Abstract
Purpose
The goals of this study were to (1) investigate the in vivo elongation behavior of the posterior cruciate ligament (PCL) during running in the uninjured knee; and (2) evaluate changes in PCL elongation during running after anatomic or non-anatomic anterior cruciate ligament (ACL) reconstruction.
Methods
Seventeen unilateral ACL-injured subjects were recruited after undergoing anatomic (n=9) or non-anatomic (n=8) ACL reconstruction. Bilateral high-resolution CT scans were obtained to produce 3D models. Anterolateral (AL) and posteromedial (PM) bundles insertion sites of the PCL were identified on the 3D CT scan reconstructions. Dynamic knee function was assessed during running using a Dynamic Stereo X-ray (DSX) system. The lengths of the AL and PM bundles were estimated from late swing through mid-stance. The contralateral knees served as normal controls.
Results
Control knees demonstrated a slight decrease in AL bundle, and a significant decrease in PM bundle length following foot strike. Length and elongation patterns of the both bundles of the PCL in the anatomic ACL reconstruction group were similar to the controls. However, the change in dynamic PCL length was significantly greater in the non-anatomic group than in the anatomic reconstruction group after foot strike (P<0.05).
Conclusion
The AL bundle length decreased slightly and the PM bundle length significantly decreased after foot strike during running in uninjured knees. Anatomic ACL reconstruction maintained normal PCL elongation patterns more effectively than non-anatomic ACL reconstruction during high-demand, functional loading. These results support the use of anatomic ACL reconstruction to achieve normal knee function in high-demand activities.
Level of evidence
Case-control study, Level III
Keywords: in vivo knee kinematics, anterior cruciate ligament (ACL), posterior cruciate ligament (PCL), soft tissue biomechanics, Anterior cruciate ligament reconstruction
Introduction
The in vivo elongation of the anterolateral (AL) and posteromedial (PM) bundles of the posterior cruciate ligament (PCL) during high-demand activities (such as running) has not previously been reported. Additionally, the effects of anterior cruciate ligament (ACL) injury and reconstruction on PCL function are unknown.
The cruciate ligaments are the primary stabilizers to anterior-posterior translation, and important secondary stabilizers to rotation of the knee. The PCL consists of the distinct AL and PM bundles, each with unique insertion site characteristics and tensions during knee motion [8,23,31,34]. Cadaveric studies have demonstrated that the AL bundle is relatively loose in knee extension and becomes progressive tensioned during knee flexion, while the PM bundle is tight in extension and loosens during knee flexion [1,6]. In vivo studies during quasi-static, low-demand activities have reported similar reciprocal bundle tightening and loosening during knee motion [20]. Additional in vivo studies have reported significant changes in knee kinematics following PCL injury during static and low-demand dynamic conditions, such as slow-speed gait [10,12,18,24]. A previous in vivo study demonstrated changes in tibio-femoral kinematics during running after PCL reconstruction surgery [26]. However, in vivo PCL elongation patterns during sports-related activities (such as running) have not been reported.
The cruciate ligaments function synergistically to guide knee motion, and contact and force transmission between the ACL and PCL have been reported for both uninjured and ACL-reconstructed knees [3,16,21,33,36]. Thus, it is reasonable to suggest that PCL function may be altered after ACL reconstruction, particularly for grafts that are positioned non-anatomically. Biomechanical studies suggest that anatomical reconstruction of the ACL is superior to non-anatomical reconstruction for restoring native knee stability [19,35,43,44,46]. However, the relative impact of the different procedures on PCL behavior has yet to be characterized (particularly during
high-demand, functional activities). A procedure to correct for damage to one structure within the knee that produces abnormal loading to another may not adequately protect the joint from further damage. Thus, understanding in vivo changes in biomechanical function of the PCL after ACL reconstruction can provide additional insights into the effectiveness of surgical treatment for restoring normal joint function and the relative merits of anatomic vs. non-anatomic graft placement, and may provide additional guidance for surgical decision-making.
The purpose of this study was to investigate the in vivo PCL elongation during high-demand running activities, and to compare effects of anatomical and non-anatomical ACL reconstruction techniques on PCL elongation. It was hypothesized that the in vivo PCL elongation patterns would vary during high-demand activities from the patterns have been previously reported for static, low-demand conditions. It was further hypothesized that ACL reconstruction would alter the elongation patterns of the native PCL, with non-anatomic ACL reconstruction demonstrating greater alterations compared to anatomical reconstruction methods.
Materials and Methods
Seventeen unilateral ACL injured subjects 6 to 12 months after surgical reconstruction were recruited for this study. Institutional Review Board approval and informed consent were obtained prior to subject enrollment. Subjects underwent either anatomic ACL reconstruction (n=9, 38±12 years old, range 21-53 years; 3 male and 6 female, average BMI=23.6, 4 SB and 5 DB ACL) or non-anatomic ACL reconstruction (n=8, 33±8 years old, range 23-44 years; 4 male and 4 female, average BMI=23.7, 3 SB and 5 DB ACL). Sports injury was the most common reason for this group of patients. Two senior surgeons preformed the reconstruction surgery used allograft in all cases of anatomic group and 6 cases of non-anatomic group, hamstring was used in the left 2 cases of non-anatomic group. All subjects underwent postoperative rehabilitation following the standardized protocols used in the department of orthopedic surgery, University of Pittsburgh. Bilateral high-resolution computerized tomography (CT) scans were obtained to produce 3D subject-specific bone models, and determination of anatomic versus non-anatomic reconstruction was based on the tunnel position assessed using CT. An anatomic coordinate system was employed to measure the tunnel position of the femur and tibia [9]. Subjects with femoral and tibia graft tunnels placed outside of the previously described native ACL footprints were included into the non-anatomic ACL group (Figure 1). [9,15,17] Based on previous studies [42,46], subjects with high AM tunnels were also included into the non-anatomic group (Figure 1).
Figure 1.
Top: The length of AL and PM bundles was determined by measuring the three-dimensional distance from the tibial to femoral insertions, as shown in this example of a typical subject (heel strike: A and B; 0.1 s after heel strike: C and D). Relative length of the graft is indicated by color (blue/green/yellow/ orange/red, from shortest to longest).
Bottom: 3D CT reconstruction of the distal femur demonstrating ACL femoral tunnel position: anatomic SB (E), anatomic DB (F), non-anatomic high tunnel SB (G) and non-anatomic vertical/high AM tunnel DB (H).
Knee kinematics were determined utilizing a previously described dynamic stereo radiography (DSX) technique [2]. Three running trials were acquired for each knee during running on a dual-belt instrumented treadmill (Bertec Corp, Columbus, OH, USA) set at a 10% decline and a speed of 2.5 m/s. The subjects were positioned so that their knees were centered within a biplane radiographic system surrounding the treadmill. Ground reaction forces during running were measured and recorded to determine the loading phase of each knee. High-speed stereo radiographic images were captured at 150 frames/second to evaluate knee kinematics during running. This measurement system has been used in prior studies of knee ligament function, and is capable of evaluating in vivo joint motion with an accuracy of within ± 0.1 mm [39,40].
The osseous geometry and landmarks of the femoral and tibial PCL insertions have been previously reported, and the application of a CT model to determine the center of each insertion has also been described [20,28,30,38]. The centroids of the individual AL and PM bundle footprints were identified on the previously obtained 3D CT scans of the femur and tibia. Briefly, on the femur side Lopes et al. [28] have extensively studied the location of the native PCL insertion site. Based on their study, the AL and PM centers were located between the articular cartilage edge and "medial intercondylar ridge". The centroids of AL and PM insertion site were identified on the femur side (Figure 2 A). Based on a previous study [38], tibial PCL insertion sites were located at the posterior intercondylar fossa between the tibial plateaus; the osseous landmarks "anterolateral slope" and "posteromedial slope" were used to locate the AL and PM bundles respectively (Figure 2B). AL and PM bundle lengths were calculated as the 3D Euclidean distances between the estimated femoral and tibial insertion site centroids. AL and PM bundles lengths were assessed from 0.1 sec before to 0.1 sec after heel strike; capturing both swing phase and initial loading response.
Figure 2.
3D CT models of the femur (A) and tibia (b), showing the landmarks used as reference for estimating insertion sites of the AL and PM bundles.
This study was approved by the IRB of the University of Pittsburgh. (PRO08030126)
Statistical analysis
Independent-samples T-tests were used to compare: the AL and PM bundle lengths among the ACL-reconstructed knees and contralateral, ACL-intact controls; the bundle length difference between the pre-heel strike and post-heel strike states; and the change in bundle length of each group. All data were analyzed with SPSS v16 software, (SPSS, Chicago, IL), with the level of significance set a priori at P<0.05. Post hoc power of these statistical tests were calculated using G*Power 3.1.9.2 (Franz Paul, Kiel, Germany). Based on the results of the independent t-test for PM-bundle length changes between the study group and the controls, an effect size of 1.2 was calculated. With this effect size and an alpha of 0.05 a power of 0.96 was achieved.
Results
In the control (uninjured) knees, AL bundle length remained relatively unchanged (relative to the length just prior to footstrike) throughout early stance (27.6 ± 3.2 mm to 26.3 ± 3.5 mm, n.s.). There was a significant decrease in the PM bundle length after footstrike in the control knees (Figure 3; 30.9 ± 3.3 mm to 27.4 ± 3.5 mm, p <0.05 for t=0.05-0.1 s). There were no significant length changes of the AL or PM bundles from just prior to footstrike though early stance in either the anatomically or non-anatomically ACL-reconstructed knees.
Figure 3.
After heel strike (time=0) a decrease in the length of the of AL and PM bundles was observed for the control knees (*P<0.05 for PM bundle).
In the non-anatomic reconstruction group, PCL bundle lengths were significantly different between ACL-reconstructed and contralateral (uninjured) knees from 0 to 0.07 seconds following foot strike for the AL bundle and from 0 to 0.06 seconds following foot strike for the PM bundle (Table 1; p<0.05). There were no significant differences in PCL bundle lengths between ACL-reconstructed and contralateral (uninjured knees) in the anatomically ACL-reconstructed group.
Table 1.
Length differences for the anterior-lateral (AL) and posterior-medial (PM) bundles of the PCL between ACL-reconstructed and contralateral (uninjured) knees following foot strike (ACLR-contralateral; mean±standard error).
| PCL Length Difference, ACL-R vs. Uninjured Knees (mm) | ||||
|---|---|---|---|---|
| Time(sec) | AL anatomic | AL non-anatomic | PM anatomic | PM non-anatomic |
| 0.00 | 0.4±0.3 | −1.2±0.5* | 0.1±0.2 | −1.4±0.6* |
| 0.01 | 0.2±0.3 | −1.4±0.5* | 0.1 ±0.2 | −1.6±0.5* |
| 0.02 | 0.0±0.3 | −1.4±0.5* | 0.0±0.2 | −1.5±0.5* |
| 0.03 | −0.1±0.4 | −1.5±0.5* | 0.1±0.2 | −1.5±0.6* |
| 0.04 | −0.2±0.4 | −1.8±0.6* | −0.1±0.4 | −1.8±0.6* |
| 0.05 | −0.3±0.3 | −1.9±0.6* | −0.1±0.5 | −1.8±0.6* |
| 0.06 | −0.3±0.2 | −1.9±0.6* | −0.1±0.3 | −1.7±0.6* |
| 0.07 | −0.4±0.2 | −1.9±0.6* | −0.1±0.3 | −1.5±0.6 |
| 0.08 | −0.5±0.3 | −1.7±0.6 | −0.3±0.3 | −1.2±0.6 |
| 0.09 | −0.4±0.3 | −1.5±0.5 | −0.4±0.4 | −1.1±0.5 |
| 0.10 | −0.2±0.3 | −1.5±0.6 | −0.5±0.5 | −0.9±0.4 |
Asterisks identify significant differences between ACL-R and control (p<0.05).
Discussion
The most significant finding of this study was the significant difference in PCL elongation behavior between uninjured knees and ACL-reconstructed knees with non-anatomical tunnel placements. This difference did not exist for knees reconstructed using more anatomically placed graft tunnels. Thus, anatomic ACL reconstruction maintained normal PCL elongation patterns more effectively than non-anatomic ACL reconstruction during a high-demand physical activity (running).
Current knowledge of the biomechanical functions of the posterior cruciate ligament (PCL) is predominantly the result of cadaveric studies [1,7,11,32]. Ahmad et al. demonstrated that the posterior tibial restraint of the PCL was greatest at the knee flexion angles where each PCL bundle reached its maximal length, with the greatest contribution of the PM bundle occurring between 0° to 45° of knee flexion and the greatest contribution of the AL bundle occurring beyond 45 degrees of knee flexion [1]. Markolf et al. observed only a small increase in posterior tibial translation following isolated transection of the PM bundle, concluding that the PM bundle provides a relatively minor role in restraining posterior tibial translation. Their experimental design was restricted to anterior-posterior laxity testing, and may have underestimated the contribution of the PM bundle to rotational stability [29].
In vivo studies of PCL biomechanics can provide a more clinically meaningful assessment of the true role of the PCL for providing AP and rotational stability during functional activities. There is currently a paucity of in vivo data on PCL behavior in the current literature. Previous authors have attempted to characterize PCL function in vivo with static images obtained during a weight-bearing lunge [23,25,27,34,41]. They demonstrated elongation of the PCL with increasing knee flexion angles, and significantly altered knee kinematics with PCL injury/insufficiency. The present study focused on the in vivo role of the PCL during a common but high-demand activity that would be applicable to a wide population, regardless of athletic status. During this high-load activity, PCL length decreased after foot strike, contrasting previous studies [23] demonstrating PCL elongation during a lunge maneuver. Those different patterns may relate to high demanding quad/hamstring activation patterns and ground reaction force, which may be different from static or low demanding activities. High demanding activity may result in femur posterior translation and contribute to kinematic changes of the knee. Future studies focusing on the mechanism under high demanding activity should be conducted. This finding may demonstrate that PCL elongation and function is dependent on activity, where rapid high impact loads may lead to shortening of the PCL in vivo while static loading of the knee may result in PCL elongation with increasing knee flexion. Possible reasons for this include the knee flexion angle during maximum load and quadriceps activation patterns during each activity. Further investigation may be able to delineate the causes for these findings.
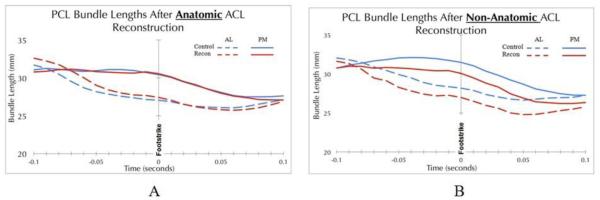
The ACL and PCL play a synergistic role in providing stability to anterior-posterior translation and rotation of the knee during activity [11], and the patterns of instability following isolated or combined injuries to the cruciate ligaments have been well-defined in the literature [1,5,12,13,22,25,37,41]. Biomechanical studies have demonstrated the improved ability of an anatomic ACL reconstruction to more closely restore the native stability of the knee compared to non-anatomic techniques [46], although differences between anatomic and non-anatomic ACL reconstruction techniques were small in some studies [4,45]. Efforts to more closely recreate the ACL anatomy during reconstruction has lead to the development of single and double bundle anatomic techniques, although prospective clinical studies have yet to demonstrate significant clinical differences, as both methods have achieved excellent results [14]. A significant amount of research has focused on the optimal treatment of ACL injury and reconstruction, but little is known about the effects of ACL reconstruction on the surrounding structures of the knee. Iriuchishima and colleagues demonstrated that high femoral tunnel placement during ACL reconstruction in porcine knees resulted in elevated impingement pressures against the neighboring PCL. They concluded that anatomical ACL reconstruction avoids impingement against the PCL while more closely restoring native knee stability [16]. The results in this study demonstrated that, in uninjured knees, the AL bundle is nearly isometric, while the PM bundle decreases in length from late swing through early stance phase of running. While the PCL bundle length change patterns were qualitatively similar in ACL-reconstructed knees (especially for the anatomically reconstructed group; Figure 4), no significant changes in length over time were identified for affected knees in either ACL reconstruction group. This may be due to the limited sample size, or possibly greater subject-to-subject variability in the reconstructed knees.
Figure 4.
Length and elongation patterns of the AL and PM bundles of the PCL in the ACL-reconstructed knees were similar to the contralateral (uninjured) knees in the anatomical ACL patients (P>0.05). However, in the non-anatomical ACL group (B), AL and PM bundle lengths and elongation patterns changed severely following foot strike (P>0.05).
While PCL elongation patterns were similar between limbs after anatomic ACL reconstruction, significant differences in PCL lengths between ACL-reconstructed and contralateral (uninjured) knees were found through out most of early stance in the non-anatomical reconstruction group for both AL and PM bundles. Thus, anatomic ACL reconstruction better maintains the normal PCL elongation patterns during running. This may be due to the improved AP stability of anatomic reconstructions, resulting in less pathological translation and PCL elongation during dynamic activity. With respect to the synergistic functions of the ACL and PCL, this finding supports the use of anatomic ACL reconstruction to more accurately restore normal knee kinematics.
Several limitations exist in this study. These results are based on measurements of the PCL calculated indirectly from points placed within the center of the estimated native PCL insertion sites utilizing modern anatomic understanding as a reference. However, individual insertion site variations may not be completely accounted for with this technique. Another limitation is the relatively small sample size; larger sample sizes in the subgroups might have enabled detection of subtle but significant differences in PCL elongation. Additionally, the changes in PCL bundle length found during running were based on the 3D Euclidean distance between the estimated femoral and tibial insertion site centroids, and did not account for any torsion, bending, or wrapping of the ligament around the ACL, femoral condyles, or other soft tissues.
In this study we found that the length of both the AL and PM bundles decreased after heel strike during running. This elongation pattern varies from previously reported in-vivo studies, which have demonstrated increasing PCL elongation with increasing knee flexion angle [23], and may demonstrate that the elongation pattern within the PCL fibers is highly activity-dependent. This has possible implications for selecting appropriate rehabilitation activities after PCL injury. Differences in PCL behavior in the non-anatomically reconstructed knees (but not after anatomical reconstruction) provides additional evidence that ACL graft placement has a complex, joint-wide effect on dynamic knee function. Anatomic ACL reconstruction is recommended for restoring normal PCL kinematics and dynamic knee function during high-demand activities after ACL injury.
Conclusion
Elongation behavior of the PCL during a demanding activity (running) was investigated after either anatomical or non-anatomical ACL reconstruction. In uninjured knees, the AL bundle length of the PCL length of the PM bundle significantly decreased after foot strike, while the AL bundle length remained relatively constant. Anatomic ACL reconstruction maintained normal PCL elongation patterns more effectively than non-anatomic ACL reconstruction during high-demand, functional loading.
Footnotes
The work was performed at the Department of Orthopaedic Surgery, University of Pittsburgh Medical Center (UPMC), USA.
Conflict of interest
The authors declare that they have no conflict of interest.
References
- 1.Ahmad CS, Cohen ZA, Levine WN, Gardner TR, Ateshian GA, Mow VC. Codominance of the individual posterior cruciate ligament bundles. An analysis of bundle lengths and orientation. Am J Sports Med. 2003;31:221–225. doi: 10.1177/03635465030310021101. [DOI] [PubMed] [Google Scholar]
- 2.Anderst W, Zauel R, Bishop J, Demps E, Tashman S. Validation of three-dimensional model-based tibio-femoral tracking during running. Med Eng Phys. 2009;31:10–16. doi: 10.1016/j.medengphy.2008.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bersini S, Sansone V, Frigo CA. A dynamic multibody model of the physiological knee to predict internal loads during movement in gravitational field. Comput Methods Biomech Biomed Engin. 2016;19:571–579. doi: 10.1080/10255842.2015.1051972. [DOI] [PubMed] [Google Scholar]
- 4.Bohn MB, Sorensen H, Petersen MK, Søballe K, Lind M. Rotational laxity after anatomical ACL reconstruction measured by 3-D motion analysis: a prospective randomized clinical trial comparing anatomic and nonanatomic ACL reconstruction techniques. Knee Surg Sports Traumatol Arthrosc. 2015;23:3473–3481. doi: 10.1007/s00167-014-3156-5. [DOI] [PubMed] [Google Scholar]
- 5.Chen K, Yin L, Cheng L, Li C, Chen C, Yang L. In Vivo Motion of Femoral Condyles During Weight-Bearing Flexion After Anterior Cruciate Ligament Rupture Using Biplane Radiography. J Sports Sci Med. 2013;12:579–587. [PMC free article] [PubMed] [Google Scholar]
- 6.Covey CD, Sapega AA. Injuries of the posterior cruciate ligament. J Bone Joint Surg Am. 1993;75:1376–1386. doi: 10.2106/00004623-199309000-00014. [DOI] [PubMed] [Google Scholar]
- 7.Covey DC, Sapega AA, Sherman GM. Testing for isometry during reconstruction of the posterior cruciate ligament. Anatomic and biomechanical considerations. Am J Sports Med. 1996;24:740–746. doi: 10.1177/036354659602400607. [DOI] [PubMed] [Google Scholar]
- 8.Edwards A, Bull AM, Amis AA. The attachments of the fiber bundles of the posterior cruciate ligament: an anatomic study. Arthroscopy. 2007;23:284–290. doi: 10.1016/j.arthro.2006.11.005. [DOI] [PubMed] [Google Scholar]
- 9.Forsythe B, Kopf S, Wong AK, Martins CA, Anderst W, Tashman S, Fu FH. The location of femoral and tibial tunnels in anatomic double-bundle anterior cruciate ligament reconstruction analyzed by three-dimensional computed tomography models. J Bone Joint Surg Am. 2010;92:1418–1426. doi: 10.2106/JBJS.I.00654. [DOI] [PubMed] [Google Scholar]
- 10.Gill TJ, Van de Velde SK, Wing DW, Oh LS, Hosseini A, Li G. Tibiofemoral and patellofemoral kinematics after reconstruction of an isolated posterior cruciate ligament injury: in vivo analysis during lunge. Am J Sports Med. 2009;37:2377–2385. doi: 10.1177/0363546509341829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Girgis FG, Marshall JL, Monajem A. The cruciate ligaments of the knee joint. Anatomical, functional and experimental analysis. Clin Orthop Relat Res. 1975;106:216–231. doi: 10.1097/00003086-197501000-00033. [DOI] [PubMed] [Google Scholar]
- 12.Goyal K, Tashman S, Wang JH, Li K, Zhang X, Harner C. In vivo analysis of the isolated posterior cruciate ligament-deficient knee during functional activities. Am J Sports Med. 2012;40:777–785. doi: 10.1177/0363546511435783. [DOI] [PubMed] [Google Scholar]
- 13.Hiraga Y, Ishibashi Y, Tsuda E, Toh HT. Biomechanical comparison of posterior cruciate ligament reconstruction techniques using cyclic loading tests. Knee Surg Sports Traumatol Arthrosc. 2006;14:13–19. doi: 10.1007/s00167-005-0633-x. [DOI] [PubMed] [Google Scholar]
- 14.Hussein M, van Eck CF, Cretnik A, Dinevski D, Fu FH. Individualized Anterior Cruciate Ligament Surgery: A Prospective Study Comparing Anatomic Single- and Double-Bundle Reconstruction. Am J Sports Med. 2012;40:1781–1788. doi: 10.1177/0363546512446928. [DOI] [PubMed] [Google Scholar]
- 15.Iriuchishima T, Ingham SJ, Tajima G, Horaguchi T, Saito A, Tokuhashi Y, Van Houten AH, Aerts MM, Fu FH. Evaluation of the tunnel placement in the anatomical double-bundle ACL reconstruction: a cadaver study. Knee Surg Sports Traumatol Arthrosc. 2010;18:1226–1231. doi: 10.1007/s00167-010-1128-y. [DOI] [PubMed] [Google Scholar]
- 16.Iriuchishima T, Tajima G, Ingham SJ, Shirakura K, Fu FH. PCL to graft impingement pressure after anatomical or non-anatomical single-bundle ACL reconstruction. Knee Surg Sports Traumatol Arthrosc. 2012;20:964–969. doi: 10.1007/s00167-011-1680-0. [DOI] [PubMed] [Google Scholar]
- 17.Iriuchishima T, Tajima G, Shirakura K, Morimoto Y, Kubomura T, Horaguchi T, Fu FH. In vitro and in vivo AM and PL tunnel positioning in anatomical double bundle anterior cruciate ligament reconstruction. Arch Orthop Trauma Surg. 2011;131:1085–1090. doi: 10.1007/s00402-011-1308-3. [DOI] [PubMed] [Google Scholar]
- 18.Iwata S, Suda Y, Nagura T, Matsumoto H, Otani T, Toyama Y. Dynamic instability during stair descent in isolated PCL-deficient knees: what affects abnormal posterior translation of the tibia in PCL-deficient knees? Knee Surg Sports Traumatol Arthrosc. 2007;15:705–711. doi: 10.1007/s00167-006-0270-z. [DOI] [PubMed] [Google Scholar]
- 19.Jarvela T. Double-bundle versus single-bundle anterior cruciate ligament reconstruction: a prospective, randomize clinical study. Knee Surg Sports Traumatol Arthrosc. 2007;15:500–507. doi: 10.1007/s00167-006-0254-z. [DOI] [PubMed] [Google Scholar]
- 20.Jeong WS, Yoo YS, Kim DY, Shetty NS, Smolinski P, Logishetty K, Ranawat A. An analysis of the posterior cruciate ligament isometric position using an in vivo 3-dimensional computed tomography-based knee joint model. Arthroscopy. 2010;26:1333–1339. doi: 10.1016/j.arthro.2010.02.016. [DOI] [PubMed] [Google Scholar]
- 21.Kropf EJ, Shen W, van Eck CF, Musahl V, Irrgang JJ, Fu FH. ACL-PCL and intercondylar notch impingement: magnetic resonance imaging of native and double-bundle ACL-reconstructed knees. Knee Surg Sports Traumatol Arthrosc. 2012;21:720–725. doi: 10.1007/s00167-012-2052-0. [DOI] [PubMed] [Google Scholar]
- 22.Kopf S, Musahl V, Bignozzi S, Irrgang JJ, Zaffagnini S, Fu FH. In vivo kinematic evaluation of anatomic double-bundle anterior cruciate ligament reconstruction. Am J Sports Med. 2014;42:2172–2177. doi: 10.1177/0363546514538958. [DOI] [PubMed] [Google Scholar]
- 23.Li G, DeFrate LE, Sun H, Gill TJ. In vivo elongation of the anterior cruciate ligament and posterior cruciate ligament during knee flexion. Am J Sports Med. 2004;32:1415–1420. doi: 10.1177/0363546503262175. [DOI] [PubMed] [Google Scholar]
- 24.Li G, Most E, DeFrate LE, Suggs JF, Gill TJ, Rubash HE. Effect of the posterior cruciate ligament on posterior stability of the knee in high flexion. J Biomech. 2004;37:779–783. doi: 10.1016/j.jbiomech.2003.09.031. [DOI] [PubMed] [Google Scholar]
- 25.Li G, Papannagari R, Li M, Bingham J, Nha KW, Allred D, Gill T. Effect of posterior cruciate ligament deficiency on in vivo translation and rotation of the knee during weightbearing flexion. Am J Sports Med. 2008;36:474–479. doi: 10.1177/0363546507310075. [DOI] [PubMed] [Google Scholar]
- 26.Lim BO, Shin HS, Lee YS. Biomechanical comparison of rotational activities between anterior cruciate ligament- and posterior cruciate ligament-reconstructed patients. Knee Surg Sports Traumatol Arthrosc. 2015;23:1231–1238. doi: 10.1007/s00167-014-2959-8. [DOI] [PubMed] [Google Scholar]
- 27.Logan M, Williams A, Lavelle J, Gedroyc W, Freeman M. The effect of posterior cruciate ligament deficiency on knee kinematics. Am J Sports Med. 2004;32:1915–1922. doi: 10.1177/0363546504265005. [DOI] [PubMed] [Google Scholar]
- 28.Lopes OV, Jr., Ferretti M, Shen W, Ekdahl M, Smolinski P, Fu FH. Topography of the femoral attachment of the posterior cruciate ligament. J Bone Joint Surg Am. 2008;90:249–255. doi: 10.2106/JBJS.G.00448. [DOI] [PubMed] [Google Scholar]
- 29.Markolf KL, Feeley BT, Tejwani SG, Martin DE, McAllister DR. Changes in knee laxity and ligament force after sectioning the posteromedial bundle of the posterior cruciate ligament. Arthroscopy. 2006;22:1100–1106. doi: 10.1016/j.arthro.2006.05.018. [DOI] [PubMed] [Google Scholar]
- 30.Moorman CT, 3rd, Murphy Zane MS, Bansai S, Cina S, Wickiewicz TL, Warren RF, Kaseta MK. Tibial insertion of the posterior cruciate ligament: a sagittal plane analysis using gross, histologic, and radiographic methods. Arthroscopy. 2008;24:269–275. doi: 10.1016/j.arthro.2007.08.032. [DOI] [PubMed] [Google Scholar]
- 31.Morgan CD, Kalman VR, Grawl DM. The anatomic origin of the posterior cruciate ligament: where is it? Reference landmarks for PCL reconstruction. Arthroscopy. 1997;13:325–331. doi: 10.1016/s0749-8063(97)90029-3. [DOI] [PubMed] [Google Scholar]
- 32.Mutnal A, Leo BM, Vargas L, Colbrunn RW, Butler RS, Uribe JW. Biomechanical analysis of posterior cruciate ligament reconstruction with aperture femoral fixation. Orthopedics. 2015;38:9–16. doi: 10.3928/01477447-20150105-50. [DOI] [PubMed] [Google Scholar]
- 33.Nishimori M, Sumen Y, Sakaridani K, Nakamura M. An evaluation of reconstructed ACL impingement on PCL using MRI. Magn Reson Imaging. 2007;25:722–726. doi: 10.1016/j.mri.2006.10.002. [DOI] [PubMed] [Google Scholar]
- 34.Papannagari R, DeFrate LE, Nha KW, Moses JM, Moussa M, Gill TJ, Li G. Function of posterior cruciate ligament bundles during in vivo knee flexion. Am J Sports Med. 2007;35:1507–1512. doi: 10.1177/0363546507300061. [DOI] [PubMed] [Google Scholar]
- 35.Petersen W, Tretow H, Weimann A, Herbort M, Fu FH, Raschke M, Zantop T. Biomechanical evaluation of two techniques for double-bundle anterior cruciate ligament reconstruction: one tibial tunnel versus two tibial tunnels. Am J Sports Med. 2007;35:228–234. doi: 10.1177/0363546506294468. [DOI] [PubMed] [Google Scholar]
- 36.Rosvold JM, Atarod M, Heard BJ, O’Brien EJ, Frank CB, Shrive NG. Ligament and meniscus loading in the ovine stifle joint during normal gait. Knee. 2016;23:70–77. doi: 10.1016/j.knee.2015.09.013. [DOI] [PubMed] [Google Scholar]
- 37.Saiegh YA, Suero EM, Guenther D, Hawi N, Decker S, Krettek C, Citak M, Omar M. Sectioning the anterolateral ligament did not increase tibiofemoral translation or rotation in an ACL-deficient cadaveric model. Knee Surg Sports Traumatol Arthrosc. 2015 doi: 10.1007/s00167-015-3787-1. Doi:10.1007/s00167-015-3787-1. [DOI] [PubMed] [Google Scholar]
- 38.Tajima G, Nozaki M, Iriuchishima T, Ingham SJ, Shen W, Smolinski P, Fu FH. Morphology of the tibial insertion of the posterior cruciate ligament. J Bone Joint Surg Am. 2009;91:859–866. doi: 10.2106/JBJS.H.00991. [DOI] [PubMed] [Google Scholar]
- 39.Tashman S, Anderst W. In-vivo measurement of dynamic joint motion using high speed biplane radiography and CT: application to canine ACL deficiency. J Biomech Eng. 2003;125:238–245. doi: 10.1115/1.1559896. [DOI] [PubMed] [Google Scholar]
- 40.Tashman S, Kolowich P, Collon D, Anderson K, Anderst W. Dynamic function of the ACL-reconstructed knee during running. Clin Orthop Relat Res. 2007;454:66–73. doi: 10.1097/BLO.0b013e31802bab3e. [DOI] [PubMed] [Google Scholar]
- 41.Van de Velde SK, Gill TJ, Li G. Dual fluoroscopic analysis of the posterior cruciate ligament-deficient patellofemoral joint during lunge. Med Sci Sports Exerc. 2009;41:1198–1205. doi: 10.1249/MSS.0b013e3181981eb5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Xu Y, Liu J, Kramer S, Martins C, Kato Y, Linde-Rosen M, Smolinski P, Fu FH. Comparison of in situ forces and knee kinematics in anteromedial and high anteromedial bundle augmentation for partially ruptured anterior cruciate ligament. Am J Sports Med. 2011;39:272–278. doi: 10.1177/0363546510383479. [DOI] [PubMed] [Google Scholar]
- 43.Yagi M, Kuroda R, Nagamune K, Yoshiya S, Kurosaka M. Double-bundle ACL reconstruction can improve rotational stability. Clin Orthop Relat Res. 2007;454:100–107. doi: 10.1097/BLO.0b013e31802ba45c. [DOI] [PubMed] [Google Scholar]
- 44.Yagi M, Wong EK, Kanamori A, Debski RE, Fu FH, Woo SL. Biomechanical analysis of an anatomic anterior cruciate ligament reconstruction. Am J Sports Med. 2002;30:660–666. doi: 10.1177/03635465020300050501. [DOI] [PubMed] [Google Scholar]
- 45.Zaffagnini S, Marcheggiani Muccioli GM, Sgnorelli C, Lopomo N, Grassi A, Bonanzinga T, Nitri M, Marcacci M. Anatomic and nonanatomic double-bundle anterior cruciate ligament reconstruction : an in vivo kinematic analysis. Am J Sports Med. 2014;42:708–715. doi: 10.1177/0363546513519070. [DOI] [PubMed] [Google Scholar]
- 46.Zantop T, Diermann N, Schumacher T, Schanz S, Fu FH, Petersen W. Anatomical and nonanatomical double-bundle anterior cruciate ligament reconstruction: importance of femoral tunnel location on knee kinematics. Am J Sports Med. 2008;36:678–685. doi: 10.1177/0363546508314414. [DOI] [PubMed] [Google Scholar]