Abstract
Introduction
Many ocular diseases require intravitreal injections of pharmacological agents. Optimizing patients’ experiences during injections is important to ensure compliance and maintenance of quality of life. The objective of this study was to identify strategies to help alleviate discomfort during intravitreal injections.
Methods
A cross-sectional study surveying 128 patients during clinic visits between 2014 and 2015 in two outpatient Retina Clinics (one academic and one private). Patients receiving an intravitreal injection(s) for any retinal disorder were given a questionnaire with 10-yes/no responses for various potential strategies. Responses were stratified by sex, age (<30 years, 30–60 years, and >60 years) and total number of prior injections (0–9 injections, 10–20 injections and >20 injections).
Results
A total of 128 patients were surveyed: 59 males, 41 females and 28 with no sex specified. Our results identified four favorable strategies as those receiving more than 50% “yes” votes. These included the presence of technician/staff during the procedure, the use of a neck pillow, a verbal warning before the injection and performing injections in both eyes on the same day. Other specific strategies were identified for females, younger patients and those with greatest experience. These included: females preferred having their hand held during injections (P = 0.001) and using a stress ball (P = 0.000) when compared to males. Stratifying by age, patients 30–60 years old preferred having their hand held (P = 0.008) and background music (P = 0.007). Stratifying by prior injections, patients with >20 prior injections preferred having their hand held (P = 0.001), using a stress ball (P = 0.021) and, if necessary, having bilateral injections performed the same day to improve comfort (P = 0.037).
Conclusions
Having an extra staff member present during the injection, having a neck pillow, having a verbal warning prior to injection and having both eyes injected on the same day were indicated as favorable strategies by over half of those surveyed. Further, specific strategies were identified for females, younger patients (30–60 years old) and those with greatest experience (>20 injections).
Keywords: Comfort, Drug delivery, Intravitreal injection, Patient, Quality improvement, Retinal disease
Introduction
Intravitreal injections provide an effective method for the administration of medications in the treatment of many different ocular diseases, including: age-related macular degeneration, diabetic retinopathy, macular edema, branch retinal vein occlusions, and ophthalmic infections [1–5]. As the number of conditions for which these injections are employed continues to grow, so does the frequency of their administration to patients in the outpatient clinic setting. For example, there was a 558-fold increase in the total number of intravitreal injections performed in Medicare patients, going from 4125 injections in 2001 to 2,354,753 injections administered in 2012 alone [6].
Despite the safety and efficacy of intravitreal injections, some patients may experience significant discomfort and anxiety while undergoing the procedure. Several studies have explored pain-reducing strategies for intravitreal injections. Two strategies frequently studied in the literature are method of anesthesia and needle gauge size. Choice of anesthetic can be viewed as the most direct way to reduce discomfort during an intravitreal injection; however, no consensus has been reached as to what type is superior overall. Three methods commonly employed are topical proparacaine drops, topical lidocaine gel and sub-conjunctival lidocaine injections. A recent study concluded that subconjunctival lidocaine was most effective in preventing pain and eye movement during injections [7]. Another study found no statistical significance between many various methods including lidocaine gel, lidocaine-soaked pledget, and topical drops, yet it advocated for the use of topical proparacaine drops given their cost effectiveness [8].
A similar trend can be seen when comparing intravitreal needle gauge size and pain reduction. One study that compared differences in pain during the injection of dexamethasone intravitreal implant (Ozurdex®, Allergan) and bevacizumab found that despite the larger needle gauge and the tunneled injection technique required to inject the dexamethasone intravitreal implant, there was no significant difference in reported pain levels between the two injections [9]. After review of the current literature, no consensus appears to exist regarding intravitreal needle gauge size and the amount of pain experienced by the patient. One study showed no difference in pain score levels between 30 and 27 gauge needles during intravitreal injections [10]. Another study comparing the two gauges found that 30 gauge needles were associated with less pain in a subset of patients studied, those less than 65 years old [11]. One study analyzing 33 vs. 30 gauge needles did not result in lower pain levels [12]. Interestingly, this study identified factors other than needle size that accounted for pain, including: distress, expectation of pain and discomfort, female gender and anticipating negative consequences [12].
Thus, addressing pain-inducing factors outside of the technical administration of injections is an important concept that may play a role in the reduction of discomfort in many patients. Our study focuses on identifying potential strategies other than type of anesthesia or injection needle gauge size for alleviating discomfort. As an added component, we directly solicited patient input from their own experience with intravitreal injections.
Very few studies have addressed alternative methods to reduce discomfort. One such study found that playing music decreased anxiety during intravitreal injections [13]. In the literature, the use of comfort strategies has been explored in much more detail for non-ocular procedures. For instance, evidence suggests that the presence of additional people, whether they are family members or additional staff, may divert attention away from pain during procedures such as venipuncture in children [14]. Handholding has likewise been shown to be a very effective coping strategy in pain perception during procedures such as blood draws, shunt placements, and peripheral chemotherapy [15]. Stress balls have been shown to be effective in reducing pain during outpatient surgeries such as endovenous thermal ablation and phlebectomies of varicose veins [16]. Physical comfort can reduce patient anxiety and stress as well as pain during peripheral intravenous cannulation, which led us to infer that providing patients with a neck pillow might be useful for intravitreal injections [17]. Use of a verbal warning was also investigated, but was not found to be very helpful in intravenous cannulation [18]. The goal of this study was to incorporate and evaluate strategies previously proven to minimize discomfort in several minor surgical procedures as well as to identify new strategies that may increase comfort in patients receiving repeat intravitreal injections.
Methods
This study was conducted at the University of Minnesota, Department of Ophthalmology Retina Clinic and at a private retina clinic, the Edina Retina Consultants. This prospective, survey-based study was approved by the Institutional Review Board (IRB) of the University of Minnesota. This study is Health Insurance Portability and Accountability Act (HIPAA) compliant, and informed consent was obtained by all participants. This study adheres to the tenets of the Declaration of Helsinki. The selection criteria included patients who were undergoing current treatment with intravitreal injections or who were going to receive their first injection at that visit. The survey was given to the patient on the same day they were receiving their injection. In our study, the anesthetic patients received varied. The majority of our patients received topical proparacaine drops only; the next most common anesthetic was lidocaine jelly, followed by subconjunctival lidocaine.
Participants were administered a ten question survey where they were prompted to indicate their preferences (by checking “yes” or “no”) for or against potential strategies to improve comfort during their intravitreal injection(s). The survey also included a section where patients could write in any other suggestions they had regarding the process of care during intravitreal injections. The survey also recorded patient age, sex and number of previous number of injections (given pre-selected ranges to choose from). Below is the list of questions included in the survey.
Having the exam room lights dimmed as much as possible.
Having a family member, friend, or significant other present during the injection.
Having a technician or staff member, in addition to the physician, present during the injection.
Having someone hold your hand during the injection.
Having a stress ball to hold during the injection.
Having music played in the background during the injection.
Having a pillow placed under neck.
Having a verbal warning or notice given just prior to the injection.
If both the eyes need to be injected, would you prefer them both to be injected on the same day?
If both the eyes need to be injected, would you prefer them both to be injected on different days?
This is an observational study where only limited descriptive analysis was performed. Qualitative statistical analysis was performed using IBM SPSS 21 Statistics software (Armonk, NY, USA). Stratification was done using Chi-squared test and Pearson coefficients. Results were measured in percentage of patients who indicated “yes” or “no” to each individual strategy. We defined favorable strategies to be those for which more than 50% of patients indicated “yes” as a response.
Results
A total of 128 patients completed the questionnaire. The questionnaire included responses from 59 males, 41 females and 28 patients with no sex indicated. Forty-three patients had previously received 0–9 injections, 20 patients had received 10–20 injections, 43 patients had received more than 20 injections, and 22 patients with no specification. Of those surveyed, none were <30 years old, 5 patients were between the ages of 30–60, 78 patients were >60 years old and 45 patients were unspecified. Patients listed their diagnosis requiring injection(s) as follows: 85 reported macular degeneration, 7 reported diabetic retinopathy, 2 reported histoplasmosis, 2 reported macular edema, 1 reported Best disease, and 25 did not specify a diagnosis. Only one diagnosis was listed for each patient.
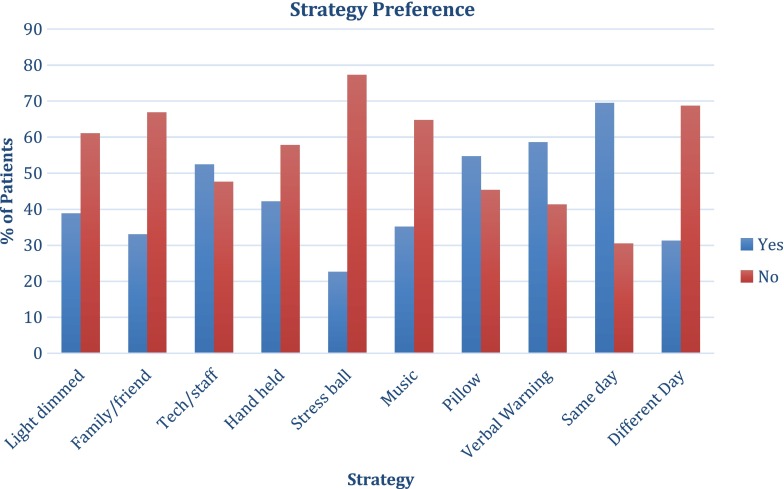
Figure 1 lists the percentage of subjects that voted “yes” for each strategy; we defined favorable strategies as those receiving more than 50% “yes” votes. Favorable strategies included the presence of Tech/Staff during the procedure, the use of a pillow, a verbal warning before the injection and performing injections in both eyes on the same day.
Fig. 1.
Illustrates patients’ preference to each of the individual strategies surveyed. Survey responses were given in a “yes” or “no” format. X-axis represents each proposed strategy and y-axis refers to the percentage of patients’ responses
The responses revealed significant differences when stratified by sex, age, and number of total injections received. Stratifying by sex (Table 1), females reported statistically significant preference for hand holding (P = 0.001) and using a stress ball (P = 0.000) when compared to males. Stratifying by age (Table 2), patients 30–60 years old reported statistically significant preference for hand holding (P = 0.008) and having background music (P = 0.007). Stratifying by the number of prior injections (Table 3), patients who had received >20 injections reported statistically significant preferences for hand holding (P = 0.001), using a stress ball (P = 0.021), and, if necessary, having bilateral injections performed the same day (P = 0.037).
Table 1.
Positive preferences for specific strategies stratified by patients’ sex
| Strategy | Male (% yes) | Female (% yes) | P value (sig < 0.05) |
|---|---|---|---|
| Light dimmed | 32.8 | 52.5 | 0.051 |
| Family/friend | 39.7 | 29.3 | 0.287 |
| Tech/staff | 44.1 | 61.5 | 0.090 |
| Hand held | 27.1 | 61.0 | 0.001 |
| Stress ball | 8.5 | 41.5 | 0.000 |
| Music | 32.2 | 43.9 | 0.233 |
| Pillow | 61.0 | 68.3 | 0.456 |
| Verbal warning | 54.2 | 73.2 | 0.055 |
| Same day | 71.2 | 63.4 | 0.413 |
| Different day | 30.5 | 36.6 | 0.525 |
Table 2.
Positive preferences for specific strategies stratified by patients’ age
| Strategy | 30–60 y/o (% yes) | >60 y/o (% yes) | P value (sig < 0.05) |
|---|---|---|---|
| Light dimmed | 80 | 39.5 | 0.075 |
| Family/friend | 40 | 35.1 | 0.823 |
| Tech/staff | 75 | 49.4 | 0.317 |
| Hand held | 100 | 39.7 | 0.008 |
| Stress ball | 40 | 21.8 | 0.348 |
| Music | 100 | 38.5 | 0.007 |
| Pillow | 100 | 67.9 | 0.130 |
| Verbal warning | 100 | 59.0 | 0.068 |
| Same day | 60 | 69.2 | 0.666 |
| Different day | 40 | 32.1 | 0.713 |
Table 3.
Preferences for specific strategies stratified by number of previous injections
| Strategy | 0–9 injections (% yes) | 10–20 injections (% yes) | >20 injections (% yes) | P value (sig < 0.05) |
|---|---|---|---|---|
| Light dimmed | 45.2 | 30 | 38.1 | 0.504 |
| Family/friend | 35.7 | 40 | 30.2 | 0.725 |
| Tech/staff | 54.8 | 45 | 57.1 | 0.661 |
| Hand held | 23.3 | 50 | 62.8 | 0.001 |
| Stress ball | 16.3 | 10 | 37.2 | 0.021 |
| Music | 41.9 | 30 | 30.2 | 0.463 |
| Pillow | 65.1 | 75 | 48.8 | 0.101 |
| Verbal warning | 62.8 | 70 | 53.5 | 0.421 |
| Same day | 55.8 | 70 | 81.4 | 0.037 |
| Different day | 44.2 | 30 | 20.9 | 0.068 |
Our survey also included a section in which patients could write in their own suggestions. Those that were commonly repeated are listed below:
Giving the injection quickly (10 patients).
Thoroughly numbing the eye prior to injection (4 patients).
Thorough eye flushing after injection (3 patients).
Minimal use of Betadine (just on eyelashes when eye is closed) (2 patients).
Discussion
Overall, tabulation of our patients’ preferences indicated four different strategies were favorable in increasing comfort during intravitreal injections. The most preferred strategy overall was having both eyes injected (69.5% of patients, Fig. 1) on the same day if bilateral injections were indicated. We found this surprising, given the informed consent process patients undergo prior to receiving an injection. From this, it can be inferred that patients understand the theoretical increased risk in having both eyes injected on the same day. This strong preference for same-day bilateral injections suggests that patients would rather accept the increased risk over the inconvenience of a return visit for injection of the opposite eye. This speaks to the burden on patients’ lifestyle of having periodic eye injections.
Other favorable strategies, preferred by >50% of patients surveyed, such as having an additional staff member or technician present in the room, in addition to the treating physician, a pillow placed underneath their head during the injection and verbal warning or count down prior to the injection, confirmed what had been previously reported in the literature in other non-ophthalmological studies related to minimize patient’s discomfort [13–18].
Interestingly, when stratified by age, sex, and number of injections, common findings were observed in all three categories. Specifically, females, patients between 30 and 60 years old and those who had received >20 injections all preferred having their hand held during the injection. A stress ball was a preferred strategy amongst females and those patients who had undergone over 20 previous injections. Younger patients (30–60 years old) preferred having background music playing during the injection. The clinical significance of these results is to demonstrate that different strategies can be tailored to certain patients based on age, sex and number of prior injections.
Patients with the most experience with injections (>20 injections) preferred having both eyes injected on the same day in comparison with those who had received fewer injections. It may be inferred that patients become more comfortable with these injections over time. As patients have successful injections with minimal to no complications, they may begin to feel more comfortable taking the risk of bilateral same-day injections in exchange for the benefit of reducing their number of clinic visits.
The demographics of our study are closely in line with those of the RVS Update Committee Database, where 93.9% were white, 67% female, and 77% over the age of 75 [6]. We feel this gives additional value to the strategies identified since they represent the patient population that frequently receive intravitreal injections.
Our goal moving forward is to implement the four strategies that were preferred by >50% of patients surveyed in our outpatient clinical practice. Some of these strategies have already been implemented by our retina specialists. Moreover, they have acknowledged a positive response by their patients, noting that by identifying patients’ personal preferences they have increased overall patient satisfaction with their eye care.
Future studies include increasing the number of patients surveyed, especially those in the younger age range, having a more equal representation of males and females and also increasing the number of patients in the 10–20 injections group. The main limitation of the study is the lack of a post-implementation survey in a randomized controlled trial to address patient’s satisfaction and to confirm the efficacy of these strategies.
Conclusion
Our study identified four main strategies that increased comfort in patients receiving intravitreal injections: same-day bilateral injection, when indicated, the presence of an extra staff member during the injection, the placement of a pillow under their neck and a verbal warning immediately before injection. Additional specific strategies were identified for females, younger patients (30–60 years old) and those with greatest experience (>20 injections). As the number of indications for intravitreal injections continues to expand, so too does the frequency of their administration. We are hopeful that the implementation of strategies to minimize patient discomfort can contribute to increased compliance with intravitreal injections and thus reduce the risk of further vision loss.
Acknowledgments
This research was supported by the Minnesota Lions Vision Foundation. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval for the version to be published.
Disclosures
J. Gomez, D. Koozekanani, A. Feng, M. Holt, P. Drayna, M. Mackley, F. van Kuijk, R. Beardsley, R. Johnston, J. Terry, S. R. Montezuma have nothing to disclose.
Compliance with Ethics Guidelines
This study was conducted at the University of Minnesota, Department of Ophthalmology Retina Clinic and at a private retina clinic, the Edina Retina Consultants. This prospective, survey-based study was approved by the Institutional Review Board (IRB) of the University of Minnesota. This study is HIPAA compliant, and informed consent was obtained by all participants. This study adheres to the tenets of the Declaration of Helsinki.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Footnotes
Enhanced content
To view enhanced content for this article go to http://www.medengine.com/Redeem/A9E4F0602982B2D6.
References
- 1.Friedman DS, O’Colmain BJ, Munoz B, et al. Prevalence of age-related macular degeneration in the United States. Arch Ophthalmol. 2004;122:564–572. doi: 10.1001/archopht.122.7.1019. [DOI] [PubMed] [Google Scholar]
- 2.Ala-Kauhaluoma M, Aho I, Ristola M, Karma A. Involvement of intraocular structures in disseminated histoplasmosis. Acta Ophthalmol. 2010;88:493–496. doi: 10.1111/j.1755-3768.2008.01454.x. [DOI] [PubMed] [Google Scholar]
- 3.World Health Organizaton. Causes of blindness and visual impairment. Available at http://www.who.int/. Accessed 20 Dec 2014.
- 4.Campochiaro P, Clark W, Boyer D, et al. Intravitreal aflibercept for macular edema following branch retinal vein occlusion: the 24-week results of the VIBRANT study. Ophthalmology. 2014;122:538–544. doi: 10.1016/j.ophtha.2014.08.031. [DOI] [PubMed] [Google Scholar]
- 5.Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group Ranibizumab and bevacizumab for treatment of neo-vascular age-related macular degeneration: two year results. Ophthalmology. 2012;119:1388–1398. doi: 10.1016/j.ophtha.2012.03.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.American Medical Association. AMA/Specialty Society RVS Update Committee Database: RBRVS DataManager Online. Available at https://commerce.ama-assn.org/store/catalog/productDetail.jsp?product_id=prod280002&navAction=push. Accessed 20 Dec 2014.
- 7.Andrade GC, Carvalho AC. Comparison of 3 different anesthetic approaches for intravitreal injections: a prospective randomized trial. Arq Bras Oftalmol. 2015;78(1):27–31. doi: 10.5935/0004-2749.20150008. [DOI] [PubMed] [Google Scholar]
- 8.Davis MJ, Pollack JS, Shott S. Comparison of topical anesthetics for intravitreal injections: a randomized clinical trial. Retina. 2012;32(4):701–705. doi: 10.1097/IAE.0b013e31822f27ca. [DOI] [PubMed] [Google Scholar]
- 9.Moisseiev E, Regenbogen M, Rabinovitch T, Barak A, Loewenstein A, Goldstein M. Evaluation of pain during intravitreal Ozurdex injections vs intravitreal bevacizumab injections. Eye. 2014;28:980–985. doi: 10.1038/eye.2014.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Haas P, Falkner-Radler C, Wimpissinger B, Malina M, Binder S. Needle size in intravitreal injections—pain evaluation of a randomized clinical trial. Acta Ophthalmol. 2016;94(2):198–202. doi: 10.1111/aos.12901. [DOI] [PubMed] [Google Scholar]
- 11.Mete G, Burak B, Musa Ç, Ali Ş, Şemsettin B. Assessment of patient pain experience during intravitreal 27-gauge bevacizumab and 30-gauge ranibizumab injection. Korean J Ophthalmol. 2015;29:190–194. doi: 10.3341/kjo.2015.29.3.190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.van Asten F, van Middendorp H, Verkerk S, Breukink MB, Lomme RM, Hoyng CB, Evers AW, Klevering BJ. Are intravitreal injections with ultrathin 33-G needles less painful than the commonly used 30-G needles? Retina. 2015;35(9):1778–1785. doi: 10.1097/IAE.0000000000000550. [DOI] [PubMed] [Google Scholar]
- 13.Chen X, Seth R, Rao V, Huang J, Adelman R. Effects of music therapy on intravitreal injections: a randomized clinical trial. J Ocul Pharmacol Ther. 2012;28:414–419. doi: 10.1089/jop.2011.0257. [DOI] [PubMed] [Google Scholar]
- 14.Gupta H, Vardhan V, Kaur A, Singla R, et al. Comparison between the analgesic effect of two techniques on the level of pain perception during venipuncture in children up to 7 years of age: a Quasi-Experimental Study. J Clin Diagn Res. 2014;8:PC01–PC04. doi: 10.7860/JCDR/2014/9731.4675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Weekes DP, Kagan SH, James K, Seboni N. The phenomenon of hand holding as a coping strategy in adolescents experiencing treatment-related pain. J Pediatr Oncol Nurs. 1993;10:19–25. doi: 10.1177/104345429301000105. [DOI] [PubMed] [Google Scholar]
- 16.Hudson B, Ogden J, Whiteley M. Randomized controlled trial to compare the effect of simple distraction interventions on pain and anxiety experienced during conscious surgery. Eur J Pain. 2015;19:1447–1455. doi: 10.1002/ejp.675. [DOI] [PubMed] [Google Scholar]
- 17.Tee F, Low C, Matizha P. Perceptions and experience of pain, anxiety and comfort during peripheral intravenous cannulation in medical wards: topical anaesthesia, effective communication, and empowerment. Int J Nurs Sci. 2015;5:41–46. [Google Scholar]
- 18.Gupta J, Bown T, Cyna A. Effect of communication on pain during intravenous cannulation: a randomized controlled trial. Br J Anesth. 2007;99:871–875. doi: 10.1093/bja/aem308. [DOI] [PubMed] [Google Scholar]