Abstract
Background and Purpose
There has been a longstanding interest in the role of menopause and its correlates in the development of cardiovascular disease (CVD) in women. Menopausal hot flashes are experienced by most midlife women; emerging data link hot flashes to CVD risk indicators. We tested whether hot flashes, measured via state-of-the-art physiologic methods, were associated with greater subclinical atherosclerosis as assessed by carotid ultrasound. We considered the role of CVD risk factors and estradiol concentrations in these associations.
Methods
295 nonsmoking women free of clinical CVD underwent ambulatory physiologic hot flash assessments; a blood draw; and carotid ultrasound measurement of IMT and plaque. Associations between hot flashes and subclinical atherosclerosis were tested in regression models controlling for CVD risk factors and estradiol.
Results
More frequent physiologic hot flashes were associated with higher carotid intima media thickness [IMT; for each additional hot flash: beta (standard error)=.004(.001), p=.0001; reported hot flash: beta (standard error)=.008(.002), p=.002, multivariable] and plaque [e.g., for each additional hot flash, odds ratio (95% confidence interval) plaque index ≥2=1.07(1.003–1.14, p=.04), relative to no plaque, multivariable] among women reporting daily hot flashes; associations were not accounted for by CVD risk factors or by estradiol. Among women reporting hot flashes, hot flashes accounted for more variance in IMT than most CVD risk factors.
Conclusions
Among women reporting daily hot flashes, frequent hot flashes may provide information about a woman’s vascular status beyond standard CVD risk factors and estradiol. Frequent hot flashes may mark a vulnerable vascular phenotype among midlife women.
Keywords: women, menopause, hot flashes, atherosclerosis, cardiovascular disease
Introduction
Cardiovascular disease (CVD) is the leading cause of death among women.1 As women typically manifest with atherosclerotic CVD postmenopausally, on average 10 years after men, there has been a longstanding interest in the role of the menopause transition and its correlates in the development of atherosclerotic CVD in women.2 The focus of this work has largely been on the hormonal changes associated with menopause. Recent data has also considered how other menopause-related factors, including menopausal symptoms, relate to CVD risk in women.
Hot flashes are the “classic” menopausal symptom, reported by >70% of midlife women.3 For a third of women, hot flashes are frequent or severe.4 The impact of hot flashes on quality of life is well-documented,5 and hot flashes are strong drivers of health care utilization.6 However, hot flashes are thought to have few implications for women’s physical health.
Newer data challenge that assumption. Posthoc analyses from large hormone therapy (HT) trials suggested that the CVD risk with HT use was highest among older women reporting moderate-severe hot flashes at baseline.7, 8 Later observations from cohort studies suggest that greater hot flash reporting may be associated with a poorer CVD risk factor profile9, 10 and higher subclinical atherosclerosis11–14 beyond standard risk factors. However, studies specifically designed to test relations among hot flashes and CVD risk are absent. The limitations in this literature, including retrospective hot flash measures vulnerable to multiple biases,15, 16 exclusion of highly symptomatic women,8, 17 the group most affected, and contradictory findings17 have limited conclusions about the precise nature of hot flash-CVD risk associations.
In a sample of nonsmoking women without clinical CVD, we used state-of-the-art ambulatory physiologic and prospective ecological momentary reports of hot flashes to test whether hot flashes (presence, frequency) were associated with subclinical atherosclerosis as assessed by carotid intima media thickness (IMT) and plaque. Carotid IMT and plaque are widely-used and well-validated indicators of subclinical atherosclerosis predictive of later clinical CVD, including among relatively low risk samples (e.g., midlife women).18–20 They are preferable to other widely used subclinical atherosclerosis indices (e.g., coronary artery calcification) that show a high rate of zero readings among midlife women.21 We tested the role of standard and novel CVD risk factors in these relations. Finally, as endogenous estradiol has been implicated in hot flashes and atherosclerosis in women,22 we considered estradiol concentrations in these relations.
Methods
Study Sample
The study sample was comprised of 304 late perimenopausal (2–12 months amenorrhea) and postmenopausal (≥12 months amenorrhea) nonsmoking women aged 40–60. By design, half of the women reported daily hot flashes or night sweats, and half reported no hot flashes or night sweats in the past three months. Of the 1929 women who underwent telephone screening, 304 were interested, eligible, and enrolled and had usable physiologic hot flash monitoring data (≥70% of 24 hours). Exclusion criteria were based upon factors having a major impact on study measures or safety and included hysterectomy and/or bilateral oophorectomy; history of heart disease, stroke, arrhythmia, ovarian/gynecological cancer, pheochromocytoma, pancreatic tumor, kidney failure, seizures, Parkinson’s disease, Raynaud’s Phenomenon; current pregnancy; or having used select medications in the past 3 months (oral/transdermal estrogen or progesterone, selective estrogen receptor modulators, selective serotonin reuptake inhibitors, serotonin norepinephrine reuptake inhibitors, gabapentin, insulin, beta blockers, calcium channel blockers, alpha-2 adrenergic agonists, other antiarrhythmic agents). Women who had undergone uterine ablation, endarterectomy, or lymph node removal or who were undergoing dialysis or chemotherapy were also excluded.
Of the 304 women, 4 women were excluded due to missing carotid data (equipment failure or poor image) and 5 women excluded due to missing blood marker data [Homeostatic Model Assessment (HOMA): N=3, low density lipoprotein cholesterol (LDL-C): N=2]. Excluded women had higher triglycerides than included women (p<.05). 295 women were included in final models.
Design and Procedures
Women were recruited from the community via advertisements, mailings, and message boards. Participants underwent physical measurements, hot flash monitoring, a blood draw, and a carotid artery ultrasound. Procedures were approved by the University of Pittsburgh Institutional Review Board. Participants provided written, informed consent.
Measures
Hot Flashes
Participants completed three days of ambulatory hot flash monitoring, the first 24 hours of which included physiologic hot flash monitoring. Women were equipped with a hot flash monitor (VU-AMS, VU University Amsterdam, Netherlands), electronic diary, and wrist actigraph. The VU-AMS is a wearable monitor that quantifies hot flashes via sternal skin conductance, a validated physiologic measure of hot flashes.23 Women reported hot flashes by completing an electronic diary (Palm Z22) and pressing event mark buttons on the VU-AMS monitor and actigraph, providing date and time-stamped hot flash reports. Participants wore the VU-AMS monitor for 24 hours, after which time they removed it and stored it in a provided case. For the remaining two days, women carried the diary and actigraph. After monitoring, hot flash data were downloaded and scored via UFI software (DPSv3.7; Morro Bay, CA) according to validated methods that have demonstrated reliability, including in the present laboratory (ĸ=.86).23, 24
Carotid Ultrasound
Trained and certified sonographers at the University of Pittsburgh’s Ultrasound Research Laboratory obtained bilateral carotid images via B-mode ultrasound using a Sonoline Antares (Siemens, Malvern, PA) high resolution duplex scanner equipped with a VF10-5 transducer. Digitized images were obtained from eight locations (four locations each from the left and right carotid arteries): near and far walls of the distal common carotid artery, far walls of the carotid bulb, and internal carotid artery. Images were read using semi-automated reading software.25 Values were obtained by electronically tracing the lumen-intima interface and the media-adventitia interface across a 1-cm segment for each of these eight segments. Average and maximal values were recorded for each of the eight locations; the mean of the average and maximal readings across the eight locations comprised mean and maximal IMT, respectively. Reproducibility of IMT measures was excellent [intraclass correlation coefficient between sonographers 0.87–0.94, between readers= 0.94–0.99].
Carotid plaque was defined as a distinct focal area protruding into the vessel lumen ≥50% thicker than the adjacent IMT.26 Sonographers evaluated the presence and extent of plaque in each of five segments of the left and right carotid artery (distal and proximal common carotid artery, carotid bulb, and proximal internal and external carotid arteries).26 Consistent with the Mannheim Consensus Statement,27 plaque was defined as a focal area protruding into the vessel lumen that was at least 50% thicker than the adjacent IMT and summarized as the presence or absence of any plaque. Additionally, for each segment the degree of plaque was graded using the following criteria: Grade 0=no observable plaque; grade 1=one small plaque (<30% of the vessel diameter); grade 2=one medium plaque (30–50% of the vessel diameter) or multiple small plaques; grade 3=one large plaque (>50% of the vessel diameter) or multiple plaques with at least one medium plaque. The grades from all segments of the combined left and right carotid artery were summed to create the plaque index,28 which was categorized as 0, 1, or ≥2 for analysis. Between sonographers agreement for carotid plaque assessment was good to excellent (kappa statistic, κ=0.78).
Covariates
Height and weight were measured via a fixed stadiometer and balance beam scale; BMI was calculated (kg/m2). Seated blood pressure was measured via a Dinamap device after 10-min rest. Demographics and medical history were assessed by standard instruments. Menopause status was obtained from reported menstrual bleeding patterns.29 Depressive symptoms were assessed by the Center for Epidemiologic Studies Depression scale.30 Sleep/wake was assessed via actigraphy and sleep diary.31 Use of medications for blood pressure-lowering, lipid-lowering, or diabetes were reported and considered as covariates.
Phlebotomy was performed after a 12-hr fast. Glucose, triglycerides, and high-density lipoprotein cholesterol (HDL-C) were measured enzymatically. Total cholesterol was determined enzymatically and LDL-C calculated.32 Insulin was measured via radioimmunoassay. HOMA, reflecting insulin resistance, was calculated.33 C-reactive protein was measured using a high sensitivity reagent set (Beckman Coulter, Brea, CA) and interleukin-6 with an R&D Systems (Minneapolis, MN) high sensitivity ELISA. Estradiol was assessed via liquid chromatography-tandem mass spectrometry, the gold standard method to measure estradiol at low postmenopausal levels (lower limit of quantitation=2.5pg/mL; lower limit of detection=1.0 pg/ml).34
Data analysis
HOMA, triglycerides, estradiol, C-reactive protein, and interleukin-6 values were natural log transformed for analysis. Hot flashes were categorized as occurring during sleep or wake according to sleep diary and actigraph. Hot flash rates were calculated as number of hot flashes/ monitoring time. Rates were standardized to a 7-hour and 17-hour sleep and wake times for ease of interpretation. 24-hour, sleep, and wake hot flash rates were considered separately. Differences between participants by hot flash status (any/none) were tested using linear regression, Wilcoxon rank sum, and chi-square tests. Associations between hot flash frequency and outcomes were evaluated using linear and multinomial logistic regression. Covariates (age, race/ethnicity, BMI, education, systolic and diastolic blood pressure, triglycerides, LDL-C, HDL-C, HOMA) were selected based upon their prior documented associations with IMT and present associations with outcomes at p<.10, with medication variables forced into models. Estradiol was added in a separate step. Interactions were tested by cross product terms in multivariable models. R2 values were derived from linear regression models. Residual analysis and diagnostic plots were conducted to verify model assumptions. Analyses were performed with SAS v9.2 (SAS Institute, Cary, NC). Models were 2-sided at α=0.05.
Results
Participants were on average 54 years of age, normotensive, overweight, and postmenopausal (Table 1). Women reporting daily hot flashes (“flashers”) were younger, less educated, more often non-White, and had a higher diastolic blood pressure than women not reporting hot flashes (“nonflashers”) during the three months prior to enrollment. Across the sample, 2422 hot flashes were physiologically-detected, and 2335 were reported. Among “flashers,” median numbers of physiologically-detected and self-reported hot flashes/24 hours were 12 and 5, respectively. Among women not reporting hot flashes (“nonflashers”), many (46%) showed evidence of physiologic hot flashes, albeit at a low frequency (24 hour: median=0, interquartile range: 0, 5).
Table 1.
Sample characteristics
| Flasher (N=147) | Non-Flasher (N=148) | |
|---|---|---|
| Age, years, M (SD)* | 53.34 (3.60) | 54.73 (4.28) |
| Race/ethnicity, N (%)* | ||
| White | 96 (65.31) | 116 (78.38) |
| African American | 45 (30.61) | 21 (14.19) |
| Other | 6 (4.08) | 11 (7.43) |
| Education* | ||
| High school, some college, vocational | 72 (48.98) | 54 (36.49) |
| College graduate | 47 (31.97) | 41 (27.70) |
| >College | 28 (19.05) | 53 (35.81) |
| Body mass index, M (SD) | 29.10 (6.55) | 28.97 (6.94) |
| Systolic blood pressure, mmHg, M (SD) | 120.16 (14.15) | 119.64 (14.79) |
| Diastolic blood pressure, mmHg, M (SD)* | 71.47 (8.74) | 68.98 (9.33) |
| Menopause stage | ||
| Perimenopausal | 30 (20.41) | 18 (12.16) |
| Postmenopausal | 117 (79.59) | 130 (87.84) |
| High density lipoprotein cholesterol, mg/dL, M (SD) | 62.60 (15.18) | 63.20 (14.44) |
| Low density lipoprotein cholesterol, mg/dL, M (SD) | 130.19 (30.88) | 130.70 (35.72) |
| Triglycerides, mg/dL, Median (IQR) | 95.00 (72, 124) | 94.50 (70, 130) |
| Homeostatic model assessment, Median (IQR) | 2.16 (1.58, 3.18) | 2.19 (1.69, 3.12) |
| Estradiol, pg/mL, Median (IQR) | 4.30 (2.00, 9.00) | 5.00 (2.00, 12.35) |
| Medications, N (%) | ||
| Lipid-lowering | 17 (11.56) | 19 (12.84) |
| Blood pressure-lowering | 25 (17.01) | 22 (14.86) |
| Diabetes medication | 4 (2.72) | 5 (3.38) |
| Physiologically detected hot flashes, Median (IQR)† | ||
| 24-hour | 12 (7, 19) | 0 (0, 5) |
| Wake | 10 (4, 15) | 0 (0, 3) |
| Sleep | 3 (1, 5) | 0 (0, 2) |
| Self-reported hot flashes, Median (IQR)† | ||
| 24-hour | 5 (3, 7) | 0 (0, 0) |
| Wake | 4 (2, 6) | 0 (0, 0) |
| Sleep | 1 (0, 2) | 0 (0, 0) |
p<0.05 differs by hot flash group (daily hot flashes versus no hot flashes, past three months prior to enrollment);
Sleep and wake values standardized to 7-hour and 17-hour sleep and wake durations for interpretation
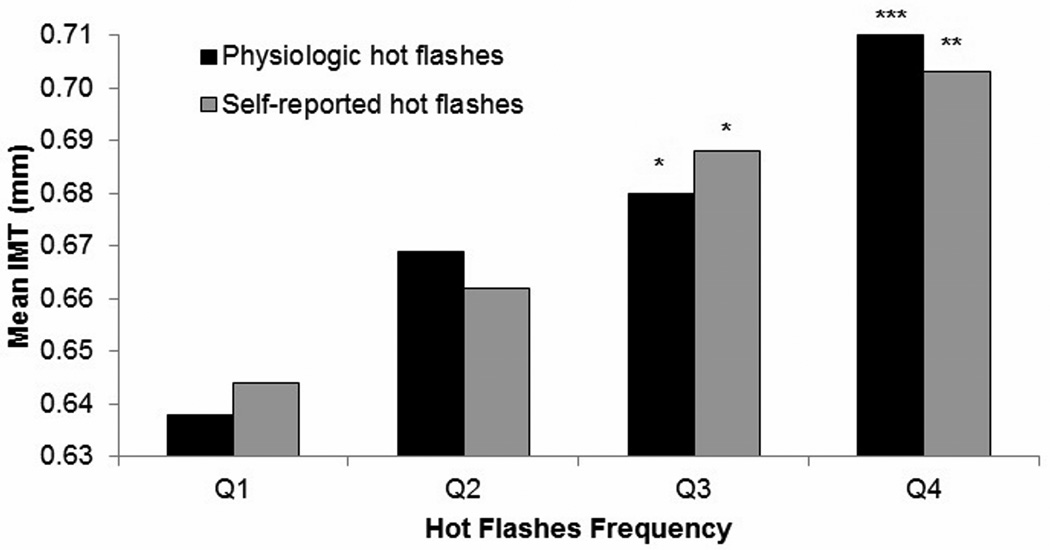
The mean and maximal IMT was .68 mm (SD=.11) and .85 (SD=.16), respectively. Mean IMT did not vary by hot flash group (“flasher” vs. “nonflasher”) in multivariable models [b(SE)=−.008 (.01), p=.46, flasher: raw mean (SD)=.67(.09), range: .51–.98, nonflasher: raw mean (SD)=.69(.12) range: .50–1.28]. Maximal IMT also did not differ by hot flash group [b(SE)=−.007(.02), p=.66]. However, significant interactions by hot flash status and hot flash frequency in relation to IMT were observed (p=.02). Among women reporting hot flashes (N=147), more frequent hot flashes were strongly associated with higher mean and maximal IMT in multivariable models (Table 2; Figure 1). Associations were not accounted for by CVD risk factors. Estradiol was not related to IMT [b(SE)=−.002 (.005), p=.75, multivariable] and did not impact hot flash-IMT associations [e.g., waking physiologic hot flashes and IMT: b(SE)=.004 (.001), p=.0001, multivariable models with estradiol]. Among women reporting hot flashes, physiologic hot flashes accounted for more variance in IMT than any other factor except race/ethnicity (Table 3).
Table 2.
Relation between hot flashes and carotid intima media thickness (IMT) and plaque among women reporting daily hot flashes
| Mean IMT | Max IMT | Plaque index | ||
|---|---|---|---|---|
| 1 | ≥2 | |||
| beta (standard error)§ |
beta (standard error)§ |
Odds ratio (confidence interval)‖ |
Odds ratio (confidence interval)‖ |
|
| Physiologic Hot Flashes | ||||
| 24-hour | .003 (.001)† | .004 (.001)† | 1.00 (.94–1.06) | 1.06 (1.001–1.12)* |
| Wake | .004 (.001)‡ | .005 (.001)‡ | 0.99 (.92–1.06) | 1.07 (1.003–1.14)* |
| Sleep | .003 (.003) | .004 (.005) | 0.96 (.77–1.21) | 1.17 (.96–1.42) |
| Self-Reported Hot Flashes | ||||
| 24-hour | .006 (.002)† | .007 (.003)* | 1.06 (.92–1.21) | 1.06 (.93–1.21) |
| Wake | .008 (.002)† | .009 (.004)* | 1.09 (.92–1.30) | 1.12 (.95–1.32) |
| Sleep | .008 (.006) | .01 (.009) | .98 (.65–1.48) | 1.00 (.70–1.42) |
Covariates: age, race, body mass index, education, high density lipoprotein cholesterol, low density lipoprotein cholesterol, triglycerides, systolic blood pressure, diastolic blood pressure, homeostatic model assessment, blood pressure-lowering medications, diabetes medications, and lipid-lowering medications
p<.05;
p<.01,
p<.001
N=147; IMT measured in mm
beta coefficients indicate mm increase in IMT for each additional hot flash
Odds ratio associated with each additional hot flash, relative to no plaque
Figure 1.
Adjusted means in average IMT by hot flash frequency among women in daily hot flash group.
Quartile (Q) 1: ≤ 4 physiologic hot flashes, ≤ 1 self-reported hot flashes; Q2: 5–9 physiologic hot flashes, 2–3 self-reported hot flashes; Q3: 10–14 physiologic hot flashes, 4–5 self-reported hot flashes; Q4: ≥ 15 physiologic hot flashes, ≥ 6 self-reported hot flashes; waking hot flashes Adjusted for age, race, body mass index, education, high density lipoprotein cholesterol, low density lipoprotein cholesterol, triglycerides, systolic blood pressure, diastolic blood pressure, homeostatic model assessment, blood pressure-lowering medications, lipid-lowering medications, diabetes medications
IMT = intima media thickness
*p<.05; **p<.01; ***p<.001 relative to Q1
Table 3.
Percent of variance (R2) in mean intima media thickness (IMT) explained by each variable among women reporting daily hot flashes (N=147)
| Variable | R2 |
|---|---|
| Age | 4.72 |
| Race | 8.75 |
| Education | 0.98 |
| Body mass index | 1.50 |
| Systolic blood pressure | 0.19 |
| Diastolic blood pressure | 0.01 |
| High-density lipoprotein cholesterol | 0.31 |
| Low-density lipoprotein cholesterol | 2.71 |
| Triglycerides | 0.91 |
| Homeostatic model assessment | 0.13 |
| Lipid-lowering medication | 0.99 |
| Blood pressure-lowering medication | 0.67 |
| Diabetes medication | 0.19 |
| Physiologic waking hot flashes | 7.86 |
We also considered carotid plaque. Over half the women (54%, N=160) showed no plaque, 21% (N=63) had a plaque index of 1, and 25% (N=73) had a plaque index of ≥2. Plaque did not differ by reported hot flash status [flasher vs. nonflasher: plaque index ≥2, OR(95%CI)= 1.06(.56–2.01), p=.85); plaque index 1, OR(95%CI)=1.08(.58–2.01), p=.82, relative to no plaque, multivariable]. However, interactions by hot flash status (p=.02) indicated that among women reporting hot flashes, more frequent waking physiologic hot flashes were associated with higher carotid plaque (Table 3). Estradiol did not impact associations (data not shown).
We conducted several additional analyses. No significant interactions by age, race, menopause stage, time since final menstrual period, and BMI for hot flash-IMT relations were observed. However, for plaque, significant interactions by age were observed in the total sample (p=.04) and in flashers (p=.04), with positive relations between hot flashes and plaque observed largely among the older (e.g., ≥54, upper median) women [e.g., for each additional physiologic hot flash among flashers: plaque index ≥2 OR(95%CI)=1.14 (1.03–1.26), p=.009, multivariable]. Further, we tested interactions for blood pressure, finding significant interactions between waking physiologic hot flashes and DBP in relation to IMT among the flashers (p=.04). Probing these interactions indicated that associations were strongest among the women at the upper median of DBP [≥70mg/dL: b=.005 (SE=.001), p<.001; <70mg/dL: b=.001 (SE=.001), p=.59]. To better understand factors that may account for hot flash-IMT relations, we considered depressive symptoms, interleukin-6, C-reactive protein as well as menopause stage as additional covariates; associations between hot flashes and IMT or plaque persisted (data not shown). Finally, neither diary-rated hot flash severity nor bother were associated with outcomes (data not shown).
Discussion
We present the results from the first study designed to test associations between menopausal hot flashes and markers of carotid atherosclerosis. Among midlife women reporting daily hot flashes, a greater frequency of hot flashes was associated with higher carotid IMT and plaque. The associations were not accounted for by CVD risk factors nor by estradiol. Among women reporting hot flashes, hot flashes accounted for more variance in IMT than most CVD risk factors.
These findings contribute to the literature on hot flashes and markers of CVD risk. Initial observations of relations between hot flashes and CVD risk arose from posthoc analysis of large HT trials7, 8. Subsequent observations from the Study of Women’s Health Across the Nation (SWAN) and other cohort studies indicated that reported hot flashes were positively associated with subclinical atherosclerosis.11–14 In other work, we have found hot flashes associated with brain white matter hyperintensities.35 Not all studies have linked hot flashes to CVD risk.17, 36 The existing literature has been limited by the heavy reliance upon retrospective measurements of hot flashes asking women to recall their hot flashes up to a decade prior.36 Highly symptomatic women are typically not differentiated from their less symptomatic counterparts; and hot flash severity, bother, and frequency are conflated, constructs that are not interchangeable, as evidenced here. In this most rigorous test of this question to date, among women reporting hot flashes, more frequent hot flashes were associated with IMT, accounting for more variance in IMT in this study than most standard CVD risk factors.
The mechanisms linking hot flashes to CVD risk are not yet clear, partly due to the limited understanding of hot flash physiology. Hot flashes may represent thermoregulatory events.37 However, other mechanisms have been implicated. Hot flashes have been linked to a poorer CVD risk factor profile,9, 10 yet risk factors did not explain observed associations. Lower estradiol is permissive to hot flash occurrence and has been linked to poorer cardiovascular health for women.22 However estradiol, measured via state-of-the-art methods that detect the low levels of estradiol observed among postmenopausal women, did not explain associations. Future work should consider other novel pathways (e.g., sympathetic nervous system, hypothalamic pituitary adrenal axis) in hot flash-CVD risk relations.
Both self-reported and physiologically-monitored hot flashes were prospectively measured here. These measures improve upon typically employed questionnaires, avoiding the influence of memory and other limitations.16 We found that women under-reported their hot flashes, consistent with prior work.38 Further, many “nonflashers” showed low frequency hot flashes on physiologic monitoring; we have previously shown these hot flashes to have a similar autonomic signature as reported hot flashes.39 Wake hot flashes showed stronger relations to subclinical atherosclerosis than sleep hot flashes, consistent with some prior work.11, 13 Finally, as opposed to low frequency hot flashes, the present findings underscore the clinical significance of frequent flashing (e.g., 10+ physiologic hot flashes/day), a frequency found in half of the women reporting hot flashes in this sample.
This work had limitations. This observational study does not allow for conclusions about directionality or causality of relations. Use of subclinical atherosclerosis indices is necessary given the rarity of clinical events in midlife women, but limits conclusions about clinical disease. Estradiol concentrations, but not estradiol fluctuations, were quantified, yet most of the women were postmenopausal, a time when estradiol typically stabilizes at low levels. Blood pressure variability, which has been linked to CVD risk beyond blood pressure levels alone,40 was not measured here and its role in these relations should be considered in future work. Although the sample was 25% nonwhite, Asian and Hispanic women were under-represented. By design, smokers, women reporting infrequent hot flashes (< daily), and women with hysterectomy or bilateral oophorectomy were excluded. Findings cannot be generalized to these groups; future work should consider these women. Hot flashes were captured once over several days; yet next steps should include a longitudinal study quantifying hot flashes over multiple time points.
Conclusions
More frequent hot flashes were associated with markers of carotid atherosclerosis among midlife women reporting daily hot flashes. This line of work may ultimately have clinical implications for women with frequent hot flashes. For women, midlife is typically decades before the emergence of clinical events and is a time in which CVD risk stratification can be challenging; with additional replication and extension of this work, hot flashes may ultimately assist in that effort. With further understanding of hot flash-CVD risk relations, hot flashes may have implications for understanding the accelerated changes in the vasculature occurring during menopause,41 changes not fully explained by reproductive hormones or aging. This body of work begins to call into question the solely incidental nature of this midlife symptom.
Acknowledgments
Sources of Funding
Supported by the National Institutes of Health, National Heart Lung and Blood Institute (R01HL105647, K24123565 to Thurston) and the University of Pittsburgh Clinical and Translational Science Institute (NIH Grant UL1TR000005).
Footnotes
Disclosures: Thurston: None; Chang: None; Barinas-Mitchell: None; Jennings: None; Landsittel: None; Santoro: Grant support: Bayer Healthcare, Stock options: Menogenix; von Känel: None; Matthews: None.
References
- 1.Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M, et al. Heart disease and stroke statistics-2016 update: A report from the american heart association. Circulation. 2016;133:e38–e360. doi: 10.1161/CIR.0000000000000350. [DOI] [PubMed] [Google Scholar]
- 2.Mehta LS, Beckie TM, DeVon HA, Grines CL, Krumholz HM, Johnson MN, et al. Acute myocardial infarction in women: A scientific statement from the american heart association. Circulation. 2016;133:916–947. doi: 10.1161/CIR.0000000000000351. [DOI] [PubMed] [Google Scholar]
- 3.Gold E, Colvin A, Avis N, Bromberger J, Greendale G, Powell L, et al. Longitudinal analysis of vasomotor symptoms and race/ethnicity across the menopausal transition: Study of women’s health across the nation (swan) Am J Public Health. 2006;96:1226–1235. doi: 10.2105/AJPH.2005.066936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Williams RE, Kalilani L, DiBenedetti DB, Zhou X, Granger AL, Fehnel SE, et al. Frequency and severity of vasomotor symptoms among peri- and postmenopausal women in the united states. Climacteric. 2008;11:32–43. doi: 10.1080/13697130701744696. [DOI] [PubMed] [Google Scholar]
- 5.Avis NE, Ory M, Matthews KA, Schocken M, Bromberger J, Colvin A. Health-related quality of life in a multiethnic sample of middle-aged women: Study of women's health across the nation (swan) Med Care. 2003;41:1262–1276. doi: 10.1097/01.MLR.0000093479.39115.AF. [DOI] [PubMed] [Google Scholar]
- 6.Williams RE, Kalilani L, DiBenedetti DB, Zhou X, Fehnel SE, Clark RV. Healthcare seeking and treatment for menopausal symptoms in the united states. Maturitas. 2007;58:348–358. doi: 10.1016/j.maturitas.2007.09.006. [DOI] [PubMed] [Google Scholar]
- 7.Huang AJ, Sawaya GF, Vittinghoff E, Lin F, Grady D. Hot flushes, coronary heart disease, and hormone therapy in postmenopausal women. Menopause. 2009;16:639–643. doi: 10.1097/gme.0b013e31819c11e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rossouw JE, Prentice RL, Manson JE, Wu L, Barad D, Barnabei VM, et al. Postmenopausal hormone therapy and risk of cardiovascular disease by age and years since menopause. JAMA. 2007;297:1465–1477. doi: 10.1001/jama.297.13.1465. [DOI] [PubMed] [Google Scholar]
- 9.Gast GC, Grobbee DE, Pop VJ, Keyzer JJ, Wijnands-van Gent CJ, Samsioe GN, et al. Menopausal complaints are associated with cardiovascular risk factors. Hypertension. 2008;51:1492–1498. doi: 10.1161/HYPERTENSIONAHA.107.106526. [DOI] [PubMed] [Google Scholar]
- 10.Thurston RC, El Khoudary SR, Sutton-Tyrrell K, Crandall CJ, Sternfeld B, Joffe H, et al. Vasomotor symptoms and insulin resistance in the study of women's health across the nation. J Clin Endocrinol Metab. 2012;97:3487–3494. doi: 10.1210/jc.2012-1410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Thurston RC, Sutton-Tyrrell K, Everson-Rose SA, Hess R, Matthews KA. Hot flashes and subclinical cardiovascular disease: Findings from the study of women's health across the nation heart study. Circulation. 2008;118:1234–1240. doi: 10.1161/CIRCULATIONAHA.108.776823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lambrinoudaki I, Augoulea A, Armeni E, Rizos D, Alexandrou A, Creatsa M, et al. Menopausal symptoms are associated with subclinical atherosclerosis in healthy recently postmenopausal women. Climacteric. 2012;15:350–357. doi: 10.3109/13697137.2011.618564. [DOI] [PubMed] [Google Scholar]
- 13.Thurston RC, Sutton-Tyrrell K, Everson-Rose SA, Hess R, Powell LH, Matthews KA. Hot flashes and carotid intima media thickness among midlife women. Menopause. 2011;18:352–358. doi: 10.1097/gme.0b013e3181fa27fd. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ozkaya E, Cakir E, Kara F, Okuyan E, Cakir C, Ustun G, et al. Impact of hot flashes and night sweats on carotid intima-media thickness and bone mineral density among postmenopausal women. Int J Gynaecol Obstet. 2011;113:235–238. doi: 10.1016/j.ijgo.2010.12.020. [DOI] [PubMed] [Google Scholar]
- 15.Erskine A, Morley S, Pearce S. Memory for pain: A review. Pain. 1990;41:255–265. doi: 10.1016/0304-3959(90)90002-U. [DOI] [PubMed] [Google Scholar]
- 16.Fu PB, Matthews KA, Thurston RC. How well do different measurement modalities estimate the number of vasomotor symptoms? Findings from the study of women's health across the nation flashes study. Menopause. 2014;21:124–130. doi: 10.1097/GME.0b013e318295a3b9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wolff EF, He Y, Black DM, Brinton EA, Budoff MJ, Cedars MI, et al. Self-reported menopausal symptoms, coronary artery calcification, and carotid intima-media thickness in recently menopausal women screened for the kronos early estrogen prevention study (keeps) Fertil Steril. 2013;99:1385–1391. doi: 10.1016/j.fertnstert.2012.11.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Stein JH, Korcarz CE, Hurst RT, Lonn E, Kendall CB, Mohler ER, et al. Use of carotid ultrasound to identify subclinical vascular disease and evaluate cardiovascular disease risk: A consensus statement from the american society of echocardiography carotid intima-media thickness task force. Endorsed by the society for vascular medicine. J Am Soc Echocardiogr. 2008;21:93–111. doi: 10.1016/j.echo.2007.11.011. quiz 189–190. [DOI] [PubMed] [Google Scholar]
- 19.Peters SA, den Ruijter HM, Bots ML, Moons KG. Improvements in risk stratification for the occurrence of cardiovascular disease by imaging subclinical atherosclerosis: A systematic review. Heart. 2012;98:177–184. doi: 10.1136/heartjnl-2011-300747. [DOI] [PubMed] [Google Scholar]
- 20.Polak JF, Szklo M, Kronmal RA, Burke GL, Shea S, Zavodni AE, et al. The value of carotid artery plaque and intima-media thickness for incident cardiovascular disease: The multi-ethnic study of atherosclerosis. JAHA. 2013;2:e000087. doi: 10.1161/JAHA.113.000087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.McClelland RL, Chung H, Detrano R, Post W, Kronmal RA. Distribution of coronary artery calcium by race, gender, and age: Results from the multi-ethnic study of atherosclerosis (mesa) Circulation. 2006;113:30–37. doi: 10.1161/CIRCULATIONAHA.105.580696. [DOI] [PubMed] [Google Scholar]
- 22.Lenfant F, Tremollieres F, Gourdy P, Arnal JF. Timing of the vascular actions of estrogens in experimental and human studies: Why protective early, and not when delayed? Maturitas. 2011;68:165–173. doi: 10.1016/j.maturitas.2010.11.016. [DOI] [PubMed] [Google Scholar]
- 23.Freedman RR. Laboratory and ambulatory monitoring of menopausal hot flashes. Psychophysiology. 1989;26:573–579. doi: 10.1111/j.1469-8986.1989.tb00712.x. [DOI] [PubMed] [Google Scholar]
- 24.Thurston RC, Matthews KA, Hernandez J, De La Torre F. Improving the performance of physiologic hot flash measures with support vector machines. Psychophysiology. 2009;46:285–292. doi: 10.1111/j.1469-8986.2008.00770.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wendelhag I, Gustavsson T, Suurkula M, Berglund G, Wikstrand J. Ultrasound measurement of wall thickness in the carotid artery: Fundamental principles and description of a computerized analysing system. Clin Physiol. 1991;11:565–577. doi: 10.1111/j.1475-097x.1991.tb00676.x. [DOI] [PubMed] [Google Scholar]
- 26.Thompson T, Sutton-Tyrrell K, Wildman RP, Kao A, Fitzgerald SG, Shook B, et al. Progression of carotid intima-media thickness and plaque in women with systemic lupus erythematosus. Arthritis Rheum. 2008;58:835–842. doi: 10.1002/art.23196. [DOI] [PubMed] [Google Scholar]
- 27.Touboul PJ, Hennerici MG, Meairs S, Adams H, Amarenco P, Bornstein N, et al. Mannheim carotid intima-media thickness and plaque consensus (2004–2006–2011). An update on behalf of the advisory board of the 3rd, 4th and 5th watching the risk symposia, at the 13th, 15th and 20th european stroke conferences, mannheim, germany, 2004, brussels, belgium, 2006, and hamburg, germany, 2011. Cerebrovasc Dis. 2012;34:290–296. doi: 10.1159/000343145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sutton-Tyrrell K, Kuller LH, Matthews KA, Holubkov R, Patel A, Edmundowicz D, et al. Subclinical atherosclerosis in multiple vascular beds: An index of atherosclerotic burden evaluated in postmenopausal women. Atherosclerosis. 2002;160:407–416. doi: 10.1016/s0021-9150(01)00591-3. [DOI] [PubMed] [Google Scholar]
- 29.Harlow SD, Gass M, Hall JE, Lobo R, Maki P, Rebar RW, et al. Executive summary of the stages of reproductive aging workshop + 10: Addressing the unfinished agenda of staging reproductive aging. J Clin Endocrinol Metab. 2012;97:1159–1168. doi: 10.1210/jc.2011-3362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Radloff LS. The ces-d scale: A self-report depression scale for research in the general population. Appl Psychol Meas. 1977;1:385–401. [Google Scholar]
- 31.Ancoli-Israel S, Cole R, Alessi C, Chambers M, Moorcroft W, Pollak CP. The role of actigraphy in the study of sleep and circadian rhythms. Sleep. 2003;26:342–392. doi: 10.1093/sleep/26.3.342. [DOI] [PubMed] [Google Scholar]
- 32.Friedewald W, Levy R, Fredrickson D. Estimation of the concentration of low density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18:499–502. [PubMed] [Google Scholar]
- 33.Matthews D, Hosker J, Rudenski A, Naylor B, Teacher D, Turner R. Homeostasis model assessment: Insulin resistance and b cell function from fasting plasma glucose and insulin concentration in man. Diabetologia. 1985;28:412–419. doi: 10.1007/BF00280883. [DOI] [PubMed] [Google Scholar]
- 34.Santen RJ, Lee JS, Wang S, Demers LM, Mauras N, Wang H, et al. Potential role of ultra-sensitive estradiol assays in estimating the risk of breast cancer and fractures. Steroids. 2008;73:1318–1321. doi: 10.1016/j.steroids.2008.05.008. [DOI] [PubMed] [Google Scholar]
- 35.Thurston RC, Aizenstein HJ, Derby CA, Sejdic E, Maki PM. Menopausal hot flashes and white matter hyperintensities. Menopause. 2016;23:27–32. doi: 10.1097/GME.0000000000000481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Szmuilowicz ED, Manson JE, Rossouw JE, Howard BV, Margolis KL, Greep NC, et al. Vasomotor symptoms and cardiovascular events in postmenopausal women. Menopause. 2011;18:603–610. doi: 10.1097/gme.0b013e3182014849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Freedman RR. Physiology of hot flashes. Am J Hum Biol. 2001;13:453–464. doi: 10.1002/ajhb.1077. [DOI] [PubMed] [Google Scholar]
- 38.Mann E, Hunter MS. Concordance between self-reported and sternal skin conductance measures of hot flushes in symptomatic perimenopausal and postmenopausal women: A systematic review. Menopause. 2011;18:709–722. doi: 10.1097/gme.0b013e318204a1fb. [DOI] [PubMed] [Google Scholar]
- 39.Thurston RC, Matthews KA, Chang Y, Santoro N, Barinas-Mitchell E, von Känel R, et al. Changes in heart rate variability during vasomotor symptoms among midlife women. Menopause. 2016;23:499–505. doi: 10.1097/GME.0000000000000586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Parati G, Ochoa JE, Lombardi C, Bilo G. Blood pressure variability: Assessment, predictive value, and potential as a therapeutic target. Curr Hypertens Rep. 2015;17:537. doi: 10.1007/s11906-015-0537-1. [DOI] [PubMed] [Google Scholar]
- 41.El Khoudary SR, Wildman RP, Matthews K, Thurston RC, Bromberger JT, Sutton-Tyrrell K. Progression rates of carotid intima-media thickness and adventitial diameter during the menopausal transition. Menopause. 2013;20:8–14. doi: 10.1097/gme.0b013e3182611787. [DOI] [PMC free article] [PubMed] [Google Scholar]