Abstract
Background
Alcohol consumption is associated with increased risk of numerous cancers, but existing evidence for an association with melanoma is equivocal. No study has evaluated the association with different anatomic locations of melanoma.
Methods
We used data from three large prospective cohort studies to investigate whether alcohol intake was associated with risk of melanoma. Alcohol intake was assessed repeatedly by food-frequency questionnaires. A Cox proportional hazards model was used to calculate multivariate-adjusted hazard ratios (HRs).
Results
A total of 1,374 cases of invasive melanoma were documented during 3,855,706 person-years of follow-up. There was an association between higher alcohol intake and incidence of invasive melanoma (pooled multivariate HR 1.14; 95% confidence interval [CI]: 1.00–1.29] per drink/d, p trend = 0.04). Among alcoholic beverages, white wine consumption was associated with an increased risk of melanoma (pooled multivariate HR 1.13 [95% CI: 1.04–1.24] per drink/d, p trend <0.01) after adjusting for other alcoholic beverages. The association between alcohol consumption and melanoma risk was stronger for melanoma in relatively UV-spared sites (trunk) versus more UV-exposed sites (head, neck, or extremities). Compared to non-drinkers, the pooled multivariate-adjusted HRs for ≥20g/d of alcohol were 1.02 (95% CI: 0.64–1.62; P trend =0.25) for melanomas of the head, neck, and extremities and 1.73 (95% CI: 1.25–2.38; P trend =0.02) for melanomas of the trunk.
Conclusions
Alcohol intake was associated with a modest increase in the risk of melanoma, particularly in UV-protected sites.
Impact
These findings further support American Cancer Society Guidelines for Cancer Prevention to limit alcohol intake.
Introduction
Melanoma incidence has been rising steadily since the 1970s,(1–12) during which time Americans’ average consumption of alcoholic beverages has increased by 71 kilocalories per person per day.(13) The concurrence of these trends tentatively suggests that they may be related, and some investigators have proposed that alcohol consumption might intensify sunburn severity and thereby increase risk of melanoma.(14–15) Known predictors of melanoma risk include a personal or family history of skin cancer, presence of numerous or atypical moles, high sun sensitivity (sunburning easily, difficulty tanning, natural red or blond hair color), immunosuppression, and intermittent ultraviolet radiation (UVR) exposure. With the exception of UVR exposure, most of these are non-modifiable host factors, which would be unlikely to drive the rising incidence of melanoma.
Alcohol consumption is a modifiable lifestyle factor associated with cancers of the aerodigestive tract, liver, pancreas, colon, rectum, and breast.(16–20) Approximately 3.6% of cancers worldwide have been attributed to alcohol use.(21–22) Alcohol causes carcinogenesis via metabolism of ethanol into acetaldehyde, a Group I human carcinogen that readily forms Schiff-base adducts with DNA and cellular proteins.(23–25) These adducts cause both point mutations and deleterious DNA-protein and DNA-DNA cross-links. In addition to the endogenous conversion of ethanol into acetaldehyde, some alcoholic beverages contain carcinogenic levels of pre-existing acetaldehyde.(26–29) Such beverages may confer greater risk than others, even at similar levels of ethanol consumption.
Experimental studies in animals suggest that the clinical course of melanoma may be more aggressive in the presence of ethanol.(30–32) This has never been demonstrated in humans, and epidemiological evidence for an association between alcohol consumption and melanoma has been equivocal.(33–45) A meta-analysis of sixteen studies (fourteen case-control and two cohort) with 6,251 cases of melanoma, found that regular alcohol consumption was associated with a 20% (pooled hazard ratio (HR) 1.20; 95% confidence interval [CI] 1.06–1.37) elevated risk of melanoma compared with non-drinkers/occasional drinkers.(46) The HRs were 1.10 (95% CI: 0.96–1.26) for drinkers consuming ≤1 drink per day and 1.18 (95% CI: 1.01–1.40) for those consuming ≥1 drink per day, suggesting a dose-response relationship. However, the data largely relied on case-control studies, although the positive association was similar in cohort studies. Furthermore, six of the studies did not adjust for UVR exposure (pooled HR 1.27, 95% CI: 1.20–1.35 among these studies), and among the ten studies that adjusted for UVR exposure, the results were not statistically significant (pooled HR 1.15, 95% CI: 0.94–1.41). A pooled analysis of eight case-control studies, which somewhat overlapped with the meta-analysis, reported a positive association between alcohol consumption and melanoma (47). Therefore, prospective data on alcohol intake and melanoma are limited with no evaluation of the association by body location of melanoma.
We therefore investigated the association between alcohol consumption and melanoma risk using prospective data from three large cohort studies.
Materials and Methods
Study Population
A total of 210,252 participants from the United States were followed for a mean of 18.3 years (3,855,706 person-years). Details of each cohort are presented in Supplementary Table S1. Briefly, the Nurses’ Health Study (NHS) started in 1976 with 121,700 married, female registered nurses aged 30–55 years. The Nurses’ Health Study II (NHS II) began in 1989 with 116,671 female nurses aged 25–42 years. The Health Professionals Follow-Up Study (HPFS) began in 1986 with 51,529 men in health professions. Participants receive biennial questionnaires, and the response rate in each follow-up cycle typically exceeds 90%. Return of the questionnaires was considered as informed consent in the studies. This study was approved by the Human Research Committee at the Brigham and Women’s Hospital and Harvard School of Public Health (Boston, MA, USA).
Assessment of Alcohol Intake
Semi-quantitative food frequency questionnaires (FFQs) were administered almost every four years from 1984–2006 in the NHS, from 1991–2007 in the NHS II, and from 1986–2006 in the HPFS. Participants were asked: “For each food listed, how often on average have you used the amount specified during the past year?” Five questions pertained to alcoholic beverages (regular beer, light beer, red wine, white wine, and liquor). Intake was reported in nine categories (number of drinks): 0 or ≤1 per month, 1–3 per month, 1 per week, 2–4 per week, 5–6 per week, 1 per day, 2–3 per day, 4–5 per day, and 6+ per day. Responses were coded as 0, 0.5, 1, 3, 5.5, 7, 17.5, 31.5, and 49 drinks per week, respectively. Beverage-specific consumption was calculated as coded above for red wine, white wine, liquor, and beer (combined light and non-light beer). For other analyses, the amount of alcohol in grams was estimated for each beverage (12.8 g for a beer, 11 g for a glass of wine, and 14 g for a shot of liquor). Total alcohol intake was computed as the sum from these sources. Due to the paucity of participants in higher intake categories, several higher consumption categories were combined for analysis. To reduce within-person variation and to better estimate long-term intake by taking advantage of multiple dietary assessments during follow-up in each study, we used the cumulative average intake of alcohol as reported on all available questionnaires up to the start of each 2-year follow-up interval. For example, 1991 alcohol intake was used for the 1991 to 1995 follow-up periods, the average of 1991 and 1995 intake was used for the 1995 to 1999 follow-up periods, and the average of all three intakes (1991, 1995, and 1999) was used for the 1999–2003 follow-up periods to maintain a strictly prospective analysis in NHSII. HRs “per drink” in the total alcohol intake analyses are reported for a standard drink containing 12.8 g of alcohol (median amount of alcohol in one drink of beer, wine, or liquor). The reliability of this questionnaire for alcohol intake has been previously documented.(48) To evaluate the validity of the reported alcohol consumption measured by the FFQ, comprehensive diet records and plasma high-density lipoprotein (HDL) levels were obtained from a subsample of participants in the NHS and HPFS. Mean daily intake of alcohol as assessed by FFQs and diet records were very similar (Spearman correlation coefficient of 0.90 in women and 0.86 in men). Moreover, the difference in plasma HDL for men reporting zero alcohol intake versus drinkers reporting 39 g/day of alcohol intake on the FFQ was 11.8 mg/dl, a magnitude similar to that determined from short term intervention studies of alcohol.
Assessment of Covariates
Participants reported their date of birth, height, current weight, smoking history, physical activity, caffeine intake (49), family history of melanoma, tanning ability, lifetime number of severe sunburns, number of moles on forearms, hair color at age 18, and place of residence on the biennial mailed questionnaires. Quintiles of metabolic equivalents were calculated from questions about physical activity. BMI was calculated from reported weight and height. Age was calculated from reported date of birth. Average annual UVB flux, a composite measure of mean UVB radiation level based on latitude, altitude, and cloud cover, was estimated for all participants according to state of residence. The accuracy of self-reported anthropometric measures was previously validated among 140 NHS participants. Self-reported and measured weights were highly correlated (Pearson r = 0.97).(50)
Assessment of Melanoma and Melanoma in situ
Cases were ascertained from participants’ responses to a question on physician-diagnosed melanoma on the biennial questionnaires. With their permission, medical records of participants’ who reported a diagnosis of melanoma were reviewed by physicians to confirm the diagnosis and to record the anatomic site affected. Only incident cases of cutaneous melanoma that were confirmed via pathology record review were included.
Statistical Analyses
Participants were excluded at baseline if they reported a personal history of cancer, including non-melanoma skin cancer, in order to avoid ascertainment bias due to closer physician follow-up of cancer patients. Participants who reported non-white race/ethnicity were also excluded, since there were too few non-white participants from which to draw statistically valid conclusions. After application of exclusion criteria, 73,545 participants in the NHS, 88,380 in the NHS II, and 48,327 in the HPFS were included. Person-years of follow-up for each participant were calculated from return of the baseline questionnaire (1984 for NHS, 1991 for NHS II, and 1986 for HPFS) to the date of melanoma diagnosis, death, or the end of follow-up (June 2012 for NHS, June 2011 for NHS II, and January 2012 for HPFS), whichever came first.
We used a Cox proportional hazards model updated by calendar time at 2-year intervals to estimate age-adjusted and multivariate-adjusted HRs and 95% confidence intervals. Covariates were updated during each follow-up cycle, if available. Beverage-specific analyses were additionally adjusted for total alcohol intake. To calculate the P value for the test for trend, participants were assigned the median value of their category of alcohol consumption, and this variable was used as a continuous variable in the study-specific regression models. To assess for effect modification, we stratified research subjects by the variable of interest (age, smoking history, caffeine intake, physical activity, hair color, BMI, anatomic site) and calculated interaction terms. Finally, we performed a sensitivity analysis by excluding cases of lentigo maligna melanoma, a melanoma subtype that is particularly UVR-dependent.(51) We used a meta-analytic approach drawing on data from all three cohorts to obtain pooled HRs for all participants. We tested the heterogeneity among studies using the Q statistic and estimated the overall association from random effects models (weighted proportionately to the inverse of the sum of the study-specific variance plus the common variance between studies) or fixed effects models (weighted proportionately to the inverse of the study-specific variance).
All statistical analyses were conducted using Statistical Analysis System software (SAS, version 9.3; SAS Institute, Cary, NC). All statistical tests were two tailed, and the significance level was set at p <0.05.
Results
We identified a total of 1,374 cases of incident invasive melanoma (490 in the NHS, 391 in the NHS II, and 493 in the HPFS). Cases of incident melanoma in situ totaled 835 (324 in the NHS, 289 in the NHS II, and 222 in the HPFS).
Table 1 shows characteristics of the study population by alcohol intake at baseline, while Supplementary Tables S1 and S2 present basic cohort information and drinking patterns by cohort, respectively. Participants with higher alcohol intake were more likely to smoke and to consume caffeine, consistent with known behavioral patterns relating the consumption of alcohol, tobacco, and caffeine.(52) Higher alcohol consumption was also associated with slightly greater physical activity and higher proportion of participants with red/blond hair color. Among women, alcohol use was associated with a greater fraction of participants reporting 6+ lifetime severe sunburns. Other factors showed no association with alcohol consumption.
Table 1.
Age-adjusted characteristics of study population according to average alcohol intake Nurses’ Health Study (NHS), Nurses’ Health Study II (NHSII) and Health Professionals Follow-Up Study (HPFS)
| Characteristics | Average alcohol intake, g per day | ||||
|---|---|---|---|---|---|
| None | 0.1–4.9 | 5.0–9.9 | 10.0–19.9 | 20.0+ | |
| NHS (women) n = 73,545 at baseline in 1984 | n = 23021 | n = 24631 | n = 8537 | n = 10587 | n = 6769 |
| Mean alcohol intake, g per day | 0.0 (0.0) | 2.2 (1.2) | 7.2 (1.4) | 13.7 (2.5) | 35.9(13.9) |
| Mean age, years | 50.6 (7.3) | 49.8 (7.2) | 49.9 (7.1) | 50.5 (7.0) | 51.2 (6.8) |
| Mean BMI, kg m−2 | 26.1 (5.4) | 25.3 (4.8) | 24.3 (3.9) | 23.9 (3.8) | 23.8 (3.8) |
| Physical Activity, metabolic equivalents per week | 12.0 (18.7) | 14.4(21.8) | 15.5 (20.1) | 15.8(22.4) | 15.4 (22.2) |
| 6+ sunburns that blistered in lifetime, % | 6 | 7 | 8 | 9 | 9 |
| 6+ moles ≥ 3mm on left forearm, % | 5 | 5 | 4 | 4 | 4 |
| Natural red/blond hair color at age 18, % | 15 | 15 | 16 | 17 | 17 |
| Family history of melanoma*, % | 3 | 3 | 3 | 3 | 3 |
| High skin sensitivity to sun**, % | 16 | 14 | 13 | 13 | 13 |
| Current smoking, % | 19 | 22 | 25 | 30 | 40 |
| Past smoking, % | 23 | 33 | 39 | 40 | 38 |
| Mean pack-years among ever smokers | 23.4 (18.7) | 20.3 (17.2) | 20.1 (17.1) | 22.1 (17.8) | 27.1 (20.1) |
| UVB flux at residence in 1986, RB counts × 10−4 | 122 (25) | 120 (23) | 121 (24) | 122 (24) | 125 (26) |
| Caffeine, mg/d | 289 (242) | 321 (232) | 346 (228) | 357 (224) | 365 (223) |
| NHS II (women) n = 88,380 at baseline in 1991 | n = 37140 | n = 34598 | n = 8791 | n = 5858 | n = 1993 |
| Mean alcohol intake, g per day | 0.0 (0.0) | 2.1 (1.2) | 7.1 (1.4) | 13.2 (2.4) | 32.5 (12.4) |
| Mean age, years | 36.2 (4.7) | 35.9 (4.7) | 35.9 (4.7) | 36.5 (4.7) | 37.4 (4.4) |
| Mean BMI, kg m−2 | 25.5 (5.9) | 24.3 (5.1) | 23.3 (4.0) | 23.3 (4.1) | 23.6 (4.1) |
| Physical Activity, metabolic equivalents per week | 18.4 (25.1) | 21.6 (27.1) | 24.0 (29.9) | 25.0 (29.2) | 25.0 (32.0) |
| 5+ sunburns that blistered in lifetime, % | 9 | 10 | 11 | 11 | 12 |
| 5+ moles ≥ 3mm on bilateral lower legs, % | 21 | 22 | 23 | 22 | 21 |
| Natural red/blond hair color at age 18, % | 20 | 20 | 22 | 23 | 24 |
| Family history of melanoma*, % | 4 | 4 | 5 | 5 | 5 |
| High skin sensitivity to sun**, % | 26 | 24 | 22 | 20 | 22 |
| Current smoking, % | 9 | 13 | 15 | 20 | 31 |
| Past smoking, % | 16 | 24 | 31 | 33 | 34 |
| Mean pack-years among ever smokers | 12.9 (9.2) | 11.4 (8.4) | 10.9 (7.9) | 11.7 (8.5) | 13.1 (9.0) |
| UVB flux at residence, RB counts × 10−4 | 125 (24) | 123 (24) | 125 (25) | 127 (26) | 130 (26) |
| Caffeine, mg/d | 198 (209) | 254 (215) | 295 (213) | 317 (214) | 350 (225) |
| HPFS (men) n = 48,327 at baseline in 1986 | n = 11270 | n = 11631 | n = 7014 | n = 9901 | n = 8511 |
| Alcohol intake, g per day | 0.0 (0.0) | 2.5 (1.2) | 7.3 (1.4) | 14.2 (2.6) | 39.1 (16.7) |
| Age, years | 54.7 (10.0) | 53.6 (10.0) | 53.5 (9.8) | 54.4 (9.6) | 55.2 (9.6) |
| BMI, kg m−2 | 25.7 (3.6) | 25.6 (3.4) | 25.4 (3.3) | 25.4 (3.2) | 25.5 (3.2) |
| Physical Activity, metabolic equivalents per week | 18.4 (26.7) | 20.0 (27.7) | 22.3 (31.5) | 22.8 (32.0) | 22.0 (30.2) |
| 6+ sunburns that blistered in lifetime | 35 | 34 | 34 | 36 | 38 |
| 6+ moles ≥3mm on bilateral forearms, % | 6 | 5 | 5 | 5 | 5 |
| Natural red/blond hair color at age 18, % | 14 | 13 | 13 | 14 | 16 |
| Family history of melanoma, % | 3 | 3 | 3 | 3 | 3 |
| High skin sensitivity to sun**, % | 27 | 26 | 24 | 24 | 23 |
| Current smoking, % | 7 | 8 | 9 | 10 | 17 |
| Past smoking, % | 33 | 41 | 45 | 50 | 55 |
| Mean pack-years among ever smokers | 27.4 (20.8) | 24.1 (18.7) | 24.1 (19.0) | 24.1 (18.0) | 28.8 (20.5) |
| UVB flux at residence in 1988, RB counts × 10−4 | 133 (28) | 128 (27) | 128 (27) | 129 (28) | 132 (28) |
| Caffeine, mg/d | 190 (228) | 210 (220) | 226 (217) | 244 (223) | 29038) |
Values are reported as mean (standard deviation) or as percentages. All variables (except age) are standardized to the age-distribution of the study population.
Defined as a skin reaction consisting of painful burning and then peeling after 1 hour in the sun
Comparing cohorts, women in the NHS reported drinking more caffeine, getting less physical activity, and smoking more than other participants. Women in the NHS II were more likely to have a family history of melanoma and more likely to have natural red or blond hair. They were also younger and had less extensive smoking histories than other participants. Conversely, men in the HPFS were older and more likely to have experienced 6+ blistering sunburns in their lifetime.
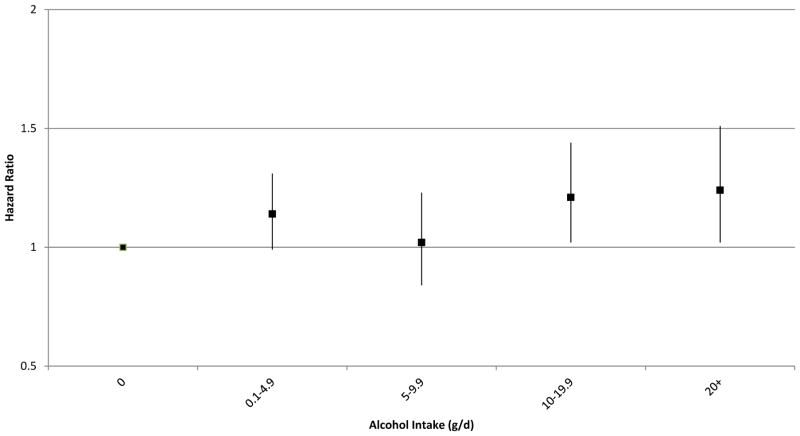
Table 2 shows the HRs for the association between alcohol intake and invasive melanoma. Significant associations were found in the NHS II (P trend =0.01) and in the pooled analysis of the three cohorts (HR 1.14 per drink per day, 95% CI: 1.00–1.29, P trend =0.04) after adjusting for multiple covariates. The NHS and HPFS cohorts showed tendencies toward elevated risk of invasive melanoma associated with alcohol use, but those results did not reach statistical significance. For the heaviest drinkers (≥20g of ethanol per day), the pooled multivariate-adjusted HR was 1.23 (95% CI: 0.96–1.59) compared to non-drinkers (P for heterogeneity by study=0.26). We also conducted a combined analysis of the three cohort studies and found that alcohol intake was similarly positively associated with melanoma risk. Compared with non-drinkers, the multivariate HRs for increasing intake of alcohol (0.1–4.9, 5–9.9, 10–19.9, 20+ g/d) were 1.14 (95% CI 0.99–1.31), 1.02 (95% CI 0.84–1.23), 1.21 (95% CI 1.02–1.44), and 1.24 (95% CI 1.02–1.51) (Figure 1).
Table 2.
Age-adjusted and multivariate-adjusted Hazard Ratios (HRs) and pooled estimates for incident invasive melanoma by amount of average alcohol intake in Nurses’ Health Study (NHS), Nurses’ Health Study II (NHSII) and Health Professionals Follow-Up Study (HPFS)
| Alcohol intake, grams per day | Number of incident cases | Age-adjusted HR (95% CI) | Multivariate* HR (95% CI) |
|---|---|---|---|
| NHS (women) | |||
| 0 | 189 | Reference | Reference |
| 0.1–4.9 | 146 | 1.04 (0.83–1.29) | 1.00 (0.80–1.25) |
| 5–9.9 | 53 | 1.10 (0.81–1.49) | 1.04 (0.76–1.42) |
| 10–19.9 | 66 | 1.14 (0.86–1.51) | 1.07 (0.80–1.44) |
| 20+ | 36 | 1.10 (0.77–1.57) | 1.05 (0.73–1.52) |
| Total | 490 | P (trend) = 0.34 | P (trend) = 0.61 |
| NHS II (women) | |||
| 0 | 126 | Reference | Reference |
| 0.1–4.9 | 156 | 1.37 (1.08–1.73) | 1.42 (1.11–1.80) |
| 5–9.9 | 44 | 1.22 (0.86–1.72) | 1.26 (0.88–1.79) |
| 10–19.9 | 45 | 1.52 (1.08–2.14) | 1.57 (1.10–2.25) |
| 20+ | 20 | 1.67 (1.04–2.69) | 1.76 (1.08–2.87) |
| Total | 391 | P (trend) = 0.01 | P (trend) = 0.01 |
| HPFS (men) | |||
| 0 | 118 | Reference | Reference |
| 0.1–4.9 | 113 | 1.02 (0.79–1.32) | 1.03 (0.80–1.34) |
| 5–9.9 | 55 | 0.83 (0.60–1.14) | 0.83 (0.60–1.15) |
| 10–19.9 | 112 | 1.12 (0.86–1.45) | 1.12 (0.86–1.46) |
| 20+ | 95 | 1.13 (0.86–1.48) | 1.17 (0.88–1.55) |
| Total | 493 | P (trend) = 0.24 | P (trend) = 0.19 |
| Meta-analysis (all cohorts) | Age-adjusted HR (95% CI) | Multivariate* HR (95% CI) | |
| 0 | 433 | Reference | Reference |
| 0.1–4.9 | 415 | 1.13 (0.94, 1.37) | 1.13 (0.91, 1.41) |
| 5–9.9 | 152 | 1.03 (0.82, 1.29) | 1.02 (0.81, 1.28) |
| 10–19.9 | 223 | 1.21 (1.02, 1.45) | 1.21 (0.97, 1.49) |
| 20+ | 151 | 1.21 (0.97, 1.50) | 1.23 (0.96, 1.59) |
| Per drink/day** | 1374 | 1.14 (1.02, 1.27) | 1.14 (1.00, 1.29) |
| P (trend) = 0.02 | P (trend) = 0.04† |
Multivariate estimates are adjusted for age, BMI, smoking status, physical activity, caffeine intake, family history of melanoma, tanning ability, lifetime number of severe sunburns, number of moles on forearms, hair color at age 18, and average annual UV-B flux at place of residence.
Relative risks per drink per day were estimated based on a standard drink containing 12.8 grams of alcohol.
P for heterogeneity by study=0.26, I2 15.
Figure 1.
Multivariate hazard ratios (95% confidence intervals) for categories of alcohol consumption and melanoma risk, compared with non-drinkers; multivariable model included the covariates listed in footnote in Table 2.
Supplementary Table S3 displays the equivalent data for melanoma in situ. After adjusting for covariates, higher alcohol consumption was positively associated with risk of melanoma in situ in the NHS (P trend <0.01), the NHS II (P trend <0.01), and the pooled analysis (HR 1.46 per drink per day, 95% CI: 1.24–1.72, P trend <0.0001). There was no association in the men’s cohort (HPFS). For the heaviest drinkers (≥20g of ethanol per day), the pooled multivariate-adjusted HR was 1.57 (95% CI: 1.12–2.22) compared to non-drinkers.
Table 3 shows HRs for the association between beer, red wine, white wine, and liquor with incident invasive melanoma after adjusting for melanoma risk factors and alcoholic beverages simultaneously. White wine consumption was associated with melanoma risk in the HPFS (P trend =0.02) and pooled analyses (HR 1.13, 95% CI: 1.04–1.24, P trend <0.01, P for heterogeneity by study=0.63), but not in the NHS or NHS II. Supplementary Table S4 shows the same type of beverage-specific analysis with incidence of melanoma in situ as the outcome of interest. White wine consumption was associated with melanoma in situ in the NHS (P trend =0.003) and NHS II (P trend =0.03), and in the pooled analysis (HR: 1.18, 95% CI: 1.03–1.35, P trend=0.02), but not in the HPFS. No associations were found between beer, red wine, or liquor and incidence of either invasive melanoma or melanoma in situ.
Table 3.
Multivariate Hazard Ratios (HRs) for invasive melanoma according to alcoholic beverage intake in Nurses’ Health Study (NHS), Nurses’ Health Study II (NHSII) and Health Professionals Follow-Up Study (HPFS)
| Drinks Consumed | Beer | Red wine | White wine | Liquor* | ||||
|---|---|---|---|---|---|---|---|---|
| Cases (n) | HR** | Cases (n) | HR** | Cases (n) | HR** | Cases (n) | HR** | |
| NHS (women) | ||||||||
| None | 399 | Ref. | 315 | Ref. | 251 | Ref. | 348 | Ref. |
| 1–3/m | 41 | 0.86 (0.62, 1.19) | 91 | 1.36 (1.04, 1.77) | 105 | 0.98 (0.76, 1.27) | 48 | 0.75 (0.55, 1.03) |
| 1/wk | 20 | 1.14 (0.72, 1.80) | 26 | 1.02 (0.66, 1.60) | 43 | 1.05 (0.72, 1.51) | 31 | 1.13 (0.77, 1.66) |
| 2–4/wk | 18 | 0.65 (0.40, 1.06) | 41 | 1.83 (1.26, 2.67) | 49 | 1.03 (0.72, 1.48) | 61 | 0.94 (0.70, 1.26) |
| ≥ 5/wk | 10 | 0.81 (0.43, 1.53) | 15 | 0.99 (0.58, 1.69) | 40 | 1.31 (0.91, 1.87) | ||
| P for trend | 0.94 | 0.90 | 0.12 | 0.85 | ||||
| Per drink/d | 1.01 (0.79, 1.30) | 1.01 (0.83, 1.23) | 1.12 (0.97, 1.28) | 0.98 (0.84, 1.16) | ||||
| NHSII (women) | ||||||||
| None | 249 | Ref. | 267 | Ref. | 198 | Ref. | 301 | Ref. |
| 1–3/m | 52 | 1.16 (0.85, 1.59) | 51 | 0.75 (0.54, 1.03) | 99 | 1.34 (1.03, 1.75) | 58 | 0.96 (0.71, 1.28) |
| 1/wk | 16 | 0.95 (0.56, 1.59) | 36 | 1.26 (0.85, 1.88) | 41 | 1.35 (0.92, 1.99) | 16 | 0.76 (0.45, 1.28) |
| 2–4/wk | 55 | 1.19 (0.87, 1.63) | 21 | 0.82 (0.50, 1.33) | 36 | 1.69 (1.13, 2.52) | 14 | 0.74 (0.42, 1.27) |
| ≥ 5/wk | 17 | 1.39 (0.84, 2.31) | 14 | 1.26 (0.72, 2.21) | 15 | 1.23 (0.71, 2.13) | ||
| P for trend | 0.70 | 0.42 | 0.60 | 0.29 | ||||
| Per drink/d | 1.10 (0.68, 1.78) | 1.10 (0.88, 1.38) | 1.06 (0.86, 1.30) | 0.84 (0.61, 1.16) | ||||
| HPFS (men) | ||||||||
| None | 216 | Ref. | 273 | Ref. | 220 | Ref. | 225 | Ref. |
| 1–3/m | 83 | 1.02 (0.78, 1.33) | 75 | 0.66 (0.49, 0.88) | 117 | 1.13 (0.87, 1.46) | 92 | 1.58 (1.21, 2.05) |
| 1/wk | 38 | 0.89 (0.62, 1.28) | 51 | 0.82 (0.58, 1.17) | 68 | 1.24 (0.90, 1.72) | 46 | 1.43 (1.02, 2.00) |
| 2–4/wk | 97 | 0.99 (0.75, 1.29) | 53 | 0.92 (0.65, 1.30) | 47 | 0.97 (0.68, 1.40) | 129 | 1.22 (0.96, 1.55) |
| ≥ 5/wk | 56 | 1.05 (0.76, 1.43) | 38 | 0.92 (0.64, 1.34) | 40 | 1.66 (1.15, 2.39) | ||
| P for trend | 0.81 | 0.70 | 0.02 | 0.73 | ||||
| Per drink/d | 1.02 (0.89, 1.16) | 1.03 (0.89, 1.19) | 1.19 (1.03, 1.37) | 1.02 (0.90, 1.16) | ||||
| Meta-analysis (all cohorts) | ||||||||
| None | 864 | Ref | 797 | Ref. | 669 | Ref. | 874 | Ref. |
| 1–3/m | 176 | 1.01 (0.85, 1.21) | 226 | 0.88 (0.55, 1.39) | 321 | 1.14 (0.95, 1.36) | 198 | 1.05 (0.68, 1.63) |
| 1/wk | 74 | 0.97 (0.76, 1.25) | 100 | 1.00 (0.78, 1.30) | 152 | 1.21 (0.98, 1.48) | 93 | 1.12 (0.81, 1.56) |
| 2–4/wk | 170 | 0.96 (0.72, 1.28) | 159 | 1.12 (0.68, 1.85) | 132 | 1.18 (0.85, 1.65) | 204 | 1.01 (0.78, 1.31) |
| ≥ 5/wk | 83 | 1.08 (0.84, 1.38) | 85 | 1.01 (0.77, 1.32) | 95 | 1.42 (1.13, 1.80) | ||
| P for trend† | 0.54 | 0.47 | <0.01 | 0.86 | ||||
| Per drink/d | 1.02 (0.91, 1.14) | 1.04 (0.94, 1.15) | 1.13 (1.04, 1.24) | 0.99 (0.90, 1.09) | ||||
The highest consumption category for liquor is “2+ drinks/wk”
Values are reported as multivariate adjusted relative risks (95% confidence intervals). The covariates in this model are age, BMI, smoking status, physical activity, caffeine intake, family history of melanoma, tanning ability, lifetime number of severe sunburns, number of moles on forearms, hair color at age 18, and average annual UV-B flux at place of residence, and intake of other alcoholic beverages.
P for heterogeneity by study=0.95 for beer, 0.86 for red wine, 0.63 for white wine, and 0.53 for liquor.
The associations were similar in stratified analyses by age, BMI, smoking history, physical activity, natural hair color, mole count, and caffeine intake (data not shown). When we evaluated alcohol intake in relation to anatomic site of tumor, melanomas of the head, neck, and extremities were not associated with alcohol intake in the pooled analysis, while melanomas of the trunk were associated with consumption of alcohol (P trend=0.02; Table 4). Compared to non-drinkers, the pooled multivariate-adjusted HRs for ≥20g/d of alcohol were 1.02 (95% CI: 0.64–1.62; P trend =0.25) for melanomas of the head, neck, and extremities and 1.73 (95% CI: 1.25–2.38; P trend =0.02) for melanomas of the trunk.
Table 4.
Multivariate-adjusted Hazard Ratios (HRs) and pooled estimates for incident invasive melanoma by anatomic site in Nurses’ Health Study (NHS), Nurses’ Health Study II (NHSII) and Health Professionals Follow-Up Study (HPFS)*
| Alcohol intake, grams/d | Cases (n) | Head, Neck, Extremities | Cases (n) | Trunk |
|---|---|---|---|---|
| NHS (women) | ||||
| 0 | 131 | Reference | 52 | Reference |
| 0.1–4.9 | 94 | 0.93 (0.71, 1.22) | 49 | 1.20 (0.80, 1.78) |
| 5–9.9 | 38 | 1.08 (0.74, 1.56) | 14 | 0.97 (0.53, 1.78) |
| 10–19.9 | 49 | 1.14 (0.81, 1.61) | 15 | 0.88 (0.48, 1.59) |
| 20+ | 18 | 0.76 (0.46, 1.26) | 16 | 1.65 (0.92, 2.96) |
| P (trend) | >0.99 | 0.52 | ||
| NHS II (women) | ||||
| 0 | 81 | Reference | 44 | Reference |
| 0.1–4.9 | 96 | 1.38 (1.02, 1.86) | 57 | 1.44 (0.96, 2.15) |
| 5–9.9 | 27 | 1.21 (0.77, 1.90) | 17 | 1.37 (0.77, 2.44) |
| 10–19.9 | 31 | 1.68 (1.09, 2.60) | 13 | 1.31 (0.68, 2.49) |
| 20+ | 13 | 1.77 (0.96, 3.25) | 7 | 1.81 (0.79, 4.14) |
| P (trend) | 0.02 | 0.27 | ||
| HPFS (men) | ||||
| 0 | 55 | Reference | 40 | Reference |
| 0.1–4.9 | 46 | 0.92 (0.62, 1.37) | 51 | 1.34 (0.88, 2.04) |
| 5–9.9 | 26 | 0.87 (0.54, 1.39) | 25 | 1.09 (0.66, 1.81) |
| 10–19.9 | 58 | 1.26 (0.86, 1.84) | 45 | 1.28 (0.82, 1.98) |
| 20+ | 33 | 0.86 (0.55, 1.35) | 49 | 1.75 (1.13, 2.71) |
| P (trend) | 0.69 | 0.04 | ||
| Meta-analysis (all cohorts) | ||||
| 0 | 267 | Reference | 136 | Reference |
| 0.1–4.9 | 236 | 1.06 (0.81, 1.39) | 157 | 1.32 (1.05–1.67) |
| 5–9.9 | 91 | 1.05 (0.82, 1.35) | 56 | 1.13 (0.82–1.56) |
| 10–19.9 | 138 | 1.30 (1.04, 1.62) | 73 | 1.16 (0.85, 1.58) |
| 20+ | 64 | 1.02 (0.64, 1.62) | 72 | 1.73 (1.25–2.38) |
| P (trend) | 0.25 | 0.02 | ||
| Per drink/d† | 1.11 (0.93, 1.34) | 1.22 (1.03, 1.45) |
All results are reported as relative risk (95% CI). Multivariate estimates are adjusted for age, BMI, smoking status, physical activity, caffeine intake, family history of melanoma, tanning ability, lifetime number of severe sunburns, number of moles on forearms, hair color at age 18, and average annual UV-B flux at place of residence.
Relative risks per drink per day were estimated based on a standard drink containing 12.8 g of alcohol. Melanomas of head, neck, and extremities; P (for heterogeneity by study)=0.09, I2 59, truncal melanomas; P >0.99, I2 0.
Although our multivariate-adjusted analyses included several measures of UVR exposure, an additional sensitivity analysis was performed in order to assess for residual confounding by UVR exposure. In this sensitivity analysis, there was no material change when lentigo maligna melanoma (LMM) cases (n=5) were excluded (data not shown).
Discussion
Our data demonstrate that alcohol consumption was associated with modest increased risk of melanoma. Among alcoholic beverages, white wine consumption was associated with increased risk of melanoma independent of other alcoholic beverages. The positive association between alcohol consumption and melanoma risk was stronger for melanoma in relatively UV-spared sites (trunk) than relatively UV-exposed sites (head, neck, or extremities).
Alcohol in general causes carcinogenesis via acetaldehyde by creating DNA adducts.(23–25) It is unclear whether the same mechanism operates in skin carcinogenesis. Alcohol may also act as a photosensitizer and the combination of UV radiation and alcohol consumption may potentiate the skin carcinogenesis (53). However, the stronger association with melanoma of UV-spared sites than UV-exposed sites supports against the mechanism.
The association between alcohol consumption and risk of melanoma in situ was stronger in women than in men, while the association with invasive melanoma was only significant in the NHSII. Men have a greater average volume of distribution and greater gastric metabolism of alcohol. Consequently, men generate lower blood concentrations of alcohol and acetaldehyde than women after imbibing equivalent doses of alcohol.(54–56) Therefore, the effective dose of alcohol and acetaldehyde reaching the melanocytes would be higher in women (on average) than in men at the same level of alcohol consumption. Since it is the effective dose reaching the melanocytes that affects the probability of developing melanoma, it is reasonable that the risk per drink per day is higher for women. Meanwhile, in women, the association was stronger in NHSII, which was based on younger women with alcohol intake assessed earlier in life than in NHS. It may suggest that alcohol intake in earlier life might be more relevant to melanoma risk.
Although we hypothesized that other life-style factors might modulate how alcohol use affects the risk of cancer(57), there was no evidence that age, smoking history, caffeine intake, physical activity, hair color, mole count or BMI modified the association between alcohol intake and melanoma when results were stratified by those variables However, heavier drinkers were more likely to be smokers, had higher caffeine intake, had a higher number of lifetime severe sunburns, and were more physically active than those who consumed less alcohol. Although each of these variables was included in the regression model as a covariate in the calculation of adjusted hazard ratios, the possibility of residual confounding remains. In addition, alcohol drinking pattern (e.g. binge drinking compared to consuming alcohol with a meal) was not evaluated, and may be an important modifier of the effect of alcohol consumption on melanoma.
When alcohol intake was investigated in relation to anatomic site of melanoma, we found that alcohol intake had a far greater effect on melanoma risk at relatively UV-spared sites such as the trunk compared to highly UV-exposed sites such as the head, neck, or extremities. Because the literature suggests that etiologies of melanoma differ by anatomic site, (58) we posit that alcohol consumption may affect those etiologic pathways differently. Alcohol’s carcinogenic effect may be more relevant to melanomas of relatively UV spared sites. A pooled analysis of eight case-control studies also evaluated the effect of alcohol by anatomic site of melanoma. The positive association between alcohol intake and melanoma was slightly stronger with truncal melanomas than melanomas of other body site (47)
Among alcoholic beverages, we believe that some types are carcinogenic beyond what would be explained by ethanol content alone.(27–28) We attribute this effect to high levels of pre-existing acetaldehyde in those beverages, which adds to the carcinogenicity of endogenously generated acetaldehyde. In some cases, the pre-existing acetaldehyde alone is above the carcinogenic level.(26–27) To isolate the effects of pre-existing acetaldehyde and other non-ethanol components of alcoholic beverages, we analyzed each alcoholic beverage (white wine, red wine, beer, and liquor) after adjusting for other alcoholic beverages simultaneously. White wine was the only beverage independently associated with risk of melanoma. This accords with research showing that wine has far higher levels of pre-existing acetaldehyde than beer or spirits.(26–27) Our findings also support those of a previous study by Kubo et al, in which preference for white wine was associated with increased risk of melanoma and non-melanoma skin cancer in the Women’s Health Initiative, a cohort of postmenopausal women.(59). A pooled analysis of eight case-control studies also found that among alcoholic beverages, wine was most strongly associated with melanoma risk (OR=1.4, 95% CI 1.1–1.8 for ever consumption vs. never) (47). Red vs. white wine was not evaluated separately. Red and white wines have similar pre-existing acetaldehyde content, but antioxidants in red wine may offset these risks; experiments show that blood acetaldehyde content and cytotoxicity are much lower after red versus white wine consumption.(60–63) Similarly, in studies of other alcohol-associated malignancies, white wine was frequently found to be more strongly associated than red wine or other forms or alcohol,(64–68) with some exceptions.(69–70)
We conducted a sensitivity analysis to test for residual confounding by UVR exposure (e.g. if white wine were preferentially consumed outdoors in the summer months compared to other beverage types). LMM is a subtype of melanoma that occurs almost exclusively on sun-damaged skin.(51) It is more tightly associated with UVR exposure than other melanoma subtypes. Therefore, if the association between white wine and melanoma were secondary to residual confounding by UVR, the association should be attenuated when LMM are excluded. Our results were not sensitive to the exclusion of LMM, so residual confounding by UVR exposure is unlikely to explain the association.
Our study was limited by the homogeneity of our study populations; all participants were white, educated, and largely worked in healthcare settings. This makes residual confounding by socioeconomic status, race/ethnicity, healthcare access, or health literacy less likely, but may limit generalizability in other populations, especially other racial/ethnic groups.(71–74) For example, among heavy drinkers, those who are heterozygous for the aldehyde dehydrogenase 2(ALDH2) gene are up to 12 times more likely to develop esophageal cancer compared to those who are homozygous for the active enzyme.(75–76) The proportion of carriers of heterozygous or homozygous forms of variant ALDH2 gene is much higher in Asian population than other populations. Additionally, we had few participants reporting heavy drinking in these cohorts and were limited to investigate the risks of higher levels of alcohol intake. Finally, we were not able to take into account some potential risk factors of melanoma such as sun protection behaviors.
Overall, our findings support carcinogenicity of alcohol on development of melanoma and are consistent with the proven carcinogenicity of alcoholic beverages in other malignancies. Furthermore, we found that the positive association between alcohol consumption and melanoma risk was stronger with melanoma of relatively UV-spared sites than UV-exposed sites. The clinical and biological significance of these findings remains to be determined, but for motivated individuals with other strong risk factors for melanoma, counseling regarding alcohol use may be an appropriate risk reduction strategy to reduce risks of melanoma as well as other cancers.
Supplementary Material
Acknowledgments
Funding
This work was supported by the National Institutes of Health (grant numbers CA186107 (M Stampfer), CA176726 (W Willett), CA167552 (W Willett), and CA198216 (E Cho).
We would like to thank the participants and staff of the NHS, NHS II, and HPFS for their valuable contributions as well as the following state cancer registries: AL, AZ, AR, CA, CO, CT, DE, FL, GA, ID, IL, IN, IA, KY, LA, ME, MD, MA, MI, NE, NH, NJ, NY, NC, ND, OH, OK, OR, PA, RI, SC, TN, TX, VA, WA, WY. The authors assume full responsibility for analyses and interpretation of these data.
Footnotes
Financial Disclosures: AAQ serves as a consultant for Abbott, Centocor, Novartis and the Centres for Disease Control and Prevention. The other authors state no conflict of interest.
References
- 1.Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30. doi: 10.3322/caac.21166. [DOI] [PubMed] [Google Scholar]
- 2.Linos E, Swetter SM, Cockburn MG, Colditz GA, Clarke CA. Increasing burden of melanoma in the United States. J Invest Dermatol. 2009;129:1666–1674. doi: 10.1038/jid.2008.423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Millen AE, Tucker MA, Hartge P, Halpern A, Elder DE, Guerry D, 4th, et al. Diet and melanoma in a case-control study. Cancer Epidemiol Biomarkers Prev. 2004;13:1042–1051. [PubMed] [Google Scholar]
- 4.Rigel DS. Epidemiology of melanoma. Semin Cutan Med Surg. 2010;29:204–209. doi: 10.1016/j.sder.2010.10.005. [DOI] [PubMed] [Google Scholar]
- 5.Rigel DS. Trends in dermatology: melanoma incidence. Arch Dermatol. 2010;146:318. doi: 10.1001/archdermatol.2009.379. [DOI] [PubMed] [Google Scholar]
- 6.Garbe C, Leiter U. Melanoma epidemiology and trends. Clin Dermatol. 2009;27:3–9. doi: 10.1016/j.clindermatol.2008.09.001. [DOI] [PubMed] [Google Scholar]
- 7.MacKie RM, Hauschild A, Eggermont AM. Epidemiology of invasive cutaneous melanoma. Ann Oncol. 2009;20(Suppl 6):vi1–7. doi: 10.1093/annonc/mdp252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Barbaric J, Znaor A. Incidence and mortality trends of melanoma in Croatia. Croatian Medical Journal. 2012;53:135–140. doi: 10.3325/cmj.2012.53.135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Koh D, Wang H, Lee J, Chia KS, Lee HP, Goh CL. Basal cell carcinoma, squamous cell carcinoma and melanoma of the skin: analysis of the Singapore Cancer Registry data 1968–97. Br J Dermatol. 2003;148:1161–1166. doi: 10.1046/j.1365-2133.2003.05223.x. [DOI] [PubMed] [Google Scholar]
- 10.Glusac EJ. The melanoma ‘epidemic’, a dermatopathologist’s perspective. J Cutan Pathol. 2011;38:264–267. doi: 10.1111/j.1600-0560.2010.01660.x. [DOI] [PubMed] [Google Scholar]
- 11.Ortiz CA, Goodwin JS, Freeman JL. The effect of socioeconomic factors on incidence, stage at diagnosis and survival of cutaneous melanoma. Medical Science Monitor. 2005;11:163–172. [PubMed] [Google Scholar]
- 12.Pollitt RA, Clarke CA, Shema SJ, Swetter SM. California Medicaid enrollment and melanoma stage at diagnosis: a population-based study. Am J Prev Med. 2008;35:7–13. doi: 10.1016/j.amepre.2008.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Duffey KJ, Popkin BM. Shift in Patterns and Consumption of Beverages Between 1965 and 2002. Obesity. 2007;15:2739–2747. doi: 10.1038/oby.2007.326. [DOI] [PubMed] [Google Scholar]
- 14.Warthan MM, Sewell DS, Marlow RA, Warthan ML, Wagner RF., Jr The economic impact of acute sunburn. Arch Dermatol. 2003;139:1003–1006. doi: 10.1001/archderm.139.8.1003. [DOI] [PubMed] [Google Scholar]
- 15.Saladi RN, Nektalova T, Fox JL. Induction of skin carcinogenicity by alcohol and ultraviolet light. Clin Exp Dermatol. 2010;35:7–11. doi: 10.1111/j.1365-2230.2009.03465.x. [DOI] [PubMed] [Google Scholar]
- 16.Bagnardi V, Blangiardo M, La Vecchia C, Corrao G. A meta-analysis of alcohol drinking and cancer risk. BJC. 2001;85:1700–1705. doi: 10.1054/bjoc.2001.2140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Poschl G, Seitz HK. Alcohol and Cancer. Alcohol Alcohol. 2004;39:155–165. doi: 10.1093/alcalc/agh057. [DOI] [PubMed] [Google Scholar]
- 18.Williams RR. Breast and thyroid cancer and malignant melanoma promoted by alcohol-induced pituitary secretion of prolactin, T.S.H. and M.S.H. Lancet. 1976;1:996–999. doi: 10.1016/s0140-6736(76)91865-1. [DOI] [PubMed] [Google Scholar]
- 19.DeLancey JO, Hannan LM, Gapstur SM, Thun MJ. Cigarette smoking and the risk of indicant and fatal melanoma in a large prospective cohort study. Cancer Causes Control. 2011;22:937–942. doi: 10.1007/s10552-011-9766-z. [DOI] [PubMed] [Google Scholar]
- 20.Kessides MC, Wheless L, Hoffman-Bolton J, Clipp S, Alani RM, Alberg AJ. Cigarette smoking and malignant melanoma: a case-control study. J Am Acad Dermatol. 2011;64:84–90. doi: 10.1016/j.jaad.2010.01.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Boffetta P, Hashibe M, La Vecchia C, Zatonski W, Rehm J. The burden of cancer attributable to alcohol drinking. Int J Cancer. 2006;119:884–887. doi: 10.1002/ijc.21903. [DOI] [PubMed] [Google Scholar]
- 22.Sigvardsson S, Hardell L, Przybeck TR, Cloninger R. Increased cancer risk among Swedish female alcoholics. Epidemiology. 1996;7:140–143. doi: 10.1097/00001648-199603000-00006. [DOI] [PubMed] [Google Scholar]
- 23.Obe G, Anderson D. International Commission for Protection against Environmental Mutagens and Carcinogens. ICPEMC Working Paper No. 15/1. Genetic Effects of Ethanol. Mutat Res. 1987:177–200. doi: 10.1016/0165-1110(87)90003-0. [DOI] [PubMed] [Google Scholar]
- 24.Brooks PJ, Theruvathu JA. DNA adducts from acetaldehyde: implications for alcohol-related carcinogenesis. Alcohol. 2005;35:187–193. doi: 10.1016/j.alcohol.2005.03.009. [DOI] [PubMed] [Google Scholar]
- 25.Seitz HK, Mueller S. Alcohol and cancer: an overview with special emphasis on the role of acetaldehyde and cytochrome P450 2E1. Adv Exp Med Biol. 2015;815:59–70. doi: 10.1007/978-3-319-09614-8_4. [DOI] [PubMed] [Google Scholar]
- 26.Lachenmeier DW, Sohnius EM. The role of acetaldehyde outside ethanol metabolism in the carcinogenicity of alcoholic beverages: Evidence from a large chemical survey. Food Chem Toxicol. 2008;46:2903–2911. doi: 10.1016/j.fct.2008.05.034. [DOI] [PubMed] [Google Scholar]
- 27.Lachenmeier DW, Kanteres F, Rehm J. Carcinogenicity of acetaldehyde in alcoholic beverages: risk assessment outside ethanol metabolism. Addiction. 2009;104:533–550. doi: 10.1111/j.1360-0443.2009.02516.x. [DOI] [PubMed] [Google Scholar]
- 28.Launoy G, Milan C, Day NE, Faivre J, Pienkowski P, Gignoux M. Oesophageal cancer in France: potential importance of hot alcoholic drinks. Int J Cancer. 1997;71:917–923. doi: 10.1002/(sici)1097-0215(19970611)71:6<917::aid-ijc1>3.0.co;2-0. [DOI] [PubMed] [Google Scholar]
- 29.Linderborg K, Joly JP, Visapää JP, Salaspuro M. Potential mechanism for Calvados-related oesophageal cancer. Food Chem Toxicol. 2008;46(2):476–479. doi: 10.1016/j.fct.2007.08.019. [DOI] [PubMed] [Google Scholar]
- 30.Tan W, Bailey AP, Shaparago M, Busby B, Covington J, Johnson JW, et al. Chronic alcohol consumption stimulates VEGF expression, tumor angiogenesis and progression of melanoma in mice. Cancer Biology & Therapy. 2007;6:1211–1217. doi: 10.4161/cbt.6.8.4406. [DOI] [PubMed] [Google Scholar]
- 31.Merimsky O, Inbar M. Alcohol intake-associated skin and mucosal cancer. Clin Dermatol. 1999;17:447–455. doi: 10.1016/s0738-081x(99)00031-0. [DOI] [PubMed] [Google Scholar]
- 32.Blank SE, Meadows GG. Ethanol modulates metastatic potential of B16BL6 melanoma and host responses. Alcohol Clin Exp Res. 1996;20:624–628. doi: 10.1111/j.1530-0277.1996.tb01664.x. [DOI] [PubMed] [Google Scholar]
- 33.Holman CD, Armstrong BK, Heenan PJ, Blackwell JB, Cumming FJ, English DR, et al. The causes of malignant melanoma: results from the West Australian Lions Melanoma Research Project. Recent Results Cancer Res. 1986;102:18–37. doi: 10.1007/978-3-642-82641-2_3. [DOI] [PubMed] [Google Scholar]
- 34.Le Marchand L, Saltzman BS, Hankin JH, Wilkens LR, Franke AA, Morris SJ, et al. Sun exposure, diet, and melanoma in Hawaii Caucasians. Am J Epidemiol. 2006;164:232–245. doi: 10.1093/aje/kwj115. [DOI] [PubMed] [Google Scholar]
- 35.Stryker WS, Stampfer MJ, Stein EA, Kaplan L, Louis TA, Sober A, et al. Diet, plasma levels of beta-carotene and alpha-tocopherol, and risk of malignant melanoma. Am J Epidemiol. 1990;131:597–611. doi: 10.1093/oxfordjournals.aje.a115544. [DOI] [PubMed] [Google Scholar]
- 36.Freedman DM, Sigurdson A, Doody MM, Rao RS, Linet MS. Risk of melanoma in relation to smoking, alcohol intake, and other factors in a large occupational cohort. Cancer Causes Control. 2003;14:847–857. doi: 10.1023/b:caco.0000003839.56954.73. [DOI] [PubMed] [Google Scholar]
- 37.Bain C, Green A, Siskind V, Alexander J, Harvey P. Diet and melanoma: An exploratory case-control study. Ann Epidemiol. 1993;3:235–238. doi: 10.1016/1047-2797(93)90024-x. [DOI] [PubMed] [Google Scholar]
- 38.Williams RR, Horm JW. Association of cancer sites with tobacco and alcohol consumption and socioeconomic status of patients: interview study from the Third National Cancer Survey. JNCI. 1997;58:525–547. doi: 10.1093/jnci/58.3.525. [DOI] [PubMed] [Google Scholar]
- 39.Williams RR, Stegens NL, Goldsmith JR. Associations of cancer site and type with occupation and industry from the Third National Cancer Survey Interview. JNCI. 1977;59:1147–1185. doi: 10.1093/jnci/59.4.1147. [DOI] [PubMed] [Google Scholar]
- 40.Kirkpatrick CS, White E, Lee JA. Case-control study of malignant melanoma in Washington State. II. Diet, alcohol, and obesity. Am J Epidemiol. 1994;139:869–880. doi: 10.1093/oxfordjournals.aje.a117093. [DOI] [PubMed] [Google Scholar]
- 41.Green A, Bain C, McLennan R, Siskind V. Risk factors for cutaneous melaonoma in Queensland. Recent Results Cancer Res. 1986;102:76–97. doi: 10.1007/978-3-642-82641-2_6. [DOI] [PubMed] [Google Scholar]
- 42.Holly EA, Aston DA, Cress RD, Ahn DK, Kristiansen JJ. Cutaneous melanoma in women. II. Phenotypic characteristics and other host-related factors. Am J Epidemiol. 1995;141:934–942. doi: 10.1093/oxfordjournals.aje.a117360. [DOI] [PubMed] [Google Scholar]
- 43.Sigvardsson S, Hardell L, Przybeck TR, Cloninger R. Increased cancer risk among Swedish female alcoholics. Epidemiology. 1996;7:140–143. doi: 10.1097/00001648-199603000-00006. [DOI] [PubMed] [Google Scholar]
- 44.Adami HO, McLaughlin JK, Hsing AW, Wolk A, Ekbom A, Holmberg L, et al. Alcoholism and cancer risk: a population-based cohort study. Cancer Causes Control. 1992;3:419–425. doi: 10.1007/BF00051354. [DOI] [PubMed] [Google Scholar]
- 45.Veierod MB, Thelle DS, Laake P. Diet and risk of cutaneous malignant melanoma: a prospective study of 50,757 Norwegian men and women. Int J Cancer. 1997;71:600–604. doi: 10.1002/(sici)1097-0215(19970516)71:4<600::aid-ijc15>3.0.co;2-f. [DOI] [PubMed] [Google Scholar]
- 46.Rota M, Pasquali E, Bellocco R, Bagnardi V, Scotti L, Islami F, et al. Alcohol drinking and cutaneous melanoma risk: a systemic review and dose-risk meta-analysis. Br J Dermatol. 2014;170:1021–1028. doi: 10.1111/bjd.12856. [DOI] [PubMed] [Google Scholar]
- 47.Miura K, Zens M, Peart T, Holly E, Berwick M, Gallagher R, et al. Alcohol consumption and risk of melanoma among women: pooled analysis of eight case control studies. Arch Dermatol Research. 2015;307:819–828. doi: 10.1007/s00403-015-1591-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Giovannucci E, Colditz G, Stampfer MJ, Rimm EB, Litin L, Sampson L, et al. The assessment of alcohol consumption by a simple self-administered questionnaire. Am J Epidemiol. 1991;133:810–817. doi: 10.1093/oxfordjournals.aje.a115960. [DOI] [PubMed] [Google Scholar]
- 49.Holick Crystal N, Smith Scott G, Giovannucci Edward, Michaud Dominique S. Coffee, tea, caffeine intake and risk of adult glioma in 3 prospective cohort studies. Cancer Epidemiol Biomarkers Prev. 2010 Jan;19(1):39–47. doi: 10.1158/1055-9965.EPI-09-0732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Rimm EB, Stampfer MJ, Colditz GA, Chute CG, Litin LB, Willett WC. Validity of self-reported waist and hip circumferences in men and women. Epidemiology. 1990;1:466–473. doi: 10.1097/00001648-199011000-00009. [DOI] [PubMed] [Google Scholar]
- 51.Smalberger GJ, Siegel DM, Khachemoune A. Lentigo Maligna. Dermatologic Therapy. 2008;21:439–446. doi: 10.1111/j.1529-8019.2008.00244.x. [DOI] [PubMed] [Google Scholar]
- 52.Istvan J, Matarazzo JD. Tobacco, alcohol, and caffeine use: A review of their interrelationships. Psychol Bull. 1984;95:301–326. [PubMed] [Google Scholar]
- 53.Saladi RN, Nektalova T, Fox JL. Induction of skin carcinogenicity by alcohol and ultraviolet light. Clin Exp Dermatol. 2010;35:7–11. doi: 10.1111/j.1365-2230.2009.03465.x. [DOI] [PubMed] [Google Scholar]
- 54.Baraona E, Abittan CS, Dohmen K, Moretti M, Pozzato G, Chayes ZW, Schaefer C, Lieber CS. Gender differences in pharmacokinetics of alcohol. Alcohol Clin Exp Res. 2001;25:502–507. [PubMed] [Google Scholar]
- 55.Frezza M, di Padova C, Pozzato G, Terpin M, Baraona E, Lieber CS. High blood alcohol levels in women. The role of decreased gastric alcohol dehydrogenase activity and first-pass metabolism. NEJM. 1990;322:95–99. doi: 10.1056/NEJM199001113220205. [DOI] [PubMed] [Google Scholar]
- 56.Fukunaga T, Sillanaukee P, Eriksson CJ. Occurrence of blood acetaldehyde in women during ethanol intoxication: preliminary findings. Alcohol Clin Exp Res. 1993;17:1198–1200. doi: 10.1111/j.1530-0277.1993.tb05228.x. [DOI] [PubMed] [Google Scholar]
- 57.Prabhu A, Obi KO, Rubenstein JH. The Synergistic Effects of Alcohol and Tobacco Consumption on the Risk of Esophageal Squamous Cell Carcinoma: A Meta-Analysis. Am J Gastroenterol. 2014;109:822–827. doi: 10.1038/ajg.2014.71. [DOI] [PubMed] [Google Scholar]
- 58.Cho E, Rosner BA, Colditz GA. Risk Factors for Melanoma by Body Site for Whites. Cancer Epidemiol Biomarkers Prev. 2005;14:1241–1244. doi: 10.1158/1055-9965.EPI-04-0632. [DOI] [PubMed] [Google Scholar]
- 59.Kubo JT, Henderson MT, Desai M, Wactawski-Wende J, Sefanick ML, Tang JY. Alcohol consumption and risk of melanoma and non-melanoma skin cancer in the Women’s Health Initiative. Cancer Causes Control. 2014;25:1–10. doi: 10.1007/s10552-013-0280-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kamei H, Hashimoto Y, Koide T, Kojima T, Hasegawa M. Anti-tumor Effect of Methanol Extracts from Red and White Wines. Cancer Biotherapy and Radiopharmaceuticals. 1998;13:447–452. doi: 10.1089/cbr.1998.13.447. [DOI] [PubMed] [Google Scholar]
- 61.Baur JA, Sinclair DA. Therapeutic potential of resveratrol: the in vivo evidence. Nature Reviews Drug Discovery. 2006;5:493–506. doi: 10.1038/nrd2060. [DOI] [PubMed] [Google Scholar]
- 62.Damianaki A, Bakogeorgou E, Kampa M, Notas G, Hatzoglou A, Panagiotou S, et al. Potent Inhibitory Action of Red Wine Polyphenols on Human Breast Cancer Cells. JCB. 2000;78:429–441. doi: 10.1002/1097-4644(20000901)78:3<429::aid-jcb8>3.0.co;2-m. [DOI] [PubMed] [Google Scholar]
- 63.Wichramasinghe SN, Hasan R, Khalpey Z. Differences in serum levels of acetaldehyde and cytotoxic acetaldehyde-albumin complexes after the consumption of red and white wine: in vitro effects of flavenoids, vitamin E, and other dietary antioxidants on cytotoxic complexes. Alcohol Clin Exp Res. 1996;20:799–803. doi: 10.1111/j.1530-0277.1996.tb05254.x. [DOI] [PubMed] [Google Scholar]
- 64.Schoonen WM, Salinas CA, Kiemeney LA, Stanford JL. Alcohol consumption and risk of prostate cancer in middle-aged men. Int J Cancer. 2005;113:133–140. doi: 10.1002/ijc.20528. [DOI] [PubMed] [Google Scholar]
- 65.Velicer CM, Kristal A, White E. Alcohol Use and the Risk of Prostate Cancer: Results From the VITAL Cohort Study. Nutr Cancer. 2006;56:50–56. doi: 10.1207/s15327914nc5601_7. [DOI] [PubMed] [Google Scholar]
- 66.Ruano-Ravina A, Figueiras A, Barros-Dios JM. Type of wine and risk of lung cancer: a case-control study in Spain. Thorax. 2004;59:981–985. doi: 10.1136/thx.2003.018861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Schurrman AG, Goldbohm RA, van den Brandt PA. A pospective cohort study on consumption of alcoholic beverages in relation to prostate cancer incidence (The Netherlands) Cancer Causes Control. 1999;10:597–605. doi: 10.1023/a:1008925103542. [DOI] [PubMed] [Google Scholar]
- 68.Murtaugh MA, Ma KN, Caan BJ, Slattery ML. Association of fluids from beverages with risk of rectal cancer. Nutr Cancer. 2004;49:25–31. doi: 10.1207/s15327914nc4901_4. [DOI] [PubMed] [Google Scholar]
- 69.Newcomb PA, Nichols HB, Beasley JM, Egan K, Titus-Ernstoff L, Hampton JM, et al. No Difference Between Red Wine or White Wine Consumption and Breast Cancer Risk. Cancer Epidemiol Biomarkers Prev. 2009;18:1007. doi: 10.1158/1055-9965.EPI-08-0801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Farrow DC, Davis S. Risk of pancreatic cancer in relation to medical history and the use of tobacco, alcohol and coffee. Int J Cancer. 1990;45:816–820. doi: 10.1002/ijc.2910450504. [DOI] [PubMed] [Google Scholar]
- 71.Yokoyama A, Tsutsumi E, Imazeki H, Suwa Y, Nakamura C, Mizukami T, et al. Salivary acetaldehyde concentration according to alcoholic beverage consumed and aldehyde dehydrogenase-2 genotype. Alcohol Clin Exp Res. 2008;32:1607–1614. doi: 10.1111/j.1530-0277.2008.00739.x. [DOI] [PubMed] [Google Scholar]
- 72.Vakevainen S, Tillonen J, Agarwal DP, Srivastava N, Salaspuro M. High salivary acetaldehyde after a moderate dose of alcohol in ALDH2-deficient subjects: strong evidence for the local carcinogenic action of acetaldehyde. Alcohol Clin Exp Res. 2000;24:873–877. [PubMed] [Google Scholar]
- 73.Vakevainen S, Tillonen J, Salaspuro M. 4-Methylpyrazole decreases salivary acetaldehyde levels in aldh2-deficient subjects but not in subjects with normal aldh2. Alcohol Clin Exp Res. 2001;25:829–834. [PubMed] [Google Scholar]
- 74.Matsuda T, Yabushita H, Kanaly RA, Shibutani S, Yokoyama A. Increased DNA damage in ALDH2-deficient alcoholics. Chem Res Toxicol. 2006;19:1374–1378. doi: 10.1021/tx060113h. [DOI] [PubMed] [Google Scholar]
- 75.Lewis SJ, Smith GD. Alcohol, ALDH2, and esophageal cancer: a meta-analysis which illustrates the potentials and limitations of a Mendelian randomization approach. Cancer Epidemiol Biomarkers Prev. 2005;14:1967–1971. doi: 10.1158/1055-9965.EPI-05-0196. [DOI] [PubMed] [Google Scholar]
- 76.Yokoyama A, Muramatsu T, Ohmori T, Yokoyama T, Okuyama K, Takahashi H, et al. Alcohol-related cancers and aldehyde dehydrogenase-2 in Japanese alcoholics. Carcinogenesis. 1998;19:1383–1387. doi: 10.1093/carcin/19.8.1383. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.