Abstract
Background
Acute myocardial infarction complicated by cardiogenic shock (AMI-CS) is associated with substantial mortality. We evaluated outcomes of patients in The Society of Thoracic Surgeons Adult Cardiac Surgery Database who underwent coronary artery bypass graft surgery (CABG) in the setting of AMI-CS.
Methods
All patients with AMI-CS who underwent nonelective CABG or CABG with ventricular assist device implantation within 7 days after myocardial infarction were enrolled. The primary analysis sample consisted of patients who underwent surgery between June 2011 and December 2013. Baseline characteristics, operative findings, outcomes, and the utilization of mechanical circulatory support (MCS) were assessed in detail in this population. We also evaluated trends in unadjusted mortality for all patients undergoing CABG or CABG with ventricular assist device for AMI-CS from January 2005 to December 2013.
Results
A total of 5,496 patients met study criteria, comprising 1.5% of all patients undergoing CABG during the study period. Overall operative mortality was 18.7%, decreasing from 19.3% in 2005 to 18.1% in 2013 (p < 0.001). Use of MCS increased from 5.8% in 2011 to 8.8% in 2013 (p = 0.008). Patients receiving MCS had a high proportion of cardiovascular risk factors or high clinical acuity. Patients requiring preoperative and patients requiring intraoperative or postoperative MCS had operative mortality of 37.2% and 58.4%, respectively. Patients undergoing CABG as a salvage procedure had an operative mortality of 53.3%, and a high incidence of reoperation (21.8%), postoperative respiratory failure requiring prolonged ventilation (59.7%), and renal failure (18.5%).
Conclusions
Most patients undergoing CABG for AMI-CS have a sizeable but not prohibitive risk. Patients who require MCS and those undergoing operation as a salvage procedure reflect higher risk populations.
Cardiogenic shock is the primary cause of hospital death after myocardial infarction (MI), with an associated mortality of roughly 40% [1]. Revascularization can improve survival after cardiogenic shock from MI [2]. In the Should We Emergently Revascularize Occluded Coronaries for Cardiogenic Shock (SHOCK) trial, patients undergoing coronary artery bypass graft surgery (CABG) had outcomes similar to those of patients undergoing PCI despite having more substantial coronary artery disease and higher rates of diabetes mellitus [3]. Despite the benefit of CABG, this therapy remains underutilized in these critically ill patients [4].
Mehta and colleagues [5] reported the outcomes of patients in The Society of Thoracic Surgeons (STS) Adult Cardiac Surgery Database (ACSD) who underwent CABG in the setting of AMI-CS between 2002 and 2005. The operative mortality ranged from 20% for isolated CABG to 33% for CABG plus valve surgery and 58% for CABG plus ventricular septal repair. Despite high mortality primarily due to pump failure, less than 3% of patients with shock received a left ventricular assist device (LVAD) [5].
Since 2005, there have been several major changes in the management of patients with AMI-CS. These include increased utilization of drug-eluting stents, an increase in the number of hospitals with 24-hour catheterization laboratory availability, new anticoagulant and antiplatelet agents with improved safety profiles, and improvements in extracorporeal membrane oxygenation (ECMO) and percutaneous and surgical ventricular assist device (VAD) technology. The utilization of percutaneous MCS devices has increased rapidly, with a 1,511% increase from 2007 to 2011 [6]. Outcomes after VAD implantation for post-MI cardiogenic shock and postcardiotomy shock have also improved [7].
The purpose of this study was to evaluate clinical characteristics and current outcomes of patients with AMI-CS undergoing CABG, with a specific emphasis on the utilization of MCS and associated outcomes.
Material and Methods
Data Source
The STS ACSD is a multicenter registry for adult cardiac surgery. It contains more than 5 million cardiac surgical records and captures 95% of all cardiac surgical procedures performed in the United States. The formation, rationale, and methodology of the STS ACSD have been described [8, 9].
Patients
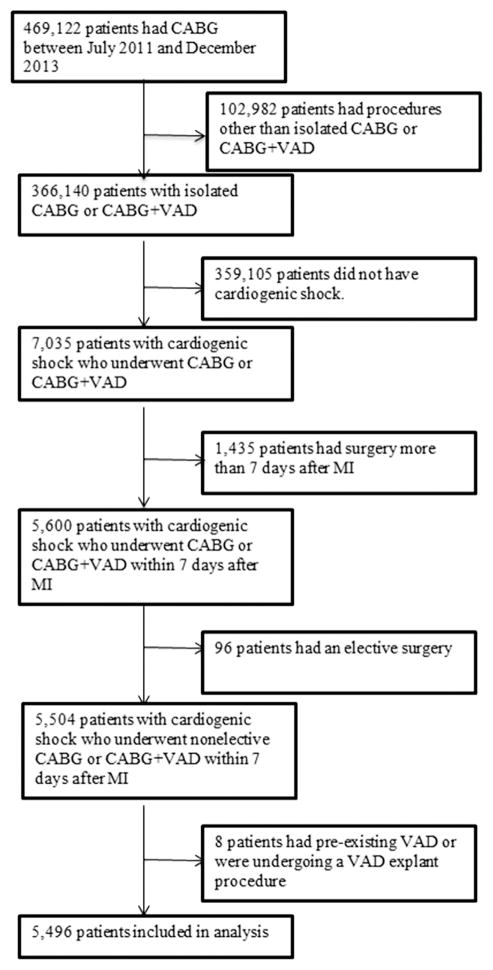
The study population included patients in the STS ACSD who had a recent MI (within 7 days before the procedure) and cardiogenic shock and underwent nonelective CABG or CABG with VAD implantation. The primary analysis sample consisted of patients who underwent surgery between July 2011 and December 2013. The 2011 start date was chosen to coincide with version 2.73 of the STS data collection form, which first started collecting detailed data on catheter-based MCS and ECMO. We also evaluated patients who had surgery for the same indications between 2005 and 2013 solely to determine trends in unadjusted mortality. Exclusion criteria included patients with concomitant surgeries, preexisting LVAD, and post-MI complications such as mitral regurgitation, free wall rupture, or ventricular septal rupture (Fig 1).
Fig 1.
Patient selection. (CABG = coronary artery bypass graft surgery; MI = myocardial infarction; VAD = ventricular assist device.)
Data Definitions
Cardiogenic shock is defined as the patient being, at the time of procedure, in a sustained (more than 30 minutes) clinical state of hypoperfusion due to cardiac failure according to the following criteria: persistent hypotension (systolic blood pressure less than 80 mm Hg or mean arterial pressure 30 mm Hg lower than baseline) with a severe reduction in cardiac index (less than 1.8 without mechanical or inotropic support or less than 2 with mechanical or inotropic support), in the setting of adequate or elevated filling pressures. Urgent operative status is defined as the procedure required during same hospitalization to minimize the chance of further clinical deterioration. Patients requiring emergent operations are defined as those having ongoing, refractory, unrelenting cardiac compromise, with or without hemodynamic instability, and not responsive to any form of therapy except cardiac surgery. Emergent salvage patients are defined as those undergoing cardiopulmonary resuscitation en route to the operating room or before anesthesia induction or having ongoing ECMO to maintain life. Operative mortality is defined as all deaths occurring during the hospitalization in which the operation was performed, or deaths occurring after discharge from the hospital but within 30 days of the procedure. Myocardial infarction is defined as per the American College of Cardiology Foundation/American Heart Association data definitions [10]. Other data definitions are provided on the STS website [11].
Analysis
Baseline characteristics, operative variables, and postoperative outcomes of the overall cohort were analyzed using standard summary statistics. Categoric variables were calculated as frequency and percentage, and continuous variables were calculated as median and interquartile range. Linear trend tests were applied to evaluate changes in mortality and MCS use over time.
Patients were categorized by MCS status into three groups: no MCS use, preoperative MCS use, and intraoperative/postoperative MCS use only. Patients who had both preoperative and postoperative MCS were grouped within the preoperative MCS group. Summary statistics were used to determine differences in baseline characteristics and outcomes associated with MCS. Finally, select patient characteristics were compared among groups using Pearson’s χ2 test for categoric variables, and the Kruskal-Wallis test for continuous variables. All analyses were performed with SAS version 9.2 (SAS Institute, Cary, NC). A p value of 0.05 was used to determine statistical significance.
Results
Baseline Characteristics
A total of 5,496 patients underwent nonelective CABG or CABG with VAD in the setting of recent AMI-CS from July 2011 to December 2013. Baseline characteristics are listed in Table 1. The majority of patients (71.7%) were male, the median age was 66 years (interquartile range: 58 to 74), and nearly one third (32.6%) were current smokers. These patients comprised approximately 1.5% of all patients undergoing CABG during the study period.
Table 1.
Baseline Characteristics and Risk Factors
| Characteristics | All Patients (n = 5,496) | Preoperative MCS (n = 129) | Intra/Postop MCS (n = 279) | No MCS (n = 5,088) | p Valuea |
|---|---|---|---|---|---|
| Age, years, median (IQR) | 66.0 (58.0–74.0) | 64.0 (57.0–71.0) | 62.0 (54.0–70.0) | 66.0 (58.0–74.0) | <0.0001 |
| Sex | |||||
| Male | 3,939 (71.7) | 87 (67.4) | 205 (73.5) | 3,647 (71.7) | 0.4518 |
| Female | 1,553 (28.3) | 42 (32.6) | 74 (26.5) | 1,437 (28.2) | |
| Current smoker | 1,793 (32.6) | 43 (33.3) | 97 (34.8) | 1,653 (32.5) | 0.7368 |
| Diabetes mellitus | 2,252 (41.0) | 59 (45.7) | 92 (33.0) | 2,101 (41.3) | 0.0121 |
| Dyslipidemia | 4,013 (73.0) | 95 (73.6) | 179 (64.2) | 3,739 (73.5) | 0.0027 |
| Renal failure on dialysis | 196 (3.6) | 3 (2.3) | 5 (1.8) | 188 (3.7) | 0.1826 |
| Lung disease | |||||
| Mild | 571 (10.4) | 7 (5.4) | 17 (6.1) | 547 (10.8) | 0.0009 |
| Moderate | 344 (6.3) | 7 (5.4) | 7 (2.5) | 330 (6.5) | |
| Severe | 299 (5.4) | 7 (5.4) | 9 (3.2) | 283 (5.6) | |
| Liver disease | 181 (3.3) | 3 (2.3) | 7 (2.5) | 171 (3.4) | 0.6299 |
| Peripheral arterial disease | 909 (16.5) | 26 (20.2) | 33 (11.8) | 850 (16.7) | 0.0542 |
| Prior CVA | 452 (8.2) | 13 (10.1) | 17 (6.1) | 422 (8.3) | 0.3157 |
| Previous cardiac intervention | 2,120 (38.6) | 68 (52.7) | 120 (43.0) | 1,932 (38.0) | 0.0009 |
| Previous CABG | 100 (1.8) | 5 (3.9) | 9 (3.2) | 86 (1.7) | 0.0368 |
| Previous PCI | 1,958 (35.6) | 59 (45.7) | 106 (38.0) | 1,793 (35.2) | 0.0347 |
From statistical tests of the null hypothesis that the distribution of the factor was the same across three groups.
Values are n (%) unless otherwise indicated.
CABG = coronary artery bypass graft surgery; CVA = cerebrovascular accident; Intra/Postop = intraoperative/postoperative; IQR = interquartile range; MCS = mechanical circulatory support; PCI = percutaneous coronary intervention.
Cardiac History
Myocardial infarction occurred within 6 hours of CABG in 31.9%, between 6 and 24 hours in 23.8%, and between 1 and 7 days in 44.4% of patients. Catheterization revealed three-vessel disease in 78.2%, two-vessel disease in 17.4%, and single-vessel disease in 4.1%. Significant left main disease (greater than 50% stenosis) was present in 51.2% and significant proximal left anterior descending artery (more than 70% stenosis) was present in 63.9%. Thirty-eight percent had previous cardiac interventions, with 35.6% having had a PCI and 1.8% having previous CABG. Among those who had PCI, 68.9% had PCI during the episode of care leading to CABG.
Severity of Illness
The STS 2008 cardiac surgery risk models predicted a mortality of 20% or greater for 30.5% of patients, mortality of 12% to 20% for 20.1%, mortality of 8% to 12% for 15.6%, and mortality between 4% and 8% for 20.7%. In the overall cohort, 38% had preoperative arrhythmias, of which 66.1% were ventricular tachycardia or fibrillation. Twenty-two percent had resuscitation for cardiac arrest. Median preoperative ejection fraction was 35%. End-organ function was preserved, with median creatinine 1.1 mg/dL and bilirubin 0.6 mg/dL.
Age, preoperative LVEF, and end-organ function including creatinine and bilirubin were clinically similar among patients with urgent (n = 1,734, 31.6%), emergent (n = 3,339, 60.8%), and salvage (n = 422, 7.7%) operative status (Table 2). Patients with salvage status had the highest incidence of preoperative ventricular arrhythmias, left main disease, and resuscitation, and the shortest duration between MI and CABG. Patients with urgent and emergent status had similar age, incidence of resuscitation LVEF, creatinine, and bilirubin. The incidence of left main disease was higher and the incidence of three-vessel disease and proximal left anterior descending artery disease was lower in the emergent group compared with the urgent group. Most patients in the urgent group (82.9%) had surgery 1 to 7 days after MI, whereas most patients in the emergent group (72.2%) had surgery within 24 hours of MI.
Table 2.
Severity of Illness
| Variables | Urgent (n = 1,734) | Emergent (n = 3,339) | Salvage (n = 422) | p Value |
|---|---|---|---|---|
| Age, years | 66.0 (58.0–74.0) | 65.0 (58.0–74.0) | 65.0 (56.0–73.0) | 0.0578 |
| Sex | ||||
| Male | 1,294 (74.6) | 2,344 (70.2) | 301 (71.3) | 0.0036 |
| Female | 438 (25.3) | 993 (29.7) | 121 (28.7) | |
| Timing of MI | ||||
| 1–7 days | 1,437 (82.9) | 929 (27.8) | 73 (17.3) | <0.0001 |
| 6–24 hours | 235 (13.6) | 995 (29.8) | 76 (18.0) | |
| ≤6 hours | 62 (3.6) | 1,415 (42.4) | 273 (64.7) | |
| Left main disease ≥50% | 826 (47.6) | 1,761 (52.7) | 228 (54.0) | 0.0011 |
| Diseased vessels | ||||
| None | 2 (0.1) | 5 (0.1) | 3 (0.7) | <0.0001 |
| One | 31 (1.8) | 168 (5.0) | 28 (6.6) | |
| Two | 218 (12.6) | 646 (19.3) | 91 (21.6) | |
| Three | 1,482 (85.5) | 2,519 (75.4) | 299 (70.9) | |
| Resuscitation | 280 (16.1) | 550 (16.5) | 413 (97.9) | <0.0001 |
| Arrhythmia typea | 344 (59.4) | 729 (64.7) | 240 (85.7) | <0.0001 |
| LVEF | 35.0 (25.0–47.0) | 35.0 (25.0–48.0) | 35.0 (20.0–50.0) | 0.1920 |
| INR | 1.1 (1.0–1.2) | 1.1 (1.0–1.2) | 1.1 (1.0–1.3) | 0.0092 |
| Total bilirubin | 0.7 (0.5–0.9) | 0.6 (0.4–0.9) | 0.6 (0.4–0.8) | <0.0001 |
| Creatinine, mg/dL | 1.0 (0.8–1.3) | 1.1 (0.9–1.4) | 1.1 (0.9–1.5) | <0.0001 |
| Predicted risk of mortality | ||||
| <4% | 564 (32.5) | 146 (4.4) | 0 (0.0) | <0.0001 |
| ≥4% and <8% | 460 (26.5) | 656 (19.6) | 9 (2.1) | |
| ≥8% and <12% | 239 (13.8) | 589 (17.6) | 28 (6.6) | |
| ≥12% and <16% | 140 (8.1) | 410 (12.3) | 49 (11.6) | |
| ≥16% and <20% | 112 (6.5) | 350 (10.5) | 43 (10.2) | |
| ≥20% | 208 (12.0) | 1,165 (34.9) | 289 (68.5) | |
Ventricular tachycardia/ventricular fibrillation (among patients with arrhythmia).
Values are median (interquartile range) or n (%).
INR = international normalized ratio; LVEF = left ventricular ejection fraction; MI = myocardial infarction.
Operative Characteristics
Operative characteristics are listed in Table 3. The majority of patients (88.7%) had cardiopulmonary bypass utilization, with a median bypass time of 100 minutes. Patients had a median of one arterial and two venous anastomoses. The left internal mammary artery was used in 75.4%.
Table 3.
Operative Characteristics
| Characteristics | All Patients (n = 5,496) | Preoperative MCS (n = 129) | Intra/Postop MCS (n = 279) | No MCS (n = 5,088) | p Valuea |
|---|---|---|---|---|---|
| Status | <0.0001 | ||||
| Urgent | 1,734 (31.6) | 22 (17.1) | 45 (16.1) | 1,667 (32.8) | |
| Emergent | 3,339 (60.8) | 83 (64.3) | 167 (59.9) | 3,089 (60.7) | |
| Salvage | 422 (7.7) | 24 (18.6) | 67 (24.0) | 331 (6.5) | |
| Missing | 1 (0.0) | 0 (0.0) | 0 (0.0) | 1 (0.0) | |
| Operative time, minutes | 303.0 (251.0–364.0) | 325.0 (258.0–379.0) | 356.0 (297.0–436.0) | 300.0 (250.0–360.0) | <0.0001 |
| CPB utilization | |||||
| None | 620 (11.3) | 16 (12.4) | 10 (3.6) | 594 (11.7) | 0.0015 |
| Combination | 123 (2.2) | 3 (2.3) | 6 (2.2) | 114 (2.2) | |
| Full | 4,753 (86.5) | 110 (85.3) | 263 (94.3) | 4,380 (86.1) | |
| CBP time, minutes | 100.0 (76.0–130.0) | 112.5 (84.0–146.5) | 136.0 (94.0–197.0) | 98.0 (76.0–128.0) | <0.0001 |
| Intraoperative blood product use | 3,582 (65.2) | 107 (82.9) | 240 (86.0) | 3,235 (63.6) | <0.0001 |
| Total ICU time, hours | 108.0 (59.0–212.1) | 136.0 (79.0–290.0) | 127.5 (47.5–330.0) | 106.0 (58.6–208.0) | 0.0088 |
| Reoperation for any reason | 759 (13.8) | 43 (33.3) | 119 (42.7) | 597 (11.7) | <0.0001 |
| Reoperation for bleeding | 270 (4.9) | 19 (14.7) | 59 (21.1) | 192 (3.8) | <0.0001 |
From statistical tests of the null hypothesis that the distribution of the factor was the same across the three groups.
Values are n (%) or median (interquartile range).
CBP = cardiopulmonary bypass; ICU = intensive care unit; Intra/Postop = intraoperative/postoperative; MCS = mechanical circulatory support.
Outcomes
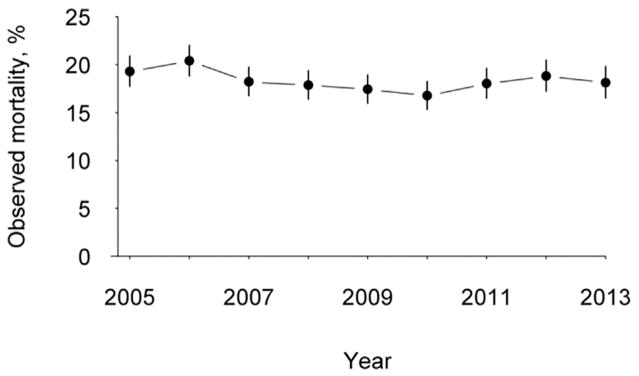
Operative mortality was 18.1%. The primary cause of death was cardiac in 75.5% of patients. The median intensive care unit time was 108 hours and the most common complications were prolonged ventilator requirement, pneumonia, renal failure, sepsis, and stroke (Table 4). The observed to expected mortality ratios for the entire cohort ranged from 1.07 to 1.09 between 2011 and 2013. Utilizing the entire cohort of patients from 2005 to 2013, there was a significant decrease in operative mortality from 19.3% in 2005 to 18.1% in 2013 (p < 0.001; Fig 2).
Table 4.
Postoperative Complications and Outcomes
| Complications and Outcomes | All Patients (n = 5,496) | Preoperative MCS (n = 129) | Intra/Postop MCS (n = 279) | No MCS (n = 5,088) |
|---|---|---|---|---|
| Stroke | 192 (3.5) | 7 (5.4) | 22 (7.9) | 163 (3.2) |
| Prolonged ventilator need | 3,017 (54.9) | 90 (69.8) | 203 (72.8) | 2,724 (53.5) |
| Pneumonia | 702 (12.8) | 26 (20.2) | 48 (17.2) | 628 (12.3) |
| Renal failure requiring dialysis | 443 (8.1) | 21 (16.3) | 68 (24.4) | 354 (7.0) |
| Multisystem organ failure | 385 (7.0) | 19 (14.7) | 64 (22.9) | 302 (5.9) |
| Cardiac arrest | 622 (11.3) | 25 (19.4) | 68 (24.4) | 529 (10.4) |
| Postoperative EF | 38.0 (28.0–50.0) | 32.0 (23.0–43.0) | 26.0 (18.0–40.0) | 40.0 (30.0–50.0) |
| Operative mortality | 1,026 (18.7) | 48 (37.2) | 163 (58.4) | 815 (16.0) |
| Primary cause of mortality | ||||
| Cardiac | 775 (75.5) | 41 (85.4) | 132 (81.0) | 602 (73.9) |
| Neurologic | 61 (5.9) | 4 (8.3) | 12 (7.4) | 45 (5.5) |
| Renal | 14 (1.4) | 0 (0.0) | 0 (0.0) | 14 (1.7) |
| Vascular | 10 (1.0) | 0 (0.0) | 1 (0.6) | 9 (1.1) |
| Infection | 16 (1.6) | 1 (2.1) | 1 (0.6) | 14 (1.7) |
| Pulmonary | 64 (6.2) | 1 (2.1) | 3 (1.8) | 60 (7.4) |
Values are n (%) or median (interquartile range).
EF = ejection fraction; Intra/Postop = intraoperative/postoperative; MCS = mechanical circulatory support.
Fig 2.
Temporal trends in operative mortality for isolated coronary artery bypass graft surgery, without or without ventricular assist device.
Patients who had urgent, emergent, and salvage operations had operative mortality of 10.3%, 18.6%, and 53.3%, respectively. Patients undergoing salvage operations had a high rate of postoperative complications, including prolonged ventilation in 59.7%, reoperations in 21.8%, renal failure in 18.5%, multisystem organ failure in 14.7%, and lower postoperative compared with preoperative LVEF (35% versus 33%). The primary cause of mortality was cardiac in 82% of salvage cases, compared with 74.8% of emergent and 69.1% of urgent cases.
Mechanical Circulatory Support
Eighty-two percent of patients had a perioperative intraaortic balloon pump (IABP). The use of MCS with a higher degree of support than the IABP is evaluated in detail in the following section; for the remainder of this article, MCS refers to circulatory support devices other than IABP. Of the 5,496 patients in the cohort, 129 (2.3%) had preoperative MCS with or without continued intraoperative/postoperative MCS, 279 (5.1%) had intraoperative or postoperative MCS but no preoperative MCS, and 5,088 (92.6%) did not have MCS (Fig 3). The operative mortality for patients without MCS, preoperative MCS, and intraoperative/postoperative MCS was 16%, 37.2%, and 58.4%, respectively. The STS mortality algorithm predicted greater than 20% mortality in 48.8% of patients in the preoperative group, 33.7% of the intraoperative/postoperative MCS group, and 29.8% of the no-MCS group.
Fig 3.
Patient subgroups by mechanical circulatory support (MCS) status.
Patients with MCS tended to be younger than patients without MCS. Patients with preoperative MCS had the highest prevalence of hyperlipidemia, cerebrovascular disease, peripheral arterial disease, prior PCI, and prior CABG. The preoperative MCS group had the lowest baseline LVEF (Tables 3 and 5). The intraoperative/postoperative MCS group had the highest measures of preoperative clinical acuity. The intraoperative/postoperative MCS group had a higher percentage (42.3%) of patients with MI 6 hours or less before surgery compared with the preoperative MCS group (36.4%) and no-MCS group (31.2%). The intraoperative/postoperative group had the highest incidence of cardiopulmonary resuscitation within 1 hour of the operative procedure (38.7%), as well as the highest proportion of salvage procedures (24%) among the three groups. The intraoperative/postoperative group also had the highest incidence of postoperative complications, including reoperations, stroke, respiratory failure, renal failure, dialysis, multisystem organ failure, and cardiac arrest (Table 4).
Table 5.
Myocardial Infarction Characteristics by Mechanical Circulatory Support Status
| Characteristics | Preoperative MCS (n = 129) | Intra/Postop MCS (n = 279) | No MCS (n = 5,088) | p Valuea |
|---|---|---|---|---|
| Timing of MI | 0.0001 | |||
| ≤6 hours | 47 (36.4) | 118 (42.3) | 1,586 (31.2) | |
| 6–24 hours | 37 (28.7) | 67 (24.0) | 1,202 (23.6) | |
| 1–7 days | 45 (34.9) | 94 (33.7) | 2,300 (45.2) | |
| Resuscitation | 43 (33.3) | 108 (38.7) | 1,092 (21.5) | <0.0001 |
| Type of arrhythmia | 0.0027 | |||
| Afib/flutter | 14 (10.9) | 15 (5.4) | 455 (8.9%) | |
| Heart block | 4 (3.1) | 7 (2.5) | 102 (2.0%) | |
| Sustained VT/VF | 36 (27.9) | 90 (32.3) | 943 (18.5%) | |
| Multiple types | 2 (1.6) | 15 (5.4) | 241 (4.7%) | |
| LV ejection fraction | 25.0 (20.0–40.0) | 28.0 (20.0–43.0) | 35.0 (25.0–48.5) | <0.0001 |
| No. of diseased vessels | ||||
| None | 0 (0.0%) | 2 (0.7%) | 8 (0.2%) | 0.0001 |
| One | 12 (9.3%) | 12 (4.3%) | 203 (4.0%) | |
| Two | 24 (18.6%) | 70 (25.1%) | 862 (16.9%) | |
| Three | 93 (72.1%) | 195 (69.9%) | 4,012 (78.9%) | |
| Left main ≥50% stenosis | 70 (54.3%) | 131 (47.0%) | 2,615 (51.4%) | 0.2713 |
| Proximal LAD ≥70% stenosis | 84 (65.1%) | 175 (62.7%) | 3,251 (63.9%) | 0.8590 |
From statistical tests of the null hypothesis that the distribution of the factor was the same across the three groups.
Values are n (%) or median (interquartile range).
Afib = atrial fibrillation; LAD = left anterior descending artery; LV = left ventricle; MCS = mechanical circulatory support; MI = myocardial infarction; VT/VF = ventricular tachycardia/ventricular fibrillation.
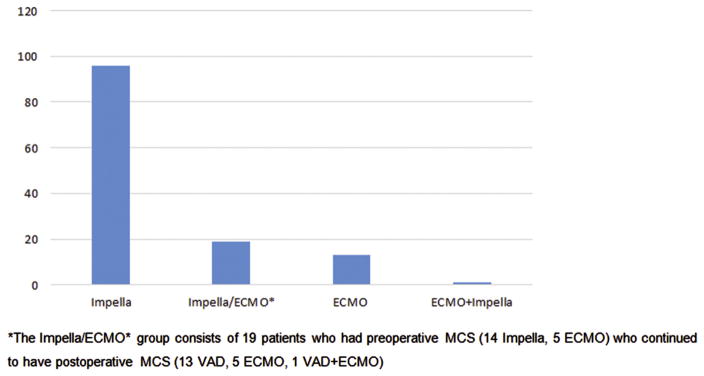
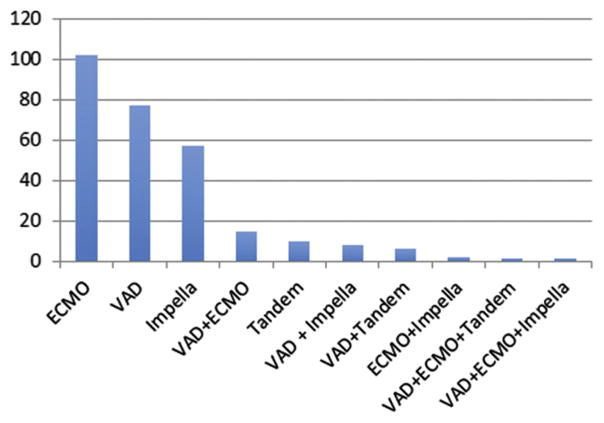
The most common preoperative MCS device was Impella (Abiomed, Danvers, MA), and the intraoperative/postoperative MCS device was most commonly ECMO (Figs 4 and 5). From July 2011 to December 2013, there was a significant increase in the use of MCS perioperatively from 5.8% to 8.8% (p = 0.008) and an increasing trend in the use of preoperative MCS from 1.3% to 2.9% (p = 0.05). The LVEF improved after operation in the preoperative MCS and no MCS groups, but declined in the group that required intraoperative/postoperative MCS.
Fig 4.
Types of preoperative mechanical circulatory support (MCS). *The Impella/extracorporeal membrane oxygenation (ECMO) group consists of 19 patients who had preoperative MCS (14 Impella, 5 ECMO) who continued to have postoperative MCS (13 ventricular assist device, 5 ECMO, and 1 ventricular assist device plus ECMO).
Fig 5.
Types of intraoperative/postoperative mechanical circulatory support: extracorporeal membrane oxygenation (ECMO), ventricular assist device (VAD), Impella, VAD plus ECMO, Tandem, VAD plus Impella, VAD plus Tandem, ECMO plus Impella, VAD plus ECMO plus Tandem, and VAD plus ECMO plus Impella. (Tandem = Tandemheart percutaneous ventricular assist device; CardiacAssist, Pittsburgh, PA.)
Comment
Cardiogenic shock is the leading cause of death after MI. Early revascularization improves survival, and CABG is an effective means of revascularization in shock, particularly for diffuse and multivessel disease, lesions not amenable to PCI, failed PCI, and for mechanical complications after MI [2]. In the IABP-SHOCK II trial, where 52% had three-vessel disease and the left main was the infarct-related artery in approximately 9% of patients, only 3.5% underwent immediate or staged CABG [1]. These findings may be related to limited referrals for emergency CABG, lack of access or capability for emergency CABG in smaller hospitals, and reluctance to operate on very high risk patients.
This analysis of a large nationwide registry shows that outcomes of patients with MI and CS undergoing CABG has improved over time, and the operative mortality of 18.1%, while substantial, is not prohibitive. For the overall cohort, current risk-prediction algorithms capture the clinical acuity, which may alleviate some concerns that operating on these high-risk patients would negatively impact surgeon and center outcome data. There are subgroups, including patients requiring MCS and patients with salvage operations who have significantly worse outcomes, and careful assessment of risk and benefit is required in these cases.
In this study, measures of clinical acuity were highly associated with mortality, with mortality ranging from 10% to 53%. Any degree of cardiogenic shock remained an important risk factor, and even patients with milder degrees of cardiogenic shock undergoing bypass with “urgent” operative status had an operative mortality of more than 10%, significantly higher than that observed in patients with MI without shock who undergo CABG [12]. Similarly, in a report of patients undergoing PCI from the national cardiovascular database registry, clinical acuity was a strong predictor of survival, and the inhospital mortality among patients with transient shock, sustained shock or salvage status, and sustained shock plus salvage status was 15.26%, 33.45%, and 63.99%, respectively [13]. Other studies have also shown that hemodynamic and clinical variables such as clinical acuity, duration of end-organ ischemia, evidence of end-organ hypoperfusion, and hypotension predict response to revascularization and survival in AMI-CS [14].
Mechanical circulatory support can improve these hemodynamic variables, reverse shock, and normalize end-organ perfusion, and various devices have been used over the years to treat AMI-CS. Many observational studies have shown improved survival for AMICS with percutaneous and surgical LVADs compared with historical controls [15]. There is also evidence that early ventricular unloading by MCS before reperfusion may improve outcomes [16, 17]. However, there have been no randomized trials that have demonstrated survival benefit, leading to a IIB guideline recommendation for MCS for AMI-CS [18]. Therefore, even though it is recognized that it is important to reverse shock before multisystem organ failure occurs, there are few data that provide guidance on timing of MCS and particular device selection, and MCS selection remains discretionary with wide variation in use among physicians and institutions [19]. Despite these uncertainties, there has been significant overall increase in utilization of temporary MCS as well as decreasing mortality and hospital costs for patients undergoing short-term MCS [6].
This study demonstrates that patients undergoing CABG with AMI-CS have a high utilization of IABP, but only a small proportion receives other MCS devices. Patients who require MCS have worse outcomes. There was a 2.3-fold difference in operative mortality between the preoperative MCS group and no-MCS group, and a 3.5-fold difference between the intraoperative/postoperative MCS group and no-MCS group. Patients receiving preoperative MCS had the highest prevalence of cardiovascular risk factors, whereas patients receiving intraoperative/postoperative MCS had the highest measures of clinical acuity, including salvage operative status, resuscitation, and ventricular arrhythmias. Given missing relevant factors such as individual hemodynamic profiles at the time of MCS implantation, institutional differences in patient selection for MCS and management of shock, and the degree of hemodynamic support provided by different MCS devices, there are insufficient data to comment on how earlier or preoperative MCS implantation, or any particular MCS device, would have influenced survival. It is possible, perhaps likely, that for patients who required intraoperative/postoperative MCS, the devices were utilized in a situation that would have resulted in patient death without mechanical support. The inability to wean from cardiopulmonary bypass, for example, is 100% fatal without MCS. Therefore, survival of 41.6% would seem to justify MCS use in this patient population.
The primary cause of mortality was cardiac in all groups, but more specific reasons, such as persistent or worsening hemodynamic instability on MCS, irreversible end-organ dysfunction, or inadequate recovery of ventricular function and ineligibility for transition to a durable device or transplant were not available. Others have shown that only approximately a third of patients who require temporary or permanent MCS for AMI-CS will have myocardial recovery despite revascularization [20]. The intraoperative/postoperative MCS group with the highest mortality was also the group in which LVEF did not improve after revascularization. Similarly, patients who underwent operation as salvage did not in general have improvement in their LVEF postoperatively, and had a high mortality with predominantly cardiac cause of death despite revascularization. Furthermore, patients who ultimately required intraoperative/postoperative MCS had a high proportion who went to the operating room as salvage status. Given the poor outcomes in these subgroups, early assessment of myocardial viability, likelihood of adequate revascularization, stabilization and assessment of end organ and neurologic function with temporary MCS and ECMO, and expedited evaluation for alternative strategies such as early planned durable LVAD (with or without CABG) may be considered.
Data collection for MCS utilization (other than IABP use) began after the most recent iteration of the STS mortality risk models were published in 2008, and these models therefore do not include MCS utilization as a risk factor for mortality. Current models accurately assess operative risks in patients that do not require MCS, but underestimate risks in patients that have MCS, particularly those who have intraoperative/postoperative MCS. Given the increased clinical acuity reflected by patients who have MCS, MCS utilization should be evaluated for future mortality prediction models.
This study had several limitations. First, patients were accepted for CABG as a matter of routine clinical practice rather than in a randomized trial setting, and they may represent a lower risk cohort among all patients who were initially referred for CABG. Second, the duration of MI before revascularization was listed, but the duration of shock before revascularization or MCS was not available. Third, given the lack of standardized criteria for MCS use, a patient with similar clinical acuity may or may not have received MCS depending on the admitting institution. Finally, the use of MCS may not reflect clinical factors alone. Larger tertiary medical centers are more likely to have availability of MCS and use it compared with smaller hospitals, or there may be an institutional history and preference for a particular device. The same factors that affect MCS utilization may also influence revascularization or adjunctive strategies, and that was not measured. Because of these limitations, the low number of events, and a heterogeneous MCS device profile, we were not able to perform a robust multivariable analysis to assess the independent effect of MCS on survival.
In conclusion, most patients undergoing CABG for AMI-CS have a sizeable but not prohibitive risk, which has slightly decreased since 2005. Patients who require MCS reflect a higher risk population. Randomized studies to assess the optimal mode of revascularization, and the optimal timing and outcomes of MCS for patients failing standard management are necessary.
Abbreviations and Acronyms
- ACSD
Adult Cardiac Surgery Database
- AMI-CS
acute myocardial infarction complicated by cardiogenic shock
- CABG
coronary artery bypass graft surgery
- ECMO
extracorporeal membrane oxygenation
- IABP
intraaortic balloon pump
- LVAD
left ventricular assist device
- LVEF
left ventricular ejection fraction
- MCS
mechanical circulatory support
- MI
myocardial infarction
- PCI
percutaneous coronary intervention
- STS
The Society of Thoracic Surgeons
- VAD
ventricular assist device
References
- 1.Thiele H, Zeymer U, Neumann FJ, et al. Intraaortic balloon support for myocardial infarction with cardiogenic shock. N Engl J Med. 2012;367:1287–96. doi: 10.1056/NEJMoa1208410. [DOI] [PubMed] [Google Scholar]
- 2.Hochman JS, Sleeper LA, Webb JG, et al. Early revascularization in acute myocardial infarction complicated by cardiogenic shock. SHOCK Investigators. Should we emergently revascularize occluded coronaries for cardiogenic shock. N Engl J Med. 1999;341:625–34. doi: 10.1056/NEJM199908263410901. [DOI] [PubMed] [Google Scholar]
- 3.White HD, Assmann SF, Sanborn TA, et al. Comparison of percutaneous coronary intervention and coronary artery bypass grafting after acute myocardial infarction complicated by cardiogenic shock: results from the Should We Emergently Revascularize Occluded Coronaries for Cardiogenic Shock (SHOCK) trial. Circulation. 2005;112:1992–2001. doi: 10.1161/CIRCULATIONAHA.105.540948. [DOI] [PubMed] [Google Scholar]
- 4.Babaev A, Frederick PD, Pasta DJ, Every N, Sichrovsky T, Hochman JS. Trends in management and outcomes of patients with acute myocardial infarction complicated by cardiogenic shock. JAMA. 2005;294:448–54. doi: 10.1001/jama.294.4.448. [DOI] [PubMed] [Google Scholar]
- 5.Mehta RH, Grab JD, O’Brien SM, et al. Clinical characteristics and in-hospital outcomes of patients with cardiogenic shock undergoing coronary artery bypass surgery: insights from The Society of Thoracic Surgeons national cardiac database. Circulation. 2008;117:876–85. doi: 10.1161/CIRCULATIONAHA.107.728147. [DOI] [PubMed] [Google Scholar]
- 6.Stretch R, Sauer CM, Yuh DD, Bonde P. National trends in the utilization of short-term mechanical circulatory support: incidence, outcomes, and cost analysis. J Am Coll Cardiol. 2014;64:1407–15. doi: 10.1016/j.jacc.2014.07.958. [DOI] [PubMed] [Google Scholar]
- 7.Hernandez AF, Grab JD, Gammie JS, et al. A decade of short-term outcomes in post-cardiac surgery ventricular assist device implantation: data from The Society of Thoracic Surgeons’ national cardiac database. Circulation. 2007;116:606–12. doi: 10.1161/CIRCULATIONAHA.106.666289. [DOI] [PubMed] [Google Scholar]
- 8.Shroyer AL, Coombs LP, Peterson ED, et al. The Society of Thoracic Surgeons: 30-day operative mortality and morbidity risk models. Ann Thorac Surg. 2003;75:1856–64. doi: 10.1016/s0003-4975(03)00179-6. [DOI] [PubMed] [Google Scholar]
- 9.Shahian DM, O’Brien SM, Filardo G, et al. The Society of Thoracic Surgeons 2008 cardiac surgery risk models: part 1—coronary artery bypass grafting surgery. Ann Thorac Surg. 2009;88(Suppl):2–22. doi: 10.1016/j.athoracsur.2009.05.053. [DOI] [PubMed] [Google Scholar]
- 10.Thygesen K, Alpert JS, Jaffe AS, et al. Third universal definition of myocardial infarction. Circulation. 2012;126:2020–35. doi: 10.1161/CIR.0b013e31826e1058. [DOI] [PubMed] [Google Scholar]
- 11.The Society of Thoracic Surgeons. Adult cardiac surgery database. Available at: http://www.sts.org/sts-national-database/database-managers/adult-cardiac-surgery-database/data-collection.
- 12.Khaladj N, Bobylev D, Peterss S, et al. Immediate surgical coronary revascularization in patients presenting with acute myocardial infarction. J Cardiothorac Surg. 2013;8:167. doi: 10.1186/1749-8090-8-167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Brennan JM, Curtis JP, Dai D, et al. Enhanced mortality risk prediction with a focus on high-risk percutaneous coronary intervention: results from 1,208,137 procedures in the NCDR. J Am Coll Cardiol Intv. 2013;6:790–9. doi: 10.1016/j.jcin.2013.03.020. [DOI] [PubMed] [Google Scholar]
- 14.Demondion P, Fournel L, Golmard JL, et al. Predictors of 30-day mortality and outcome in cases of myocardial infarction with cardiogenic shock treated by extracorporeal life support. Eur J Cardiothorac Surg. 2014;45:47–54. doi: 10.1093/ejcts/ezt207. [DOI] [PubMed] [Google Scholar]
- 15.Acharya D, Loyaga-Rendon RY, Tallaj JA, Pamboukian SV, Sasse MF. Circulatory support for shock complicating myocardial infarction. J Invasive Cardiol. 2014;26:E109–14. [PubMed] [Google Scholar]
- 16.O’Neill WW, Schreiber T, Wohns DH, et al. The current use of Impella 2. 5 in acute myocardial infarction complicated by cardiogenic shock: results from the USPella registry. J Interv Cardiol. 2014;27:1–11. doi: 10.1111/joic.12080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Boning A, Buschbeck S, Roth P, et al. IABP before cardiac surgery: clinical benefit compared with intraoperative implantation. Perfusion. 2013;28:103–8. doi: 10.1177/0267659112471577. [DOI] [PubMed] [Google Scholar]
- 18.O’Gara PT, Kushner FG, Ascheim DD, et al. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction. Circulation. 2013;127:e362–425. doi: 10.1161/CIR.0b013e3182742cf6. [DOI] [PubMed] [Google Scholar]
- 19.Chamogeorgakis T, Rafael A, Shafii AE, Nagpal D, Pokersnik JA, Gonzalez-Stawinski GV. Which is better: a miniaturized percutaneous ventricular assist device or extracorporeal membrane oxygenation for patients with cardiogenic shock? ASAIO J. 2013;59:607–11. doi: 10.1097/MAT.0b013e3182a8baf7. [DOI] [PubMed] [Google Scholar]
- 20.Truby L, Naka Y, Kalesan B, et al. Important role of mechanical circulatory support in acute myocardial infarction complicated by cardiogenic shock. Eur J Cardiothorac Surg. 2015;48:322–8. doi: 10.1093/ejcts/ezu478. [DOI] [PubMed] [Google Scholar]