Abstract
Liver fibrosis is the end-stage of many chronic liver diseases and is a significant health threat. The only effective therapy is liver transplantation, which still has many problems, including the lack of donor sources, immunological rejection, and high surgery costs, among others. However, the use of cell therapy is becoming more prevalent, and mesenchymal stem cells (MSCs) seem to be a promising cell type for the treatment of liver fibrosis. MSCs have multiple differentiation abilities, allowing them to migrate directly into injured tissue and differentiate into hepatocyte-like cells. Additionally, MSCs can release various growth factors and cytokines to increase hepatocyte regeneration, regress liver fibrosis, and regulate inflammation and immune responses. In this review, we summarize the current uses of MSC therapies for liver fibrosis and suggest potential future applications.
Keywords: Liver fibrosis, Cell therapy, Mesenchymal stem cells
1. Introduction
Liver fibrosis is a pathologic process that occurs between liver injury and liver cirrhosis. After liver injury, several processes occur including cell apoptosis, inflammation, and scarring, resulting in the deposition of the extracellular matrix (ECM). In the early stages, the ECM deposition can be hydrolyzed by proteolytic enzymes such as matrix metalloproteinases (MMPs). However, continuous damage will ultimately lead to excessive matrix deposition and the alteration of the normal liver structure (Lichtinghagen et al., 2001).
Liver fibrosis can be triggered by viruses, alcohol abuse, drug abuse, and auto-immunity, among other conditions. When liver fibrosis is not well controlled, it can develop into liver cirrhosis, which is the end-stage of liver disease. Currently, liver transplantation is the main effective therapy for liver cirrhosis, but this treatment method is associated with many problems, such as immunological rejection, donor shortages, surgical complications, and high costs (Eom et al., 2015). Therefore, finding new therapeutic strategies for liver fibrosis is essential.
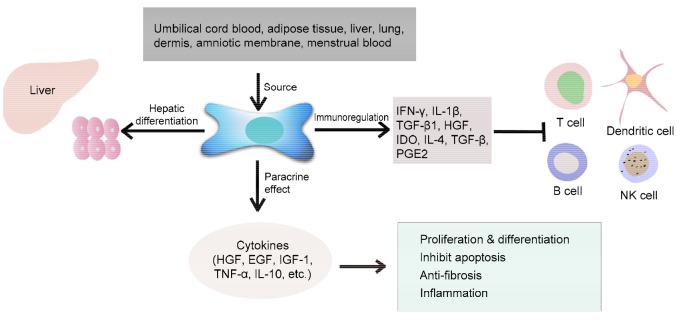
In addition to liver transplantation, cell therapy is a common treatment method for liver disease. For instance, hepatocyte transplantation can be used to restore liver function because of the regeneration abilities of these cells. However, the utility of this treatment is limited because hepatocytes easily lose their viability and function when they are cultured in vitro or when they are preserved cryogenically (Eom et al., 2015). Thus, other types of cells have been explored in an effort to find an ideal treatment for liver diseases. Such research has shown that stem cell transplantation is an effective therapy for liver fibrosis (Kakinuma et al., 2009; Kharaziha et al., 2009). Among the different types of stem cells, mesenchymal stem cells (MSCs) in particular have obvious advantages in regenerative repair because of their high potential for multipotent differentiation, capacity for self-renewal, and low immunogenicity (Jang et al., 2014). MSCs are fibroblast-like and plate-adhering cells, which have the ability to self-renew and to differentiate into adult cells from different germ layers, such as neurocytes from ectoderm, osteoblasts and myocytes from mesoderm, and hepatocyte-like cells from endoderm (Chan et al., 2014). Recently, MSCs have been isolated from a variety of tissues including umbilical cord blood, adipose tissue, liver, lung, dermis, amniotic membrane, and menstrual blood (Erices et al., 2000; Campagnoli et al., 2001; Jiang et al., 2002; de Ugarte et al., 2003; Mou et al., 2013). Additionally, MSCs can secrete a series of cytokines and signaling molecules, such as hepatocyte growth factor (HGF), interleukin 6 (IL-6), tumor necrosis factor alpha (TNF-α), epidermal growth factor (EGF), nitric oxide, prostaglandin E2 (PGE2), and indoleamine 2,3-dioxygenase (Ortiz et al., 2007; Kiss et al., 2008; Puglisi et al., 2011), which can regulate inflammatory responses, stimulate hepatocyte proliferation, and maintain hepatocyte function (Lin et al., 2011; Sharma et al., 2014) (Fig. 1).
Fig. 1.
Sources and function of mesenchymal stem cells (MSCs)
The sources of MSCs are abundant, including umbilical cord blood, adipose tissue, liver, lung, dermis, amniotic membrane, and menstrual blood, among others. MSCs can differentiate into hepatocytes, secrete various cell factors, and participate in immunoregulation. IFN-γ: interferon gamma; IL: interleukin; TGF: transforming growth factor; HGF: hepatocyte growth factor; IDO: indoleamine 2,3-dioxygenase; PGE2: prostaglandin E2; EGF: epidermal growth factor; IGF: insulin-like growth factor; TNF: tumor necrosis factor; NK cells: natural killer cells
In general, MSC therapy for liver fibrosis is effective and promising, and many studies have been performed in this field. Thus, in this review, we discuss the current research regarding the mechanisms and uses of MSC therapy for liver fibrosis and the associated limitations, and we suggest some potential future applications of this therapy.
2. Mechanisms of fibrogenesis in the liver
The mechanisms of liver fibrosis are complex and involve a variety of cytokines, growth factors, and signaling pathways. Although many studies have been performed, the exact mechanisms of fibrogenesis remain unknown. It has been established that an imbalance between ECM production and degradation is the precipitating cause of liver fibrosis (Ek et al., 2007). However, how the imbalance happens is unclear.
The universally accepted key mechanism involved in ECM accumulation is the activation of transforming growth factor beta (TGF-β)/Smad signaling (Wrana, 1999; Berardis et al., 2015), which is mediated by transmembrane serine/threonine kinase receptors including type I and type II. In the injured liver, the microenvironment promotes the activation of Kupffer cells, which in turn exert proinflammatory cytokines such as TNF-α, TGF-α, TGF-β, and platelet-derived growth factor (PDGF), among others. The increased TGF-β, combined with the type II receptor, activates the type I receptor and forms a complex. Then, the complex phosphorylates, for the downstream signal transduction molecules Smad2/3, are translocated into the nucleus where they regulate transcriptional responses such as collagens.
The activated hepatic stellate cells (HSCs), which can transform into myofibroblast-like cells, also play a critical role in the production of ECM (Berardis et al., 2015). When stimulated by lipid peroxides, products from injured hepatocytes, or biochemical signals from Kupffer cells, HSCs can become activated and exhibit the following: high expression of alpha smooth muscle actin (α-SMA), tissue inhibitors of metalloproteinases (TIMPs)-1/2, the secretion of collagen-1, and increased proliferation ability (Iredale et al., 1992; Friedman, 1993; Benyon et al., 1996). The activation can occur through autocrine and paracrine signaling pathways; one of the main pathways is the PDGF-β signaling pathway. The PDGF-β signaling pathway can activate other pathways, such as the Ras-mitogen-activated protein kinase, phosphoinositide 3-kinase-AKT/protein kinase B, and protein kinase C pathways, resulting in HSC proliferation (Kelly et al., 1991). In addition to HSCs, other cell types, such as circulating fibrocytes, portal fibroblasts, and bone marrow-derived cells, are believed to contribute to ECM deposition (Forbes et al., 2004; Wells et al., 2004).
Recent studies have shown that the development of liver fibrosis is accompanied by the expression of MMPs (Lichtinghagen et al., 2001). MMPs are critical for the regression of fibrogenesis in which they can degrade collagens and are involved in the early stages of tissue remodeling (Milani et al., 1994; Benyon et al., 1996). Moreover, TIMPs, which can be produced by activated HSCs, are believed to induce ECM deposition by slowing the breakdown of collagens (Arthur, 1995; 1997). It is known that the expression of TIMPs is mainly induced by inflammation responses. Inflammation factors like IL-1β and TNF-α can promote TIMP expression. Thus, the expression level and activity of TIMPs can be used as indicators to measure the disease process. In general, the balance between MMPs and TIMPs plays an important role in liver fibrosis (Bӧker et al., 2000).
Thus, it is clear that the TGF-β/Smad signaling pathway plays a critical role in ECM accumulation. When this pathway is activated, the downstream factors notably increase, which induces the expression of fibrosis-related genes including the genes for collagen-1, α-SMA (a surface marker of HSCs), and TIMPs. As a result, the number of activated HSCs increases, whereupon the degradation cannot match the production of ECM, resulting in liver fibrosis (Fig. 2).
Fig. 2.
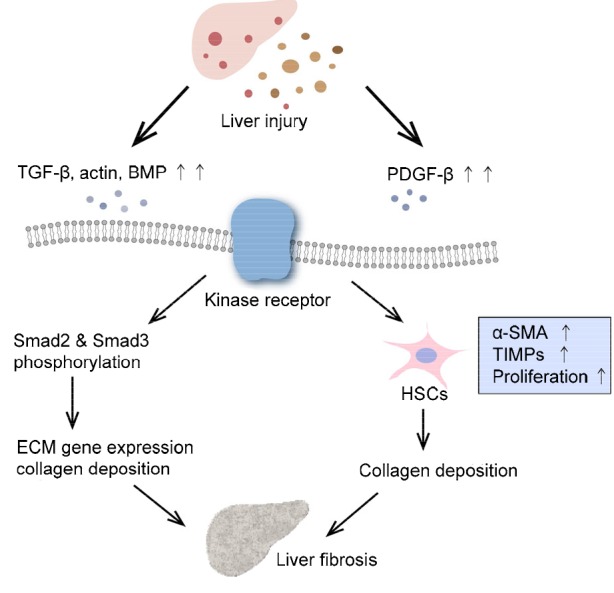
Signaling pathway of liver fibrogenisis
The injured liver produces various cell factors, which can activate transforming growth factor beta (TGF-β) and platelet-derived growth factor beta (PDGF-β), and bone morphogenetic protein (BMP). Through the TGF-β signaling pathway, Smad2/3 are phosphorylated and activate downstream molecules such as extracellular matrix (ECM) genes, resulting in the deposition of collagen. Hepatic stellate cells (HSCs) are greatly increased through the PDGF-β pathway and they induce the secretion of collagens and tissue inhibitors of metalloproteinases (TIMPs). Thus, the degradation of collagens decreases and the secretion increases, leading to the deposition of collagens, which results in worsened liver disease. α-SMA: alpha smooth muscle actin
3. Differentiation ability of MSCs
It is known that MSCs have the capacity to differentiate into various progenitor cells from different cell lines, including hepatic progenitor cells. Indeed, a variety of studies (Banas et al., 2007; Ishii et al., 2008; Kakinuma et al., 2009; Puglisi et al., 2011; Hang et al., 2014) have demonstrated the ability of MSCs to differentiate into hepatocyte-like cells by examining the expression of specific hepatocyte markers such as albumin, α-fetoprotein, and cytokeratin-19, among others.
The ability of MSCs to differentiate into hepatocyte-like cells makes them an ideal alternative method for treating liver fibrosis. Therefore, many studies have examined the mechanisms underlying the differentiation ability of MSCs. Several recent studies (Yoshida et al., 2007; Ishii et al., 2008; Liu et al., 2015) have demonstrated that Wnt/β-catenin signaling plays an important role in regulating the hepatic differentiation of human MSCs. Upon Wnt signaling activation, β-catenin will translocate into the nucleus and coactivate downstream transcription factors to regulate the differentiation of MSCs. Furthermore, mesenchymal-epithelial transition and the reverse, epithelial-mesenchymal transition are critical developmental processes that play fundamental roles in the differentiation of multiple tissues (Hay, 2005). Epigenetic modifications, such as DNA methylation and histone acetylation, have also been shown to participate in the differentiation of MSCs (Snykers et al., 2007).
Additionally, some studies have tried to enhance the efficiency of MSC differentiation, since during regular differentiation, MSCs have low metabolic activity and low expression of functional proteins (Ek et al., 2007). For instance, Mohsin et al. (2011) demonstrated that pretreating MSCs with injured liver tissue enhances their differentiation ability owing to the growth factors and cytokines that are released by the injured tissue, such as HGF, insulin-like growth factor (IGF), EGF, and basic fibroblast growth factor (bFGF), among others (Liu et al., 2015).
While it is clear that MSCs have multi-differentiation abilities including the ability to differentiate into hepatocyte-like cells, it remains unclear whether MSCs can adopt a mature hepatic fate, as no reliable and detailed results of mature hepatocytic gene expression have been reported. Many researchers believe that MSCs can become hepatocytes both morphologically and functionally. For instance, Banas et al. (2007) and Yin et al. (2015) demonstrated that adipose tissue-derived MSCs could be induced into transplantable and mature hepatocyte-like cells both in vivo and in vitro. Moreover, these studies showed that differentiated MSCs express hepatocyte-specific markers including albumin and α-fetoprotein and share liver functions such as low-density lipoprotein uptake, glucose storage, and ammonia detoxification.
In contrast, other researchers have opposed the opinion that MSCs can adopt a mature hepatic fate by claiming that only early specific markers have been detected and noting that little credible data on the detection of mature hepatocyte markers exist. Campard et al. (2008) conducted a study to detect the differentiation ability of umbilical cord matrix stem cells; the results showed that the differentiated umbilical cord matrix stem cells exhibited hepatocyte-like morphologies, specific liver markers (e.g., albumin, α-fetoprotein, cytokeratin-19, connexin-32), and some hepatic functions including glucose storage, low-density lipoprotein uptake, and urea production. However, the cells did not express hepatocyte nuclear factor 4 or HepPar1, two specific hepatic makers. In addition, the differentiated MSCs still contained some MSC-specific makers. Collectively, these findings suggested that the differentiated MSCs did not express enough markers of mature hepatocytes, implying that MSCs cannot fully become hepatocytes. Lian et al. (2006) demonstrated that bone marrow (BM) hematopoietic stem cells expressed several hepatic markers but could not be efficiently converted into hepatocyte-like cells, as one of the mature hepatic markers (anti-trypsin) was not detected. Another study (Hengstler et al., 2005), based on drug metabolism, showed that it is unlikely that MSCs fully differentiate into hepatocytes, and it has also noted that the use of different protocols for hepatic differentiation and different detection methods are problematic. Therefore, specific criteria are needed to define hepatocyte-like cells derived from MSCs. It has been suggested that the definition should not only be based on qualitative analyses but also on quantitative analyses including analyses of enzyme activity.
Overall, MSCs have the potential to differentiate into immature hepatocyte-like cells that exhibit some early specific hepatic markers and functions. Regardless, MSCs are an optimal choice for treating liver fibrosis because of their paracrine effects and immunologic regulation in addition to their multi-differentiation potential.
4. Paracrine effect of MSCs
MSCs have the ability to migrate into injured tissues via chemotaxis due to cytokines that are released from the injured organ or tissues (Golzar et al., 2015; Lourenco et al., 2015). Under stimulation of the microenvironment in injured tissue, like some inflammation factors, MSCs can release various growth factors and cytokines, which promote the proliferation of endogenous hepatocytes, reduce hepatocyte apoptosis, enhance liver function, and repress inflammatory responses (Zhou et al., 2009; Lin et al., 2011).
Research shows that MSCs secrete cytokines, such as HGF, EGF, IL-6, and TNF-α, which can stimulate hepatocyte proliferation and enhance liver function, as indicated by the high levels of albumin and urea secretion. For instance, Kim et al. (2014) overexpressed HGF by transducing MSCs with an adenovirus vector carrying the HGF gene. Their results showed a decrease in collagen and lower mRNA levels of the fibrogenic cytokines PDGF-bb and TGF-β1, suggesting that MSCs that overexpress HGF are effective in the treatment of liver fibrosis. Additionally, MSCs can release other cytokines such as IGF-1, stromal cell-derived factor-1 (SDF-1), and a vascular endothelial growth factor (VEGF), which inhibit cell apoptosis mainly by regulating the SDF-1/CX chemokine receptor-4 (CXCR-4) axis (Lin et al., 2011). IGF-1 is an important factor in body metabolism, and has been demonstrated to be anti-apoptotic to hepatocytes and increase the secretion of HGF in the cirrhotic liver (Bonefeld and Møller, 2011). Fiore et al. (2015) used a recombinant adenovirus overexpressing IGF-1 in BM-MSCs to ameliorate liver fibrosis in mice. The application of BM-derived AdIGF-I-MSCs resulted in the reduced activation of HSCs, increased IGF-I and HGF expression, reduced fibrogenesis, and increased hepatocyte proliferation.
Moreover, researches (Siller-López et al., 2004; Snykers et al., 2007) have demonstrated that MSCs overexpressing MMPs promote the regression of liver fibrosis. MSCs have the potential to reverse the fibrotic process by inhibiting collagen deposition through high levels of MMPs including MMP-8, MMP-9, and MMP-13. MMPs have been shown to degrade the ECM directly in order to balance the increased TIMPs that are induced by activated HSCs, thus contributing to the regression of fibrogenesis (Lin et al., 2011). In addition, the blockades of MSC-derived IL-10 and TNF-α exhibit minimal inhibitory effects on HSC proliferation and collagen synthesis, demonstrating the anti-fibrogenic effects of IL-10 and TNF-α (Parekkadan et al., 2007).
It is known that cytokines are important factors that participate in inflammation, as they can mediate inflammatory responses and prevent inflammatory effects. TNF-α, IL-1, and IL-6 are familiar proinflammatory factors that play critical roles in activating immunocytes and in regulating tissue metabolism (Liu et al., 2013; Huang et al., 2015). In injured tissue, TNF-α is one of the first factors to be released, which then activates neutrophil granulocytes and lymphocytes and induces the secretion of other inflammation factors. In a lung injury model, MSCs have been shown to express an IL-1 receptor antagonist that blocks the release of TNF-α from activated macrophages, thus preventing tissue damage (Ortiz et al., 2007).
5. MSC therapy and immunoregulation
It has been established that MSCs possess remarkable immunosuppressive properties that inhibit the proliferation and function of immune cells from both the adaptive and innate immune systems (Shi et al., 2011). The immunomodulatory effects of MSCs are mediated through both a cell-cell contact and secreted factors such as PGE2, nitric oxide, and TGF-β. MSCs can also inhibit the proliferation of T lymphocytes through cell contact (Tse et al., 2003; Sotiropoulou et al., 2006) and through soluble cytokines such as HGF, IL-1β, TGF-β1, interferon gamma (IFN-γ), and indoleamine 2,3-dioxygenase (di Nicola et al., 2002; Meisel et al., 2004; Groh et al., 2005; Krampera et al., 2006), which is indicated by an increase in the number of cells in the G0/G1 phase (Glennie et al., 2005) and by the up-regulated expression of p27 (Krampera et al., 2003). Further, MSCs can inhibit CD4 + T cells, CD8 + T cells (Glennie et al., 2005), T-helper lymphocytes (Th1/Th17) (Aggarwal and Pittenger, 2005; Zappia et al., 2005), and cytotoxic T cells (Potian et al., 2003; Rasmusson et al., 2003). The suppression effect of MSCs on T cells can indirectly act on B lymphocytes because B cell activation mainly depends on T cells. Additionally, MSCs can directly inhibit the proliferation of B lymphocytes, the production of antibodies, and chemotaxis when co-stimulating with anti-immunoglobulin antibodies, anti-CD40L and IL-4 in humans (Corcione et al., 2006).
MSCs also have suppression effects on cells belonging to the innate immune system, including natural killer (NK) cells, dendritic cells (DCs), monocytes, and macrophages. Studies have shown that MSCs can only partially suppress the proliferation of activated NK cells. Some cell factors such as TGF-β1 and PGE2 are believed to participate in the suppression of NK cell proliferation (Rasmusson et al., 2003; Krampera et al., 2006). In addition, MSCs can affect the production of DCs by inhibiting the differentiation of monocytes, as MSCs can block the maturation signals and co-stimulatory molecules (Zhang et al., 2004; Jiang et al., 2005; Nauta et al., 2006). On the other hand, MSCs reduce the proinflammatory ability of DCs by decreasing the secretion of TNF-α, IFN-γ, and IL-12 and increasing IL-10 secretion (Zhang et al., 2004; Jiang et al., 2005).
In summary, the ability of MSCs to regulate immune responses is an important advantage for cell therapy and allogeneic transplantation. It is known that MSCs have low immunogenicity because they lack human leukocyte antigen class II and co-stimulatory molecules such as CD80, CD86, and CD40 in the cytomembrane (Reinders et al., 2013). In addition, the sources of MSCs are various and abundant. MSCs also have direct migration abilities and a high differentiation capacity. Considering all of these characteristics, MSCs are the ideal transplant donors in regeneration diseases.
6. MSC therapy and CRISPR/Cas9
Currently, genome editing is widely used in studies involving functional genomics, transgenic animals, and gene therapy. Genome editing is based on programmable and highly specific nucleases, which generate site-specific cleavage and subsequently induce cellular DNA repair (Zhang et al., 2014). Multiple artificial nuclease systems have been developed for genome editing, including zinc-finger nucleases, transcription activator-like effector nucleases, and clustered regularly interspaced short palindromic repeats (CRISPR) and the CRISPR-associated (Cas) protein 9. Zinc-finger nucleases and transcription activator-like effector nucleases, based on protein-DNA interactions, are more complex and time-consuming compared with CRISPR/Cas9, which is easier and more efficient when using guide RNA (gRNA) and DNA targeting.
CRISPR/Cas9 is widely used in genetic modification, transcription regulation, and gene therapy studies. Researches have demonstrated that CRISPR/Cas9 can be used to conduct genomic editing in many organisms, including in bacteria (Jiang et al., 2013), drosophila (Gratz et al., 2013), zebrafish (Hruscha et al., 2013), mice (Wang H. et al., 2013), Caenorhabditis elegans (Friedland et al., 2013), and Bombyx mori (Wang Y. et al., 2013). Furthermore, in terms of the development of stem cell therapy, CRISPR/Cas9 has been widely applied in the accurate and complex genetic manipulation of stem cells to enhance their reprogramming, differentiation, and other functions. Mandal et al. (2014) successfully silenced the expression of the genes B2M and CCR5 in human hematopoietic cells using CRISPR/Cas9 with minimal off-target mutagenesis. Additionally, Wettstein et al. (2016) transfected two paired CRISPR single guide RNAs (sgRNAs)-Cas9 plasmids into mouse embryonic stem cells, which resulted in the knock-out of the targeted gene.
CRISPR/Cas9 provides us with a more efficient way to optimize MSC therapy for liver fibrosis. We can transform MSCs using different aspects to enhance their vitality and function, including their proliferation and differentiation ability, chemotaxis for injured tissue, and anti-inflammatory capacity. To aid in this, Schmidt et al. (2015) successfully built an arrayed sgRNA library that can target one critical exon of almost every protein-coding gene in humans. Therefore, by using the sgRNA library, we can find genes related to the various characteristics of MSCs, and then knockout the specific gene to optimize the MSC function.
It is also possible to take advantage of homologous recombination to overexpress targeted genes through CRISPR/Cas9. As mentioned above, genetically engineered MSCs that overexpress certain genes such as the genes for HGF and IGF-1 have therapeutic effects on liver fibrosis. However, it is unclear how we can overexpress specific genes stably without affecting the MSC function or the expression of other genes. This problem is critical. Currently, the use of recombinant virus infection is fervent, including the use of non-integrating viruses like RNA viruses, modified lentiviruses, and integrating adenoviruses (Seah et al., 2015). The efficiency of virus infection and the level of gene expression are both high; however, there are still some problems with this method. Non-integrating viruses will not integrate into the cell genome; therefore, the heterologous gene will not be stably expressed as cell proliferation. Thus, integrated adenoviruses are a good vector for targeted gene overexpression. However, adenoviruses, lentiviruses, and RNA viruses are all viruses, meaning that they are associated with pathogenic risks in clinical treatments. Therefore, finding a new method is necessary.
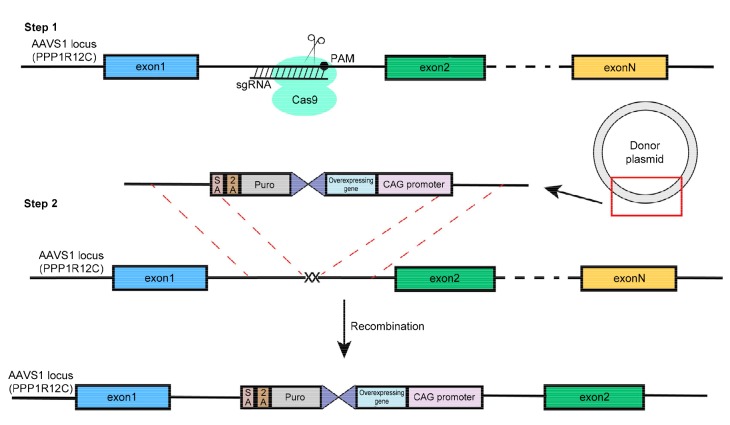
CRISPR/Cas9 is a promising tool that may allow us to transform MSCs in order to overexpress targeted genes. Currently, our lab is performing some related experiments. We have constructed a donor vector that contains the targeted gene, and next we will transfect it with the CRISPR sgRNAs-Cas9 plasmid into MSCs.Taking advantage of homology-direct repair, targeted genes can be combined into the genomic DNA of the MSCs and stably expressed through proliferation (Fig. 3). Our goal is to obtain the targeted gene in a stably expressed cell line, which can then be used to treat liver fibrosis. However, the transfection efficiency is not high; hence, additional research is needed to improve the efficiency.
Fig. 3.
Overexpressing gene in targeted site of genome through CRISPR/Cas9
The cleavage induced by CRISPR/Cas9 produces a double strand break, which will trigger cellular DNA repair processes, including non-homologous end-joining and homology-directed repairs. The AAVS1 locus is a safe harbor for insertion, and does not interfere with the expression of the inserted gene or other genes. We constructed a plasmid containing homologous arms of AAVS1 and inserted genes. Taking advantage of homology directed repair, we can insert certain genes into the specific site and obtain a stably expressed cell line
In general, CRISPR/Cas9 can be used to reform stem cells. Additionally, stem cell therapy combined with genomic editing will be a promising method for many diseases in the future.
7. Current problems and future prospects
The transplantation of MSCs for the treatment of liver fibrosis is an effective and promising method, considering the targeted migration ability, release capacity, and low immunogenicity of MSCs. MSCs can directly interact with the fibrogenic liver by differentiating into hepatocyte-like cells or by fusing with hepatocytes. Additionally, MSCs have the potential to release different growth factors and cytokines, which can regulate the microenvironment and immune system to enhance their therapeutic effects on liver fibrosis. MSCs can also be combined with gene engineering to create a new method that can obviously regress fibrogenesis, promote regeneration, and restore the liver function. Therefore, MSC therapy for liver fibrosis is an optimal choice. However, many issues with these methods still need to be resolved. For instance, several different types of MSCs exist, which each have their respective advantages and disadvantages. The isolation of BM-MSCs is strenuous and traumatic. In contrast, MSCs derived from adipose tissue-derived MSCs are abundant and easily obtained, but the therapeutic effect is inferior to that of BM-MSCs (Liu et al., 2015). Moreover, we still do not fully understand the mechanisms underlying the therapeutic effects of MSCs. Therefore, the oncogenic potential and the risks of using MSCs remain unknown. In addition, when combining MSCs with gene engineering, the transfection problem exists, which will require finding a better transfection condition to increase the efficiency. In general, there is still much for us to explore regarding the use of MSCs in the treatment of liver fibrosis.
Acknowledgements
We thank Dr. Qiu-rong DING (Chinese Academy of Sciences in Shanghai, China) for his guidance on genome editing techniques. Additionally, we thank Editage (https://www. editage.com) for assistance with the English language editing.
Footnotes
Project supported by the National High-Tech R & D Program (863) of China (No. 2015AA020306)
Compliance with ethics guidelines: Yang GUO, Bo CHEN, Li-jun CHEN, Chun-feng ZHANG, and Charlie XIANG declare that they have no conflict of interest.
This article does not contain any studies with human or animal subjects performed by any of the authors.
References
- 1.Aggarwal S, Pittenger MF. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood. 2005;105(4):1815–1822. doi: 10.1182/blood-2004-04-1559. (Available from: http://dx.doi.org/10.1182/blood-2004-04-1559) [DOI] [PubMed] [Google Scholar]
- 2.Arthur MJ. Collagenases and liver fibrosis. J Hepatol. 1995;22(2 Suppl.):43–48. [PubMed] [Google Scholar]
- 3.Arthur MJ. Matrix degradation in liver: a role in injury and repair. Hepatology. 1997;26(4):1069–1071. doi: 10.1002/hep.510260440. (Available from: http://dx.doi.org/10.1053/jhep.1997.v26.ajhep0261069) [DOI] [PubMed] [Google Scholar]
- 4.Banas A, Teratani T, Yamamoto Y, et al. Adipose tissue-derived mesenchymal stem cells as a source of human hepatocytes. Hepatology. 2007;46(1):219–228. doi: 10.1002/hep.21704. (Available from: http://dx.doi.org/10.1002/hep.21704) [DOI] [PubMed] [Google Scholar]
- 5.Benyon RC, Iredale JP, Goddard S, et al. Expression of tissue inhibitor of metalloproteinases 1 and 2 is increased in fibrotic human liver. Gastroenterology. 1996;110(3):821–831. doi: 10.1053/gast.1996.v110.pm8608892. [DOI] [PubMed] [Google Scholar]
- 6.Berardis S, Dwisthi Sattwika P, Najimi M, et al. Use of mesenchymal stem cells to treat liver fibrosis: current situation and future prospects. World J Gastroenterol. 2015;21(3):742–758. doi: 10.3748/wjg.v21.i3.742. (Available from: http://dx.doi.org/10.3748/wjg.v21.i3.742) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bӧker KH, Pehle B, Steinmetz C, et al. Tissue inhibitors of metalloproteinases in liver and serum/plasma in chronic active hepatitis C and HCV-induced cirrhosis. Hepatogastroenterology. 2000;47(33):812–819. [PubMed] [Google Scholar]
- 8.Bonefeld K, Møller S. Insulin-like growth factor-I and the liver. Liver Int. 2011;31(7):911–919. doi: 10.1111/j.1478-3231.2010.02428.x. (Available from: http://dx.doi.org/10.1111/j.1478-3231.2010.02428.x) [DOI] [PubMed] [Google Scholar]
- 9.Campagnoli C, Roberts IA, Kumar S, et al. Identification of mesenchymal stem/progenitor cells in human first-trimester fetal blood, liver, and bone marrow. Blood. 2001;98(8):2396–2402. doi: 10.1182/blood.v98.8.2396. (Available from: http://dx.doi.org/10.1182/blood.V98.8.2396) [DOI] [PubMed] [Google Scholar]
- 10.Campard D, Lysy PA, Najimi M, et al. Native umbilical cord matrix stem cells express hepatic markers and differentiate into hepatocyte-like cells. Gastroenterology. 2008;134(3):833–848. doi: 10.1053/j.gastro.2007.12.024. (Available from: http://dx.doi.org/10.1053/j.gastro.2007.12.024) [DOI] [PubMed] [Google Scholar]
- 11.Chan TM, Harn HJ, Lin HP, et al. Improved human mesenchymal stem cell isolation. Cell Transplant. 2014;23(4-5):399–406. doi: 10.3727/096368914X678292. (Available from: http://dx.doi.org/10.3727/096368914X678292) [DOI] [PubMed] [Google Scholar]
- 12.Corcione A, Benvenuto F, Ferretti E, et al. Human mesenchymal stem cells modulate B-cell functions. Blood. 2006;107(1):367–372. doi: 10.1182/blood-2005-07-2657. (Available from: http://dx.doi.org/10.1182/blood-2005-07-2657) [DOI] [PubMed] [Google Scholar]
- 13.de Ugarte DA, Morizono K, Elbarbary A, et al. Comparison of multi-lineage cells from human adipose tissue and bone marrow. Cells Tissues Organs. 2003;174(3):101–109. doi: 10.1159/000071150. (Available from: http://dx.doi.org/10.1159/000071150) [DOI] [PubMed] [Google Scholar]
- 14.di Nicola M, Carlo-Stella C, Magni M, et al. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood. 2002;99(10):3838–3843. doi: 10.1182/blood.v99.10.3838. (Available from: http://dx.doi.org/10.1182/blood.V99.10.3838) [DOI] [PubMed] [Google Scholar]
- 15.Ek M, Soderdahl T, Küppers-Munther B, et al. Expression of drug metabolizing enzymes in hepatocyte-like cells derived from human embryonic stem cells. Biochem Pharmacol. 2007;74(3):496–503. doi: 10.1016/j.bcp.2007.05.009. (Available from: http://dx.doi.org/10.1016/j.bcp.2007.05.009) [DOI] [PubMed] [Google Scholar]
- 16.Eom YW, Kim G, Baik SK. Mesenchymal stem cell therapy for cirrhosis: present and future perspectives. World J Gastroenterol. 2015;21(36):10253–10261. doi: 10.3748/wjg.v21.i36.10253. (Available from: http://dx.doi.org/10.3748/wjg.v21.i36.10253) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Erices A, Conget P, Minguell JJ. Mesenchymal progenitor cells in human umbilical cord blood. Br J Haematol. 2000;109(1):235–242. doi: 10.1046/j.1365-2141.2000.01986.x. (Available from: http://dx.doi.org/10.1046/j.1365-2141.2000.01986.x) [DOI] [PubMed] [Google Scholar]
- 18.Fiore EJ, Bayo JM, Garcia MG, et al. Mesenchymal stromal cells engineered to produce IGF-I by recombinant adenovirus ameliorate liver fibrosis in mice. Stem Cells Dev. 2015;24(6):791–801. doi: 10.1089/scd.2014.0174. (Available from: http://dx.doi.org/10.1089/scd.2014.0174) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Forbes SJ, Russo FP, Rey V, et al. A significant proportion of myofibroblasts are of bone marrow origin in human liver fibrosis. Gastroenterology. 2004;126(4):955–963. doi: 10.1053/j.gastro.2004.02.025. (Available from: http://dx.doi.org/10.1053/j.gastro.2004.02.025) [DOI] [PubMed] [Google Scholar]
- 20.Friedland AE, Tzur YB, Esvelt KM, et al. Heritable genome editing in C. elegans via a CRISPR-Cas9 system. Nat Methods. 2013;10(8):741–743. doi: 10.1038/nmeth.2532. (Available from: http://dx.doi.org/10.1038/nmeth.2532) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Friedman SL. Seminars in medicine of the Beth Israel Hospital, Boston. The cellular basis of hepatic fibrosis. Mechanisms and treatment strategies. N Engl J Med. 1993;328(25):1828–1835. doi: 10.1056/NEJM199306243282508. (Available from: http://dx.doi.org/10.1056/NEJM199306243282508) [DOI] [PubMed] [Google Scholar]
- 22.Glennie S, Soeiro I, Dyson PJ, et al. Bone marrow mesenchymal stem cells induce division arrest anergy of activated T cells. Blood. 2005;105(7):2821–2827. doi: 10.1182/blood-2004-09-3696. (Available from: http://dx.doi.org/10.1182/blood-2004-09-3696) [DOI] [PubMed] [Google Scholar]
- 23.Golzar F, Javanmard SH, Bahrambeigi V, et al. The effect of Kisspeptin-10 on mesenchymal stem cells migration in vitro and in vivo. Adv Biomed Res. 2015;4:20. doi: 10.4103/2277-9175.149851. (Available from: http://dx.doi.org/10.4103/2277-9175.149851) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gratz SJ, Cummings AM, Nguyen JN, et al. Genome engineering of Drosophila with the CRISPR RNA-guided Cas9 nuclease. Genetics. 2013;194(4):1029–1035. doi: 10.1534/genetics.113.152710. (Available from: http://dx.doi.org/10.1534/genetics.113.152710) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Groh ME, Maitra B, Szekely E, et al. Human mesenchymal stem cells require monocyte-mediated activation to suppress alloreactive T cells. Exp Hematol. 2005;33(8):928–934. doi: 10.1016/j.exphem.2005.05.002. (Available from: http://dx.doi.org/10.1016/j.exphem.2005.05.002) [DOI] [PubMed] [Google Scholar]
- 26.Hang H, Yu Y, Wu N, et al. Induction of highly functional hepatocytes from human umbilical cord mesenchymal stem cells by HNF4α transduction. PLOS ONE. 2014;9(8):e104133. doi: 10.1371/journal.pone.0104133. (Available from: http://dx.doi.org/10.1371/journal.pone.0104133) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hay ED. The mesenchymal cell, its role in the embryo, and the remarkable signaling mechanisms that create it. Dev Dyn. 2005;233(3):706–720. doi: 10.1002/dvdy.20345. (Available from: http://dx.doi.org/10.1371/10.1002/dvdy.20345) [DOI] [PubMed] [Google Scholar]
- 28.Hengstler JG, Brulport M, Schormann W, et al. Generation of human hepatocytes by stem cell technology: definition of the hepatocyte. Expert Opin Drug Metab Toxicol. 2005;1(1):61–74. doi: 10.1517/17425255.1.1.61. (Available from: http://dx.doi.org/10.1517/17425255.1.1.61) [DOI] [PubMed] [Google Scholar]
- 29.Hruscha A, Krawitz P, Rechenberg A, et al. Efficient CRISPR/Cas9 genome editing with low off-target effects in zebrafish. Development. 2013;140(24):4982–4987. doi: 10.1242/dev.099085. (Available from: http://dx.doi.org/10.1242/dev.099085) [DOI] [PubMed] [Google Scholar]
- 30.Huang X, Liu LI, Liu N. Molecular mechanism of tumor necrosis factor-alpha monoclonal antibody in hepatopulmonary syndrome in rats. Chin J Hepatol. 2015;23(6):458–463. doi: 10.3760/cma.j.issn.1007-3418.2015.06.013. (in Chinese) [DOI] [PubMed] [Google Scholar]
- 31.Iredale JP, Murphy G, Hembry RM, et al. Human hepatic lipocytes synthesize tissue inhibitor of metalloproteinases-1. Implications for regulation of matrix degradation in liver. J Clin Invest. 1992;90(1):282–287. doi: 10.1172/JCI115850. (Available from: http://dx.doi.org/10.1172/JCI115850) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ishii K, Yoshida Y, Akechi Y, et al. Hepatic differentiation of human bone marrow-derived mesenchymal stem cells by tetracycline-regulated hepatocyte nuclear factor 3β. Hepatology. 2008;48(2):597–606. doi: 10.1002/hep.22362. (Available from: http://dx.doi.org/10.1002/hep.22362) [DOI] [PubMed] [Google Scholar]
- 33.Jang YO, Kim MY, Cho MY, et al. Effect of bone marrow-derived mesenchymal stem cells on hepatic fibrosis in a thioacetamide-induced cirrhotic rat model. BMC Gastroenterol. 2014;14(1):198. doi: 10.1186/s12876-014-0198-6. (Available from: http://dx.doi.org/10.1186/s12876-014-0198-6) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jiang WY, Bikard D, Cox D, et al. RNA-guided editing of bacterial genomes using CRISPR-Cas systems. Nat Biotechnol. 2013;31(3):233–239. doi: 10.1038/nbt.2508. (Available from: http://dx.doi.org/10.1038/nbt.2508) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jiang XX, Zhang Y, Liu B, et al. Human mesenchymal stem cells inhibit differentiation and function of monocyte-derived dendritic cells. Blood. 2005;105(10):4120–4126. doi: 10.1182/blood-2004-02-0586. (Available from: http://dx.doi.org/10.1182/blood-2004-02-0586) [DOI] [PubMed] [Google Scholar]
- 36.Jiang YH, Vaessen B, Lenvik T, et al. Multipotent progenitor cells can be isolated from postnatal murine bone marrow, muscle, and brain. Exp Hematol. 2002;30(8):896–904. doi: 10.1016/s0301-472x(02)00869-x. [DOI] [PubMed] [Google Scholar]
- 37.Kakinuma S, Nakauchi H, Watanabe M. Hepatic stem/progenitor cells and stem-cell transplantation for the treatment of liver disease. J Gastroenterol. 2009;44(3):167–172. doi: 10.1007/s00535-008-2297-z. (Available from: http://dx.doi.org/10.1007/s00535-008-2297-z) [DOI] [PubMed] [Google Scholar]
- 38.Kelly JD, Haldeman BA, Grant FJ, et al. Platelet-derived growth factor (PDGF) stimulates PDGF receptor subunit dimerization and intersubunit trans-phosphorylation. J Biol Chem. 1991;266(14):8987–8992. [PubMed] [Google Scholar]
- 39.Kharaziha P, Hellstrom PM, Noorinayer B, et al. Improvement of liver function in liver cirrhosis patients after autologous mesenchymal stem cell injection: a phase I‒II clinical trial. Eur J Gastroenterol Hepatol. 2009;21(10):1199–1205. doi: 10.1097/MEG.0b013e32832a1f6c. (Available from: http://dx.doi.org/10.1097/MEG.0b013e32832a1f6c) [DOI] [PubMed] [Google Scholar]
- 40.Kim MD, Kim SS, Cha HY, et al. Therapeutic effect of hepatocyte growth factor-secreting mesenchymal stem cells in a rat model of liver fibrosis. Exp Mol Med. 2014;46:e110. doi: 10.1038/emm.2014.49. (Available from: http://dx.doi.org/10.1038/emm.2014.49) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kiss J, Urbán VS, Dudics V, et al. Mesenchymal stem cells and the immune system–immunosuppression without drugs . Orv Hetil. 2008;149(8):339–346. doi: 10.1556/OH.2008.28291. in Hungarian. (Available from: http://dx.doi.org/10.1556/OH.2008.28291) [DOI] [PubMed] [Google Scholar]
- 42.Krampera M, Glennie S, Dyson J, et al. Bone marrow mesenchymal stem cells inhibit the response of naive and memory antigen-specific T cells to their cognate peptide. Blood. 2003;101(9):3722–3729. doi: 10.1182/blood-2002-07-2104. (Available from: http://dx.doi.org/10.1182/blood-2002-07-2104) [DOI] [PubMed] [Google Scholar]
- 43.Krampera M, Cosmi L, Angeli R, et al. Role for interferon-γ in the immunomodulatory activity of human bone marrow mesenchymal stem cells. Stem Cells. 2006;24(2):386–398. doi: 10.1634/stemcells.2005-0008. (Available from: http://dx.doi.org/10.1634/stemcells.2005-0008) [DOI] [PubMed] [Google Scholar]
- 44.Lian GW, Wang CY, Teng CB, et al. Failure of hepatocyte marker-expressing hematopoietic progenitor cells to efficiently convert into hepatocytes in vitro. Exp Hematol. 2006;34(3):348–358. doi: 10.1016/j.exphem.2005.12.004. (Available from: http://dx.doi.org/10.1016/j.exphem.2005.12.004) [DOI] [PubMed] [Google Scholar]
- 45.Lichtinghagen R, Michels D, Haberkorn CI, et al. Matrix metalloproteinase (MMP)-2, MMP-7, and tissue inhibitor of metalloproteinase-1 are closely related to the fibroproliferative process in the liver during chronic hepatitis C. J Hepatol. 2001;34(2):239–247. doi: 10.1016/s0168-8278(00)00037-4. (Available from: http://dx.doi.org/10.1016/S0168-8278(00)00037-4) [DOI] [PubMed] [Google Scholar]
- 46.Lin H, Xu R, Zhang Z, et al. Implications of the immunoregulatory functions of mesenchymal stem cells in the treatment of human liver diseases. Cell Mol Immunol. 2011;8(1):19–22. doi: 10.1038/cmi.2010.57. (Available from: http://dx.doi.org/10.1038/cmi.2010.57) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Liu WH, Song FQ, Ren LN, et al. The multiple functional roles of mesenchymal stem cells in participating in treating liver diseases. J Cell Mol Med. 2015;19(3):511–520. doi: 10.1111/jcmm.12482. (Available from: http://dx.doi.org/10.1111/jcmm.12482) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Liu Y, Zuo GQ, Zhao L, et al. Effect of inflammatory stress on hepatic cholesterol accumulation and hepatic fibrosis in C57BL/6J mice. Chin J Hepatol. 2013;21(2):116-120(in Chinese):116–120 (in Chinese). doi: 10.3760/cma.j.issn.1007-3418.2013.02.010. (Available from: http://dx.doi.org/10.3760/cma.j.issn.1007-3418.2013.02.010) [DOI] [PubMed] [Google Scholar]
- 49.Lourenco S, Teixeira VH, Kalber T, et al. Macrophage migration inhibitory factor-CXCR4 is the dominant chemotactic axis in human mesenchymal stem cell recruitment to tumors. J Immunol. 2015;194(7):3463–3474. doi: 10.4049/jimmunol.1402097. (Available from: http://dx.doi.org/10.4049/jimmunol.1402097) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mandal PK, Ferreira LM, Collins R, et al. Efficient ablation of genes in human hematopoietic stem and effector cells using CRISPR/Cas9. Cell Stem Cell. 2014;15(5):643–652. doi: 10.1016/j.stem.2014.10.004. (Available from: http://dx.doi.org/10.1016/j.stem.2014.10.004) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Meisel R, Zibert A, Laryea M, et al. Human bone marrow stromal cells inhibit allogeneic T-cell responses by indoleamine 2,3-dioxygenase-mediated tryptophan degradation. Blood. 2004;103(12):4619–4621. doi: 10.1182/blood-2003-11-3909. (Available from: http://dx.doi.org/10.1182/blood-2003-11-3909) [DOI] [PubMed] [Google Scholar]
- 52.Milani S, Herbst H, Schuppan D, et al. Differential expression of matrix-metalloproteinase-1 and -2 genes in normal and fibrotic human liver. Am J Pathol. 1994;144(3):528–537. [PMC free article] [PubMed] [Google Scholar]
- 53.Mohsin S, Shams S, Ali Nasir G, et al. Enhanced hepatic differentiation of mesenchymal stem cells after pretreatment with injured liver tissue. Differentiation. 2011;81(1):42–48. doi: 10.1016/j.diff.2010.08.005. (Available from: http://dx.doi.org/10.1016/j.diff.2010.08.005) [DOI] [PubMed] [Google Scholar]
- 54.Mou XZ, Lin J, Chen JY, et al. Menstrual blood-derived mesenchymal stem cells differentiate into functional hepatocyte-like cells. J Zhejiang Univ-Sci B (Biomed & Biotechnol) 2013;14(11):961–972. doi: 10.1631/jzus.B1300081. (Available from: http://dx.doi.org/10.1631/jzus.B1300081) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Nauta AJ, Kruisselbrink AB, Lurvink E, et al. Mesenchymal stem cells inhibit generation and function of both CD34+-derived and monocyte-derived dendritic cells. J Immunol. 2006;177(4):2080–2087. doi: 10.4049/jimmunol.177.4.2080. (Available from: http://dx.doi.org/10.4049/jimmunol.177.4.2080) [DOI] [PubMed] [Google Scholar]
- 56.Ortiz LA, DuTreil M, Fattman C, et al. Interleukin 1 receptor antagonist mediates the antiinflammatory and antifibrotic effect of mesenchymal stem cells during lung injury. PNAS. 2007;104(26):11002–11007. doi: 10.1073/pnas.0704421104. (Available from: http://dx.doi.org/10.1073/pnas.0704421104) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Parekkadan B, van Poll D, Megeed Z, et al. Immunomodulation of activated hepatic stellate cells by mesenchymal stem cells. Biochem Biophys Res Commun. 2007;363(2):247–252. doi: 10.1016/j.bbrc.2007.05.150. (Available from: http://dx.doi.org/10.1016/j.bbrc.2007.05.150) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Potian JA, Aviv H, Ponzio NM, et al. Veto-like activity of mesenchymal stem cells: functional discrimination between cellular responses to alloantigens and recall antigens. J Immunol. 2003;171(7):3426–3434. doi: 10.4049/jimmunol.171.7.3426. [DOI] [PubMed] [Google Scholar]
- 59.Puglisi MA, Tesori V, Lattanzi W, et al. Therapeutic implications of mesenchymal stem cells in liver injury. J Biomed Biotechnol. 2011;2011:860578. doi: 10.1155/2011/860578. (Available from: http://dx.doi.org/10.1155/2011/860578) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Rasmusson I, Ringdén O, Sundberg B, et al. Mesenchymal stem cells inhibit the formation of cytotoxic T lymphocytes, but not activated cytotoxic T lymphocytes or natural killer cells. Transplantation. 2003;76(8):1208–1213. doi: 10.1097/01.TP.0000082540.43730.80. (Available from: http://dx.doi.org/10.1097/01.TP.0000082540.43730.80) [DOI] [PubMed] [Google Scholar]
- 61.Reinders ME, Rabelink TJ, de Fijter JW. The role of mesenchymal stromal cells in chronic transplant rejection after solid organ transplantation. Curr Opin Organ Transplant. 2013;18(1):44–50. doi: 10.1097/MOT.0b013e32835c2939. (Available from: http://dx.doi.org/10.1097/MOT.0b013e32835c2939) [DOI] [PubMed] [Google Scholar]
- 62.Schmidt T, Schmid-Burgk JL, Hornung V. Synthesis of an arrayed sgRNA library targeting the human genome. Sci Rep. 2015;5:14987. doi: 10.1038/srep14987. (Available from: http://dx.doi.org/10.1038/srep14987) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Seah YFS, el Farran CA, Warrier T, et al. Induced pluripotency and gene editing in disease modelling: perspectives and challenges. Int J Mol Sci. 2015;16(12):28614–28634. doi: 10.3390/ijms161226119. (Available from: http://dx.doi.org/10.3390/ijms161226119) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sharma RR, Pollock K, Hubel A, et al. Mesenchymal stem or stromal cells: a review of clinical applications and manufacturing practices. Transfusion. 2014;54(5):1418–1437. doi: 10.1111/trf.12421. (Available from: http://dx.doi.org/10.1111/trf.12421) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Shi M, Liu ZW, Wang FS. Immunomodulatory properties and therapeutic application of mesenchymal stem cells. Clin Exp Immunol. 2011;164(1):1–8. doi: 10.1111/j.1365-2249.2011.04327.x. (Available from: http://dx.doi.org/10.1111/j.1365-2249.2011.04327.x) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Siller-López F, Sandoval A, Salgado S, et al. Treatment with human metalloproteinase-8 gene delivery ameliorates experimental rat liver cirrhosis. Gastroenterology. 2004;126(4):1122–1133; discussion 1949. doi: 10.1053/j.gastro.2003.12.045. (Available from: http://dx.doi.org/10.1053/j.gastro.2003.12.045) [DOI] [PubMed] [Google Scholar]
- 67.Snykers S, Vanhaecke T, de Becker A, et al. Chromatin remodeling agent trichostatin A: a key-factor in the hepatic differentiation of human mesenchymal stem cells derived of adult bone marrow. BMC Dev Biol. 2007;7:24. doi: 10.1186/1471-213X-7-24. (Available from: http://dx.doi.org/10.1186/1471-213X-7-24) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Sotiropoulou PA, Perez SA, Gritzapis AD, et al. Interactions between human mesenchymal stem cells and natural killer cells. Stem Cells. 2006;24(1):74–85. doi: 10.1634/stemcells.2004-0359. (Available from: http://dx.doi.org/10.1634/stemcells.2004-0359) [DOI] [PubMed] [Google Scholar]
- 69.Tse WT, Pendleton JD, Beyer WM, et al. Suppression of allogeneic T-cell proliferation by human marrow stromal cells: implications in transplantation. Transplantation. 2003;75(3):389–397. doi: 10.1097/01.TP.0000045055.63901.A9. (Available from: http://dx.doi.org/10.1097/01.TP.0000045055.63901.A9) [DOI] [PubMed] [Google Scholar]
- 70.Wang H, Yang H, Shivalila CS, et al. One-step generation of mice carrying mutations in multiple genes by CRISPR/Cas-mediated genome engineering. Cell. 2013;153(4):910–918. doi: 10.1016/j.cell.2013.04.025. (Available from: http://dx.doi.org/10.1016/j.cell.2013.04.025) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Wang Y, Li Z, Xu J, et al. The CRISPR/Cas system mediates efficient genome engineering in Bombyx mori . Cell Res. 2013;23(12):1414–1416. doi: 10.1038/cr.2013.146. (Available from: http://dx.doi.org/10.1038/cr.2013.146) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wells RG, Kruglov E, Dranoff JA. Autocrine release of TGF-β by portal fibroblasts regulates cell growth. FEBS Lett. 2004;559(1-3):107–110. doi: 10.1016/S0014-5793(04)00037-7. (Available from: http://dx.doi.org/10.1016/S0014-5793(04)00037-7) [DOI] [PubMed] [Google Scholar]
- 73.Wettstein R, Bodak M, Ciaudo C. Generation of a knockout mouse embryonic stem cell line using a paired CRISPR/Cas9 genome engineering tool. Methods Mol Biol. 2016;1341:321–343. doi: 10.1007/7651_2015_213. (Available from: http://dx.doi.org/10.1007/7651_2015_213) [DOI] [PubMed] [Google Scholar]
- 74.Wrana JL. Transforming growth factor-β signaling and cirrhosis. Hepatology. 1999;29(6):1909–1910. doi: 10.1002/hep.510290641. (Available from: http://dx.doi.org/10.1002/hep.510290641) [DOI] [PubMed] [Google Scholar]
- 75.Yin LB, Zhu YH, Yang JG, et al. Adipose tissue-derived mesenchymal stem cells differentiated into hepatocyte-like cells in vivo and in vitro. Mol Med Rep. 2015;11(3):1722–1732. doi: 10.3892/mmr.2014.2935. (Available from: http://dx.doi.org/10.3892/mmr.2014.2935) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Yoshida Y, Shimomura T, Sakabe T, et al. A role of Wnt/β-catenin signals in hepatic fate specification of human umbilical cord blood-derived mesenchymal stem cells. Am J Physiol Gastrointest Liver Physiol. 2007;293(5):G1089–G1098. doi: 10.1152/ajpgi.00187.2007. (Available from: http://dx.doi.org/10.1152/ajpgi.00187.2007) [DOI] [PubMed] [Google Scholar]
- 77.Zappia E, Casazza S, Pedemonte E, et al. Mesenchymal stem cells ameliorate experimental autoimmune encephalomyelitis inducing T-cell anergy. Blood. 2005;106(5):1755–1761. doi: 10.1182/blood-2005-04-1496. (Available from: http://dx.doi.org/10.1182/blood-2005-04-1496) [DOI] [PubMed] [Google Scholar]
- 78.Zhang F, Wen Y, Guo X. CRISPR/Cas9 for genome editing: progress, implications and challenges. Hum Mol Genet. 2014;23(R1):R40–R46. doi: 10.1093/hmg/ddu125. (Available from: http://dx.doi.org/10.1093/hmg/ddu125) [DOI] [PubMed] [Google Scholar]
- 79.Zhang W, Ge W, Li CH, et al. Effects of mesenchymal stem cells on differentiation, maturation, and function of human monocyte-derived dendritic cells. Stem Cells Dev. 2004;13(3):263–271. doi: 10.1089/154732804323099190. (Available from: http://dx.doi.org/10.1089/154732804323099190) [DOI] [PubMed] [Google Scholar]
- 80.Zhou P, Hohm S, Olusanya Y, et al. Human progenitor cells with high aldehyde dehydrogenase activity efficiently engraft into damaged liver in a novel model. Hepatology. 2009;49(6):1992–2000. doi: 10.1002/hep.22862. (Available from: http://dx.doi.org/10.1002/hep.22862) [DOI] [PMC free article] [PubMed] [Google Scholar]