Abstract
Objective
Fetal/neonatal alloimmune thrombocytopenia (FNAIT) is a severe bleeding disorder which can result in intracranial hemorrhage (ICH), leading to death or neurological sequelae. In Caucasians, maternal anti-HPA- 1a antibodies (abs) are responsible for the majority of cases. No predictive factors for ICH are available to guide prophylactic treatment during pregnancy. In this study, we investigated abs from mothers with ICH-positive FNAIT and with ICH-negative FNAIT in order to identify serological and functional differences between the groups.
Approach and Results
In an antigen capture assay, we observed a stronger binding of +ICH abs to endothelial cell (EC)-derived αvβ3. By absorption experiments, we subsequently identified anti-HPA-1a abs of anti-αvβ3 specificity in the +ICH, but not in the −ICH cohort. Only the anti-αvβ3 subtype, but not the anti-β3 subtype, induced EC apoptosis of HPA-1a positive ECs by caspase-3/7 activation, and mediated by reactive oxygen species. In addition, only the anti-αvβ3 subtype, but not the anti-β3 subtype, interfered with EC adhesion to vitronectin and with EC tube formation.
Conclusions
We conclude that the composition of the anti-HPA-1a antibody subtype(s) of the mother may determine whether ICH occurs. Analysis of anti-HPA-1a abs of the anti-αvβ3 subtype in maternal serum has potential in the diagnostic prediction of ICH development and may allow for modification of prophylactic treatment in FNAIT.
Introduction
Fetal/neonatal alloimmune thrombocytopenia (FNAIT) is a bleeding disorder of the fetus and newborn in which maternal alloantibodies bind to the infant’s platelets and cause their destruction during pregnancy and after birth. In Caucasians, approximately 80% of FNAIT cases are induced by antibodies against human platelet antigen 1a (HPA-1a).1,2 The most devastating risk of FNAIT is intracranial hemorrhage (ICH) leading to death or persistent neurological sequelae in approximately 10% of the clinically symptomatic cases.3,4 It is currently unclear which factors determine whether ICH will occur. Modality of birth, birth weight, and the presence of other bleeding symptoms are not associated with ICH, and the association between low platelet counts and ICH is loose.5,6 In subsequent pregnancies of mothers immunized against HPA-1a, only sibling history, but no laboratory test has been shown to be predictive for the risk of ICH.7,8
HPA-1a is formed by a single amino acid substitution (Leu33Pro) located on the flexible PSI-domain of the integrin β3 chain.9 On platelets, the β3 chain forms heterodimers either with αIIb or with αv, which functions as fibrinogen or vitronectin receptor, respectively.10 In contrast to αIIbβ3, αvβ3 is also found on endothelial cells, smooth muscle cells, and different cultured cells.11 Several studies demonstrated that HPA-1a is constitutively expressed on endothelial αvβ3.12,13 It has also been shown that anti-HPA-1a antibodies can affect endothelial integrity and the spreading capability of these cells14, indicating that vascular damage may be involved in the pathomechanism of FNAIT. However, others have reported no effect of anti-HPA-1a antibodies on endothelial activation and/or integrity.15 Recently, Yougbaré et al.16 demonstrated that anti-β3 isoantibodies produced in β3 knockout mice can induce ICH in pups by impairment of angiogenesis rather than by thrombocytopenia. The relevance of this finding for the development of ICH in humans is not clear, especially since ICH is far less frequent in humans than observed in the animal model, and a “booster” effect as described in mice is absent in men, i.e. likelihood of ICH does not increase with the number of pregnancies in humans.17 It appears likely that these differences are related to the fact that iso-antibodies (in the murine model) and allo-antibodies (in humans) are not alike. Furthermore, frequency and natural history of ICH in humans suggests that the composition of the maternal anti-HPA-1a antibody repertoire could diverge between FNAIT cases with and without ICH.
In this study, we asked the question whether a specific anti-HPA-1a antibody subtype exists in FNAIT cases with ICH, in comparison to FNAIT cases without ICH. Our results show that anti-HPA-1a antibodies in FNAIT cases with ICH bind specifically to the αvβ3 complex, trigger endothelial apoptosis via reactive oxygen species, and interfere with angiogenesis.
Materials and Methods
Materials and methods are available in the online-only Data Supplement
Results
Anti-HPA-1a antibodies from +ICH and −ICH cases show different binding patterns
The binding of anti-HPA-1a antibodies derived from −ICH cases (n=18; Table 1) and +ICH cases (n=18; Table 2) to αIIbβ3 and αvβ3 integrins derived from HPA-1aa platelets and ECs was investigated in an antigen capture assay. As shown in Figure 1, no significant difference between the two cohorts was observed in their binding to platelet-derived αIIbβ3 (a). However, a significant difference was observed when binding to both platelet-derived αIIbβ3 and αvβ3 was analysed (b). The differences between both cohorts became more significant when sera were tested against EC-derived αvβ3 immobilized by moabs against αvβ3 (c) and β3 (d). These results indicate that +ICH anti-HPA-1a might contain additional antibody specificity, most probably against αvβ3 compound epitope(s).
Table 1.
Clinical and laboratory details of −ICH cases
| No. | Mother | Newborn | anti-αvβ3 (OD) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| gravida/para | antibody specificity | week of gestation | sex | platelet nadir (G/l) | ICH | other bleeding symptoms | bleeding excluded by | ||
| 1 | II/I | HPA-1a | 28+4 | M | 12 | no | H | ultrasound | 0.300 |
| 2 | II/II | HPA-1a | 38+4 | F | 22 | no | P, H | ultrasound | 0.033 |
| 3 | NR | HPA-1a | 36+5 | M | 29 | no | P | ultrasound | 0.048 |
| 4 | I/I | HPA-1a | 40+0 | M | 6 | no | P | ultrasound | 0.320 |
| 5 | NR | HPA-1a | 40+2 | F | 11 | no | P | ultrasound | 0.030 |
| 6 | NR | HPA-1a | 28+0 | F | 24 | no | P | ultrasound | 0.152 |
| 7 | III/III | HPA-1a | 36+5 | F | 13 | no | P | ultrasound | 0.324 |
| 8 | I/I | HPA-1a | 41+0 | F | 5 | no | P | ultrasound | 0.273 |
| 9 | I/I | HPA-1a | 38+0 | M | 15 | no | P | ultrasound | 0.238 |
| 10 | I/I | HPA-1a | 41+2 | M | 24 | no | P | ultrasound | 0.215 |
| 11 | I/I | HPA-1a | 38+2 | M | 15 | no | P, H | ultrasound | 0.089 |
| 12 | I/I | HPA-1a | 40+0 | F | 3 | no | P, H | ultrasound | 0.174 |
| 13 | III/II | HPA-1a | 37+5 | F | 13 | no | P, H | ultrasound | 0.084 |
| 14 | III/III | HPA-1a | 39+3 | F | 18 | no | H | ultrasound | 0.098 |
| 15 | I/I | HPA-1a | 38+4 | M | 13 | no | P, H | ultrasound | 0.167 |
| 16 | IV/II | HPA-1a | 34+5 | NR | 7 | no | P | ultrasound | 0.204 |
| 17 | IV/III | HPA-1a | 40+0 | M | 18 | no | P | ultrasound | 0.289 |
| 18 | II/II | HPA-1a | 40+0 | M | 9 | no | P, U | ultrasound | 0.245 |
NR = not reported; P = petechiae; H = hematoma; U = hematuria
Table 2.
Clinical and laboratory details of +ICH cases
| No. | Mother | Newborn | anti-αvβ3 (OD) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| gravida/para | antibody specificity | week of gestation | sex | platelet nadir (G/l) | ICH | bleeding localization | other bleeding symptoms | ||
| 1 | I/I | HPA-1a | 38+0 | F | 18 | yes | parenchymal, left fronto-parietal lobe | no | 0.674 |
| 2 | I/I | HPA-1a | 38+6 | F | 21 | yes | parenchymal, left side | P, H | 0.488 |
| 3 | II/II | HPA-1a | 40+3 | M | 3 | yes | NR | P, H | 0.664 |
| 4 | VI/V | HPA-1a | 39+0 | M | 5 | yes | parenchymal, left temporal lobe | P | 0.800 |
| 5 | II/II | HPA-1a | 37+6 | F | 5 | yes | subependymal, left side | P, H | 0.777 |
| 6 | II/I | HPA-1a | 40+0 | M | 30 | yes | left thalamus and internal capsule | P | 0.402 |
| 7 | I/I | HPA-1a | 39+6 | M | 10 | yes | periventricular, right side, and plexus cyst | P, H | 0.999 |
| 8 | IV/II | HPA-1a | 34+4 | NR | 5 | yes | intra- and periventricular | 0.549 | |
| 9 | III/II | HPA-1a | 36+6 | F | 45 | yes | parenchymal, right temporal lobe, grade IV | P | 0.685 |
| 10 | III/III | HPA-1a | 41+2 | F | 7 | yes | parenchymal, left parieto-occipital lobe | P,H | 0.721 |
| 11 | II/I | HPA-1a | 36+0 | F | 14 | yes | parenchymal, left parieto-temporal lobe | P | 0.689 |
| 12 | II/I | HPA-1a | 38+0 | M | 17 | yes | parenchymal, right temporal lobe | H, U | 0.780 |
| 13 | II/II | HPA-1a | 34+5 | M | 24 | yes | intraventricular | P | 0.548 |
| 14 | NR | HPA-1a | NR | F | 12 | yes | parenchymal | no | 0.369 |
| 15 | III/I | HPA-1a | 41+3 | F | 12 | yes | parenchymal, left parieto-occipital lobe and right frontal lobe | P, H | 0.995 |
| 16 | NR | HPA-1a | 39+0 | M | 2 | yes | parenchymal | P, H | 0.565 |
| 17 | I/I | HPA-1a | 38+0 | M | 10 | yes | porencephalic cyst, right frontal lobe | P, H | 0.686 |
| 18 | II/I | HPA-1a | NR | M | 20 | yes | parenchymal, left and right hemisphere | no | 0.206 |
NR = not reported; P = petechiae; H = hematoma; U = hematuria; IU = in utero
Figure 1. Analysis of maternal anti-HPA-1a derived from −ICH (n=18) and +ICH (n=18) in an antigen capture assay.
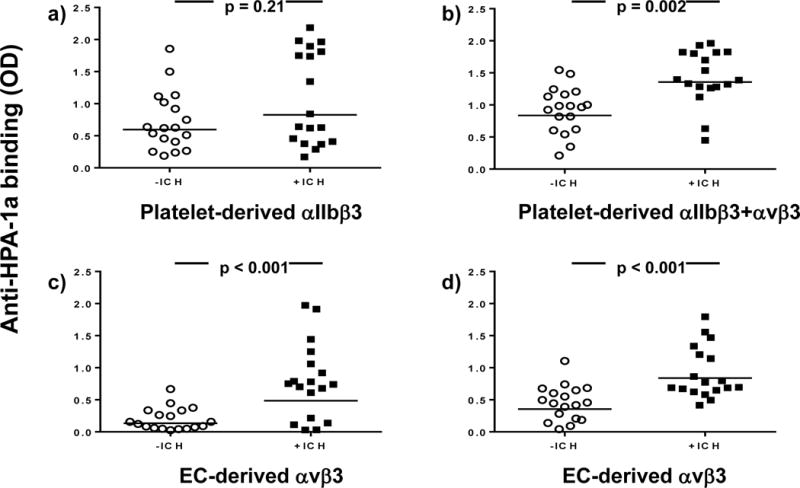
HPA-1aa platelets (upper panel) or endothelial cells (bottom panel) were incubated with anti-HPA-1a sera and moabs against aIIbb3 (a), avb3 (c) and b3 (b, d). After cell lysis, the moab-(aIIbb3/avb3/b3)-anti-HPA-1a trimolecular complex was immobilized on microtiter wells coated with anti-mouse IgG. Binding of anti-HPA-1a antibodies to platelet-derived aIIbb3, aIIbb3 + avb3 and endothelial-derived avb3 was detected with enzyme labelled anti-human IgG. Statistical analysis was performed by Mann-Whitney U Test.
Anti-HPA-1a antibodies from +ICH cases contain a specific anti-αvβ3 subtype
To prove this hypothesis, we aimed to isolate anti-αvβ3 by absorbing +ICH sera with αIIbβ3-coated beads. For the evaluation of this approach, moabs against αIIbβ3, β3 and αvβ3 complex were first absorbed with these beads and tested with CHO cells expressing either αIIbβ3 or αvβ3. Absorption removed completely anti-β3 and anti-αIIbβ3, but not anti-αvβ3 (Supplemental figure 1). Subsequently, all human sera were absorbed with αIIbβ3-coated beads, and the remaining anti-HPA-1a antibodies (absorbate) were re-tested by antigen capture assay (Figure 2A). Absorbates from both cohorts became largely non-reactive with platelet-derived αIIbβ3 and αvβ3, with no differences between the cohorts (upper panel). In contrast, only sera from +ICH, but not from −ICH cases, showed significant reaction with EC-derived αvβ3 confirming our assumption that +ICH sera contain anti-HPA-1a specifically reactive with αvβ3 complex. Note that absorption reduced overall reactivity for both cohorts significantly (in comparison to Figure 1), indicating that relevant amounts of anti-αIIbβ3 and anti-β3, present in sera from both cohorts, were removed by absorption.
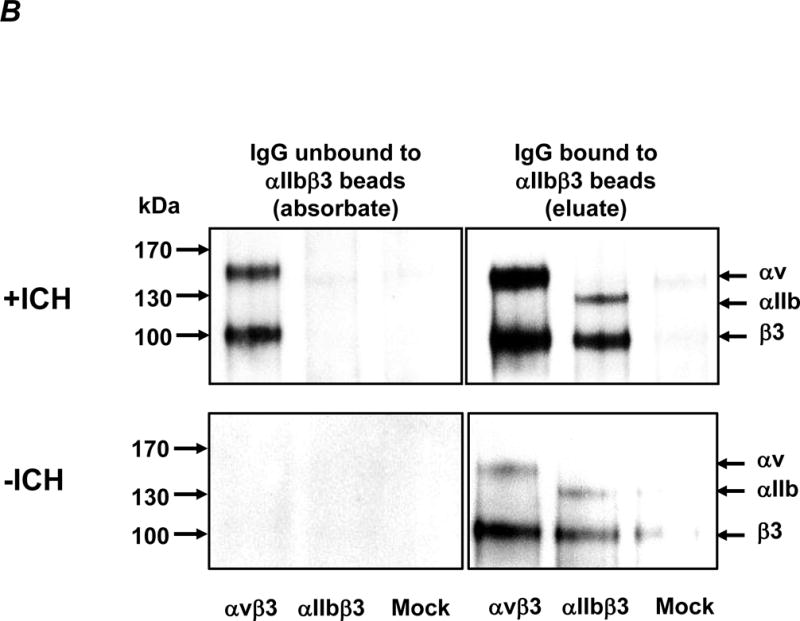
Figure 2. Identification of an anti-HPA-1a avb3-specific subtype in +ICH cases after absorption of other subtypes with αIIbb3 beads.
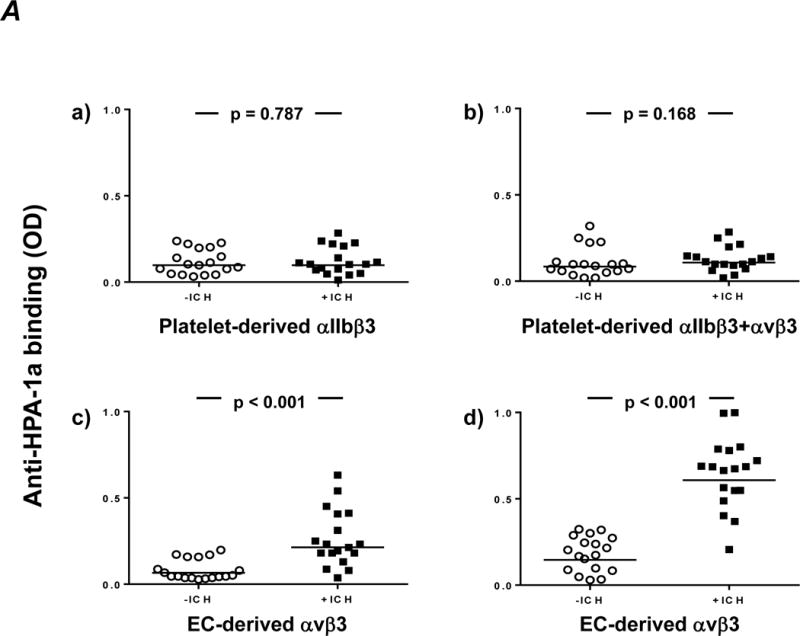
A. Maternal anti-HPA-1a antibodies (n=18 per cohort) were pre-absorbed with aIIbb3 beads to remove HPA-1a antibodies against aIIbb3 and b3. Afterwards, the absorbate was incubated with HPA-1aa platelets (upper panel) or endothelial cells (bottom panel) and moabs against aIIbb3 (a), avb3 (c) or b3 (b,d). After cell lysis, the trimolecular antigen-antibody complex was immobilized on microtiter wells coated with anti-mouse IgG. Binding of anti-HPA-1a antibodies was detected with horseradish peroxidase (HRP)-labeled anti-human IgG. Statistical analysis was performed by Mann-Whitney U Test. Note that after absorption, avb3-specific anti-HPA-1a remains detectable in the +ICH cohort only (c,d). This antibody specificity reacts more readily with avb3 immobilized with moab AP3 (d) than with avb3 immobilized with moab 23C6 (c), possibly indicating epitope interference.
B. Maternal anti-HPA-1a antibodies from the +ICH (top) and −ICH (bottom) cohorts were absorbed with aIIbb3 beads. Unbound IgG (the absorbates) and bead-bound IgG (the eluates) were incubated with biotin-labelled transfected (avb3, aIIbb3) or non-transfected (mock) CHO cells, as indicated. After washings, CHO cells were lysed, the antigen-antibody complex was precipitated with protein-G coupled beads and separated on 10% SDS-PAGE under non-reducing conditions. After blotting, precipitated proteins were visualized by the use of enzyme-labeled streptavidin and a chemiluminescence system. In the –ICH cohort (bottom panel), all antibodies were removed by the beads as indicated by a non-reactive absorbate (left). Antibodies of anti-aIIbb3/anti-b3 specificity could be eluted from these beads (right). Note that anti-b3 is capable to pull down aIIbb3 and avb3 integrins. In contrast, in the +ICH cases (top panel), removal of anti-aIIbb3/anti-b3 antibodies leaves antibodies of anti-avb3 specificity behind (left). The eluate is reactive with both integrins, indicating presence of anti-b3/anti-aIIbb3 (right). One representative gel from n=9 independent experiments is shown.
To further confirm the presence of anti-αvβ3 in the +ICH cohort, anti-HPA-1a antibodies present in absorbates as well as antibodies eluted from αIIbβ3-coated beads (eluates) were investigated by immunoprecipitation using biotin-labelled αIIbβ3 or αvβ3 transfected CHO cells (Figure 2B). In the absorbates, anti-αvβ3 reactivity could be detected in +ICH, but not in –ICH cohort, and this antibody specificity did not show cross reactivity with αIIbβ3 (neither anti-αIIbβ3 nor anti-β3). In contrast, cross-reactive antibodies against αvβ3 and αIIbβ3 were found in eluates from both cohorts. All 18 immunoblots were evaluated by integrity density measurement, demonstrating that the difference between the +ICH cohort and the –ICH cohort for anti-αvβ3 was significant (p=0.00042) in the absorbate, but non-significant for αvβ3 (p=0.13) and αIIbβ3 (p=0.48) in eluates. Taken together, our results suggest that three different subtypes of anti-HPA-1a antibodies can exist in FNAIT sera: anti-αIIbβ3, anti-β3, and anti-αvβ3. In contrast to −ICH cases, sera from +ICH cases contain significant amounts of the anti-αvβ3 subtype.
The anti-αvβ3 subtype of anti-HPA-1a interferes with EC functions
Based on the fact that the anti-αvβ3 subtype is present in sera from +ICH cases and reacts predominantly with EC, we sought to investigate whether this antibody type interferes with EC function. All functional experiments were performed with IgG fractions obtained from n=9 human sera from each cohort, both before (pre) and after (post) absorption with αIIbβ3-coated beads.
First, interference of receptor-ligand binding was analysed by cell adhesion assay. ECs were incubated with isolated IgG fractions prior to adhesion onto vitronectin-coated wells. As shown in Figure 3A, +ICH IgG significantly inhibited EC adhesion (black), whereas −ICH IgG did not (white). Removal of anti-HPA-1a antibodies of the anti-β3 and anti-αIIbβ3 subtype (Figure 3A, post) had no influence on this effect, demonstrating that this effect was mediated by the anti-αvβ3 subtype.
Figure 3. The avb3-specific subtype of anti-HPA-1a interferes with endothelial function.
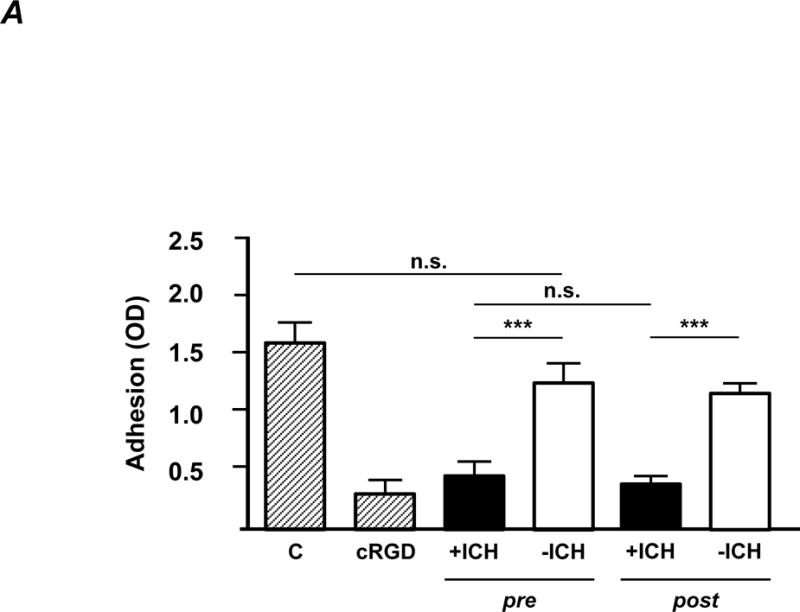
IgG was purified from maternal sera from +ICH cases (n=9; black columns) and from −ICH cases (n=9; white columns) either before (pre) or after (post) absorption with aIIbb3 beads and then further studied.
A. Cell adhesion. Endothelial cells were incubated with purified IgG (20 μg/ml) and added onto microtiter wells pre-coated with vitronectin. After washings, adherent cells were stained with crystal violet and measured in an ELISA reader. Statistical analysis was performed by 1-way ANOVA followed by Bonferroni’s post-hoc test; n.s. = not significant. C = control IgG from healthy donors; cRGD was used as a positive control. Note that only antibodies from +ICH cases hinder endothelial cell adhesion, with no difference between pre- and post-absorption experiments.
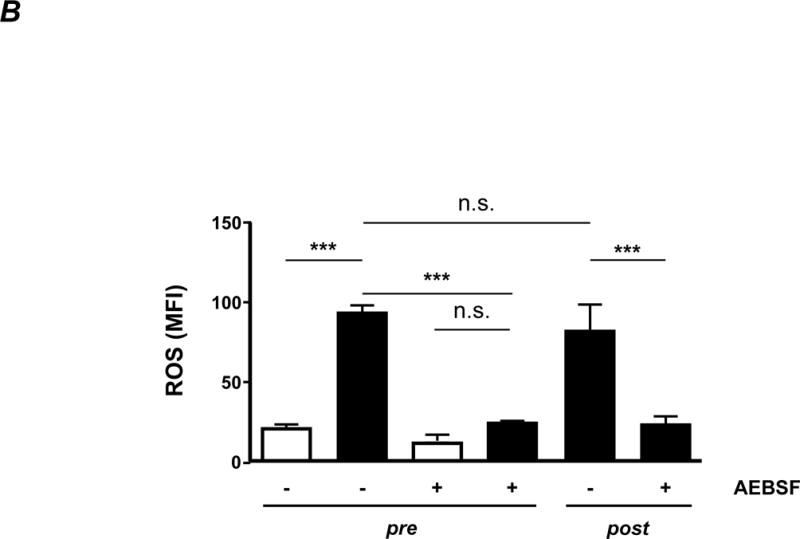
B. ROS production. Endothelial cells were incubated with purified IgG (20 μg/ml) in the presence of DCFDA. The production of fluorescent DCF correlates with cellular ROS production and was measured by flow cytometry. AEBSF was used as an antioxidant in some experiments, as indicated. Statistical analysis was performed by 1-way ANOVA followed by Bonferroni’s post-hoc test. n.s. = not significant. Note that only antibodies from +ICH cases induce endothelial ROS, with no difference between pre- and post-absorption experiments.
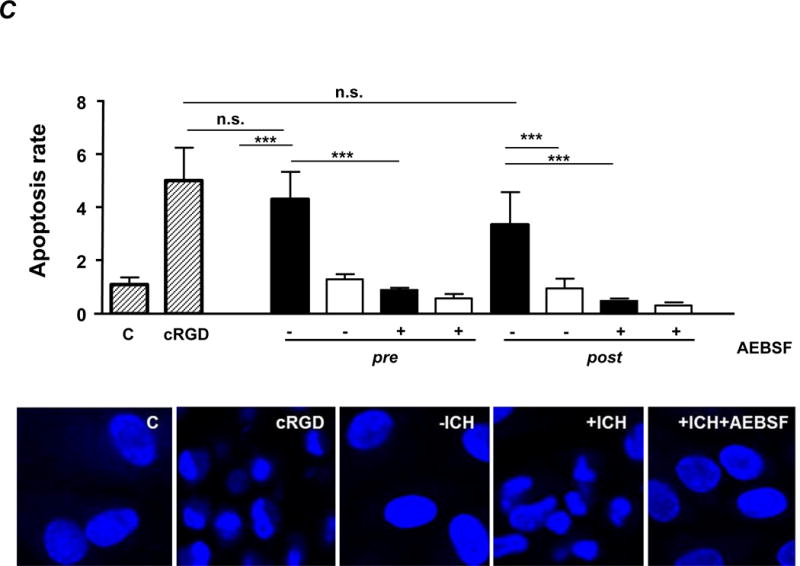
C. Cell apoptosis. Purified IgG (20 μg/ml) was added to endothelial monolayers and caspase 3/7 activity was measured by luminometry (upper panel). Some experiments were performed in the presence of a ROS inhibitor (AEBSF). Statistical analysis was performed by 1-way ANOVA followed by Bonferroni’s post-hoc test; n.s. = not significant; C = control IgG from healthy donors. cRGD was used as positive control. Note that only antibodies from +ICH cases induce apoptosis, with no difference between pre- and post-absorption experiments. Nucleus staining of endothelial cells with DAPI after incubation with anti-HPA-1a IgG post-absorption and controls was performed (lower panel). Representative pictures (60fold magnification) from n=9 independent experiments per cohort are shown.
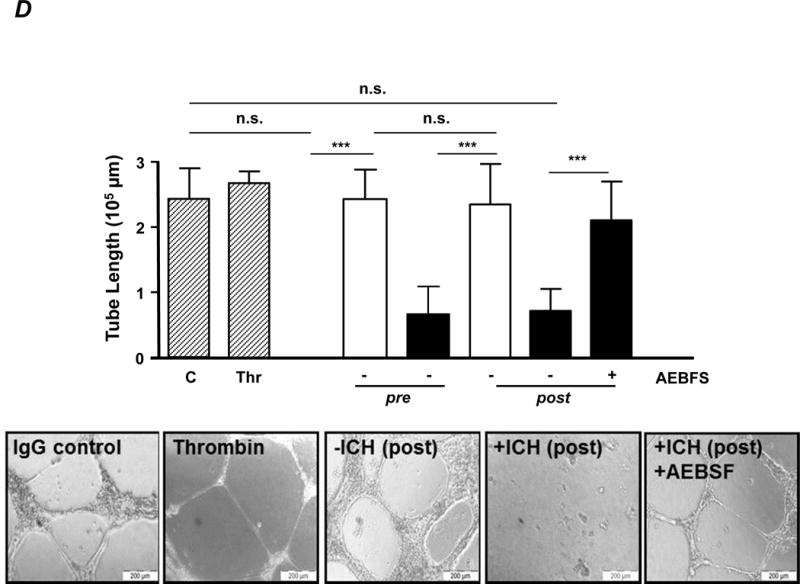
D. Tube formation. Purified IgG (40 μg/ml) was added to endothelial cells, and tube formation was investigated by microscopy. Data are given as mean of tube length in μm +SD (upper panel). Some experiments were performed in the presence of a ROS inhibitor (AEBSF) as indicated. Statistical analysis was performed by 1-way ANOVA followed by Bonferroni’s post-hoc test; n.s. = not significant. C = control IgG; Thr = thrombin. Note that only antibodies from +ICH cases reduce tube length, with no difference between pre- and post-absorption experiments. Representative microphotographs of endothelial tube formation assays as outlined are given in the lower panel.
It is known that disruption of EC adhesion onto extracellular matrix(ECM) results in detachment-induced apoptosis, termed anoikis, which is associated with increased intracellular ROS level.18,19 To analyse whether antibodies of the anti-αvβ3 subtype can also trigger endothelial anoikis, the generation of intracellular ROS induced by anti-HPA-1a antibodies was first measured by oxidation of DCFDA using flow cytometry. As shown in Figure 3B, only +ICH IgG (black) induced ROS, both pre and post absorption, indicating that the anti-αvβ3 subtype triggers ROS production. In the presence of AEBSF, a specific inhibitor of NADPH oxidase20, ROS production was abrogated. Similar results were obtained with N-acetylcysteine (data not shown).
In accordance with these observations, binding of the anti-αvβ3 subtype triggered endothelial apoptosis as measured by caspase 3/7 assay (Figure 3C). In particular, the apoptosis rate induced by +ICH IgG remained unchanged after removal of the other subtypes (pre vs. post, p=0.106). Apoptosis was confirmed by morphological assessment of chromatin DNA cleavage (Figure 3C).
Subsequently, an endothelial tube formation assay was performed to analyse whether the anti-αvβ3 antibody subtype would also affect angiogenesis (Figure 3D). +ICH IgG (black), but not −ICH IgG (white), significantly reduced tube length. Again, only the anti-αvβ3 subtype mediated the biological effect; removal of the other antibody subtypes by absorption had no influence on tube formation. The biological effect was ROS-dependent and could be abrogated by AEBSF. Representative microphotographs are shown in Figure 3D (bottom panel). Finally, HPA-1bb ECs remained unaffected in all experiments in the presence of any of the IgG preparations (data not shown). Taken together, the anti-αvβ3 subtype bound specifically to HPA-1a expressed on ECs, inhibits cellular adhesion to vitronectin, caused cell apoptosis (anoikis), and consequently disturbed angiogenesis.
Detection of the anti-αvβ3 subtype as a potential predictive parameter for ICH
Once anti-HPA-1a of the β3-subtype has been removed from the serum, clinically relevant anti-HPA-1a of the anti-αvβ3 subtype can be demonstrated in an antigen capture assay (Figure 2A, lower panel). We noticed that the use of some monoclonal capture antibodies results in reduced sensitivity (Figure 2A, lower panel, left diagram), most probably caused by competitive inhibition between monoclonal and human antibodies. Adjusting the current assay (right diagram) to 100% specificity (no false-positive detection of anti-αvβ3 subtype in the −ICH cohort), 1/18 anti-HPA-1a of the anti-αvβ3 subtype cannot be identified in the +ICH cohort, resulting in a negative predictive value of 94.7%.
Discussion
The reason how and why ICH occurs in newborns with FNAIT is still unknown. The association between platelet count and ICH is weak, and ICH has been reported in FNAIT cases where the platelet count was within the reference range.6,21 This indicates that biological effects of anti-HPA-1a antibodies other than increased platelet turnover may be responsible for the development of ICH.
In this study, we demonstrate that maternal sera from +ICH cases, but not −ICH cases, contain significant amounts of anti-HPA-1a antibodies which exclusively react with the αvβ3 complex. Binding of this antibody subtype to endothelial cells hinders endothelial adhesion to vitronectin, leads to cell anoikis, and interferes with angiogenesis in a ROS dependent manner. Our results indicate that presence of the anti-αvβ3 subtype in maternal serum is the critical cause for the development of fetal ICH in FNAIT.
It is known that the polymorphic residue Leu33Pro residing on the PSI domain of the β3 integrin subunit controls the formation HPA-1a epitopes9. However, little is known about the contribution of αIIb and αv subunits to the formation of these epitopes. Recent studies demonstrated that some anti-HPA-1a antibodies bound discretely to the β3 chain, and some recognized complex (or compound) epitopes formed by αIIb and β3.22–25 Similar to αIIbβ3, point mutation Leu33Pro together with αv subunit may create HPA-1a compound dependent HPA-1a antigenic determinant. Our structural analysis of the PSI domain of αIIbβ3 and αvβ3 shows distinct conformational states that could in theory be differentially recognized by the immune system (Supplemental figure 2). However, other mechanisms may play a role.
We found in our cohorts a new anti-HPA-1a antibody subtype that reacts with compound epitopes formed by αv and β3 subunits. Accordingly, three different anti-HPA-1a subtypes may exist: anti-αIIbβ3 (reactive with platelets only), anti-β3 (reactive with platelets and endothelial cells), and anti-αvβ3 (predominantly reactive with endothelial cells). The third subtype is produced by FNAIT mothers with fetal/neonatal ICH.
It has become evident that integrin αvβ3-mediated adhesion to extracellular matrix (ECM) is essential for endothelial cell growth and survival, whereas αvβ3 antagonism can induce endothelial apoptosis during angiogenesis.26 In fact, disruption of αvβ3 ligation with moabs or peptide antagonist of αvβ3 caused detachment-induced endothelial anoikis19 via activation of caspase-3 cascade27, and impaired thereby angiogenesis.28–30 In line with these observations, we show in this study that only the anti-αvβ3 subtype of anti-HPA-1a (that impaired αvβ3 ligation) could induce endothelial anoikis and affect angiogenesis.
Recently, Yougbaré and coworkers demonstrated that anti-β3 antibodies induce ICH in pups by impairing angiogenesis in mouse model of FNAIT16. In this model, maternal isoantibodies were developed by transfusion of β3 knockout mice with wild-type platelets, and ICH frequency was increased with subsequent immunizations. In humans, however, severity of FNAIT does usually not increase with subsequent pregnancies17 and a high rate of ICH-reoccurrence is found in previous siblings with ICH7,8. Unfortunately, analysis of different antibody subtypes was not investigated in this murine model. It is conceivable that, similar to humans, anti-αvβ3 impaired angiogenesis in these pups.
Vessel development in the brain depends on cross-talk between endothelial cells and perivascular astrocytes.31 For this process, αv integrins play an important role through regulation of TGF-β activation and expression of TGF-β responsive genes that promote vessel differentiation and stabilization.32–34 A critical period for ICH in FNAIT is before week 28 of gestation.35 Of note, rapid development of premature medullary veins into shower-like numerous vessels associated with extensive angiogenesis has been observed between week 19 and 24 of gestation.36 Therefore, it is feasible that antibodies against αvβ3 interfere with these critical processes to provoke ICH, especially in the brain within this period. To which extent thrombocytopenia further modifies bleeding probability and severity in the fetus requires further research.
In summary, our study provides evidence that human anti-HPA-1a of anti-αvβ3, but not of anti-β3, specificity can affect fetal vessel wall integrity, a mechanism that appears to be responsible for FNAIT-associated ICH. Our findings contribute not only to our understanding of FNAIT pathology in humans, but also opens the way for new diagnostic testing and treatment strategies for immunized women in subsequent pregnancies. The absence of the anti-αvβ3 subtype could indicate a lower risk (or no risk) for ICH and might allow for modification (or cessation) of prophylactic FNAIT treatment. However, larger clinical studies are necessary to evaluate the clinical value of the proposed diagnostic test.
Supplementary Material
Highlights.
Intracranial hemorrhage (ICH) is the most serious complication of severe fetal/neonatal alloimmune thrombocytopenia (FNAIT), caused by maternal alloantibodies against human platelet antigen-1a (HPA-1a).
We show here that a specific anti-HPA-1a antibody subtype reactive with αvβ3 compound epitopes expressed on endothelial cells is present in ICH cases.
This antibody subtype impairs endothelial function, indicating that anti-endothelial rather than anti-platelet activity is responsible for the development of ICH in FNAIT.
Our finding has the potential of changing diagnostic and therapeutic strategies in FNAIT.
Acknowledgments
U.J. Sachs and S. Santoso designed the study; H. Wihadmadyatami, B. Bayat, S. Werth performed the experiments; H. Wihadmadyatami, T. Bakchoul., B. Bayat, N. Al-Fakhri, G. Bein and P.J. Newman analyzed and interpreted the data; V. Kiefel and J. Zhu provided essential material; T. Bakchoul, B. Bayat, U.J. Sachs and S. Santoso interpreted the data and wrote the manuscript. We thank Heike Berghöfer (Giessen, Germany) for excellent technical assistance, and K.T. Preissner (Giessen, Germany) for helpful discussions.
Source of funding
This work was supported by grants from the Else Kröner-Fresenius-Stiftung, Bad Homburg vor der Höhe, Germany (to U.J. Sachs and B. Bayat) and from the Deutsche Forschungsgemeinschaft (DFG), Excellence Cluster Cardiopulmonary System (to S. Santoso). This paper contains parts of the doctoral thesis of H. Wihadmadyatami.
Nonstandard Abbreviations and Acronyms
- EC
Endothelial Cells
- FNAIT
Fetal Neonatal Alloimmune Thrombocytopenia
- HPA
Human Platelet Alloantigen
- ICH
Intracranial Hemorrhage
- ROS
Reactive Oxygen Species
Footnotes
Statement of prior presentation: Some of the data from this manuscript have been orally presented at the 56th Annual Meeting of the American Society of Hematology, San Francisco, CA, USA.
Disclosure
None
References
- 1.Mueller-Eckhardt C, Kiefel V, Grubert A, Kroll H, Weisheit M, Schmidt S, Mueller-Eckhardt D, Santoso S. 348 cases of suspected neonatal alloimmune thrombocytopenia. Lancet. 1989;1:363–366. doi: 10.1016/s0140-6736(89)91733-9. [DOI] [PubMed] [Google Scholar]
- 2.Kjeldsen-Kragh J, et al. A screening and intervention program aimed to reduce mortality and serious morbidity associated with severe neonatal alloimmune thrombocytopenia. Blood. 2007;110:833–839. doi: 10.1182/blood-2006-08-040121. [DOI] [PubMed] [Google Scholar]
- 3.Spencer JA, Burrows RF. Feto-maternal alloimmune thrombocytopenia: a literature review and statistical analysis. Aust N Z J Obstet Gynaecol. 2001;41:45–55. doi: 10.1111/j.1479-828x.2001.tb01293.x. [DOI] [PubMed] [Google Scholar]
- 4.Bussel J. Diagnosis and management of the fetus and neonate with alloimmune thrombocytopenia. J Thromb Haemost. 2009;7(Suppl 1):253–257. doi: 10.1111/j.1538-7836.2009.03380.x. [DOI] [PubMed] [Google Scholar]
- 5.Thude H, Schorner U, Helfricht C, Loth M, Maak B, Barz D. Neonatal alloimmune thrombocytopenia caused by human leucocyte antigen-B27 antibody. Transfus Med. 2006;16:143–149. doi: 10.1111/j.1365-3148.2006.00634.x. [DOI] [PubMed] [Google Scholar]
- 6.Kroll H, Penke G, Santoso S. Functional heterogeneity of alloantibodies against the human platelet antigen (HPA)-1a. Thromb Haemost. 2005;94:1224–1229. doi: 10.1160/TH05-03-0159. [DOI] [PubMed] [Google Scholar]
- 7.Birchall JE, Murphy MF, Kaplan C, Kroll H. European collaborative study of the antenatal management of feto-maternal alloimmune thrombocytopenia. Br J Haematol. 2003;122:275–288. doi: 10.1046/j.1365-2141.2003.04408.x. [DOI] [PubMed] [Google Scholar]
- 8.Radder CM, Brand A, Kanhai HH. Will it ever be possible to balance the risk of intracranial haemorrhage in fetal or neonatal alloimmune thrombocytopenia against the risk of treatment strategies to prevent it? Vox Sang. 2003;84:318–325. doi: 10.1046/j.1423-0410.2003.00302.x. [DOI] [PubMed] [Google Scholar]
- 9.Newman PJ, Derbes RS, Aster RH. The human platelet alloantigens, PlA1 and PlA2, are associated with a leucine33/proline33 amino acid polymorphism in membrane glycoprotein IIIa, and are distinguishable by DNA typing. J Clin Invest. 1989;83:1778–1781. doi: 10.1172/JCI114082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bennett JS, Berger BW, Billings PC. The structure and function of platelet integrins. J Thromb Haemost. 2009;7(Suppl 1):200–205. doi: 10.1111/j.1538-7836.2009.03378.x. [DOI] [PubMed] [Google Scholar]
- 11.Cheresh DA, Smith JW, Cooper HM, Quaranta V. A novel vitronectin receptor integrin is responsible for distinct adhesive properties of carcinoma cells. Cell. 1989;57:59–69. doi: 10.1016/0092-8674(89)90172-4. [DOI] [PubMed] [Google Scholar]
- 12.Leeksma OC, Giltay JC, Zandbergen-Spaargaren J, Modderman PW, van Mourik JA, von dem Borne AE. The platelet alloantigen Zwa or PlA1 is expressed by cultured endothelial cells. Br J Haematol. 1987;66:369–373. doi: 10.1111/j.1365-2141.1987.tb06925.x. [DOI] [PubMed] [Google Scholar]
- 13.Giltay JC, Leeksma OC, Von dem Borne AE, van Mourik JA. Alloantigenic composition of the endothelial vitronectin receptor. Blood. 1988;72:230–233. [PubMed] [Google Scholar]
- 14.Van Gils JM, Stutterheim J, van Duijn TJ, Zwaginga JJ, Porcelijn L, de Haas M, Hordijk PL. HPA-1a alloantibodies reduce endothelial cell spreading and monolayer integrity. Mol Immunol. 2009;46:406–415. doi: 10.1016/j.molimm.2008.10.015. [DOI] [PubMed] [Google Scholar]
- 15.Radder CM, Beekhuizen H, Kanhai HH, Brand A. Effect of maternal anti-HPA-1a antibodies and polyclonal IVIG on the activation status of vascular endothelial cells. Clin Exp Immunol. 2004;137:216–222. doi: 10.1111/j.1365-2249.2004.02496.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yougbaré I, et al. Maternal anti-platelet β3 integrins impair angiogenesis and cause intracranial hemorrhage. J Clin Invest. 2015;125:1545–1556. doi: 10.1172/JCI77820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tiller H, Husebekk A, Skogen B, Kjeldsen-Kragh J, Kjaer M. True risk of fetal/neonatal alloimmune thrombocytopenia in subsequent pregnancies: a prospective observational follow-up study. BJOG. 2016;123:738–744. doi: 10.1111/1471-0528.13343. [DOI] [PubMed] [Google Scholar]
- 18.Frisch SM, Screaton RA. Anoikis mechanism. Curr Opin Cell Biol. 2001;13:555–562. doi: 10.1016/s0955-0674(00)00251-9. [DOI] [PubMed] [Google Scholar]
- 19.Li AE, Ito H, Rovira II, Kim KS, Takeda K, Yu ZY, Ferrans VJ, Finkel T. A role for reactive oxygen species in endothelial anoikis. Circ Res. 1999;85:304–310. doi: 10.1161/01.res.85.4.304. [DOI] [PubMed] [Google Scholar]
- 20.Bernardini A, Brockmeier U, Metzen E, Berchner-Pfannschmidt U, Harde E, Acker-Palmer A, Papkovsky D, Acker H, Fandrey J. Measurement of ROS levels and membrane potential dynamics in the intact carotid body ex vivo. Adv Exp Med Biol. 2015;860:55–59. doi: 10.1007/978-3-319-18440-1_7. [DOI] [PubMed] [Google Scholar]
- 21.Bussel JB, Berkowitch RL, Hung C, Kolb EA, Wissert M, Primiani A, Tsaur FW, Macfarland JG. Intracranial hemorrhage in alloimmune thrombocytopenia: stratified management to prevent recurrence in the subsequent affected fetus. Am J Obstet Gynecol. 2010;203:135.e1–135.e14. doi: 10.1016/j.ajog.2010.03.011. [DOI] [PubMed] [Google Scholar]
- 22.Valentin N, Visentin GP, Newman PJ. Involvement of the cysteine-rich domain of glycoprotein IIIa in the expression of the human platelet alloantigen, PlA1: evidence for heterogeneity in the humoral response. Blood. 1995;85:3028–3033. [PubMed] [Google Scholar]
- 23.Honda S, Honda Y, Bauer B, Ruan C, Kunicki TJ. The impact of three-dimensional structure on the expression of PIA alloantigens on human integrin beta 3. Blood. 1996;86:234–242. [PubMed] [Google Scholar]
- 24.Liu LX, Nardi MA, Casella JF, Karpatkin S. Inhibition of binding of anti-PLA1 antibodies to platelets with monoclonal antibody LK-4. Evidence for multiple PLA1 receptor sites on platelet GPIIIa. Blood. 1996;89:3601–3607. [PubMed] [Google Scholar]
- 25.Allen DL, Abrahamsson S, Murphy MF, Roberts DJ. Human platelet antigen 1a epitopes are dependent on the cation-regulated conformation of integrin α(IIb)β(3) (GPIIb/IIIa) J Immunol Methods. 2012;375:166–175. doi: 10.1016/j.jim.2011.10.008. [DOI] [PubMed] [Google Scholar]
- 26.Erdreich-Epstein A, Tran LB, Cox OT, Huang EY, Laug WE, Shimada H, Millard M. Endothelial apoptosis induced by inhibition of integrins alphavbeta3 and alphavbeta5 involves ceramide metabolic pathways. Blood. 2005;105:4353–4361. doi: 10.1182/blood-2004-08-3098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang WJ. Acurhagin-C, an ECD disintegrin, inhibits integrin alphavbeta3-mediated human endothelial cell functions by inducing apoptosis via caspase-3 activation. Br J Pharmacol. 2010;160:1338–1351. doi: 10.1111/j.1476-5381.2010.00781.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Brooks PC, Clark RA, Cheresh DA. Requirement of vascular integrin alpha v beta 3 for angiogenesis. Science. 1994;264:569–571. doi: 10.1126/science.7512751. [DOI] [PubMed] [Google Scholar]
- 29.Gutheil JC, Campbell TN, Pierce PR, Watkins JD, Huse WD, Bodkin DJ, Cheresh DA. Targeted antiangiogenic therapy for cancer using Vitaxin: a humanized monoclonal antibody to the integrin alphavbeta3. Clin Cancer Res. 2000;6:3056–3061. [PubMed] [Google Scholar]
- 30.Zhang D, Pier T, McNeel DG, Wilding G, Friedl A. Effects of a monoclonal anti-alpha v beta 3 integrin antibody on blood vessels - a pharmacodynamic study. Invest New Drugs. 2007;25:49–55. doi: 10.1007/s10637-006-9013-8. [DOI] [PubMed] [Google Scholar]
- 31.Ballabh P, Braun A, Nedergaard M. The blood-brain barrier: an overview: structure, regulation, and clinical implications. Neurobiol Dis. 2004;16:1–13. doi: 10.1016/j.nbd.2003.12.016. [DOI] [PubMed] [Google Scholar]
- 32.Cambier S, Gline S, Mu D, Collins R, Araya J, Dolganov G, Einheber S, Boudreau N, Nishimura SL. Integrin αvβ8-mediated activation of transforming growth factor-β by perivascular astrocytes: an angiogenic control switch. Am J Pathol. 2005;166:1883–1894. doi: 10.1016/s0002-9440(10)62497-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Scaffidi AK, Petrovic N, Moodley YP, Fogel-Petrovic M, Kroeger KM, Seeber RM, Eidne KA, Thompson PJ, Knight DA. αvβ3 integrin interacts with the transforming growth factor B (TGFB) type II receptor to potentiate the proliferative effects of TGFB1 in living human lung fibroblasts. J Biol Chem. 2004;279:37726–37733. doi: 10.1074/jbc.M403010200. [DOI] [PubMed] [Google Scholar]
- 34.Wipff PJ, Hinz B. Integrins and the activation of latent transforming growth factor beta1 - an intimate relationship. Eur J Cell Biol. 2008;87:601–615. doi: 10.1016/j.ejcb.2008.01.012. [DOI] [PubMed] [Google Scholar]
- 35.Tiller H, Kamphuis MJ, Flodmark O, Papadogiannakis N, David AL, Sainio S, Koskinen S, Javela K, Wikman AT, Kekomaki R, Kanhai HH, Oepkes D, Husebekk A, Westgren M. Fetal intracranial haemorrhages caused by fetal and neonatal alloimmune thrombocytopenia: an observational cohort study of 43 cases from an international multicentre registry. BMJ Open. 2013;3:e002490. doi: 10.1136/bmjopen-2012-002490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pooh RK, Kurjak A. Fetal brain vascularity visualized by conventional 2D and 3D power doppler technology. DSJUOG. 2010;4:249–258. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


