Abstract
Neurexins are cell adhesion molecules involved in synaptic formation and synaptic transmission. Mutations in neurexin genes are linked to autism spectrum disorders (ASDs), which are frequently associated with sleep problems. However, the role of neurexin-mediated synaptic transmission in sleep regulation is unclear. Here, we show that lack of the Drosophila α-neurexin homolog significantly reduces the quantity and quality of nighttime sleep and impairs sleep homeostasis. We report that neurexin expression in Drosophila mushroom body (MB) αβ neurons is essential for nighttime sleep. We demonstrate that reduced nighttime sleep in neurexin mutants is due to impaired αβ neuronal output, and show that neurexin functionally couples calcium channels (Cac) to regulate synaptic transmission. Finally, we determine that αβ surface (αβs) neurons release both acetylcholine and short neuropeptide F (sNPF), whereas αβ core (αβc) neurons release sNPF to promote nighttime sleep. Our findings reveal that neurexin regulates nighttime sleep by mediating the synaptic transmission of αβ neurons. This study elucidates the role of synaptic transmission in sleep regulation, and might offer insights into the mechanism of sleep disturbances in patients with autism disorders.
Sleep is an essential and evolutionarily conserved behavior from worm to human1,2. Sleep disturbances are frequently observed in children with autism spectrum disorders (ASDs), a group of neurodevelopmental disorders3. Numerous studies have indicated that sleep disturbances occur in approximately 40–70% of children with ASDs4, and primarily include delayed sleep onset, frequent waking during the night, early morning waking, and decreased total sleep time. Sleep traits are largely affected by genetic factors, for example, specific genetic polymorphisms5,6. The identification of numerous mutations that affect sleep quantity and quality has been crucial to further our understanding of the regulation and functions of sleep1,7.
Many genetic factors and mutations that contribute to the development of ASDs have been identified. Neurexins and neuroligins are cell adhesion molecules that function in synapse formation8,9,10 and synaptic transmission11,12. Mutations in neurexin and neuroligin genes have been linked to autism13,14. Previous studies demonstrated that neuroligin 1 knockout mice and neuroligin 4 mutant flies exhibit defective sleep behaviors15,16. A recent study showed that neurexin mutant flies exhibit fragmented sleep, whereas overexpression of neurexin can consolidate nighttime sleep, which might be due to synaptic growth. However, the potential roles and underlying mechanisms of neurexin-mediated synaptic transmission in sleep regulation are largely unknown, and the molecular and neuroanatomical underpinnings of neurexin-regulated sleep are not clear.
Drosophila exhibits sleep-like behavior and is a useful model system for genetic studies17,18. Therefore, Drosophila has been used to dissect the molecular regulation and mechanism of sleep19,20,21,22. A number of genes20,23, circuits21,22, and biological processes24 that affect sleep have been identified. Drosophila sleep is governed by both circadian and homeostatic regulation19,25, similarly as in mammals. The large ventral lateral neurons (l-LNvs) mediate light-driven arousal through the release of pigment dispersing factor (PDF), whereas GABAergic inputs to l-LNvs promote sleep. The mushroom bodies (MBs), which contain 7 types of Kenyon cells (KCs), 21 types of output neurons, and 20 types of dopaminergic neurons, also are involved in sleep regulation21,22,26. Previous studies showed that blocking synaptic output from KCs can either increase or decrease sleep depending on the GAL4 driver used21,22. Convincingly, activation of different types of MB output neurons can either repress or promote sleep26,27. These results suggest that different KC populations play diverse roles in sleep regulation.
Neurexins are encoded by three genes in mammals, each of which has two promoters to generate α- and β-Neurexins28. In Drosophila, only a single α-neurexin gene (CG7050) was identified9,10. Here, we show that the Drosophila homolog of α-neurexin is essential for nighttime sleep. We demonstrate that neurexin controls sleep quantity and sleep homeostasis by mediating the synaptic transmission of αβ neurons. We further reveal that neurexin functionally couples Cac channels to regulate synaptic transmission. Our findings indicate that neurexin-mediated synaptic transmission is crucial for nighttime sleep regulation.
Results
Neurexin mutant flies exhibit reduced nighttime sleep and impaired sleep homeostasis
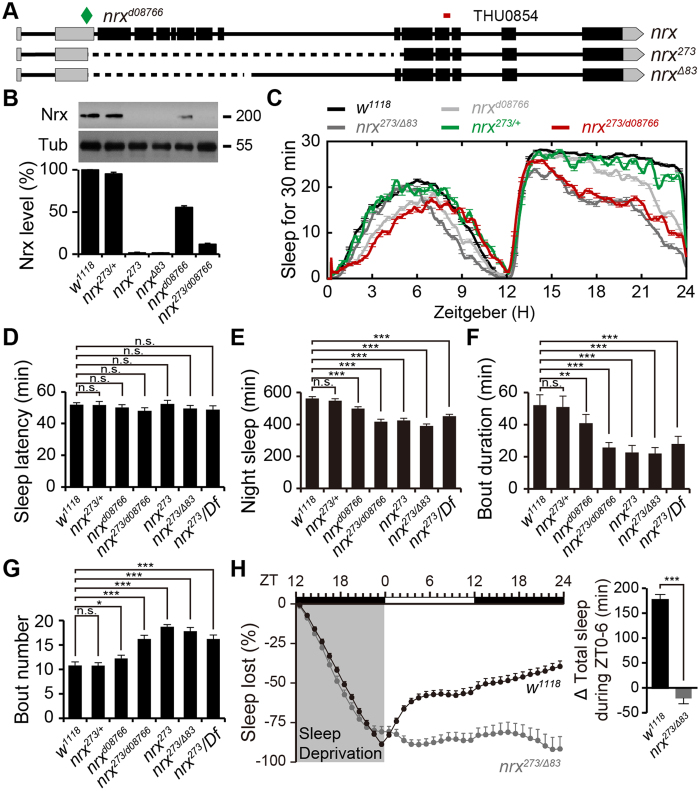
To examine the potential roles of neurexin in Drosophila sleep, we obtained two null mutant alleles, nrxΔ83 and nrx273, and a hypomorphic allele, P{XP}Nrxd08766, which contains a transposon inserted in the 5′ untranslated region of the neurexin gene (Fig. 1A). After six generations of out-crosses with the w1118 strain, all homozygous neurexin mutants were viable. Western blots confirmed the complete loss of neurexin in two neurexin null alleles (nrxΔ83 and nrx273), and significant reduction of neurexin in the hypomorphic allele (P{XP}Nrxd08766) (Fig. 1B). Next, we assessed the sleep behavior of these mutants during 12 h light/dark cycles. nrx273 mutant flies showed normal sleep onset compared with w1118 flies (49.6 ± 1.9 vs. 51.9 ± 1.2 min, Fig. 1C,D). However, compared with w1118 flies, nrx273 mutant flies exhibited a progressive reduction in nighttime sleep (416.3 ± 16.6 vs. 562.5 ± 10.9 min, Fig. 1E) accompanied by shortened episode length (25.7 ± 3.5 vs. 52.1 ± 6.9 min, Fig. 1F) and increased episode number (16.2 ± 0.8 vs. 10.8 ± 0.7, Fig. 1G). These data reflected the poorly consolidated nighttime sleep in neurexin mutants. To further eliminate potential genetic background effects, we generated nrxΔ83/273 trans-heterozygotes by combining two out-crossed neurexin null mutant alleles, nrxΔ83 and nrx273. Convincingly, similar sleep profiles were observed in nrxΔ83/273 flies and nrx273-deficient flies, in which the mutant allele was combined with Df(3R)BSC685 (lacking the entire neurexin gene) (Fig. 1C and E). These combined observations indicate that neurexin is essential for nighttime sleep.
Figure 1. neurexin mutant flies exhibit reduced nighttime sleep.
(A) Annotated transcription of the neurexin gene. The P element XPd08766 insertion site is marked with a diamond; the RNAi recognition site is labeled at the top. The deleted regions of neurexin mutant alleles (nrxΔ83 and nrx273) are indicated by dashed lines. (B) Western blot analysis showing neurexin protein levels in w1118 control and neurexin mutant flies. Tubulin was used as a loading control. Quantification of the relative protein level for each genotype is presented in the lower panel. (C) Sleep profiles of control w1118 (black, n = 308), nrxd08766 (light grey, n = 53), nrx273/+ (green, n = 32), nrx273/d08766 (red, n = 63), and nrxΔ83/273 (grey, n = 192) flies, which is plotted as a 30 min moving average. (D–G) Quantification of sleep onset latency after lights off, total nighttime sleep, average sleep episode length, and number of sleep episodes per night for each genotype. (H) After sleep deprivation overnight for 12 h, nrxΔ83/273 mutants regain a significantly lower percentage of their lost sleep than w1118 control flies (mean ± SEM, n = 32 flies per group). Quantification of total sleep time during the first 6 h recovery period is presented in the right panel.
To further test whether specific levels of neurexin are required for sleep regulation, we compared neurexin protein levels and sleep behavior in nrx273/+ heterozygotes, P{XP}nrxd08766 mutants, and p{XP}nrxd08766/273 combination flies. Although nrx273/+ heterozygotes showed a slight reduction in neurexin protein level compared with that of w1118 flies (95.1 ± 2.6%, Fig. 1B), they exhibited normal nighttime sleep (548.0 ± 13.4 vs. 562.5 ± 10.9 min, Fig. 1C and E). P{XP}nrxd08766 mutants express 53.4 ± 1.8% of the normal amount of neurexin protein expressed by w1118 flies (Fig. 1B), and exhibited reduced nighttime sleep compared with w1118 flies (499 ± 8.2 vs. 562.5 ± 10.9 min, Fig. 1C and 1E). The p{XP}nrxd08766/273 combination flies contained only 11.2 ± 1.9% of the normal amount of neurexin proteins compared with the levels in w1118 flies, and displayed more severe reductions in nighttime sleep (425 ± 14.3 vs. 562.5 ± 10.9 min, Fig. 1C and E). These combined observations suggest that neurexin mediates nighttime sleep in a dosage-dependent manner.
The timing and amount of sleep are governed by cooperation of circadian and homeostatic mechanisms. We first excluded the possibility that abnormal nighttime sleep in the neurexin mutant was due to an altered circadian clock, as nrxΔ83/273 mutants exhibit comparable behavioural periods with those of wild-type flies under constant dark conditions (23.75 ± 0.42 vs. 23.77 ± 0.33 h, Fig. S1). Next, we examined whether reduced sleep in neurexin mutants might be due to defective homeostatic regulation. We mechanically deprived flies of sleep for 12 h overnight, and then measured the amount of sleep that was regained during the following 24 h. The nrxΔ83/273 mutants recovered significantly less of the lost sleep than that of w1118 controls (Fig. 1H). These data demonstrate that neurexin mutants exhibit defective homeostatic sleep regulation, and suggest that the reduced nighttime sleep in neurexin mutants is likely due to alterations in the neural circuits or molecular pathways that govern homeostatic control of sleep.
Neurexin is expressed in mushroom body neurons and is crucial for nighttime sleep
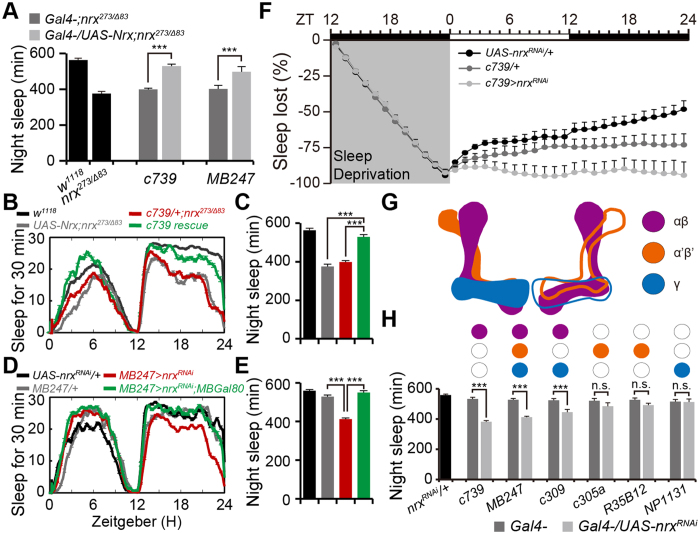
To investigate how neurexin mediates nighttime sleep, we generated p[UAS-Neurexin] transgenic flies and conducted rescue experiments to map the anatomical requirements for neurexin in nighttime sleep using the GAL4/UAS system. Endogenous neurexin is broadly expressed; therefore, nine GAL4 lines that cover neurexin expression patterns or are involved in sleep regulation were selected for the rescue experiments. Both of the c739-GAL4 and MB247-GAL4 lines, which label MB neurons29, largely recovered nighttime sleep in neurexin mutants (Fig. 2A–C). By contrast, other GAL4 lines that label extra-MB regions (e.g., c316-GAL4, pdf-GAL4, 104y-GAL4, and c57-GAL4) failed to restore impaired nighttime sleep in neurexin mutants (Fig. S2A). The glia-specific GAL4 lines (repo-GAL4 and gcm-GAL4) also failed to rescue the impaired sleep (Fig. S2A).
Figure 2. Neurexin expression in mushroom body neurons is essential for nighttime sleep.
(A) Total nighttime sleep in flies with rescued neurexin expression using anatomically restricted GAL4 drivers. All rescue experiments were performed in the nrx273/Δ83 background; flies carry one copy of the indicated drivers (n = 32). (B,C) Average sleep profiles for rescued and control flies, with curves plotted as 30 min moving averages (B); quantification of total nighttime sleep in rescued and control flies (C); n = 61. (D,E) Average sleep profiles for MB247-GAL4/UAS-nrxRNAi and control flies, plotted as a 30 min moving average (D), and quantification of total nighttime sleep for each genotype (E); n = 32. (F) Cumulative sleep lost during 12 h of sleep deprivation and regained during subsequent recovery for 24 h in c739-GAL4/+;UAS-nrxRNAi/+ flies and control flies (n = 16). (G) Model of the fly MB illustrating the different subsets of intrinsic Kenyon cells (KCs) within the lobes. Purple, αβ lobes; orange, α′β′ lobes; blue, γ lobes. (H) Total nighttime sleep in flies with depleted neurexin. Each GAL4 expression pattern is summarized at the top. A single copy of the driver was used for each GAL4 line (n = 32).
To further validate the anatomical requirements of neurexin for nighttime sleep, we attempted to recapture the sleep deficits in neurexin mutants using RNAi against neurexin in specific neuronal subpopulations. We found that MB247-GAL4, c739-GAL4, and c309-GAL4 significantly reduced nighttime sleep (Fig. 2D,E and Fig. S2B). The reduced sleep in MB247-GAL4 was eliminated by combination with MB-GAL80, which suppresses GAL4 activity in MBs (Fig. 2D,E). We failed to significantly repress nighttime sleep using other drivers with more restricted neuronal expression, including 104y-GAL4 in fan-shaped body neurons, c819-GAL4 and GMR15C02-GAL4 in ellipsoid body (EB) neurons, c929-GAL4 and pdf-GAL4 in large and small ventral lateral (LnV) neurons, and Ilp-GAL4 in pars intercerebralis (PI) neurons (Fig. S2B). We further investigated whether neurexin expression in MB neurons was required for sleep homeostatic regulation. We found that c739-GAL4-mediated ablation of neurexin in MB neurons resulted in impaired homeostatic response to sleep loss (Fig. 2F). These combined observations suggest that neurexin expression in MB neurons is essential for nighttime sleep and homeostatic regulation.
MBs contain Kenyon cells (KCs) and MB output neurons. KCs have been grouped into three classes, γ, α′/β′, and α/β, with each class projecting axons to the eponymous lobe (Fig. 2G)30. Fluorescent labeling studies of MB247-GAL4, c739-GAL4, and c309-GAL4 show that all three overlap on MB αβ neurons; therefore, our observations indicate that neurexin expressed in MB αβ neurons is essential for nighttime sleep. To further investigate whether neurexin expressed in MB γ and α′β′ neurons is required for nighttime sleep, we depleted neurexin from these neurons using the GAL4 lines that label γ (NP1131-GAL4) and α′β′ (c305a-GAL4 and GMR35B12-GAL4) neurons (Fig. 2H). However, none of these lines significantly repressed nighttime sleep (Fig. 2H). These observations strongly suggest that neurexin expressed in MB αβ neurons is essential for nighttime sleep.
Neurexin expressed in MB αβcs neurons promotes nighttime sleep
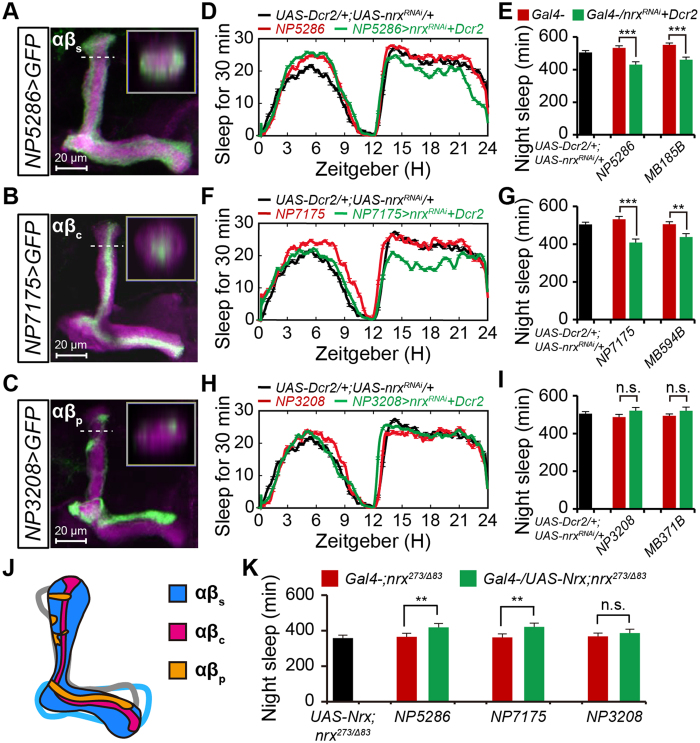
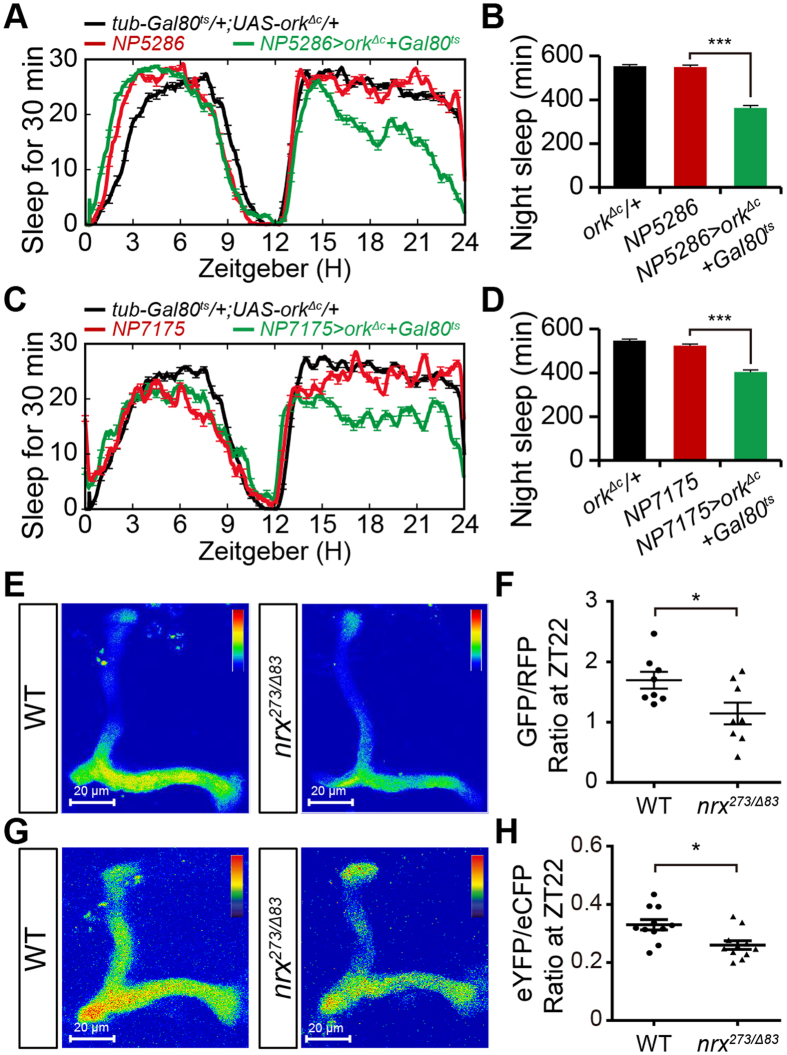
MB αβ neurons have been classified into three types according to the parallel axon fibers occupying different layers within the αβ lobes, αβc, αβs, and αβp26. To further explore the potential roles of neurexin expressed in each αβ neuron type for nighttime sleep, we obtained three GAL4 lines that label different subsets of αβ neurons. Previous reports showed that NP7175-GAL4, NP5286-GAL4, and NP3208-GAL4 were expressed in αβc, αβs, and αβp neurons, respectively29,31. Our confocal projection images confirmed the restricted expression of these lines in each αβ neuron type (Fig. 3A–C). To enhance the transgenic RNAi effects, we overexpressed a component of the RNAi machinery, Dicer232. We depleted neurexin from individual αβ-subset neurons, and then assessed nighttime sleep in these flies. Both NP5286-GAL4 and NP7175-GAL4 resulted in reduced nighttime sleep (Fig. 3D–G). By contrast, NP3208-GAL4 did not significantly affect nighttime sleep (Fig. 3H,I). To further confirm these observations, we obtained a set of validated split-GAL4 lines specific for each αβ subset26 and depleted neurexin using these GAL4 lines. Convincingly, both αβs-specific MB185B-GAL4 and αβc-specific MB594B-GAL4 dramatically reduced nighttime sleep (Fig. 3E and G), whereas αβp-specific MB371B-GAL4 flies exhibited normal nighttime sleep (Fig. 3I).
Figure 3. Neurexin functions in αβsc neurons to promote nighttime sleep.
(A–C) Projection views of confocal stacks at the level of the right MB lobes from NP5286-αβs (A), NP7175-αβc (B), and NP3208-αβp (C) flies driving mCD8::GFP (green). In all panels, the αβ lobes are labeled with anti-FasII (magenta). The inset shows a horizontal cross-section through the vertical collateral at the level of the dashed line in each panel. Scale bar = 20 μm. (D,E) Average sleep profiles for UAS-Dicer2/NP5286-GAL4;UAS-nrxRNAi/+ (n = 53) and control (n = 32) flies, plotted as a 30 min moving average (D), and quantification of total nighttime sleep for each genotype (E). Note that Dicer-2 was used to enhance the transgenic RNAi effect in panels D, F, and H. (F,G) Average sleep profiles for NP7175-GAL4/Y;UAS-Dicer2/+;UAS-nrxRNAi/+ (n = 37) and control (n = 32) flies, plotted as a 30 min moving average (F), and quantification of total nighttime sleep for each genotype (G). (H,I) Average sleep profiles for NP3208-GAL4/Y;UAS-Dicer2/+;UAS-nrxRNAi/+ (n = 42) and control (n = 32) flies, plotted as a 30 min moving average (H), and quantification of total nighttime sleep for each genotype (I). (J) Model of the MB αβ neurons illustrating three subsets of neurons within the αβ lobes. Purple, αβc neurons; blue, αβs neurons; yellow, αβp neurons. (K) Total nighttime sleep in flies with rescued neurexin expression using specific GAL4 drivers for each αβ subset. Rescue experiments were conducted in the nrx273/Δ83 background; flies bear one copy of the indicated drivers (n = 16).
To further validate the role of neurexin in nighttime sleep, we performed rescue experiments by expressing neurexin in individual αβ-subset neurons. Convincingly, expression of neurexin with either NP5286-GAL4 or NP7175-GAL4 largely increased nighttime sleep, whereas expression of neurexin with NP3208-GAL4 had no clear rescue effects on nighttime sleep (Fig. 3J,K). Taken together, these data demonstrate that neurexin functions in αβsc neurons to promote nighttime sleep.
Knockdown of neurexin expression in adult flies reduces nighttime sleep
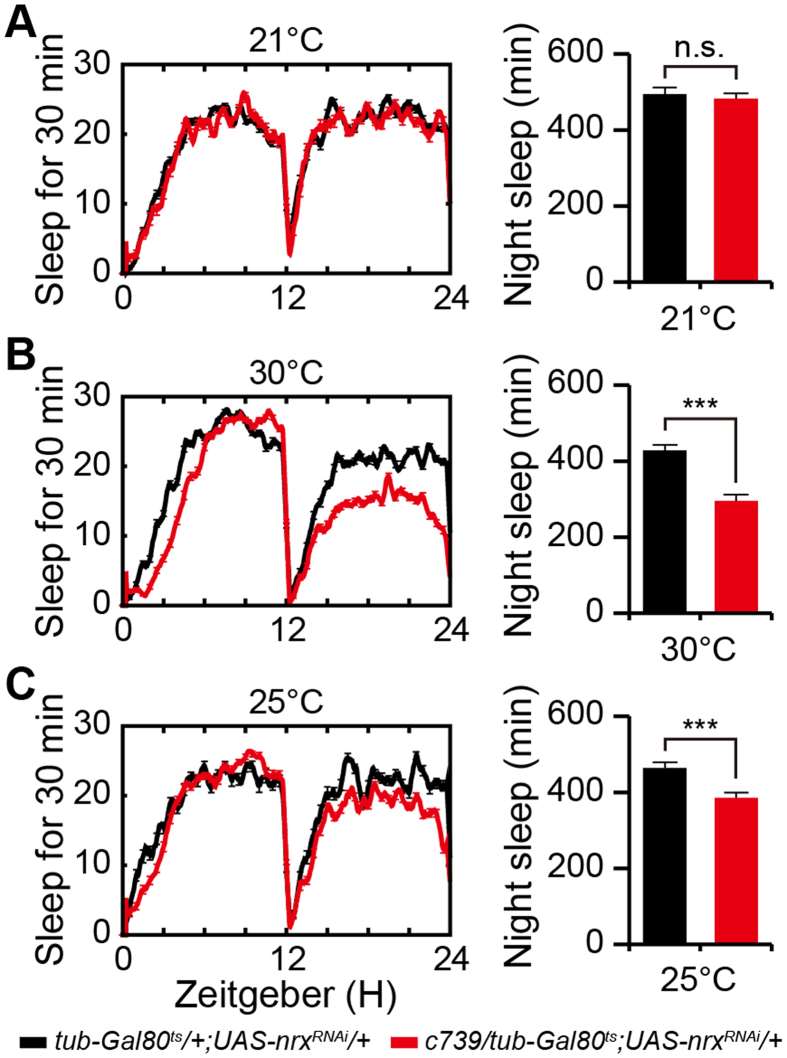
The abnormal sleep observed in neurexin mutants might be due to developmental defects or impaired synaptic function in adult flies. To distinguish these possibilities, we generated tubulin-GAL80ts/c739-GAL4;UAS-nrxRNAi/+ flies and analyzed their sleep behavior. In these flies, RNAi was suppressed during development by the ubiquitous expression of temperature-sensitive GAL80ts, but was selectively induced at adulthood by exposure to 30 °C. Flies were reared at 21 °C during development. Adult flies were entrained for 2 days in light/dark at 21 °C, treated with 30 °C for 3 days to inactivate temperature-sensitive GAL80ts, and then returned to 25 °C. Compared with the control flies, tubulin-GAL80ts/c739-GAL4;UAS-nrxRNAi/+ flies exhibited a reduction in nighttime sleep after switching to 30 °C (Fig. 4A,B). Two days after the return to 25 °C, tubulin-GAL80ts/c739-GAL4;UAS-nrxRNAi/+ flies still exhibited reduced nighttime sleep (Fig. 4C). These data indicate that knockdown of neurexin expression in adult flies is sufficient to reduce nighttime sleep.
Figure 4. Knockdown of neurexin expression in adult flies reduces nighttime sleep.
(A–C) Sleep profiles for c739-GAL4/tub-GAL80ts;UAS-nrxRNAi/+ (black) and c739-GAL4/tub-GAL80ts control (red) flies at 21 °C (A), 30 °C (B), and 25 °C (C). Quantification of total nighttime sleep at each temperature is presented in the right panel (n = 16).
Defective nighttime sleep in neurexin mutants is due to impaired αβ neuron synaptic transmission
Previous studies showed that blocking the synaptic output of αβ neurons reduced nighttime sleep, whereas hyperpolarization of these neurons has no clear promotion effects on nighttime sleep22,27. To further clarify the role of each αβ-subset neuron in nighttime sleep, we silenced each subset by expressing a mutant form of the open rectifier potassium channel (dORKΔC)33. To exclude the potential roles of dORKΔC in neural development, its expression was suppressed during development by the ubiquitous expression of temperature-sensitive GAL80ts, and then selectively induced in adult flies by exposure to 30 °C34. Compared with control flies, ectopic expression of dORKΔC under the control of either NP5286-GAL4 or NP7175-GAL4 significantly reduced sleep (Fig. 5A–D), resembling the defective sleep patterns observed in flies with neurexin selectively depleted from the corresponding αβ-subset neurons. We also expressed the temperature-sensitive cation channel TrpA1 to increase neural excitability and subsequently reinforce αβ neuron synaptic transmission35. Consistent with a previous report36, activation of TrpA1 by exposure to elevated temperature did not significantly affect nighttime sleep in each αβ-subset neuron (Fig. S3).
Figure 5. Blocking synaptic output from αβsc neurons reduces nighttime sleep.
(A,B) Average sleep profiles for NP5286/tub-GAL80ts;UAS-Ork1ΔC/+ and control flies, plotted as a 30 min moving average (A), and quantification of total nighttime sleep in NP5286/tub-GAL80ts;UAS-Ork1ΔC/+ and control flies (B) (n = 16). For (A) and (C), all flies were reared at 18 °C throughout development, and adults were entrained at 30 °C for 6 hours every day for 2 days. Then, the flies’ sleep was measured. (C,D) Average sleep profiles for NP7175/Y;tub-GAL80ts/+;UAS-Ork1ΔC/+ and control flies, plotted as a 30 min moving average (C), and quantification of total nighttime sleep in NP7175/Y;tub-GAL80ts/+;UAS-Ork1ΔC/+ and control flies (D) (n = 24). (E) Representative images of GCaMP6 fluorescence in both w1118 control and nrx273/Δ83 mutant αβ lobes. Images show the sum of fluorescence intensity from multiple layers primarily consisting of αβ lobes at ZT22. (F) Quantification of GCaMP6/RFP fluorescence ratios in w1118 control and nrx273/Δ83 mutant αβ neurons at ZT22. RFP fluorescence was used as control (n = 8). (G) Averaged eYFP/eCFP fluorescence ratio in both w1118 control and nrx273/Δ83 mutant αβ lobes at ZT22. (H) Quantification of eYFP/eCFP ratios in w1118 control and nrx273/Δ83 mutant αβ neurons at ZT22 (n = 11).
These observations suggest that defective nighttime sleep in neurexin mutant flies might be due to reduced synaptic transmission of αβ neurons. To compare neural activities, we expressed GCaMP6.0 in αβ neurons and subsequently performed Ca2+ imaging analysis. The αβ neurons were visualized by membrane expression of an RFP-tagged mCD8. Compared with wild-type αβ neurons, neurexin mutant αβ neurons showed reduced fluorescence intensity of GCaMP6.0 at ZT22 (Fig. 5E,F). To further validate these observations, we applied the calcium sensor Cameleon 2.1 (Cam2.1), which enables ratiometric measurements of calcium-induced fluorescence resonance transfer (FRET) between enhanced cyan fluorescent protein (eCFP) and enhanced yellow fluorescent protein (eYFP)37. Convincingly, compared with wild-type αβ neurons, neurexin mutant αβ neurons showed increased eCFP signals and reduced eYFP signals at ZT22 (Fig. 5G,H). These data demonstrate that neural activity and subsequent synaptic transmission are impaired in neurexin mutant αβ neurons.
Neurexin regulates synaptic transmission through functional coupling of Cac channels
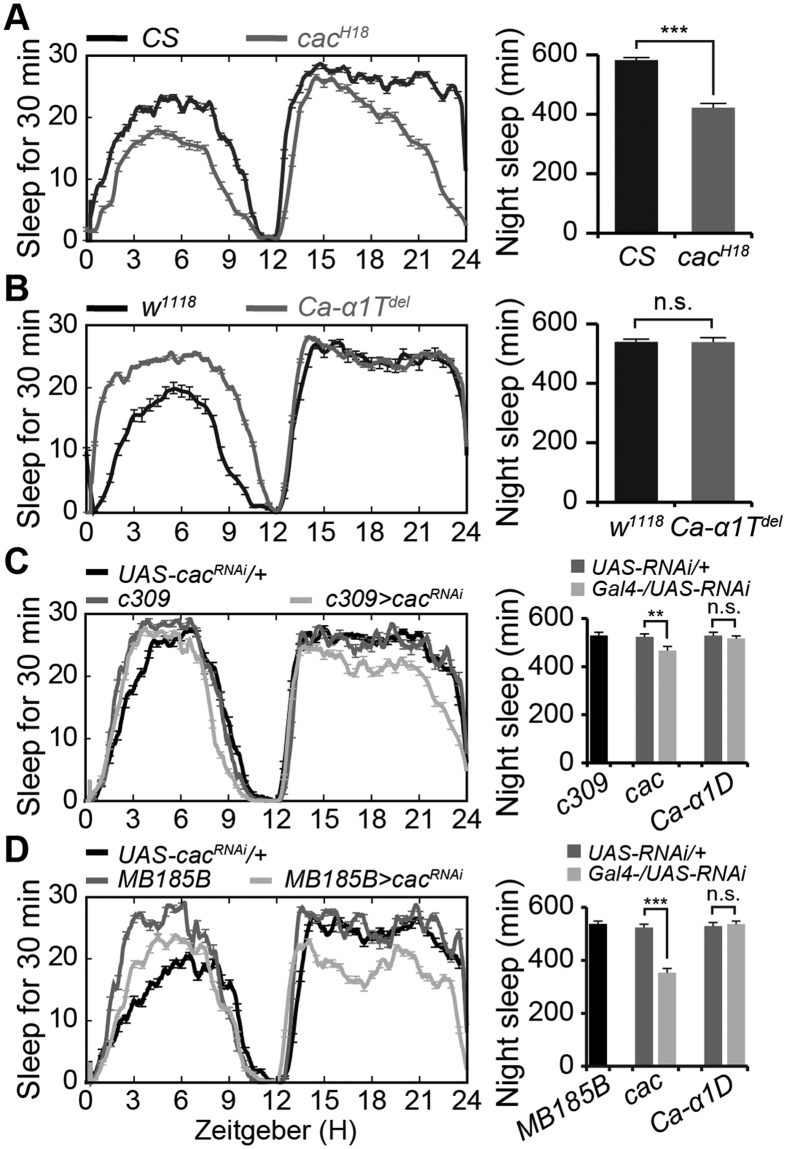
Previous studies suggest that synaptic transmission may depend on Ca2+ entry via voltage-gated Ca2+ channels (VGCCs)38. We attempted to identify which Ca2+ channel was regulated by neurexin in αβ neurons. The Drosophila genome contains only three putative homologs of vertebrate voltage-gated Ca2+ channels (Ca-α1D, Cac, and Ca-α1T)39. Cacophony channel mutant (cacH18) flies exhibited reduced nighttime sleep compared with that of control flies (422.1 ± 15.3 vs. 582.3 ± 9.4, Fig. 6A), similar to the defective nighttime sleep pattern observed in neurexin mutants. Although Ca-α1Tdel mutant flies exhibited increased daytime sleep compared with that of control flies (458 ± 10.8 vs. 298 ± 9.7 min), they exhibited normal nighttime sleep (538.4 ± 7.9 vs. 549.7 ± 12.3 min, Fig. 6B), consistent with a previous report40. In addition, αβ neuron-specific RNAi against cac but not Ca-α1D mimicked the defective nighttime sleep in neurexin mutant flies (Fig. 6C), and αβs neuron-specific RNAi against cac, but not Ca-α1D, recaptured the reduced nighttime sleep (Fig. 6D).
Figure 6. Neurexin functionally couples with calcium channels to mediate synaptic transmission.
(A,B) Averaged total sleep per night in cacH18 mutants and Canton-S (CS) flies (A), and in Ca-α1Tdel mutants and w1118 control flies (B), plotted as a 30-min moving average (n = 64). Quantification of total nighttime sleep is presented in the right panel of each figure. Note that CS flies were used as the control flies for cacH18 mutants, which were generated in the CS background. (C) Averaged total sleep per night in c309-GAL4/+;UAS-cacRNAi/+ and control flies, plotted as a 30 min moving average (n = 32). Quantification of total nighttime sleep in each genotype is presented in the right panel. (D) Averaged total sleep per night in MB185B-GAL4/+;UAS-cacRNAi/+ and control flies, plotted as a 30 min moving average (n = 20). Quantification of total nighttime sleep in each genotype is presented in the right panel.
Defective synaptic transmission in neurexin mutants could be due to either abnormal Cac channel coupling or altered Cac channel distribution. To distinguish between these possibilities, we compared the levels and distribution of Cac channels in wild-type and mutant Kenyon neurons. Ca2+ channels are extremely sensitive to fixation; therefore, we utilized a GFP-tagged Cac transgene (Cac-GFP) and captured live images to visualize Cac channels in vivo10,41. Although reduced levels of active zone protein Brp were observed in the mutant α-lobes (Fig. S4A), Cac-GFP levels appeared normal in neurexin mutant α-lobes compared with those in controls (Fig. S4B), which was consistent with observations from neurexin mutant neuromuscular junctions10. Convincingly, N-type Ca2+ channels showed normal levels and affinities in several α-neurexin knockout mice11. Furthermore, overexpression of Cac channels failed to restore defective sleep in mutant MB neurons (Fig. S4C). Taken together, these results indicate that impaired synaptic transmission in neurexin mutants is most likely due to changes in Ca2+ channel coupling rather than Ca2+ channel distribution.
The αβs neurons release both acetylcholine and short neuropeptide F, whereas α β c neurons use sNPF to promote nighttime sleep
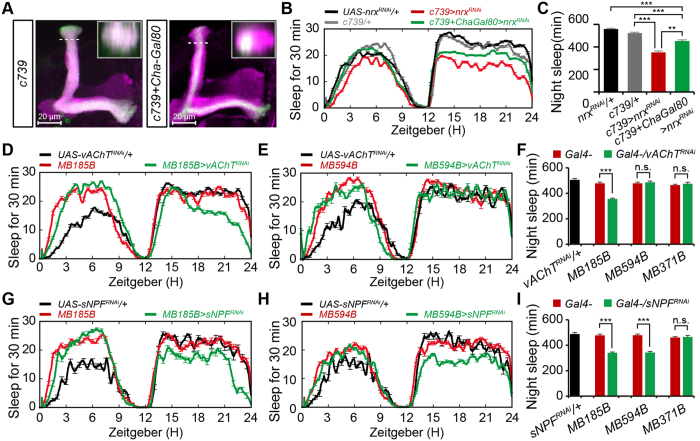
Finally, we investigated which neurotransmitter was released by each αβ-subset neuron to promote sleep. Down-regulation of acetylcholine (ACh) signaling in MBs can inhibit sleep42; therefore, we first investigated which type of αβ neuron was cholinergic. Cha-GAL80, which suppresses GAL4 activity in cholinergic neurons, successfully repressed the GAL4 activity of c739-GAL4 in αβsp but not αβc subset neurons (Fig. 7A). Compared with the sleep profile of c739-GAL4/+ ;UAS-nrxRNAi/+ flies, those that also expressed Cha-GAL80 exhibited increased nighttime sleep (452.6 ± 10.7 vs. 351.3 ± 13.1 min, Fig. 7B and C). These observations suggest that αβs neurons are cholinergic and their ACh signaling promotes nighttime sleep. However, the combination of Cha-GAL80 in c739-GAL4/+;UAS-nrxRNAi/+ flies did not fully restore the reduced nighttime sleep (Fig. 7B and C). These observations suggest that some non-cholinergic c739 neurons may promote nighttime sleep, or some cholinergic neurons may be mislabeled by Cha-GAL80.
Figure 7. The αβs neurons release both acetylcholine and short neuropeptide F to promote nighttime sleep, whereas α β c neurons release sNPF to maintain nighttime sleep.
(A) Expression of c739 in αβsp neurons is blocked by Cha-GAL80. The inset shows a horizontal cross-section through the vertical collateral at the level of the dashed line in each panel. Scale bar = 20 μm. (B,C) Averaged total sleep per night in c739-GAL4/+;UAS-nrxRNAi/+ and c739-GAL4i/+;Cha-GAL80/UAS-nrxRNAi flies, plotted as a 30 min moving average (B), and quantification of total nighttime sleep in each genotype (C) (n = 64). (D,E) Averaged total sleep per night in MB185B-GAL4/UAS-vAChTRNAi and control flies (D) (n = 64), and in MB594B-GAL4/UAS-vAChTRNAi and control flies (E) (n = 24), plotted as a 30 min moving average. (F) Quantification of total nighttime sleep in vAChT-RNAi flies and control flies. (G,H) Averaged total sleep per night in MB185B-GAL4/UAS-sNPFRNAi and control flies (G) (n = 32), and in MB594B-GAL4/UAS-sNPFRNAi and control flies (H) (n = 32), plotted as a 30 min moving average. (I) Quantification of total nighttime sleep in sNPFRNAi flies and control flies.
To further examine whether ACh signaling from αβs neurons promotes nighttime sleep, we depleted vesicular acetylcholine transporter (vAChT) required for acetylcholine transport and release. Depletion of vAChT in αβs neurons, but not αβc and αβp neurons, resulted in reduced nighttime sleep (Fig. 7D–F), similar to the defective sleep observed in neurexin mutant flies. By contrast, down-regulation of other neurotransmitters in αβs neurons, including glutamate and gamma-aminobutyric acid, did not significantly affect nighttime sleep (Fig. S5A). Taken together, these results demonstrate that αβs neurons release ACh to promote nighttime sleep.
Neuropeptides are important intercellular signaling molecules in animals, and some genes that encode neuropeptides and their receptors are involved in sleep in Drosophila43,44. Given that down-regulation of all identified neurotransmitters in αβc neurons still resulted in normal nighttime sleep, we next examined whether αβc neurons use neuropeptides to maintain sleep by investigating the sleep-promoting roles of four potential neuropeptides known to be expressed in MBs, including FMRFamides (FMRFa), sulfakinins (XSK), short neuropeptide F (sNPF), and tachykinin-related peptide (TKRP). The results showed that αβs neuron-specific depletion of sNPF but not the other neuropeptides caused reduced nighttime sleep (Fig. 7G, and Fig. S5B). In addition, depletion of sNPF in αβc neurons also reduced nighttime sleep (Fig. 7H and I, Fig. S5C), whereas down-regulation of sNPF in αβp neurons resulted in normal nighttime sleep (Fig. 7I). These observations indicate that αβc neurons use sNPF to promote nighttime sleep.
Taken together, our results demonstrate that neurexin promotes nighttime sleep by regulating synaptic transmission. Using neurexin mutants, we revealed that αβs neurons release both ACh and sNPF, whereas αβc neurons release sNPF to promote nighttime sleep.
Discussion
Sleep is correlated with neural activity, and numerous mutations that alter neuronal excitability and the balance of inhibitory over excitatory neurotransmission have been shown to significantly affect sleep quantity and quality. In the present study, we showed that neurexin mutant flies exhibited reduced and fragmented nighttime sleep. A recent study reported that combinations of hypomorphic neurexin alleles led to fragmented sleep, but the total time of night sleep did not differ among these flies45. Given that combinations of hypomorphic neurexin alleles lead to a partial reduction in neurexin mRNA levels45, it is reasonable that neurexin null mutants exhibit more severe sleep deficits than combinations of hypomorphic alleles. Convincingly, we showed that flies with the hypomorphic neurexin allele P{XP}nrxd08766, which produce 53% of the neurexin level of wild-type flies, exhibited reduced nighttime sleep. We also showed that neurexin mutant flies exhibited impaired sleep homeostasis, and identified the anatomical requirements and mechanism of neurexin in nighttime sleep regulation.
The Drosophila genome contains only a single α-neurexin gene, and immunostaining analysis reveals its ubiquitous expression in nervous system9,10. Our RNAi knockdown and rescue experiments showed that α-neurexin expression in MBs is essential for nighttime sleep, consistent with previous studies showing that MB neurons are involved in sleep regulation21,22. Our anatomically restricted manipulations of neurexin further revealed that neurexin expressed in αβsc neurons, but not αβp neurons, controls nighttime sleep. Given that expression of neurexin in MB neurons does not fully restore defective nighttime sleep in neurexin null mutant flies, we suspected that neurexin expressed in other neurons also may function in sleep regulation. We excluded the possibility that neurexin regulated nighttime sleep through several identified sleep-promoting neurons, including fan-shaped body neurons, EB neurons, larger LNv neurons, and PI neurons46,47. Elucidating the roles of neurexin in other potential sleep-promoting neurons will require additional study.
Neurexins are cell adhesion molecules that function in various neural circuits through mediating synapse formation and synaptic transmission8,48. A recent report suggested that defective sleep in neurexin mutants is not solely due to developmental effects because it can be induced acutely in adults and there is evidence that it functions in synaptic growth45. Our study showed that knockdown of neurexin in adult flies reduced nighttime sleep, and neurexin regulated nighttime sleep by modulating synaptic transmission. Our Ca2+ imaging analyses revealed that neurexin mutant αβ neurons had reduced Ca2+ levels compared with those of wild-type neurons, consistent with electrophysiological recordings showing impaired synaptic transmission in neurexin mutant neuromuscular junctions10. Neurotransmitter release is mediated by the fusion of synaptic vesicles triggered by Ca2+, and α-neurexin functionally couples Ca2+ channels to the presynaptic machinery to mediate synaptic vesicle exocytosis11. In this study, we show that Cac channels are expressed in αβ neurons and mediate synaptic transmission. Therefore, impaired synaptic transmission is correlated with reduced nighttime sleep in neurexin mutant flies. This conclusion is consistent with previous reports, which show that sleep behavior is closely correlated with synaptic function and transmission15,16.
As in mammals, Drosophila sleep is coordinated by the interaction between sleep-promoting and wake-promoting neurons. Previous studies showed that blocking synaptic output from KCs can either increase or decrease sleep depending on the GAL4 driver used21,22,36. In the present study, we used anatomically restricted GAL4 lines to demonstrate that blocking synaptic output from αβsc neurons resulted in reduced nighttime sleep, whereas silencing αβp neurons did not significantly affect nighttime sleep. Consistent with previous observations22,36,43, activation of αβ neurons resulted in normal nighttime sleep. Furthermore, we showed that down-regulation of ACh signaling from αβs neurons inhibited nighttime sleep, whereas depletion of sNPF from αβsc neurons decreased nighttime sleep. The αβsp neurons have been identified as putative cholinergic neurons42,49, and down-regulation of ACh signaling inhibits nighttime sleep42. In addition, sNPF is highly expressed in MBs, and sNPF mutant flies display reduced sleep, indicating that sNPF plays an essential role to promote sleep in Drosophila50,51.
Our study finds that different αβ neuron populations release variant neurotransmitters and/or neuropeptides to promote nighttime sleep. The different sleep-promoting roles of αβc and αβs neurons might be attributable to the anatomy of neural circuits within the MB lobes. Each MB contains ~2,000 KCs that extend their axonal fibers to relevant lobes and synapse with a relatively small number of MB output neurons (MBONs)26,30,52. Thus, the convergence of a large number of KCs onto a small number of MBONs may integrate signals from both sleep-promoting and wake-promoting neural circuits. MB-M8 dopamine neurons arborize throughout the βs and βc regions and are positively reinforcing during odor stimulation, whereas aversive-reinforcing MB-M3 and MB-MP1 dopamine neurons preferentially arborize in the αβs layer and exhibit no or much weaker αβc innervation49. A recent report revealed that activation of different MBON populations either suppresses or promotes sleep26,27. However, the process of signaling integration in different αβ-subset neurons under physiological conditions requires further investigation.
Methods
Fly stocks and genetics
Flies were maintained on standard medium at 25 °C, 60–80% relative humidity, with a 12 h light/dark cycle unless otherwise stated. The wild-type flies used in this study were w1118, except for those in Fig. 6A that were Canton-S flies. The α-neurexin (CG7050) null mutant allele nrx273 was obtained from Dr. Manzoor A. Bhat’s lab10; it also carries a mutation in the DNA repair gene mutagen-sensitive 309 (mus309) located at 86E17. The α-neurexin null mutant allele, nrxΔ83 was generated by imprecise excision of the P-element using Δ2–3 as a transposase source according to standard procedures9. P{XP}nrxd08766 was obtained from the Bloomington Stock Center. To delete the extra mutants and standardize the background, all neurexin mutants were outcrossed for more than six generations with the w1118 strain. To further eliminate potential genetic background effects, two out-crossed neurexin null mutant allele nrxΔ83/273 were combined for use in this study. The full-length neurexin transgene p[UAS-Neurexin] was generated as previously described9,53. The mushroom body split-GAL4 lines MB185B, MB594B, and MB371B were kindly provided by Yoshi Aso. UAS-RNAi lines were ordered from the Tsinghua Fly Center and Vienna Drosophila RNAi Center. Other lines used in this work were obtained from the Bloomington Stock Center.
Sleep and circadian assays
Sleep assays were performed as described previously19. For sleep measurements, 3- to 5-day-old male flies raised in light/dark (LD)-entrained cultures were placed in 65 × 5 mm glass tubes containing 5% sucrose and 2% agar. Locomotor activity was measured for 5–7 days at 25 °C during LD cycles using the Drosophila Activity Monitoring System (DAM2, Trikinetics) in a DigiTherm CircKinetics incubator (Tritech Research). Data were collected in 1-min bins, and a sliding window was applied. Sleep was defined as 5 consecutive minutes of inactivity as described previously54. Data were analyzed with pySolo software. For analysis of circadian behavior, locomotor activity was measured using DAM2 monitors (Trikinetics), and activity counts were collected in 30 min bins in LD for 4 days, and then in DD during a 7 day period, and analyzed using ClockLab (Actimetrics). Power is a measure of rhythm amplitude and corresponds to the height of the periodogram peak above the significance line55.
For temperature-shift experiments, flies expressing UAS-dTrpA1 were raised at 21 °C throughout their development, and dTrpA1 was activated at 28 °C. Flies expressing Tubulin-GAL80ts were reared at 18 °C throughout their development, and adults were entrained at 30 °C for 6 hours every day for 2 days. Then, sleep was measured. To exclude the potential role of elevated temperature in sleep, sleep data collected at the elevated temperature were compared with those collected at the normal temperature.
Sleep deprivation experiments were performed as described previously56,57. Briefly, male flies were subjected to sleep deprivation using the mechanical Sleep Nullifying Apparatus (SNAP) method while housed in TriKinetics DAM2 systems. Cumulative difference plots were calculated for each individual fly by comparing the percentage of sleep lost during overnight sleep deprivation to that of the immediately preceding unperturbed night. Individual sleep rebound was quantified every hour for 24 h by dividing the cumulative amount of sleep regained by the total amount of sleep lost during deprivation. We found that 9 h of sleep deprivation was sufficient to cause a 70% loss of sleep in w1118 flies and more than 60 min of sleep loss in some mutant flies. To exclude the flies escaping from mechanical stimulations, individual flies were excluded from rebound analysis if 12 h of sleep deprivation was less than 70% effective in control flies or if mutant flies lost less than 60 min of sleep57. Statistical significance was assessed by two-way repeated-measures ANOVA.
Antibodies
The anti-neurexin antibody was generated as described previously9,53. Briefly, purified 6 × His fusion fragment (amino acids 1,534–1,690) of neurexin protein was used as antigen. An affinity column was created by coupling GST-neurexin fusion protein to CNBr-activated Sepharose 4B, and was used to purify the antibody. Other antibodies were obtained from Developmental Studies Hybridoma Bank (Tubulin, Fas II, and nc82).
Western blots
Fly heads were homogenized in SDS sample buffer. The proteins were fractionated by SDS-PAGE and transferred to polyvinylidene fluoride (PVDF) membranes (Pall) in Tris-glycine buffer. After blocking, the membranes were probed with anti-neurexin (1:200) or anti-tubulin (1:1,000) antibody at room temperature for 2 h. After three washes with phosphate-buffered saline containing Tween, the membranes were probed with either anti-rabbit or anti-mouse IgG-peroxidase conjugate (GE Healthcare) at room temperature for 2 h. The probe signals were detected using enhanced chemiluminescence reagents (Amersham Biosciences).
Immunostaining
After dissection and fixation, fly heads were stained with anti-GFP (1:100), anti-FasII (1:100, Developmental Studies Hybridoma Bank), and anti-Brp (1:100, nc82, Developmental Studies Hybridoma Bank) antibodies. Samples were imaged on an LSM 710 confocal microscope (Zeiss). For Cac-GFP live imaging, dissected adult heads were mounted on the slides and the images were acquired from live α lobes using an LSM 710 confocal microscope (Zeiss).
In vivo calcium imaging
In vivo GCaMP imaging experiments were performed on 3- to 7-day-old male adult flies. GCaMP6.0 and mCD8-RFP were expressed in αβ-subset neurons under c739-GAL4 control; mCD8-RFP was used as a control. Adult flies were entrained under a 12-h LD cycle, and the fly brains were rapidly dissected at ZT21 under a light intensity of 400 lux for <90 s. After resting in complete darkness for more than 30 min, GCaMP6.0 and mCD8-RFP signals from the αβ lobe were imaged at ZT22. The GFP/RFP ratio is presented.
In vivo Cam2.1 imaging experiments were performed as described previously58. Briefly, fluorescence signals were monitored on an LSM 710 confocal microscope (Zeiss). Each experiment was performed with more than 8 flies, and averaged responses were presented. Data acquisition and evaluation were performed as described previously58.
Statistical analysis
Data are presented as mean ± SEM. Two-tailed Student’s t-tests were used to compare genotypes for statistical analysis of sleep parameters with normal distributions. Mann-Whitney U-tests were used to analyze sleep episode length, which did not have normal distributions. Signal intensities of western blots and calcium images were measured with ImageJ software (National Institute of Health, USA), and data from eight independent experiments were averaged. Statistical significance was set as p < 0.05, p < 0.01, p < 0.001, or no significance.
Additional Information
How to cite this article: Tong, H. et al. Neurexin regulates nighttime sleep by modulating synaptic transmission. Sci. Rep. 6, 38246; doi: 10.1038/srep38246 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Material
Acknowledgments
We thank Dr. Manzoor A. Bhat for providing the nrx273 flies; Dr. Wei Xie for providing the nrxΔ83 flies; Dr. Yoshinori Aso for providing the split GAL4 flies; Dr. Li Liu for providing 104y-GAL4 flies; the Bloomington stock center, VDRC stock center, and Tsinghua Fly Center for providing flies; and members of the Han laboratory for their critical comments on the manuscript. This work was supported by grants from the Ministry of Science and Technology (2012CB517903 and 2014CB942803), the National Natural Science Foundation of China (31471031), the Research Project of Chinese Ministry of Education (413032), the Excellent Youth Foundation of Jiangsu Province of China (BK20140024 to J.H.), the Nature Science Fundation of Jiangsu Province (BK20130606 to Z.C.Z.), the Fundamental Research Funds for the Central Universities, and Funding of Jiangsu Innovation Program for Graduate Education (KYLX_0088).
Footnotes
Author Contributions H.T. and J.H. designed the experiments. H.T., Q.L., and Y.L. performed the experiments and analyzed the data. J.H. and Z.-C.Z. wrote the paper.
References
- Cirelli C. The genetic and molecular regulation of sleep: from fruit flies to humans. Nat. Rev. Neurosci. 10, 549–560 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andretic R., Franken P. & Tafti M. Genetics of sleep. Annu. Rev. Genet. 42, 361–388 (2008). [DOI] [PubMed] [Google Scholar]
- Association. A. P. In 4th ed. Text Revision. (American Psychiatric Association, Washington, DC, 2000). [Google Scholar]
- Souders M. C. et al. Sleep behaviors and sleep quality in children with autism spectrum disorders. Sleep 32, 1566–1578 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maret S. et al. Retinoic acid signaling affects cortical synchrony during sleep. Science 310, 111–113 (2005). [DOI] [PubMed] [Google Scholar]
- Tafti M. et al. Deficiency in short-chain fatty acid beta-oxidation affects theta oscillations during sleep. Nat. Genet. 34, 320–325 (2003). [DOI] [PubMed] [Google Scholar]
- Sehgal A. & Mignot E. Genetics of sleep and sleep disorders. Cell 146, 194–207 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dean C. et al. Neurexin mediates the assembly of presynaptic terminals. Nat. Neurosci. 6, 708–716 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeng X. et al. Neurexin-1 is required for synapse formation and larvae associative learning in Drosophila. FEBS Lett. 581, 2509–2516 (2007). [DOI] [PubMed] [Google Scholar]
- Li J., Ashley J., Budnik V. & Bhat M. A. Crucial role of Drosophila neurexin in proper active zone apposition to postsynaptic densities, synaptic growth, and synaptic transmission. Neuron 55, 741–755 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Missler M. et al. Alpha-neurexins couple Ca2 + channels to synaptic vesicle exocytosis. Nature 423, 939–948 (2003). [DOI] [PubMed] [Google Scholar]
- Li T. et al. The Neurexin/N-Ethylmaleimide-sensitive Factor (NSF) Interaction Regulates Short Term Synaptic Depression. J. Biol. Chem. 290, 17656–17667 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim H. G. et al. Disruption of neurexin 1 associated with autism spectrum disorder. Am. J. Hum. Genet. 82, 199–207 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zahir F. R. et al. A patient with vertebral, cognitive and behavioural abnormalities and a de novo deletion of NRXN1alpha. J. Med. Genet. 45, 239–243 (2008). [DOI] [PubMed] [Google Scholar]
- El Helou J. et al. Neuroligin-1 links neuronal activity to sleep-wake regulation. Proc. Natl. Acad. Sci. USA. 110, 9974–9979 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y. et al. Drosophila neuroligin 4 regulates sleep through modulating GABA transmission. J. Neurosci. 33, 15545–15554 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nitz D. A., van Swinderen B., Tononi G. & Greenspan R. J. Electrophysiological correlates of rest and activity in Drosophila melanogaster. Curr. Biol. 12, 1934–1940 (2002). [DOI] [PubMed] [Google Scholar]
- van Swinderen B., Nitz D. A. & Greenspan R. J. Uncoupling of brain activity from movement defines arousal States in Drosophila. Curr. Biol. 14, 81–87 (2004). [PubMed] [Google Scholar]
- Shaw P. J., Cirelli C., Greenspan R. J. & Tononi G. Correlates of sleep and waking in Drosophila melanogaster. Science 287, 1834–1837 (2000). [DOI] [PubMed] [Google Scholar]
- Cirelli C. et al. Reduced sleep in Drosophila Shaker mutants. Nature 434, 1087–1092 (2005). [DOI] [PubMed] [Google Scholar]
- Joiner W. J., Crocker A., White B. H. & Sehgal A. Sleep in Drosophila is regulated by adult mushroom bodies. Nature 441, 757–760 (2006). [DOI] [PubMed] [Google Scholar]
- Pitman J. L., McGill J. J., Keegan K. P. & Allada R. A dynamic role for the mushroom bodies in promoting sleep in Drosophila. Nature 441, 753–756 (2006). [DOI] [PubMed] [Google Scholar]
- Bushey D., Huber R., Tononi G. & Cirelli C. Drosophila Hyperkinetic mutants have reduced sleep and impaired memory. J. Neurosci. 27, 5384–5393 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ganguly-Fitzgerald I., Donlea J. & Shaw P. J. Waking experience affects sleep need in Drosophila. Science 313, 1775–1781 (2006). [DOI] [PubMed] [Google Scholar]
- Hendricks J. C. et al. Rest in Drosophila is a sleep-like state. Neuron 25, 129–138 (2000). [DOI] [PubMed] [Google Scholar]
- Aso Y. et al. The neuronal architecture of the mushroom body provides a logic for associative learning. eLife 3, e04577 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aso Y. et al. Mushroom body output neurons encode valence and guide memory-based action selection in Drosophila. eLife 3, e04580 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Missler M. & Sudhof T. C. Neurexins: three genes and 1001 products. Trends Genet. 14, 20–26 (1998). [DOI] [PubMed] [Google Scholar]
- Aso Y. et al. The mushroom body of adult Drosophila characterized by GAL4 drivers. J. Neurogenet. 23, 156–172 (2009). [DOI] [PubMed] [Google Scholar]
- Crittenden J. R., Skoulakis E. M., Han K. A., Kalderon D. & Davis R. L. Tripartite mushroom body architecture revealed by antigenic markers. Learn. Mem. 5, 38–51 (1998). [PMC free article] [PubMed] [Google Scholar]
- Murthy M., Fiete I. & Laurent G. Testing odor response stereotypy in the Drosophila mushroom body. Neuron 59, 1009–1023 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dietzl G. et al. A genome-wide transgenic RNAi library for conditional gene inactivation in Drosophila. Nature 448, 151–156 (2007). [DOI] [PubMed] [Google Scholar]
- Nitabach M. N., Blau J. & Holmes T. C. Electrical silencing of Drosophila pacemaker neurons stops the free-running circadian clock. Cell 109, 485–495 (2002). [DOI] [PubMed] [Google Scholar]
- McGuire S. E., Mao Z. & Davis R. L. Spatiotemporal gene expression targeting with the TARGET and gene-switch systems in Drosophila. Sci. STKE 2004, pl6 (2004). [DOI] [PubMed] [Google Scholar]
- Hamada F. N. et al. An internal thermal sensor controlling temperature preference in Drosophila. Nature 454, 217–220 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sitaraman D. et al. Propagation of Homeostatic Sleep Signals by Segregated Synaptic Microcircuits of the Drosophila Mushroom Body. Curr. Biol. 25, 2915–2927 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diegelmann S., Fiala A., Leibold C., Spall T. & Buchner E. Transgenic flies expressing the fluorescence calcium sensor Cameleon 2.1 under UAS control. Genesis 34, 95–98 (2002). [DOI] [PubMed] [Google Scholar]
- Sun J. et al. A dual-Ca2 + -sensor model for neurotransmitter release in a central synapse. Nature 450, 676–682 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Littleton J. T. & Ganetzky B. Ion channels and synaptic organization: analysis of the Drosophila genome. Neuron 26, 35–43 (2000). [DOI] [PubMed] [Google Scholar]
- Jeong K. et al. Ca-alpha1T, a fly T-type Ca2 + channel, negatively modulates sleep. Sci. Rep. 5, 17893 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawasaki F., Zou B., Xu X. & Ordway R. W. Active zone localization of presynaptic calcium channels encoded by the cacophony locus of Drosophila. J. Neurosci. 24, 282–285 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yi W. et al. A subset of cholinergic mushroom body neurons requires Go signaling to regulate sleep in Drosophila. Sleep 36, 1809–1821 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shang Y. et al. Short neuropeptide F is a sleep-promoting inhibitory modulator. Neuron 80, 171–183 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parisky K. M. et al. PDF cells are a GABA-responsive wake-promoting component of the Drosophila sleep circuit. Neuron 60, 672–682 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larkin A. et al. Neurexin-1 regulates sleep and synaptic plasticity in Drosophila melanogaster. Eur. J. Neurosci. 42, 2455–2466 (2015). [DOI] [PubMed] [Google Scholar]
- Donlea J. M., Thimgan M. S., Suzuki Y., Gottschalk L. & Shaw P. J. Inducing sleep by remote control facilitates memory consolidation in Drosophila. Science 332, 1571–1576 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shang Y., Griffith L. C. & Rosbash M. Light-arousal and circadian photoreception circuits intersect at the large PDF cells of the Drosophila brain. Proc. Natl. Acad. Sci. USA. 105, 19587–19594 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Craig A. M. & Kang Y. Neurexin-neuroligin signaling in synapse development. Curr. Opin. Neurobiol. 17, 43–52 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perisse E. et al. Different kenyon cell populations drive learned approach and avoidance in Drosophila. Neuron 79, 945–956 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johard H. A. et al. Intrinsic neurons of Drosophila mushroom bodies express short neuropeptide F: relations to extrinsic neurons expressing different neurotransmitters. J. Comp. Neurol. 507, 1479–1496 (2008). [DOI] [PubMed] [Google Scholar]
- Chen W. et al. Regulation of sleep by the short neuropeptide F (sNPF) in Drosophila melanogaster. Insect Biochem. Mol. Biol. 43, 809–819 (2013). [DOI] [PubMed] [Google Scholar]
- Ito K. et al. The organization of extrinsic neurons and their implications in the functional roles of the mushroom bodies in Drosophila melanogaster Meigen. Learn. Mem. 5, 52–77 (1998). [PMC free article] [PubMed] [Google Scholar]
- Tian Y. et al. Neurexin regulates visual function via mediating retinoid transport to promote rhodopsin maturation. Neuron 77, 311–322 (2013). [DOI] [PubMed] [Google Scholar]
- Huber R. Sleep homeostasis in Drosophila melanogaster. Sleep 27, 628–639 (2004). [DOI] [PubMed] [Google Scholar]
- Liu X. et al. The strength and periodicity of D. melanogaster circadian rhythms are differentially affected by alterations in period gene expression. Neuron 6, 753–766 (1991). [DOI] [PubMed] [Google Scholar]
- Shaw P. J., Tononi G., Greenspan R. J. & Robinson D. F. Stress response genes protect against lethal effects of sleep deprivation in Drosophila. Nature 417, 287–291 (2002). [DOI] [PubMed] [Google Scholar]
- Donlea J. M., Pimentel D. & Miesenbock G. Neuronal machinery of sleep homeostasis in Drosophila. Neuron 81, 860–872 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiala A. & Spall T. In vivo calcium imaging of brain activity in Drosophila by transgenic cameleon expression. Sci. STKE 2003, PL6 (2003). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.