoddinott M. P., Vinti G. and Partridge L. (2011). Lifespan extension by increased expression of the Drosophila homologue of the IGFBP7 tumour suppressor. Aging Cell 10, 137-147. 10.1111/j.1474-9726.2010.00653.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amoyel M., Sanny J., Burel M. and Bach E. A. (2013). Hedgehog is required for CySC self-renewal but does not contribute to the GSC niche in the Drosophila testis. Development 140, 56-65. 10.1242/dev.086413 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amoyel M., Simons B. D. and Bach E. A. (2014). Neutral competition of stem cells is skewed by proliferative changes downstream of Hh and Hpo. EMBO J. 33, 2295-2313. 10.15252/embj.201387500 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amoyel M., Anderson J., Suisse A., Glasner J. and Bach E. A. (2016). Socs36E controls Niche competition by repressing MAPK signaling in the Drosophila testis. PLoS Genet. 12, e1005815 10.1371/journal.pgen.1005815 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arquier N., Geminard C., Bourouis M., Jarretou G., Honegger B., Paix A. and Leopold P. (2008). Drosophila ALS regulates growth and metabolism through functional interaction with insulin-like peptides. Cell Metab. 7, 333-338. 10.1016/j.cmet.2008.02.003 [DOI] [PubMed] [Google Scholar]
- Brand A. H. and Perrimon N. (1993). Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 118, 401-415. [DOI] [PubMed] [Google Scholar]
- Britton J. S., Lockwood W. K., Li L., Cohen S. M. and Edgar B. A. (2002). Drosophila's insulin/PI3-kinase pathway coordinates cellular metabolism with nutritional conditions. Dev. Cell 2, 239-249. 10.1016/S1534-5807(02)00117-X [DOI] [PubMed] [Google Scholar]
- Brogiolo W., Stocker H., Ikeya T., Rintelen F., Fernandez R. and Hafen E. (2001). An evolutionarily conserved function of the Drosophila insulin receptor and insulin-like peptides in growth control. Curr. Biol. 11, 213-221. 10.1016/S0960-9822(01)00068-9 [DOI] [PubMed] [Google Scholar]
- Busada J. T., Niedenberger B. A., Velte E. K., Keiper B. D. and Geyer C. B. (2015). Mammalian target of rapamycin complex 1 (mTORC1) is required for mouse spermatogonial differentiation in vivo. Dev. Biol. 407, 90-102. 10.1016/j.ydbio.2015.08.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen T., Shen L., Yu J., Wan H., Guo A., Chen J., Long Y., Zhao J. and Pei G. (2011). Rapamycin and other longevity-promoting compounds enhance the generation of mouse induced pluripotent stem cells. Aging Cell 10, 908-911. 10.1111/j.1474-9726.2011.00722.x [DOI] [PubMed] [Google Scholar]
- Fabrizio J. J., Boyle M. and DiNardo S. (2003). A somatic role for eyes absent (eya) and sine oculis (so) in Drosophila spermatocyte development. Dev. Biol. 258, 117-128. 10.1016/S0012-1606(03)00127-1 [DOI] [PubMed] [Google Scholar]
- Fairchild M. J., Smendziuk C. M. and Tanentzapf G. (2015). A somatic permeability barrier around the germline is essential for Drosophila spermatogenesis. Development 142, 268-281. 10.1242/dev.114967 [DOI] [PubMed] [Google Scholar]
- Flaherty M. S., Salis P., Evans C. J., Ekas L. A., Marouf A., Zavadil J., Banerjee U. and Bach E. A. (2010). chinmo is a functional effector of the JAK/STAT pathway that regulates eye development, tumor formation, and stem cell self-renewal in Drosophila. Dev. Cell 18, 556-568. 10.1016/j.devcel.2010.02.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gingras A.-C., Gygi S. P., Raught B., Polakiewicz R. D., Abraham R. T., Hoekstra M. F., Aebersold R. and Sonenberg N. (1999). Regulation of 4E-BP1 phosphorylation: a novel two-step mechanism. Genes Dev. 13, 1422-1437. 10.1101/gad.13.11.1422 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grewal S. S. (2009). Insulin/TOR signaling in growth and homeostasis: a view from the fly world. Int. J. Biochem. Cell Biol. 41, 1006-1010. 10.1016/j.biocel.2008.10.010 [DOI] [PubMed] [Google Scholar]
- Hardy R. W., Tokuyasu K. T., Lindsley D. L. and Garavito M. (1979). The germinal proliferation center in the testis of Drosophila melanogaster. J. Ultrastruct. Res. 69, 180-190. 10.1016/S0022-5320(79)90108-4 [DOI] [PubMed] [Google Scholar]
- He J., Kang L., Wu T., Zhang J., Wang H., Gao H., Zhang Y., Huang B., Liu W., Kou Z. et al. (2012). An elaborate regulation of Mammalian target of rapamycin activity is required for somatic cell reprogramming induced by defined transcription factors. Stem Cells Dev. 21, 2630-2641. 10.1089/scd.2012.0015 [DOI] [PubMed] [Google Scholar]
- Hobbs R. M., Seandel M., Falciatori I., Rafii S. and Pandolfi P. P. (2010). Plzf regulates germline progenitor self-renewal by opposing mTORC1. Cell 142, 468-479. 10.1016/j.cell.2010.06.041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Honegger B., Galic M., Kohler K., Wittwer F., Brogiolo W., Hafen E. and Stocker H. (2008). Imp-L2, a putative homolog of vertebrate IGF-binding protein 7, counteracts insulin signaling in Drosophila and is essential for starvation resistance. J. Biol. 7, 10 10.1186/jbiol72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsu H.-J. and Drummond-Barbosa D. (2009). Insulin levels control female germline stem cell maintenance via the niche in Drosophila. Proc. Natl. Acad. Sci. USA 106, 1117-1121. 10.1073/pnas.0809144106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsu H.-J., LaFever L. and Drummond-Barbosa D. (2008). Diet controls normal and tumorous germline stem cells via insulin-dependent and -independent mechanisms in Drosophila. Dev. Biol. 313, 700-712. 10.1016/j.ydbio.2007.11.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inaba M., Yuan H. and Yamashita Y. M. (2011). String (Cdc25) regulates stem cell maintenance, proliferation and aging in Drosophila testis. Development 138, 5079-5086. 10.1242/dev.072579 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Issigonis M., Tulina N., de Cuevas M., Brawley C., Sandler L. and Matunis E. (2009). JAK-STAT signal inhibition regulates competition in the Drosophila testis stem cell niche. Science 326, 153-156. 10.1126/science.1176817 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacinto E., Loewith R., Schmidt A., Lin S., Ruegg M. A., Hall A. and Hall M. N. (2004). Mammalian TOR complex 2 controls the actin cytoskeleton and is rapamycin insensitive. Nat. Cell Biol. 6, 1122-1128. 10.1038/ncb1183 [DOI] [PubMed] [Google Scholar]
- Kapuria S., Karpac J., Biteau B., Hwangbo D. and Jasper H. (2012). Notch-mediated suppression of TSC2 expression regulates cell differentiation in the Drosophila intestinal stem cell lineage. PLoS Genet. 8, e1003045 10.1371/journal.pgen.1003045 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiger A. A., White-Cooper H. and Fuller M. T. (2000). Somatic support cells restrict germline stem cell self-renewal and promote differentiation. Nature 407, 750-754. 10.1038/35037606 [DOI] [PubMed] [Google Scholar]
- Kiger A. A., Jones D. L., Schulz C., Rogers M. B. and Fuller M. T. (2001). Stem cell self-renewal specified by JAK-STAT activation in response to a support cell cue. Science 294, 2542-2545. 10.1126/science.1066707 [DOI] [PubMed] [Google Scholar]
- Kirilly D., Wang S. and Xie T. (2011). Self-maintained escort cells form a germline stem cell differentiation niche. Development 138, 5087-5097. 10.1242/dev.067850 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klein A. M. and Simons B. D. (2011). Universal patterns of stem cell fate in cycling adult tissues. Development 138, 3103-3111. 10.1242/dev.060103 [DOI] [PubMed] [Google Scholar]
- LaFever L. and Drummond-Barbosa D. (2005). Direct control of germline stem cell division and cyst growth by neural insulin in Drosophila. Science 309, 1071-1073. 10.1126/science.1111410 [DOI] [PubMed] [Google Scholar]
- LaFever L., Feoktistov A., Hsu H.-J. and Drummond-Barbosa D. (2010). Specific roles of Target of rapamycin in the control of stem cells and their progeny in the Drosophila ovary. Development 137, 2117-2126. 10.1242/dev.050351 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laplante M. and Sabatini D. M. (2012). mTOR signaling in growth control and disease. Cell 149, 274-293. 10.1016/j.cell.2012.03.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leatherman J. L. and Dinardo S. (2008). Zfh-1 controls somatic stem cell self-renewal in the Drosophila testis and nonautonomously influences germline stem cell self-renewal. Cell Stem Cell 3, 44-54. 10.1016/j.stem.2008.05.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leatherman J. L. and Dinardo S. (2010). Germline self-renewal requires cyst stem cells and stat regulates niche adhesion in Drosophila testes. Nat. Cell Biol. 12, 806-811. 10.1038/ncb2086 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee T. and Luo L. (1999). Mosaic analysis with a repressible cell marker for studies of gene function in neuronal morphogenesis. Neuron 22, 451-461. 10.1016/S0896-6273(00)80701-1 [DOI] [PubMed] [Google Scholar]
- Li M. A., Alls J. D., Avancini R. M., Koo K. and Godt D. (2003). The large Maf factor Traffic Jam controls gonad morphogenesis in Drosophila. Nat. Cell Biol. 5, 994-1000. 10.1038/ncb1058 [DOI] [PubMed] [Google Scholar]
- Loewith R. and Hall M. N. (2011). Target of rapamycin (TOR) in nutrient signaling and growth control. Genetics 189, 1177-1201. 10.1534/genetics.111.133363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Losick V. P., Morris L. X., Fox D. T. and Spradling A. (2011). Drosophila stem cell niches: a decade of discovery suggests a unified view of stem cell regulation. Dev. Cell 21, 159-171. 10.1016/j.devcel.2011.06.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo L., Wang H., Fan C., Liu S. and Cai Y. (2015). Wnt ligands regulate Tkv expression to constrain Dpp activity in the Drosophila ovarian stem cell niche. J. Cell Biol. 209, 595-608. 10.1083/jcb.201409142 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGuire S. E., Mao Z. and Davis R. L. (2004). Spatiotemporal gene expression targeting with the TARGET and gene-switch systems in Drosophila. Sci. STKE 2004, pl6 10.1126/stke.2202004pl6 [DOI] [PubMed] [Google Scholar]
- McLeod C. J., Wang L., Wong C. and Jones D. L. (2010). Stem cell dynamics in response to nutrient availability. Curr. Biol. 20, 2100-2105. 10.1016/j.cub.2010.10.038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michel M., Kupinski A. P., Raabe I. and Bokel C. (2012). Hh signalling is essential for somatic stem cell maintenance in the Drosophila testis niche. Development 139, 2663-2669. 10.1242/dev.075242 [DOI] [PubMed] [Google Scholar]
- Miron M., Lasko P. and Sonenberg N. (2003). Signaling from Akt to FRAP/TOR targets both 4E-BP and S6K in Drosophila melanogaster. Mol. Cell. Biol. 23, 9117-9126. 10.1128/MCB.23.24.9117-9126.2003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagarkar-Jaiswal S., Lee P.-T., Campbell M. E., Chen K., Anguiano-Zarate S., Gutierrez M. C., Busby T., Lin W.-W., He Y., Schulze K. L. et al. (2015). A library of MiMICs allows tagging of genes and reversible, spatial and temporal knockdown of proteins in Drosophila. eLife 4, e05338 10.7554/elife.05338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okamoto N., Yamanaka N., Yagi Y., Nishida Y., Kataoka H., O'Connor M. B. and Mizoguchi A. (2009). A fat body-derived IGF-like peptide regulates postfeeding growth in Drosophila. Dev. Cell 17, 885-891. 10.1016/j.devcel.2009.10.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quan Z., Sun P., Lin G. and Xi R. (2013). TSC1/2 regulates intestinal stem cell maintenance and lineage differentiation through Rheb-TORC1-S6K but independently of nutritional status or Notch regulation. J. Cell Sci. 126, 3884-3892. 10.1242/jcs.125294 [DOI] [PubMed] [Google Scholar]
- Reddy P., Liu L., Adhikari D., Jagarlamudi K., Rajareddy S., Shen Y., Du C., Tang W., Hamalainen T., Peng S. L. et al. (2008). Oocyte-specific deletion of Pten causes premature activation of the primordial follicle pool. Science 319, 611-613. 10.1126/science.1152257 [DOI] [PubMed] [Google Scholar]
- Sanchez C. G., Teixeira F. K., Czech B., Preall J. B., Zamparini A. L., Seifert J. R. K., Malone C. D., Hannon G. J. and Lehmann R. (2016). Regulation of ribosome biogenesis and protein synthesis controls germline stem cell differentiation. Cell Stem Cell 18, 276-290. 10.1016/j.stem.2015.11.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schulz C., Wood C. G., Jones D. L., Tazuke S. I. and Fuller M. T. (2002). Signaling from germ cells mediated by the rhomboid homolog stet organizes encapsulation by somatic support cells. Development 129, 4523-4534. [DOI] [PubMed] [Google Scholar]
- Shields A. R., Spence A. C., Yamashita Y. M., Davies E. L. and Fuller M. T. (2014). The actin-binding protein profilin is required for germline stem cell maintenance and germ cell enclosure by somatic cyst cells. Development 141, 73-82. 10.1242/dev.101931 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Signer R. A. J., Magee J. A., Salic A. and Morrison S. J. (2014). Haematopoietic stem cells require a highly regulated protein synthesis rate. Nature 509, 49-54. 10.1038/nature13035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slaidina M., Delanoue R., Gronke S., Partridge L. and Leopold P. (2009). A Drosophila insulin-like peptide promotes growth during nonfeeding states. Dev. Cell 17, 874-884. 10.1016/j.devcel.2009.10.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stine R. R., Greenspan L. J., Ramachandran K. V. and Matunis E. L. (2014). Coordinate regulation of stem cell competition by Slit-Robo and JAK-STAT signaling in the Drosophila testis. PLoS Genet. 10, e1004713 10.1371/journal.pgen.1004713 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun P., Quan Z., Zhang B., Wu T. and Xi R. (2010). TSC1/2 tumour suppressor complex maintains Drosophila germline stem cells by preventing differentiation. Development 137, 2461-2469. 10.1242/dev.051466 [DOI] [PubMed] [Google Scholar]
- Tatar M., Kopelman A., Epstein D., Tu M.-P., Yin C.-M. and Garofalo R. S. (2001). A mutant Drosophila insulin receptor homolog that extends life-span and impairs neuroendocrine function. Science 292, 107-110. 10.1126/science.1057987 [DOI] [PubMed] [Google Scholar]
- Terry N. A., Tulina N., Matunis E. and DiNardo S. (2006). Novel regulators revealed by profiling Drosophila testis stem cells within their niche. Dev. Biol. 294, 246-257. 10.1016/j.ydbio.2006.02.048 [DOI] [PubMed] [Google Scholar]
- Tran J., Brenner T. J. and DiNardo S. (2000). Somatic control over the germline stem cell lineage during Drosophila spermatogenesis. Nature 407, 754-757. 10.1038/35037613 [DOI] [PubMed] [Google Scholar]
- Tulina N. and Matunis E. (2001). Control of stem cell self-renewal in Drosophila spermatogenesis by JAK-STAT signaling. Science 294, 2546-2549. 10.1126/science.1066700 [DOI] [PubMed] [Google Scholar]
- Ueishi S., Shimizu H. and Inoue Y. H. (2009). Male germline stem cell division and spermatocyte growth require insulin signaling in Drosophila. Cell Struct. Funct. 34, 61-69. 10.1247/csf.08042 [DOI] [PubMed] [Google Scholar]
- Upadhyay M., Martino Cortez Y., Wong-Deyrup S., Tavares L., Schowalter S., Flora P., Hill C., Nasrallah M. A., Chittur S. and Rangan P. (2016). Transposon dysregulation modulates dWnt4 signaling to control germline stem cell differentiation in Drosophila. PLoS Genet. 12, e1005918 10.1371/journal.pgen.1005918 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang L., McLeod C. and Jones D. L. (2011). Regulation of adult stem cell behavior by nutrient signaling. Cell Cycle 10, 2628-2634. 10.4161/cc.10.16.17059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang S., Gao Y., Song X., Ma X., Zhu X., Mao Y., Yang Z., Ni J., Li H., Malanowski K. E. et al. (2015). Wnt signaling-mediated redox regulation maintains the germ line stem cell differentiation niche. eLife 4, e08174 10.7554/elife.08174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yilmaz O. H., Valdez R., Theisen B. K., Guo W., Ferguson D. O., Wu H. and Morrison S. J. (2006). Pten dependence distinguishes haematopoietic stem cells from leukaemia-initiating cells. Nature 441, 475-482. 10.1038/nature04703 [DOI] [PubMed] [Google Scholar]
- Ying Q.-L., Wray J., Nichols J., Batlle-Morera L., Doble B., Woodgett J., Cohen P. and Smith A. (2008). The ground state of embryonic stem cell self-renewal. Nature 453, 519-523. 10.1038/nature06968 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y., Billington C. J. Jr, Pan D. and Neufeld T. P. (2006). Drosophila target of rapamycin kinase functions as a multimer. Genetics 172, 355-362. 10.1534/genetics.105.051979 [DOI] [PMC free article] [PubMed] [Google Scholar]