Abstract
Background and objective
There is limited knowledge about the prevalence of pain and its relation to comorbidities, medication, and certain lifestyle factors in older adults. To address this limitation, this cross-sectional study examined the spreading of pain on the body in a sample of 6611 subjects ≥65 years old (mean age = 75.0 years; standard deviation [SD] = 7.7) living in southeastern Sweden.
Methods
Sex, age, comorbidities, medication, nicotine, alcohol intake, and physical activity were analyzed in relation to the following pain categories: local pain (LP) (24.1%), regional pain medium (RP-Medium) (20.3%), regional pain heavy (RP-Heavy) (5.2%), and widespread pain (WSP) (1.7%).
Results
RP-Medium, RP-Heavy, and WSP were associated more strongly with women than with men (all p<0.01). RP-Heavy was less likely in the 80–84 and >85 age groups compared to the 65–69 age group (both p<0.01). Traumatic injuries, rheumatoid arthritis/osteoarthritis, and analgesics were associated with all pain categories (all p<0.001). An association with gastrointestinal disorders was found in LP, RP-Medium, and RP-Heavy (all p<0.01). Depressive disorders were associated with all pain categories, except for LP (all p<0.05). Disorders of the central nervous system were associated with both RP-Heavy and WSP (all p<0.05). Medication for peripheral vascular disorders was associated with RP-Medium (p<0.05), and hypnotics were associated with RP-Heavy (p<0.01).
Conclusion
More than 50% of older adults suffered from different pain spread categories. Women were more likely to experience greater spreading of pain than men. A noteworthy number of common comorbidities and medications were associated with increased likelihood of pain spread from LP to RP-Medium, RP-Heavy, and WSP. Effective management plans should consider these observed associations to improve functional deficiency and decrease spreading of pain-related disability in older adults.
Keywords: pain spreading, chronic pain, older adults, aging, medication, comorbidity
Introduction
Pain is a common cause of disability in older adults.1–6 Among its consequences are risk of future fall injuries,7 reduced life satisfaction and quality of life, increased mood disturbances, impairments in activities of daily living, and increased health care utilization and costs.2,4,8–10 Previous studies on pain in older adults have reported prevalence from 24% to 72%,1,10–14 a variability that may be due to methodological dissimilarities, case definitions, and cultural differences across studies.9,11,15,16 While it is broadly accepted that pain prevalence is higher in the elderly than in younger ages,17,18 a recent review argues that a general estimation of pain prevalence in the elderly is not feasible.19 Hence, there is a need for epidemiological studies to investigate the prevalence of pain in the elderly.
On the other hand, pain intensity, pain frequency, and the extent of spreading of pain on the body (i.e., the spatial distribution) influence the impact and the consequences of pain.12,20–28 Widespread pain (WSP), in particular, is associated with lower levels of quality of life,28 decreased functionality and work status,29,30 and greater future disability.30 However, recent evidence suggests that not only WSP but also conditions with more limited pain spread can promote significant disabilities in the elderly.24 In fact, the majority of patients with pain cannot be classified into the WSP subgroup, and in order to cover the whole spectrum of “widespreadness”, some studies have used the number of pain sites based on a body manikin.30 As pointed out by Grimby-Ekman et al,24 identifying the number of painful sites may have advantages in epidemiological studies but lacks validity in a clinical setting. Spatial categories of pain used clinically are local pain (LP), regional pain (RP), and WSP.31,32 A recent epidemiological study found that the vast majority of patients with pain had a regional pain condition; the study also suggested that future analyses should split this group into two categories, an approach used by a large study from the United Kingdom.33 Few studies have investigated the prevalence of pain spread categories in the elderly, and even fewer studies have performed in-depth analyses of the associations of pain spread.1,10,15,16,34,35
The link between pain and sex is unambiguous; women demonstrate higher prevalence of pain than men.1,12,29,36–38 However, the relationship between age and pain remains controversial; some studies demonstrate that pain prevalence decreases with age, while others support an opposing association.10,12,34 Some others, though, suggest that there is no concrete association between pain and age.1,39 Furthermore, several studies indicate that pain, especially WSP, is associated with increased prevalence of psychological comorbidities (e.g., depression and anxiety) as well as somatic conditions (e.g., cardiovascular disorders [CVDs], hypertension, and diabetes).33,40–42 It is not known if such conditions are comorbidities to pain, including different pain categories, also in the elderly. Pain and possible comorbidities in older adults are often associated with medications and lifestyle factors such as nicotine, alcohol intake, and physical activity, factors that are usually not addressed in the epidemiological studies.1,34
Awareness of the spreading of pain on the body in older adults is crucial for clinical evaluation and for pain prevention and management. To this end, this study aimed to estimate the prevalence of different pain categories based on the pain spread on the body and to analyze how sex, age, comorbidities, medications, nicotine, alcohol intake, and physical activity are associated with these pain categories.
Methods
Throughout this article, the terms older adults and/or elderly will refer to individuals who are ≥65 years old.43
Subjects and procedure
This cross-sectional study collected data from a sampling frame based on the Swedish Total Population Register (STPR) for the two largest cities (Linköping and Norrköping) of Östergötland County, located in southeastern Sweden. The STPR consists of ~49320 older adults. From this registry, a stratified sample of 2000 subjects was randomly selected based on five age strata (65–69 years, 70–74 years, 75–79 years, 80–84 years, and 85 years and older). This procedure resulted in a total sample of 10000 eligible subjects.8 A postal survey was conducted in October 2012 followed by up to two postal reminders (at 2-week intervals). The collection of surveys closed in January 2013. Data were collected by Statistics Sweden (SCB). The study was approved by the Regional Ethics Research Committee in Linköping, Sweden (Dnr: 2012/154-31). Completion of postal survey was deemed to be agreement of patient informed consent.
Assessments
Pain categories based on spreading of pain on the body
The subjects marked the site of their pain on a body manikin divided into a total of 45 sections on the front and on the back.24 Based on these 45 sections, 23 anatomical regions were determined and a total pain index score, ranging from 0 to 23, was calculated. Using a slightly modified definition developed by MacFarlane et al,44 we defined WSP as pain in at least two sections in two contralateral limbs and the axial skeleton that were equally marked on the front and the back of the manikin. MacFarlane et al44 defined WSP in limbs to be present “if there are at least two painful sections (in two contralateral limbs)”, a definition that does not require pain to be marked equally on the front and back of the manikin. Therefore, our study employs a more rigorous definition of WSP than the American College of Rheumatology (ACR) criteria.45
In addition to WSP, we defined the following pain categories: 1) no pain (NP) if the participants reported zero anatomical sites with pain (i.e., pain index = 0) – zero duration of pain in months and zero pain intensity (this group serves as a control group); 2) LP if the participants reported 1–2 anatomical sites with pain; 3) regional pain medium (RP-Medium) if the participants reported 3–6 anatomical sites with pain; and 4) regional pain heavy (RP-Heavy) if the participants reported 7–17 anatomical pain sites with pain but not fulfilling the WSP criteria. WSP – a common clinical entity – depends both on the number of pain sites and on their spatial distribution. A minority of subjects with RP-Heavy could have a higher number of pain sites than some of the subjects with WSP24; however, the 95% confidence intervals for the number of pain sites clearly differed between RP-Heavy and WSP. Subsequently, we also captured the duration of pain in months. Respondents who reported subacute pain (i.e., pain <3 months) were also included in the aforementioned pain categories. Additionally, we assessed the mean pain intensity using an eleven-point Numeric Rating Scale (NRS7d): 0 = no pain and 10 = worst imaginable pain.46
Basic demographic data
Sex and age were recorded from the respondents’ answers in the postal survey.
Comorbidities
The evaluation of comorbidities was based on a 12-item self-reported questionnaire covering a wide range of common comorbidities as follows: 1) traumatic injuries; 2) rheumatoid arthritis and osteoarthritis; 3) CVDs (including hypertension and coronary artery diseases); 4) pulmonary disorders; 5) depressive disorders; 6) anxiety disorders; 7) gastrointestinal disorders; 8) disorders of the central nervous system (CNS), including ophthalmological and ear–nose–throat disorders; 9) urogenital disorders; 10) skin disorders; 11) tumors and cancer; and 12) metabolic disorders (including diabetes, obesity, anorexia, bulimia, and goiter).
These comorbidities were reported on a five-point scale: 1) No; 2) Yes, according to both my own and my doctor’s opinions; 3) Yes, according to my own opinion; 4) Yes, according to my doctor’s opinion; and 5) Do not know. We combined the answers from 2, 3, and 4 into the category “Yes” in order to have a robust measurement of the presence of a certain comorbidity vs the answer option “No”. The answer option “Do not know” was recorded as missing.
Medications
The assessment of medications was divided into two parts. In the first part, the subjects were asked to report the prescribed medications they had taken during the previous 2 weeks: 1) analgesics; 2) gastroesophageal reflux medication; 3) constipation medication; 4) hypnotics; 5) anxiolytics; 6) antidepressants; 7) anti-inflammatory medication; 8) cardiovascular medication; 9) medication for peripheral vascular disorders; 10) medication for skin disorders; and 11) insulin. In the second part, we recorded the non-prescribed medications during the previous 2 weeks: 1) analgesics; 2) gastroesophageal reflux medication; 3) constipation medication; 4) anti-inflammatory medication; 5) medication for skin disorders; 6) asthma medication; 7) general improvement of vitality supplements; 8) nutrition deficiencies (including B, C, and D vitamins); and 9) herbal remedies used for specific complaints and health problems. Each item had three alternatives: “No”, “Yes, occasionally”, and “Yes, more or less every day”. In the analyses, these items were dichotomized – “Yes, more or less every day” vs the two other alternatives.
Nicotine and alcohol intake
From the instrument the Health Curve (Hälsokurvan), two questions were chosen concerning nicotine intake and frequency. The following questions were asked: 1) “Do you smoke?” and 2) “Do you use snuff?” The alternative answers were coded on a five-point scale: 1 = never and 5 = daily habit.47 For analytical purposes, we combined smoking and snuff habits into the category nicotine intake, and these answers were dichotomized as follows: “never” vs the four other alternatives. One question concerned alcohol intake: “Do you drink alcohol regularly?” The alternative answers were “Yes” and “No”.
Physical activity
Physical activity was measured with the Godin Leisure-Time Exercise Questionnaire (GLTEQ).48 The GLTEQ contains four questions where the respondents state how many times a week they participate in “strenuous”, “moderate”, and “mild” exercise. A total leisure activity score is calculated by the times per week stated for the different intensities multiplied by nine for strenuous, five for moderate, and three for mild. A high score indicates higher intensity and higher frequency of weekly leisure-time activities.48
Data analysis
Data were analyzed using SPSS version 23.0 for Windows (IBM Corporation, Armonk, NY, USA). All tests were two-tailed and used a conventional threshold of a=0.05 for significance. All statistical analyses were weighted to account for unequal possibilities of sample selection by weighting cases regarding age strata, sex, and city. The sampling weights were calculated by Statistics Sweden (SCB). Descriptive statistics were used to present all the variables of interest. We presented mean ± standard deviation (SD) for continuous variables and n (%) for categorical variables. Analysis of variance, Student’s unpaired t-test, and Fisher’s exact test were used where appropriate.
Logistic regression models were used to estimate odds ratios (ORs) and 95% confidence intervals (CIs) on the effects of sex and age, comorbidities, medications, nicotine, alcohol intake, and physical activity in each pain category vs no pain. To determine which variables should be entered into the full multivariate model, univariate logistic regression analyses were performed with each variable one at a time (single-predictor model). Only variables with p<0.05 in the single-predictor model were included in the full multivariate model. We further examined problems of multicollinearity among the independent categorical variables using the phi coefficient (Φ). In the case of high correlation (Φ≥0.30)49 between pairs of variables, only one variable was entered into the full multivariate model. In all the regression models, however, we excluded the pain intensity, non-prescribed analgesics, as well as non-prescribed gastroesophageal reflux medication, constipation medication, anti-inflammatory medication, and medication for skin disorders due to the high correlation with the pain categories and these prescribed medications. The result of this procedure was examined by reanalyzing the multivariate model with all variables together (correlated variables and not) in one model; both analyses were compared. The goodness of fit of the full multivariate model was examined using the Hosmer–Lemeshow test.
Results
Descriptive characteristics
The final sample consisted of 6611 respondents (response rate: 66.1%; 128 excluded because of missing data). The average age was 75.0 years (SD=7.7), and 3554 respondents (53.8%) were women. There was a predominance of nonsmokers/non-snuff users (84.9%) and alcohol users (72.0%). The mean score of physical activity was 21.5 (SD=20.9), and the mean pain intensity was 4.3 (SD=2.4) (Table 1). The most common comorbidities were CVDs (53.8%), rheumatoid arthritis/osteoarthritis (37.0%), and disorders of the CNS (39.5%). The most common medications were prescribed cardiovascular medication (31.3%) and non-prescribed nutritional deficiency medications (21.3%) (Table 2).
Table 1.
Descriptive characteristics of the study in the total sample, in the different pain categories, and comparisons among them.
| Variables (n, %) (otherwise stated) |
Total sample (n=6611) |
NP (n=3227) | LP (n=1585) | RP-Medium (n=1344) |
RP-Heavy (n=341) |
WSP (n=114) | Differences among pain categories (p-value) |
|---|---|---|---|---|---|---|---|
| Sex (n=6611) | <0.001 | ||||||
| Men | 3057 (46.2) | 1665 (50.0) | 755 (46.3) | 519 (37.1) | 104 (28.1) | 24 (22.2) | |
| Women | 3554 (53.8) | 1572 (50.0) | 830 (53.7) | 825 (62.9) | 237 (71.9) | 90 (77.8) | |
| Age strata, years (n=6611) | 0.209 | ||||||
| 65–69 | 1488 (31.6) | 724 (31.5) | 377 (33.3) | 292 (30.5) | 70 (28.7) | 25 (30.4) | |
| 70–74 | 1496 (22.7) | 730 (22.8) | 341 (21.5) | 320 (23.9) | 82 (24.2) | 23 (20.2) | |
| 75–79 | 1397 (17.4) | 655 (16.6) | 337 (17.4) | 279 (17.2) | 90 (21.9) | 36 (26.4) | |
| 80–84 | 1251 (13.6) | 622 (13.8) | 305 (13.7) | 258 (14.0) | 50 (10.5) | 16 (10.6) | |
| ≥85 | 979 (14.7) | 496 (15.3) | 225 (14.1) | 195 (14.4) | 49 (14.7) | 14 (12.4) | |
| NRS7d (n=3660) | |||||||
| Mean (±SD) | 4.3 (±2.4) | 0.0 (0.0) | 4.3 (±1.9) | 5.0 (±1.9) | 5.6 (±1.9) | 6.0 (±2.3) | <0.001 |
| Range | 0–10 | 0–0 | 1–10 | 1–10 | 1–10 | 1–10 | |
| Nicotine intake (n=3868) | 0.892 | ||||||
| Yes | 552 (15.1) | 281 (15.5) | 129 (14.7) | 108 (15.2) | 24 (12.9) | 10 (13.5) | |
| No | 3316 (84.9) | 1643 (84.5) | 785 (85.3) | 643 (84.8) | 182 (87.1) | 63 (86.5) | |
| Alcohol intake (n=6474) | <0.001 | ||||||
| Yes | 4572 (72.0) | 2272 (73.3) | 1121 (74.0) | 908 (70.0) | 215 (65.1) | 56 (51.0) | |
| No | 1902 (28.0) | 887 (26.7) | 429 (26.0) | 412 (30.0) | 117 (34.9) | 57 (49.0) | |
| Physical activity (n=5073) | |||||||
| Mean (±SD) | 21.5 (±20.9) | 22.9 (±21.6) | 20.5 (±19.2) | 20.3 (±19.8) | 19.6 (±19.7) | 17.5 (±18.8) | <0.001 |
| Range | 0–255 | 0–255 | 0–158 | 0–144 | 0–119 | 0–90 | |
Notes: NRS7d, pain intensity for the previous 7 days measured by numeric rating scale. Significant p-values are in bold.
Abbreviations: NP, no pain; LP, local pain; RP-Medium, regional pain medium; RP-Heavy, regional pain heavy; WSP, widespread pain; SD, standard deviation.
Table 2.
Prevalence of comorbidities and medications (prescribed and non-prescribed) in the total sample, in the different pain categories, and comparisons among them
| Variables (n, %) | Total sample (n=6611) |
NP (n=3227) | LP (n=1585) | RP-Medium (n=1344) |
RP-Heavy (n=341) |
WSP (n=114) |
Differences among pain categories (p-value) |
|---|---|---|---|---|---|---|---|
| Comorbidities | |||||||
| Traumatic injuries | 636 (10.8) | 158 (5.2) | 165(11.6) | 215 (19.2) | 71 (24.1) | 27 (23.1) | <0.001 |
| Rheumatoid arthritis and osteoarthritis | 2165 (37.0) | 474 (16.0) | 587 (42.4) | 772 (66.0) | 240 (79.0) | 92 (87.8) | <0.001 |
| Cardiovascular disorders | 3319 (53.8) | 1502 (50.0) | 791(53.8) | 745 (58.8) | 209 (66.1) | 72 (63.7) | <0.001 |
| Pulmonary disorders | 879 (14.5) | 332 (11.2) | 194(13.3) | 241 (19.5) | 83 (27.8) | 29 (27.9) | <0.001 |
| Depressive disorders | 789 (13.6) | 277 (9.5) | 196 (13.8) | 221 (19.7) | 64 (21.8) | 31 (31.9) | <0.001 |
| Anxiety disorders | 995 (16.7) | 355 (11.9) | 229 (16.0) | 279 (23.8) | 84 (27.8) | 48(47.1) | <0.001 |
| Gastrointestinal disorders | 1255 (20.9) | 363 (12.1) | 327 (22.8) | 383 (32.0) | 132 (42.7) | 50 (46.6) | <0.001 |
| Disorders of CNS | 2524 (39.5) | 1040 (33.0) | 614 (40.6) | 627 (48.0) | 180 (55.5) | 63 (60.8) | <0.001 |
| Urogenital disorders | 651 (10.1) | 236 (7.4) | 167 (11.1) | 163 (12.2) | 61 (19.2) | 24 (22.1) | <0.001 |
| Skin disorders | 712 (11.8) | 257 (9.3) | 185 (12.5) | 172 (14.0) | 61 (19.9) | 19 (19.4) | <0.001 |
| Tumors and cancer | 505 (8.1) | 215 (7.0) | 127 (8.5) | 116 (9.3) | 38 (12.5) | 9 (8.6) | 0.007 |
| Metabolic disorders | 1059 (17.0) | 423 (13.9) | 247 (16.2) | 270 (21.5) | 85 (27.8) | 34 (31.8) | <0.001 |
| Prescribed medication | |||||||
| Analgesics | 1031 (15.7) | 149 (4.5) | 272 (17.0) | 391 (29.4) | 149 (44.0) | 70 (63.0) | <0.001 |
| Gastroesophageal reflux medication | 537 (8.7) | 156 (5.1) | 124 (8.2) | 178 (14.2) | 57 (17.7) | 22 (22.4) | <0.001 |
| Constipation medication | 364 (5.8) | 121 (3.9) | 75 (4.9) | 118 (9.3) | 35 (10.7) | 15 (13.2) | <0.001 |
| Hypnotics | 504 (8.0) | 176 (5.8) | 122 (7.9) | 134 (10.5) | 50 (15.3) | 22 (20.3) | <0.001 |
| Anxiolytics | 333 (5.4) | 110 (3.6) | 80 (5.4) | 98 (7.9) | 27 (8.8) | 18 (17.0) | <0.001 |
| Antidepressants | 291 (4.8) | 107 (3.7) | 68 (4.6) | 79 (6.4) | 21 (6.8) | 16 (15.2) | <0.001 |
| Anti-inflammatory medication | 442 (7.3) | 99 (3.3) | 106 (7.1) | 144 (11.6) | 66 (21.8) | 27 (26.3) | <0.001 |
| Cardiovascular medication | 2015 (31.3) | 861 (27.3) | 498 (32.5) | 481 (36.8) | 128 (38.3) | 47 (39.7) | <0.001 |
| Medication for peripheral vascular disorders | 645 (10.3) | 241 (7.9) | 153 (10.2) | 183 (14.4) | 51 (15.5) | 17 (15.1) | <0.001 |
| Medication for skin disorders | 429 (6.9) | 166 (5.4) | 103 (6.7) | 109 (8.9) | 34 (10.6) | 17 (15.9) | <0.001 |
| Insulin agents | 429 (6.8) | 174 (5.7) | 104 (6.9) | 106 (8.5) | 33 (10.3) | 12 (10.9) | <0.001 |
| Non-prescribed medication | |||||||
| Analgesics | 603 (9.4) | 65 (2.0) | 190 (12.3) | 233 (17.6) | 80 (23.8) | 35 (34.3) | <0.001 |
| Gastroesophageal reflux medication | 259 (4.1) | 74 (2.3) | 64 (4.0) | 87 (6.8) | 24 (7.2) | 10 (11.0) | <0.001 |
| Constipation medication | 258 (4.0) | 84 (2.7) | 58 (3.8) | 84 (6.3) | 25 (7.6) | 7 (6.7) | <0.001 |
| Anti-inflammatory medication | 166 (2.7) | 29 (0.9) | 43 (2.8) | 60 (4.9) | 21 (6.9) | 13 (14.9) | <0.001 |
| Medication for skin disorders | 315 (5.1) | 113 (3.7) | 74 (4.8) | 84 (7.1) | 31 (9.0) | 13 (13.8) | <0.001 |
| Asthma medication | 214 (3.5) | 69 (2.2) | 48 (3.2) | 68 (5.5) | 22 (7.4) | 7 (6.4) | <0.001 |
| Vitality supplements | 159 (2.7) | 57 (2.0) | 37 (2.5) | 39 (3.2) | 21 (7.2) | 5 (4.8) | <0.001 |
| Nutritional deficiencies | 1342 (21.3) | 535(17.5) | 334 (22.0) | 323 (25.3) | 112 (34.6) | 38 (34.6) | <0.001 |
| Herbal remedies | 305 (5.1) | 91 (3.2) | 82 (5.5) | 91(7.6) | 32 (10.2) | 9 (9.8) | <0.001 |
Note: Significant p-values are in bold.
Abbreviations: NP, no pain; LP, local pain; RP-Medium, regional pain medium; RP–Heavy, regional pain heavy; WSP, widespread pain; CNS, central nervous system.
Prevalence of pain and distribution of pain categories
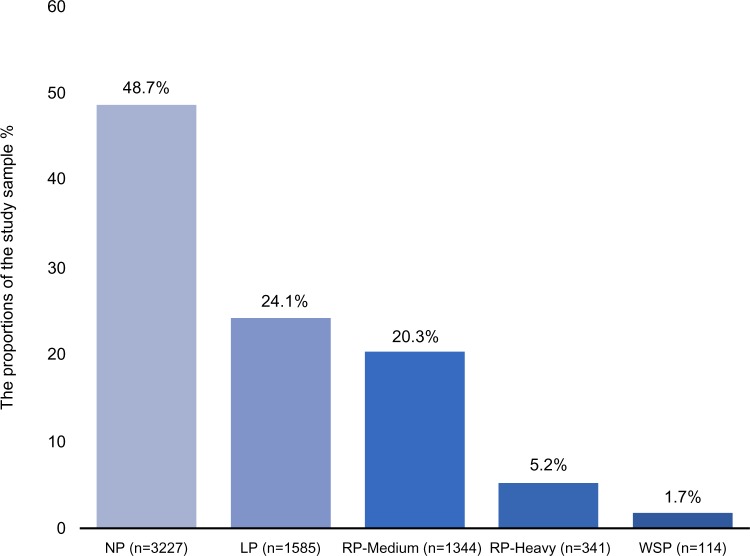
The overall fraction of pain was 51.3%. The criteria for the LP were met by 1585 respondents, a prevalence of 24.1% among all respondents. RP-Medium was reported by 1344 respondents, corresponding to a prevalence of 20.3%. RP-Heavy was reported by 341 respondents, corresponding to a prevalence of 5.2%. With a prevalence of 1.7%, 114 respondents met the criteria of the WSP (Figure 1). Women reported a higher prevalence of all pain categories compared to men (p<0.001). Age distribution did not differ among the five pain categories (p=0.21). As expected, statistically significant differences were found among the pain categories with respect to pain intensity (p<0.001) (Table 1).
Figure 1.
Distribution of pain categories based on spreading of pain on the body.
Abbreviations: NP, no pain; LP, local pain, RP-Medium, regional pain medium; RP-Heavy, regional pain heavy; WSP, widespread pain.
Lifestyle factors in the pain categories
Significant overall differences were found among the five pain categories with regard to alcohol intake and physical activity (both p<0.001). Nicotine intake did not differ significantly among the pain categories (Table 1).
Comorbidities and prescribed and non-prescribed medications in the pain categories
Prominent and significant differences were found between the five pain categories and the percentages of the comorbidities and the medications (almost all p<0.001) (Table 2). Hence, in WSP, higher prevalence of comorbidities and medications was observed compared to the other categories with few exceptions (Table 2).
Results of single-predictor analysis
The single-predictor analysis is presented in Table 3. As expected, spreading of pain was associated with women more strongly than with men (all p<0.001). In respondents 75–79 years old, compared to those 65–69 years old, PR-Heavy was more common than in those without pain (p=0.02). All pain categories were positively associated with almost all comorbidities and medications. Nicotine intake was not associated with the pain categories. Alcohol intake was lower in the pain groups than in those without pain. Physical activity was negatively associated with all pain categories.
Table 3.
Results of logistic regression analyses: single-predictor models of association of each pain category with sex, age strata, comorbidities, medications, nicotine and alcohol intake, and physical activity
| Variables | LP, OR (95% CI) |
p-Value | RP-Medium, OR (95% CI) | p-Value | RP-Heavy, OR (95% CI) | p-Value | WSP, OR (95% CI) | p-Value |
|---|---|---|---|---|---|---|---|---|
| Women (vs men) | 1.16 (1.03–1.31) | <0.001 | 1.69 (1.48–1.92) | <0.001 | 2.56 (1.99–3.26) | <0.001 | 3.49 (2.24–5.47) | <0.001 |
| Age strata, years | ||||||||
| 70–74 (vs 65–69) | 1.12 (0.91–1.36) | 0.280 | 1.09 (0.92–1.29) | 0.341 | 1.71 (0.86–1.59) | 0.313 | 0.92 (0.54–1.57) | 0.762 |
| 75–79 (vs 65–69) | 1.09 (0.88–1.36) | 0.401 | 1.07 (0.88–1.29) | 0.509 | 1.45 (1.05–1.98) | 0.023 | 1.64 (0.99–2.71) | 0.051 |
| 80–84 (vs 65–69) | 1.14 (0.91–1.42) | 0.275 | 1.05 (0.85–1.28) | 0.661 | 0.83 (0.56–1.24) | 0.369 | 0.88 (0.41–1.54) | 0.503 |
| ≥85 (vs 65–69) | 1.07 (0.83–1.36) | 0.583 | 0.98 (0.81–1.20) | 0.553 | 1.07 (0.75–1.52) | 0.730 | 0.82 (0.45–1.58) | 0.601 |
| Comorbidities (vs no) | ||||||||
| Traumatic injuries | 2.38 (1.89–3.00) | <0.001 | 4.33 (3.47–5.39) | <0.001 | 5.77 (4.22–7.89) | <0.001 | 7.41 (4.57–11.97) | <0.001 |
| Rheumatoid arthritis and osteoarthritis | 3.85 (3.31–4.64) | <0.001 | 10.15 (8.67–11.88) | <0.001 | 19.07 (14.69–26.28) | <0.001 | 37.75 (20.85–68.32) | <0.001 |
| Cardiovascular disorders | 1.16 (1.03–1.32) | 0.018 | 1.42 (1.24–1.63) | <0.001 | 1.94 (1.52–2.49) | <0.001 | 1.75 (1.18–2.61) | 0.005 |
| Pulmonary disorders | 1.21 (1.01–1.47) | 0.046 | 1.91 (1.59–2.30) | <0.001 | 3.05 (2.30–4.03) | <0.001 | 3.07 (1.95–4.83) | <0.001 |
| Depressive disorders | 1.52 (1.24–1.85) | <0.001 | 2.32 (1.91–2.81) | <0.001 | 2.65 (1.94–3.61) | <0.001 | 4.44 (2.83–6.97) | <0.001 |
| Anxiety disorders | 1.41 (1.17–1.68) | <0.001 | 2.31 (1.93–2.47) | <0.001 | 2.83 (2.14–3.4) | <0.001 | 6.55 (4.36–9.85) | <0.001 |
| Gastrointestinal disorders | 2.14 (1.81–2.53) | <0.001 | 3.43 (2.91–4.05) | <0.001 | 5.41 (4.20–6.89) | <0.001 | 6.36 (4.27–9.46) | <0.001 |
| Disorders of CNS | 1.39 (1.22–1.58) | <0.001 | 1.87 (1.63–2.14) | <0.001 | 2.53 (2.00–3.21) | <0.001 | 3.15 (2.11–4.69) | <0.001 |
| Urogenital disorders | 1.56 (1.26–1.95) | <0.001 | 1.75 (1.28–1.70) | <0.001 | 2.99 (2.17–4.12) | <0.001 | 3.56 (2.19–5.79) | <0.001 |
| Skin disorders | 1.38 (1.13–1.69) | 0.001 | 1.58 (1.29–1.93) | <0.001 | 2.40 (1.48–2.68) | <0.001 | 2.33 (1.40–3.88) | 0.001 |
| Tumors and cancer | 1.24 (0.98–1.58) | 0.072 | 1.36 (1.06–1.73) | 0.013 | 1.19 (1.30–2.79) | 0.001 | 1.26 (0.61–2.60) | 0.530 |
| Metabolic disorders | 1.99 (1.01–1.42) | 0.040 | 1.69 (1.43–2.01) | <0.001 | 2.39 (1.83–3.13) | <0.001 | 2.90 (1.89–4.45) | <0.001 |
| Prescribed medication (vs no) | ||||||||
| Analgesics | 4.32 (3.47–5.37) | <0.001 | 8.79 (7.12–10.85) | <0.001 | 16.63 (12.61–21.52) | <0.001 | 35.98 (23.49–55.11) | <0.001 |
| Gastroesophageal reflux medication | 1.66 (1.29–2.12) | <0.001 | 3.06 (2.43–3.85) | <0.001 | 3.96 (2.84–5.15) | <0.001 | 5.32 (3.26–6.76) | <0.001 |
| Constipation medication | 1.25 (0.92–1.69) | 0.143 | 2.50 (1.91–3.27) | <0.001 | 2.93 (1.96–4.39) | <0.001 | 3.72 (2.05–6.73) | <0.001 |
| Hypnotics | 1.39 (1.09–1.77) | 0.007 | 1.89 (1.49–2.40) | <0.001 | 2.92 (2.08–4.12) | <0.001 | 4.13 (2.51–6.77) | <0.001 |
| Anxiolytics | 1.51 (1.13–2.04) | 0.006 | 2.28 (1.72–3.03) | <0.001 | 2.56 (1.66–3.95) | <0.001 | 5.49 (3.18–9.49) | <0.001 |
| Antidepressants | 1.23 (0.98–1.72) | 0.136 | 1.79 (1.32–2.39) | <0.001 | 1.89 (1.17–3.05) | <0.001 | 4.67 (2.65–8.24) | <0.001 |
| Anti-inflammatory medication | 2.22 (1.67–2.95) | <0.001 | 3.78 (2.89–4.39) | <0.001 | 8.05 (5.76–11.26) | <0.001 | 10.28 (6.34–16.65) | <0.001 |
| Cardiovascular medication | 1.28 (1.11–1.47) | <0.001 | 1.55 (1.34–1.78) | <0.001 | 1.65 (1.30–2.10) | <0.001 | 1.75 (1.18–2.59) | 0.005 |
| Medication for peripheral vascular disorders | 1.32 (1.07–1.65) | 0.011 | 1.97 (1.61–2.43) | <0.001 | 2.14 (1.53–2.99) | <0.001 | 2.08 (1.19–3.62) | 0.010 |
| Medication for skin disorders | 1.25 (0.96–1.62) | 0.091 | 1.69 (1.31–2.17) | <0.001 | 2.06 (1.39–3.04) | <0.001 | 3.29 (1.92–5.63) | <0.001 |
| Insulin agents | 1.22 (0.95–1.58) | 0.115 | 1.54 (1.19–1.98) | 0.001 | 1.99 (1.29–2.85) | <0.001 | 2.04 (1.08–3.84) | 0.027 |
| Non-prescribed medication (vs no) | ||||||||
| Asthma medication | 1.45 (0.99–2.13) | 0.052 | 2.58(1.82–3.65) | <0.001 | 3.55 (2.18–5.79) | <0.001 | 3.04 (1.34–6.91) | 0.008 |
| Vitality supplements | 1.26 (0.84–1.92) | 0.263 | 1.63 (1.09–2.45) | <0.001 | 3.78 (2.30–6.23) | <0.001 | 2.48 (0.99–6.23) | 0.053 |
| Nutritional deficiencies | 1.33 (1.14–1.55) | <0.001 | 1.60 (1.36–1.87) | <0.001 | 2.50 (1.95–3.20) | <0.001 | 2.51 (1.66–3.78) | <0.001 |
| Herbal remedies | 1.76 (1.30–2.39) | <0.001 | 2.47 (1.84–3.31) | <0.001 | 3.41 (2.25–5.18) | <0.001 | 3.27 (1.68–6.38) | 0.001 |
| Nicotine intake (vs no) | 0.94 (0.75–1.17) | 0.597 | 0.98 (0.77–1.24) | 0.883 | 0.81 (0.53–1.24) | 0.335 | 0.85 (0.43–1.67) | 0.645 |
| Alcohol intake (vs no) | 1.03 (0.90–1.19) | 0.598 | 0.85 (0.73–0.98) | 0.026 | 0.68 (0.54–0.87) | 0.002 | 0.38 (0.26–0.55) | <0.001 |
| Physical activity | 0.99 (0.98–0.99) | 0.001 | 0.99 (0.98–0.99) | 0.001 | 0.99 (0.98 –0.99) | 0.016 | 0.98 (0.97–0.99) | 0.021 |
Note: Significant p-values are in bold.
Abbreviations: LP, local pain; OR, odds ratio; CI, confidence interval; RP-Medium, regional pain medium; RP-Heavy, regional pain heavy; WSP, widespread pain; CNS, central nervous system.
Results of multivariate analysis
The results of the full multivariate model are presented in Table 4. Being a woman was independently associated with RP-Medium, RP-Heavy, and WSP. RP-Heavy was less likely to occur in 80–84 year-olds and >85 year-olds compared to 65–69 year-olds. Traumatic injuries, rheumatoid arthritis/osteoarthritis, and analgesics were positively associated with all pain categories. An association with gastrointestinal disorders was found in LP, RP-Medium, and RP-Heavy. Depressive disorders were positively associated with all pain categories, except for LP. Disorders of the CNS were positively associated with the RP-Heavy and WSP. Medication for peripheral vascular disorders was positively associated with RP-Medium, and hypnotics were positively associated with RP-Heavy.
Table 4.
Results of logistic regression analyses: multivariate models of association of each pain category with sex, age strata, comorbidities, medications, alcohol intake, and physical activity
| Variables | LP, OR (95% CI) | p-Value | RP-Medium, OR (95% CI) | p-Value | RP-Heavy, OR (95% CI) | p-Value | WSP, OR (95% CI) | p-Value |
|---|---|---|---|---|---|---|---|---|
| Women (vs men) | 1.09 (0.90–1.31) | 0.382 | 1.15 (1.18–1.94) | 0.001 | 2.76 (1.63–4.65) | <0.001 | 3.80 (1.59–9.07) | 0.003 |
| Age strata, years | ||||||||
| 70–74 (vs 65–69) | Not included (p>0.05) | Not included (p>0.05) | 0.87 (0.46–1.65) | 0.677 | Not included (p>0.05) | |||
| 75–79 (vs 65–69) | Not included (p>0.05) | Not included (p>0.05) | 0.81 (0.41–1.61) | 0.557 | Not included (p>0.05) | |||
| 80–84 (vs 65–69) | Not included (p>0.05) | Not included (p>0.05) | 0.24 (0.09–0.59) | 0.002 | Not included (p>0.05) | |||
| ≥85 (vs 65–69) | Not included (p>0.05) | Not included (p>0.05) | 0.23 (0.09–0.55) | 0.001 | Not included (p>0.05) | |||
| Comorbidities (vs no) | ||||||||
| Traumatic injuries | 1.89 (1.30–2.66) | 0.001 | 3.29 (2.22–4.89) | <0.001 | 3.75 (1.96–7.20) | <0.001 | 5.64 (2.17–14.63) | <0.001 |
| Rheumatoid arthritis and osteoarthritis | 3.78 (3.07–4.66) | <0.001 | 8.27 (6.46–10.57) | <0.001 | 10.82 (6.66–17.57) | <0.001 | 10.02 (4.23–22.68) | <0.001 |
| Cardiovascular disorders | 1.06 (0.88–1.28) | 0.535 | 1.13 (0.88–1.44) | 0.341 | 1.12 (0.67–1.86) | 0.663 | 0.58 (0.26–1.32) | 0.198 |
| Pulmonary disorders | 0.83 (0.61–1.21) | 0.224 | 1.33 (0.95–1.86) | 0.097 | 1.23 (0.66–2.28) | 0.518 | 0.75 (0.27–2.10) | 0.591 |
| Depressive disorders | 1.28 (0.94–1.74) | 0.110 | 1.77 (1.23–2.55) | 0.002 | 2.05 (1.08–3.88) | 0.028 | 1.66 (1.23–4.52) | <0.001 |
| Gastrointestinal disorders | 1.42 (1.15–1.75) | 0.001 | 1.96 (1.50–2.55) | <0.001 | 2.14 (1.28–3.55) | 0.003 | 1.50 (0.68–3.34) | 0.313 |
| Disorders of CNS | 1.07 (0.88–1.30) | 0.514 | 1.23 (0.96–1.58) | 0.110 | 1.77 (1.06–2.93) | 0.027 | 2.30 (1.04–5.09) | 0.040 |
| Urogenital disorders | 1.19 (0.85–1.66) | 0.290 | 1.01 (0.64–1.57) | 0.998 | 1.47 (0.66–3.27) | 0.335 | 1.44 (0.49–4.21) | 0.501 |
| Skin disorders | 1.02 (0.77–1.35) | 0.860 | 1.21 (0.86–1.71) | 0.266 | 0.98 (0.51–1.91) | 0.965 | 1.94 (0.77–4.93) | 0.160 |
| Tumors and cancer | Not included (p>0.05) | 1.19 (0.87–1.45) | 0.102 | 1.03 (0.45–2.37) | 0.948 | Not included (p>0.05) | ||
| Metabolic disorders | 0.92 (0.71–1.19) | 0.520 | 1.33 (0.97–1.82) | 0.078 | 1.31 (0.72–2.35) | 0.370 | 1.92 (0.80–4.62) | 0.142 |
| Prescribed medication (vs no) | ||||||||
| Analgesics | 3.58 (2.69–4.77) | <0.001 | 6.22 (4.50–8.85) | <0.001 | 13.95 (8.34–23.35) | <0.001 | 15.03 (6.81–33.19) | <0.001 |
| Constipation medication | Not included (p>0.05) | 1.14 (0.67–1.94) | 0.621 | 1.11 (0.46–2.67) | 0.813 | 0.87 (0.20–3.82) | 0.858 | |
| Hypnotics | 1.25 (0.85–1.85) | 0.252 | 0.99 (0.59–1.64) | 0.962 | 2.83 (1.33–6.03) | 0.007 | 2.16 (0.65–7.12) | 0.204 |
| Medication for peripheral vascular disorders | 1.19 (0.86–1.64) | 0.301 | 1.52 (1.03–2.26) | 0.036 | 1.39 (0.66–2.95) | 0.386 | 0.95 (0.23–3.16) | 0.937 |
| Non-prescribed medication (vs no) | ||||||||
| Vitality supplements | Not included (p>0.05) | 1.00 (0.45–2.19) | 0.999 | 2.38 (0.75–1.77) | 0.135 | Not included (p>0.05) | ||
| Nutritional deficiencies | 0.96 (0.75–1.22) | 0.742 | 1.23 (0.91–1.66) | 0.182 | 1.44 (0.83–2.51) | 0.192 | 1.12 (0.49–2.58) | 0.783 |
| Herbal remedies | 1.15 (0.72–1.82) | 0.554 | 1.43 (0.85–2.40) | 0.169 | 0.95 (0.33–2.69) | 0.922 | 2.71 (0.77–9.53) | 0.120 |
| Alcohol intake (vs no) | Not included (p>0.05) | 1.25 (0.95–1.65) | 0.116 | 1.13 (0.65–1.97) | 0.660 | 0.91 (0.40–2.06) | 0.918 | |
| Physical activity | 0.99 (0.94–1.01) | 0.576 | 1.00 (0.99–1.01) | 0.940 | 0.99 (0.98–1.01) | 0.864 | 1.01 (0.99–1.03) | 0.069 |
| Nagelkerke R2 | 0.184 | 0.451 | 0.567 | 0.541 | ||||
| Hosmer and Lemeshow test (p-value) | 0.058 | 0.257 | 0.245 | 0.665 | ||||
| Classification percentage correct | 73.0 | 83.0 | 94.5 | 97.4 | ||||
Note: Significant p-values are in bold.
Abbreviations: LP, local pain; OR, odds ratio; CI, confidence interval; RP-Medium, regional pain medium; RP-Heavy, regional pain heavy; WSP, widespread pain; CNS, central nervous system.
Discussion
This large-scale cross-sectional study demonstrated an illustrative depiction of the prevalence of pain categories based on pain spread and its relation to basic demographic characteristics, comorbidities, medication, and lifestyle factors in the elderly. The stratified age and the random sampling methodology implemented in this survey are an additional strength. We found a high overall prevalence of spreading of pain (both subacute and chronic); the highest prevalence was observed in LP and the lowest in WSP. Pain intensity was higher in WSP. Female sex had strong correlation with all pain categories, with the exception of LP. Numerous comorbidities and medications were associated with spreading of pain; however, only traumatic injuries, rheumatoid arthritis/osteoarthritis, and analgesics were related to all pain categories. Evidently, the ratios of these associations increased with the pain spread from LP to RP-Medium, RP-Heavy, or WSP.
We found that >50% of older individuals report pain, a rate quite similar compared to the prevalence reported in some other studies of older adults.1,12,14 The highest, and also similar, prevalence was found for LP and RP-Medium, results that are partly consistent with earlier investigations based on spreading of pain.24,31,40 The observation of the remarkably lower prevalence of WSP relative to those reported by other studies24,31,35,40 was somehow unexpected. The Manchester definition of WSP may account for this variability. Indeed, studies using the Manchester criteria have found lower prevalence of WSP than studies using the American College of Rheumatology criteria.15,24,31,40,45 Environmental and genetic factors could also explain the discrepancies in the rate of WSP.50
We further observed that women were more likely to experience greater spreading of pain (i.e., RP-Medium, RP-Heavy, and WSP) than men. This finding is supported by both epidemiological1,12,29,37 and review studies.36,38 Although the exact etiological basis of sex differences regarding pain experience remains unknown, there is evidence supporting that it is due to multiple biological, psychosocial, and cultural variations.36,38 As with some other studies,1,29,34 we found no evidence of an association between age and pain. Despite this lack of evidence, we found peaks for significantly decreased ratios of RP-Heavy in the old old (i.e., 75–84 years old) and oldest old (i.e., ≥85 years old) individuals. Such a decrease in symptomatology might coincide with the idea of fall-off of symptoms with advancing age.50,51 It is reasonable to assume that a certain proportion of the oldest old individuals with severe comorbidities are likely to have passed away at the time of this study. Thus, the survivors might represent a comparatively healthier group where a high prevalence of WSP on the body is less likely to occur.
Not surprisingly, traumatic injuries, rheumatoid arthritis, and osteoarthritis were associated with all pain categories. Regarding the other comorbidities, an association with gastrointestinal disorders was found in LP, RP-Medium, and RP-Heavy. Depressive disorders were associated with all pain categories, except for LP. Disorders of the CNS were associated with the greatest pain spread categories (i.e., RP-Heavy and WSP). These results are consistent with data obtained in cross-sectional and review studies that linked those comorbidities with the development and/or maintenance of WSP.11,16,23,24,27,33,40–42,52,53 The observed strong relationship between those comorbidities and the pain categories can be partly explained by the fact that greater levels of spreading of pain trigger a tendency in health-seeking behavior more frequently than the more limited spreading of pain.24 Another factor might be that WSP (including fibromyalgia syndrome) is associated with the most prominent central alterations (i.e., central hyperexcitability). The correlation between depressive disorders and the spreading of pain in our study might further be attributed to the fact that older adults seem to be vulnerable to depressive symptoms that often correspond to somatic symptoms as a part of aging.34,51,54
Our results do not support previous observations of a clear association between the spreading of pain, cancer, and CVDs.24,31,40 This finding is quite intriguing because there is an ongoing discussion in the literature about their relationship.24,31,40 One possible explanation is that our study evaluates the existence of cancer and CVDs in relation to spreading of pain, rather than examining the mortality risk from those disorders related to regional pain and WSP, as other studies focused on.31,40 Unlike our study, these studies did not take into evaluation medication and other comorbidities that might be independently related to pain, cancer, and CVD mortality.31,40,44,47,55 Additionally, some other factors such as smoking may be associated with pain and both cancer and CVD mortality. There is some evidence that pain is associated with smoking behaviors and vice versa,56,57 a habit that contributes to worsening health status, including cancer and CVD morbidity.56 However, in our study nicotine intake was not associated with pain categories. This finding could reflect the fact that our sample had a relatively low prevalence of nicotine intake. These differences can be partly explained by the population studied and the study design; we examined only older adults using a cross-sectional methodology, and this may account for the observed variations regarding cancer, smoking, CVDs, and pain. Nevertheless, these results require more attention and should be tested longitudinally in future studies.
Various medications such as analgesics, hypnotics, and medication for peripheral vascular disorders were independently associated with different pain spread categories. These results seem to support other empirical studies that have shown that the prevalence of pain is associated with polypharmacy.11,58 Notably, WSP had the strongest relationship with analgesics. It appears that older adults with greater pain spread and multi-comorbidity might have a higher prescription of use of such agents. A second explanation can be related to the fact that the medications concern self-reports and that the respondents may have mixed up analgesics and anti-inflammatory medication. Other lifestyle factors (e.g., alcohol intake and physical activity) did not seem to be important factors of spreading of pain in older adults.
Of note, we found that the associations of pain categories with comorbidities and medications were largely reduced when all the comorbidities and medications were simultaneously controlled for (Table 4). This finding could imply that the factors associated with spreading of pain in older adults are multifactorial. Even if each one of the examined comorbidities and medications seem to be important factors for spreading of pain, it remains unclear how these factors are combined to affect the experience of pain in this population. These results are important issues for future research.
Study limitations
Due to the cross-sectional study design, interpretations on causal associations between studied variables and different pain categories cannot be confirmed. However, our large sample size provides adequate statistical power to derive concrete interpretations. Another limitation is the use of postal questionnaires and the self-reported measurement of pain as well as the use of other examined variables. Obviously, these methods are generally less precise than a doctor’s diagnosis and clinical evaluation.11 Nonetheless, several studies have used analogous approaches to assess the prevalence of pain.6,7,22,24,26,37,40 Finally, we did not examine the role of cognitive function, other socioeconomic factors, or the important interaction between studied variables that might be associated with the pain in older adults.2,11
Conclusion
This study suggests that older adults with compromised health conditions tend to experience greater spreading of pain. Women appear to be more vulnerable to spreading of pain compared to men. Advancing age per se did not play an important role. Therapeutic plans should take into account these associations to improve pain treatments that inhibit functional deficiency and pain spread-related disability in this population. Given that half of older adults in this study had spreading of pain most often with additional comorbidities under multiple medications, there is an apparent need to further explore the nature of the associations reported here.
Acknowledgments
This study was sponsored by a grant from Grünenthal Sweden AB. The sponsor of the study had no role in study design, data collection, data analysis, data interpretation, writing of the report, or the decision to submit for publication. The authors had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Footnotes
Author contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Patel KV, Guralnik JM, Dansie EJ, Turk DC. Prevalence and impact of pain among older adults in the United States: findings from the 2011 National Health and Aging Trends Study. Pain. 2013;154(12):2649–2657. doi: 10.1016/j.pain.2013.07.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Onder G, Cesari M, Russo A, Zamboni V, Bernabei R, Landi F. Association between daily pain and physical function among old-old adults living in the community: results from the ilSIRENTE study. Pain. 2006;121(1–2):53–59. doi: 10.1016/j.pain.2005.12.003. [DOI] [PubMed] [Google Scholar]
- 3.Rice AS, Smith BH, Blyth FM. Pain and the global burden of disease. Pain. 2016;157(4):791–796. doi: 10.1097/j.pain.0000000000000454. [DOI] [PubMed] [Google Scholar]
- 4.Andrews JS, Cenzer IS, Yelin E, Covinsky KE. Pain as a risk factor for disability or death. J Am Geriatr Soc. 2013;61(4):583–589. doi: 10.1111/jgs.12172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Leveille SG, Fried L, Guralnik JM. Disabling symptoms: what do older women report? J Gen Intern Med. 2002;17(10):766–773. doi: 10.1046/j.1525-1497.2002.20229.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Melzer D, Gardener E, Guralnik JM. Mobility disability in the middle-aged: cross-sectional associations in the English Longitudinal Study of ageing. Age Ageing. 2005;34(6):594–602. doi: 10.1093/ageing/afi188. [DOI] [PubMed] [Google Scholar]
- 7.Munch T, Harrison SL, Barrett-Connor E, et al. Pain and falls and fractures in community-dwelling older men. Age Ageing. 2015;44(6):973–979. doi: 10.1093/ageing/afv125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bernfort L, Gerdle B, Rahmqvist M, Husberg M, Levin LÅ. Severity of chronic pain in an elderly population in Sweden – impact on costs and quality of life. Pain. 2015;156(3):521–527. doi: 10.1097/01.j.pain.0000460336.31600.01. [DOI] [PubMed] [Google Scholar]
- 9.Covinsky KE, Lindquist K, Dunlop DD, Yelin E. Pain, functional limitations, and aging. J Am Geriatr Soc. 2009;57(9):1556–1561. doi: 10.1111/j.1532-5415.2009.02388.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jakobsson U, Klevsgård R, Westergren A, Hallberg IR. Old people in pain: a comparative study. J Pain Symptom Manage. 2003;26(1):625–636. doi: 10.1016/s0885-3924(03)00145-3. [DOI] [PubMed] [Google Scholar]
- 11.Helme RD, Gibson SJ. The epidemiology of pain in elderly people. Clin Geriatr Med. 2001;17(3):417–431. doi: 10.1016/s0749-0690(05)70078-1. [DOI] [PubMed] [Google Scholar]
- 12.Andersson HI, Ejlertsson G, Leden I, Rosenberg C. Chronic pain in a geographically defined general population: studies of differences in age, gender, social class, and pain localization. Clin J Pain. 1993;9(3):174–182. doi: 10.1097/00002508-199309000-00004. [DOI] [PubMed] [Google Scholar]
- 13.Blyth FM, March LM, Brnabic AJ, Jorm LR, Williamson M, Cousins MJ. Chronic pain in Australia: a prevalence study. Pain. 2001;89(2–3):127–134. doi: 10.1016/s0304-3959(00)00355-9. [DOI] [PubMed] [Google Scholar]
- 14.Jackson T, Thomas S, Stabile V, Han X, Shotwell M, McQueen K. Prevalence of chronic pain in low-income and middle-income countries: a systematic review and meta-analysis. Lancet. 2015;385(Suppl 2):S10. doi: 10.1016/S0140-6736(15)60805-4. [DOI] [PubMed] [Google Scholar]
- 15.Mansfield KE, Sim J, Jordan JL, Jordan KP. A systematic review and meta-analysis of the prevalence of chronic widespread pain in the general population. Pain. 2016;157(1):55–64. doi: 10.1097/j.pain.0000000000000314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Vellucci R. Heterogeneity of chronic pain. Clin Drug Investig. 2012;32(Suppl 1):3–10. doi: 10.2165/11630030-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 17.Harker J, Reid KJ, Bekkering GE, et al. Epidemiology of chronic pain in Denmark and Sweden. Pain Res Treat. 2012;2012:371248. doi: 10.1155/2012/371248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gereau RW, 4th, Sluka KA, Maixner W, et al. A pain research agenda for the 21st century. J Pain. 2014;15(12):1203–1214. doi: 10.1016/j.jpain.2014.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fejer R, Leboeuf-Yde C. Does back and neck pain become more common as you get older? A systematic literature review. Chiropr Man Therap. 2012;20(1):24. doi: 10.1186/2045-709X-20-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jones GT, Macfarlane GJ. Epidemiology of pain in older persons. In: Gibson SJ, Weiner DK, editors. Pain in Older Persons. Seattle: IASP Press; 2005. pp. 3–22. [Google Scholar]
- 21.Bromley Milton M, Borsbo B, Rovner G, Lundgren-Nilsson A, Stibrant-Sunnerhagen K, Gerdle B. Is pain intensity really that important to assess in chronic pain patients? A study based on the Swedish quality registry for pain rehabilitation (SQRP) PLoS One. 2013;8(6):e65483. doi: 10.1371/journal.pone.0065483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.de Waal MW, den Elzen WP, Achterberg WP, Gussekloo J, Blom JW. A postal screener for pain and need for treatment in older persons in primary care. J Am Geriatr Soc. 2014;62(10):1832–1837. doi: 10.1111/jgs.13064. [DOI] [PubMed] [Google Scholar]
- 23.Denkinger MD, Lukas A, Nikolaus T, Peter R, Franke S, ActiFE Study Group Multisite pain, pain frequency and pain severity are associated with depression in older adults: results from the ActiFE Ulm study. Age Ageing. 2014;43(4):510–514. doi: 10.1093/ageing/afu013. [DOI] [PubMed] [Google Scholar]
- 24.Grimby-Ekman A, Gerdle B, Bjork J, Larsson B. Comorbidities, intensity, frequency and duration of pain, daily functioning and health care seeking in local, regional, and widespread pain – a descriptive population-based survey (SwePain) BMC Musculoskelet Disord. 2015;16:165. doi: 10.1186/s12891-015-0631-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Walton DM, Pretty J, MacDermid JC, Teasell RW. Risk factors for persistent problems following whiplash injury: results of a systematic review and meta-analysis. J Orthop Sports Phys Ther. 2009;39(5):334–350. doi: 10.2519/jospt.2009.2765. [DOI] [PubMed] [Google Scholar]
- 26.Lacey RJ, Belcher J, Rathod T, Wilkie R, Thomas E, McBeth J. Pain at multiple body sites and health-related quality of life in older adults: results from the North Staffordshire Osteoarthritis Project. Rheumatology (Oxford) 2014;53(11):2071–2079. doi: 10.1093/rheumatology/keu240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kamaleri Y, Natvig B, Ihlebaek CM, Benth JS, Bruusgaard D. Number of pain sites is associated with demographic, lifestyle, and health-related factors in the general population. Eur J Pain. 2008;12(6):742–748. doi: 10.1016/j.ejpain.2007.11.005. [DOI] [PubMed] [Google Scholar]
- 28.Kamaleri Y, Natvig B, Ihlebaek CM, Bruusgaard D. Localized or widespread musculoskeletal pain: does it matter? Pain. 2008;138(1):41–46. doi: 10.1016/j.pain.2007.11.002. [DOI] [PubMed] [Google Scholar]
- 29.Gerdle B, Bjork J, Coster L, Henriksson C, Bengtsson A. Prevalence of widespread pain and associations with work status: a population study. BMC Musculoskelet Disord. 2008;9:102. doi: 10.1186/1471-2474-9-102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kamaleri Y, Natvig B, Ihlebaek CM, Bruusgaard D. Does the number of musculoskeletal pain sites predict work disability? A 14-year prospective study. Eur J Pain. 2009;13(4):426–430. doi: 10.1016/j.ejpain.2008.05.009. [DOI] [PubMed] [Google Scholar]
- 31.Mundal I, Gråwe RW, Bjørngaard JH, Linaker OM, Fors EA. Prevalence and long-term predictors of persistent chronic widespread pain in the general population in an 11-year prospective study: the HUNT study. BMC Musculoskelet Disord. 2014;15:213. doi: 10.1186/1471-2474-15-213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Thompson JM. Exercise in muscle pain disorders. PM R. 2012;4(11):889–893. doi: 10.1016/j.pmrj.2012.08.004. [DOI] [PubMed] [Google Scholar]
- 33.Nicholl BI, Mackay D, Cullen B, et al. Chronic multisite pain in major depression and bipolar disorder: cross-sectional study of 149,611 participants in UK Biobank. BMC Psychiatry. 2014;14:350. doi: 10.1186/s12888-014-0350-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bicket MC, Mao J. Chronic pain in older adults. Anesthesiol Clin. 2015;33(3):577–590. doi: 10.1016/j.anclin.2015.05.011. [DOI] [PubMed] [Google Scholar]
- 35.Hunt IM, Silman AJ, Benjamin S, McBeth J, Macfarlane GJ. The prevalence and associated features of chronic widespread pain in the community using the ‘Manchester’ definition of chronic widespread pain. Rheumatology (Oxford) 1999;38(3):275–279. doi: 10.1093/rheumatology/38.3.275. [DOI] [PubMed] [Google Scholar]
- 36.Bartley EJ, Fillingim RB. Sex differences in pain: a brief review of clinical and experimental findings. Br J Anaesth. 2013;111(1):52–58. doi: 10.1093/bja/aet127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bassols A, Bosch F, Campillo M, Cañellas M, Baños JE. An epidemiological comparison of pain complaints in the general population of Catalonia (Spain) Pain. 1999;83(1):9–16. doi: 10.1016/s0304-3959(99)00069-x. [DOI] [PubMed] [Google Scholar]
- 38.Fillingim RB, King CD, Ribeiro-Dasilva MC, Rahim-Williams B, Riley JL., 3rd Sex, gender, and pain: a review of recent clinical and experimental findings. J Pain. 2009;10(5):447–485. doi: 10.1016/j.jpain.2008.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Boggero IA, Geiger PJ, Segerstrom SC, Carlson CR. Pain intensity moderates the relationship between age and pain interference in chronic orofacial pain patients. Exp Aging Res. 2015;41(4):463–474. doi: 10.1080/0361073X.2015.1053770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.McBeth J, Symmons DP, Silman AJ, et al. Musculoskeletal pain is associated with a long-term increased risk of cancer and cardiovascular-related mortality. Rheumatology (Oxford) 2009;48(1):74–77. doi: 10.1093/rheumatology/ken424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Morales-Espinoza EM, Kostov B, Salami DC, et al. Complexity, comorbidity, and health care costs associated with chronic widespread pain in primary care. Pain. 2016;157(4):818–826. doi: 10.1097/j.pain.0000000000000440. [DOI] [PubMed] [Google Scholar]
- 42.Kato K, Sullivan PF, Evengard B, Pedersen NL. Chronic widespread pain and its comorbidities: a population-based study. Arch Intern Med. 2006;166(15):1649–1654. doi: 10.1001/archinte.166.15.1649. [DOI] [PubMed] [Google Scholar]
- 43.Erber JT. Aging and Older Adulthood. 3rd ed. Chichester, West Sussex: Wiley-Blackwell; 2013. [Google Scholar]
- 44.MacFarlane GJ, Croft PR, Schollum J, Silman AJ. Widespread pain: is an improved classification possible? J Rheumatol. 1996;23(9):1628–1632. [PubMed] [Google Scholar]
- 45.Wolfe F, Smythe HA, Yunus MB, et al. The American College of Rheumatology 1990 criteria for the classification of fibromyalgia. Report of the Multicenter Criteria Committee. Arthritis Rheum. 1990;33(2):160–172. doi: 10.1002/art.1780330203. [DOI] [PubMed] [Google Scholar]
- 46.Ferreira-Valente MA, Pais-Ribeiro JL, Jensen MP. Validity of four pain intensity rating scales. Pain. 2011;152(10):2399–2404. doi: 10.1016/j.pain.2011.07.005. [DOI] [PubMed] [Google Scholar]
- 47.Persson LG, Lindström K, Lingfors H, Bengtsson C. A study of men aged 33-42 in Habo, Sweden with special reference to cardiovascular risk factors. Design, health profile and characteristics of participants and non-participants. Scand J Soc Med. 1994;22(4):264–272. doi: 10.1177/140349489402200405. [DOI] [PubMed] [Google Scholar]
- 48.Godin G, Shephard RJ. A simple method to assess exercise behavior in the community. Can J Appl Sport Sci. 1985;10(3):141–146. [PubMed] [Google Scholar]
- 49.Fleiss JL, Levin BA, Paik MC. Statistical Methods for Rates and Proportions. 3rd ed. Hoboken, NJ; Chichester: Wiley-Interscience; 2003. [Google Scholar]
- 50.Mourao AF, Blyth FM, Branco JC. Generalised musculoskeletal pain syndromes. Best Pract Res Clin Rheumatol. 2010;24(6):829–840. doi: 10.1016/j.berh.2010.10.005. [DOI] [PubMed] [Google Scholar]
- 51.Cimmino MA, Ferrone C, Cutolo M. Epidemiology of chronic musculoskeletal pain. Best Pract Res Clin Rheumatol. 2011;25(2):173–183. doi: 10.1016/j.berh.2010.01.012. [DOI] [PubMed] [Google Scholar]
- 52.de Heer EW, Gerrits MM, Beekman AT, et al. The association of depression and anxiety with pain: a study from NESDA. PLoS One. 2014;9(10):e106907. doi: 10.1371/journal.pone.0106907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hanssen DJ, Naarding P, Collard RM, Comijs HC, Oude Voshaar RC. Physical, lifestyle, psychological, and social determinants of pain intensity, pain disability, and the number of pain locations in depressed older adults. Pain. 2014;155(10):2088–2096. doi: 10.1016/j.pain.2014.07.019. [DOI] [PubMed] [Google Scholar]
- 54.Mitchell R, Draper B, Harvey L, Brodaty H, Close J. The association of physical illness and self-harm resulting in hospitalisation among older people in a population-based study. Aging Ment Health. 2015;15:1–10. doi: 10.1080/13607863.2015.1099610. [DOI] [PubMed] [Google Scholar]
- 55.Kittisupamongkol W. Comment on: musculoskeletal pain is associated with a long-term increased risk of cancer and cardiovascular-related mortality. Rheumatology (Oxford) 2009;48(5):595. doi: 10.1093/rheumatology/kep032. [DOI] [PubMed] [Google Scholar]
- 56.Parkerson HA, Zvolensky MJ, Asmundson GJ. Understanding the relationship between smoking and pain. Expert Rev Neurother. 2013;13(12):1407–1414. doi: 10.1586/14737175.2013.859524. [DOI] [PubMed] [Google Scholar]
- 57.Andersson H, Ejlertsson G, Leden I. Widespread musculoskeletal chronic pain associated with smoking. An epidemiological study in a general rural population. Scand J Rehabil Med. 1998;30(3):185–191. [PubMed] [Google Scholar]
- 58.O’Dwyer M, Peklar J, McCallion P, McCarron M, Henman MC. Factors associated with polypharmacy and excessive polypharmacy in older people with intellectual disability differ from the general population: a cross-sectional observational nationwide study. BMJ Open. 2016;6(4):e010505. doi: 10.1136/bmjopen-2015-010505. [DOI] [PMC free article] [PubMed] [Google Scholar]