Abstract
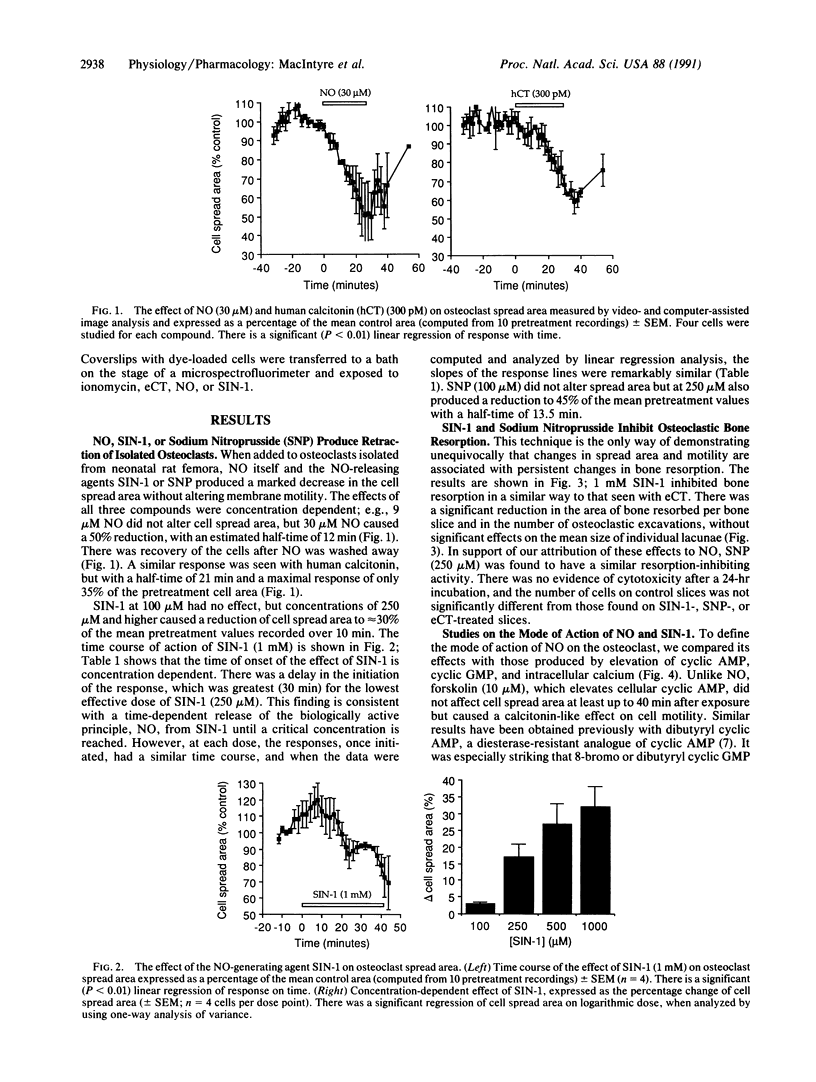
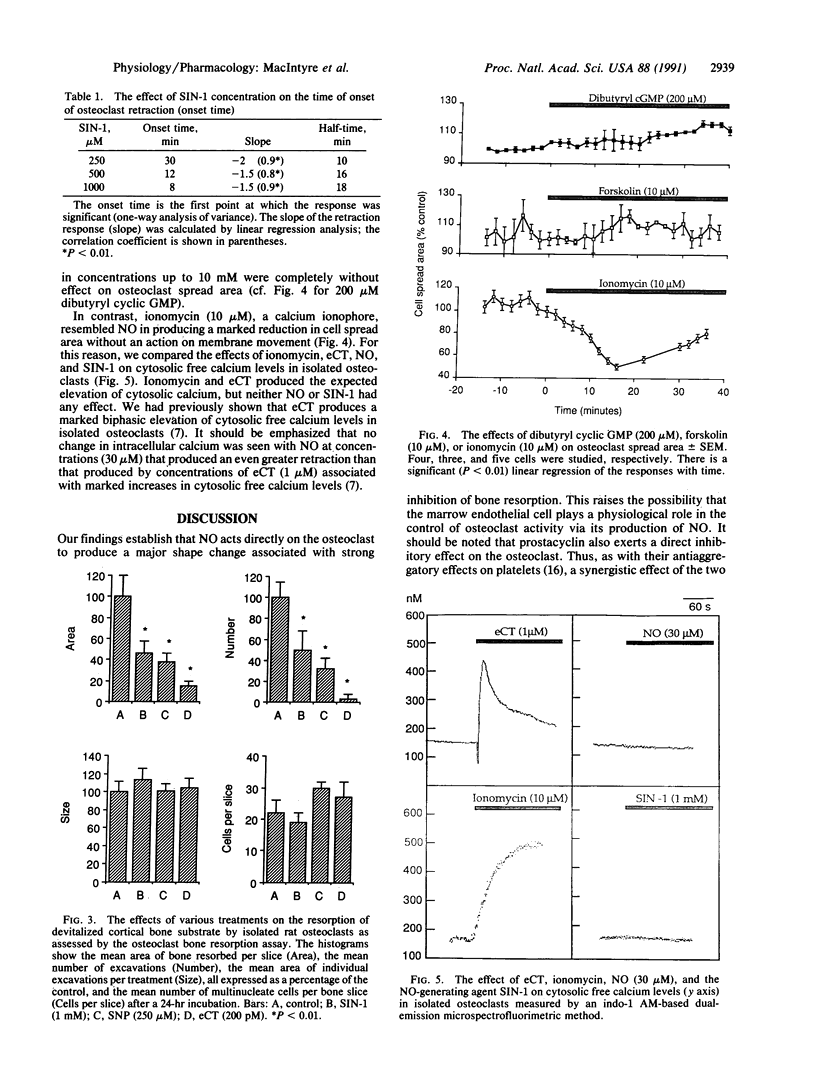
The osteoclast is unique in its ability to resorb bone, and excessive osteoclastic activity has been implicated in osteoporosis, Paget disease of bone, rheumatoid arthritis, and the growth of metastases in bone. The activity of this cell is controlled by the main circulating inhibitor, calcitonin, in association with locally produced modulators. We show that nitric oxide (NO) may be an important member of the latter group. NO is produced by the vascular endothelium and nervous system and is involved in both neurotransmission and the regulation of blood pressure. However, our results show that the autocoid is also a potent inhibitor of osteoclast function. NO (30 microM) produced a decrease to approximately 50% of the original osteoclast spread area. Similar effects were also produced by 3-morpholinosydnonimine or sodium nitroprusside, reagents that spontaneously release NO. These shape changes were associated with a reduction of bone resorption after a 24-hr incubation of isolated osteoclasts on devitalized bone slices. NO is thought to act by stimulating guanylate cyclase, with a consequent increase in cyclic GMP, but a different mode of action is likely in the osteoclast since dibutyryl or 8-bromo cyclic GMP have no effect. It should be noted that calcitonin can produce similar changes in shape and activity but is associated with an increase in osteoclast intracellular calcium and cessation of membrane movement; neither of these is produced by NO, suggesting that its mode of action is different. The abundance of NO-producing endothelial cells in bone marrow and their proximity to osteoclasts suggests that marrow endothelial cells may play a physiological role in the regulation of osteoclastic activity.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Breimer L. H., MacIntyre I., Zaidi M. Peptides from the calcitonin genes: molecular genetics, structure and function. Biochem J. 1988 Oct 15;255(2):377–390. doi: 10.1042/bj2550377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brüne B., Lapetina E. G. Activation of a cytosolic ADP-ribosyltransferase by nitric oxide-generating agents. J Biol Chem. 1989 May 25;264(15):8455–8458. [PubMed] [Google Scholar]
- Chambers T. J., Horton M. A. Failure of cells of the mononuclear phagocyte series to resorb bone. Calcif Tissue Int. 1984 Sep;36(5):556–558. doi: 10.1007/BF02405365. [DOI] [PubMed] [Google Scholar]
- Chambers T. J. Osteoblasts release osteoclasts from calcitonin-induced quiescence. J Cell Sci. 1982 Oct;57:247–260. doi: 10.1242/jcs.57.1.247. [DOI] [PubMed] [Google Scholar]
- Chambers T. J. The pathobiology of the osteoclast. J Clin Pathol. 1985 Mar;38(3):241–252. doi: 10.1136/jcp.38.3.241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Datta H. K., MacIntyre I., Zaidi M. Intracellular calcium in the control of osteoclast function. I. Voltage-insensitivity and lack of effects of nifedipine, BAYK8644 and diltiazem. Biochem Biophys Res Commun. 1990 Feb 28;167(1):183–188. doi: 10.1016/0006-291x(90)91748-h. [DOI] [PubMed] [Google Scholar]
- Datta H. K., MacIntyre I., Zaidi M. The effect of extracellular calcium elevation on morphology and function of isolated rat osteoclasts. Biosci Rep. 1989 Dec;9(6):747–751. doi: 10.1007/BF01114813. [DOI] [PubMed] [Google Scholar]
- Garg U. C., Hassid A. Nitric oxide-generating vasodilators inhibit mitogenesis and proliferation of BALB/C 3T3 fibroblasts by a cyclic GMP-independent mechanism. Biochem Biophys Res Commun. 1990 Aug 31;171(1):474–479. doi: 10.1016/0006-291x(90)91417-q. [DOI] [PubMed] [Google Scholar]
- Garrett I. R., Boyce B. F., Oreffo R. O., Bonewald L., Poser J., Mundy G. R. Oxygen-derived free radicals stimulate osteoclastic bone resorption in rodent bone in vitro and in vivo. J Clin Invest. 1990 Mar;85(3):632–639. doi: 10.1172/JCI114485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katsuki S., Arnold W., Mittal C., Murad F. Stimulation of guanylate cyclase by sodium nitroprusside, nitroglycerin and nitric oxide in various tissue preparations and comparison to the effects of sodium azide and hydroxylamine. J Cyclic Nucleotide Res. 1977 Feb;3(1):23–35. [PubMed] [Google Scholar]
- Lancaster J. R., Jr, Hibbs J. B., Jr EPR demonstration of iron-nitrosyl complex formation by cytotoxic activated macrophages. Proc Natl Acad Sci U S A. 1990 Feb;87(3):1223–1227. doi: 10.1073/pnas.87.3.1223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moonga B. S., Moss D. W., Patchell A., Zaidi M. Intracellular regulation of enzyme secretion from rat osteoclasts and evidence for a functional role in bone resorption. J Physiol. 1990 Oct;429:29–45. doi: 10.1113/jphysiol.1990.sp018242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radomski M. W., Palmer R. M., Moncada S. The anti-aggregating properties of vascular endothelium: interactions between prostacyclin and nitric oxide. Br J Pharmacol. 1987 Nov;92(3):639–646. doi: 10.1111/j.1476-5381.1987.tb11367.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomson B. M., Mundy G. R., Chambers T. J. Tumor necrosis factors alpha and beta induce osteoblastic cells to stimulate osteoclastic bone resorption. J Immunol. 1987 Feb 1;138(3):775–779. [PubMed] [Google Scholar]
- Vane J. R., Anggård E. E., Botting R. M. Regulatory functions of the vascular endothelium. N Engl J Med. 1990 Jul 5;323(1):27–36. doi: 10.1056/NEJM199007053230106. [DOI] [PubMed] [Google Scholar]
- Yang J. T., Saxton W. M., Stewart R. J., Raff E. C., Goldstein L. S. Evidence that the head of kinesin is sufficient for force generation and motility in vitro. Science. 1990 Jul 6;249(4964):42–47. doi: 10.1126/science.2142332. [DOI] [PubMed] [Google Scholar]
- Zaidi M. "Calcium receptors" on eukaryotic cells with special reference to the osteoclast. Biosci Rep. 1990 Dec;10(6):493–507. doi: 10.1007/BF01116610. [DOI] [PubMed] [Google Scholar]
- Zaidi M., Chambers T. J., Bevis P. J., Beacham J. L., Gaines Das R. E., MacIntyre I. Effects of peptides from the calcitonin genes on bone and bone cells. Q J Exp Physiol. 1988 Jul;73(4):471–485. doi: 10.1113/expphysiol.1988.sp003168. [DOI] [PubMed] [Google Scholar]
- Zaidi M., Datta H. K., Moonga B. S., MacIntyre I. Evidence that the action of calcitonin on rat osteoclasts is mediated by two G proteins acting via separate post-receptor pathways. J Endocrinol. 1990 Sep;126(3):473–481. doi: 10.1677/joe.0.1260473. [DOI] [PubMed] [Google Scholar]
- Zaidi M., Datta H. K., Patchell A., Moonga B., MacIntyre I. 'Calcium-activated' intracellular calcium elevation: a novel mechanism of osteoclast regulation. Biochem Biophys Res Commun. 1989 Sep 29;163(3):1461–1465. doi: 10.1016/0006-291x(89)91143-1. [DOI] [PubMed] [Google Scholar]
- Zaidi M. Modularity of osteoclast behaviour and "mode-specific" inhibition of osteoclast function. Biosci Rep. 1990 Dec;10(6):547–556. doi: 10.1007/BF01116615. [DOI] [PubMed] [Google Scholar]