Abstract
A latent viral reservoir that resides in resting CD4+ T cells represents a major barrier for eradication of HIV infection. We test here the impact of HIV protease inhibitor (PI) based combination anti-retroviral therapy (cART) over nonnucleoside reverse transcriptase inhibitor (NNRTI)-based cART on HIV-1 reactivation and integration in resting CD4+ T cells. This is a prospective cohort study of patients with chronic HIV-1 infection treated with conventional cART with an undetectable viremia. We performed a seven-year study of 47 patients with chronic HIV-infection treated with cART regimens and with undetectable plasma HIV-1 RNA levels for at least 1 year. Of these 47 patients treated with cART, 24 were treated with a PI-based regimen and 23 with a NNRTI-based regimen as their most recent treatment for more than one year. We evaluated the HIV-1 reservoir using reactivation assay and integrated HIV-1 DNA, respectively, in resting CD4+ T cells. Resting CD4+ T cells isolated from PI-treated patients compared to NNRTI-treated patients showed a limited HIV-1 reactivation upon T-cell stimulation (p = 0·024) and a lower level of HIV-1 integration (p = 0·024). Our study indicates that PI-based cART could be more efficient than NNRTI-based cART for limiting HIV-1 reactivation in aviremic chronically infected patients.
Among the main features of human immunodeficiency virus-1 (HIV-1) infection are immune suppression and viral persistence1. Combination anti-retroviral therapy (cART) drives the viral load down to undetectable levels2. However, using the ultrasensitive assays, low levels of active viral replication can be detected in the majority of subjects successfully treated with cART3. Indeed, the persistence of latent reservoirs of replication-competent proviruses remains a major obstacle in HIV-1 eradication4,5. Latent reservoirs are established early during acute viral infection and include among others, macrophages and latently infected resting CD4+ T cells, these later being the main viral reservoir6,7,8.
Most of the studies so far have addressed the effect of cART on the decrease of HIV-1 viremia under limit of detection using the classical assays. Usually, a better virologic performance of nonnucleoside reverse transcriptase inhibitor-(NNRTI)-based cART compared to protease inhibitor-(PI)-including regimens has been reported9. Although PI-based regimen have lower rates of HIV suppression compared with the NNRTI-based treatments, greater CD4 cell increases are seen in patients on PI arm and there is also less development of major drug resistance mutations in patients failing PI-based therapy10,11. Recently, cART intensification was assessed and did not reduce residual HIV-1 viremia in patients on cART, indicating that its potential to eradicate the virus appears limited12.
In contrast to the measurement of viremia in patients on cART, the impact of cART on the size of the cellular reservoirs of HIV-1 has been much less studied. Initiation of cART during primary HIV infection may limit the establishment of viral reservoirs, and very early cART limits the seeding of the HIV reservoir in long-lived central memory CD4+ T cells6,13,14. By contrast, the impact of cART on the HIV reservoir especially on viral reactivation from resting CD4+ T cells in aviremic chronically infected patients is so far unknown. We report here a study indicating a higher efficiency of PI-based cART over NNRTI-based cART for limiting HIV-1 reactivation in CD4+ T cells from aviremic chronically HIV-1 infected patients.
Results
Forty-seven patients with chronic HIV-1 infection treated with cART (treatment range: 2 years to 16 years) and with undetectable plasma HIV-1 RNA levels (<40 copies/ml) for at least 1 year were included in the study between 2008 and 2014. Of these 47 patients (mean age 47.4 years; range 27–93 years) treated with cART, 24 were treated with PI-based cART and 23 with NNRTI-based cART (as their most recent treatment) for more than one year (Table 1, Supplementary Tables S1 and S2). We did not observe significant differences for nadir median CD4 counts (298·106 versus 333·106 cells/l, P = 0.579), median CD4 counts at initiation of treatment (384·106 versus 349·106 cells/l, P = 0·903), median CD4 counts at the last point (607·106 versus 624·106 cells/l, P = 0·647), and median HIV RNA load at zenith (4·92 versus 4·83 log/ml, P = 0·892) between PI-treated and NNRTI-treated patients (Table 1). We did not find significant differences between PI-treated and NNRTI-treated patients for the number of failures of treatment (1 versus 1, P = 0·985) and the duration of therapy (8·50 versus 8·52 years, P = 0·936). The duration with undetectable plasma HIV-1 RNA levels was not significantly different in the PI-treated arm compared to the NNRTI-treated arm (4·83 versus 5·39 years, P = 0·581) (Table 1). At the last treatment, in 51·1% of individuals cART included a PI (fosamprenavir; saquinavir; atazanavir; lopinavir/ritonavir; darunavir) and in 48·9% a NNRTI (nevirapine; efavirenz; rilpivirine). Of note, among the 47 patients treated with cART, 17 were treated only with PI and 7 only with NNRTI (Supplementary Tables S1 and S2).
Table 1. Comparison of biological and virological characteristics of HIV+ patient groups under PI-cART and NNRTI-cART.
| Biological characteristics | PI | NNRTI | P value |
|---|---|---|---|
| (n = 24) | (n = 23) | ||
| Age (years), mean (±s.d.) | 48.6 ± 12.7 | 46.2 ± 10.7 | 0.495 |
| CD4+ T cell counts at nadir, absolute (cells per μl), median (IQR) | 298 (201–375) | 333 (246–419) | 0.579 |
| CD4+ T cell counts at initiation of treatment, absolute (cells per μl), median (IQR) | 384 (285–461) | 349 (265–574) | 0.903 |
| CD4+ T cell counts at the last point, absolute (cells per μl), median (IQR) | 607 (480–832) | 624 (492–801) | 0.647 |
| pVL at zenith (log copies per ml), median (IQR) | 4.92 (4.23–5.07) | 4.83 (4.47–5.16) | 0.892 |
| Previous treatment failure, median (IQR) | 1·00 (0–2.25) | 1·00 (0–4) | 0.985 |
| Time with therapy (years), mean ± s.d. | 8.50 ± 3.63 | 8.52 ± 3.49 | 0.936 |
| Time with undetectable pVL (years), mean ± s.d. | 4.83 ± 2.97 | 5.39 ± 2.73 | 0.581 |
| HIV-1 reactivation assay | |||
| Resting CD4+T cells | (n = 10) | (n = 11) | |
| HIV-1 RNA levels after reactivation assay (log copies per ml), median (IQR) | 3.01 (1.84–3.45) | 3.93 (3.31–4.87) | 0.024* |
| HIV-1 integration assay | |||
| Resting CD4+ T cells | (n = 14) | (n = 12) | |
| Integrated HIV-1 DNA (log copies per 106 cells), median (IQR) | 2.09 (1.53–2.52) | 2.98 (2.55–3.48) | 0.024* |
IQR, interquartile range; pVL, plasma viral load *P < 0·05.
In order to evaluate the qualitative and quantitative patient-specific variations in the ex vivo reactivation capacity of the HIV-infected cells depending of cART treatment, we analyzed the effect of a known HIV inducer (anti-CD2+anti-CD28 antibodies)15,16 on viral reactivation in ex vivo cultures of purified resting CD4+ T cells isolated from HIV+ patients treated with PI-based cART and with NNRTI-based cART. Since latently infected resting CD4+ T cells that harbour integrated replication-competent viral DNA represent the primary long-lived source of persistent HIV-1 in patients under cART5,6, we decided to analyze the impact of cART regimen on resting CD4+ T cells isolated from HIV+ patients. We observed, following reactivation of HIV-1 from latency in purified resting CD4+ T cells, that HIV-1 recovery was 0·92 log of HIV RNA copies/ml (3·01 versus 3·93 log copies/ml, P = 0·024) lower in HIV-1-infected patients treated with PI compared to those treated with NNRTI as their lastly administered treatment (Tables 1 and 2, and Fig. 1A). The impact of PI versus NNRTI treatment, taking into account the last treatment administered, on latently infected CD4+ T cells was further assessed by quantifying integrated HIV-1 DNA. The levels of integrated HIV-1 DNA was 0·89 log (2·09 versus 2·98 log copies/106 cells, P = 0·024) lower in purified resting CD4+ T cells isolated from HIV-1 patients treated with PI-based cART compared to HIV-1-infected patients treated with NNRTI-based cART (Tables 1 and 3, and Fig. 1B).
Table 2. Reactivation assay on HLA DR− CD25− CD69− CD4+ T cells (resting CD4+ T cells) isolated from HIV+ patients with undetectable viremia treated with PI (n = 10) and NNRTI (n = 11) as their lastly administered treatment.
| Patients | CD4+ at last point (cells/microl) | Last treatment | Duration of therapy (years) | Duration with undetectable plasma HIV-1 RNA levels (years) | HIV-1 RNA levels after reactivation assay (copies/ml) | HIV-1 RNA levels after reactivation assay (log copies/ml) |
|---|---|---|---|---|---|---|
| PI at the last treatment (n = 10) | ||||||
| WSH 229 | 617 | ABC 3TC SQV | 13 | 12 | 25862 | 4.41 |
| WSH 231 | 770 | ABC 3TC ATV | 10 | 7 | 40 | 1.60 |
| WSH 232 | 899 | TDF FTC fAPV | 7 | 1 | 3106 | 3.49 |
| WSH 233 | 465 | ATV EFV | 8 | 8 | 40 | 1.60 |
| WSH 235 | 797 | DDI AZT 3TC fAPV | 15 | 5 | 1108 | 3.04 |
| WSH 236 | 402 | TDF FTC ATV | 13 | 1 | 2862 | 3.46 |
| WSH 238 | 1240 | TDF FTC ATV | 5 | 5 | 40 | 1.60 |
| WSH 239 | 999 | TDF FTC ATV | 9 | 1 | 954 | 2.98 |
| WSH 240 | 663 | TDF FTC ATV | 9 | 4 | 2548 | 3.41 |
| WSH 241 | 524 | TDF FTC fAPV | 2 | 2 | 372 | 2.57 |
| Mean | 738 | 9.10 | 4.60 | 3693 | 2.82 | |
| SD | 246 | 3.73 | 3.44 | 7477 | 0.91 | |
| Median | 717 | 9.00 | 4.50 | 1031 | 3.01 | |
| 1st Quartile | 547 | 7.25 | 1.25 | 123 | 1.84 | |
| 3rd Quartile | 874 | 12.3 | 6.50 | 2784 | 3.45 | |
| NNRTI at the last treatment (n = 11) | ||||||
| WSH 227 | 733 | EFV FTC TDF | 9 | 8 | 146 | 2.16 |
| WSH 228 | 1324 | TDF FTC NVP | 10 | 7 | 229674 | 5.36 |
| WSH 230 | 444 | EFV FTC TDF | 8 | 1 | 8492 | 3.93 |
| WSH 242 | 428 | DDI TDF ABC 3TC EFV | 10 | 3 | 1668 | 3.22 |
| WSH 243 | 794 | EFV FTC TDF | 6 | 1 | 2514 | 3.40 |
| WSH 244 | 479 | EFV FTC TDF | 8 | 8 | 122268 | 5.09 |
| WSH 245 | 478 | TDF FTC NVP | 12 | 3 | 43614 | 4.64 |
| WSH 246 | 629 | EFV FTC TDF | 3 | 1 | 374578 | 5.57 |
| WSH 247 | 498 | ABC 3TC NVP | 16 | 7 | 31976 | 4.50 |
| WSH 248 | 1017 | ABC 3TC EFV | 10 | 7 | 4224 | 3.63 |
| WSH 249 | 519 | AZT 3TC NVP | 13 | 11 | 668 | 2.82 |
| Mean | 668 | 9.55 | 5.18 | 74529 | 4.03 | |
| SD | 270 | 3.31 | 3.33 | 116748 | 1.05 | |
| Median | 519 | 10.0 | 7.00 | 8492 | 3.93 | |
| 1st Quartile | 479 | 8.00 | 2.00 | 2091 | 3.31 | |
| 3rd Quartile | 764 | 11.0 | 7.50 | 82941 | 4.87 | |
| MW p-value | 0.461 | 0.768 | 0.684 | 0.024 | 0.024 | |
Figure 1. Resting CD4+ T cells isolated from PI-treated patients compared to NNRTI-treated patients showed a limited HIV-1 reactivation upon T-cell stimulation and a lower level of HIV-1 integration.
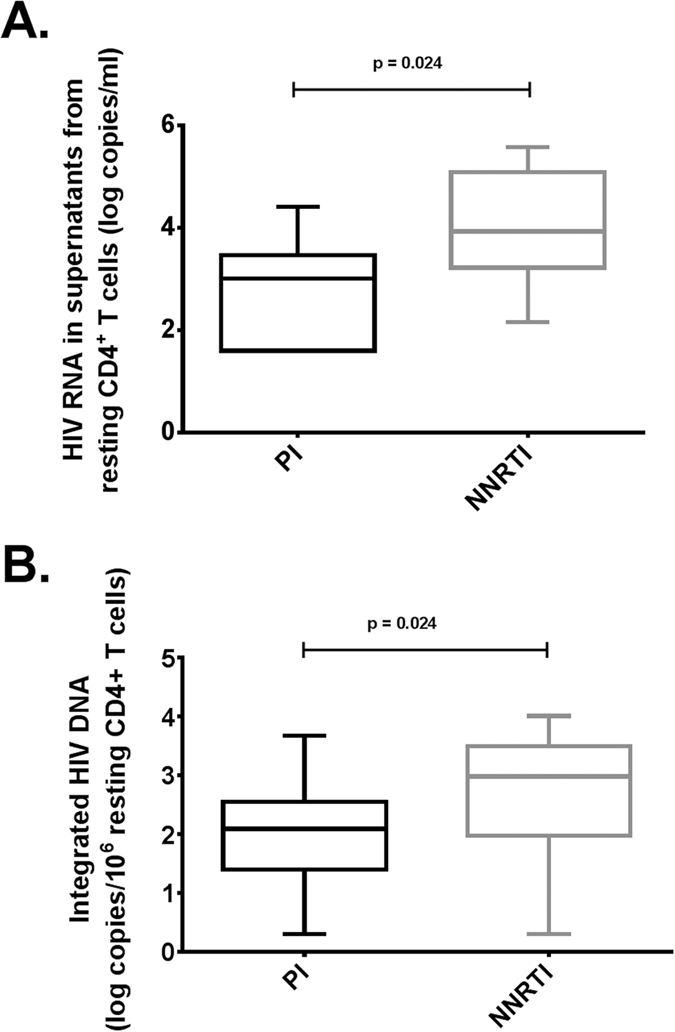
(A) Comparison of reactivation assay on resting CD4+ T cells isolated from HIV+ patients with undetectable viremia treated with PI (n = 10) and NNRTI (n = 11) as their lastly administered treatment. (B) Comparison of measurement of integrated HIV-1 DNA in resting CD4+ T cells isolated from HIV+ patients with undetectable viremia treated with PI (n = 14) and NNRTI (n = 12) as their lastly administered treatment.
Table 3. Measurement of integrated HIV-1 DNA in HLA DR− CD25− CD69− CD4+ T cells (resting CD4+ T cells) isolated from HIV+ patients with undetectable viremia treated with PI (n = 14) and NNRTI (n = 12) as their lastly administered treatment.
| Patients | CD4+ at the last point (cells/microl) | Last treatment | Duration of therapy (years) | Duration with undetectable plasma HIV-1 RNA levels (years) | Integrated HIV-1 DNA (copies/10^6 cells) | Integrated HIV-1 DNA (log copies/10^6 cells) |
|---|---|---|---|---|---|---|
| PI at the last treatment (n = 14) | ||||||
| B9 | 485 | TDF FTC ATV | 11 | 8 | 95 | 1.97 |
| B13 | 512 | ABC 3TC ATV | 14 | 8 | 85 | 1.92 |
| B15 | 995 | TDF FTC ATV | 12 | 8 | 4731 | 3.67 |
| B27 | 529 | ABC 3TC ATV | 3 | 2 | 4 | 0.6 |
| B33 | 404 | TDF FTC DRV | 8 | 2 | 175 | 2.24 |
| B34 | 127 | TDF FTC DRV | 2 | 1 | 17 | 1.23 |
| B38 | 423 | TDF FTC ATV | 8 | 5 | 2 | 0.3 |
| B39 | 997 | ABC 3TC DRV | 8 | 3 | 409 | 2.61 |
| B40 | 394 | fAPV AZT 3TC | 8 | 5 | 56 | 1.74 |
| B41 | 596 | TDF FTC DRV | 3 | 2 | 29 | 1.46 |
| B42 | 638 | DRV TDF ABC 3TC | 8 | 5 | 307 | 2.48 |
| B43 | 505 | ABC 3TC ATV | 10 | 8 | 163 | 2.21 |
| B45 | 809 | ABC TDF DRV | 12 | 8 | 344 | 2.53 |
| B46 | 906 | ABC 3TC ATV | 6 | 5 | 635 | 2.8 |
| Mean | 594 | 8.07 | 5.00 | 504 | 1.98 | |
| SD | 242 | 3.49 | 2.56 | 1186 | 0.85 | |
| Median | 521 | 8.00 | 5.00 | 129 | 2.09 | |
| 1st Quartile | 439 | 6.50 | 2.25 | 35.8 | 1.53 | |
| 3rd Quartile | 766 | 10.8 | 8.00 | 335 | 2.52 | |
| NNRTI at the last treatment (n = 12) | ||||||
| B2 | 548 | EFV FTC TDF | 5 | 4 | 6174 | 3.79 |
| B3 | 927 | ABC 3TC NVP | 7 | 5 | 2 | 0.3 |
| B6 | 479 | NVP TDF FTC | 8 | 7 | 682 | 2.83 |
| B8 | 975 | EFV FTC TDF | 3 | 2 | 985 | 2.99 |
| B10 | 580 | EFV TDF FTC | 6 | 5 | 2963 | 3.47 |
| B14 | 486 | NVP TDF FTC | 5 | 4 | 944 | 2.97 |
| B16 | 928 | TDF FTC NVP | 15 | 8 | 2879 | 3.46 |
| B18 | 624 | TDF FTC RPV | 5 | 4 | 10218 | 4.01 |
| B21 | 607 | TDF FTC RPV | 12 | 8 | 49 | 1.69 |
| B22 | 793 | TDF FTC NVP | 5 | 4 | 38 | 1.58 |
| B29 | 807 | TDF FTC RPV | 10 | 8 | 3185 | 3.5 |
| B37 | 790 | TDF FTC RPV | 10 | 8 | 771 | 2.88 |
| Mean | 712 | 7.58 | 5.58 | 2408 | 2.79 | |
| SD | 171 | 3.38 | 2.02 | 2931 | 1.04 | |
| Median | 707 | 6.50 | 5.00 | 965 | 2.98 | |
| 1st Quartile | 572 | 5.00 | 4.00 | 524 | 2.55 | |
| 3rd Quartile | 837 | 10.0 | 8.00 | 3019 | 3.48 | |
| MW p-value | 0.210 | 0.583 | 0.719 | 0.024 | 0.024 | |
Discussion
To diminish the size of the viral reservoir, several studies have shown that an early treatment during HIV-1 primo-infection could be beneficial13,14, and recently on-demand preexposure prohylaxis has been shown to be highly efficient to prevent HIV transmission17. We report here a study indicating that PI-based cART could be more efficient than NNRTI-based cART for limiting HIV-1 reactivation in aviremic chronically infected patients.
Although the respective use of NNRTI-based cART and PI-based cART have been extensively studied in regard to residual viremia, only little is known on their impact on cellular reservoirs of HIV-1. By two approaches, reactivation assay and the Alu-PCR assay that allow evaluating in a qualitative and quantitative manner, respectively, the amount of integrated provirus, we observed that PI-based cART was superior to NNRTI-based cART for limiting the size of the HIV-1 reservoir. Recent studies indicate that raltegravir intensification of PI-based cART results in a specific and transient increase in episomal HIV DNA that might represent lower efficiency of HIV integration under PI18,19. Recent publications also indicate that the size of the HIV-1 reservoir measured by total cell-associated HIV-1 DNA correlates positively with the frequency of positive recovery measurements in response to various latency-reversing agents20. Additionally, the low frequency of infected cells in the HIV-1 controllers is associated with less efficient viral reactivation21. Our data are in line with these findings indicating that limited reactivation could be a consequence of lower integration events.
Several molecular mechanisms could explain the lower viral reactivation under PIs. First, low frequency of viral integration in transcriptionally active sites could explain the limited reactivation observed under PIs. Second, since integrase is produced by protease-mediated cleavage of the Pr160Gag-Pol precursor protein, PIs might inhibit HIV-1 integration by preventing the formation of a functional integrase19. Impaired integrase activity and/or impairment of the integrase-LEDGF/p75 interaction could result in a decreased number of integrated provirus in transcriptionally active sites, but also could favor the viral integration in less transcriptionally active sites which will result in lower viral reactivation22. Third, HIV PIs have been reported to block Akt activation23, and we recently observed that PIs, by blocking Akt activation especially triggered by Nef, limit HIV-1 recovery from latently-infected T cells24. Additionally HIV integration and the establishment of latency in CCL19-treated resting CD4+ T cells has been shown to require activation of NF-kB and increased HIV integrase stability25. Interestingly, PIs block NF-kB activation induced by Toll-like receptor 2 (TLR2) and TLR426. We also observed that PIs, but not NNRTIs, block Akt activation in PBLs treated with recombinant HIV-1 Nef in vitro and that the blockade of Nef-mediated Akt activation by Akt inhibitors decreased dramatically NF-kB activation24. Therefore, we cannot exclude that PIs by blocking HIV-mediated Akt activation in T cells decrease NF-kB activation and ultimately limit HIV integration in T cells.
Since Akt activation is critical for cell survival, the blockade of Akt activation by PIs in latently infected resting CD4+ T cells could help to clear these cells through accelerated cell death. Additionally, PIs have been shown to trigger the death of CD4+ T cells through the generation of Casp8p41, a fragment obtained by cleavage of procaspase 8 by PIs, that lacks caspase activity but nonetheless contributes to T cell apoptosis27. Thus PIs could participate to decrease the size of the viral reservoir in infected patients through the clearance of infected CD4+ T cells.
Low HIV-1 reservoirs found in HIV-1 controllers contributes to temporary control of infection without antiretroviral therapy21. Along this line, control of HIV-1 viremia or delayed viral rebound after discontinuation of antiretroviral treatment has been consistently associated with low levels of cell associated HIV-DNA at the time of treatment interruption28,29. Therefore, lower levels of integrated HIV-1 in purified resting CD4+ T cells isolated from HIV-1 patients treated with PI-based cART compared to HIV-1-infected patients treated with NNRTI-based cART could be critical to achieve control of infection.
In conclusion, in aviremic chronically HIV-1-infected patients our study indicates that PI-based cART limits HIV-1 recovery from latently infected resting CD4+ T cells and curtails the amount of integrated HIV-1 DNA. Altogether, our results support the need to further assess the therapeutic use of HIV-1 protease inhibitors in view of their impact on persistence of HIV-1 reservoirs and might provide and/or accompany new therapeutic strategies for a remission.
Methods
Ethics approval and consent to participate
All the patients who were enrolled at the St-Pierre University Hospital (Brussels, Belgium) and the Besançon University Hospital (France) gave their written informed consent to participate in the study according to the Helsinki declaration. The study was approved by the local ethics committees of the Saint-Pierre University Hospital (Brussels, Belgium) and the Besancon University Hospital (Besançon, France) (CPP EST-2). To perform our study, we used blood samples that were part of a routine care. It was an observational study and not a clinical trial. According to the French Regulatory Authority for clinical studies, prospective and retrospective studies with observation analysis only are not evaluated by Human Protection Committees. The Human Protection Committee East Area II from France was consulted and issued a formal waiver of approval. This study did not rely solely on medical records. The authors did not have any contact with the study subjects and performed tests on patient blood samples that were part of a routine care. The blood samples were anonymized before being used by the authors.
Patients
We enrolled 47 chronically HIV-1 infected individuals at the St-Pierre Hospital (Brussels, Belgium) and the Besançon University Hospital (Besançon, France). The patients were treated with cART (treatment range: 2 years to 16 years) and had undetectable plasma HIV-1 RNA levels (<40 copies/ml) for at least 1 year. Characteristics of these patients were well documented and presented in Supplementary Tables S1 and S2. Of these 47 patients treated with cART, 24 were treated with PI and 23 with NNRTI as their most recent treatment for more than one year.
Isolation of resting CD4+ T lymphocytes
Purified CD25−, CD69−, HLA-DR− CD4+ T cells (resting CD4+ T cells) were obtained from the peripheral blood of HIV+ patients using a negative selection assay using magnetic beads (LD columns, Miltenyi Biotec, Bergisch Gladbach, Germany) as described previously30.
Viral reactivation assay
One day after isolation, 5 × 105 purified CD25−, CD69−, HLA-DR− CD4+ T cells (resting CD4+ T cells) were mock-treated or treated with anti-CD2+ anti-CD28 antibodies as a positive control for global T-cell activation15,16. For the measurement of reactivation of HIV-1 expression in resting CD4+ T cells isolated from the peripheral blood of HIV-infected patients, six days after treatment, culture supernatants were tested for quantitative HIV-1 RNA levels using the COBAS AmpliPrep/COBAS Amplicor HIV-1 Monitor Test, according to the manufacturer’s instructions.
Quantification of integrated HIV-1 DNA
Integrated HIV-1 DNA was measured by using a previously described Alu-gag PCR with some modifications31. This assay detects only integrated provirus because it relies on initial amplification in which one primer hybridizes with conserved sequences in Alu elements that are present in the human genome. The level of HIV-1 integration was measured by comparing the detection signal to an integration standard curve that was prepared from U1 cells. ACH2 cells were used for calibration. The sequences of first step PCR amplification primers were as follows: genomic Alu forward, 5′-GCCTCCCAAAGTGCTGGGATTACAG-3′; and HIV-1 gag reverse, 5′-GCTCTCGCACCCATCTCTCTCC-3′. The reaction conditions were 1x Tfi PCR reaction buffer, 1.5 mM MgCl2, 1 mM dNTPs, 100 nM Alu forward primer, 600 nM gag reverse primer and 5 U of Tfi DNA polymerase (Invitrogen). The thermal cycler (Eppendorf AG, Hamburg, Germany) was programmed to perform a 2 min hot start at 94 °C, followed by 20 cycles of following: denaturation at 94 °C for 30 sec, annealing at 50 °C for 1 min, and extension at 72 °C for 1 min 40 sec. The second round of real time quantitative PCR was performed using 21 μl of Nested Alu-gag PCR along with standard curve amplification products. The sequences of the primers were as follows: LTR forward, 5′-GCCTCAATAAAGCTTGCCTTGA-3′; and LTR reverse, 5′-TCCACACTGACTAAAAGGGTCTGA-3′. The LTR Taqman probe, labeled at its 5′ terminus with the reporter fluorophore 6-carboxyfluorescein (FAM) and at its 3′ terminus with the quencher 4-(4-dimethylamino-phenylazo)-benzene (DABCYL), had the following sequence: 5′-FAM-GCGAGTGCCCGTCTGTTGTGTGACTCTGGTAACTAGCTCGC-DABCYL-3′. The reaction was carried out in a volume of 50 μl containing 1 X Taqman Master mix (Applied Biosystems, Foster City, CA), 250 nM concentration of LTR forward and reverse primers, and 200 nM Taqman probe. The reactions were performed on Stratagene Mx2005 P. The thermal program was 2 min hot start at 95 °C, followed by 40 cycles of denaturation at 95 °C for 15 sec and annealing and extension at 60 °C for 1 min.
Data analysis
The program used for plotting and statistical analysis was Prism version 6.0 (GraphPad Software). Results from ex vivo reactivation studies and HIV integration using patient cell cultures of resting CD4+ T cells are shown as medians. Data sets were analyzed using an unpaired nonparametric t-test (Mann-Whitney test). Differences were considered significant at a value of p < 0.05.
Additional Information
How to cite this article: Kumar, A. et al. Limited HIV-1 Reactivation in Resting CD4+ T cells from Aviremic Patients under Protease Inhibitors. Sci. Rep. 6, 38313; doi: 10.1038/srep38313 (2016).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Material
Acknowledgments
We thank the HIV-1-infected patients for their willingness to participate in this study and Elodie Goudeseune, Joelle Cailleau and Annick Caestecker who cared for the patients. We thank Christelle Cardona, Kashif Khan, Audrey Varin, Laurie Coquard, Caroline Vanhulle, Nadège Delacourt, and Laurence Colin for excellent technical assistance. This work was supported by grants (to GH) from the University of Franche-Comté, the Région Franche-Comté (RECH-FON12–000013), the France Recherche Nord & Sud Sida-HIV Hépatites (ANRS, n°13543 and 13544) and HIVERA 2013 (EURECA project), (to CVL) from the Université Libre de Bruxelles (ULB), the Belgian Fund for Scientific Research (FRS-FNRS, Belgium), the NEAT program (European AIDS treatment network), the “Fondation Roi Baudouin” (Belgium), the Wallon region (CIBLES Excellence programm) and the ANRS (France). W.A. is a recipient of a doctoral scholarship from the Higher Education Commission, Pakistan. A.K. is a recipient of a postdoctoral grant of Région Franche-Comté (N° 2012C-06102). S.B. is fellowship from the Belgian ‘Fonds pour la Recherche dans l′Industrie et l′Agriculture’ (FRIA). J.S.G. was a fellow of the Excellence program ‘Cibles’. C.V.L. is Research Director of the FRS-FNRS. The University of Franche-Comté, the Région Franche-Comté, ANRS, HIVERA 2013, the Université Libre de Bruxelles, FRS-FNRS, NEAT, the “Fondation Roi Baudouin”, and the Wallon region; the funders had no role in the data collection, analysis, patient recruitment, or decision to publish.
Footnotes
Author Contributions G.H. and C.V.L. conceived and designed the project. A.K., W.A., S.B., S.P. and J.S.G. performed experiments. G.H., K.K., N.C. and S.D.W. participated to the enrolment of HIV-infected patients. Quantification of the integrated viral reservoir: W.A. G.H. and C.V.L. wrote the manuscript.
References
- Chun T. W., Moir S. & Fauci A. S. HIV reservoirs as obstacles and opportunities for an HIV cure. Nat. Immunol. 16, 584–589 (2015). [DOI] [PubMed] [Google Scholar]
- Perelson A. S. et al. Decay characteristics of HIV-1-infected compartments during combination therapy. Nature 387, 188–191 (1997). [DOI] [PubMed] [Google Scholar]
- Palmer S. et al. Low-level viremia persists for at least 7 years in patients on suppressive antiretroviral therapy. Proc. Natl. Acad. Sci. USA. 105, 3879–3884 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho Y. C. et al. Replication-competent noninduced proviruses in the latent reservoir increase barrier to HIV-1 cure. Cell 155, 540–551 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Lint C., Bouchat S. & Marcello A. HIV-1 transcription and latency: an update. Retrovirology 10, 67 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siliciano R. F. Opening fronts in HIV vaccine development: targeting reservoirs to clear and cure. Nat. Med. 20, 480–481 (2014). [DOI] [PubMed] [Google Scholar]
- Le Douce V. et al. Molecular mechanisms of HIV-1 persistence in the monocyte-macrophage lineage. Retrovirology 7, 32 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar A., Darcis G., Van Lint C. & Herbein G. Epigenetic control of HIV-1 post integration latency: implications for therapy. Clin. Epigenetics 7, 103 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- INITIO Trial International Co-ordinating Committee et al. Virological and immunological outcomes at 3 years after starting antiretroviral therapy with regimens containing non-nucleoside reverse transcriptase inhibitor, protease inhibitor, or both in INITIO: open-label randomised trial. Lancet 368, 287–298 (2006). [DOI] [PubMed] [Google Scholar]
- Cummins N. W., Sainski A. M., Natesampillai S., Bren G. D. & Badley A. D. Choice of antiretroviral therapy differentially impacts survival of HIV-infected CD4 T cells. Mol. Cell. Ther. 2, 1 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chéret A. et al. Combined ART started during acute HIV infection protects central memory CD4+ T cells and can induce remission. J. Antimicrob. Chemother. 70, 2108–2120 (2015). [DOI] [PubMed] [Google Scholar]
- Dinoso J. B. et al. Treatment intensification does not reduce residual HIV-1 viremia in patients on highly active antiretroviral therapy. Proc. Natl. Acad. Sci. USA. 106, 9403–9408 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sáez-Cirión A. et al. Post-treatment HIV-1 controllers with a long-term virological remission after the interruption of early initiated antiretroviral therapy ANRS VISCONTI study. Plos Pathog. 9, e1003211 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buzón M. J. et al. HIV-1 replication and immune dynamics are affected by raltegravir intensification of HAART-suppressed subjects. Nat. Med. 16, 460–465 (2010). [DOI] [PubMed] [Google Scholar]
- Costello R. et al. Activation of primary human T-lymphocytes through CD2 plus CD28 adhesion molecules induces long-term nuclear expression of NF-kappa B. Cell Growth Differ. 4, 329–339 (1993). [PubMed] [Google Scholar]
- Pierrès A. et al. Triggering CD28 molecules synergize with CD 2 (T 11.1 and T 11.2)-mediated T cell activation. Eur. J. Immunol. 18, 685–690 (1988). [DOI] [PubMed] [Google Scholar]
- Molina J. M. et al. On-demand preexposure prophylaxis in men at high risk for HIV-1 infection. N. Engl. J. Med. 373, 2237–2246 (2015). [DOI] [PubMed] [Google Scholar]
- Hatano H. et al. Increase in 2-long terminal repeat circles and decrease in D-dimer after raltegravir intensification in patients with treated HIV infection: a randomized, placebo-controlled trial. J. Infect. Dis. 208, 1436–1442 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rabi S. A. et al. Multi-step inhibition explains HIV-1 protease inhibitor pharmacodynamics and resistance. J. Clin. Invest. 123, 3848–3860 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Darcis G. et al. Reactivation capacity by latency-reversing agents correlates with the size of the HIV-1 reservoir. AIDS PMID: 27755105 (2016). [DOI] [PubMed] [Google Scholar]
- Noel N. et al. Long-term spontaneous control of HIV-1 relates to low frequency of infected cells and inefficient viral reactivation. J. Virol. 90, 6148–6158 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vranckx L. S. et al. LEDGIN-mediated inhibition of integrase-LEDGF/p75 interaction reduces reactivation of residual latent HIV. EBioMedicine 8, 248–264 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kourjian G. et al. HIV protease inhibitor-induced cathepsin modulation alters antigen processing and cross-presentation. J. Immunol. 196, 3595–3607 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar A. et al. Fine tuning of AKT-pathway by Nef and its blockade by protease inhibitors results in limited recovery in latently HIV infected T-cell line. Sci Rep. 6, 24090 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saleh S. et al. HIV integration and the establishment of latency in CCL19-treated resting CD4+ T cells require activation of NF-kB. Retrovirology 13, 49 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Equils O. et al. Human immunodeficiency virus type 1 protease inhibitors block Toll-like receptor 2 (TLR2)- and TLR4-induced NF-kappaB activation. Antimicrob. Agents Chemother. 48, 3905–3911 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sainski A. M. et al. Casp8p41 generated by HIV protease kill CD4 T cells through direct Bak activation. J Cell Biol 206, 867–876 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Assoumou L. et al. A low HIV-DNA level in peripheral blood mononuclear cells at antiretroviral treatment interruption predicts a higher probability of maintaining viral control. AIDS 29, 2003–2007 (2015). [DOI] [PubMed] [Google Scholar]
- Goujard C. et al. HIV-1 control after transient antiretroviral treatment initiated in primary infection: role of patient characteristics and effect of therapy. Antivir. Ther. 17, 1001–1009 (2012). [DOI] [PubMed] [Google Scholar]
- Bouchat S. et al. Histone methyltransferase inhibitors induce HIV-1 recovery in resting CD4(+) T cells from HIV-1-infected HAART-treated patients. AIDS 26, 1473–1482 (2012). [DOI] [PubMed] [Google Scholar]
- Liszewski M. K., Yu J. J. & O’Doherty U. Detecting HIV-1 integration by repetitive-sampling Alu-gag PCR. Methods 47, 254–260 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


