Significance
Urinary tract infections (UTIs) are primarily caused by uropathogenic Escherichia coli (UPEC), and 1 in 40 women experience chronic UTIs during their lifetime. The antibiotic courses required to treat infections promote antibiotic resistance, and current vaccine options offer limited protection. We have pioneered a strategy using small iron-chelating compounds called siderophores as vaccine antigens. These siderophores are not produced by commensal bacteria and are required for UTI. The siderophore vaccines reported here are easy to formulate and reduce bacterial burdens in a murine model of UTI. This report highlights the untapped resource of bacteria-specific small molecules as potential vaccine antigens and provides a proof of principle for incorporating these compounds into multicomponent vaccines for the prevention of bacterial infections.
Keywords: vaccine conjugate, urinary tract infection, siderophore, UPEC, iron acquisition
Abstract
Uropathogenic Escherichia coli (UPEC) is the primary cause of uncomplicated urinary tract infections (UTIs). Whereas most infections are isolated cases, 1 in 40 women experience recurrent UTIs. The rise in antibiotic resistance has complicated the management of chronic UTIs and necessitates new preventative strategies. Currently, no UTI vaccines are approved for use in the United States, and the development of a highly effective vaccine remains elusive. Here, we have pursued a strategy for eliciting protective immunity by vaccinating with small molecules required for pathogenesis, rather than proteins or peptides. Small iron-chelating molecules called siderophores were selected as antigens to vaccinate against UTI for this vaccine strategy. These pathogen-associated stealth siderophores evade host immune defenses and enhance bacterial virulence. Previous animal studies revealed that vaccination with siderophore receptor proteins protects against UTI. The poor solubility of these integral outer-membrane proteins in aqueous solutions limits their practical utility. Because their cognate siderophores are water soluble, we hypothesized that these bacterial-derived small molecules are prime vaccine candidates. To test this hypothesis, we immunized mice with siderophores conjugated to an immunogenic carrier protein. The siderophore–protein conjugates elicited an adaptive immune response that targeted bacterial stealth siderophores and protected against UTI. Our study has identified additional antigens suitable for a multicomponent UTI vaccine and highlights the potential use of bacterial-derived small molecules as antigens in vaccine therapies.
Both the physical and financial burdens of urinary tract infections (UTIs) are staggering. Half of all women experience a symptomatic UTI in their lifetime (1). And of those women, almost half suffer a reoccurrence within the next year (1). In the United States, where the annual societal cost of UTI is likely underestimated at $3.5 billion (2), 4 million women have UTIs continuously (3). Uropathogenic Escherichia coli (UPEC) is a subclass of extraintestinal pathogenic E. coli (ExPEC) and is the etiological agent for 80% of all uncomplicated UTIs (1). In 2006, there were 11 million physician visits, over 1.7 million emergency room visits, and 479,000 hospitalizations of both men and women in the United States for UTI (2, 4). Altogether, these estimates place UTIs first among kidney and urologic diseases in terms of total cost.
UTIs occur when bacteria, most commonly UPEC (5), contaminate the periurethral area and traverse the urethra to colonize the bladder and its underlying epithelium, causing cystitis (6, 7). If left untreated, UPEC may ascend the ureters and establish a secondary infection in the kidney parenchyma, causing pyelonephritis. At this juncture, UPEC can elicit serious complications, including renal scarring, septicemia, and death.
UTIs are routinely treated with antibiotic therapy, including trimethroprim–sulfamethoxazole (TMP–SMX) and ciprofloxacin. Women experiencing at least two UTIs per year are frequently given antibiotics prophylactically (8). Not surprisingly, the rates of resistance to these antibiotics in UPEC strains have steadily risen over the past few decades. In the United States, Canada, and elsewhere, ∼10–25% of uncomplicated UTI isolates are resistant to TMP–SMX (9–11). This trend is forcing physicians to reach for more expensive and sometimes less effective drugs to treat UTIs (10, 12, 13). Even more troubling is the rise in multidrug resistance among UPEC strains, as a recent international study found that over 10% of E. coli cystitis isolates are resistant to at least three different classes of antimicrobial agents (14). These trends challenge the prescription choices of physicians to address shifting microbial susceptibilities (15).
To compound the danger of antibiotic resistance, there are no currently licensed vaccines in the United States to combat recurrent UTIs in women. In Europe, two vaccines against UTIs called SolcoUrovac and Uro-Vaxom are licensed for use in women with recurrent UTIs (16). SolcoUrovac is a vaginal suppository containing 10 heat-killed UPEC strains that provides relatively poor protection in the absence of frequent administration (17). Uro-Vaxom is an oral capsule containing a lyophilized mixture of membrane proteins from 18 UPEC isolates that is expected to be taken daily. Although this vaccine offers protection against UPEC, its success is limited due to toxicity and poor adherence to the daily regimen (18). Due to these drawbacks, no vaccines are licensed for use in the United States (19). Given the paucity of effective vaccines, the increasing rate of UPEC antibiotic resistance, the decline in novel antibiotic scaffolds, and the need to reduce healthcare expenditures, new therapeutic strategies to manage UTIs must be explored.
Previous work using unbiased genomic and proteomic screens identified bacterial targets that are expressed in vivo by UPEC during UTIs in women, reside on the surface of the bacterium, are immunogenic, and carry out a critical function for survival of E. coli in the host (20). Six bacterial iron acquisition system proteins met all criteria. These findings are supported by a rich history of genomic, transcriptomic, and proteomic studies that have also identified iron acquisition systems as prime anti-UTI targets (21–27).
Iron is an essential cofactor in many biological processes, including DNA synthesis, electron transfer, and central metabolism (28). Iron acquisition is generally required for bacterial growth during infection (28, 29). One facet of innate immunity, coined “nutritional immunity,” restricts bacterial infections by limiting access to critical metal cofactors (28, 30). The mammalian host limits intracellular and freely circulating iron by sequestering iron in proteins such as lactoferrin, transferrin, ferritin, and hemoglobin (31). Notably, the primary site of UPEC infection, the bladder, has lower iron levels than serum (32). Thus, it is not surprising that over 14 gene clusters implicated in iron acquisition have been identified as important virulence factors in UPEC strains (33–37); these gene clusters encode up to four siderophore biosynthesis and uptake systems as well as receptors for the acquisition of heme, ferric citrate, and ferrous iron. Of the many classes of siderophores, UPEC strains typically encode at least three of the following siderophores: yersiniabactin (Ybt), aerobactin (Aer), enterobactin (Ent), and the glucosylated Ent, salmochelin (Glc-Ent) (38, 39).
Bacterial iron acquisition is a natural target of the host immune system. For example, serum albumin and lipocalin-2 bind and inactivate Ent (40, 41). To evade host immunity, pathogenic E. coli strains typically encode a combination of Ybt, Aer, and Glc-Ent stealth siderophores, which are not recognized by host defenses (39). By evading host defenses to secure nutrient iron, Ybt, Aer, and Glc-Ent serve as urovirulence factors (33, 36). Notably, Ybt and Aer are more prevalent among pathogenic E. coli strains than commensal isolates (39). Moreover, previous studies that systematically assessed the use of surface-exposed iron receptors as potential vaccine antigens found that two of the stealth siderophore receptors, those that recognize Ybt and Aer, protect against UTI (20, 42, 43). The hydrophobic nature of these outer-membrane receptors, however, makes these antigens insoluble in water, complicating the purification and formulation of the vaccines. Here, we examine the potential use of the small-molecule siderophores Ybt and Aer as protective vaccine antigens that could bolster the efficacy of other immunoprotective strategies.
Results
By virtue of the confirmed importance of Ybt and Aer in uropathogenesis (33–37), the proven efficacy of their receptors in experimental vaccines (20, 42), their increased prevalence among pathogenic E. coli (39), and amenable biochemical features, we hypothesized that Ybt and Aer could represent valid vaccine candidates. Because the siderophores are small (<564 Da) and unlikely to be immunogenic, Ybt and Aer were conjugated to cationized BSA (cBSA), an immunogenic carrier protein that has aminoethyl-capped carboxylic acids (44–46). The positive surface charge of cBSA increases vaccine binding to immune cells, and the aminoethyl modifications improve the coupling reaction by both eliminating carboxylic acids and providing additional primary amines on the carrier protein (44–46). Both Ybt and Aer have carboxylic acid moieties; thus, standard amide coupling conditions were used to prepare the cBSA–siderophore conjugates using 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC) as the coupling reagent (SI Appendix, Fig. S1 A and B). cBSA incubated with EDC in the absence of siderophore was used as a negative control.
Liquid chromatography and tandem mass spectrometry on trypsin-digested cBSA–siderophore conjugates confirmed linkage of the siderophores to cBSA. For the cBSA–Aer conjugate, peptide fragment ions supporting the linkage of Aer to Lys266, aminoethyl-Asp474, and aminoethyl-Glu267 were detected (SI Appendix, Fig. S2 A–C and Tables S1–S3). For cBSA–Ybt, peptide ions supporting the linkage of Ybt to aminoethyl-Asp272 and Lys437 were detected (SI Appendix, Fig. S2D and Tables S4 and S5). Altogether, these data confirm at least two conjugation sites on cBSA for each siderophore.
To assess the efficacy of vaccination with the stealth siderophore conjugates, mice were immunized intranasally with 10 μg of vaccine conjugate prepared in 20 μL of PBS (SI Appendix, Fig. S1C). Three vaccine groups were examined along with a cBSA control, including cBSA–Ybt, cBSA–Aer, or a 1:1 mixture of cBSA–Ybt:cBSA–Aer, which was composed of 5 μg of each conjugate. Previous reports have shown that intranasal vaccination provides the most consistent protection in murine UTI vaccine studies (20, 42, 47). Therefore, mice were boosted intranasally with 20 μL of PBS containing 2.5 μg of vaccine conjugate 7 and 14 d postimmunization. UPEC strain HM69, a strain recently isolated from a patient with uncomplicated cystitis, was selected for challenge because it encodes Ent, Ybt, and Aer (38). On day 21 postimmunization, mice were transurethrally inoculated with 108 colony-forming units (cfus) of HM69, and after 48 h, the bacterial burdens in the urinary tract were quantified.
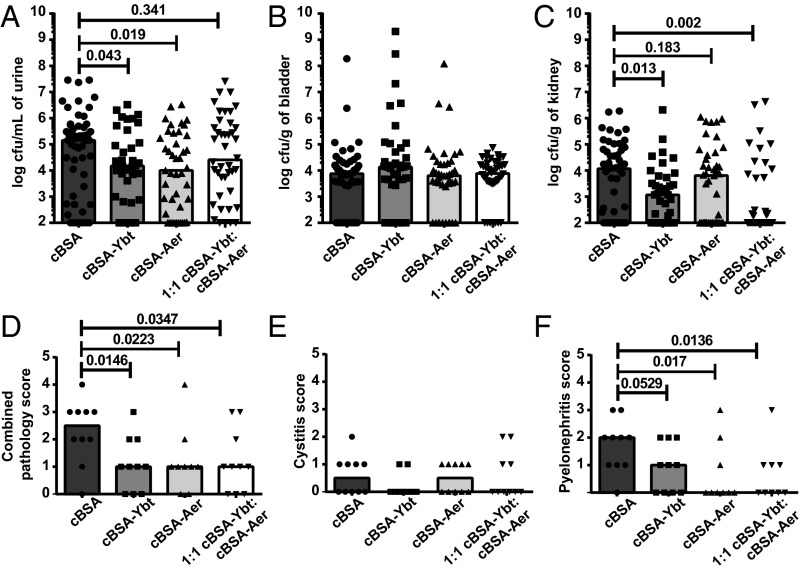
Vaccination with cBSA–Ybt reduced bacterial burden by 12-fold in the urine (P = 0.04) and 10-fold in the kidneys (P = 0.01), whereas cBSA–Aer reduced bacterial burden by 19-fold (P = 0.02) in the urine (Fig. 1 A–C). Coimmunization with 1:1 cBSA–Ybt:Aer also decreased bacterial burdens in the urine by 14-fold (P = 0.3) and, most dramatically, reduced bacterial burden in the kidneys by 126-fold (P = 0.002) (Fig. 1 A–C). Altogether these data demonstrate that the siderophore–protein conjugates significantly reduce the bacterial burden in experimental UTI, particularly dissemination to the kidneys. At the time of sacrifice, a subset of the kidneys and bladders from infected mice were fixed in neutral buffered formalin, embedded in paraffin, sectioned, and stained with hematoxylin and eosin (H&E). Five-micrometer histologic sections were randomized and blindly scored for neutrophilic inflammation by a board-certified veterinary pathologist. Scores were semiquantitative and ranged from 0 (no inflammation) to 3 (severe inflammation) (SI Appendix, Fig. S3). Vaccination with cBSA–Ybt, cBSA–Aer, and 1:1 cBSA–Ybt:cBSA–Aer significantly reduced pyelonephritis and overall inflammation scores (Fig. 1 D–F).
Fig. 1.
Vaccination with siderophores reduces bacterial burdens and inflammation in the urine and kidneys during UTI. CBA/J mice were vaccinated with cBSA, cBSA–Ybt, cBSA–Aer, or 1:1 cBSA–Ybt:cBSA–Aer. At 21 d after the first immunization, mice were transurethrally inoculated with 108 cfus of wild-type HM69. After 48 h, cfus in the (A) urine, (B) bladder, and (C) kidneys were enumerated by serial dilution and plating on LB agar. Limit of detection was 102 cfu/mL of urine or cfu/g of organs. Bladders and kidneys were fixed, embedded in paraffin, sectioned, and H&E stained. Tissue sections were randomized and blindly scored for inflammation in the (E) bladders and (F) kidneys. The pathology scores in E and F were combined to quantify overall inflammation in D. Significance was calculated using a one-tailed Mann–Whitney test. In all panels, bars represent the median; in A–C, n ≥ 38; D–F, n ≥ 8.
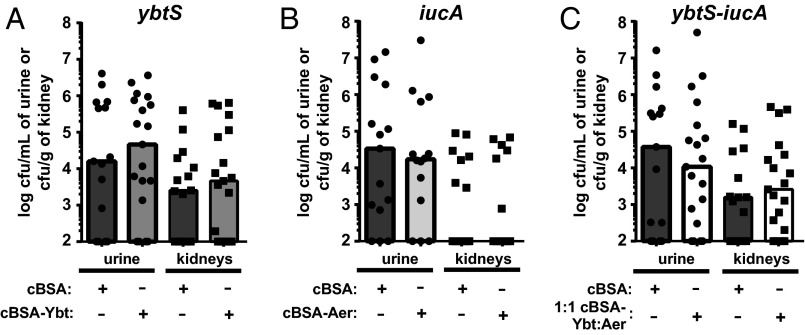
To ascertain whether the vaccine specifically targets Ybt and Aer, three isogenic mutants were generated; E. coli HM69 ybtS::cam (ybtS) and iucA::kan (iucA) have the Ybt and Aer biosynthetic machinery disrupted, respectively. In addition, both biosynthetic operons were disrupted in a ybtS–iucA double mutant. We hypothesized that if the vaccines specifically target Ybt or Aer during infection, then infection with each siderophore biosynthesis mutant would result in loss of protection by the corresponding vaccine. Mice were vaccinated with either cBSA or cBSA–Ybt (SI Appendix, Fig. S1C) and then transurethrally challenged with ybtS. After 48 h, there were no significant differences in the bacterial burdens of ybtS between cBSA- and cBSA–Ybt-vaccinated mice (Fig. 2A and SI Appendix, Fig. S4A). Similar experiments were conducted by vaccinating mice with cBSA–Aer and then challenging with iucA, as well as by covaccinating with 1:1 cBSA–Ybt:cBSA–Aer and then challenging with ybtS–iucA (Fig. 2 B and C and SI Appendix, Fig. S4 B and C). In all instances, no significant differences were identified between the vaccinated groups and the cBSA control mice (Fig. 2 and SI Appendix, Fig. S4), indicating that the vaccines elicit an immune response specifically targeting Ybt and Aer during UTI.
Fig. 2.
Protection provided by siderophore vaccines requires the biosynthesis of bacterial stealth siderophores. CBA/J mice were vaccinated according to the protocol but transurethrally inoculated with the corresponding siderophore biosynthesis mutant. (A) cBSA–Ybt-vaccinated mice were inoculated with HM69 ybtS, (B) cBSA–Aer-vaccinated mice were inoculated with HM69 iucA, and (C) 1:1 cBSA–Ybt:cBSA–Aer-vaccinated mice were infected with HM69 ybtS–iucA. For each experiment, cBSA was used as the negative control (dark gray bars). In all instances, mice were transurethrally inoculated with 108 cfus of the indicated strain. After 48 h, cfus in the urine and kidneys were enumerated by serial dilution and plating on LB agar. Statistical analyses using a one-tailed Mann–Whitney test identified no significant differences between negative control and siderophore-vaccinated groups (0.2336 < P < 0.4761). Limit of detection was 102 cfu/mL of urine or cfu/g of kidneys; bars represent the median; and n ≥ 14.
The unexpected differences in kidney colonization for iucA and ybtS–iucA mutants (Fig. 2 B and C) compared with the cBSA–Aer- and 1:1 cBSA–Ybt:cBSA–Aer-vaccinated and infected with WT HM69 (Fig. 1C) could be due to disparities between the effects of genetically disrupting intracellular siderophore biosynthesis and immunologically targeting extracellular siderophore activity. In the case of iucA (Fig. 2B) and cBSA–Aer (Fig. 1C), it is possible that the immune response does not inactivate Aer-mediated iron acquisition as well as genetically disrupting Aer biosynthesis. Whereas in the case of ybtS–iucA (Fig. 2C) and 1:1 cBSA–Ybt:cBSA–Aer (Fig. 1C), ybtS–iucA may colonize the kidneys better than WT in 1:1 cBSA–Ybt:cBSA–Aer-vaccinated mice because of increased virulence or Ent production in ybtS–iucA. Studies have shown that disrupting siderophore biosynthesis impacts central metabolism, which could affect the elaboration of other virulence factors and ultimately pathogenesis (48). Alternatively, elevated Ent levels have been detected in bacterial strains with mutations in the Aer and Ybt pathways, which could improve pathogenesis (49, 50). Elevated Ent levels in ybtS–iucA may also explain the surprising increase in kidney colonization of cBSA-vaccinated mice infected with ybtS–iucA compared with iucA (Fig. 2 B and C), as the iucA and ybtS–iucA mutants have been rigorously confirmed to be genetically correct and found to grow similarly in vitro (SI Appendix, Fig. S5). Despite these confounding factors presented by the use of isogenic siderophore biosynthesis mutants, the data presented in Fig. 2 reveal that the bacteria must encode the targeted siderophore to be susceptible to the corresponding cBSA–siderophore vaccine. This emphasizes that the protection elicited by vaccination with Ybt and Aer is target-specific, suggestive of an adaptive immune response.
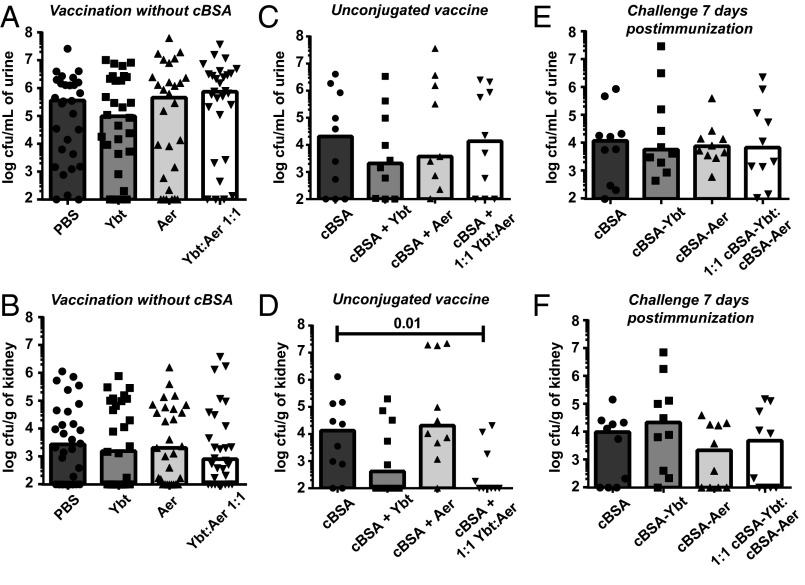
Because the molecular masses of Ybt and Aer are relatively small (481 Da and 564 Da, respectively), we hypothesized that Ybt and Aer are unable to stimulate an immune response alone and are acting as haptens. To test the immunogenicity of Ybt and Aer, mice were vaccinated with Ybt, Aer, or a 1:1 mixture of Ybt:Aer. Based on the theoretical maximum amount of siderophores administered in the cBSA–conjugate vaccine, mice were immunized with 0.75 μg of siderophore and boosted with 0.19 μg of siderophore on days 7 and 14 postimmunization (SI Appendix, Fig. S1C). On day 21 postimmunization, mice were challenged with 108 cfu of HM69 and bacterial burdens were quantified 48 h later. Without a carrier protein, immunization with Ybt and Aer no longer protected against UTI (Fig. 3 A and B and SI Appendix, Fig. S6A), revealing that Ybt and Aer require cBSA to elicit a protective immune response.
Fig. 3.
Protection requires repeated exposure to siderophore and an immunogen. The requirement of the cBSA carrier protein was examined by intranasally administering the unconjugated apo-siderophores (A and B) in PBS and (C and D) with cBSA. (E and F) To determine if the vaccine requires time adequate for generating an adaptive immune response, mice were vaccinated with 10 μg of cBSA, cBSA–Ybt, cBSA–Aer, or 1:1 cBSA–Ybt:cBSA–Aer and transurethrally inoculated 1 wk after immunization. No booster doses were administered. In all experiments, mice were transurethrally inoculated with wild-type HM69. After 48 h, cfus in the urine (A, C, and E) and kidneys (B, D, and F) were enumerated by serial dilution and plating on LB agar. Statistical analyses were calculated using a one-tailed Mann–Whitney test. The range of unlabeled P values is 0.0627–0.4933. Limit of detection was 102 cfu/mL of urine or cfu/g of kidneys; bars represent the median; and n ≥ 10.
To fully address whether the siderophores are acting as haptens, we next vaccinated the mice with cBSA unconjugated to Ybt, Aer, or a 1:1 mixture of Ybt:Aer. We hypothesized that if the siderophores are acting as haptens, then protection will be lost when the mice are vaccinated with cBSA and unconjugated siderophore. The mice were immunized with 0.75 μg of siderophore plus 10 μg of cBSA on day 0 and boosted with 0.19 μg of siderophore plus 2.5 μg of cBSA on days 7 and 14 postimmunization (SI Appendix, Fig. S1C). On day 21 postimmunization, mice were transurethrally inoculated with 108 cfu of HM69, and after 48 h, the bacterial burdens in the urinary tract were quantified. In a small pilot experiment, bacterial burdens were diminished in the urine and kidneys (Fig. 3 C and D and SI Appendix, Fig. S6B) following the same trend as the conjugated vaccine (Fig. 1), indicating that the siderophores are not acting as haptens and do not require conjugation to cBSA to protect against pyelonephritis.
Because Ybt and Aer require cBSA as an immunogen to elicit a protective immune response and bacterial production of the targeted siderophore, we hypothesized that protection is mediated by the adaptive immune system. Because the highly cationic surface charge of cBSA interferes with analysis by ELISA (51), we examined this notion using our murine model of ascending UTI. Mice were immunized once with 10 μg of cBSA, cBSA–Ybt, cBSA–Aer, or a 1:1 mixture of cBSA–Ybt:cBSA–Aer and then challenged transurethrally with E. coli HM69 7 d postimmunization. Challenging shortly after immunization precludes the development of an effective adaptive immune response (52). After 48 h, the bacterial burdens in the urinary tract were enumerated, and as predicted, no protection was observed (Fig. 3 E and F and SI Appendix, Fig. S6C), suggesting that protection is mediated by an adaptive immune response.
Discussion
These findings provide a significant conceptual advance in which bacterial-derived small molecules have been successfully formulated in a vaccine regimen that reduces both inflammation and the bacterial burden in the kidneys and urine in a murine model of UTI (Fig. 1). To protect against pyelonephritis, the bacteria used for challenge must encode the targeted stealth siderophores (Fig. 2). The siderophores do not need to be conjugated to the immunogen to elicit protection, indicating that despite their small molecular weights (481 Da and 564 Da), Ybt and Aer are large enough to stimulate a protective immune response (Fig. 3 A–D). In addition, successful vaccination with siderophores requires repeated coadministration with the immunogen cBSA over the course of 3 wk (Fig. 3 E and F). Altogether, these data suggest that the cBSA–siderophore conjugates elicit an adaptive immune response that specifically targets pathogens encoding stealth siderophores. We propose that Aer and Ybt are suitable for inclusion in multicomponent vaccines due to their amenable biochemical features, specificity for pathogenic bacteria compared with commensal organisms, and synergistic protection in the kidneys.
We have been unable to confirm, however, that the protective immune response is antibody-mediated, as we do not detect siderophore-specific antibodies by direct or sandwich ELISAs or ELISpot using the cBSA-conjugated siderophores as capture antigens. Because cBSA interferes with ELISAs (51) and the siderophores are not suited for adsorption to ELISA plates, we have also probed for anti-siderophore antibodies by direct and sandwich ELISAs with Ybt and Aer conjugated to cholera toxin or BSA; neither revealed siderophore-specific antibodies. It is possible that these carrier proteins are poorly haptenized or antisiderophore antibodies do not recognize the siderophores in the context of these carrier proteins. Despite our best efforts, we have been unable to identify siderophore-specific antibodies, however the data presented here suggest that the vaccine is specific (Fig. 2) and acts in a time frame consistent with adaptive immunity (Fig. 3 E and F). It is possible that siderophore-specific antibodies are either absent or present and effective but below the limit of detection. Nonetheless, the utility of small molecules formulated for use as vaccine antigens is intriguing.
Previous publications describing the use of siderophores in vaccines against human pathogens primarily focus on eliciting an antibody response and either did not observe protection against infection or did not use an animal model to evaluate the effectiveness of the vaccine. The first report describing the development of monoclonal antibodies (MAbs) against a siderophore targeted pseudobactin, the siderophore of the plant pathogen Pseudomonas putida. These MAbs were primarily used for studying iron acquisition in the plant pathogen and were not developed for therapeutic applications (53). Shortly thereafter, another group elicited mouse MAbs against ferric Aer (FeAer) and demonstrated that these antibodies restricted E. coli growth in calf serum (54). These FeAer-specific MAbs were useful for detecting Aer-producing Enterobacteriaceae but did not provide protection in three tested models of infection (55, 56). A lethal i.p. infection model with E. coli and Klebsiella pneumoniae was used to assess the protection provided upon either passive or active immunization of Swiss mice (55). In both cases, vaccination did not significantly improve the survival of the mice despite the confirmed presence of anti-FeAer antibodies. The same vaccine preparation was also used to assess protection in a passive transovarian immunization chick model challenged with E. coli, and despite the presence of Aer-specific antibodies, the chicks succumbed to infection similar to the unvaccinated controls (55). The last published study regarding siderophore vaccine preparation successfully elicited vibriobactin-specific antibodies in mice, but the protective effects of these antibodies were never reported (57). Building upon these studies, we have shown that vaccination with siderophores is protective in a mouse model of ascending UTI. The success of the vaccine strategy described here compared with previous publications using siderophore vaccines may be due to the intranasal immunization route and an animal model that better recapitulates natural infection.
It has been documented that vaccination with outer-membrane proteins up-regulated during iron limitation protects against E. coli infections (43). Specifically, our laboratory investigated the immunoprotective properties of the outer-membrane protein receptors for Ybt and Aer: namely, FyuA and IutA, respectively (20, 42). Immunization with FyuA resulted in a 1.5 log decrease in the bacterial burden in the kidneys (urine counts were not reported), and immunization with IutA reduced bacterial burden in the urine, bladders, and kidneys by 1.3, 1.0, and 0.9 logs, respectively. Studies reported here recapitulate these findings with the exception of the protection observed in the bladder with the IutA vaccine. In our extensive experience with developing UTI vaccines, we have found that protection in the bladder is more difficult to achieve in our murine model than protection in the kidneys, likely due to low levels of antigen-specific antibody detected in the bladder (20, 42). This may be due to technical issues implicit in a murine model of UTI or the lower abundance of antibodies in the urine compared with the sera (20, 58–60). Alternatively, the lack of protection in the bladder in response to targeting Aer-mediated iron acquisition could be due to the different immune response elicited by vaccination with the low-molecular mass siderophores compared with the proteinaceous receptors.
Building upon these prior studies, we now show that vaccination with siderophores protects against pyelonephritis in a mouse model of ascending UTI and circumvents the biochemical challenges associated with using outer-membrane proteins as antigens (Fig. 1). Moreover, Aer and Ybt may be suitable for a multicomponent vaccine due to their amenable biochemical features, specificity for pathogenic bacteria over commensal organisms, and synergistic protection in the kidneys. The practical utility of cBSA as an immunogen in human vaccines remains to be evaluated in future studies. Combining Aer and Ybt with other protective antigens such as adhesins, toxins, and other iron receptors may prove to be a successful vaccination strategy (19). Adding Aer and Ybt to such multicomponent vaccine preparations should prove simple due to their amenable biochemical properties and their ability to elicit protection without being conjugated to a carrier protein (Fig. 3D). Notably, highly evolved iron acquisition strategies, including Aer and Ybt, are critical virulence factors for ExPEC and other pathogenic species within the Enterobacteriaceae (61, 62). Therefore, by defining the protective efficacy of two stealth siderophores prevalent among pathogenic E. coli (39), we have determined that Ybt and Aer are valuable antigens that have not heretofore been included in any UTI vaccines and, more generally, exemplify the untapped resource of bacteria-specific small molecules as potential vaccine antigens.
Online Methods
Descriptions of the bacterial strains, culture methods, and recombineering; cBSA–siderophore conjugation; tandem mass spectrometry; and histologic scoring are in SI Appendix, SI Materials and Methods.
Vaccination Procedures.
All mouse studies were conducted in accordance with protocol 00005052 approved by the Institutional Animal Care and Use Committee at the University of Michigan. This protocol is in compliance with the National Institutes of Health guidelines for the humane use and care of laboratory animals.
Vaccination was performed as previously described (SI Appendix, Fig. S1C) (20). All vaccine doses were prepared in advance from 2 mg/mL stocks. Aliquots were flash-frozen and stored at –80 °C until the time of vaccination. Briefly, 6–7-wk-old CBA/J mice were intranasally immunized with 20 μL of vaccine conjugate (10 μL/nare). On day 0, the mice received 10 μg of the appropriate vaccine conjugate (0.5 mg/mL in PBS, pH 7.4). On days 7 and 14, mice were boosted with 2.5 μg of vaccine conjugate (0.125 mg/mL in PBS, pH 7.4). Mice were challenged, as described below, on day 21.
One exception to this protocol was for vaccinations that used unconjugated siderophores. When PBS pH 7.4 or unconjugated cBSA was used as controls, mice were immunized with 0.75 μg siderophore (37.5 µg/mL in PBS pH 7.4) and boosted with 0.19 μg of siderophore (9.4 µg/mL in PBS pH 7.4). The amounts of siderophores used were calculated based on the maximum amount of siderophore the mice could potentially receive from the cBSA–vaccine conjugates. A second exception to the above protocol was when mice were transurethrally challenged 1 wk after the initial immunization. No boosters were given to assess the requirement for adaptive responses to elicit protection.
Murine Model of Ascending UTI.
A previously described murine model of ascending UTI was used to assess vaccine efficacy (20, 58). Unless noted otherwise, female CBA/J mice were transurethrally challenged with 50 μL of 4 × 109 cfu/mL of E. coli HM69 resuspended in PBS, resulting in an inoculum of 108 cfu per mouse. The inoculum was delivered using a sterile 0.28-mm inner-diameter polyethylene catheter connected to an infusion pump (Harvard Apparatus) over a period of 20 s. Infection progressed for 48 h, after which bacterial burdens in urine as well as bladder and kidney homogenates were enumerated. Tissues were homogenized in PBS with a GLH homogenizer (Omni International) and plated on LB agar using an Autoplate 4000 spiral plater (Spiral Biotech). A QCount automated plate counter (Spiral Biotech) was used to enumerate colony counts on the plates.
Statistical Analysis.
Unpaired, nonparametric analysis was performed using the Mann–Whitney test calculated in GraphPad Prism 6.01 for Windows. Results were considered significant if the P value was less than or equal to 0.05.
Supplementary Material
Acknowledgments
The authors thank Matthew Schaller, Jackson Turner, Irina Grigorova, Ashootosh Tripathi, Marilia Cascalho, and members of the Michael Bachman and H.L.T.M. laboratories for helpful discussions and assistance with pilot experiments; faculty members of the University of Michigan Microbiology and Immunology Department for critical feedback on the manuscript; MS Bioworks LLC for experimental processing and data analysis of the liquid chromatography and tandem mass spectrometry (LC–MS/MS) experiments; the University of Michigan Proteomics and Peptide Synthesis Core for interpretation and discussion of the LC–MS/MS data; and David Friedman for providing the spectinomycin-resistant pKD46. This work was supported by Public Health Service Grants R01 AI116791 and R01 DK097362 from the National Institutes of Health (NIH). L.A.M. was supported by NIH Grant T32 AI007528 and is a Research Scholars Fellow for the American Urological Association administered by the Urology Care Foundation.
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1606324113/-/DCSupplemental.
References
- 1.Barber AE, Norton JP, Spivak AM, Mulvey MA. Urinary tract infections: Current and emerging management strategies. Clin Infect Dis. 2013;57(5):719–724. doi: 10.1093/cid/cit284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Litwin MS, Saigal CS. Introduction. In: Litwin MS, Saigal CS, editors. Urologic Diseases in America. US Department of Health and Human Services, Public Health Service, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases; Washington, DC: 2007. pp. 3–7. [Google Scholar]
- 3.Foxman B. The epidemiology of urinary tract infection. Nat Rev Urol. 2010;7(12):653–660. doi: 10.1038/nrurol.2010.190. [DOI] [PubMed] [Google Scholar]
- 4.DeFrances CJ, Lucas CA, Bule VC, Golosinskly A. National Health Statistics Reports. National Center for Health Statistics; Hyattsville, MD: 2008. 2006 National Hospital Discharge Survey. [PubMed] [Google Scholar]
- 5.Wiles TJ, Kulesus RR, Mulvey MA. Origins and virulence mechanisms of uropathogenic Escherichia coli. Exp Mol Pathol. 2008;85(1):11–19. doi: 10.1016/j.yexmp.2008.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Justice SS, et al. Differentiation and developmental pathways of uropathogenic Escherichia coli in urinary tract pathogenesis. Proc Natl Acad Sci USA. 2004;101(5):1333–1338. doi: 10.1073/pnas.0308125100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rosen DA, Hooton TM, Stamm WE, Humphrey PA, Hultgren SJ. Detection of intracellular bacterial communities in human urinary tract infection. PLoS Med. 2007;4(12):e329. doi: 10.1371/journal.pmed.0040329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Beerepoot MA, et al. Cranberries vs antibiotics to prevent urinary tract infections: A randomized double-blind noninferiority trial in premenopausal women. Arch Intern Med. 2011;171(14):1270–1278. doi: 10.1001/archinternmed.2011.306. [DOI] [PubMed] [Google Scholar]
- 9.Gupta K, Hooton TM, Stamm WE. Increasing antimicrobial resistance and the management of uncomplicated community-acquired urinary tract infections. Ann Intern Med. 2001;135(1):41–50. doi: 10.7326/0003-4819-135-1-200107030-00012. [DOI] [PubMed] [Google Scholar]
- 10.Moura A, Nicolau A, Hooton T, Azeredo J. Antibiotherapy and pathogenesis of uncomplicated UTI: Difficult relationships. J Appl Microbiol. 2009;106(6):1779–1791. doi: 10.1111/j.1365-2672.2008.04115.x. [DOI] [PubMed] [Google Scholar]
- 11.Zhanel GG, et al. NAUTICA Group Antibiotic resistance in outpatient urinary isolates: Final results from the North American Urinary Tract Infection Collaborative Alliance (NAUTICA) Int J Antimicrob Agents. 2005;26(5):380–388. doi: 10.1016/j.ijantimicag.2005.08.003. [DOI] [PubMed] [Google Scholar]
- 12.Gupta K, et al. Infectious Diseases Society of America European Society for Microbiology and Infectious Diseases International clinical practice guidelines for the treatment of acute uncomplicated cystitis and pyelonephritis in women: A 2010 update by the Infectious Diseases Society of America and the European Society for Microbiology and Infectious Diseases. Clin Infect Dis. 2011;52(5):e103–e120. doi: 10.1093/cid/ciq257. [DOI] [PubMed] [Google Scholar]
- 13.Foxman B, Ki M, Brown P. Antibiotic resistance and pyelonephritis. Clin Infect Dis. 2007;45(3):281–283. doi: 10.1086/519267. [DOI] [PubMed] [Google Scholar]
- 14.Schito GC, et al. The ARESC study: An international survey on the antimicrobial resistance of pathogens involved in uncomplicated urinary tract infections. Int J Antimicrob Agents. 2009;34(5):407–413. doi: 10.1016/j.ijantimicag.2009.04.012. [DOI] [PubMed] [Google Scholar]
- 15.Karlowsky JA, Hoban DJ, Decorby MR, Laing NM, Zhanel GG. Fluoroquinolone-resistant urinary isolates of Escherichia coli from outpatients are frequently multidrug resistant: Results from the North American Urinary Tract Infection Collaborative Alliance-Quinolone Resistance study. Antimicrob Agents Chemother. 2006;50(6):2251–2254. doi: 10.1128/AAC.00123-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sivick KE, Mobley HL. Waging war against uropathogenic Escherichia coli: Winning back the urinary tract. Infect Immun. 2010;78(2):568–585. doi: 10.1128/IAI.01000-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Uehling DT, Hopkins WJ, Elkahwaji JE, Schmidt DM, Leverson GE. Phase 2 clinical trial of a vaginal mucosal vaccine for urinary tract infections. J Urol. 2003;170(3):867–869. doi: 10.1097/01.ju.0000075094.54767.6e. [DOI] [PubMed] [Google Scholar]
- 18.Tammen H. The German Urinary Tract Infection Study Group Immunobiotherapy with Uro-Vaxom in recurrent urinary tract infection. Br J Urol. 1990;65(1):6–9. doi: 10.1111/j.1464-410x.1990.tb14649.x. [DOI] [PubMed] [Google Scholar]
- 19.Brumbaugh AR, Mobley HL. Preventing urinary tract infection: Progress toward an effective Escherichia coli vaccine. Expert Rev Vaccines. 2012;11(6):663–676. doi: 10.1586/erv.12.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Alteri CJ, Hagan EC, Sivick KE, Smith SN, Mobley HLT. Mucosal immunization with iron receptor antigens protects against urinary tract infection. PLoS Pathog. 2009;5(9):e1000586. doi: 10.1371/journal.ppat.1000586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vigil PD, Alteri CJ, Mobley HL. Identification of in vivo-induced antigens including an RTX family exoprotein required for uropathogenic Escherichia coli virulence. Infect Immun. 2011;79(6):2335–2344. doi: 10.1128/IAI.00110-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Subashchandrabose S, et al. Host-specific induction of Escherichia coli fitness genes during human urinary tract infection. Proc Natl Acad Sci USA. 2014;111(51):18327–18332. doi: 10.1073/pnas.1415959112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hagan EC, Lloyd AL, Rasko DA, Faerber GJ, Mobley HL. Escherichia coli global gene expression in urine from women with urinary tract infection. PLoS Pathog. 2010;6(11):e1001187. doi: 10.1371/journal.ppat.1001187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Walters MS, Mobley HL. Identification of uropathogenic Escherichia coli surface proteins by shotgun proteomics. J Microbiol Methods. 2009;78(2):131–135. doi: 10.1016/j.mimet.2009.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Snyder JA, et al. Transcriptome of uropathogenic Escherichia coli during urinary tract infection. Infect Immun. 2004;72(11):6373–6381. doi: 10.1128/IAI.72.11.6373-6381.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Alteri CJ, Mobley HL. Quantitative profile of the uropathogenic Escherichia coli outer membrane proteome during growth in human urine. Infect Immun. 2007;75(6):2679–2688. doi: 10.1128/IAI.00076-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Alteri CJ, Smith SN, Mobley HL. Fitness of Escherichia coli during urinary tract infection requires gluconeogenesis and the TCA cycle. PLoS Pathog. 2009;5(5):e1000448. doi: 10.1371/journal.ppat.1000448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Skaar EP, Raffatellu M. Metals in infectious diseases and nutritional immunity. Metallomics. 2015;7(6):926–928. doi: 10.1039/c5mt90021b. [DOI] [PubMed] [Google Scholar]
- 29.Frey PA, Reed GH. The ubiquity of iron. ACS Chem Biol. 2012;7(9):1477–1481. doi: 10.1021/cb300323q. [DOI] [PubMed] [Google Scholar]
- 30.Cassat JE, Skaar EP. Iron in infection and immunity. Cell Host Microbe. 2013;13(5):509–519. doi: 10.1016/j.chom.2013.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Miethke M, Marahiel MA. Siderophore-based iron acquisition and pathogen control. Microbiol Mol Biol Rev. 2007;71(3):413–451. doi: 10.1128/MMBR.00012-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Howard RL, Buddington B, Alfrey AC. Urinary albumin, transferrin and iron excretion in diabetic patients. Kidney Int. 1991;40(5):923–926. doi: 10.1038/ki.1991.295. [DOI] [PubMed] [Google Scholar]
- 33.Garcia EC, Brumbaugh AR, Mobley HL. Redundancy and specificity of Escherichia coli iron acquisition systems during urinary tract infection. Infect Immun. 2011;79(3):1225–1235. doi: 10.1128/IAI.01222-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Torres AG, Redford P, Welch RA, Payne SM. TonB-dependent systems of uropathogenic Escherichia coli: Aerobactin and heme transport and TonB are required for virulence in the mouse. Infect Immun. 2001;69(10):6179–6185. doi: 10.1128/IAI.69.10.6179-6185.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hagan EC, Mobley HL. Haem acquisition is facilitated by a novel receptor Hma and required by uropathogenic Escherichia coli for kidney infection. Mol Microbiol. 2009;71(1):79–91. doi: 10.1111/j.1365-2958.2008.06509.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Russo TA, et al. IroN functions as a siderophore receptor and is a urovirulence factor in an extraintestinal pathogenic isolate of Escherichia coli. Infect Immun. 2002;70(12):7156–7160. doi: 10.1128/IAI.70.12.7156-7160.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Johnson JR, et al. The IrgA homologue adhesin Iha is an Escherichia coli virulence factor in murine urinary tract infection. Infect Immun. 2005;73(2):965–971. doi: 10.1128/IAI.73.2.965-971.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Subashchandrabose S, Hazen TH, Rasko DA, Mobley HL. Draft genome sequences of five recent human uropathogenic Escherichia coli isolates. Pathog Dis. 2013;69(1):66–70. doi: 10.1111/2049-632X.12059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Searle LJ, Méric G, Porcelli I, Sheppard SK, Lucchini S. Variation in siderophore biosynthetic gene distribution and production across environmental and faecal populations of Escherichia coli. PLoS One. 2015;10(3):e0117906. doi: 10.1371/journal.pone.0117906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Goetz DH, et al. The neutrophil lipocalin NGAL is a bacteriostatic agent that interferes with siderophore-mediated iron acquisition. Mol Cell. 2002;10(5):1033–1043. doi: 10.1016/s1097-2765(02)00708-6. [DOI] [PubMed] [Google Scholar]
- 41.Flo TH, et al. Lipocalin 2 mediates an innate immune response to bacterial infection by sequestrating iron. Nature. 2004;432(7019):917–921. doi: 10.1038/nature03104. [DOI] [PubMed] [Google Scholar]
- 42.Brumbaugh AR, Smith SN, Mobley HL. Immunization with the yersiniabactin receptor, FyuA, protects against pyelonephritis in a murine model of urinary tract infection. Infect Immun. 2013;81(9):3309–3316. doi: 10.1128/IAI.00470-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Bolin CA, Jensen AE. Passive immunization with antibodies against iron-regulated outer membrane proteins protects turkeys from Escherichia coli septicemia. Infect Immun. 1987;55(5):1239–1242. doi: 10.1128/iai.55.5.1239-1242.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Muckerheide A, Apple RJ, Pesce AJ, Michael JG. Cationization of protein antigens. I. Alteration of immunogenic properties. Journal of Immunology. 1987;138(3):833–837. [PubMed] [Google Scholar]
- 45.Muckerheide A, Domen PL, Michael JG. Cationization of protein antigens. II. Alteration of regulatory properties. Journal of Immunology. 1987;138(9):2800–2804. [PubMed] [Google Scholar]
- 46.Border WA, Ward HJ, Kamil ES, Cohen AH. Induction of membranous nephropathy in rabbits by administration of an exogenous cationic antigen. J Clin Invest. 1982;69(2):451–461. doi: 10.1172/JCI110469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Li X, et al. Development of an intranasal vaccine to prevent urinary tract infection by Proteus mirabilis. Infect Immun. 2004;72(1):66–75. doi: 10.1128/IAI.72.1.66-75.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Su Q, Guan T, He Y, Lv H. Siderophore biosynthesis governs the virulence of uropathogenic Escherichia coli by coordinately modulating the differential metabolism. J Proteome Res. 2016;15(4):1323–1332. doi: 10.1021/acs.jproteome.6b00061. [DOI] [PubMed] [Google Scholar]
- 49.Bachman MA, et al. Klebsiella pneumoniae yersiniabactin promotes respiratory tract infection through evasion of lipocalin 2. Infect Immun. 2011;79(8):3309–3316. doi: 10.1128/IAI.05114-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Caza M, Lépine F, Dozois CM. Secretion, but not overall synthesis, of catecholate siderophores contributes to virulence of extraintestinal pathogenic Escherichia coli. Mol Microbiol. 2011;80(1):266–282. doi: 10.1111/j.1365-2958.2011.07570.x. [DOI] [PubMed] [Google Scholar]
- 51.Pesce AJ, Apple R, Sawtell N, Michael JG. Cationic antigens. Problems associated with measurement by ELISA. J Immunol Methods. 1986;87(1):21–27. doi: 10.1016/0022-1759(86)90339-x. [DOI] [PubMed] [Google Scholar]
- 52. Janeway CA Jr, Travers PT, Walport M, Shlomchik MJ (2001) Adaptive immunity to infection. Immunobiology: The Immune System in Health and Disease, ed Gibbs S (Garland Science, New York), pp 412–420.
- 53.Buyer JS, Kratzke MG, Sikora LJ. A method for detection of pseudobactin, the siderophore produced by a plant-growth-promoting pseudomonas strain, in the barley rhizosphere. Appl Environ Microbiol. 1993;59(3):677–681. doi: 10.1128/aem.59.3.677-681.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.le Roy D, et al. Activity and specificity of a mouse monoclonal antibody to ferric aerobactin. Infect Immun. 1992;60(3):768–772. doi: 10.1128/iai.60.3.768-772.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Le Roy D, et al. Results of passive and active immunization directed against ferric aerobactin in experimental enterobacterial infections in mice and chickens. Res Microbiol. 1995;146(2):167–174. doi: 10.1016/0923-2508(96)80894-1. [DOI] [PubMed] [Google Scholar]
- 56.Le Roy D, Bouchet A, Saulnier P, Pecquet S, Andremont A. Comparison of chemical assay, bioassay, enzyme-linked immunosorbent assay, and dot blot hybridization for detection of aerobactin in members of the family Enterobacteriaceae. Appl Environ Microbiol. 1993;59(3):942–944. doi: 10.1128/aem.59.3.942-944.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bergeron RJ, et al. Vibriobactin antibodies: A vaccine strategy. J Med Chem. 2009;52(12):3801–3813. doi: 10.1021/jm900119q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Hagberg L, et al. Ascending, unobstructed urinary tract infection in mice caused by pyelonephritogenic Escherichia coli of human origin. Infect Immun. 1983;40(1):273–283. doi: 10.1128/iai.40.1.273-283.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Weichhart T, Haidinger M, Hörl WH, Säemann MD. Current concepts of molecular defence mechanisms operative during urinary tract infection. Eur J Clin Invest. 2008;38(Suppl 2):29–38. doi: 10.1111/j.1365-2362.2008.02006.x. [DOI] [PubMed] [Google Scholar]
- 60.Vázquez S, et al. Kinetics of antibodies in sera, saliva, and urine samples from adult patients with primary or secondary dengue 3 virus infections. Int J Infect Dis. 2007;11(3):256–262. doi: 10.1016/j.ijid.2006.05.005. [DOI] [PubMed] [Google Scholar]
- 61.Garénaux A, Caza M, Dozois CM. The ins and outs of siderophore mediated iron uptake by extra-intestinal pathogenic Escherichia coli. Vet Microbiol. 2011;153(1-2):89–98. doi: 10.1016/j.vetmic.2011.05.023. [DOI] [PubMed] [Google Scholar]
- 62.Porcheron G, Garénaux A, Proulx J, Sabri M, Dozois CM. Iron, copper, zinc, and manganese transport and regulation in pathogenic Enterobacteria: Correlations between strains, site of infection and the relative importance of the different metal transport systems for virulence. Front Cell Infect Microbiol. 2013;3:90. doi: 10.3389/fcimb.2013.00090. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.