Abstract
We previously demonstrated that decreased miR-17∼92 cluster expression was 1) present in lungs from human infants who died with bronchopulmonary dysplasia (BPD); 2) inversely correlated with DNA methyltransferase (DNMT) expression and promoter methylation; and 3) correlated with a subsequent diagnosis of BPD at 36 wk gestational age. We tested the hypothesis that plasma miR-17 levels would be lowest in infants who ultimately develop severe BPD. Secondly, we utilized our well-characterized murine model of severe BPD that combines perinatal inflammation with postnatal hyperoxia to test the hypothesis that alterations in lung miR-17∼92, DNMT, and promoter methylation in our model would mirror our findings in tissues from premature human infants. Plasma was obtained during the first 5 days of life from premature infants born ≤32 wk gestation. Lung tissues were harvested from mice exposed to maternal inflammation and neonatal hyperoxia for 14 days after birth. miR-17∼92 cluster expression and DNA methyltransferase expression were measured by qRT-PCR, and promoter methylation was assessed by Methyl-Profiler assay. Plasma miR-17 levels are significantly lower in the first week of life in human infants who develop severe BPD compared with mild or moderate BPD. Data from our severe BPD murine model reveal that lung miR-17∼92 cluster expression is significantly attenuated, and levels inversely correlated with DNMT expression and miR-17∼92 cluster promoter methylation. Collectively, our data support a plausible role for epigenetically altered miR-17∼92 cluster in the pathogenesis of severe BPD.
Keywords: bronchopulmonary dysplasia, DNMT, methylation, microRNA, miR-17∼92
bronchopulmonary dysplasia (BPD) is a chronic lung disease that afflicts ∼14,000 premature infants in the United States each year. Infants with BPD often require prolonged respiratory support and/or supplemental oxygen therapy and display persistent morbidities (8, 16–18). The miR-17∼92 cluster is processed as a single transcript that, once posttranscriptionally modified, results in six individual miRNAs, miRs-17, -18a, -19a, -19b, -20a, and -92, that are highly expressed in the developing lung. Expression is regulated, in part, by methylation of promoter CpG islands via the actions of DNA methyltransferase (DNMT) enzymes including DNMT-1, -3a, and -3b (3). The miR-17∼92 promoter has ∼80% CG content in a CpG island spanning −3.3 to −1.2 kb (relative to the first nucleotide of the pre-miR-17 sequence) (10). Three DNMTs are expressed in humans: DNMT-1 is a maintenance methyltransferase while DNMT-3a and -3b encode de novo methyl transferases. There is significant functional overlap for the DNMTs, and individual knockout mice have demonstrated that all three are essential for mammalian viability (5, 11).
In our previous publication, we demonstrated that all members of the miR-17∼92 cluster were suppressed in samples obtained from infants that died with BPD compared with infants that died of other causes (13). We also observed increases in methylation within the promoter region of miR-17∼92 and this methylation correlated with increases in the expression of DNMT-1, 3a, and 3b. Furthermore, receiver operating characteristic curves indicated that decreased plasma miR-17 levels in the first week of life directly correlated with a subsequent diagnosis of any form of BPD at 36 wk postmenstrual age (13). For the present studies, we tested the hypothesis that plasma miR-17 levels within the first week of life would correlate with the severity of BPD diagnosis at term-corrected age.
While most premature infants diagnosed with chronic lung disease develop mild to moderate disease with delayed alveolarization, infants that develop severe BPD are often plagued with lifelong morbidities resulting from severe alveolarization abnormalities and interstitial lung fibrosis leading to permanently altered respiratory physiology (4, 15, 23). Most murine BPD models recapitulate the developmental lung growth deficits seen in human patients with mild to moderate BPD but few address the morbidities associated with severe BPD. Our extensively characterized murine model demonstrates the synergistic pathophysiology of perinatal maternal lipopolysaccharide (LPS)-induced inflammation and neonatal hyperoxic exposure on lung development and recapitulates the severe BPD phenotype (Table 1) (1, 19–21). Our data demonstrate increased expression of matrix proteins, as well as overall increases in TGFβ and SMAD 2/3 (Table 1). Members of the miR-17∼92 cluster are predicted to regulate the expression of several genes implicated in the pathogenesis of BPD (3, 9), including those previously characterized in our model. Therefore, the present studies were also designed to test the hypothesis that miR-17∼92 cluster expression is decreased in our mouse model of severe BPD and that altered expression was associated with epigenetic alterations involving DNMTs.
Table 1.
Characteristics of the LPS/O2 murine model
| Alterations: LPS/O2 vs. Saline/RA | |
|---|---|
| Increased | Decreased |
| Morphometrics | |
| septal thickness | alveolar number |
| alveolar area | |
| Pulmonary function | |
| tissue resistance | compliance |
| airway resistance | inspiratory capacity |
| Gene expression | |
| vimentin | IkB |
| collagen 1 & 3 | p53 |
| TGFβ | MMP-2 |
| SMAD 2/3 | MMP-3 |
| DNMT 3a and 3b | |
| αSMA | |
These data define the characteristics of the LPS/O2 murine model relevant to the BPD phenotype. RA, room air. See Refs. 1, 19–21.
METHODS
Human plasma.
Samples were obtained under protocols approved by the Institutional Review Board of Nationwide Children's Hospital. Patients were enrolled from neonatal intensive care units at Nationwide Children's Hospital and Ohio State University Hospital by written, informed consent of the parents between May 2005 and December 2010. The study was HIPAA compliant. Data from this collection, including eligibility guidelines, exclusion criteria, and methodology of sample collection/storage, have been previously published (12, 14). The BPD groups were defined, according to the NICHD/NHLBI 2001 workshop definition, based on the need for additional respiratory support at 36 wk corrected gestational age. Specifically, severe BPD was defined as the need for >30% oxygen, with or without positive-pressure ventilation or continuous positive pressure at 36 wk postmenstrual age (6).
Preparation of cationic lipoplex nanoparticles containing molecular beacons.
Levels of plasma miR-17 expression were determined as previously described (13, 24). Briefly, tethered cationic lipoplex nanoparticles containing molecular beacons for miR-17 were prepared, glass slides were incubated with an avidin derivative (NeutrAvidin; Thermo Scientific, Waltham, MA), and total internal reflection fluorescence microscopy was used to detect the fluorescence signals on the analyzed images (13, 24).
Animals and exposure.
The Institutional Animal Care and Use Committee at The Research Institute at Nationwide Children's Hospital approved animal study protocols. Our well-characterized lung injury model, which includes both male and female offspring, combines prenatal inflammatory stress (intraperitoneal LPS injection) with postnatal hyperoxic exposure (85% for 14 days) and was used to generate murine tissues as previously published (19, 20).
Quantitative real-time polymerase chain reaction analyses.
RNA was isolated using Qiagen RNA Easy kit (Qiagen, Valencia, CA) or homogenization in TRIzol reagent according to manufacturer's instructions (Invitrogen, Carlsbad, CA) from murine tissue samples and was subjected to polymerase chain reaction analysis for mRNA and miR expression as previously described (3). For miRNA analysis, endogenous controls small nucleolar RNA (sno), RNU38B, RNU6, or sno202 were evaluated for each experiment. The endogenous control with the most consistent cycle threshold value and little variation was used for normalization as indicated. For mRNA analysis, the endogenous control adenylyl cyclase-associated protein-1 was used for normalization.
DNA methylation studies.
DNA was isolated from tissue samples using QIAamp DNA Mini Kit and methylation was analyzed using Methyl-Profiler DNA methylation qPCR Primer Assays (SABiosciences/Qiagen, Frederick, MD) according to manufacturer instructions.
Statistics.
Data were tested for homogeneity of variances, log transformed when indicated, and analyzed by one-way ANOVA with Tukey post hoc analysis where appropriate using GraphPad PRISM 6 (GraphPad, La Jolla, CA). Data are expressed as means ± SD with significance noted at P < 0.05.
RESULTS
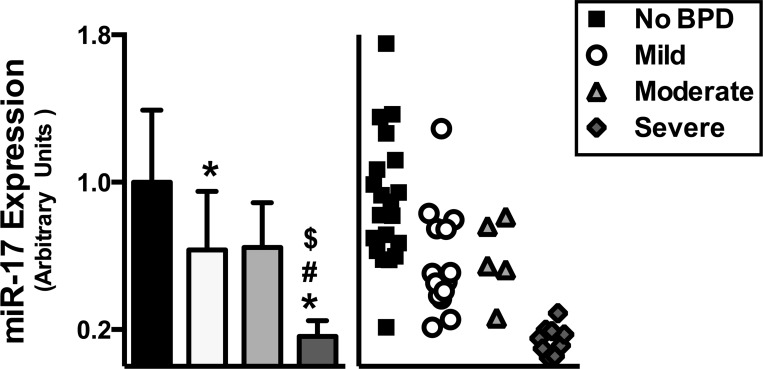
First week plasma miR-17 levels are lowest in infants who develop severe BPD.
Plasma miR-17 levels were measured using Molecular Beacon technology. While infants diagnosed with mild or moderate BPD exhibited decreased miR-17 expression, infants diagnosed with severe BPD experienced the most significant decrease (Fig. 1).
Fig. 1.
Plasma miR-17 levels in the first week of life are decreased in infants who subsequently develop severe BPD. Data, expressed as means ± SD (n = 5–20), were analyzed by one-way ANOVA followed by Tukey's post hoc analysis (*P < 0.01 vs. No BPD; #P = 0.0071 vs. mild; $P = 0.046 vs. moderate).
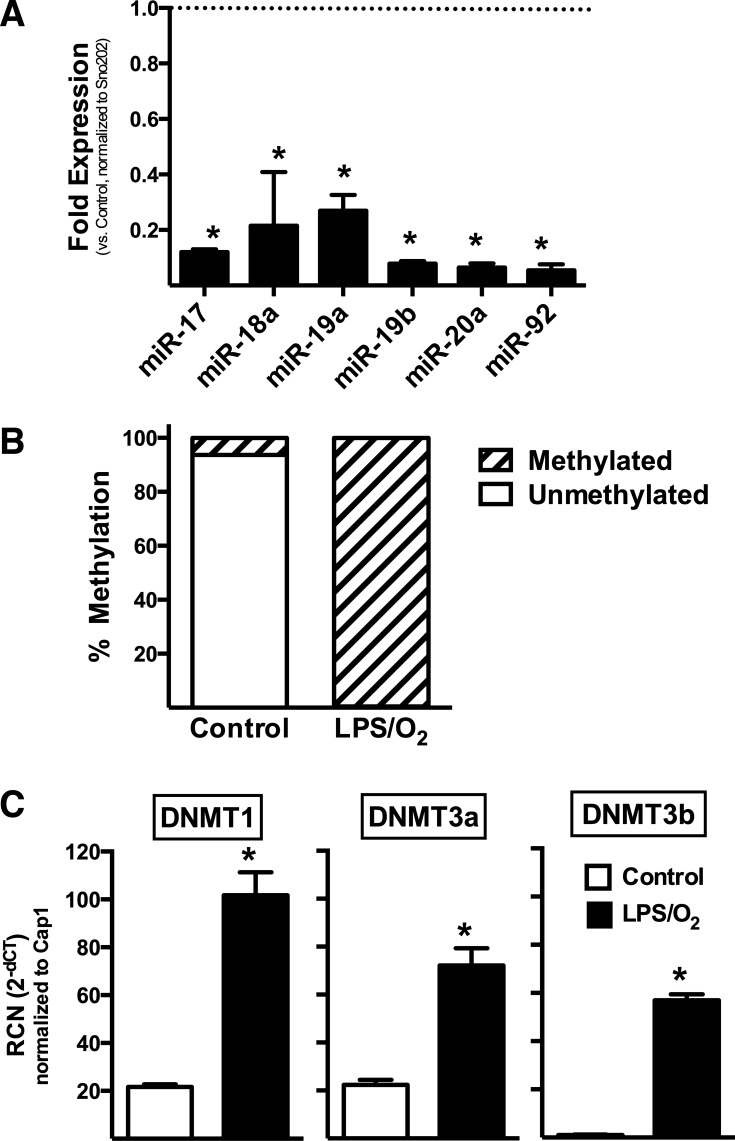
Pulmonary expression of the miR-17∼92 cluster is decreased in mice exposed to perinatal inflammation and neonatal hyperoxia.
We performed qRT-PCR using lung tissues from mouse pups born to LPS-treated dams and raised in 85% O2 (LPS/O2) from birth to 14 days. Control tissues were taken from mice born to saline-treated dams and raised in room air after birth (Control). Compared with control tissues, expression of all members of the miR-17∼92 cluster were significantly decreased (Fig. 2A).
Fig. 2.
Lung miR-17∼92 cluster expression is decreased and promoter methylation and DNMT expression increased at 14 days of life in a murine model of severe BPD. RCN, relative copy number. Transcript expression (means ± SD, n = 3–4) was determined by qRT-PCR and was analyzed by one-way ANOVA followed by Tukey's post hoc analysis (*P < 0.0001). Promoter methylation in the same samples was determined by Methyl-Profiler assay.
Lung miR-17∼92 cluster promoter methylation is increased in our severe BPD model.
Promoter methylation analysis revealed ∼2% promoter methylation in the Control mice. Conversely, promoter methylation was 98% in lungs from LPS/O2 mice (Fig. 2B).
Lung DNMT expression is increased in our severe BPD model.
To determine the impact of antenatal inflammatory and neonatal hyperoxic exposure on the expression of DNMTs in newborn murine lungs, qRT-PCR analyses were performed for DNMT-1, -3a, and -3b using lung tissues from Control and LPS/O2-exposed pups. DNMT-1, -3a, and -3b expression was significantly increased in lungs from mice exposed to the combination of antenatal LPS and postnatal hyperoxia (Fig. 2C).
DISCUSSION
Proper expression of the miR-17∼92 cluster is necessary for normal lung growth and development and alterations in expression have been reported in other pulmonary diseases (2, 7). Our previous data in human autopsy tissue and current data in plasma samples indicate that infants diagnosed with BPD have suppressed expression of the miR-17∼92 cluster and coincident increases in DNMT-1, -3a, and -3b (13). The present investigation demonstrates a correlation in the degree of early miR-17∼92 cluster suppression in patients with the severity of the BPD diagnosed at 36 wk. Although unable to differentiate between mild and moderate disease, these data support the necessity of future prospective studies into the use of the miR-17∼92 cluster as a potential early marker of severe disease, to identify patients early to target therapies without having to await 36 wk corrected for the clinical diagnosis. The present studies utilized lung samples generated using a newborn murine model of lung disease that combines perinatal inflammation and neonatal hyperoxic exposure. The major lung findings in this double-exposure model are that 1) expression of the miR-17∼92 is substantially decreased; 2) methylation of the miR-17∼92 cluster promoter is significantly increased; and 3) expression of DNMT-1, -3a and -3b is significantly increased.
The individual members of the miR-17∼92 cluster have been shown to regulate many matrix remodeling proteins, as well as TGFβ and SMAD2/3. Similarly, changes in matrix composition can give rise to altered alveolarization and pulmonary function as observed in infants with severe BPD and in our murine model (4, 9, 20, 23). While methylation is certainly involved with suppression of miR-17∼92 expression, others studies have recently identified histone deacetylase-3 (Hdac3) as a repressive regulator of miR-17∼92 and, therefore, an upstream regulator of TGFβ expression. Hdac3 also regulates normal type 1 epithelial cell differentiation and spreading; abnormal Hdac3 levels in addition to miR-17∼92 promoter methylation may be involved in the decreased alveolarization observed in our model and the interaction between these epigenetic regulating mechanisms is a potential future avenue of investigation (22).
Thus our data are the first to describe circulating plasma levels of miR-17 as a predictor of disease severity as early as 5 days of life and prior to onset of diagnostic disease criteria. Furthermore, our data are the first to demonstrate the presence of altered expression of the miR-17∼92 cluster and DNMT expression in experimental BPD. Our data are consistent with a model in which alterations in miR-17∼92 cluster expression are mediated by enhanced promoter methylation via the actions of DNMTs.
GRANTS
This work was supported by National Heart, Lung, and Blood Institute Grant R01HL119280 (T. E. Tipple); Office of Dietary Supplements Grant R01AT006880 (L. K. Rogers); National Institute of Child Health and Human Development Grant 5T32HD043003-08 (M. E. Robbins); and National Center for Advancing Translational Sciences Grant UL1TR001070 (T. E. Tipple).
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the author(s).
AUTHOR CONTRIBUTIONS
M.E.R., D.D., C.B.M., L.K.R., and T.E.T. conception and design of research; M.E.R., D.D., L.K.R., and T.E.T. performed experiments; M.E.R., D.D., L.K.R., and T.E.T. analyzed data; M.E.R., D.D., C.B.M., L.K.R., and T.E.T. interpreted results of experiments; M.E.R., L.K.R., and T.E.T. prepared figures; M.E.R., L.K.R., and T.E.T. drafted manuscript; M.E.R., L.K.R., and T.E.T. edited and revised manuscript; M.E.R., D.D., C.B.M., L.K.R., and T.E.T. approved final version of manuscript.
REFERENCES
- 1.Ali M, Heyob KM, Velten M, Tipple TE, Rogers LK. DHA suppresses chronic apoptosis in the lung caused by perinatal inflammation. Am J Physiol Lung Cell Mol Physiol 309: L441–L448, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bonauer A, Dimmeler S. The microRNA-17-92 cluster: still a miracle? Cell Cycle 8: 3866–3873, 2009. [DOI] [PubMed] [Google Scholar]
- 3.Dakhlallah D, Batte K, Wang Y, Cantemir-Stone CZ, Yan P, Nuovo G, Mikhail A, Hitchcock CL, Wright VP, Nana-Sinkam SP, Piper MG, Marsh CB. Epigenetic regulation of miR-17∼92 contributes to the pathogenesis of pulmonary fibrosis. Am J Respir Crit Care Med 187: 397–405, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fawke J, Lum S, Kirkby J, Hennessy E, Marlow N, Rowell V, Thomas S, Stocks J. Lung function and respiratory symptoms at 11 years in children born extremely preterm: the EPICure study. Am J Respir Crit Care Med 182: 237–245, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jin B, Robertson KD. DNA methyltransferases, DNA damage repair, and cancer. Adv Exp Med Biol 754: 3–29, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jobe AH, Bancalari E. Bronchopulmonary dysplasia. Am J Respir Crit Care Med 163: 1723–1729, 2001. [DOI] [PubMed] [Google Scholar]
- 7.Lu Y, Okubo T, Rawlins E, Hogan BL. Epithelial progenitor cells of the embryonic lung and the role of microRNAs in their proliferation. Proc Am Thorac Soc 5: 300–304, 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.McLeod A, Ross P, Mitchell S, Tay D, Hunter L, Hall A, Paton J, Mutch L. Respiratory health in a total very low birthweight cohort and their classroom controls. Arch Dis Child 74: 188–194, 1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Morty RE, Konigshoff M, Eickelberg O. Transforming growth factor-beta signaling across ages: from distorted lung development to chronic obstructive pulmonary disease. Proc Am Thorac Soc 6: 607–613, 2009. [DOI] [PubMed] [Google Scholar]
- 10.Pospisil V, Vargova K, Kokavec J, Rybarova J, Savvulidi F, Jonasova A, Necas E, Zavadil J, Laslo P, Stopka T. Epigenetic silencing of the oncogenic miR-17-92 cluster during PU.1-directed macrophage differentiation. EMBO J 30: 4450–4464, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Robertson KD, Uzvolgyi E, Liang G, Talmadge C, Sumegi J, Gonzales FA, Jones PA. The human DNA methyltransferases (DNMTs) 1, 3a and 3b: coordinate mRNA expression in normal tissues and overexpression in tumors. Nucleic Acids Res 27: 2291–2298, 1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rogers LK, Graf AE, Bhatia A, Leonhart KL, Oza-Frank R. Associations between maternal and infant morbidities and sRAGE within the first week of life in extremely preterm infants. PLoS One 8: e82537, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rogers LK, Robbins M, Dakhlallah D, Yang Z, Lee LJ, Mikhail M, Nuovo G, Pryhuber GS, McGwin G, Marsh CB, Tipple TE. Attenuation of miR-17-92 cluster in bronchopulmonary dysplasia. Ann Am Thorac Soc 12: 1506–1513, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rogers LK, Young CM, Pennell ML, Tipple TE, Leonhart KL, Welty SE. Plasma lipid metabolites are associated with gestational age but not bronchopulmonary dysplasia. Acta Paediatr 101: e321–e326, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shi W, Bellusci S, Warburton D. Lung development and adult lung diseases. Chest 132: 651–656, 2007. [DOI] [PubMed] [Google Scholar]
- 16.Thebaud B, Abman SH. Bronchopulmonary dysplasia: where have all the vessels gone? Roles of angiogenic growth factors in chronic lung disease. Am J Respir Crit Care Med 175: 978–985, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Thebaud B, Ladha F, Michelakis ED, Sawicka M, Thurston G, Eaton F, Hashimoto K, Harry G, Haromy A, Korbutt G, Archer SL. Vascular endothelial growth factor gene therapy increases survival, promotes lung angiogenesis, and prevents alveolar damage in hyperoxia-induced lung injury: evidence that angiogenesis participates in alveolarization. Circulation 112: 2477–2486, 2005. [DOI] [PubMed] [Google Scholar]
- 18.Van Marter LJ. Epidemiology of bronchopulmonary dysplasia. Semin Fetal Neonatal Med 14: 358–366, 2009. [DOI] [PubMed] [Google Scholar]
- 19.Velten M, Britt RD Jr, Heyob KM, Welty SE, Eiberger B, Tipple TE, Rogers LK. Prenatal inflammation exacerbates hyperoxia-induced functional and structural changes in adult mice. Am J Physiol Regul Integr Comp Physiol 303: R279–R290, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Velten M, Heyob KM, Rogers LK, Welty SE. Deficits in lung alveolarization and function after systemic maternal inflammation and neonatal hyperoxia exposure. J Appl Physiol 108: 1347–1356, 2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Velten M, Hutchinson KR, Gorr MW, Wold LE, Lucchesi PA, Rogers LK. Systemic maternal inflammation and neonatal hyperoxia induces remodeling and left ventricular dysfunction in mice. PLoS One 6: e24544, 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wang Y, Frank DB, Morley MP, Zhou S, Wang X, Lu MM, Lazar MA, Morrisey EE. HDAC3-dependent epigenetic pathway controls lung alveolar epithelial cell remodeling and spreading via miR-17-92 and TGF-beta signaling regulation. Dev Cell 36: 303–315, 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wong PM, Lees AN, Louw J, Lee FY, French N, Gain K, Murray CP, Wilson A, Chambers DC. Emphysema in young adult survivors of moderate-to-severe bronchopulmonary dysplasia. Eur Respir J 32: 321–328, 2008. [DOI] [PubMed] [Google Scholar]
- 24.Wu Y, Kwak KJ, Agarwal K, Marras A, Wang C, Mao Y, Huang X, Ma J, Yu B, Lee R, Vachani A, Marcucci G, Byrd JC, Muthusamy N, Otterson G, Huang K, Castro CE, Paulaitis M, Nana-Sinkam SP, Lee LJ. Detection of extracellular RNAs in cancer and viral infection via tethered cationic lipoplex nanoparticles containing molecular beacons. Anal Chem 85: 11265–11274, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]