Abstract
Background
The majority of differentiated thyroid cancer tends to present with limited locoregional disease, leading to excellent long-term survival after operative treatment. Even patients with advanced local disease may survive for long periods with appropriate treatment. The aim of this study is to present our institutional experience of the management of locally advanced differentiated thyroid cancer and to analyze factors predictive of outcome.
Methods
We reviewed our institutional database of 3,664 previously untreated patients with differentiated thyroid cancer operated between 1986 and 2010. A total of 153 patients had tumor extension beyond the thyroid capsule that invaded the subcutaneous soft tissues, recurrent laryngeal nerve, larynx, trachea, or esophagus. Details on extent of operation and adjuvant therapy were recorded. Disease-specific survival and locoregional recurrence-free probability were determined by the Kaplan-Meier method. Factors predictive of outcome were determined by multivariate analysis.
Results
The median age of the 153 patients with tumor extension beyond the thyroid capsule was 55 years (range 11–91 years). Eighty-nine patients (58.2%) were female. Twenty-three patients (15.0%) were staged as M1 at presentation, and 122 (79.7%) had pathologically involved lymph nodes. The most common site of extrathyroidal extension was the recurrent laryngeal nerve (51.0%) followed by the trachea (46.4%) and esophagus (39.2%). Sixty-three patients (41%) required resection of the recurrent laryngeal nerve due to tumor involvement. After surgery, 20 patients (13.0%) had gross residual disease (R2), 63 (41.2%) had a positive margin of resection (R1), and 70 (45.8%) had complete resection with negative margins (R0). With a median follow-up of 63.9 months, 5-year, disease-specific survival, when stratified by R0/R1/R2 resection, was 94.4%, 87.6%, and 67.9%, respectively (P = .030). The data do not demonstrate a statistical difference in survival between R0 versus R1 (P = .222). The 5-year distant recurrence-free probability for M0 patients was 90.8%, 90.3%, and 70.7% (P = .410). The locoregional recurrence-free probability was 85.8% for R0 patients and 85.5% for R1 patients (P = .593).
Conclusion
With an appropriate operative strategy, patients with locally advanced thyroid cancer with an R0 or R1 resection have excellent survival outcome.
The Majority of Patients With Differentiated Thyroid Cancers (DTCs) tend to present with limited locoregional disease1-3 and have excellent long-term outcome.1,4,5 The incidence of DTC in the United States is increasing,6,7 and this has been attributed to increased detection of early stage disease. In addition to an increase in these low-risk cases, a simultaneous increase in the number of larger tumors (>4 cm) with adverse features, such as extrathyroid extension (ETE), has been noted by some, but the cause of this increased incidence, however, is unclear.8,9 Although locally advanced DTC with gross ETE is rare, when detected, it presents an operative challenge for both the clinician and patient. The aim of treatment is to control disease in the central compartment by removing all gross tumor followed by adjuvant radioactive iodine (RAI) and in select cases, external beam radiation therapy (EBRT).10
It is well recognized that completeness of resection is critical and that an operation that achieves negative margins (R0) gives the best chance of cure.11 In locally advanced DTC, involvement of the upper aerodigestive tract does not always necessitate resection of the larynx or esophagus. In the absence of mucosal involvement, partial thickness resection of esophageal muscle and shave of tracheal cartilage to achieve R0/1 margins may be adequate in select patients to avoid the morbidity of more extensive resection. Debate continues, however, in the literature regarding the need for radical resection, with some authors reporting improved results related to more aggressive resections, while others report similar results for carefully selected, conservative operative procedures as long as all gross tumor is removed (R0/R1).11-20 The aim of this study is to report our experience at the Memorial Sloan Kettering Cancer Center (MSKCC) with the management of locally advanced DTC and to further analyze factors predictive of outcome within this group.
Patients and Methods
A retrospective review of a single institutional operative database of 3,664 previously untreated, consecutive DTC patients identified 153 patients (4.2%) with pT4 DTC between 1986 and 2010. Patients who underwent treatment elsewhere prior to referral and those considered to have inoperable disease at the time of the operation were excluded. Data collection included patient demographics and operative details, including the presence of gross ETE. Histopathologic details included tumor histology, primary tumor size, presence and extent of ETE, histologic margin status, and the presence of metastatic lymph nodes. Postoperative treatment details regarding the use of RAI and EBRT were also captured.
T4a disease was defined by the International Union Against Cancer (UICC) as a tumor of any size extending beyond the thyroid capsule to invade subcutaneous soft tissues, larynx, trachea, esophagus, and recurrent laryngeal nerve (RLN), and T4b was defined as extension into the carotid artery, great vessels, or prevertebral fascia.21 Minimal ETE limited to the sternothyroid muscle or perithyroid soft tissues is considered T3 disease.
For the purposes of this study, operative findings were taken as the primary indicator of ETE and required confirmation on histopathology. Completeness of resection was determined from the operative and pathology reports; a resection was called R0 in patients with no gross residual disease and negative margins on histopathology; R1 was assigned in patients with no gross residual disease but positive margins on histopathology; and R2 was assigned for patients with gross residual disease irrespective of the histopathologic margin.
At our institution, we do not carry out prophylactic central neck or lateral neck dissection in patients with a clinically N0 neck. Assessment of the lateral neck is performed by palpation at the preoperative clinic visit or by preoperative ultrasonography. The central compartment is assessed by intraoperative evaluation of the central compartment lymph nodes at the time of thyroidectomy. If no suspicious nodes are present in the central compartment, then an elective central neck dissection is not performed. If nodes are palpable or confirmed to have cancer on frozen section, then a central compartment neck dissection is carried out.
Assessment of the presence of distant metastases (M1) was based on evidence of metastases prior to an operation (on imaging or biopsy) or detection on postoperative RAI scanning (within 6 months of the initial operation). Postoperative thyroid stimulating hormone suppression was practiced for all patients, aiming for a thyroid stimulating hormone level of between 0.1–0.5 mU/L.
Local and regional recurrences were determined by clinical examination supplemented with imaging and fine-needle aspiration biopsy if recurrence was suspected. Locoregional recurrence was defined as recurrent disease located in the operated thyroid bed or cervical lymph nodes. Distant recurrence was determined by imaging studies, including radioiodine uptake scans, computed tomography (CT), positron emission tomography scans, or cytologic and histopathologic evidence, where available. During the study period, serum thyroglobulin and annual ultrasound (US) were not used routinely to detect recurrence; however, this approach is now standard practice.
Disease-specific outcomes were calculated using the date of last follow-up with the disease management team at MSKCC. Overall survival outcomes were not analyzed due to the relatively slow disease progression in thyroid cancer. Details of death were determined from the social security index and hospital records. All patients who had evidence of active structural disease at the time of last follow-up and who died during follow-up were considered to have died of disease.
Disease-specific survival (DSS) was calculated from the time of cancer diagnosis to the date of first disease recurrence. Distant recurrence-free probability (DRFP) was calculated from diagnosis to date of first distant disease recurrence, while locoregional recurrence-free probability (LRRFP) was calculated from diagnosis to the date of first thyroid bed or cervical lymph node recurrence. The median follow-up was 64 months (range 1–290 months). Outcomes data were calculated at 5 years. Recurrence events were recorded for all patients. Because progression of local disease in patients who had an R2 resection is not comparable with patients with R0 and R1 resections, patients with R2 resections were excluded from analysis of locoregional recurrence.
Statistical analysis was carried out using SPSS (Version 21, IBM Corp, Armonk, NY). Variables were compared between R0, R1, and R2 groups using the Pearson χ2 test. DSS, DRFP, and LRRFP were analyzed using the Kaplan-Meier method. Factors predictive of outcome were determined by univariate analysis using the log-rank test and by multivariate analysis using the Cox proportional hazards method.
Results
The median age of the 153 patients with extrathyroidal DTC was 55 years (range 11–91 years), 64 men (41.8%) and 89 women (58.2%). Twenty-three patients were M1 (15.0%), and 130 patients were considered M0 (85.0%) after initial therapy. Nine of the M1 patients were identified preoperatively (39.1%) and 14 postoperatively on RAI scan within 6 months of thyroidectomy (60.1%).
Operative management
A total of 140 patients underwent total thyroidectomy (91.5%), 8 had thyroid lobectomy (5.2%), and 5 patients (3.3%) underwent subtotal thyroidectomy with a portion of the contralateral lobe preserved. Less than total thyroidectomy was performed to avoid damage to the contralateral recurrent nerve after an extensive ipsilateral dissection. On histopathology, the median size of the primary lesion was 3 cm (range 0.6–9 cm). There were 3 cases of Hurthle cell carcinoma (2.0%), and the remaining cases were papillary carcinoma in 150 (98.0%) patients, of which 50 were tall cell variant.
The extrathyroidal structures involved by local extension are described in Fig 1. The details of the operative management of the surrounding structures involved by disease are detailed in Table I. Sixty-four patients (41.8%) required resection of the RLN; 11 (7.2%) had full-thickness, partial, or circumferential tracheal resections; 1 required cervical esophagectomy; and 5 (3.2%) had partial or total laryngectomy. All 7 sleeve resections of the trachea were repaired with primary, end-to-end anastomosis. Of these, 2 required a temporary tracheostomy. One patient had a total laryngectomy with cervical esophagectomy, reconstructed using a jejunal, free tissue transfer. In addition, 58 patients (37.9%) had partial thickness tracheal shave excision, 57 (37.3%) had partial thickness esophageal or pharyngeal excision of the muscular wall, and 41 patients (26.8%) had partial thickness excision of part of the laryngeal structure.
Figure 1.
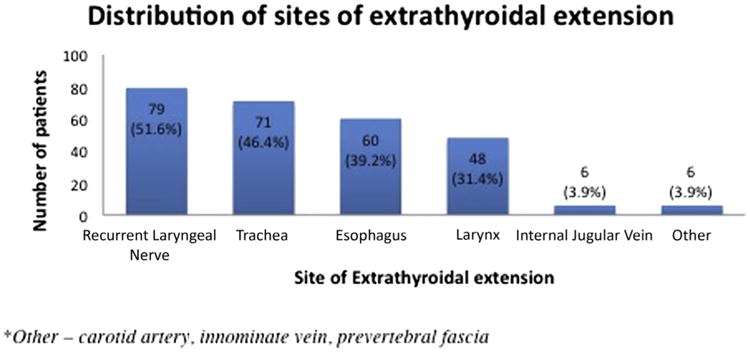
Sites of extrathyroidal extension in 153 patients with T4 differentiated thyroid cancer. Other indicates carotid artery, innominate vein, and prevertebral fascia.
Table I. Operative management of involved structures in 153 patients with T4 differentiated thyroid cancer.
| Structure involved | Procedure | n (%) |
|---|---|---|
| Recurrent laryngeal nerve | Not resected | 15 (19.0%) |
| Resected | 64 (81.0%) | |
| Trachea | None (gross residual disease left) | 2 (2.8%) |
| Cartilage shave | 58 (81.7%) | |
| Partial resection | 4 (5.6%) | |
| Sleeve resection | 7 (9.9%) | |
| Esophagus/pharynx | Not resected | 2 (3.3%) |
| Muscle wall excision | 57 (95%) | |
| Cervical esophagectomy | 1 (1.7%) | |
| Larynx | None (gross residual disease left) | 2 (4.2%) |
| Shave excision | 41 (85.4%) | |
| Partial laryngectomy | 3 (6.2%) | |
| Total laryngectomy | 2 (4.2%) | |
| Internal jugular vein | Not resected | 1 |
| Resected | 5 | |
| Other | ||
| Carotid artery | Shave excision | 3 |
| Innominate vein | Sternotomy and resection | 1 |
| Prevertebral fascia | Excision and resection of sympathetic trunk | 2 |
In addition to the thyroid operation, 27 patients (17.6%) had a central neck dissection alone, 18 (11.8%) had a lateral neck dissection alone, 63 (41.2%) had a central and lateral neck dissection, and 45 (29.4%) had no neck dissection. Thirty-one patients (21.3%) were classified as pN0/X, 43 (28.1%) were pN1a, and 79 (51.6%) were pN1b.
Of the 153 patients, 20 (13.1%) had gross residual disease at the completion of the operation (R2 resection). Of the 133 patients who were considered free of gross disease, 63 (41.2%) had disease present at the operative margins (R1 resection), and 70 (45.8%) had negative margins (R0 resection).
Adjuvant therapy
After the operation, 12 patients (7.8%) received no adjuvant therapy, 123 (80.4%) received postoperative RAI only, 5 (3.3%) received EBRT only, and 13 (8.5%) received both RAI and EBRT. EBRT was used after 10% (7/70) of R0 resections, 11% (7/63) of R1 resections, and 20% (4/20) of R2 resections. Although patients with R2 resections are recommended routinely to undergo EBRT, several R2 patients did not receive EBRT despite this recommendation because of medical comorbidities or patient refusal.
Oncologic outcomes
With a median follow-up of 63.9 months (range 1–290 months), 49 patients died, 23 of whom died with active disease. There were 5 local recurrences, 23 cervical nodal recurrences, and 24 distant recurrences.
For patients who were cleared of all gross disease at the end of an operation (R0/R1 and M0), the 5-year DSS, LRRFP, and DRFP were excellent at 97.8%, 86.3%, and 90.6%, respectively. The overall DSS for M1 patients was poorer compared with M0 patients (46.9% vs 97.1%, P < .001). Similarly, when stratified by R status (R0/R1/R2), the 5-year DSS was poorer for patients with residual disease at 94.4%, 87.6%, and 67.9%, respectively (P = .030; Fig 2, A). Factors predictive of DSS are shown in Table II; age >45 years, tumor size >4 cm, R2 resection, and the presence of distant metastases were predictive of worse DSS on univariate analysis.
Figure 2.
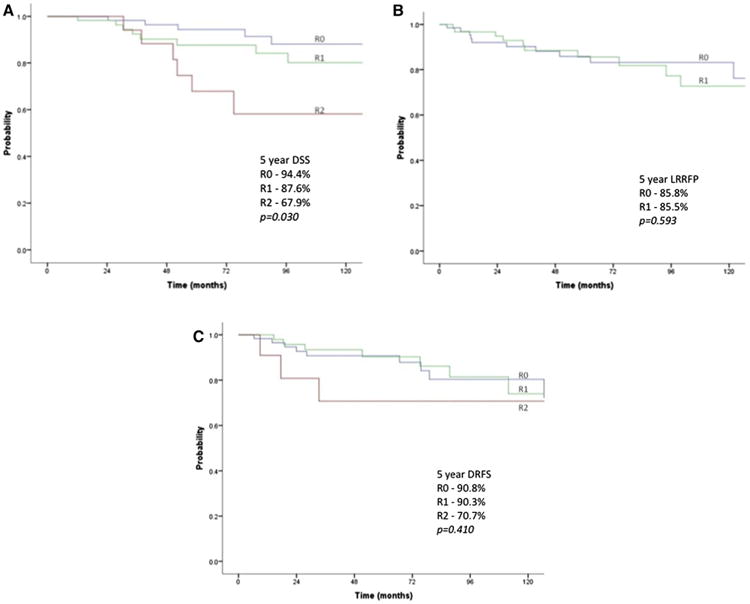
Survival and types of recurrence. (A) DSS by R stage. (B) LRRFP by R stage (excluding R2 patients. (C) DRFP by R stage (M0 patients).
Table II. Factors predictive of disease-specific survival by univariate analysis.
| Variable | n (%) | 5-y DSS | Univariate P value |
|---|---|---|---|
| Age | |||
| <45 | 45 (29%) | 100% | .001 |
| ≥45 | 108 (71%) | 83% | |
| Sex | |||
| Female | 89 (58%) | 90% | .625 |
| Male | 64 (42%) | 86% | |
| Operation | |||
| Less than total | 13 (8%) | 90% | .913 |
| Total thyroid | 140 (92%) | 88% | |
| R status | |||
| R0 | 70 (46%) | 94% | .030 |
| R1 | 63 (41%) | 88% | |
| R2 | 20 (13%) | 68% | |
| Neck dissection | |||
| None | 45 (29%) | 83% | .484 |
| Central | 27 (18%) | 90% | |
| Lateral | 81 (53%) | 89% | |
| pT size | |||
| ≤4 cm | 115 (74%) | 94% | .024 |
| >4 cm | 37 (25%) | 72% | |
| pN status | |||
| N0/NX | 31 (20%) | 75% | .887 |
| N1a | 43 (28%) | 98% | |
| N1b | 79 (52%) | 88% | |
| M status | |||
| M0 | 130 | 97% | <.001 |
| M1 | 23 | 47% | |
| RAI therapy | |||
| No | 17 (11%) | 80% | .613 |
| Yes | 136 (89%) | 89% | |
| EBRT | |||
| No | 135 (88%) | 87% | .852 |
| Yes | 18 (12%) | 94% | |
pN, Pathologic nodal; pT, pathologic tumor.
The 5-year, Kaplan-Meier (KM) plots of LRRFP for patients clear of all gross local disease (R0/R1) are shown in Fig 2, B; interestingly, the LRRFP of R0 and R1 patients are comparable at 85.8% and 85.5% (P = .593). Analysis of LRFFP excluding R2 patients (Table III) showed age greater than 45 years was a prognostic factor for locoregional recurrence on univariate analysis. While patients who received RT had no locoregional recurrences, this was not significant on univariate analysis. On multivariate analysis adjusting for age, tumor size, and administration of RT, age remained prognostic (HR 4.56, 95% confidence interval, 1.03–20.21; P = .046). Tumor size and RT were not prognostic of locoregional recurrence (P = .620, .125, respectively).
Table III. Factors predictive of locoregional recurrence by univariate analysis (R0 and R1 only, n = 133).
| Variable | N (%) | 5-y LRRFP | Univariate P value |
|---|---|---|---|
| Age | |||
| <45 | 38 | 94% | .019 |
| ≥45 | 95 | 82% | |
| Sex | |||
| Female | 77 | 86% | .590 |
| Male | 56 | 86% | |
| Operation | |||
| Less than total | 10 | 86% | .560 |
| Total thyroid | 123 | 86% | |
| R status | |||
| R0 | 70 | 86% | .593 |
| R1 | 63 | 86% | |
| Neck dissection | |||
| None | 44 | 87% | .530 |
| Central | 22 | 72% | |
| Lateral | 67 | 89% | |
| pT size | |||
| ≤4 cm | 101 | 85% | .984 |
| >4 cm | 32 | 87% | |
| pN status | |||
| N0/NX | 29 | 79% | .140 |
| N1a | 40 | 75% | |
| N1b | 64 | 94% | |
| M status | |||
| M0 | 118 | 86% | .713 |
| M1 | 15 | 81% | |
| RAI therapy | |||
| No | 14 | 76% | .689 |
| Yes | 119 | 87% | |
| EBRT | |||
| No | 119 | 84% | .227 |
| Yes | 14 | 100% | |
pN, Pathologic nodal; pT, pathologic tumor.
The KM plots for DRFP in patients with no distant metastases at presentation (M0) are shown in Fig 2, C; the 5-year DRFP of R0, R1, and R2 patients were 90.8%, 90.3%, and 70.7%, respectively (P = .410). Again, the DRFP of R0 and R1 patients appears comparable and in excess of 90%. Age greater than 45 years and tumor size greater than 4 cm were prognostic for poorer DRFP (Table IV) on univariate analysis.
Table IV. Factors predictive of distant recurrence by univariate analysis (M0 only, n = 130).
| Variable | N (%) | 5-y DRFP | Univariate P value |
|---|---|---|---|
| Age | |||
| ≥45 | 37 | 93% | .030 |
| >45 | 93 | 87% | |
| Sex | |||
| Female | 77 | 91% | .066 |
| Male | 53 | 86% | |
| Operation | |||
| Less than total | 11 | 100% | .363 |
| Total thyroid | 119 | 88% | |
| R status | |||
| R0 | 64 | 91% | .410 |
| R1 | 54 | 90% | |
| R2 | 12 | 71% | |
| Neck dissection | |||
| None | 39 | 100% | .167 |
| Central | 23 | 95% | |
| Lateral | 68 | 81% | |
| pT size | |||
| ≤4 cm | 104 | 93% | .013 |
| >4 cm | 25 | 74% | |
| pN status | |||
| N0/NX | 26 | 95% | .364 |
| N1a | 41 | 100% | |
| N1b | 63 | 79% | |
| RAI therapy | |||
| No | 16 | 91% | .699 |
| Yes | 114 | 89% | |
| EBRT | |||
| No | 113 | 89% | .791 |
| Yes | 17 | 88% | |
pN, Pathologic nodal; pT, pathologic tumor.
Discussion
Locally advanced (T4) DTC is rare. We report an incidence of locally advanced disease of 4.1% (153/3664) in our series that spans 25 years in a single tertiary-care referral institution. It is well recognized that gross ETE predicts worse survival.13,14,22 In contrast, in patients with T3 disease due to minimal extension to the overlying strap muscles, en bloc removal of strap muscles with the underlying thyroid gland achieves equivalent outcomes to patients with T1/T2 disease.23 While disease confined to the thyroid gland (T1-2) or minimal ETE in the sternothyroid muscle (T3) can be resected with minimal morbidity, resection of the vital structures surrounding the thyroid gland can result in a substantial impact on airway, voice, and swallowing. For this reason, multidisciplinary management teams must select the most appropriate treatment strategies to optimize both oncologic and functional outcome.
Shin et al24 proposed 5 stages of tracheal invasion: stage 0 when there is no ETE, stage I for extrathyroidal disease abutting the airway, stage II when invasion of the cartilage rings is present, stage III when disease invades the submucosa of the trachea, and stage IV when disease involves the mucosa of the trachea. Although some groups have suggested that aggressive operative resections, including full thickness resection of the airway, yield optimal results,11,16,18,19 others have demonstrated that in select patients with disease abutting the tracheal cartilage but without invasion of the submucosa, less aggressive tracheal resections can result in comparable oncologic outcomes while decreasing postoperative morbidity.10,12,14,15,25
Nishida et al15 advocated airway resection for stages II–IV while reserving shave excision for stage I only. McCaffery,13 however, recommended full thickness resection for stages III and IV and shave excision for stages I and II disease, reporting similar outcomes as long as all gross disease is resected. Despite these slight differences, all authors agree that achieving a complete resection with removal of all gross disease is crucial to optimize outcome in this group of patients.
Regardless of the approach, accurate, preoperative assessment of patients with suspected advanced DTC is crucial. Assessment includes identifying a history of changes in voice, compromise of the airway, dysphagia, or hemoptysis. The examination should include looking for a mass fixed to the airway, paralysis of the vocal cord, or intraluminal disease visible on flexible laryngoscopy. Preoperative investigation should include US routinely. However, US is not sensitive in assessing ETE, particularly involvement of structures posterior to the gland.
All patients with a worrisome clinical history or examination for ETE should undergo preoperative, contrast-enhanced CT of the neck. Although the use of iodine-based contrast media will affect the duration between an operation and the start of adjuvant RAI administration, accurate evaluation to facilitate a negative gross resection has far greater impact on oncologic and functional outcomes than a delay of a few weeks for the administration of RAI therapy. The use of intravenous contrast in preoperative CT is now recommended in the 2015 guidelines of the American Thyroid Association.26 After a CT scan that is suspicious for involvement of the airway, tracheoscopy under general anesthetic should be performed to allow direct visualization of the tracheal epithelium to complete the assessment. Similarly, esophagoscopy may be required to assess the extent of esophageal involvement, although intraluminal disease is extremely rare.
In the event that there is no evidence of disease penetration through the submucosa of the trachea (stages I–II), shave excision of the tracheal cartilage will provide adequate clearance while still preserving the tracheal integrity. A more extensive disease involving the submucosa or extending through the mucosa mandates full-thickness tracheal resection. A decision regarding the need for circumferential tracheal resection or laryngectomy has to be made on a case-by-case basis. Features that must be considered include patient-related factors, such as additional comorbidities and the degree of pulmonary reserve, and disease-related factors, including extent of circumferential involvement, specific sites of distant metastatic disease, and status of the RLNs.
The presence of distant metastatic disease is not an absolute contraindication for a resection to achieve local control for a symptom-free extension of life, because close to one half of these patients were still alive at 5 years in our study. In cases where resection may be considered palliative, particularly in the older age group, the treating clinician must not only consider the duration of survival but also the mode of death; mortality related to uncontrolled disease in the central compartment of the neck leading to hemorrhage and asphyxiation are especially traumatic for the patient, relatives, and clinicians.
Our study confirms the findings of Breaux et al16 that the RLN and trachea are the most commonly affected structures of upper aerodigestive tract, followed by esophagus and larynx. After investigation, about 90% of cases can be managed without entry into the airway, with only 4% of patients requiring partial and 7% circumferential resection of the trachea, larynx, and/or esophagus. The vast majority of patients (98%) in our series presented with papillary carcinoma, and in later years, tall cell variant of papillary carcinoma was noted in up to 38% of cases. This is a far greater rate than the reported general incidence of tall cell carcinoma within standard groups of DTC patients27 and is congruent with its recognized association with ETE and increasing age.28
In terms of factors prognostic of oncologic outcome in this group of patients, Rosa Pelizzo et al18 found that older age, more extensive disease, less than total thyroidectomy, and no neck dissection were all independently predictive of poor outcome. In patients with operatively resectable disease, our data confirm that older age, tumor size >4 cm, R2 resection, and the presence of distant metastases were predictive of worse DSS on univariate analysis. In addition, our study confirms that age greater than 45 years and tumor size greater than 4 cm were prognostic for poorer DRFP in M0 patients. For patients considered free of all gross local disease at time of resection (pathologically R0/R1), only age >45 years predicted poorer LRRFP.
Our analysis suggests that resection status was predictive of outcome. For patients with no gross residual disease in the thyroid bed, the 5-year LRRFP was similar for R0 and R1 patients (85.8% vs 85.5%). For these R0 and R1 patients, the 5-year DRFP was also similar at 90.8% and 90.3%. R2 patients, however, were much less likely to be free of distant disease, with 5-year DRFP of just 70.7%. Together, these findings emphasize that resection of all gross locoregional disease as judged by the surgeon is crucial to improve recurrence-free survival in this cohort of T4 patients.
When considering the 5-year DSS of patients based on resection status (R0/R1/R2), the DSS decreased incrementally across the 3 resection statuses (94.4%, 87.6%, and 67.9%; P = .030). It appears that while the presence of microscopic residual disease does not affect recurrence-free survival, it does have a detrimental impact on survival, associated with a significant difference in DSS between that of R0 and R2 status patients. The current data do not demonstrate a statistically significant difference in survival between R0 versus R1 (94.4% vs 87.6%, P = .222); however, with longer follow-up, the impact of R1 disease could be apparent. Alternatively, the lack of any observed difference could be because some of the R0 patients were really R1 but were not visualized on pathology. Together, these findings suggest that operative clearance of disease can mitigate effectively the risk of disease recurrence but only partly offset the aggressive disease biology and associated DSS of such patients.
Subsites of DTC that are locally invasive may represent dedifferentiated areas that were completely differentiated previously. These aggressive areas of disease are likely to be RAI refractory and more likely drivers of poor prognosis in such patients. Resection of grossly normal, contralateral thyroid tissue aids in administration of RAI; however, the efficacy of RAI in patients with locally advanced disease may be decreased, further emphasizing the importance of achieving R0/R1 margins and the rationale for adjuvant EBRT in some patients.
Due to the retrospective nature of this study, several limitations warrant discussion. First, this is an operative series of patients undergoing primary thyroidectomy for management of T4 DTC, and as such, only patients considered amenable to resection were included. Furthermore, patients were managed at a high-volume center with a dedicated, thyroid cancer, multidisciplinary team. Therefore, these results may not be generalizable to all patients with advanced disease.
Second, the patients in this series were managed over a long time period, from 1986 to 2010. Histologic subtype of the cohort was based on the original histopathology report. The histologic subtype of thyroid cancer may have been changed if re-reviewed; however, this was not possible for us due to the slides being unavailable from the early periods of the study. Furthermore, over this long time period, aspects of management of DTC have also evolved. In particular, the quality of imaging has improved, and more efficacious, targeted therapy using tyrosine kinase inhibitors are now available for management of progressive distant disease; these differences may lead to heterogeneity of the patients presented in this cohort.
Third, our results will be influenced by selection bias by treating physicians with regard to adjuvant therapies based on histopathologic findings. Although there were no significant differences between use of adjuvant RAI and EBRT therapies and resection status (R0/1/2; P ≥ .46 each), the decision to administer or withhold adjuvant RAI and EBRT in patients is prone to selection bias. Due to the rarity of locally advanced DTC at presentation, the limitations highlighted here are difficult to overcome, and future prospective studies are unlikely to be feasible.
Notwithstanding these limitations, our data demonstrate that patients with locally advanced DTC can be managed successfully with a 5-year DSS in excess of 90.0% for those patients considered free of gross disease after the initial resection (R0/R1, M0). Patients who have distant metastatic disease at presentation can expect significantly worse survival, although even among this group, 46% of patients were alive at 5 years. In these patients, achieving disease control in the central compartment is crucial to achieving a better quality of life.
In conclusion, attaining comprehensive operative clearance with the aim of achieving an R0/R1 resection is critical and associated with superior outcomes in T4 disease. Gross residual disease in the central compartment is associated with poorer outcomes and should be avoided. In patients in whom all gross disease has been removed but microscopic residual disease is evident due to positive operative margins, we recommend adjuvant therapy with RAI or EBRT to try to obtain equivalent outcomes to an R0 resection. Appropriate patient selection, thorough assessment of disease extent, fastidious resection of gross disease, and involvement of a multidisciplinary team are essential to ensure the best outcome in this complex patient group.
Acknowledgments
Supported in part through the NIH/NCI Cancer Center Support Grant P30 CA008748.
References
- 1.Hay ID, Hutchinson ME, Gonzalez-Losada T, McIver B, Reinalda ME, Grant CS, et al. Papillary thyroid microcarcinoma: A study of 900 cases observed in a 60-year period. Surgery. 2008;144:980–7. doi: 10.1016/j.surg.2008.08.035. discussion 987-8. [DOI] [PubMed] [Google Scholar]
- 2.Hassanain M, Wexler M. Conservative management of well-differentiated thyroid cancer. Can J Surg. 2010;53:109–18. [PMC free article] [PubMed] [Google Scholar]
- 3.Shah JP, Loree TR, Dharker D, Strong EW, Begg C, Vlamis V. Prognostic factors in differentiated carcinoma of the thyroid gland. Am J Surg. 1992;164:658–61. doi: 10.1016/s0002-9610(05)80729-9. [DOI] [PubMed] [Google Scholar]
- 4.Hundahl SA, Cady B, Cunningham MP, Mazzaferri E, McKee RF, Rosai J, et al. Initial results from a prospective cohort study of 5583 cases of thyroid carcinoma treated in the United States during 1996. U.S. and German Thyroid Cancer Study Group An American College of Surgeons Commission on Cancer Patient Care Evaluation study. Cancer. 2000;89:202–17. doi: 10.1002/1097-0142(20000701)89:1<202::aid-cncr27>3.0.co;2-a. [DOI] [PubMed] [Google Scholar]
- 5.Hughes DT, Haymart MR, Miller BS, Gauger PG, Doherty GM. The most commonly occurring papillary thyroid cancer in the United States is now a microcarcinoma in a patient older than 45 years. Thyroid. 2011;21:231–6. doi: 10.1089/thy.2010.0137. [DOI] [PubMed] [Google Scholar]
- 6.Enewold L, Zhu K, Ron E, Marrogi AJ, Stojadinovic A, Peoples GE, et al. Rising thyroid cancer incidence in the United States by demographic and tumor characteristics, 1980-2005. Cancer Epidemiol Biomarkers Prev. 2009;18:784–91. doi: 10.1158/1055-9965.EPI-08-0960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chen AY, Jemal A, Ward EM. Increasing incidence of differentiated thyroid cancer in the United States, 1988-2005. Cancer. 2009;115:3801–7. doi: 10.1002/cncr.24416. [DOI] [PubMed] [Google Scholar]
- 8.Morris LG, Myssiorek D. Improved detection does not fully explain the rising incidence of well-differentiated thyroid cancer: A population-based analysis. Am J Surg. 2010;200:454–61. doi: 10.1016/j.amjsurg.2009.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Davies L, Welch HG. Increasing incidence of thyroid cancer in the United States, 1973-2002. JAMA. 2006;295:2164–7. doi: 10.1001/jama.295.18.2164. [DOI] [PubMed] [Google Scholar]
- 10.McCaffrey JC. Evaluation and treatment of aerodigestive tract invasion by well-differentiated thyroid carcinoma. Cancer Control. 2000;7:246–52. doi: 10.1177/107327480000700304. [DOI] [PubMed] [Google Scholar]
- 11.Gaissert HA, Honings J, Grillo HC, Donahue DM, Wain JC, Wright CD, et al. Segmental laryngotracheal and tracheal resection for invasive thyroid carcinoma. Ann Thorac Surg. 2007;83:1952–9. doi: 10.1016/j.athoracsur.2007.01.056. [DOI] [PubMed] [Google Scholar]
- 12.Cody HS, 3rd, Shah JP. Locally invasive, well-differentiated thyroid cancer 22 years' experience at Memorial Sloan-Kettering Cancer Center. Am J Surg. 1981;142:480–3. doi: 10.1016/0002-9610(81)90379-2. [DOI] [PubMed] [Google Scholar]
- 13.McCaffrey JC. Aerodigestive tract invasion by well-differentiated thyroid carcinoma: Diagnosis, management, prognosis, and biology. Laryngoscope. 2006;116:1–11. doi: 10.1097/01.MLG.0000200428.26975.86. [DOI] [PubMed] [Google Scholar]
- 14.Wada N, Nakayama H, Masudo Y, Suganuma N, Rio Y. Clinical outcome of different modes of resection in papillary thyroid carcinomas with laryngotracheal invasion. Langenbecks Arch Surg. 2006;391:545–9. doi: 10.1007/s00423-006-0106-9. [DOI] [PubMed] [Google Scholar]
- 15.Nishida T, Nakao K, Hamaji M. Differentiated thyroid carcinoma with airway invasion: Indication for tracheal resection based on the extent of cancer invasion. J Thorac Cardiovasc Surg. 1997;114:84–92. doi: 10.1016/S0022-5223(97)70120-X. [DOI] [PubMed] [Google Scholar]
- 16.Breaux GP, Jr, Guillamondegui OM. Treatment of locally invasive carcinoma of the thyroid: How radical? Am J Surg. 1980;140:514–7. doi: 10.1016/0002-9610(80)90202-0. [DOI] [PubMed] [Google Scholar]
- 17.Nishida T, Nakao K, Hamaji M, Kamiike W, Kurozumi K, Matsuda H. Preservation of recurrent laryngeal nerve invaded by differentiated thyroid cancer. Ann Surg. 1997;226:85–91. doi: 10.1097/00000658-199707000-00012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rosa Pelizzo M, Toniato A, Boschin IM, Piotto A, Bernante P, Pagetta C, et al. Locally advanced differentiated thyroid carcinoma: A 35-year mono-institutional experience in 280 patients. Nucl Med Commun. 2005;26:965–8. doi: 10.1097/01.mnm.0000184936.75628.77. [DOI] [PubMed] [Google Scholar]
- 19.Friedman M, Danielzadeh JA, Caldarelli DD. Treatment of patients with carcinoma of the thyroid invading the airway. Arch Otolaryngol Head Neck Surg. 1994;120:1377–81. doi: 10.1001/archotol.1994.01880360071013. [DOI] [PubMed] [Google Scholar]
- 20.Grillo HC, Suen HC, Mathisen DJ, Wain JC. Resectional management of thyroid carcinoma invading the airway. Ann Thorac Surg. 1992;54:3–9. doi: 10.1016/0003-4975(92)91131-r. discussion 9-10. [DOI] [PubMed] [Google Scholar]
- 21.Sobin L, Gospodarowicz M, Wittekind C, editors. International Union Against Cancer (UICC) TNM Classification of Malignant Tumors. 7th. Hoboken NJ: Wiley-Blackwell; 2009. [Google Scholar]
- 22.Machens A, Hinze R, Lautenschläger C, Thomusch O, Dralle H. Thyroid carcinoma invading the cervicovisceral axis: Routes of invasion and clinical implications. Surgery. 2001;129:23–8. doi: 10.1067/msy.2001.108699. [DOI] [PubMed] [Google Scholar]
- 23.Nixon IJ, Ganly I, Patel S, Palmer FL, Whitcher MM, Tuttle RM, et al. The impact of microscopic extrathyroid extension on outcome in patients with clinical T1 and T2 well-differentiated thyroid cancer. Surgery. 2011;150:1242–9. doi: 10.1016/j.surg.2011.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Shin DH, Mark EJ, Suen HC, Grillo HC. Pathologic staging of papillary carcinoma of the thyroid with airway invasion based on the anatomic manner of extension to the trachea: A clinicopathologic study based on 22 patients who underwent thyroidectomy and airway resection. Hum Pathol. 1993;24:866–70. doi: 10.1016/0046-8177(93)90136-5. [DOI] [PubMed] [Google Scholar]
- 25.Segal K, Shpitzer T, Hazan A, Bachar G, Marshak G, Popovtzer A. Invasive well-differentiated thyroid carcinoma: Effect of treatment modalities on outcome. Otolaryngol Head Neck Surg. 2006;134:819–22. doi: 10.1016/j.otohns.2005.11.040. [DOI] [PubMed] [Google Scholar]
- 26.Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, et al. 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid. 2016;26:1–133. doi: 10.1089/thy.2015.0020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Terry JH, St John SA, Karkowski FJ, Suarez JR, Yassa NH, Platica CD, et al. Tall cell papillary thyroid cancer: Incidence and prognosis. Am J Surg. 1994;168:459–61. doi: 10.1016/s0002-9610(05)80099-6. [DOI] [PubMed] [Google Scholar]
- 28.Morris LG, Shaha AR, Tuttle RM, Sikora AG, Ganly I. Tall-cell variant of papillary thyroid carcinoma: A matched-pair analysis of survival. Thyroid. 2010;20:153–8. doi: 10.1089/thy.2009.0352. [DOI] [PMC free article] [PubMed] [Google Scholar]


