Abstract
Both environmental and genetic factors contribute to schizophrenia; however, the exact etiology of this disorder is not known. Animal models are utilized to better understand the mechanisms associated with neuropsychiatric diseases, including schizophrenia. One of these involves gestational administration of methylazoxymethanol acetate (MAM) to induce a developmental disruption, which in turn produces a schizophrenia-like phenotype in post-pubertal rats. The mechanisms by which MAM produces this phenotype are not clear; however, we now demonstrate that MAM induces differential DNA methylation, which may be heritable. Here we demonstrate that a subset of both second (F2) and third (F3) filial generations of MAM-treated rats displays a schizophrenia-like phenotype and hypermethylation of the transcription factor, Sp5. Specifically, ventral tegmental area of dopamine neuron activity was examined using electrophysiology as a correlate for the dopamine hyperfunction thought to underlie psychosis in patients. Interestingly, only a subset of F2 and F3 MAM rats exhibited increases in dopamine neuron population activity, indicating that this may be a unique model with a susceptibility to develop a schizophrenia-like phenotype. An increase in dopamine system function in rodent models has been previously associated with decreases in hippocampal GABAergic transmission. In line with these observations, we found a significant correlation between hippocampal parvalbumin expression and dopamine neuron activity in F2 rats. These data therefore provide evidence that offspring born from MAM-treated rats possess a susceptibility to develop aspects of a schizophrenia-like phenotype and may provide a useful tool to investigate gene–environment interactions.
INTRODUCTION
Schizophrenia is a complex and heterogeneous disorder with an unknown etiology (Tsuang and Faraone, 1995). Evidence supports that both genetic (Greenwood et al, 2013; Kiyomi Ota et al, 2013; Sacchetti et al, 2013) and environmental factors (Brown et al, 2000; Gerard and Houston, 1953; Kendell and Kemp, 1989) contribute to the development of the disease. The dopamine (Snyder, 1976; van Rossum, 1966), glutamate (Javitt and Zukin, 1991), and various genetic (Craddock et al, 2007; McClellan et al, 2007) and environmental (Brown et al, 2000; Cannon et al, 2002; Cantor-Graae, 2007) hypotheses of schizophrenia help explain phenomena observed in patients; however, it is a combination of some, or all, of these factors that likely leads to schizophrenia pathophysiology. An increased baseline activity within hippocampal subfields is commonly observed in brain imaging studies of schizophrenia patients and is associated with the severity of positive symptoms (Heckers et al, 1998; Lahti et al, 2006; Schobel et al, 2009). Likewise, animal models display aberrant ventral hippocampal (vHipp) signaling that leads to augmented dopamine system function and a behavioral phenotype associated with positive symptoms of the disease (Lodge and Grace, 2007; Perez et al, 2013). This hippocampal hyperactivity likely stems from a deficit in GABAergic transmission within the hippocampus, particularly within the subtype of interneurons containing the calcium-binding protein parvalbumin (PV; Boley et al, 2014; Konradi et al, 2011; Lewis et al, 2005; Lodge et al, 2009a; Shah and Lodge, 2013). Post-mortem studies on schizophrenia patients report a decreased PV expression in the prefrontal cortex and hippocampus (Beasley et al, 2002; Hashimoto et al, 2003; Knable et al, 2004). In addition, numerous animal models also report region-specific decreases in PV expression (Amitai et al, 2012; Chen et al, 2014; Francois et al, 2009; Jenkins et al, 2010; Lodge et al, 2009a; Penschuck et al, 2006; Wang et al, 2008). Moreover, we have recently demonstrated that a decrease in PV interneuron function is sufficient to augment hippocampal signaling and produce downstream changes in dopamine system function and behavior (Boley et al, 2014; Shah and Lodge, 2013).
Gestational administration of methylazoxymethanol acetate (MAM) produces a developmental disruption model of schizophrenia, which recapitulates a wide variety of phenotypes observed in schizophrenic patients (for review see Lodge and Grace (2009b)). However, the exact mechanisms by which MAM induces a schizophrenia-like phenotype are still unknown. MAM is a mitotoxin that selectively affects the developing central nervous system by interfering with mitosis and DNA methylation (Cattabeni and Di Luca, 1997; Matsumoto and Higa, 1966). Given the lack of significant cell loss observed in this model (Moore et al, 2006), it is likely that the effects of MAM may be secondary to aberrant DNA methylation. Indeed, here we demonstrate bidirectional alterations in DNA promoter methylation within the hippocampus of MAM-treated rats. Furthermore, some of these epigenetic alterations are heritable (ie, the hypermethylation of Sp5) and may contribute to the decrease in PV expression and augmented dopamine neuron activity observed in the second (F2) and third (F3) filial generations of MAM-treated rats.
MATERIALS AND METHODS
All experiments were performed in accordance with the guidelines outlined in the USPH Guide for the Care and Use of Laboratory Animals and were approved by the Institutional Animal Care and Use Committee of the University of Texas Health Science Center at San Antonio.
Animals
As described previously (Lodge, 2013; Moore et al, 2006), MAM treatments were performed on timed pregnant female Sprague–Dawley rats obtained from Harlan Laboratories on gestational day (GD) 16 and housed individually in plastic tubs. MAM (diluted in saline, 25 mg/kg, i.p.) was administered on GD17, whereas control rats received injections of saline (1 ml/kg, i.p.). Male pups were weaned on postnatal day 21 and were housed with littermates in groups of two to three until adulthood (>PD 60). All experiments were performed on multiple litters of MAM- and saline-treated rats. F2 generation rats were obtained by crossing saline (♂) × saline (♀), saline (♂) × MAM (♀), MAM (♂) × saline (♀), and MAM (♂) × MAM (♀). A subpopulation of MAM (♀) offspring was cross-fostered by control dams to ensure that any observable effects were not associated with aberrant maternal care. To generate F3 rats, female rats were crossed with either MAM- or saline-F2 male rats to produce multiple litters of F3 generation rats.
Methylation Array
First-generation, adult MAM- and saline-treated rats (n=3 per group) were anesthetized with sodium pentobarbital (60 mg/kg, i.p.) and rapidly decapitated. The vHipp was dissected on ice, rapidly frozen, and stored at −80 °C. Microarray analysis of DNA methylation was performed using methylated DNA immunoprecipitation (MeDIP)-chip by a commercial vendor (ArrayStar, Rockville, Maryland). In brief, genomic DNA was extracted and sonicated to random fragments of 200–1000 bp. Immunoprecipitation of methylated DNA was performed using Biomag magnetic bead-coupled mouse monoclonal antibody against 5-methylcytidine. Given that a fully methylated profile is required to determine an absolute methylation score, DNA from each sample was pooled and treated with CpG methyltransferase (M.SssI, NEB). The immunoprecipitated DNA was eluted and purified by phenol–chloroform extraction and ethanol precipitation. The total input and immunoprecipitated DNA were labeled with Cy3- and Cy5-labeled random 9-mers, respectively, and were hybridized to NimbleGen Rat DNA Methylation 385 K Promoter Plus CpG Island Array, which is a 385-k format array design containing 15 809 CpG Islands and all well-characterized promoter regions (from about −1300 to +500 bp of the TSSs). Scanning was performed with the Axon GenePix 4000B microarray scanner. Raw data for the fully methylated sample and test samples were median-centered and quantile-normalized using Bioconductor packages Ringo and limma. Then MEDME (modeling experimental data with MeDIP enrichment) was performed to calculate an absolute methylation score. A t-test was performed to calculate the p-values for each probe, and those containing both a p-value<0.05 and an AMS difference greater than eight were used to identify differentially methylated regions (DMRs) that were mapped to genomic features.
Methylation Sequencing
As the microarrays used to examine DNA methylation in the F1 generation were discontinued, a complementary method, MeDIP-seq, was used to analyze DNA methylation in the F2 rats. Adult MAM and saline F2 rats (n=4 per group) were anesthetized with sodium pentobarbital (60 mg/kg, i.p.) and rapidly decapitated. The vHipp was dissected on ice, rapidly frozen, and stored at −80 °C. DNA samples were extracted from the rat brain using the DNeasy Blood and Tissue Kit (Qiagen, Valencia, CA) using the manufacturer's protocol with slight modifications (addition of 60 μl proteinase K and incubation for 4 h; elution with 2 × 75 μl AE buffer). For each sample, 1.1 μg of DNA normalized to 20 ng/μl was subjected to shearing by the Covaris S220 Ultra Sonicator (Covaris, Woburn, MA), using the following conditions: duty cycle of 10%, an intensity of 5200 cycles per burst, 120 s, frequency sweeping mode, 23 W power, and temperature 6–8 °C. The NEBNext UltraTM DNA Library Prep Kit for Illumina (New England Biolabs, Ipswich, MA) was used to perform end repair, adapter ligation, and sample cleanup without size selection using 1 μg DNA, according to the manufacturers' protocol. Unmethylated adapters were obtained from the NEBNext Multiplex Oligos for Illumina Kit to allow for multiplexing of samples. Adapter-ligated samples underwent immunoprecipitation with a 5-methylcytosine antibody using the MagMeDIP Kit (Diagenode, Denville, NJ), according to the manufacturers' instructions with the modification that the starting volume of DNA for the IP incubation mix was 23 μl (volume of water adjusted). Owing to the high pH of the elution buffer used, we performed a pH adjustment by adding 8 μl of 100 mM Tris-HCl to the 100 μl volume of sample and then performed sample cleanup using Agencourt AMPure XP beads (Beckman Coulter, Brea, CA) according to the standard protocols (starting with 1.8 × the sample volume for the resuspended bead mix, including two 70% ethanol washes and resuspension in 26 μl 10 mM Tris-HCl). PCR amplification (12 cycles) and cleanup were carried out using the NEBNext UltraTM DNA Library Prep Kit from Illumina, incorporating the primer provided in the NEBNext Multiplex Oligos for Illumina Kit, according to the manufacturers' protocol. Amplified products were verified for size (~300–1000 bp, peak at 500 bp) using the Agilent High Sensitivity DNA Kit (Agilent Technologies, Santa Clara, CA) and samples were pooled in equimolar quantities for sequencing analysis. Samples underwent 200-bp paired-end sequencing runs on the Illumina HiSeq2500 instrument (Illumina, San Diego, CA), according to the manufacturer's instructions.
Raw sequence data were processed by the Illumina base-calling pipeline and de-multiplexed. Sequence quality was ascertained using FastQC (http://www.bioinformatics.bbsrc.ac.uk/projects/fastqc), and reads were trimmed (10 bp at 5′ and 5 bp at 3′) and filtered for quality (>30% N calls or >10% of the sequence with Phred quality <20 were omitted) using CutAdapt (Martin, 2011). The R environment was used for all subsequent downstream analyses. Trimmed filtered reads were aligned to the reference genome (BSgenome.Rnorvegicus.UCSC.rn5.masked) using QuasR (Gaidatzis et al, 2014) with Rbowtie as the aligner (Langmead et al, 2009) set to default parameters. Aligned paired reads (mean total per sample 17 162 721, s=2 694 841) were used for subsequent DMR identification. The mean gap between paired reads among all samples was 263, s=6.6.
The Bioconductor package MEDIPS (Lienhard et al, 2014) was used to identify DMRs from the MeDIP-seq data. Initially, each sample was verified to have sufficient read saturation for reproducibility, and then analyzed for CpG content and CpG enrichment. Two enrichment scores were reported by MEDIPS: the average RelH score, based on the relative frequency of CpGs within the regions, was 1.91875 (s=0.053721); the average GoGe, based on the observed/expected ratio of CpGs within the regions, was 1.2825 (s=0.026339), indicating that the IP experiment successfully enriched the samples for methylated cytosine. Commands were executed with a genome window size of 100 nt (ws=100) and the extend parameter at 300 nt (extend=300).
DMRs between saline- and MAM-treated rats were identified using the edgeR functionality in MEDIPS (Robinson et al, 2010) with Bonferroni correction for multiple testing and a p-value threshold of 0.1. Adjacent locations were merged using the mr.edgeR.s.m command, followed by annotation of the results. Annotation data were obtained from the Rattus norvegicus genes (Rnor_5.0) data set from biomaRt (http://www.biomart.org) and the rno5 refFlat file from UCSC Genome Browser (Karolchik et al, 2014).
Extracellular Recordings
F2 and F3 generation offspring (300–500 g) were anesthetized with 8% chloral hydrate (400 mg/kg, i.p.). This anesthetic drug was used as it does not significantly depress dopamine neuron activity (Hyland et al, 2002). Rats were then placed in a stereotaxic apparatus and a core body temperature of 37 °C was maintained using a thermostatically controlled heating pad (TCAT-2LV; Kopf Instruments, USA). Anesthesia was maintained by supplemental administration of chloral hydrate as required to maintain suppression of limb compression withdrawal reflex. An extracellular glass microelectrode (impedance 6–14 MΩ) was lowered into the ventral tegmental area (VTA; A/P ±5.3, M/L ±0.6 from bregma and D/V −6.5 to −9.0 mm ventral of brain surface) to measure dopamine neuron activity. Spontaneously active dopamine neurons were identified with open filter settings (low pass: 30 Hz; high pass: 30 kHz) using previously established electrophysiological criteria (Grace and Bunney, 1983). Once isolated, dopamine neuron activity was recorded for 2–3 min. Three parameters of dopamine neuron activity were measured: population activity (defined as the number of spontaneously active dopamine neurons encountered while making six to nine vertical passes, separated by 200 μm in a predetermined pattern, to sample equivalent regions of the VTA); basal firing rate; and the proportion of action potentials occurring in bursts (Grace and Bunney, 1984).
Western Blot
F2 generation rats (300–500 g) were killed by rapid decapitation immediately following electrophysiological recordings. The vHipp was dissected on ice and homogenized in ice-cold homogenization buffer (50 mM Tris-HCl, 150 mM NaCl, 0.1% Triton X-100, and pH 7.4) containing a protease inhibitor cocktail (Sigma-Aldrich, St Louis, MO, USA). Homogenates were centrifuged at 14 000 r.p.m. for 10 min and the supernatant containing protein fractions was analyzed for PV and GAPDH expression levels. Proteins (20 μg per well) were separated (35–45 min at 200 V) on Mini-PROTEAN TGX Any kD gels (Bio-Rad, Hercules, CA, USA) in Tris/glycine/SDS buffer (Bio-Rad) and subsequently transferred on nitrocellulose/filter paper sandwiches, 0.2 μm (Bio-Rad) with transfer buffer (25 mM Tris-HCl, 192 mM glycine, 20% w/v ethanol; pH 8.3) for 1 h at 100 V. Membranes were blocked for 30 min (PV—2% milk in TBST or GAPDH—5% BSA in TBST; TBST—15 mM Tris-HCl, 137 mM NaCl, and 0.1% Tween 20) before incubation with primary anti-PV (1 : 5000) or anti-GAPDH antibody (1 : 1000; 1 h; 4 °C). Next, membranes were washed three times in TBST (10 min each) and incubated with secondary antibodies (1 : 5000) at room temperature (1 h; PV: goat anti-rabbit conjugated to HRP; GAPDH: goat anti-mouse conjugated to HRP). Lastly, membranes were treated with Pierce ECL Western Blotting Substrate (1 min; Thermo Scientific, Rockford, IL, USA; #32106) and exposed to high-performance chemiluminescence film (GE Healthcare; Amersham Hyperfilm ECL; 28906839) before developing, scanning, and quantification with ImageJ. Membranes were then washed three times in TBST (10 min) and stripped with Restore Western Blot Stripping Buffer (Thermo Scientific) following PV probing to allow for re-probing for GAPDH.
Analysis
The MEDME analysis and statistics from the F1 generation were performed using ArrayStar that resulted in the identification of 1436 DMRs. Not surprisingly, the sequencing from the F2 generation yielded a lower number of DMRs (181). These data were utilized for functional, canonical, and pathway analyses with IPA (Ingenuity Systems, www.ingenuity.com). Statistical examinations of the pathway analysis accounted for multiple corrections by utilizing the Benjamini–Hochberg method (Benjamini and Hochberg, 1995). Electrophysiological analysis of dopamine neuron activity was performed with commercially available computer software (LabChart version 7.1; ADInstruments, Chalgrove, Oxfordshire, UK) and was analyzed using the Prism software (GraphPad Software, San Diego, CA). Western blot films were scanned on an Epson Perfection V700 and optical density measured using ImageJ. Data are represented as the mean±SEM unless stated otherwise, with n-values representing the number of neurons recorded or animals per experimental group, where indicated. All statistics were calculated using the Prism software (GraphPad Software) or SigmaPlot (Systat Software, Chicago, IL, USA). Nonparametric tests were used when data failed the Shapiro–Wilk test for normality (p<0.001).
Materials
MAM was purchased from the Midwest Research Institute (Kansas City, MO, USA). Chloral hydrate, protease inhibitor cocktail (P8340), and anti-mouse IgG-HRP (A4416) were purchased from Sigma-Aldrich. Anti-PV (Rb pAb to PV; ab11427), anti-GAPDH (Ms mAb to GAPDH; ab9484), and goat pAb to Rb IgG (HRP; ab6721) were purchased from ABCAM. Laemmli sample buffer (#161–0737), 10 × Tris/glycine/SDS buffer (#161-0732), Mini-PROTEAN TGX Any kD gels (#456-9035), and nitrocellulose/filter paper sandwiches, 0.2 μm (#162-0213), were purchased from Bio-Rad. Pierce ECL western blotting substrate (#32106) and Restore western blot stripping buffer (#21059) were purchased from Thermo Scientific. High-performance chemiluminescence film (Amersham Hyperfilm ECL; 28906839) was purchased from GE Healthcare. All other chemicals and reagents were of either analytical or laboratory grade, and were purchased from standard suppliers.
RESULTS
DNA Methylation of F1 and F2 Rats
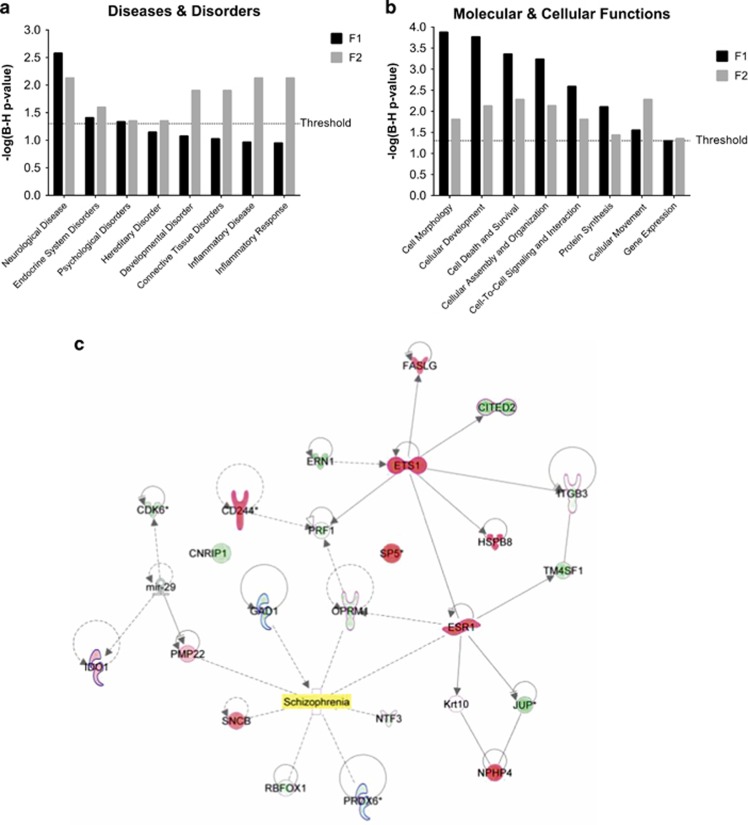
MeDIP-chip was utilized to examine the absolute methylation state of DNA promoters throughout the vHipp of F1 MAM- and saline-treated rats. Of the 385-k probes examined, only 1436 DMRs were observed (Supplementary Table 1). Given that these microarrays were discontinued, we utilized MeDIP-seq to examine DNA methylation state in the F2 hippocampus, which yielded 181 DMRs (Supplementary Table 1). It is important to note that both approaches utilize the MeDIP purification technique to enrich for methylated DNA sequences and that there is a high concordance between data obtained using sequencing and microarray approaches (Wang et al, 2014). Software-driven, independent comparative analyses of the DMRs in F1 and F2 rats demonstrated significant overlapping alterations associated with neurological disease, developmental disorders, and cell signaling (Figure 1a and b). In addition, the pathway analyses demonstrated significant alterations in F2 rats that included a number of systems associated with schizophrenia (Figure 1c). It is important to note that one gene, the transcription factor Sp5, was similarly hypermethylated in both the F1 and F2 rats and may demonstrate a heritable epigenetic alteration in MAM-treated rats.
Figure 1.
Methylazoxymethanol acetate (MAM) administration on gestational day (GD) 17 produces alterations in hippocampal DNA promoter methylation in F1 (detected by methylated DNA immunoprecipitation (MeDIP)-chip) and F2 (detected by MeDIP-seq) rats. Computer-assisted pathway analysis demonstrates similar statistically significant associations with specific diseases and disorders (a), as well as molecular and cellular functions (b). Select genes in the F2 population determined by independent pathway analysis as being related to schizophrenia are shown in c. F2 MAM rats display either increased (red) or decreased (green) absolute methylation scores relative to F2 saline controls. It is important to note that the transcription factor, Sp5, was similarly hypermethylated in both F1 and F2 generations.
Electrophysiology
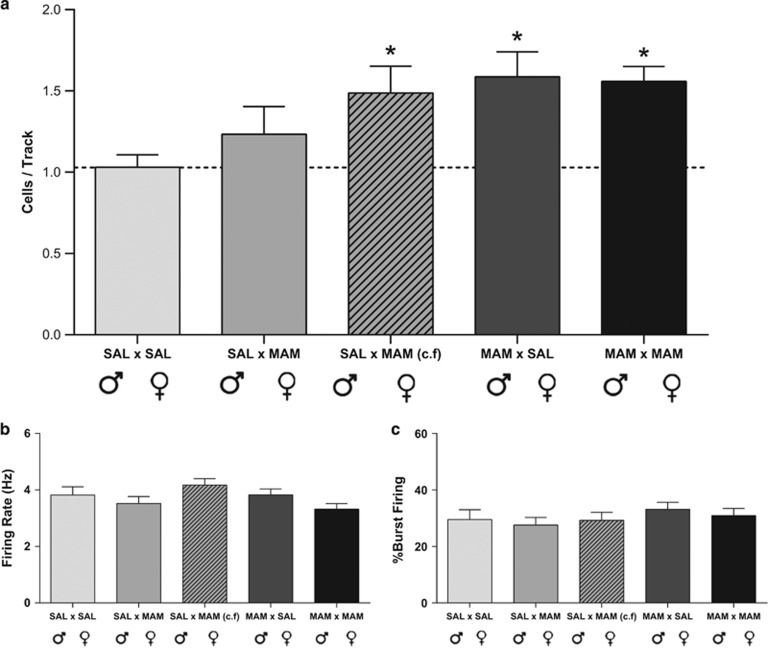
MAM-treated rats consistently exhibit elevated VTA dopamine neuron population activity when compared with saline-treated rats (Lodge and Grace, 2007; Perez et al, 2013). To investigate whether rats born from MAM- and saline-parents could inherit this schizophrenia-like phenotype, we recorded spontaneously active VTA dopamine neurons. Saline (♂) × saline (♀) F2 rats (n=11; Figure 2a) displayed an average of 1.03±0.08 spontaneously active dopamine neurons per track, which is consistent with previous findings in both untreated and saline-treated rats (Lodge and Grace, 2007; Perez et al, 2013). Saline (♂) × MAM (♀) F2 rats (n=10) displayed an average of 1.24±0.59 spontaneously active dopamine neurons and were not significantly different from controls. Cross-fostered saline (♂) × MAM (♀) F2 rats (n=10; 1.49±0.17, one-wayANOVA; Holm–Sidak; t=2.35; p=0.05), MAM (♂) × saline (♀) F2 rats (n=12; 1.59±0.53; t=3.00; p=0.02), and MAM (♂) × MAM (♀) F2 rats (n=12; 1.56±0.31; t=2.85; p=0.02) all displayed a significantly greater number of spontaneously active dopamine neurons when compared with saline control rats (Figure 2a). There were no significant differences between any of the groups with regards to average firing rates (n=68–114 neurons/group; Kruskal–Wallis ANOVA; Figure 2b) or bursting activity (n=67–114 neurons/group; Kruskal–Wallis ANOVA; Figure 2c).
Figure 2.
Offspring born from methylazoxymethanol acetate (MAM)-treated parents exhibit enhanced ventral tegmental area (VTA) dopamine neuron activity. F2 generation rats born from MAM-treated fathers, regardless of the phenotype of the mother, display a significant increase in dopamine neuron population activity (a) when compared with F2 rats born from saline parents. Cross-fostered (c.f., dashed bar) rats born from MAM-treated mothers, but raised by control dams, also demonstrated an increase in dopamine neuron population activity. No significant differences were observed in the firing rate (b) or burst firing (c) of dopamine neurons between any crosses. *represents a significant difference from saline (♂) × saline (♀).
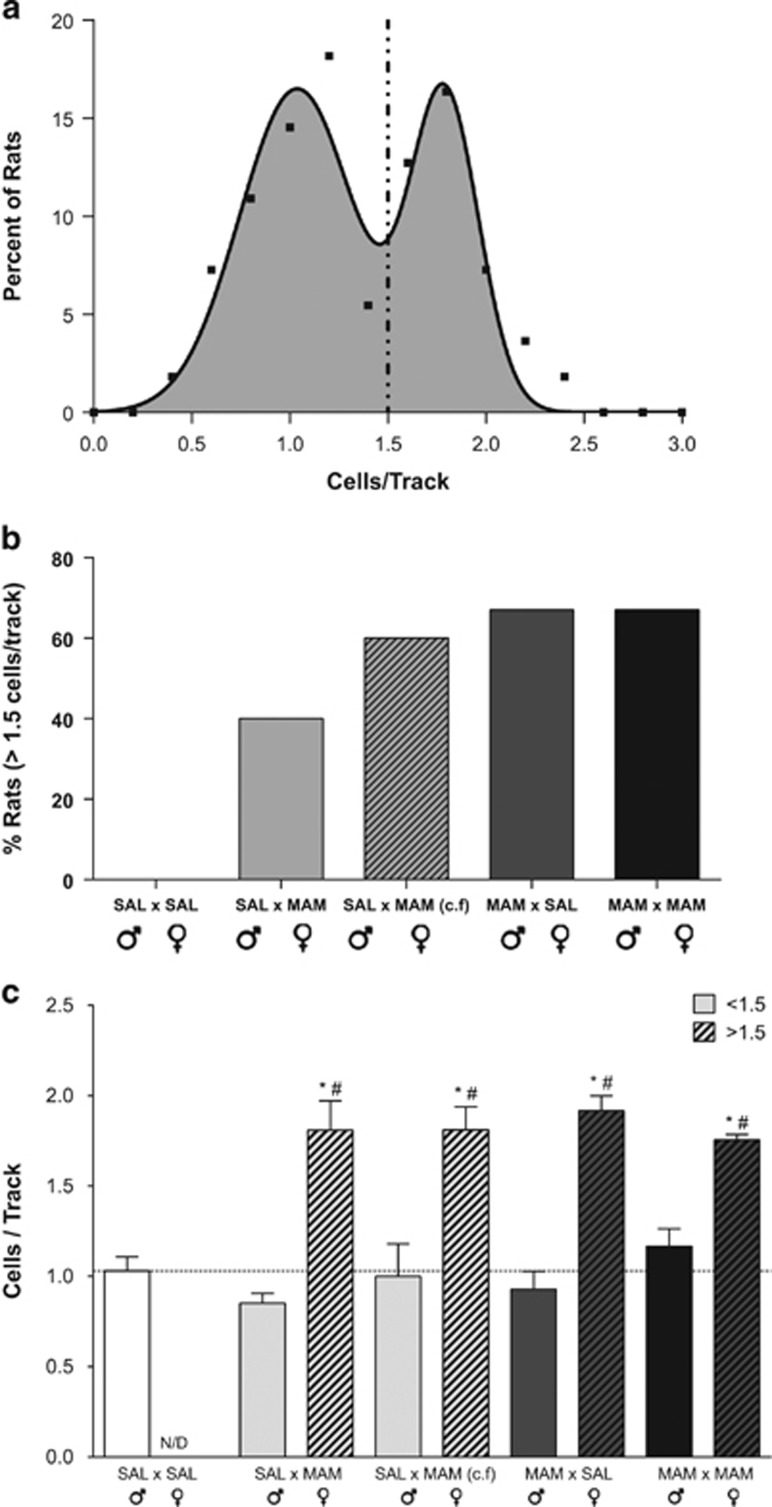
Further analysis of VTA dopamine neuron activity revealed a clear bimodal distribution with an R2 of 0.92 (Figure 3a). On the basis of this distribution, two distinct groups of offspring were identified: those that inherited (⩾1.5 cells/track) and those that did not (<1.5 cells/track) display a schizophrenia-like phenotype (ie, elevated dopamine neuron population activity). The percentage of F2 rats displaying ⩾1.5 cells/track (Figure 3b) was not significantly different whether born from a MAM-treated father or mother, or whether both parents were exposed to MAM (saline (♂) × MAM (♀)=40% cross-fostered saline (♂) × MAM (♀)=60% MAM (♂) × saline (♀)=67% MAM (♂) × MAM (♀)=67% Figure 3b). As mentioned previously, all saline (♂) × saline (♀) F2 rats (n=11; Figure 3c) displayed an average of 1.03±0.08 spontaneously active dopamine neurons/track, whereas in other groups this differed. Saline (♂) × MAM (♀) F2 rats with <1.5 cells/track (n=6) displayed an average of 0.85±0.05, whereas those with ⩾1.5 cells/track (n=4) had an average of 1.81±0.16 (one-way ANOVA; Holm–Sidak; t=6.241, p<0.05); cross-fostered saline (♂) × MAM (♀) F2 rats with <1.5 cells/track (n=4) displayed an average of 1.00±0.18, whereas those with ⩾1.5 cells/track (n=6) had an average of 1.81±0.13 (t=5.282, p<0.05); MAM (♂) × saline (♀) with <1.5 cells per track (n=4) displayed an average of 0.93±0.10 and those with ⩾1.5 cell/track (n=8) displayed an average of 1.92±0.06 (t=6.783, p<0.05); whereas MAM (♂) × MAM (♀) with <1.5 cell/track (n=4) displayed an average of 1.17±0.10 and ⩾1.5 cells per track (n=8) displayed an average of 1.76±0.03 (t=4.045, p<0.05). An all pairwise multiple comparison using the Holm–Sidak method also revealed significant differences between saline (♂) × saline (♀) offspring when compared with offspring with a MAM parent displaying ⩾1.5 cells/track (saline (♂) × MAM (♀), t=5.610; cross-fostered saline (♂) × MAM (♀), t=6.467; MAM (♂) × saline (♀), t=8.019; and MAM (♂) × MAM (♀), t=6.565; all p<0.05).
Figure 3.
A subset of F2 generation rats displays an augmented dopamine neuron population activity. Examination of the data demonstrated that dopamine population activity followed a bimodal distribution with an R2 of 0.93 (a). The percentage of F2 generation rats displaying a population activity ⩾1.5 is presented in (b) and the number of cells/track recorded for each subset is depicted in (c). *represents a significant difference from respective <1.5 cells/track population activity; #represents a significant difference from saline (♂) × saline (♀) rats.
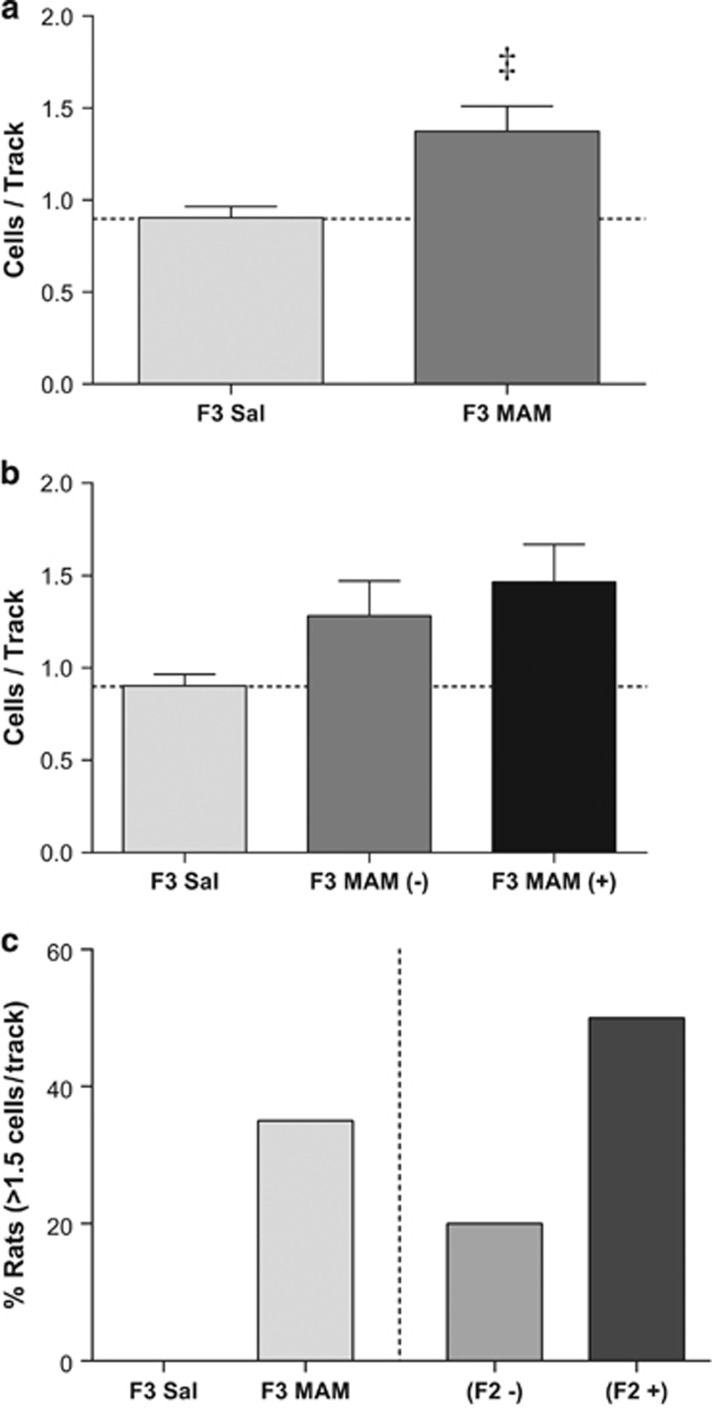
Although a subpopulation of the F2 rats clearly demonstrates a schizophrenia-like phenotype, this is not strictly evident for a transgenerational phenotype per se. To examine transgenerational inheritance, F2 males were bred with control females to produce an F3 generation. F2 males were utilized regardless of whether they displayed a schizophrenia-like phenotype. Consistent with the results obtained in the F2 generation, F3 MAM rats displayed a significant increase in dopamine neuron population activity (Figure 4a: n=20; 1.38±0.14; rank-sum test; U=35.00; p=0.05) when compared with saline control rats (n=7; 0.90±0.06). Interestingly, this was observed regardless of whether the MAM F2 parent displayed the phenotype of ⩾1.5 cells/track (Figure 4b and c). Thus, these data are consistent with a transgenerational inheritance of the schizophrenia-like phenotype.
Figure 4.
A subset of F3 generation rats displays an augmented dopamine neuron population activity. F3 generation rats born from F2 methylazoxymethanol acetate (MAM) fathers display a significant increase in dopamine neuron population activity (a) when compared with F3 rats born from F2 saline parents. Interestingly, this phenotype was observed regardless of whether the F2 MAM father displayed an increase in dopamine neuron population activity (MAM (+)) or no change in activity (MAM (−)) (b). The percentage of F3 generation rats displaying a population activity ⩾1.5 is presented in c. ‡p=0.05 from F3 saline rats.
PV Expression
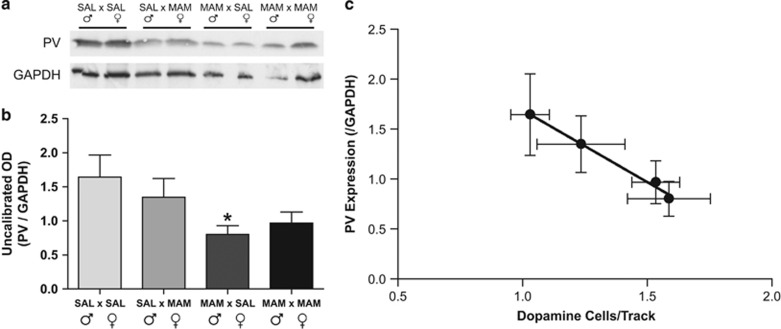
As mentioned previously, MAM-treated rats exhibit a decrease in vHipp PV expression (Chen et al, 2014; Lodge et al, 2009a), which is thought to underlie the hippocampal hyperactivity and corresponding augmented dopamine neuron population activity (Boley et al, 2014; Shah and Lodge, 2013). To examine whether this was also observed in the F2 generation from MAM- and saline-treated rats, we examined PV expression throughout the vHipp of these offspring. F2 rats born from MAM-treated parents showed reduced PV expression in the vHipp, with a significant decrease in the MAM (♂) × saline (♀) group when compared with saline (♂) × saline (♀) group (n=10–14 rats/group in duplicate, one-way ANOVA; Holm–Sidak; t=2.660 p<0.05; Figure 5b). Further analysis indicated a robust and significant negative correlation between the expression of PV/GAPDH and the number of dopamine cells/track, in that with decreasing PV expression F2 rats exhibited an increase in the population activity of dopamine neurons (Pearson's correlation R2=0.99; p=0.005; Figure 5c).
Figure 5.
F2 generation rats display decreased levels of ventral hippocampal (vHipp) parvalbumin (PV). Representative films demonstrating PV levels in the vHipp are depicted in a, whereas the quantification of these data is presented in b. *represents a significant difference from saline (♂) × saline (♀) rats. There is a significant correlation between PV expression in the vHipp and the population activity of dopamine neurons in the ventral tegmental area (VTA; Pearson correlation; R2=0.9908; c).
DISCUSSION
In the MAM rodent model of schizophrenia, augmented dopamine system function (measured by directly recording spontaneous dopamine neuron activity in the VTA) and aberrant behaviors, such as the hyper-responsivity to psychomotor stimulants and deficits in cognitive flexibility, are mediated by increased hippocampal activity (Gill et al, 2011; Lodge and Grace, 2007; Perez et al, 2013). Similarly, schizophrenia patients exhibit enhanced baseline hippocampal activity that is correlated with the severity of positive symptoms (Schobel et al, 2009). When compared with saline-treated rats, MAM-treated rats consistently display a twofold increase in dopamine neuron population activity, defined as the number of spontaneously active neurons observed per electrode track, without any changes in the frequency or bursting pattern of these neurons (Lodge and Grace, 2007; Perez et al, 2013). Dopamine neuron population activity is thought to provide a gain of function to the dopamine system. Thus, an aberrant increase in population activity may confer high salience to typically nonsalient stimuli. In this study, we showed that an increase in dopamine neuron population activity is inherited by rats born from MAM-treated parents. Interestingly, only a subpopulation of offspring appeared to exhibit the phenotype. Thus, rather than a standard Mendelian inheritance, it appears that the F2 generation displays a susceptibility to develop a schizophrenia-like phenotype, as measured by dopamine neuron activity. The F2 MAM generation may therefore provide a novel animal model to examine the role of environmental factors in the development of schizophrenia. Indeed, environmental factors have been demonstrated to contribute to the disease in humans (Vereczkei and Mirnics, 2011). Specifically, fetal hypoxia (Mittal et al, 2008), maternal stress (Malaspina et al, 2008; Van Os and Selten, 1998), and adolescent cannabis exposure (Caspi et al, 2005), to name a few, are risk factors for schizophrenia.
It should be noted that rearing conditions of the F2 offspring may contribute to their schizophrenia-like phenotype. To rule out this confounder, we cross-fostered a subset of pups from a MAM-treated mother and found that the increase in dopamine neuron activity was still observed in these animals. Interestingly, the most robust phenotypes were observed in F2 offspring of MAM fathers that were mated with saline-treated females (rats that were nurtured by control female rats throughout the pre-weaning period). Taken together, these data suggest that the phenotype observed in the F2 generation is likely associated with epigenetic alterations rather than environmental factors, such as maternal neglect.
We previously suggested that a decrease in interneuron function contributes to the augmented hippocampal activity (Lodge et al, 2009a; Lodge and Grace, 2011) that underlies the aberrant dopamine system function in MAM-treated rats (Lodge and Grace, 2007). Decreases in PV-positive interneurons have been repeatedly reported in MAM-treated rats (Chen et al, 2014; Lodge et al, 2009a; Penschuck et al, 2006), as well as in schizophrenia patients (Konradi et al, 2011; Lewis et al, 2005). Furthermore, the selective degradation of vHipp perineuronal nets, which is the support structure of PV interneurons, is sufficient to produce a schizophrenia-like phenotype comparable to that seen in the MAM model (Shah and Lodge, 2013). Similarly, a loss of vHipp PV expression itself produces comparable effects (Boley et al, 2014). Here we demonstrated that PV protein in vHipp is significantly reduced in the offspring of MAM-treated rats; moreover, we found a significant correlation between the expression of PV and the population activity of dopamine neurons, such that the F2 generation rats with decreased levels of PV throughout the vHipp displayed significantly higher dopamine neuron population activity in the VTA.
The exact mechanisms by which MAM administration causes a schizophrenia-like phenotype in rats is not known. However, given that MAM is a potent alkylating agent (Matsumoto and Higa, 1966), we examined the effect of GD17 MAM administration on the epigenetic regulation of DNA promoters throughout the vHipp using microarray. Interestingly, MAM administration resulted in specific and bidirectional alterations in DNA methylation throughout a relatively small number of promoters (<1% of the probes analyzed). Analysis of these DMRs identified a number of genes consistently implicated in schizophrenia, including markers associated with glutamatergic and GABAergic signaling. To determine whether these epigenetic alterations could be inherited by subsequent generations, we examined DNA methylation in F2 MAM rats using MeDIP-seq. Similar to that observed in F1 MAM-treated rats, the F2 generation displayed bidirectional alterations in DNA methylation, albeit in a smaller number of promotors. Independent pathway analysis demonstrated similar associations with neurological disease, psychological disorders, and developmental disorders between the F1 and F2 groups. Moreover, alterations associated with cellular development, morphology, and signaling were also observed in both groups of rats. It is important to note that one gene, Sp5, was hypermethylated in both F1 and F2 MAM rats. Sp5 belongs to the family of Sp1-like transcription factors and is the known regulator of embryonic development (Zhao and Meng, 2005). Thus, it is possible that aberrant Sp5 methylation may be inherited by subsequent generations to confer a susceptibility to develop a schizophrenia-like phenotype; however, this remains to be conclusively demonstrated.
Although we have not examined genetic alterations that may be occurring at the level of the male germline, it is well established that paternal age is a major risk factor for schizophrenia in humans, even in families with no history of psychosis (Malaspina, 2001; Malaspina et al, 2002). It has been previously suggested that the genes associated with increased risk in familial cases of schizophrenia may be the same ones that are affected with increasing paternal age (Malaspina, 2001). It has also been documented that there is a dose-dependent increase in the risk of developing schizophrenia with advancing paternal age (Zammit et al, 2003). Given that gonocytes are present on GD17 (Guraya and Uppal, 1977), it is possible that the prenatal administration of MAM produces genetic alterations in the testes that contribute to a schizophrenia-like phenotype. Indeed, F2 offspring from MAM-treated fathers display greater dopamine neuron activity and decreased PV expression when compared with offspring from saline-treated fathers.
Taken as whole, our study provides evidence that a schizophrenia-like phenotype is inherited by the F2 and F3 generations following a gestational disruption with MAM. Given that only a subpopulation of F2 and F3 rats display an increase in dopamine neuron activity, we posit that this may represent a novel animal model that has an increased susceptibility for a schizophrenia-like phenotype. Such a model is likely to be beneficial for future studies examining gene–environment interactions contributing to the etiology of schizophrenia.
FUNDING AND DISCLOSURE
This work was supported by R01 (MH090067), R01 (MH091130), and F31 (MH098564) from the NIH. The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies the paper on the Neuropsychopharmacology website (http://www.nature.com/npp)
Supplementary Material
References
- Amitai N, Kuczenski R, Behrens MM, Markou A (2012). Repeated phencyclidine administration alters glutamate release and decreases GABA markers in the prefrontal cortex of rats. Neuropharmacology 62: 1422–1431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beasley CL, Zhang ZJ, Patten I, Reynolds GP (2002). Selective deficits in prefrontal cortical GABAergic neurons in schizophrenia defined by the presence of calcium-binding proteins. Biol Psychiatry 52: 708–715. [DOI] [PubMed] [Google Scholar]
- Benjamini Y, Hochberg Y (1995). Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B (Methodol) 57: 289–300. [Google Scholar]
- Boley AM, Perez SM, Lodge DJ (2014). A fundamental role for hippocampal parvalbumin in the dopamine hyperfunction associated with schizophrenia. Schizophr Res 157: 238–243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown AS, Schaefer CA, Wyatt RJ, Goetz R, Begg MD, Gorman JM et al (2000). Maternal exposure to respiratory infections and adult schizophrenia spectrum disorders: a prospective birth cohort study. Schizophr Bull 26: 287–295. [DOI] [PubMed] [Google Scholar]
- Cannon M, Jones PB, Murray RM (2002). Obstetric complications and schizophrenia: historical and meta-analytic review. Am J Psychiatry 159: 1080–1092. [DOI] [PubMed] [Google Scholar]
- Cantor-Graae E (2007). The contribution of social factors to the development of schizophrenia: a review of recent findings. Can J Psychiatry 52: 277–286. [DOI] [PubMed] [Google Scholar]
- Caspi A, Moffitt TE, Cannon M, McClay J, Murray R, Harrington H et al (2005). Moderation of the effect of adolescent-onset cannabis use on adult psychosis by a functional polymorphism in the catechol-O-methyltransferase gene: longitudinal evidence of a gene X environment interaction. Biol Psychiatry 57: 1117–1127. [DOI] [PubMed] [Google Scholar]
- Cattabeni F, Di Luca M (1997). Developmental models of brain dysfunctions induced by targeted cellular ablations with methylazoxymethanol. Physiol Rev 77: 199–215. [DOI] [PubMed] [Google Scholar]
- Chen L, Perez SM, Lodge DJ (2014). An augmented dopamine system function is present prior to puberty in the methylazoxymethanol acetate rodent model of schizophrenia. Dev Neurobiol 74: 907–917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Craddock N, O'Donovan MC, Owen MJ (2007). Phenotypic and genetic complexity of psychosis. Invited commentary on... Schizophrenia: a common disease caused by multiple rare alleles. Br J Psychiatry 190: 200–203. [DOI] [PubMed] [Google Scholar]
- Francois J, Ferrandon A, Koning E, Angst MJ, Sandner G, Nehlig A (2009). Selective reorganization of GABAergic transmission in neonatal ventral hippocampal-lesioned rats. Int J Neuropsychopharmacol 12: 1097–1110. [DOI] [PubMed] [Google Scholar]
- Gaidatzis D, Lerch A, Hahne F, Stadler MB (2014). QuasR: quantification and annotation of short reads in R. Bioinformatics 31: 1130–1132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerard DL, Houston LG (1953). Family setting and the socila ecology of schizophrenia. Psychiatr Q 27: 90–101. [DOI] [PubMed] [Google Scholar]
- Gill KM, Lodge DJ, Cook JM, Aras S, Grace AA (2011). A novel α5GABAAR-positive allosteric modulator reverses hyperactivation of the dopamine system in the MAM model of schizophrenia. Neuropsychopharmacology 36: 1903–1911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grace AA, Bunney BS (1983). Intracellular and extracellular electrophysiology of nigral dopaminergic neurons—1. Identification and characterization. Neuroscience 10: 301–315. [DOI] [PubMed] [Google Scholar]
- Grace AA, Bunney BS (1984). The control of firing pattern in nigral dopamine neurons: burst firing. J Neurosci 4: 2877–2890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenwood TA, Swerdlow NR, Gur RE, Cadenhead KS, Calkins ME, Dobie DJ et al (2013). Genome-wide linkage analyses of 12 endophenotypes for schizophrenia From the Consortium on the Genetics of Schizophrenia. Am J Psychiatry 170: 521–532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guraya SS, Uppal J (1977). Morphological and histochemical observations on the prenatal and postnatal testes of the field rat (Millardia meltada). Andrologia 9: 371–378. [DOI] [PubMed] [Google Scholar]
- Hashimoto T, Volk DW, Eggan SM, Mirnics K, Pierri JN, Sun Z et al (2003). Gene expression deficits in a subclass of GABA neurons in the prefrontal cortex of subjects with schizophrenia. J Neurosci 23: 6315–6326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heckers S, Rauch SL, Goff D, Savage CR, Schacter DL, Fischman AJ et al (1998). Impaired recruitment of the hippocampus during conscious recollection in schizophrenia. Nat Neurosci 1: 318–323. [DOI] [PubMed] [Google Scholar]
- Hyland BI, Reynolds JN, Hay J, Perk CG, Miller R (2002). Firing modes of midbrain dopamine cells in the freely moving rat. Neuroscience 114: 475–492. [DOI] [PubMed] [Google Scholar]
- Javitt DC, Zukin SR (1991). Recent advances in the phencyclidine model of schizophrenia. Am J Psychiatry 148: 1301–1308. [DOI] [PubMed] [Google Scholar]
- Jenkins TA, Harte MK, Reynolds GP (2010). Effect of subchronic phencyclidine administration on sucrose preference and hippocampal parvalbumin immunoreactivity in the rat. Neurosci Lett 471: 144–147. [DOI] [PubMed] [Google Scholar]
- Karolchik D, Barber GP, Casper J, Clawson H, Cline MS, Diekhans M et al (2014). The UCSC Genome Browser database: 2014 update. Nucleic Acids Res 42: D764–D770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kendell RE, Kemp IW (1989). Maternal influenza in the etiology of schizophrenia. Arch Gen Psychiatry 46: 878–882. [DOI] [PubMed] [Google Scholar]
- Kiyomi Ota V, Gadelha A, Pellegrino R, Leite Santoro M, Teixeira Bellucco F, Maria Christofolini D et al (2013). Candidate genes for schizophrenia in a mixed Brazilian population using pooled DNA. Psychiatry Res 208: 201–202. [DOI] [PubMed] [Google Scholar]
- Knable MB, Barci BM, Webster MJ, Meador-Woodruff J, Torrey EF (2004). Molecular abnormalities of the hippocampus in severe psychiatric illness: postmortem findings from the Stanley Neuropathology Consortium. Mol Psychiatry 9: 609–620. [DOI] [PubMed] [Google Scholar]
- Konradi C, Yang CK, Zimmerman EI, Lohmann KM, Gresch P, Pantazopoulos H et al (2011). Hippocampal interneurons are abnormal in schizophrenia. Schizophr Res 131: 165–173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lahti AC, Weiler MA, Holcomb HH, Tamminga CA, Carpenter WT, McMahon R (2006). Correlations between rCBF and symptoms in two independent cohorts of drug-free patients with schizophrenia. Neuropsychopharmacology 31: 221–230. [DOI] [PubMed] [Google Scholar]
- Langmead B, Trapnell C, Pop M, Salzberg SL (2009). Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol 10: R25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewis DA, Hashimoto T, Volk DW (2005). Cortical inhibitory neurons and schizophrenia. Nat Rev Neurosci 6: 312–324. [DOI] [PubMed] [Google Scholar]
- Lienhard M, Grimm C, Morkel M, Herwig R, Chavez L (2014). MEDIPS: genome-wide differential coverage analysis of sequencing data derived from DNA enrichment experiments. Bioinformatics 30: 284–286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lodge DJ (2013). The MAM rodent model of schizophrenia. Curr Protoc Neurosci Chapter 9: 9.43.1–9.43.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lodge DJ, Behrens MM, Grace AA (2009. a). A loss of parvalbumin-containing interneurons is associated with diminished oscillatory activity in an animal model of schizophrenia. J Neurosci 29: 2344–2354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lodge DJ, Grace AA (2007). Aberrant hippocampal activity underlies the dopamine dysregulation in an animal model of schizophrenia. J Neurosci 27: 11424–11430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lodge DJ, Grace AA (2009. b). Gestational methylazoxymethanol acetate administration: a developmental disruption model of schizophrenia. Behav Brain Res 7: 306–312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lodge DJ, Grace AA (2011). Hippocampal dysregulation of dopamine system function and the pathophysiology of schizophrenia. Trends Pharmacol Sci 32: 507–513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malaspina D (2001). Paternal factors and schizophrenia risk: de novo mutations and imprinting. Schizophr Bull 27: 379–393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malaspina D, Brown A, Goetz D, Alia-Klein N, Harkavy-Friedman J, Harlap S et al (2002). Schizophrenia risk and paternal age: a potential role for de novo mutations in schizophrenia vulnerability genes. CNS Spectr 7: 26–29. [DOI] [PubMed] [Google Scholar]
- Malaspina D, Corcoran C, Kleinhaus KR, Perrin MC, Fennig S, Nahon D et al (2008). Acute maternal stress in pregnancy and schizophrenia in offspring: a cohort prospective study. BMC Psychiatry 8: 71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin M (2011). Cutadapt removes adapter sequences from high-throughput sequencing reads (2011) EMBnet J 17: 10–12.
- Matsumoto H, Higa HH (1966). Studies on methylazoxymethanol, the aglycone of cycasin: methylation of nucleic acids in vitro. Biochem J 98: 20C–22C. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McClellan JM, Susser E, King MC (2007). Schizophrenia: a common disease caused by multiple rare alleles. Br J Psychiatry 190: 194–199. [DOI] [PubMed] [Google Scholar]
- Mittal VA, Ellman LM, Cannon TD (2008). Gene-environment interaction and covariation in schizophrenia: the role of obstetric complications. Schizophr Bull 34: 1083–1094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore H, Jentsch JD, Ghajarnia M, Geyer MA, Grace AA (2006). A neurobehavioral systems analysis of adult rats exposed to methylazoxymethanol acetate on E17: implications for the neuropathology of schizophrenia. Biol Psychiatry 60: 253–264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penschuck S, Flagstad P, Didriksen M, Leist M, Michael-Titus AT (2006). Decrease in parvalbumin-expressing neurons in the hippocampus and increased phencyclidine-induced locomotor activity in the rat methylazoxymethanol (MAM) model of schizophrenia. Eur J Neurosci 23: 279–284. [DOI] [PubMed] [Google Scholar]
- Perez SM, Shah A, Asher A, Lodge DJ (2013). Hippocampal deep brain stimulation reverses physiological and behavioural deficits in a rodent model of schizophrenia. Int J Neuropsychopharmacol 16: 1331–1339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson MD, McCarthy DJ, Smyth GK (2010). edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26: 139–140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sacchetti E, Scassellati C, Minelli A, Valsecchi P, Bonvicini C, Pasqualetti P et al (2013). Schizophrenia susceptibility and NMDA-receptor mediated signalling: an association study involving 32 tagSNPs of DAO, DAOA, PPP3CC, and DTNBP1 genes. BMC Med Genet 14: 33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schobel SA, Kelly MA, Corcoran CM, Van Heertum K, Seckinger R, Goetz R et al (2009). Anterior hippocampal and orbitofrontal cortical structural brain abnormalities in association with cognitive deficits in schizophrenia. Schizophr Res 114: 110–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shah A, Lodge DJ (2013). A loss of hippocampal perineuronal nets produces deficits in dopamine system function: relevance to the positive symptoms of schizophrenia. Transl Psychiatry 3: e215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snyder SH (1976). The dopamine hypothesis of schizophrenia: focus on the dopamine receptor. Am J Psychiatry 133: 197–202. [DOI] [PubMed] [Google Scholar]
- Tsuang MT, Faraone SV (1995). The case for heterogeneity in the etiology of schizophrenia. Schizophr Res 17: 161–175. [DOI] [PubMed] [Google Scholar]
- Van Os J, Selten JP (1998). Prenatal exposure to maternal stress and subsequent schizophrenia. The May 1940 invasion of The Netherlands. Br J Psychiatry 172: 324–326. [DOI] [PubMed] [Google Scholar]
- van Rossum JM (1966). The significance of dopamine-receptor blockade for the mechanism of action of neuroleptic drugs. Arch Int Pharmacodyn Ther 160: 492–494. [PubMed] [Google Scholar]
- Vereczkei A, Mirnics K (2011). Genetic predisposition to schizophrenia: what did we learn and what does the future hold? Neuropsychopharmacol Hung 13: 205–210. [PubMed] [Google Scholar]
- Wang C, Gong B, Bushel PR, Thierry-Mieg J, Thierry-Mieg D, Xu J et al (2014). The concordance between RNA-seq and microarray data depends on chemical treatment and transcript abundance. Nat Biotechnol 32: 926–932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang CZ, Yang SF, Xia Y, Johnson KM (2008). Postnatal phencyclidine administration selectively reduces adult cortical parvalbumin-containing interneurons. Neuropsychopharmacology 33: 2442–2455. [DOI] [PubMed] [Google Scholar]
- Zammit S, Allebeck P, Dalman C, Lundberg I, Hemmingson T, Owen MJ et al (2003). Paternal age and risk for schizophrenia. Br J Psychiatry 183: 405–408. [DOI] [PubMed] [Google Scholar]
- Zhao C, Meng A (2005). Sp1-like transcription factors are regulators of embryonic development in vertebrates. Dev Growth Differ 47: 201–211. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.