Abstract
Dual-energy contrast-enhanced spectral mammography (CESM) represents a relatively new diagnostic tool adjunct to mammography. The aim of this study was to strengthen the breast imaging-reporting and data system (BIRADS) classification score in order to improve early breast cancer diagnosis. For this reason, we propose a sum score, termed malignancy potential score (MPS), incorporating the standard BIRADS score and our proposed CESM score. From September 2014 to September 2015, 216 females (age range, 26–85 years; mean age 54.6 years) underwent CESM evaluation of mammographic findings that were primarily assessed as BIRADS 2–5. 10 of these patients had bilateral findings; a total of 226 lesions were examined. High-energy image evaluation was based on the intensity of contrast enhancement of the lesion compared with background enhancement, categorized as Type -1, 0, 1 or 2 enhancement. Histopathology reports were compared with imaging assessment. 98 of 226 lesions were malignant and 128 of 226 lesions were benign. The area under the curve was 0.843, 0.888 and 0.917 for mammographic BIRADS score, CESM score and MPS, respectively, with p-value < 0.05. The sensitivity, specificity and accuracy rates were 91.83, 80.47 and 85.40%, respectively, when a best MPS cut-off point of 4 was used. The malignancy potential score (MPS) has higher diagnostic performance than digital mammography or CESM alone. MPS empowers the credibility of the digital mammography BIRADS score and our proposed type of enhancement in dual-energy CESM and is a diagnostic tool that increases the accuracy rate in early breast cancer diagnosis.
INTRODUCTION
Breast cancer is the most common cancer in females worldwide.1 It is of great clinical value to find an accurate and cost-effective way to detect and diagnose early breast cancers in females.
While mammography remains the standard for breast cancer screening, image sensitivity and specificity can be affected by several factors. Specifically, in dense breasts, sensitivity has been reported to be as low as 30–48%.2,3
In the past years, new methods have been developed using contrast media for the detection of angiogenesis by tracking contrast agent uptake and washout in suspicious lesions. Contrast-enhanced spectral mammography (CESM) is one of them. CESM provides information about the degree of vascularization of the lesion in addition to the morphological information provided by conventional mammography. According to recent studies, CESM is superior to conventional mammography in breast cancer detection and seems to decrease false negatives especially for females with dense breasts.4 A large clinical study by Dromain et al5 showed that digital mammography plus CESM had higher sensitivity.
These two imaging modalities (CESM and digital mammography) can be combined in order to obtain a score which increases the accuracy rate in early breast cancer detection. This sum score is of great clinical importance. The aim of this prospective study was to provide a cut-off score which results from the sum of our proposed CESM degree enhancement and standard breast imaging-reporting and data system (BIRADS) category in order to increase the accuracy rate of early breast cancer diagnosis.
METHODS AND MATERIALS
Patients included in the study
All enrolled patients provided written informed consent and the study was approved by the institutional review board. From September 2014 to September 2015, 216 females (age range, 26–85 years; mean age 54.6 years) underwent screening or diagnostic mammograms, followed by CESM. 10 of these females had bilateral findings; so, 226 lesions were examined.
Exclusion criteria were pregnancy or possible pregnancy, history of allergic reaction to iodinated contrast agents, breast implants and renal insufficiency. Also, one patient was excluded from the study after exhibiting mild allergic symptoms (mild discomfort and regional rush) during the injection of the iodine substance. In this case, the examination was ceased and antiallergic treatment was provided.
In our prospective study, data from females with BIRADS 3–5 findings were collected, while females with BIRADS 0 or 1, 2, 6 findings were excluded. Each mammography finding was evaluated using established BIRADS criteria (0–6).6 Subsequently, all patients underwent CESM and the findings were evaluated based on the intensity of lesion enhancement compared with background enhancement.
All lesions were evaluated by two independent and experienced radiologists with 15 and 4 years' interpretation experience in mammography and CESM. The evaluation was blinded; radiologists interpreted a similar number of examinations and each modality independently, with a time gap of 4–7 days between reviews for the two different modalities. Sum scores were evaluated prospectively.
Digital mammography
Digital mammography was performed in all patients [including craniocaudal (CC) and mediolateral oblique (MLO) views] and all mammograms were evaluated retrospectively to CESM interpretation.
Contrast-enhanced spectral mammography examination
All examinations were performed using a commercially available full-field digital mammography system (Senographe Essential; GE Healthcare) that was modified to shape the X-ray spectrum specifically for CESM.
The radiographer in charge ensured that the patient had no contraindications for iodine contrast injection and explained the steps of the procedure. A nurse prepared for the i.v. injection and placed a catheter into the antecubital vein of the arm contralateral to the affected breast. The iodinated contrast agent, iopromide (Ultravist) at 300 mg I ml−1, was administrated by using an automatic power injector (CT 9000 ADV; Covidien) at a rate of 2–3 ml sec−1 with a bolus chaser and total volume of 1.5 ml kg−1.
2 min after the initiation of the contrast agent administration, the “normal” breast was compressed in a CC view and a pair of low- and high-energy images was acquired. The procedure was followed by CC and MLO views of the breast with the BIRADS 3–5 categories and finally, an MLO view of the normal breast was performed. After the CESM examination, patients were observed for 30 min to confirm that they had no allergic reaction to the iodinated contrast agent.
Low-energy images were acquired with molybdenum (Mo) or rhodium (Rh) target and Mo or Rh filter automatically, at peak kilovoltage values ranging from 26 to 31 kVp, ensuring that the entire X-ray spectrum was below the k-edge of iodine (33.2 keV). High-energy images were acquired with Mo target and a filter of copper, at 45–49 kVp, ensuring that the average energy of the X-ray spectrum was just above the k-edge of iodine. Iodine-enhanced images were generated from the combination low- and high-energy images. High-energy images showed the enhanced lesions by nullifying the non-enhanced areas of the breast.12 According to literature, glandular dose values ranged between 0.96 and 1.45 mGy (low and high energy). Glandular dose values for a breast thickness of 5.0 cm and a glandular fraction of 50%, for CC and MLO views, were 1.12 mGy (low-energy image contribution is 0.98 mGy) and 1.07 mGy (low-energy image contribution is 0.95 mGy), respectively (data on publication by Yakoumakis et al)13.
Patients were observed for 30 min after the examination to ensure that they had no allergic reaction to the iodinated contrast agent. Compression time for each view was a maximum of 15 s. The total duration of the examination was 7 min.
Image analysis
Digital mammography images were reviewed using the BIRADS assessment scale.
Processed images were transferred directly to the workstation for review by the radiologist. A dedicated hanging protocol and specific tools were used for easy identification and side-by-side display of low-energy and contrast-enhanced images. Analysis of the low-energy images was completed concerning the following information: breast density, location and type of each finding (asymmetry, calcification, mass, density, calcification ± mass, asymmetry ± calcification, architectural distortions, architectural distortions ± calcification, intramammary lymph nodes and scars/distortions) according to the BIRADS (ACR) probability of malignancy classification.
High-energy image evaluation was based on the grade of contrast enhancement intensity compared with background enhancement, according to our proposed quantitative scale of contrast enhancement intensity, categorized into four types as follows:
(a) Type −1 = negative enhancement; the intensity of the lesion is eliminated compared with the background enhancement (these cases, n = 4, in our study were always connected to cystic lesions confirmed by ultrasound or abscesses).
(b) Type 0 = no enhancement of the lesion; the intensity of the lesion is similar to background enhancement
(c) Type 1 = moderate enhancement; moderate homogeneous or heterogeneous enhancement of the lesion
(d) Type 2 = intense enhancement; significantly increased enhancement of the lesion compared with background enhancement (Figure 1).
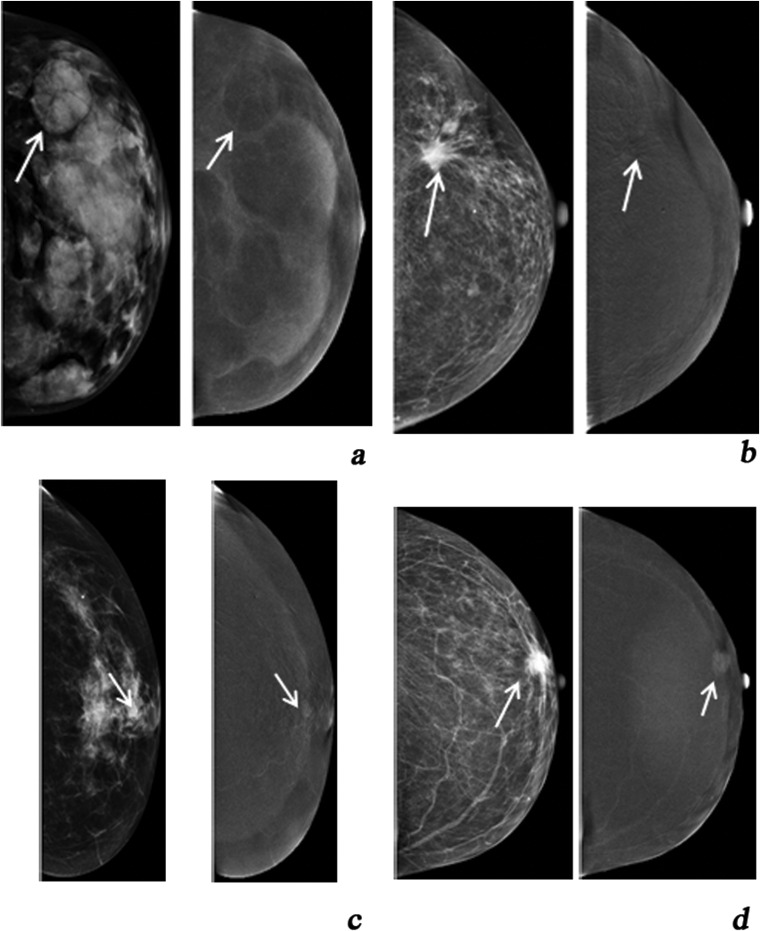
Figure 1.
(a) Type −1 enhancement (negative enhancement), (b) Type 0 = no enhancement of the lesion, (c) Type 1 = moderate enhancement and (d) Type 2 = intense enhancement.
Types −1 and 0 enhancement findings were considered to be probably benign, while Types 1 and 2 enhancement findings were considered as indicating malignancy. Each finding was scored according to the scale (−1, 0, 1, 2) proposed above and the standard BIRADS score.
Then, we thought that having two different scoring systems is confusing in clinical decision-making, while integrating these into one could be of diagnostic value. Thus, the score of BIRADS was added to the score of CESM. This new score served as the final “malignancy potential score” (MPS). In our study, the minimum MPS is: −1 (type of enhancement) + 3 (BIRADS) = 2 and the maximum MPS is: 2 (type of enhancement) + 5 (BIRADS) = 7.
Biopsy (surgical or core) and histopathological analysis were performed and pathology results were compared with the predicted values for each lesion.
Statistical analysis
For the statistical analysis, a standard IBM computer running the SPSS v. 20.0 (IBM Corp., New York, NY; formerly SPSS Inc., Chicago, IL) and R 3.1.0 software with pROC package was utilized.7 Receiver-operating characteristic (ROC) curves were used to determine the sensitivity and specificity and also to extrapolate the optimum cut-off value for MPS. The DeLong test8 for two correlated ROC curves was applied for the comparison of the areas under the curve (AUCs).
RESULTS
Of the 226 breast lesions in 216 patients (10 patients had bilateral findings) in this study, 98 (43%) lesions were malignant and 128 (57%) lesions were benign. The benign masses included sclerosing adenosis with or without microcalcifications and fibrocystic changes (n = 89), atypical hyperplastic conditions (n = 2), fibroadenomas (n = 22), papillomas (n = 6), radial scar (n = 1), hamartomas (n = 2), intramammary lymph node (n = 1), cysts (n = 2), inflammatory conditions (n = 2) and nipple Paget's disease (n = 1).
The malignant lesions included ductal carcinoma in situ (n = 19), invasive ductal carcinoma grades I (n = 12), II (n = 22) and III (n = 37), invasive lobular carcinoma (n = 4) and tubular carcinoma (n = 4).
2 of 4 lesions with Type −1 enhancement were cysts (confirmed by ultrasound as mentioned previously) and 2 of 4 lesions were abscesses.
87 of 93 lesions with Type 0 enhancement were fibroadenomas (n = 10), papillomas (n = 4), intramammary lymph node (n = 1), atypical hyperplastic conditions (n = 2), nipple Paget's disease (n = 1), hamartomas (n = 2), radial scar (n = 1) and sclerosing adenosis with or without microcalcifications and fibrocystic changes (n = 66), whereas 6 of 93 lesions were malignant, namely ductal carcinoma in situ (n = 4) and invasive ductal carcinoma (n = 2).
21 of 46 lesions with Type 1 enhancement were ductal carcinoma in situ, invasive ductal and lobular carcinomas, whereas 25 of 46 lesions were benign, namely fibroadenomas (n = 11), papilloma (n = 1) and sclerosing adenosis with or without microcalcifications and fibrocystic changes (n = 13).
71 of 83 lesions with Type 2 enhancement were ductal carcinoma in situ, invasive ductal and lobular carcinomas (n = 67) and tubular carcinoma (n = 4), whereas 12 of 83 lesions were benign, namely fibroadenomas (n = 1), papilloma (n = 1) and sclerosing adenosis with or without microcalcifications and fibrocystic changes (n = 10).
The sensitivity and specificity of MPS value 4.5 were 0.913 and at 0.806, respectively, while at the value 3.5, the sensitivity and specificity were 0.978 and 0.476. The cut-off point that combined the optimal sensitivity and specificity was value 4 (Figures 2–4).
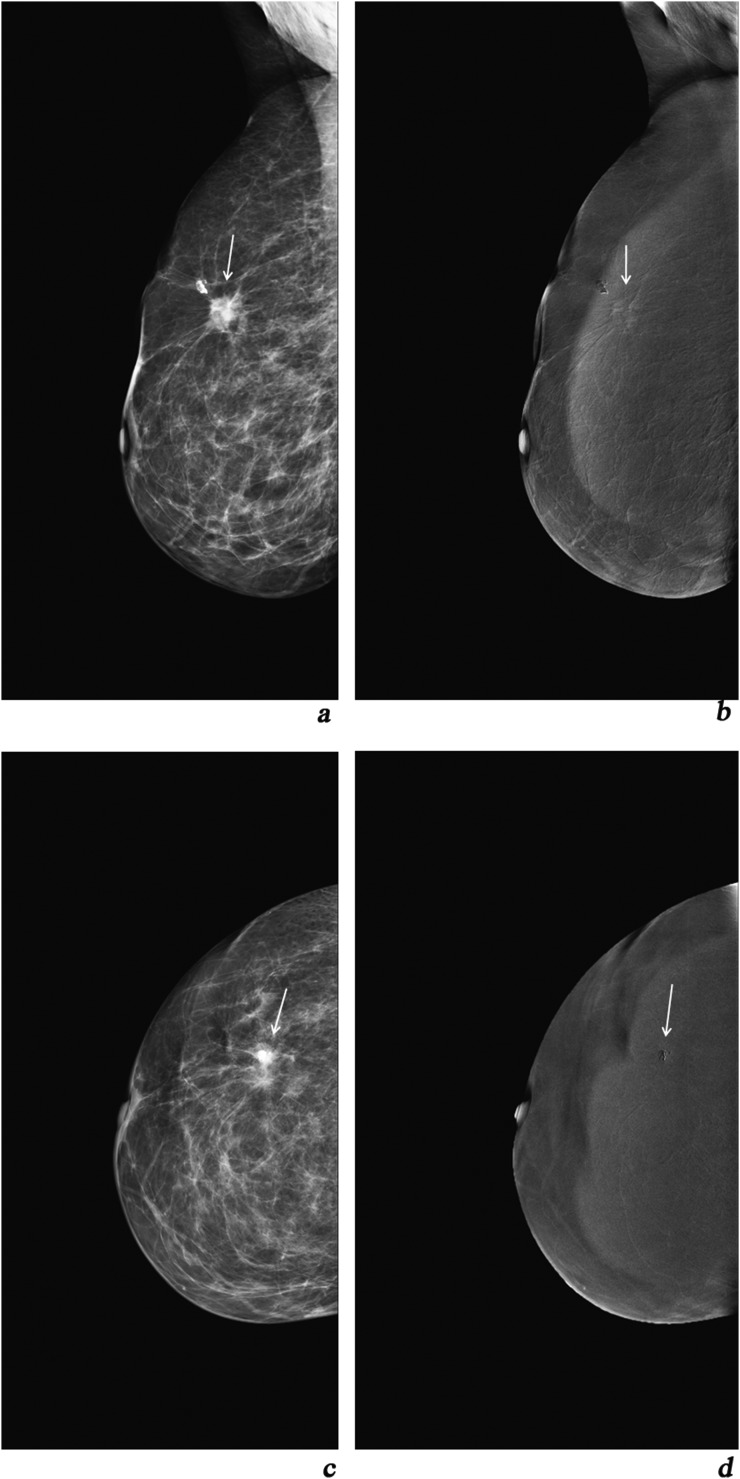
Figure 2.
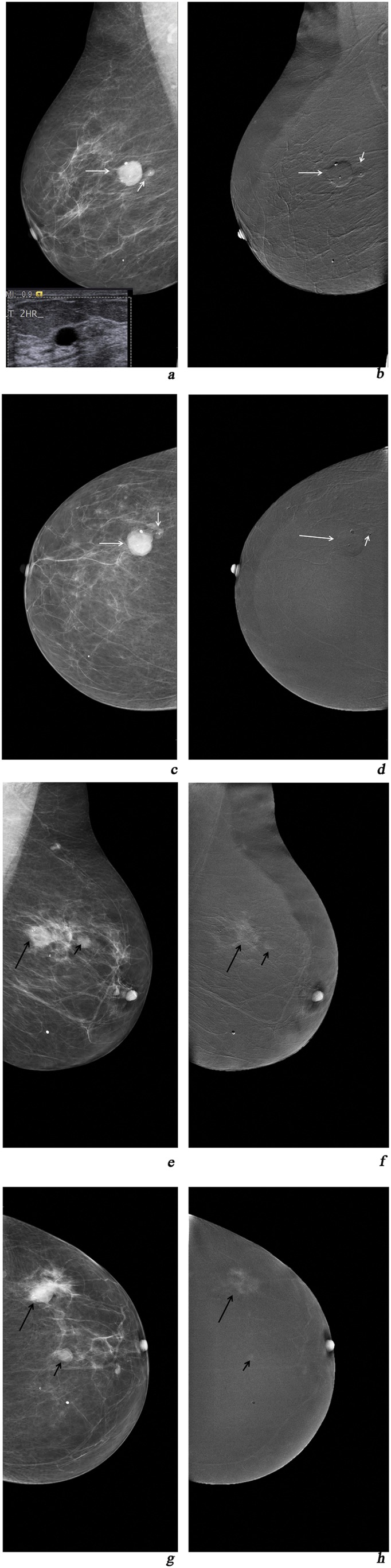
(a, c) Mediolateral oblique (MLO) and craniocaudal (CC) views of digital mammography depicting two lesions of the right breast categorized as breast imaging-reporting and data system (BIRADS) 3; (b, d) MLO and CC views of contrast-enhanced spectral mammography (CESM): Type −1 enhancement of the lesion, MPS = 3 + (−)1= 2 < 4. On ultrasound, two cysts were depicted; (e, g) MLO and CC digital mammography views of the left breast depicting a small BIRADS 4 lesion with microcalcifications (small arrows) and a bigger BIRADS 4 lesion (long arrows); (f, h) MLO and CC views of CESM Type 2 enhancement (long arrows) and Type 1 enhancement (small arrows). MPS1 for the small lesion (small arrows): 4 + 1 = 5 and MPS2 for the lesion (long arrows) = 6 > 4. Histopathology result (left breast): two foci of ductal carcinoma grades III and II, respectively.
Figure 4.
(a, c) Mediolateral oblique (MLO) and craniocaudal (CC) views of digital mammography depicting a breast imaging-reporting and data system 5 lesion of the right breast; (b, d) MLO and CC views of contrast-enhanced spectral mammography depicting a Type −1 enhancement lesion. MPS= 5 + (−1) = 4. The lesion was a post-surgery scar.
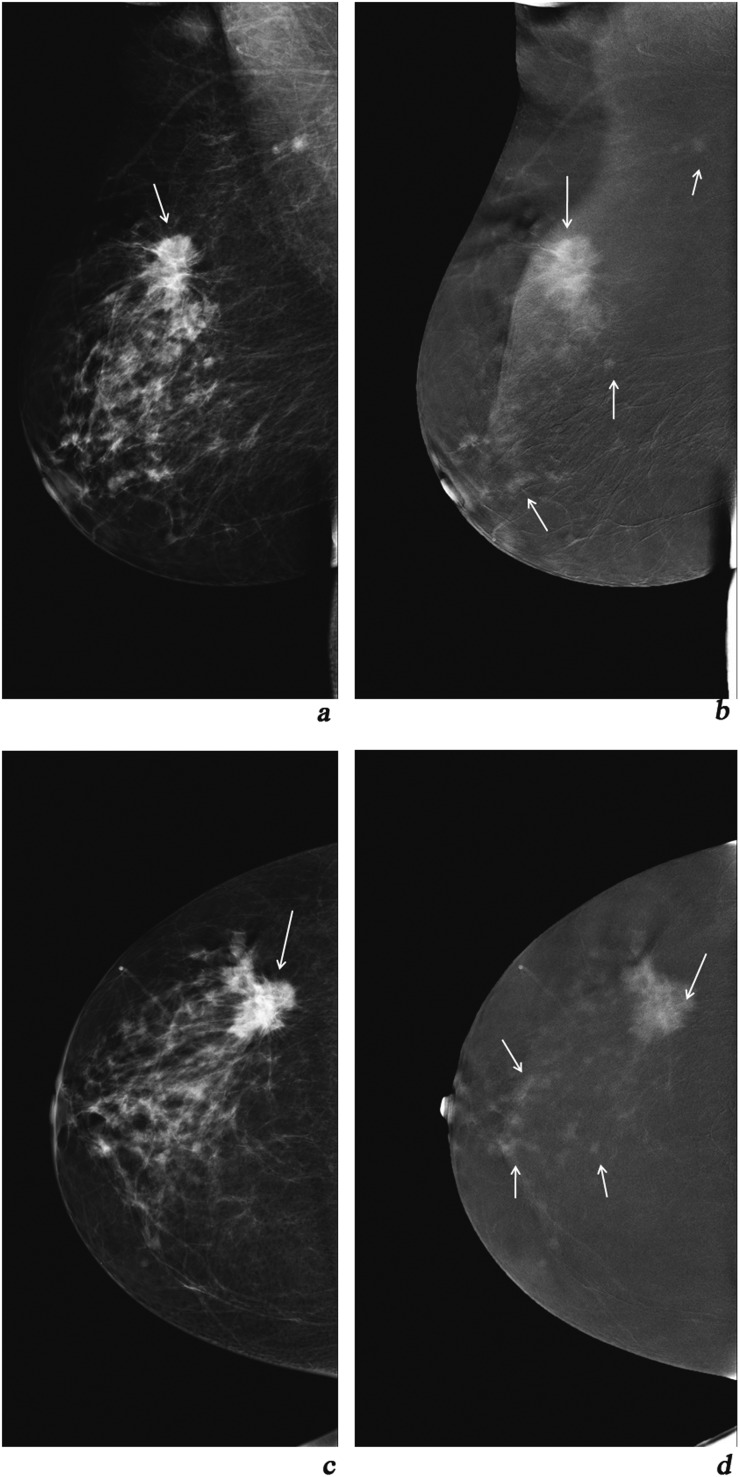
Figure 3.
(a, c) Mediolateral oblique (MLO) and craniocaudal (CC) views of digital mammography of the left breast depicting a breast imaging-reporting and data system 5 lesion; (b, d) MLO and CC views of contrast-enhanced spectral mammography depicting the lesion with Type 2 enhancement as well as many other Type 2 enhancement lesions which do not appear in digital mammography. MPS (big arrow) = 5 + 2 = 7. Histopathology result: multifoci lobular carcinoma grade III.
Table 1 shows malignancy rates in breast lesions for each MPS separately. Table 2 shows malignancy rates for MPS ≤ 4 and MPS > 4 cumulative.
Table 1.
Malignancy rates according to each MPS score separately
| MPS score | Benign (n = 128) | Malignant (n = 98) | Malignancy rate (%) |
|---|---|---|---|
| 2 | 8 | 0 | 0/8 (0) |
| 3 | 51 | 2 | 2/53 (3) |
| 4 | 44 | 6 | 6/50 (12) |
| 5 | 14 | 15 | 15/19 (52) |
| 6 | 9 | 31 | 31/40 (78) |
| 7 | 2 | 44 | 44/47 (94) |
Table 2.
Malignancy rates when 4 is used as best cut-off point
| MPS | Benign | Malignant | Malignancy rate (%) |
|---|---|---|---|
| ≤4 | 103 | 8 | 8/11 (7) |
| >4 | 25 | 90 | 90/115 (78.2) |
| Total | 128 | 98 | 226 |
When the best cut-off point 4 was used, the MPS method yielded 25 false-positive and 8 false-negative results. 9 of 25 false-positive scores were fibroadenomas, 1of 25 scores was radial scar, 1 of 25 scores was papilloma and 14 of 25 scores were sclerosing adenosis with or without microcalcifications and fibrocystic changes. 5 of 8 false negatives were in situ ductal carcinomas and 3 of 8 false negatives were invasive ductal carcinomas.
The AUC was bigger when the MPS was incorporated (AUC: 0.843, 0.888 and 0.917 for mammographic BIRADS score, CESM score and MPS, respectively, with p-value < 0.05), meaning that this new combined score of mammography BIRADS and CESM 4-step scale shows increased specificity without compromising sensitivity. As a result, accuracy rate was higher (85.40%) for the added value score (MPS) when comparing with each mode alone (73.89% for mammography and 80.97% for CESM) (Table 3). The diagnostic accuracy of MPS was superior to that of digital mammography or CESM alone.
Table 3.
Values of diagnostic tests
| Sum (MPS) | Mammogram | CESM | |
|---|---|---|---|
| Accuracy | 85.40% | 73.89% | 80.97% |
| Specificity | 80.47% | 59.37% | 71.31% |
| Sensitivity | 91.83% | 92.85% | 93.87% |
CESM, contrast-enhanced spectral mammography.
The cumulative score of BIRADS and CESM estimation (MPS) conferred a higher specificity rate of 80.47%. Specificity had lower rate in diagnostic mammography (59.37%), while in the CESM, it was 71.3%.
Mammography, CESM and sum values had comparable sensitivity rates to define malignant lesions, namely 92.85, 93.87 and 91.83%, respectively (Table 3).
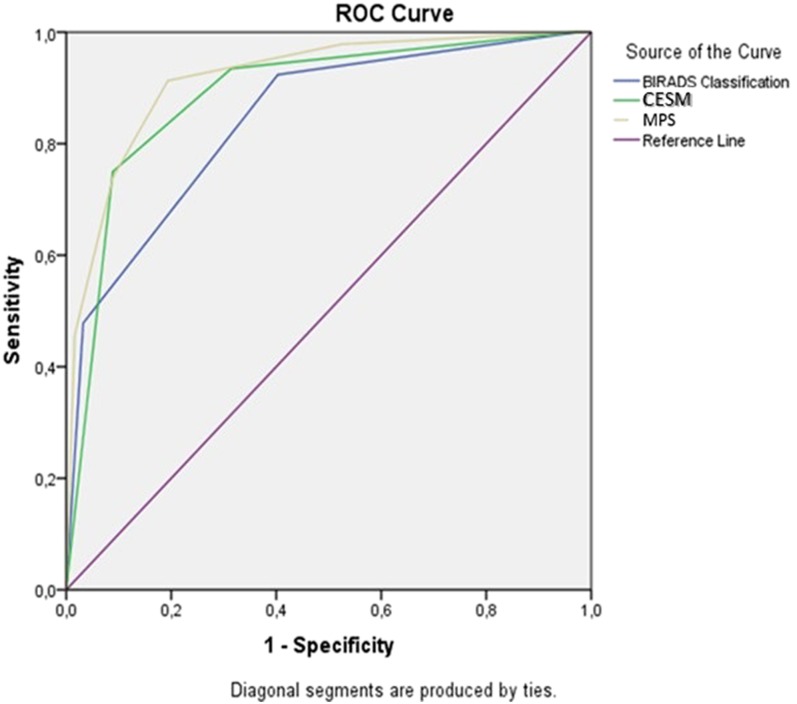
The overall diagnostic performance is shown in Figure 5 (ROC curve). Figure 5 illustrates the ROC curves of the three diagnostic methods. The statistical analysis showed statistically significant difference (p < 0.05) between the areas under the ROC curves of MPS and BIRADS (AUC: 0.917, 95% CI = 0.880–0.995, vs AUC = 0.843, 95% CI = 0.790–0.895).
Figure 5.
Receiver-operating characteristic curve for mammographic breast imaging-reporting and data system (BIRADS) classification, contrast-enhanced spectral mammography (CESM) classification and malignancy potential score (MPS). Area under the curve: 0.843, 0.888 and 0.917 for mammographic BIRADS, CESM and MPS, respectively; p-value < 0.05.
DISCUSSION
Mammography is a widely approved screening method with the primary end point of identifying the objects for further investigation,9 whereas CESM offers superior clinical performance than conventional digital mammography by decreasing false negatives, especially in females with dense breasts. CESM is cheap, can be quickly performed (total duration of the method is about 7 min) and is easily accessible to females.
The use of iodinated contrast agents is not completely devoid of risk, but most adverse side effects are rare and minor and have been decreased considerably with the use of low-osmolality contrast media.10 Patients with a history of allergic reactions to iodinated contrast agents, breast implants and renal insufficiency as well as pregnant or possibly pregnant females were excluded from this study.
Our study results showed that CESM presents higher diagnostic accuracy than digital mammography, 80.97 and 73.89%, respectively. This is in line with other studies reporting a higher diagnostic performance for CESM even when compared with a combination of mammography and ultrasound.11 The increased sensitivity (92.85%) of mammographic examination in our study is due to the fact that the study was designed to include only mammograms with findings.
In clinical practice, it is important to assess and quantify the improvement in the detection of a malignancy offered by these imaging modalities. From this point of view, our aim was to strengthen the BIRADS classification score incorporating two different scores: the standard BIRADS score that is based on well-established mammographic criteria14,15 and the CESM score that is based on enhanced areas of blood flow, according to our proposed scale (type of enhancement scale: −1, 0, 1, 2). The scale of enhancement arose by comparing lesion enhancement with background enhancement.
The sum served as malignancy potential score (MPS). To obtain the correct MPS, both the BIRADS and CESM scores must be evaluated correctly by experienced radiologists.
According to our study results, 4 was the best cut-off point that combined the optimal sensitivity (91.83%) and specificity (80.47%) and enabled the best distinction between benign and malignant lesions.
90 of 98 malignant lesions appeared with MPS > 4, whereas there were 8 false-negative results (5 in situ ductal carcinomas and 3 invasive ductal carcinomas); 103 of 128 benign lesions appeared with MPS ≤ 4, whereas there were 25 false-positive results (9 fibroadenomas, 1 radial scar, 1 papilloma and 14 sclerosing adenosis with or without microcalcifications and fibrocystic changes).
MPS has been proven to exert a higher diagnostic performance than each modality alone, as depicted in the area under the ROC curve in Figure 5. This finding is of great clinical value and suggests that MPS can be helpful for the differentiation of benign and malignant breast masses. Lesions with MPS ≤ 4 are very likely to be benign.
MPS is a diagnostic tool that seems to maintain and combine the advantages of both methods and to provide an accuracy benefit. Thus, MPS may improve patient selection for biopsy. MPS may provide fast and accurate breast lesion detection and characterization.
Further studies and software amelioration could expand the potential benefit of this combined modality.
Our study has some limitations. Firstly, benign but hyperplastic lesions, namely fibroadenomas or hyperplasia, may enhance with Type 1 enhancement (25 of 46 lesions with Type 1 enhancement were fibroadenomas or other hyperplastic lesions). Secondly, although two experienced radiologists evaluated the digital mammography and CESM findings and subsequently MPS, we did not evaluate intraobserver and interobserver variability.
CONCLUSION
Combining the well-documented experience of digital mammography with the power of type of enhancement of CESM into one score improves diagnostic accuracy compared with digital mammography or CESM alone. The “potential malignancy score” (MPS) is a new score which can be estimated quickly by the sum of BIRADS and CESM scores by experienced radiologists. According to our study results, MPS seems to be an efficient tool in clinical management. However, further studies with a larger number of patients are necessary to confirm these conclusions.
Contributor Information
Alexandra Tsigginou, Email: atsigginou@outlook.com.gr.
Christina Gkali, Email: chr.gkal@gmail.com.
Athanasios Chalazonitis, Email: red-rad@ath.forthnet.gr.
Eleni Feida, Email: elenifei@hotmail.com.
Dimitrios Efthymios Vlachos, Email: vlachos.dg@gmail.com.
Flora Zagouri, Email: florazagouri@yahoo.co.uk.
Ioannis Rellias, Email: giannisrellias@hotmail.com.
Constantine Dimitrakakis, Email: dimitrac@ymail.com.
REFERENCES
- 1.Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin 2013; 63: 11–30. doi: 10.3322/caac.21166 [DOI] [PubMed] [Google Scholar]
- 2.Mandelson MT, Oestreicher N, Porter PL, White D, Finder CA, Taplin SH, et al. Breast density as a predictor of mammographic detection: comparison of interval- and screen-detected cancers. J Natl Cancer Inst 2000; 92: 1081–7. doi: 10.1093/jnci/92.13.1081 [DOI] [PubMed] [Google Scholar]
- 3.Kolb TM, Lichy J, Newhouse JH. Comparison of the performance of screening mammography, physical examination, and breast US and evaluation of factors that influence them: an analysis of 27,825 patient evaluations. Radiology 2002; 225: 165–75. doi: 10.1148/radiol.2251011667 [DOI] [PubMed] [Google Scholar]
- 4.Mori M, Akashi-Tanaka S, Suzuki S, Daniels MI, Watanabe C, Hirose M, et al. Diagnostic accuracy of contrast-enhanced spectral mammography in comparison to conventional full-field digital mammography in a population of women with dense breasts Breast Cancer 2016. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 5.Dromain C, Thibault F, Muller S, Rimareix F, Delaloge S, Tardivon A, et al. Dual-energy contrast-enhanced digital mammography: initial clinical results. Eur Radiol 2011; 21: 565–74. doi: 10.1007/s00330-010-1944-y [DOI] [PubMed] [Google Scholar]
- 6.D'Orsi CJ, Sickles EA, Mendelson EB, Morris EA. Acr BI-RADS® Atlas, breast imaging reporting and data system. Reston, VA: American College of Radiology; 2013. [Google Scholar]
- 7.Robin X, Turck N, Hainard A, Tiberti N, Lisacek F, Sanchez JC, et al. pROC: an open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinformatics 2011; 12: 77. doi: 10.1186/1471-2105-12-77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 1988; 44: 837–45. doi: 10.2307/2531595 [DOI] [PubMed] [Google Scholar]
- 9.Tabár L, Vitak B, Chen HH, Yen MF, Duffy SW, Smith RA, et al. Beyond randomized controlled trials: organized mammographic screening substantially reduces breast carcinoma mortality. Cancer 2001; 91: 1724–31. [DOI] [PubMed] [Google Scholar]
- 10.Freund H, Biran S, Laufer N, Eyal Z. Letter: breast cancer arising in thoracotomy scars. Lancet 1976; 1: 97. doi: 10.1016/S0140-6736(76)90200-2 [DOI] [PubMed] [Google Scholar]
- 11.Badr S, Laurent N, Régis C, Boulanger L, Lemaille S, Poncelet E, et al. Dual-energy contrast-enhanced digital mammography in routine clinical practice in 2013. Diagn Interv Imaging 2014; 95: 245–58. doi: 10.1016/j.diii.2013.10.002 [DOI] [PubMed] [Google Scholar]
- 12.Evans TR, Stein RC, Ford HT, Gazet JC, Chamberlain GV, Coombes RC. Lactic acidosis. A presentation of metastatic breast cancer arising in pregnancy. Cancer 1992; 69: 453–6. [DOI] [PubMed] [Google Scholar]
- 13.Tzamicha E, Yakoumakis E, Tsalafoutas IA, Dimitriadis A, Georgiou E, Tsapaki V, Chalazonitis A. Dual-energy contrast-enhanced digital mammography: Glandular dose estimation using a Monte Carlo code and voxel phantom. Phys Med. 2015 Nov; 31(7): 785–91. [DOI] [PubMed] [Google Scholar]
- 14.Balleyguier C, Ayadi S, Van Nguyen K, Vanel D, Dromain C, Sigal R. BIRADS classification in mammography. Eur J Radiol 2007; 61: 192–4. doi: 10.1016/j.ejrad.2006.08.033 [DOI] [PubMed] [Google Scholar]
- 15.Boyer B, Canale S, Arfi-Rouche J, Monzani Q, Khaled W, Balleyguier C. Variability and errors when applying the BIRADS mammography classification. Eur J Radiol 2013; 82: 388–97. doi: 10.1016/j.ejrad.2012.02.005 [DOI] [PubMed] [Google Scholar]