ABSTRACT
Adult T-cell leukemia/lymphoma is caused by infection with HTLV-1, following a long latent period. Immunotherapy targeting Aurora kinase A, a tumor-associated antigen over-expressed in adult T-cell leukemia/lymphoma, holds great therapeutic potential. We review the evidence in favor of a therapeutic strategy combining vaccination and TCR-gene transfer against this target.
KEYWORDS: Adult T-cell leukemia/lymphoma, aurora kinase A, engineered T cell, immunotherapy, T-cell receptor gene
Adult T-cell leukemia/lymphoma (ATL) is caused by infection with human T-lymphotropic virus 1 (HTLV-1), following a latent period typically spanning many decades.1 Although a recently introduced therapeutic agent, mogamulizumab, a monoclonal antibody for anti-C-C chemokine receptor 4 (CCR4), is expected to improve the survival of ATL patients,2 in daily practice, ATL still remains refractory to conventional chemo-radiotherapies, producing a poor prognosis.3
On the other hand, allogeneic haematopoietic stem cell transplantation (allo-HSCT) has been favored as a viable option for effective treatment of ATL. The anti-ATL tumor effect mediated by allo-HSCT is largely associated with engrafted donor-derived T cells reactive to allogeneic disparity, i.e., graft versus host disease (GVHD).3 In practice, however, the advanced age-related difficulties among patients with ATL largely limit their eligibility for allo-HSCT. Those difficulties include limited acquisition of suitable and timely donors and comorbidities, resulting in an increased risk of treatment-related mortality.1 Despite these difficulties, the durable disease-free survival mediated by allo-HSCT strongly suggests that a T-cell immunity-based treatment strategy is valid for the treatment of ATL. As a corollary, target antigens for T cells mediating the anti-ATL effect have been examined, with a particular emphasis on the HLTV-1-related viral proteins. HTLV-1 Tax and HTLV-1 basic leucine zipper factor (HBZ) were shown to be immunogenic, leading to the elimination of lymphocytes positive for either or both. However, it is as yet unknown whether Tax and HBZ are sufficiently immunogenic to eradicate ATL tumor cells, particularly as primary ATL tumor cells express both Tax and HBZ proteins at low levels.1 Instead, tumor-associated antigens that are abundantly expressed by ATL tumor cells have come under scrutiny, as they might offer alternative targets for immunotherapy against ATL. It has been reported that ATL cells express numerous tumor antigens, including NY-ESO-1, MAGE-A3, MAGE-A4, each of which are recognized by T cells.1
We previously demonstrated that human Telomerase Reverse Transcriptase (hTERT) is also upregulated in ATL cells,4 and that circulating hTERT-specific T cells responsive to ATL tumor cells were detectable in the peripheral blood of ATL patients. Unfortunately, we found previously that hTERT peptide vaccination resulted in limited clinical responses. Furthermore, it proved difficult to expand hTERT-specific T cells in vivo due to the regulatory T cell-like immunosuppressive activity mediated by ATL tumor cells with a CD4+CD25+ phenotype.5 In response to these challenges, we focused upon adoptive transfer therapy using gene-modified T cells with hTERT-specific T-cell receptor (TCR) alpha and beta genes obtained from our previously established CTL clone.4 These gene-modified T cells successfully and specifically killed hTERT-positive ATL tumor cells without unwanted adverse effect upon normal cells, including “steady state” (non-proliferating) haematopoietic progenitor cells.4
Of the alternate ATL tumor-associated antigens, Aurora kinase A (AURKA) , expressed by ATL tumor cells express at supra-normal levels, is now under investigation.6 AURKA is a serine/threonine kinase that plays a crucial role in regulating mitosis. While AURKA is upregulated in many types of malignant cells, expression is limited in normal cells except for testicular tissue. Ectopic overexpression of AURKA is sufficient for transformation of fibroblasts in vitro. High AURKA expression in tumor cells is associated with resistance to conventional therapies, resulting in disease progression and a shorter survival.7 Based on these findings, small-molecule inhibitors specific for AURKA are being developed, and many clinical trials using Aurora kinase-specific inhibitors have been ongoing. However, one of the challenges of molecular-targeting drugs is that multiple agent-resistant cancer stem cells can remain difficult to eradicate.8 By contrast, in our experience, immunotherapy targeting AURKA appeared to have the potential to eradicate cancer stem cells. We have previously identified the immunogenic epitope derived from AURKA, which could induce leukemia-cidal CTLs. Such AURKA-specific CTLs were able to be detected in the circulation of leukemia patients and killed AURKA-positive primary leukemia cells, including leukemia progenitor cells. These results demonstrated the antigenicity of AURKA peptide, and the immunotherapeutic potential of targeting AURKA.9 Moreover, transferring AURKA-specific TCR alpha/beta genes endowed peripheral T cells with AURKA-specific reactivity.10 Thus, adoptive transfer employing AURKA-specific TCR gene-modified T cells might open the door to a novel treatment strategy for patients with relapsed and refractory ATL patients.
While cancer vaccines seem attractive, potentially offering safety, and having ease of production and administration, it is as yet unknown whether AURKA peptide vaccination would be sufficient to initiate anti-AURKA immune responses, to mediate antitumor activity in patients with ATL. However, combination therapy of adoptive transfer of AURKA-specific TCR-transduced T cells with in vivo delivery of the AURKA peptide by vaccination would be expected to facilitate engraftment and augment the antitumor reactivity of those gene-modified T cells in vivo (Fig. 1).
Figure 1.
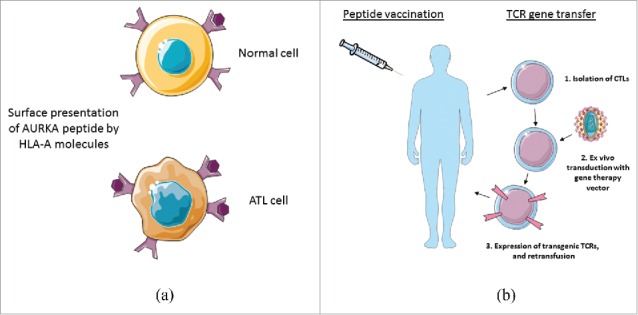
(A) Overexpression of AURKA in ATL tumor cells is assumed that epitopes derived from AURKA would largely been presented in the context of particular HLA molecules, reflecting the increased sensitivity of ATL tumor cells to cytocidal activity mediated by AURKA-specific CTLs. (B) AURKA peptide vaccination in combination with adoptive transfer of AURKA-specific TCR-transduced T cells might be able to enhance the durable engraftment of AURKA-specific T cells, which might provide a long-lasting remission of ATL.
In many cases, ATL cells are positive for CCR4, and may also possess regulatory T-cell function.5 Since CCR4 is also expressed by normal Treg cells, mogamulizumab might also have the benefit of depleting the immunosuppressive effect by Treg cells.2 In addition, immune checkpoint inhibitors, ipilimumab (anti-CTLA-4) and pembrolizumab (anti-PD-1) are demonstrating great promise for the treatment of refractory malignancies. Taken together, these antibodies, by limiting immunosuppressive factors, might be beneficially combined with AURKA-directed immunotherapies, offering considerable promise of improved outcomes for patients with refractory ATL.
Disclosure of potential conflicts of interest
No potential conflicts of interest were disclosed.
References
- 1.Nishikawa H, Maeda Y, Ishida T, Gnjatic S, Sato E, Mori F, Sugiyama D, Ito A, Fukumori Y, Utsunomiya A et al.. Cancer/testis antigens are novel targets of immunotherapy for adult T-cell leukemia/lymphoma. Blood 2012; 119:3097-104; PMID:22323448; http://dx.doi.org/ 10.1182/blood-2011-09-379982 [DOI] [PubMed] [Google Scholar]
- 2.Remer M, Al-Shamkhani A, Glennie M, Johnson P. Mogamulizumab and the treatment of CCR4-positive T-cell lymphomas. Immunotherapy 2014; 6:1187-206; PMID:25496334; http://dx.doi.org/ 10.2217/imt.14.94 [DOI] [PubMed] [Google Scholar]
- 3.Kato K, Akashi K. Recent Advances in Therapeutic Approaches for Adult T-cell Leukemia/Lymphoma. Viruses 2015; 7:6604-12; PMID:26694446; http://dx.doi.org/ 10.3390/v7122960 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Miyazaki Y, Fujiwara H, Asai H, Ochi F, Ochi T, Azuma T, Ishida T, Okamoto S, Mineno J, Kuzushima K et al.. Development of a novel redirected T-cell-based adoptive immunotherapy targeting human telomerase reverse transcriptase for adult T-cell leukemia. Blood 2013; 121:4894-901; PMID:23641014; http://dx.doi.org/ 10.1182/blood-2012-11-465971 [DOI] [PubMed] [Google Scholar]
- 5.Yano H, Ishida T, Inagaki A, Ishii T, Kusumoto S, Komatsu H, Iida S, Utsunomiya A, Ueda R. Regulatory T-cell function of adult T-cell leukemia/lymphoma cells. Int J Cancer 2007; 120:2052-7; PMID:17278106; http://dx.doi.org/ 10.1002/ijc.22536 [DOI] [PubMed] [Google Scholar]
- 6.Tomita M, Mori N. Aurora A selective inhibitor MLN8237 suppresses the growth and survival of HTLV-1-infected T-cells in vitro. Cancer Sci 2010; 101:1204-11; PMID:20180813; http://dx.doi.org/ 10.1111/j.1349-7006.2010.01499.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Malumbres M, Perez de Castro I. Aurora kinase A inhibitors: promising agents in antitumoral therapy. Expert Opin Ther Targets 2014; 18:1377-93; PMID:25200357; http://dx.doi.org/ 10.1517/14728222.2014.956085 [DOI] [PubMed] [Google Scholar]
- 8.Kim Y, Joo KM, Jin J, Nam DH. Cancer stem cells and their mechanism of chemo-radiation resistance. Int J Stem Cells 2009; 2:109-14; PMID:24855529; http://dx.doi.org/ 10.15283/ijsc.2009.2.2.109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ochi T, Fujiwara H, Suemori K, Azuma T, Yakushijin Y, Hato T, Kuzushima K, Yasukawa M. Aurora-A kinase: a novel target of cellular immunotherapy for leukemia. Blood 2009; 113:66-74; PMID:18820130; http://dx.doi.org/ 10.1182/blood-2008-06-164889 [DOI] [PubMed] [Google Scholar]
- 10.Nagai K, Ochi T, Fujiwara H, An J, Shirakata T, Mineno J, Kuzushima K, Shiku H, Melenhorst JJ, Gostick E et al.. Aurora kinase A-specific T-cell receptor gene transfer redirects T lymphocytes to display effective antileukemia reactivity. Blood 2012; 119:368-76; PMID:22025529; http://dx.doi.org/ 10.1182/blood-2011-06-360354 [DOI] [PubMed] [Google Scholar]


