Abstract
Background
Hemoglobin is a rich source of biological peptides. As a byproduct and even wastewater of poultry-slaughtering facilities, chicken blood is one of the most abundant source of hemoglobin.
Results
In this study, the chicken hemoglobin antimicrobial peptides (CHAP) were isolated and the antimicrobial and bactericidal activities were tested by the agarose diffusion assay, minimum inhibitory concentration (MIC) analysis, minimal bactericidal concentration (MBC) analysis, and time-dependent inhibitory and bactericidal assays. The results demonstrated that CHAP had potent and rapid antimicrobial activity against 19 bacterial strains, including 9 multidrug-resistant bacterial strains. Bacterial biofilm and NaCl permeability assays, transmission electron microscopy (TEM) and scanning electron microscopy (SEM) were further performed to detect the mechanism of its antimicrobial effect. Additionally, CHAP showed low hemolytic activity, embryo toxicity, and high stability in different temperatures and animal plasma.
Conclusion
CHAP may have great potential for expanding production and development value in animal medication, the breeding industry and environment protection.
Electronic supplementary material
The online version of this article (doi:10.1186/s12866-016-0904-3) contains supplementary material, which is available to authorized users.
Keywords: Antimicrobial peptides, Hemoglobin, Hydrolysis, Antimicrobial activity, Bactericidal activity
Background
Due to the widespread use and even abuse of conventional antibiotics, antibiotic resistance is rampant all over the world, which limits the lifespan of commercial antibiotics and results in the urgent demand of new platforms for efficient antibiotic discovery [1, 2].
As an essential part of innate immunity, antimicrobial peptides (AMPs) have been receiving increasing attention because of their unique antimicrobial mechanism against both Gram-positive and Gram-negative bacteria, and even including some multidrug-resistant strains over recent decades [3, 4]. Antimicrobial peptides are ubiquitous in all living organisms. More than 5000 AMPs (http://www.camp.bicnirrh.res.in/index.php) have been identified and 2593 peptides have been derived naturally (http://aps.unmc.edu/AP/main.php) since the discovery of the lysozyme by Alexander Fleming in 1922 [5]. Although substantial AMPs have been discovered over the past decades, only a small part of them have been used because of high costs and potential cytotoxicity [1, 6]. Hence, finding efficient, nontoxic and low-cost AMPs is urgent in promoting AMPs’ practical applications.
The whole blood is a mixture of cells (erythrocytes, leucocytes and platelets) and plasma (colloids and crystalloids), which delivers nourishment and oxygen to and removes waste products from all parts of the body [7]. Components in blood, such as platelet concentrates [8], defensins [3], leukocyte extracts [9], also play important roles in antimicrobial host defense. Hemoglobin is the main component of the erythrocyte [10]. Aside from the basic function of transporting oxygen, hemoglobin has been found as a source of various biological peptides [11–13]. Many AMPs called hemocidins have been isolated from hemoglobin cleavage in vivo [14] or from hemoglobin hydrolysis by chemical reagents, physical methods, or enzymes in vitro [15, 16]. To date, the hemocidins derived from human beings [17], bovines [12], rabbits [18], swine [15], crocodiles [19], fish [20], and shellfish [21] have been reported and most of them are made up of 2 to 60 residues, characterized by a common random coil structural and broad-spectrum antimicrobial activity [22–24]. As a byproduct and even one of the major dissolved pollutants in slaughter house wastewater [25], appropriate treatment of chicken blood is of great benefit to both environmental protection and economic development. However, hemocidins from poultry have not been documented yet. In this study, the hemocidins from chickens were isolated and their antimicrobial and bactericidal activities were further detected.
Methods
Materials and chemicals
All common chemical reagents and biological products were of analytical grade from commercial sources. Papain (2000 IU g−1) was purchased from Sigma Chemical Co. (St. Louis, Mo, America).
Preparation of CHAP
The chicken hemoglobin antimicrobial peptides (CHAP) were prepared as modified method described before [26]. In brief, fresh chicken blood (Beijing Huadu Broiler Corporations, Beijing, China) was collected with heparin and then centrifuged with 2,000 × g at 4 °C for 10 min. The upper liquid and white cells were removed and washed with sterilized saline. The procedures described above were repeated 3 times. The cells were frozen, thawed, stirred and homogenized in deionized water (pH 7.0) with papain (1:1,000 w/v) proteolysis at 70 °C for 8 h. The digested suspensions were added with ice-cold aqueous 5% acetic acid solution (1:1 v/v) and extracted overnight at 4 °C. After being centrifuged at 8,000 × g for 30 min at 4 °C, the suspensions were collected as crude extracts, and the protein concentration was detected by NanoDrop 2000 UV–vis Spectrophotometer (Thermo Fisher Scientific, Massachusetts, America). The pH of the extracts was adjusted to 6.0 with sodium hydroxide. The crude extracts were loaded onto 10 × 300 mm Sephadex G-100 column and eluted by 0.2 mol L−1 sodium acetate buffer (pH 6.0) with the speed of 12.0 mL cm−2 h -1 . Each elution was analyzed by agarose diffusion assay [27] with Escherichia coli ATCC 25922 as the indicator organism. The fractions with potent antibacterial activity were collected and detected with Tricine SDS-PAGE [28] and then subjected to mass spectrometry (Beijing Protein Innovation Co., Ltd., Beijing, China).
Bacterial strains and growth conditions
Staphylococcus aureus ATCC25923, Staphylococcus aureus ATCC 29213, Staphylococcus albus ATCC01331, Escherichia coli ATCC 25922, Escherichia coli O78, Escherichia coli C83922, Escherichia coli C83901, Pseudomonas aeruginosa ATCC27853, Pasteurellae gallinarum C48-3 were purchased from the China Veterinary Culture Collection Center (CVCC). Aeromonas hydrophila, Bacillus cereus and Escherichia coli were clinically isolated from crucian carps, pigeon and equines respectively by Laboratory of Veterinary Pathology and Public Health of the College of Veterinary Medicine, China Agricultural University. Staphylococcus aureus MR-L22, MR-QD-CD10, Enterococcus faecalis 53A, 52A, 37 N and Pseudomonas aeruginosa M140 and Escherichia coli T50 were all multi-resistance strains of clinics, and obtained from Beijing Key Laboratory of Detection Technology for Animal Food safety of the College of Veterinary Medicine. All the above Gram-negative strains were grown in Luria-Bertani (LB) agar and the Gram-positive bacteria were grown in brain heart infusion (BHI) agar.
Determination of antimicrobial and bactericidal activities
Agarose diffusion assay
The primary antibacterial activities of CHAP elution (100 μg mL−1) were detected by modified agarose diffusion assay as described before [27]. Briefly, the single colony of each bacterial strain was grown in trypticase soy broth (TSB, 30 g L−1) overnight at 37 °C under aerobic conditions. 2 × 108 CFU mL−1 bacteria culture of each strain was added to warm (50–55 °C) sterile agarose [1% agarose (low EEO, Sigma, St. Louis, MO), 0.03% nutrient broth, and 10 mM PBS buffer, pH 7.4] (1:100 v/v). 10 μL samples were added to 3 mm wells punched by agar punch (BioRad Laboratories, Hercules, Canda). 0.2 mol L−1 sodium acetate (solvent) and 20000 IU penicillin-streptomycin solution of the same volume were added as negative and positive control, respectively. After being incubated overnight at 37 °C, the diameter of the each clean zone of growth inhibition was measured as the antibacterial activity of CHAP against different strains.
Minimum inhibitory concentration (MIC) analysis
A micro dilution assay was employed to determine MIC according to the broth micro dilution guideline of Clinical and Laboratory Standards Institute (CLSI) [29]. Briefly, 50 μL of twofold serial dilutions of CHAP (25 to 0.20 μg mL−1) was placed into wells of sterile 96-well cell culture plates. The 50 μL of bacterial suspensions (1 × 105 CFU mL−1) were added to the peptides. The wells were added with 50 μL of Mueller- Hinto (MH) broth and 50 μL of bacterial culture was treated as positive and negative control, respectively. After 24 h incubation, the MICs were determined at 492 nm by spectrophotometer (Thermo Multiskan MK3, Thermo Fisher Scientific, Massachusetts, America).
Time-dependent inhibitory assay
A 500 μL aliquot of CHAP with 2 × MIC of the bacterium was added respectively to 500 μL bacterial suspensions (1 × 105 CFU mL−1) in the sterilized 1.5 mL tubes as the treated groups. Bacteria treated with 500 μL solvent (0.2 mol L−1 sodium acetate) were set as the control groups. After being incubated for 30 min, 100 μL aliquot of the suspensions were pipetted into to a sterilized 1.5 mL tube. After centrifugation at 1,000 × g for 5 min, the supernatant was removed, and the pellet was resuspended in 100 μL MHB medium. Tenfold serially diluted suspension was placed on agar plates and incubated at 37 °C until viable colonies could be seen and the numbers of colony-forming units (CFU) were counted. The inhibitory rate of each bacterium was calculated according to the following formula: the inhibitory rate = [(colonies of the treated group - colonies of the treated group)/colonies of the control group] × 100%.
In order to further detect the process and speed of the antimicrobial activity of CHAP, the time growth curves and inhibitory rates of Escherichia coli ATCC 25922, Staphylococcus aureus ATCC29213, Staphylococcus aureus MR-L22, Enterococcus faecalis 52A, Pseudomonas aeruginoda M140 and Escherichia coli T50 were achieved after the treated suspensions were incubated for 0, 5, 10, 30, 90 min respectively.
Minimal bactericidal concentration (MBC) analysis
The MBC values were determined in 96-well plates, which was similar to the method of MIC. MBC values were further confirmed by plating 100 μL samples of each well with no visible turbidity onto the MHB medium. The least concentration showing no visible growth on the plates was considered as the MBC value.
Time-dependent bactericidal assay
The time depending bactericidal curves of Escherichia coli ATCC 25922 were determined as the time-dependent inhibitory assay mentioned above by adding CHAP with concentration of its MBC value to the bacterial cultures grown to early and late exponential phase as the reference [2].
In order to detect the bacteriolysis against bacteria in stationary phase, 10 ml of bacterial culture (2 × 10 9 CFU mL−1) was treated with 10 × MIC of CHAP. The culture treated with solvent (0.2 mol L−1 sodium acetate) was set as the control group. After 24 h incubation, 2 ml of each culture was added to a glass tube and was photographed [2].
Bacterial biofilms assay
Crystal violet staining method was applied to detect the effect of CHAP on the biofilm formation [30]. Briefly, Staphylococcus aureus ATCC29213 were cultured in TSB overnight. 100 μL bacterial suspensions (1 × 106 CFU mL−1) with 2 × MIC, 1 × MIC, 1/2 × MIC, 1/4 × MIC, 1/8 × MIC, 1/16 × MIC of CHAP were added to 96-well plates and the bacterial suspensions with no CHAP and the sterilized TSB were treated as control groups. After static culture at 37 °C for 24 h or shake culture (50 rmp) at 37 °C for 72 h, the contents were aspirated and the wells were washed by 200 μL PBS for three times, methanol fixed for 1 h and stained with 200 μL crystal violet (5 g L−1) for 30 min. The wells were washed by running water and air dried. The plates were determined at 600 nm by spectrophotometer.
NaCl permeability assay
The effect of CHAP on the NaCl permeability of bacteria was detected as modified protocol as follows [31]. 100 μL of bacterial suspensions (1 × 106 CFU mL−1) with 1/2 × MIC of CHAP were added to 96-well plates and the bacterial suspensions with no CHAP as control. 100 μL of NaCl solutions with different concentrations (80, 100, 120, 140, 160, 180, 200 g L−1) were added into each wells and incubated at 37 °C for 12 h. The bacterial concentration of each well was determined by measuring the optical density at 600 nm (OD600).
Electron microscopy observations
Both transmission electron microscopy (TEM) and scanning electron microscopy (SEM) were conducted as previously described [32–34]. Briefly, Escherichia coli ATCC 25922 and Staphylococcus aureus ATCC29213 were cultured overnight, 107 CFU ml−1 bacteria were incubated with 1 × MIC of CHAP or diluents of the same volume at 37 °C for 30 min. All the samples were fixed and proceeded for the TEM and SEM respectively.
Hemolytic assay and embryotoxicity assay
The hemolytic activity was evaluated as previously described [35]. 4% (vol/vol) fresh chicken erythrocyte suspensions were added to a 96-well plate and incubated with CHAP at 360, 180, 90, 45, 22.5, 11.25 μg mL−1 individually at 37 °C for 1 h. Wells treated with PBS and 0.1% Triton X-100 of the same volume were taken as 0 and 100% hemolysis. The wells were determined by measuring the optical density at 492 nm (OD492).
The embryotoxicity of CHAP was detected as the following measures. The 10-days-old-chicken embryos were randomly divided into 5 groups, each of 10 eggs, 0.2 mL of CHAP of 1 × MIC, 2 × MIC, 4 × MIC, 6 × MIC dose against Escherichia coli ATCC25922 were injected into the chorio-allantoic cavity, and embryos treated with the same volume of solvent (0.2 mol L−1 sodium acetate) were used as controls. The eggs were put in a hatching machine and hatchability and weight of the eggs were observed regularly until hatching.
Stability in different temperatures and in 50% plasma
CHAP (100 μg mL−1) was treated with different temperatures varying from 30 °C, 40 °C, 50 °C, 60 °C, 70 °C, 80 °C, 90 °C, 100 °C, 121 °C for 30 min. The antimicrobial activities of these treated aliquots were determined with agarose diffusion assay and were compared with CHAP stored in 4 °C.
The stability of CHAP in 50% plasma was evaluated as previously described [29] with some modifications. Briefly, the plasma of chicken and rabbit was determined with no antimicrobial activity before the test. Then 640 μg ml−1 CHAP was diluted 1:1 in fresh chicken and rabbit plasma and pre-incubated at 37 °C for 0, 3, and 6 h respectively. After incubation, the antimicrobial activity of each sample was determined by agarose diffusion assay. The effect of CHAP diluted by its solvent was regarded as the 100%, and the effect of the treated samples was demonstrated as percentages.
Statistical Analysis
Experiments were conducted with biological replicates and experimental data were expressed as mean ± standard deviation of at least three determinations and analyzed by one-way ANOVA using SPSS 20.0 (SPSS Inc., Cary, NC, USA). Differences were considered to be statistically significant at P < 0.05 or P < 0.01.
Results
Preparation of CHAP
The crude extracts of CHAP were light yellow and the protein concentration was adjusted to 5 mg ml−1 before loading on the Sephadex G-100 column. There were two main peaks after the elution of Sephadex G-100 gelatin (Fig. 1a) and the tubes from 9 to 16 in the left half of the second peak showed potent antimicrobial activity (Fig. 1b). Detected by Tricine SDS –PAGE, the collected CHAP showed band around 3.3 KDa (Fig. 1c). This band was further analyzed and peptides of gallus hemoglobin subunit alpha were confirmed by mass spectrum (see Additional file 1).
Fig. 1.

Preparation of CHAP. a Sephadex G-100 gelatin separation of the extract from chicken blood. b Antibacterial activity detection of elution of Sephadex G-100 gelatin by agarose diffusion assay against Escherichia coli ATCC25922. c Tricine–SDS-PAGE of the interested elution and the band was around 3.3 KDa (arrow)
Determination of antimicrobial and bactericidal activity
Antimicrobial effect of CHAP
The results of antibacterial activity of CHAP detected via agarose diffusion assay, MIC assay and inhibitory rate assay are shown in the columns 2–4 in Table 1.These results demonstrated that CHAP performed potent antimicrobial activities against both Gram-negative bacteria and Gram-positive bacteria, including 9 multidrug-resistant strains.
Table 1.
Antibacterial activity and bactericidal activity of Chicken hemoglobin fragment peptides
| Strains | D (mm) | MIC (μg mL−1) | IR (%) | MBC (μg mL−1) |
|---|---|---|---|---|
| Gram-negative bacteria | ||||
| Escherichia coli ATCC 25922 | 24.5 | 6.25 | 83.00 | 80 |
| Escherichia coli C83901 | 18 | 12.5 | 66.53 | 80 |
| Escherichia coli C83922, | 20 | 6.25 | 55.24 | 80 |
| Escherichia coli O78 | 18 | 6.25 | 47.83 | 80 |
| Aeromonas hydrophila(crucian carp) | 13 | 3.13 | 52.05 | >160 |
| Pseudomonas aeruginosa ATCC27853 | 15 | 3.13 | 52.94 | 80 |
| Pasteurellae gallinarum C48-3 | 12 | 3.13 | 63.63 | 80 |
| MR- Escherichia coli (equine) | 33.5 | 6.25 | 62.73 | 80 |
| MR- Pseudomonas aeruginosa M140 | 19 | 1.56 | 85.56 | 5 |
| MR- Escherichia coli T50(swine) | 11 | 6.25 | 89.92 | 80 |
| Gram-positive bacteria | ||||
| Staphylococcus aureus ATCC25923 | 27 | 3.13 | 50.00 | >160 |
| Staphylococcus aureus ATCC 29213 | 21 | 3.13 | 94.26 | 5 |
| Staphylococcus albus ATCC01331 | 14.5 | 1.56 | 73.36 | 40 |
| MR- Bacillus cereus (pigeon) | 13.5 | 3.13 | 73.36 | 20 |
| MR- Staphylococcus aureus L22(swine) | 15.5 | 6.25 | 88.6 | >160 |
| MR -Staphylococcus aureus QD-CD10 (swine) | 15.5 | 6.25 | 56.93 | >160 |
| MR-Enterococcus faecalis 53A(pet) | 22 | 1.56 | 91.10 | >160 |
| MR-Enterococcus faecalis 52A(pet) | 13.5 | 6.25 | 35.00 | >160 |
| MR-Enterococcus faecalis 37 N(pet) | 29 | 3.13 | 55.00 | >160 |
MR multidrug resistance (in bold), D diameter of inhibition zone, MIC minimum inhibitory concentration, IR inhibitory rate in 30 min, MBC minimal bactericidal concentration
The time-dependent growth inhibitory activities of CHAP are shown in Fig. 2a-d. The results showed that CHAP not only significantly inhibited the growth of standard strains (P < 0.05), but also effectively inhibited the multi-resistant ones (P < 0.05) in 10 min (Fig. 2a and b). The inhibitory rates of all six strains reached 100% in 90 min (Fig. 2c and d). Although there were different growth inhibitory curves, CHAP showed more than 50% inhibitory rate against all six strains in 10 min.
Fig. 2.

a-b Time-dependent inhibitory curves of bacteria treated with CHAP and the bacteria treated with the solvent as control. a Gram-negative bacteria. b Gram-positive bacteria. c-d Time-dependent growth inhibitory rate curves of bacteria treated with CHAP and the bacteria treated with the solvent as control. c Gram-positive bacteria. d Gram-positive bacteria. e-f Time dependent bactericidal curves of Escherichia coli ATCC 25922 treated with CHAP and the bacteria treated with the solvent as control. e At early exponential phase. f At late exponential phase
Bactericidal effect of CHAP
The values of MBC are shown in column 5 of Table 1. By analyzing the values, most bacteria were killed by CHAP at concentrations ranging from 5 μg mL−1 to 80 μg mL−1. However, 7 strains showed no obvious bactericidal effect with the maximum concentration of 160 μg mL−1. The time-dependent bactericidal curves in Fig. 2e and f further revealed that the significant bactericidal effect of CHAP on both the bacteria grown to early and late exponential phases from 10–240 min (P < 0.01). Especially the early exponential phase bacteria, they were killed completely in only 10 min. The bacteria in stationary phase resulted in lysis after being treated with CHAP for 24 h (Fig. 3a).
Fig. 3.
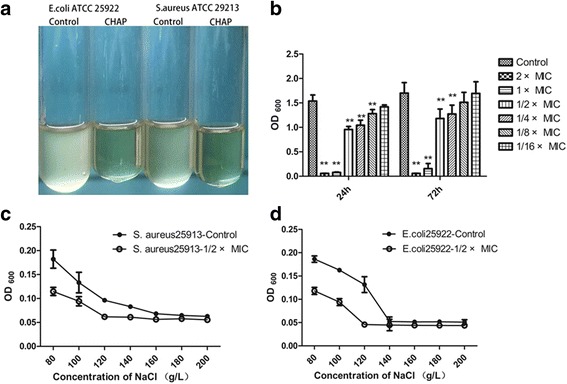
a Bacteriolysis analysis. Bacteria in stationary phase treated with CHAP resulted in lysis with the solvent as control. b The inhibitory effect of CHAP on bacterial biofilm for 24 h and 72 h. c-d The effect of CHAP on NaCl permeability of Escherichia coli ATCC 25922 and Staphylococcus aureus ATCC29213
Bacterial biofilms assay
The formation of biofilms of Staphylococcus aureus ATCC29213 was decreased with the increase of the concentration of CHAP (Fig. 3b). The 2 × MIC, 1 × MIC, 1/2 × MIC, 1/4 × MIC, 1/8 × MIC of CHAP could inhibited the formation of biofilms (P < 0.01) in 24 h and 2 × MIC, 1 × MIC, 1/2 × MIC, 1/4 × MIC of CHAP could significantly decrease the formation of biofilms (P < 0.01) in 72 h. Remarkably, in the 2 × MIC and 1 × MIC groups, there were almost no biofilm formation.
NaCl permeability assay
As shown in Fig. 3c and d, the values of OD600 of Escherichia coli ATCC 25922 and Staphylococcus aureus ATCC29213 cultures decreased with the increased concentration of NaCl solution and reached to their minimum values at concentration above 160 g L−1 and above 140 g L−1, respectively. By adding CHAP, the value of OD600 in both Escherichia coli ATCC 25922 and Staphylococcus aureus ATCC29213 groups decreased to the lowest value at concentration of NaCl above 120 g L−1.
Electron microscopy observations
The morphology of the Escherichia coli ATCC 25922 and Staphylococcus aureus ATCC29213 investigated by SEM is shown in the Fig. 3. Compared to the smooth, straight and unbroken surface of the control cells (Fig. 4a and c), the strains treated with CHAP for 30 min appeared severely damaged (Fig. 3b and d). The TEM images further demonstrated that the bacterial surfaces were damaged by the effect of CHAP (Fig. 5a and c) compared with the control group (Fig. 4b and d).
Fig. 4.

The morphology of Escherichia coli ATCC 25922 and Staphylococcus aureus ATCC29213 were investigated by scanning electron microscopy. a-b The control group. c-d The Escherichia coli ATCC 25922 treated with CHAP. Viscous substances were adhering to almost all CHAP treated cells, which got large number of bacteria together (arrowheads); Some bacteria showed variable length, rough cell surfaces or globular protrusions on their surfaces, and even appeared to collapse (arrows)
Fig. 5.
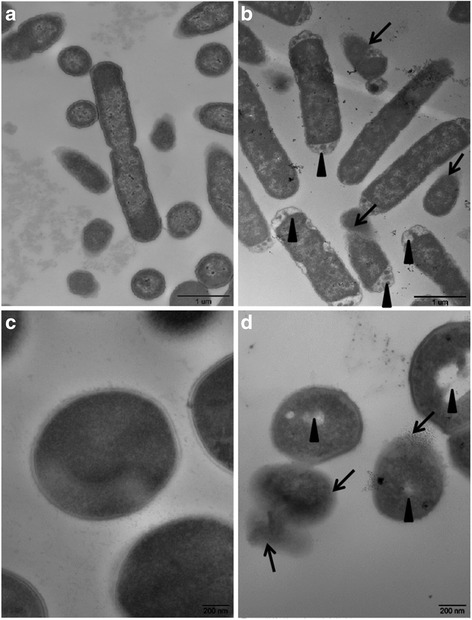
The morphology of Escherichia coli ATCC 25922 Staphylococcus aureus ATCC29213 were investigated by transmission electron microscopy. a-b The control group. c-d The Escherichia coli ATCC 25922 treated with CHAP. Most of the bacteria were translucent and pores were evident on walls especially at the two terminals of each cell (arrowheads). There was some intracellular substance released from many bacteria (arrows).
Hemolytic assay and embryotoxicity assay
The hemolysis of CHAP was 38.9% at the concentration of 360 μg mL−1 which was more than 50 times higher than the MIC values for all the detected bacteria (see Additional file 2). And the embryotoxicity assay showed that even CHAP of 6 × MIC dose against Escherichia coli ATCC25922 did not induce toxicity toward chicken embryos, that is, there was no dead or significant decrease of body weight compared to the control group (P > 0.05) (see Additional file 3).
Stability in different temperatures and in 50% plasma
The antimicrobial activity of CHAP did not decrease in different temperatures even when it was treated in 121 °C for 30 min compared with CHAP stored in 4 °C (P > 0.05) (Fig. 6a). It well proved that CHAP was capable of stability in various temperatures.
Fig. 6.

The stability of CHAP. a The stability of the CHAP in different temperatures. b The stability of the CHAP in chicken and rabbit plasma
Compared with CHAP diluted in the solvent, the antimicrobial activity of CHAP showed no change in the treatment of chicken plasma and a slight but no significant decrease in treatment of rabbit plasma (P >0.05) (Fig. 6b), which demonstrated that CHAP was of well stability in the plasma.
Discussion
Since the first anti-Streptococcus peptide was identified from the cow’s milk [36], the enzyme strategy of isolating AMPs on a large scale has been a feasible method as Bolscher postulated [37]. Our lab isolated the peptides from the hemoglobin of chickens by using a simple and practical way and studied the antimicrobial activities against 19 bacterial strains, including 9 multidrug-resistant bacteria. At the same time, the properties such as hemolytic activity, embryotoxicity and stability in different temperatures and plasma were detected, which laid a foundation for its further employment in agricultural production, public health and medication.
Antibiotics have been helping humans to fight against hazardous infections since Alexander Fleming discovered the first antibiotic, penicillin, in 1928 [38]. However, resistance to most antibiotics was discovered shortly after their applications. For example, penicillin resistance arose in 1946 just one year after its introduction to clinics [1]. The main target of most AMPs is cell membrane and there are several models for explaining the process such as barrel-stave pore model, thoroidal pore model and carpet model [3, 29, 39], and it tends to be difficult for bacteria to totally change this basic structure to resist the effect of AMPs [3]. Given the unique antimicrobial mechanism of AMPs, it was not surprising that most AMPs induced little or no resistance [40, 41]. Although there are reports and doubts about the resistance of some peptides [41, 42], there is no report about the natural ones so far, which means the development of natural AMPs is high in potential.
In our study, both the antimicrobial and bactericide results showed that CHAP was capable of strong and rapid activities against various bacteria and even some multi-resistant strains, implying its wider utility in the prevention and treatment of infectious agents, which was similar with the hemosidins reported before [16, 17]. By specifically analyzing the results above, there was no obvious difference between the antibacterial activity against Gram-negative bacteria and Gram-positive bacteria, even between the standard strains and the multi-resistant strains, suggesting that the target of CHAP is the common component of bacteria such as the cell membrane like most AMPs reported before [29, 43]. The biofilm and the NaCl permeability results showed that CHAP could inhibit the formation of bacterial biofilms and change the permeability of some Gram-negative bacteria and Gram-positive bacteria to some extent. With the confirmation of the mechanism of most AMPs, the EM observations further revealed that CHAP could accumulate copious pathogens nearby and punch through their cell surfaces swiftly [29, 32]. However the specific mechanism needs to be further investigated.
As a double-edged sword, the unique mechanism of targeting cell membranes could also lead to the low selection of some AMPs [3]. Hence, toxicity especially hemolysis and safety problems, are constantly an obstacle to their final applications [1, 40, 44]. According to our study, CHAP demonstrated low hemolysis and embryotoxicity even at rather high concentrations, which further implied that there was relative high selectivity of CHAP between eukaryote cells and prokaryote cells.
Good stability also plays an important role in the application of any biological product. As for the AMPs, the substances such as ions and proteolytic enzymes in the serum may reduce their biological ability to a large extent [29, 45]. In this study, CHAP kept high antimicrobial activity in two kinds of animal serum and different temperatures, suggesting its convenient application, transportation and storage.
Conclusions
In summary, this study firstly reported a practical method of isolating chicken hemosidins (CHAP) from the byproduct and even the pollutant of chicken-slaughtering industries. CHAP has an attractive antimicrobial and bactericidal ability with low hemolysis, low or none in toxicity and good temperature resistance and high stability in serum, which well accounts for their potential of expanding production and high development value in animal medication, breeding industry and environment protection.
Acknowledgements
We would like to thank the National Natural Science Foundation of China, Beijing Key Laboratory of Detection Technology for animal Food safety of the College of Veterinary Medicine, China Agricultural University, Beijing Protein Institute and Beijing Huadu CO.
Funding
This study was funded by the National Natural Science Foundation of China (31072110, 31272515).
Availability of data and materials
All data supporting our findings are included within the article and supplemental files.
Authors’ contributions
RShe conceived and supervised the study; HF and RShe designed experiments; HF, WQ, SS, YZ, YY, MZ and FD performed experiments; HF, WQ, SS analyzed and interpreted the data; HF wrote the manuscript; RShe, MHS and RShi made manuscript revisions. All authors read and approved the final manuscript.
Competing interests
The authors declare that they have no competing interest.
Consent for publication
Not applicable.
Ethics approval and consent to participate
All the animal experiments were approved by the Animal Care and Use Committee of China Agricultural University (permit number: 20150115–058). We followed the guidelines of the China Agricultural University Animal Care and Use Committee when handling the experimental animals during this study (The certificate of Beijing Laboratory Animal employee, ID: 114120800171).
Abbreviations
- AMPs
Antimicrobial peptides
- BHI
Brain heart infusion
- CFU
Colony-forming units
- CHAP
Chicken hemoglobin antimicrobial peptides
- CLSI
Clinical and Laboratory Standards Institute
- CVCC
China Veterinary Culture Collection Center
- LB
Luria-Bertani
- MBC
Minimal bactericidal concentration
- MH
Mueller- Hinto
- MIC
Minimum inhibitory concentration
- OD
Optical density
- SEM
Scanning electron microscopy
- TEM
Transmission electron microscopy
- TSB
Trypticase soy broth
Additional files
The mass spectrum result of CHAP. (PDF 18 kb)
Hemolysis of CHAP. (PDF 143 kb)
Results of the embryotoxicity assay. (PDF 112 kb)
Contributor Information
Fengjiao Hu, Email: hufnegjiaopepper@163.com.
Qiaoxing Wu, Email: 924813953@qq.com.
Shuang Song, Email: 1372510487@qq.com.
Ruiping She, Email: sheruiping@126.com.
Yue Zhao, Email: tsulimoon@126.com.
Yifei Yang, Email: yangyifei1987@163.com.
Meikun Zhang, Email: zmk5678@126.com.
Fang Du, Email: zizhoudufang@126.com.
Majid Hussain Soomro, Email: majidsoomro@hotmail.com.
Ruihan Shi, Email: ruihan6005@163.com.
References
- 1.Lewis K. Platforms for antibiotic discovery. Nat Rev Drug Discov. 2013;12(5):371–87. doi: 10.1038/nrd3975. [DOI] [PubMed] [Google Scholar]
- 2.Ling LL, Schneider T, Peoples AJ, Spoering AL, Engels I, Conlon BP, Mueller A, Schaberle TF, Hughes DE, Epstein S, et al. A new antibiotic kills pathogens without detectable resistance. Nature. 2015;517(7535):455–9. doi: 10.1038/nature14098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zasloff M. Antimicrobial peptides of multicellular organisms. Nature. 2002;415(6870):389–95. doi: 10.1038/415389a. [DOI] [PubMed] [Google Scholar]
- 4.Nizet V. Antimicrobial peptide resistance mechanisms of human bacterial pathogens. Curr Issues Mol Biol. 2006;8(1):11–26. [PubMed] [Google Scholar]
- 5.Gallo RL. The birth of innate immunity. Exp Dermatol. 2013;22(8):517. doi: 10.1111/exd.12197. [DOI] [PubMed] [Google Scholar]
- 6.Ginsburg I, Koren E. Are cationic antimicrobial peptides also ‘double-edged swords’? Expert Rev Anti Infect Ther. 2008;6(4):453–62. doi: 10.1586/14787210.6.4.453. [DOI] [PubMed] [Google Scholar]
- 7.Basu D, Kulkarni R. Overview of blood components and their preparation. Indian J Anaesth. 2014;58(5):529–37. doi: 10.4103/0019-5049.144647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Drago L, Bortolin M, Vassena C, Taschieri S, Del Fabbro M. Antimicrobial activity of pure platelet-rich plasma against microorganisms isolated from oral cavity. BMC Microbiol. 2013;13:47. doi: 10.1186/1471-2180-13-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Merchant ME, Leger N, Jerkins E, Mills K, Pallansch MB, Paulman RL, Ptak RG. Broad spectrum antimicrobial activity of leukocyte extracts from the American alligator (Alligator mississippiensis) Vet Immunol Immunopathol. 2006;110(3–4):221–8. doi: 10.1016/j.vetimm.2005.10.001. [DOI] [PubMed] [Google Scholar]
- 10.Hamidi M, Tajerzadeh H. Carrier erythrocytes: an overview. Drug Deliv. 2003;10(1):9–20. doi: 10.1080/713840329. [DOI] [PubMed] [Google Scholar]
- 11.Brantl V, Gramsch C, Lottspeich F, Mertz R, Jaeger KH, Herz A. Novel opioid peptides derived from hemoglobin: hemorphins. Eur J Pharmacol. 1986;125(2):309–10. doi: 10.1016/0014-2999(86)90044-0. [DOI] [PubMed] [Google Scholar]
- 12.Zhao QY, Piot JM, Gautier V, Cottenceau G. Isolation and characterization of a bacterial growth-stimulating peptide from a peptic bovine hemoglobin hydrolysate. Appl Microbiol Biotechnol. 1996;45(6):778–84. doi: 10.1007/s002530050762. [DOI] [PubMed] [Google Scholar]
- 13.Karelin AA, Philippova MM, Ivanov VT. Proteolytic degradation of hemoglobin in erythrocytes leads to biologically active peptides. Peptides. 1995;16(4):693–7. doi: 10.1016/0196-9781(95)00029-J. [DOI] [PubMed] [Google Scholar]
- 14.Fogaca AC, da Silva PI, Jr, Miranda MT, Bianchi AG, Miranda A, Ribolla PE, Daffre S. Antimicrobial activity of a bovine hemoglobin fragment in the tick Boophilus microplus. J Biol Chem. 1999;274(36):25330–4. doi: 10.1074/jbc.274.36.25330. [DOI] [PubMed] [Google Scholar]
- 15.Alvarez C, Rendueles M, Diaz M. Production of porcine hemoglobin peptides at moderate temperature and medium pressure under a nitrogen stream. Functional and antioxidant properties. J Agric Food Chem. 2012;60(22):5636–43. doi: 10.1021/jf300400k. [DOI] [PubMed] [Google Scholar]
- 16.Mak P, Wojcik K, Silberring J, Dubin A. Antimicrobial peptides derived from heme-containing proteins: hemocidins. Antonie Van Leeuwenhoek. 2000;77(3):197–207. doi: 10.1023/A:1002081605784. [DOI] [PubMed] [Google Scholar]
- 17.Liepke C, Baxmann S, Heine C, Breithaupt N, Standker L, Forssmann WG. Human hemoglobin-derived peptides exhibit antimicrobial activity: a class of host defense peptides. J Chromatogr B Analyt Technol Biomed Life Sci. 2003;791(1–2):345–56. doi: 10.1016/S1570-0232(03)00245-9. [DOI] [PubMed] [Google Scholar]
- 18.Patgaonkar M, Aranha C, Bhonde G, Reddy KV. Identification and characterization of anti-microbial peptides from rabbit vaginal fluid. Vet Immunol Immunopathol. 2011;139(2–4):176–86. doi: 10.1016/j.vetimm.2010.10.012. [DOI] [PubMed] [Google Scholar]
- 19.Srihongthong S, Pakdeesuwan A, Daduang S, Araki T, Dhiravisit A, Thammasirirak S. Complete amino acid sequence of globin chains and biological activity of fragmented crocodile hemoglobin (Crocodylus siamensis) Protein J. 2012;31(6):466–76. doi: 10.1007/s10930-012-9424-7. [DOI] [PubMed] [Google Scholar]
- 20.Fernandes JM, Smith VJ. Partial purification of antibacterial proteinaceous factors from erythrocytes of Oncorhynchus mykiss. Fish Shellfish Immunol. 2004;16(1):1–9. doi: 10.1016/S1050-4648(03)00027-5. [DOI] [PubMed] [Google Scholar]
- 21.Gambacurta A, Piro MC, Ascoli F. Cooperative homodimeric hemoglobin from Scapharca inaequivalvis. cDNA cloning and expression of the fully functional protein in E. coli. FEBS Lett. 1993;330(1):90–4. doi: 10.1016/0014-5793(93)80926-L. [DOI] [PubMed] [Google Scholar]
- 22.Mak P, Wojcik K, Wicherek L, Suder P, Dubin A. Antibacterial hemoglobin peptides in human menstrual blood. Peptides. 2004;25(11):1839–47. doi: 10.1016/j.peptides.2004.06.015. [DOI] [PubMed] [Google Scholar]
- 23.Nedjar-Arroume N, Dubois-Delval V, Adje EY, Traisnel J, Krier F, Mary P, Kouach M, Briand G, Guillochon D. Bovine hemoglobin: an attractive source of antibacterial peptides. Peptides. 2008;29(6):969–77. doi: 10.1016/j.peptides.2008.01.011. [DOI] [PubMed] [Google Scholar]
- 24.Adje EY, Balti R, Kouach M, Dhulster P, Guillochon D, Nedjar-Arroume N. Obtaining antimicrobial peptides by controlled peptic hydrolysis of bovine hemoglobin. Int J Biol Macromol. 2011;49(2):143–53. doi: 10.1016/j.ijbiomac.2011.04.004. [DOI] [PubMed] [Google Scholar]
- 25.Kundu P, Debsarkar A, Mukherjee S. Treatment of slaughter house wastewater in a sequencing batch reactor: performance evaluation and biodegradation kinetics. Biomed Res Int. 2013;2013:134872. doi: 10.1155/2013/134872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhang Y, She R, Liu T, Wengui LI, Jia J. Studies on isolation, purification and antibacterial activities of antibacterial peptides in swine blood. Science & Technology Review (China) 2008;26(2):33–7. [Google Scholar]
- 27.Bao H, She R, Liu T, Zhang Y, Peng KS, Luo D, Yue Z, Ding Y, Hu Y, Liu W, et al. Effects of pig antibacterial peptides on growth performance and intestine mucosal immune of broiler chickens. Poult Sci. 2009;88(2):291–7. doi: 10.3382/ps.2008-00330. [DOI] [PubMed] [Google Scholar]
- 28.Schagger H, von Jagow G. Tricine-sodium dodecyl sulfate-polyacrylamide gel electrophoresis for the separation of proteins in the range from 1 to 100 kDa. Anal Biochem. 1987;166(2):368–79. doi: 10.1016/0003-2697(87)90587-2. [DOI] [PubMed] [Google Scholar]
- 29.Hou Z, Lu J, Fang C, Zhou Y, Bai H, Zhang X, Xue X, Chen Y, Luo X. Underlying mechanism of in vivo and in vitro activity of C-terminal-amidated thanatin against clinical isolates of extended-spectrum beta-lactamase-producing Escherichia coli. J Infect Dis. 2011;203(2):273–82. doi: 10.1093/infdis/jiq029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Nair S, Desai S, Poonacha N, Vipra A, Sharma U. Antibiofilm activity and synergistic inhibition of S. aureus biofilms by bactericidal protein P128 in combination with antibiotics. Antimicrob Agents Chemother. 2016;60(12):7280-9. [DOI] [PMC free article] [PubMed]
- 31.De Oliveira MV, Intorne AC, Vespoli Lde S, Madureira HC, Leandro MR, Pereira TN, Olivares FL, Berbert-Molina MA, De Souza Filho GA. Differential effects of salinity and osmotic stress on the plant growth-promoting bacterium Gluconacetobacter diazotrophicus PAL5. Arch Microbiol. 2016;198(3):287–94. doi: 10.1007/s00203-015-1176-2. [DOI] [PubMed] [Google Scholar]
- 32.Shi J, Ross CR, Chengappa MM, Sylte MJ, McVey DS, Blecha F. Antibacterial activity of a synthetic peptide (PR-26) derived from PR-39, a proline-arginine-rich neutrophil antimicrobial peptide. Antimicrob Agents Chemother. 1996;40(1):115–21. doi: 10.1128/aac.40.1.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cao L, Dai C, Li Z, Fan Z, Song Y, Wu Y, Cao Z, Li W. Antibacterial activity and mechanism of a scorpion venom peptide derivative in vitro and in vivo. PLoS One. 2012;7(7):e40135. doi: 10.1371/journal.pone.0040135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ding Y, Zou J, Li Z, Tian J, Abdelalim S, Du F, She R, Wang D, Tan C, Wang H, et al. Study of histopathological and molecular changes of rat kidney under simulated weightlessness and resistance training protective effect. PLoS One. 2011;6(5):e20008. doi: 10.1371/journal.pone.0020008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Stark M, Liu LP, Deber CM. Cationic hydrophobic peptides with antimicrobial activity. Antimicrob Agents Chemother. 2002;46(11):3585–90. doi: 10.1128/AAC.46.11.3585-3590.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jones FS, Simms HS. The bacterial growth inhibitor (lactenin) of milk : i. the preparation in concentrated form. J Exp Med. 1930;51(2):327–39. doi: 10.1084/jem.51.2.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bolscher JG, van der Kraan MI, Nazmi K, Kalay H, Grun CH, Van’t Hof W, Veerman EC, Nieuw Amerongen AV. A one-enzyme strategy to release an antimicrobial peptide from the LFampin-domain of bovine lactoferrin. Peptides. 2006;27(1):1–9. doi: 10.1016/j.peptides.2005.06.012. [DOI] [PubMed] [Google Scholar]
- 38.Fleming AG. Responsibilities and Opportunities of the Private Practitioner in Preventive Medicine. Can Med Assoc J. 1929;20(1):11–3. [PMC free article] [PubMed] [Google Scholar]
- 39.Pompilio A, Crocetta V, Scocchi M, Pomponio S, Di Vincenzo V, Mardirossian M, Gherardi G, Fiscarelli E, Dicuonzo G, Gennaro R, et al. Potential novel therapeutic strategies in cystic fibrosis: antimicrobial and anti-biofilm activity of natural and designed alpha-helical peptides against Staphylococcus aureus, Pseudomonas aeruginosa, and Stenotrophomonas maltophilia. BMC Microbiol. 2012;12:145. doi: 10.1186/1471-2180-12-145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hancock RE, Sahl HG. Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat Biotechnol. 2006;24(12):1551–7. doi: 10.1038/nbt1267. [DOI] [PubMed] [Google Scholar]
- 41.Bell G, Gouyon PH. Arming the enemy: the evolution of resistance to self-proteins. Microbiology. 2003;149(Pt 6):1367–75. doi: 10.1099/mic.0.26265-0. [DOI] [PubMed] [Google Scholar]
- 42.Habets MG, Brockhurst MA. Therapeutic antimicrobial peptides may compromise natural immunity. Biol Lett. 2012;8(3):416–8. doi: 10.1098/rsbl.2011.1203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Melo MN, Ferre R, Castanho MA. Antimicrobial peptides: linking partition, activity and high membrane-bound concentrations. Nat Rev Microbiol. 2009;7(3):245–50. doi: 10.1038/nrmicro2095. [DOI] [PubMed] [Google Scholar]
- 44.Marr AK, Gooderham WJ, Hancock RE. Antibacterial peptides for therapeutic use: obstacles and realistic outlook. Curr Opin Pharmacol. 2006;6(5):468–72. doi: 10.1016/j.coph.2006.04.006. [DOI] [PubMed] [Google Scholar]
- 45.Bowdish DM, Davidson DJ, Lau YE, Lee K, Scott MG, Hancock RE. Impact of LL-37 on anti-infective immunity. J Leukoc Biol. 2005;77(4):451–9. doi: 10.1189/jlb.0704380. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data supporting our findings are included within the article and supplemental files.


