Abstract
Network meta-analysis (NMA) expands the scope of a conventional pairwise meta-analysis to simultaneously compare multiple treatments, which has an inherent appeal for clinicians, patients, and policy decision makers. Two recent reports have shown that the impact of excluding a treatment on NMAs can be substantial. However, no one has assessed the impact of excluding a trial from NMAs, which is important because many NMAs selectively include trials in the analysis. This article empirically examines the impact of trial exclusion using both the arm-based (AB) and contrast-based (CB) approaches, by reanalyzing 20 published NMAs involving 725 randomized controlled trials and 449,325 patients. For the population-averaged absolute risk estimates using the AB approach, the average fold changes across all networks ranged from 1.004 (with standard deviation 0.004) to 1.072 (with standard deviation 0.184); while the maximal fold changes ranged from 1.032 to 2.349. In 12 out of 20 NMAs, a 1.20-fold or larger change is observed in at least one of the population-averaged absolute risk estimates. In addition, while excluding a trial can substantially change the estimated relative effects (e.g., log odds ratios), there is no systematic difference in terms of changes between the two approaches. Changes in treatment rankings are observed in 7 networks and changes in inconsistency are observed in 3 networks. We do not observe correlations between changes in treatment effects, treatment rankings and inconsistency. Finally, we recommend rigorous inclusion and exclusion criteria, logical study selection process, and reasonable network geometry to ensure robustness and generalizability of the results of NMAs.
Introduction
In clinical practice, and at a wider societal level, treatment decisions need to consider all available evidence. Network meta-analysis (NMA) expands the scope of a conventional pairwise meta-analysis to simultaneously compare multiple treatment options [1–4] by collectively synthesizing direct evidence within trials and indirect evidence across trials. In the simplest case, one may be interested in comparing two treatments A and B. Direct evidence comes from randomized controlled trials (RCTs) comparing A and B, while indirect evidence comes from RCTs of either A or B versus a common comparator C. NMA has an inherent appeal for clinicians, patients, and policy decision makers because it enables simultaneous inference of multiple treatments and strengthens inference by including indirect evidence [5].
However, meta-analysts undertaking an NMA often selectively choose trials to include in the systematic reviews due to certain preference. For instance, some NMAs exclude trials with placebo or no treatment due to the belief that the placebo or no treatment may vary over time or be set in favorable conditions to appease regulatory authorities [6]; whereas some NMAs exclude trials without a placebo- or no treatment-arm (i.e., exclude trials comparing solely active treatments) [7]. In addition, some NMAs may include only trials available in a particular location or time period for convenience. It is generally difficult and tedious to include all existing trials that meet the inclusion/exclusion criteria due to some technical issues (i.e., some trials may be published using other languages) in NMA. Intuitively, if the omitted trials are similar to the included trials and there is sufficient number of included trials, the failure to include these omitted trials will only result in less information (i.e., bigger standard errors and wider confidence intervals), but will not have any systematic impact on the estimates. However, if the omitted trials happen to be different from the included, or if the number of included trials is too small to provide robust estimation, then omission of these trials may have profound influence. The exploration of impact of exclusion of trials helps make better sense of a network meta-analysis and guide future design and conduct of trials and meta-analyses.
A recent publication by Mills et al. [8] investigated the impact of removing a treatment arm (including placebo / no treatment) on the estimated effect sizes for NMAs by reanalyzing 18 NMAs, and concluded that excluding a treatment could have substantial influence on estimated effect sizes. They consequently stated that selection of treatment arms should be carefully considered when applying NMAs. Another publication by Lin et al. [9] further explored the sensitivity to excluding treatments using both the armed-based (AB) [1] and contrast-based (CB) [2] NMA approaches. They found that when a treatment was removed under the CB framework, it was also necessary to exclude the other treatment in two-arm studies that investigated the excluded treatment, while such additional exclusions were not necessary in the AB framework. To the best of our knowledge, no previous works, thus far, have empirically studied the impact of removing a trial in NMAs.
The primary objective of this article is to obtain empirical evidence of the impact of removing a trial on the effect size estimates. We investigate both the AB [1, 4] and CB [3] (a more general version than in [2]) NMA approaches by reanalyzing 20 published NMAs with binary outcomes. The impact on treatment rankings and inconsistency between direct and indirect evidence are also assessed based on the AB approach. This article is organized as follows. First, we describe the characteristics of the 20 network meta-analyses. Second, we briefly introduce the two NMA approaches and our procedures assessing the impact of excluding a trial. Fold changes are used in evaluating the impact on estimated population-averaged absolute risks from the AB approach, and changes in log odds ratios (log ORs) are used to compare the results from the AB and CB approaches. We close with a brief discussion with some suggestions for future conduct of NMAs and several limitations of our empirical study.
Materials and Methods
Data source and extraction
We reviewed the NMAs studied by Veroniki et al. [10], which searched in PubMed for articles published between March 1997 and February 2011 in which any form of indirect comparison was applied, according to the articles’ titles or abstracts. The authors initially identified 817 articles and after the screening process they ended up with 40 networks. They screened the articles according to 1) whether the networks include at least four treatments, (2) whether the networks contain one closed loop, (3) whether indirect comparisons are included, (4) whether the major outcomes are dichotomous, (5) whether the articles are research papers instead of discussing / commentary papers. We selected 20 networks in our analysis. Nineteen of them were excluded according to our inclusion criterion that each treatment should be compared in at least two trials; otherwise, the networks are poorly connected at that treatment node. Furthermore, a treatment that is only compared in one trial would disappear from the sensitivity analysis if that trial is excluded, disabling the possibility to investigate the impact on any effect sizes related to that specific treatment. A network by Brown et al. [11] was also excluded because zero events were observed in many arms, which would bring bias proportional to the rarity of the event under study [12, 13]. Finally 20 networks involving 725 randomized controlled trials and 449,325 patients were selected; they are Ara 2009 [14], Baker 2009 [15], Ballesteros 2005 [16], Bansback 2009 [17], Bucher 1997 [18], Cipriani 2009 [19], Eisenberg 2008 [20], Elliott 2007 [21], Govan 2009 [22], Lu 2006 [3], Lu 2009 [2], Macfayden 2005 [23], Middleton 2010 [24], Mills 2009 [25], Picard 2000 [7], Puhan 2009 [26], Thijs 2008 [27], Trikalinos 2009 [28], Wang 2010 [29], and Yu 2006 [30].
Table 1 presents the characteristics of the individual networks. Specifically, the first column in Table 1 lists the IDs of these networks. The second column shows the author and year of publication for each NMA. The third to the fifth columns list the type of diseases, the primary outcomes of interest, and the multiple investigated treatments (and their abbreviations) studied in each network. We had preference of efficacy outcome over others for studies that considered more than one outcome, as was done in Veroniki et al. [10]. The sixth column presents the number of trials and treatments contained in each network, from which we can see that each NMA has four or more treatments and more than twice as many studies as treatments. Networks range in size from 9 trials on 4 treatments to 111 trials on 12 treatments. The last column shows the minimum and maximum frequencies for treatments (i.e., the number of trials that contain a treatment) for each network. For example, in the first network Ara 2009 [14], treatments are compared in at least 3 but no more than 7 trials. The frequencies across all networks range from 2 to 89.
Table 1. Characteristics of the 20 network meta-analyses.
| ID | Network* | Condition/Disease | Outcome | Treatment names (abbreviations) | No. of trials (treatment) | Frequency (min/max)† |
|---|---|---|---|---|---|---|
| 1 | Ara 2009 [14] | Hypercholesterolaemia | Effectiveness in reducing LDL-c. | 1 Placebo; 2 Simvastatin 40 mg/day (SIM 40); 3 Atorvastatin 80 mg/day (ATO 80); 4 Simvastatin 80 mg/day (SIM 80); 5 Rosuvastatin 40 mg/day (ROS 40) | 11 (5) | 3/7 |
| 2 | Baker 2009 [15] | Chronic obstructive pulmonary disease (COPD) | Exacerbation episodes in Chronic Obstructive Pulmonary Disease (COPD> = 1) | 1 Placebo; 2 Fluticasone (FLU); 3 Budesonide (BUD); 4 Salmeterol (SAL); 5 Formoterol (FOR); 6 Tiotropium (TIO); 7 Fluticasone+Salmeterol (FLU+SAL); 8 Budesonide+Formoterol (BUD+FOR) | 38 (8) | 2/34 |
| 3 | Ballesteros 2005 [16] | Dysthymia | Efficacy of antidepressants in dysthymia | 1 Placebo; 2 Tricyclic antidepressant (TCA); 3 Selective serotonin reuptake inhibitor (SSRI); 4 Monoamine oxidase inhibitor (MAOI) | 9 (4) | 3/9 |
| 4 | Bansback 2009 [17] | Moderate to severe plaque psoriasis | Efficacy—PASI 75 response score for the treatment of psoriasis | 1 Placebo; 2 Etanercept (ETA); 3 Infliximab (INF); 4 Adalimumab (ADA); 5 Efalizumab (EFA); 6 Alefacept (ALE); 7 Cyclosporine (CYC); 8 Methotrexate (MET) | 22 (8) | 2/21 |
| 5 | Bucher 1997 [18] | Pseudocystis carinii | Number of Pseudocystis Carinii pneumonia (prophylaxis against Pneumocystis carinii in HIV infected patients) | 1 Aerosolized pentamidine (AP); 2 Trimethoprim-sulphamethoxazole (TMP-SMX); 3 Dapsone (D); 4 Dapsone/pyrimethamine (D/P) | 18 (4) | 4/14 |
| 6 | Cipriani 2009 [19] | Unipolar major depression | Efficacy—the proportion of patients who responded to the allocated treatment | 1 Fluoxetine (FLU); 2 Sertraline (SER); 3 Citalopram (CIT); 4 Eescitalopram (ESC); 5 Paroxetine (PAR); 6 Fluvoxamine (FVX); 7 Milnacipran (MIL); 8 Venlafaxine (VEN); 9 Reboxetine (REB); 10 Bupropion (BUP); 11 Mirtazapine (MIR); 12 Duloxetine (DUL) | 111 (12) | 6/52 |
| 7 | Eisenberg 2008 [20] | Smoking | Smoking abstinence | 1 Placebo; 2 Buprobion (BUP); 3 Nicotine gum (NG); 4 Transdermal nicotine (TN); 5 Varenicline (VAR) | 61 (5) | 6/61 |
| 8 | Elliott 2007 [21] | Hypertension | Effect of antihypertensives on incidence diabetes mellitus-proportion of patients who developed diabetes | 1 Placebo; 2 Thiazide diuretic (TD); 3 Angiotensin- converting enzyme (ACE) inhibitor; 4 Calcium-channel blockers (CCB); 5 Angiotensinreceptor blockers (ARB); 6 β-blocker (BB) | 22 (6) | 5/9 |
| 9 | Govan 2009 [22] | Stroke | Death by the end of scheduled follow up | 1 Stroke ward (SW); 2 General medical ward (GMW); 3 Mixed rehabilitation ward (MRW); 4 Mobile stroke team (MST); 5 Acute (semi-intensive) ward (AW) | 28 (5) | 2/24 |
| 10 | Lu 2006 [3] | Smoking | Smoking cessation | 1 No contact; 2 Self-help; 3 Inidividual counselling (IC); 4 Group counselling (GC) | 24 (4) | 6/19 |
| 11 | Lu 2009 [2] | Gastroesophageal reflux disease | The number of healed patients at one or more follow-up times | 1 Placebo; 2 Prokinetic agents (PA); 3 H2 receptor antagonists (H2RA); 4 H2RA double dose (H2RA-D); 5 Proton pump inhibitors (PPI); 6 PPI double dose (PPI-D) | 40 (6) | 4/32 |
| 12 | Macfayden 2005 [23] | Chronically discharging ears with underlying eardrum perforations | Resolution of discharge | 1 No treatment (No Trt); 2 Topical quinolone antibiotic (TQA); 3 Topical non-quinolone antibiotic (TNQA); 4 Topical antiseptic (TA) | 13 (4) | 2/11 |
| 13 | Middleton 2010 [24] | Heavy menstrual bleeding | Efficacy as second line treatment for heavy menstrual bleeding—dissatisfaction at 12 months | 1 “First generation” endometrial destruction techniques (FG); 2 Hysterectomy (HYST); 3 “Second generation” endometrial destruction techniques (SG); 4 Mirena (MIR) | 20 (4) | 4/17 |
| 14 | Mills 2009 [25] | Smoking | Smoking Abstinence at approximately 4 weeks post-target quit date (TQD) | 1 Control; 2 Nicotine replacement therapy (NRT); 3 Bupropion (BUP); 4 Varenicline (VAR) | 89 (4) | 9/89 |
| 15 | Picard 2000 [7] | Pain on injection | Analgecic efficacy of proplylactic interventions for the prevention of pain on injection with propofol—no pain | 1 Placebo; 2 No treatment (No Trt); 3 Lidocaine (mg) given before the injection of propofol (LIDb); 4 Lidocaine (mg) mixed with propofol 200 mg (LIDm); 5 Lidocaine (mg) with tourniquet (LID+TOU); 6 Opioids (OPI); 7 Metoclopramide (MET); 8 Temperature (TEM). | 43 (8) | 4/34 |
| 16 | Puhan 2009 [26] | Chronic obstructive pulmonary disease (COPD) | Exacerbation in patients with chronic obstructive pulmonary disease | 1 Placebo; 2 Long-acting beta-agonists (BA); 3 Long-acting anticholinergics (AC); 4 Inhaled corticosteroids (IC); 5 Combined treatment with a long-acting beta-agonist and an inhaled corticosteroid (CT) | 34 (5) | 8/31 |
| 17 | Thijs 2008 [27] | Stroke | Efficacy of antiplatelet in the prevention of serious vascular events after transient ischaemic attack or stroke | 1 Placebo; 2 Thienopyridines (ticlopidin or clopidogrel) + Aspirin (THI+ASA); 3 ASA; 4 Aspirin and dipyridamole (ASA+DP); 5 THI | 23 (5) | 3/19 |
| 18 | Trikalinos 2009 [28] | Coronary artery disease | Coronary artery disease—death | 1 Medical therapy (MT); 2 Percutaneous transluminal balloon coronary angioplasty (PTCA); 3 Bare-metal stents (BMS); 4 Drugeluting stents (DES). | 62 (4) | 13/52 |
| 19 | Wang 2010 [29] | Catheter-related infections | Catheter colonisation | 1 Standard; 2 Chlorhexidine and silver sulfadiazine + (CHSS+); 3 Benzalkonium chloride (BC); 4 Silver iontophoretic (SIT); 5 Minocycline-rifampicin (MI); 6 CHSS; 7 Silver alloy-coated (SAC); 8 Silver-impregnated (SIP); 9 Heparin-bonded (HB) | 43 (9) | 2/40 |
| 20 | Yu 2006 [30] | Cardiac surgery | Cardiac ischemic complications and mortality | 1 Control; 2 Enflurane (ENF); 3 Isoflurane (ISO); 4 Halothane (HAL); 5 Sevoflurane (SEV); 6 Desflurane (DES) | 14 (6) | 2/14 |
* Network shows the author and year of each published NMA.
† Frequency reports the smallest and largest number of trials that contain a treatment in each network.
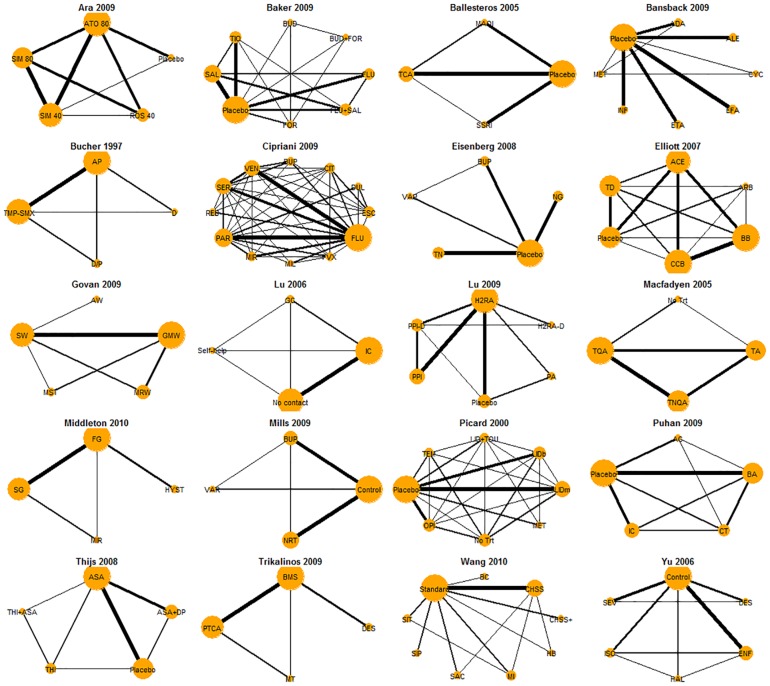
Fig 1 graphically displays the 20 networks. In each network plot, the thickness of each link is proportional to the number of trials investigaing the relation, and the size of each treatment node is proprotional to the number of direct comparisons that contain that treatment. Neither the number of trials for each pairwise comparison nor the number of direct comparisons for each treatment are balanced in all networks. The pool includes various constructions of networks, where one of which (i.e., Lu 2006 [3]) contains direct information for all pairwise comparisions while the rest do not.
Fig 1. Graphacial representations for the 20 NMAs.
The thickness of each link is proportional to the number of trials investigaing the relation, and the size of each treatment node is proprotional to the number of direct comparisons that contain that treatment.
Statistical models for NMA with binary data
Now, we briefly introduce both the AB and CB approaches using Bayesian hierarchical models. The AB approach focuses on absolute risks for each treatment arm, while the CB approach focuses on relative effects (e.g., ORs under binary case). Existing literature [1–4, 31, 32] has explored and discussed the model assumptions and model fit of the two approaches, and two recent discussion papers have further provided detailed comparisons on their strengths and limitations; see [33, 34].
We consider an NMA of I trials and K treatments of interest. Since most of the trials only compare a subset of the treatments of interest, we let Si denote the set of treatments that are compared in the ith trial, whose cardinality is equal to or smaller than (in most cases) K. Let nik be the total number of subjects, yik be the number of events, and pik be the corresponding probability of events for the kth treatment in the ith trial. We denote all observed data by D.
For the AB approach proposed by Zhang et al. [1, 4], it specifies yik~Bin(nik, pik), k∈Si, i = 1,…,I, and Φ−1(pik) = μk + σvik, (vi1,…,vik)T ~ MVN(0, RK). Here μk is the fixed treatment effect for the kth treatment, σ is the standard deviation for the random effects vik, and RK is an exchangeable correlation matrix. The population-averaged treatment-specific event rate πk has a closed form based on the above model: , where ϕ() is the density function and Φ() is the cumulative density function of the standard normal distribution. The ranking of treatments is calculated based on πk. When the outcome has a positive interpretation (say, efficacy), the posterior probability of being the best (Pbest) is P(k is the best treatment | D) = P(rank(πk) = 1 | D); while when the outcome has a negative interpretation (say, adverse event), it is P(k is the best treatment | D) = P(rank(πk) = K | D). The marginal ORs are then defined as ORkl = [πk/(1 − πk)]/[πl/(1 − πl)] for a pairwise comparison between Treatments k and l (k≠l). We report ORs in addition to event rates in this paper in order to be consistent with the CB approach.
Zhao et al. [35] proposed methods to detect inconsistency based on the AB approach. To measure inconsistency between Treatments k1 and k2, trials are divided into four groups: (i) trials that include both k1 and k2, (ii) trials include k1 but not k2, (iii) trials include k2 but not k1, (iv) trials that include neither k1 nor k2. Then the discrepancy of direct and indirect evidence can be tested by computing the posterior distribution of the discrepancy factor . If zero is in the far tail of this posterior distribution, then inconsistency is found. Note that each pair of treatments needs to be assessed in a separate model and a pair with no information of group (i), (ii) or (iii) is ineligible for inconsistency detection.
For the CB approach proposed by Lu & Ades [3], it is based on the following hierarchical specification: logit(pik) = μi + Xikδib(i)k, where μi is the baseline effect in Trial i, Xik’s are indicators for baseline treatments taking value 0 if k = b and 1 if k≠b, and δib(i)k represents the relative random effect of Treatment k versus b(i) on log odds scale in the ith trial. In the next step, the vector (δib(i)k) is assumed to have a |Si| − 1 dimensional normal distribution (a univariate normal distribution if the ith trial contains two arms or a multivariate normal distribution if the ith trial contains multiple arms) with mean vector (db(i)k) and covariance matrix , i.e., . A very common is a homogeneous-variance exchangeable matrix with correlation 1/2, i.e., δib(i)k ~ N(db(i)k, σ2) and cov(δib(i)k, δib(i)h) = σ2/2. The model in addition assumes exchangeability, i.e., dkl = dbk − dbl. Finally .
Sensitivity analysis of excluding a trial
Regardless of the approaches used in the original publications of the 20 NMAs, we reanalyzed them in this paper using both the AB and CB approaches described in the previous Section. Five steps were applied to each NMA to analyze the impact of omission of trials on the estimated treatment effects and two more steps were conducted to assess the influence on treatment ranks and inconsistency. The details are as follows:
Fit the AB and CB NMA Bayesian hierarchical models separately to the complete data of each NMA. For the AB approach, both absolute risks for each treatment arm and ORs for all pairwise comparisons are recorded; while for the CB approach, only ORs are recorded.
Remove each trial within each NMA and reanalyze the data using both the AB and CB approaches. Same statistical summaries are recorded as in Step 1.
Calculate fold changes in the estimated absolute risks (from the AB approach) to evaluate the impact of exclusion of trials. The fold change for Treatment k from omission of Trial i is equal to if ; otherwise it is equal to . Here is the estimated absolute risk for Treatment k after exclusion of Trial i. In other words, fold changes are always expressed as a value greater than 1.00. Take a simple example, if a specific event rate is 0.70 in the full network and 0.50 in the network with one trial excluded, then the change is 0.70/0.50 = 1.40-fold; if the event rates are 0.40 and 0.60 instead in the full and incomplete networks respectively, then the change is 0.60/0.40 = 1.50-fold. The larger the fold change is, the larger the impact is.
Compute the log OR changes after excluding a trial using the AB and CB approaches (i.e., and , respectively). Here and represent the ORs estimated from the AB and CB approaches without Trial i. If and are around 0, then there is subtle impact of excluding Trial i. The further they are from 0, the larger the impact of excluding Trial i is.
Compare the difference between and through graphical tools (e.g., scatter plot and Bland-Altman plot) and statistical tests. The average of of all pairwise comparisons using all eligible trial exclusions across all networks from the AB approach is compared with that from the CB approach (i.e., the average of ). Bootstrap resampling technique [36] is applied to compute the 95% confidence intervals (CIs) and the p-value for testing difference. Note that 10,000 bootstrap samples are constructed at the network level; that is, each sample contains 20 resampled networks, drawn with replacement from the original 20 networks.
Assess whether the best treatment and the corresponding Pbest of that treatment change after omission of trials, using the AB approach.
Evaluate the influence of omission of trials on inconsistency using the AB approach.
Step 3 evaluates the impact of omission of trials on the estimated absolute risks using the AB approach, and Steps 4 and 5 compare the results of impact based on the AB and CB approaches. Steps 1–5 investigate the impact on treatment effects, while Steps 6–7 further explore the influence on treatment ranks and inconsistency.
Analyses were conducted via Markov chain Monte Carlo (MCMC) methods using JAGS [37] and the R package “rjags” [38]. The S1 Appendix provides the JAGS codes for both approaches. The convergence of the MCMC chains was assessed by the Gelman-Rubin convergence statistic [39] and a visual inspection of the chains.
Results
Fold changes in event rates estimated from the AB approach
The average and maximal fold changes for each network from the AB approach are reported in Table 2. The average fold changes across all networks ranged from 1.004 (with standard deviation 0.004) to 1.072 (with standard deviation 0.184); while the maximal fold changes ranged from 1.032 to 2.349. In 8 of 20 networks, the maximal changes were below 1.200-fold; while 5 of them obtained maximal changes below 1.100-fold. Mills et al. [8] suggested considering relative changes exceeding 1.20-fold as substantial. Using this threshold, 12 out of the 20 networks had relative changes larger than 1.20-fold observed in at least one of the population-averaged absolute risk estimates. It suggests that omission of trials may have substantial impact on the estimation.
Table 2. Summary of fold changes in terms of estimated event rates using the AB approach.
| Network | Fold Change | Proportions of fold changes within each magnitude category* | ||||||
|---|---|---|---|---|---|---|---|---|
| Average (sd) | Maximal | 1.00–1.10 | 1.10–1.20 | 1.20–1.30 | 1.30–1.40 | 1.40–1.50 | >1.50 | |
| Ara 2009 [14] | 1.054 (0.049) | 1.215 | 0.836 | 0.145 | 0.018 | 0.000 | 0.000 | 0.000 |
| Baker 2009 [15] | 1.025 (0.030) | 1.388 | 0.987 | 0.010 | 0.000 | 0.003 | 0.000 | 0.000 |
| Ballesteros 2005 [16] | 1.038 (0.034) | 1.174 | 0.972 | 0.028 | 0.000 | 0.000 | 0.000 | 0.000 |
| Bansback 2009 [17] | 1.033 (0.099) | 1.835 | 0.949 | 0.028 | 0.006 | 0.000 | 0.000 | 0.017 |
| Bucher 1997 [18] | 1.044 (0.052) | 1.219 | 0.861 | 0.125 | 0.014 | 0.000 | 0.000 | 0.000 |
| Cipriani 2009 [19] | 1.004 (0.004) | 1.057 | 1.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Eisenberg 2008 [20] | 1.010 (0.009) | 1.057 | 1.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Elliott 2007 [21] | 1.034 (0.033) | 1.130 | 0.909 | 0.091 | 0.000 | 0.000 | 0.000 | 0.000 |
| Govan 2009 [22] | 1.028 (0.117) | 2.349 | 0.986 | 0.000 | 0.000 | 0.007 | 0.000 | 0.007 |
| Lu 2006 [3] | 1.028 (0.040) | 1.241 | 0.948 | 0.042 | 0.010 | 0.000 | 0.000 | 0.000 |
| Lu 2009 [2] | 1.016 (0.035) | 1.329 | 0.962 | 0.033 | 0.000 | 0.004 | 0.000 | 0.000 |
| Macfayden 2005 [23] | 1.034 (0.038) | 1.156 | 0.904 | 0.096 | 0.000 | 0.000 | 0.000 | 0.000 |
| Middleton 2010 [24] | 1.035 (0.048) | 1.338 | 0.938 | 0.050 | 0.000 | 0.012 | 0.000 | 0.000 |
| Mills 2009 [25] | 1.006 (0.005) | 1.032 | 1.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Picard 2000 [7] | 1.013 (0.020) | 1.222 | 0.988 | 0.009 | 0.003 | 0.000 | 0.000 | 0.000 |
| Puhan 2009 [26] | 1.028 (0.019) | 1.093 | 1.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Thijs 2008 [27] | 1.018 (0.013) | 1.044 | 1.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| Trikalinos 2009 [28] | 1.020 (0.030) | 1.336 | 0.988 | 0.008 | 0.000 | 0.004 | 0.000 | 0.000 |
| Wang 2010 [29] | 1.017 (0.035) | 1.498 | 0.982 | 0.013 | 0.003 | 0.000 | 0.003 | 0.000 |
| Yu 2006 [30] | 1.072 (0.184) | 2.303 | 0.881 | 0.036 | 0.012 | 0.048 | 0.000 | 0.024 |
* sd represents standard deviation. Cells are in bold if all fold changes in the network fall in [1.00, 1.10]; cells are italic if fold changes > 1.20. 1.00–1.10 = [1.00, 1.10]; 1.10–1.20 = (1.10, 1.20]; 1.20–1.30 = (1.20, 1.30]; 1.30–1.40 = (1.30, 1.40]; 1.40–1.50 = (1.40, 1.50].
Table 2 also summarizes the proportions of fold changes in the estimated event rates falling in [1.00, 1.10], (1.10, 1.20], (1.20, 1.30], (1.30, 1.40], (1.40, 1.50], and (1.50, +∞) intervals for the 20 NMAs. Five networks, which were Cipriani 2009 [19], Eisenberg 2008 [20], Mills 2009 [25], Puhan 2009 [26], and Thijs 2008 [27], obtained fold changes of estimated event rates all smaller than 1.10-fold. Fold changes in another three networks, Ballesteros 2005 [16], Elliott 2007 [21], and Macfayden 2005 [23], were all smaller than 1.20 with some larger than 1.10. Nine networks obtained relative changes all smaller than 1.50-fold with some exceeding 1.20-fold; they were Ara 2009 [14], Baker 2009 [15], Bucher 1997 [18], Lu 2006 [3], Lu 2009 [2], Middleton 2010 [24], Picard 2000 [7], Trikalinos 2009 [28] and Wang 2010 [29]. The rest three networks, i.e., Bansback 2009 [17], Govan 2009 [22] and Yu 2006 [30], contained changes in estimated event rates larger than 1.50-fold.
We further explore the features of the three networks with fold changes larger than 1.50. In Bansback 2009 [17], exclusion of Trials 21 and 22 led to 1.805-fold and 1.835-fold changes in the estimated event rate for Treatment 7 (Cyclosporine), respectively. This observation is understandable because Trials 21 and 22 were the only two trials containing Cyclosporine, whereas the crude event rates (observed number of events / observed total number of subjects) of Cyclosporine in the two trials were 0.200 and 0.714, respectively. Thus excluding either trial would lead to substantial changes in estimation. In addition, exclusion of Trial 10 in this network resulted in a 1.541-fold change in event rate for Treatment 8 (Methotrexate), which were compared in only Trials 10 and 22 with sample sizes 110 and 43 and crude event rates 0.364 and 0.605, respectively. In Govan 2009 [22], Treatment 5 (Acute ward) was compared in only Trials 25 and 26, and the exclusion of Trial 26 resulted in a fold change of value 2.349 in the estimated event rate for Acute ward. Though crude event rates in those two trials were not significantly different, Trial 26 contained a much larger sample size of 134 in contrast to 27 for Trial 25. In Yu 2006 [30], Treatment 4 (Halothane) was compared in only Trials 1 and 8 with sample sizes 253 and 14 and crude event rates 0.036 and 0.071, and Treatment 6 (Desflurane) was compared in only Trials 11, 13 and 14 with sample sizes 80, 100, 25 and crude event rates 0.013, 0.040 and 0.000. The exclusion of Trials 1 and 13 produced 1.951-fold and 2.303-fold changes in the estimated event rates for Treatments 4 and 6, respectively. In summary, the most influential trials typically contain larger sample sizes among the few trials that compare treatments with small frequencies (in other words, treatments that are compared in small numbers of trials) and sometimes report different crude event rates from the rest. Omission of those trials may bring larger impact in the estimation of treatment effects, thus may influence treatment comparison and decision making. It further implies the importance of network geometry.
Comparison of the results from the AB and CB approaches
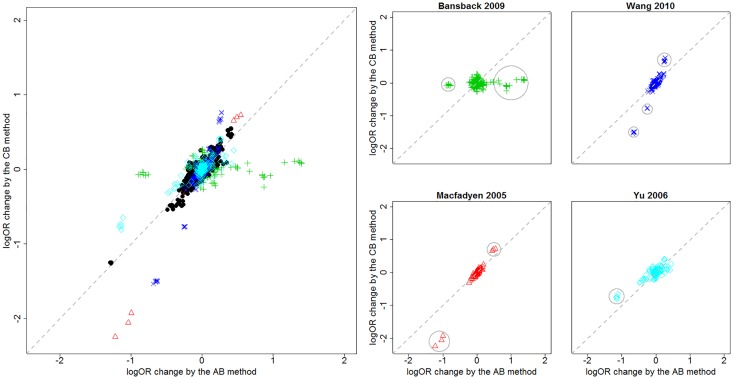
and were recorded and used to compare the performance of the AB and CB approaches. The left panel in Fig 2 presents the scatter plots of against pooled from the 20 networks across all pairwise comparisons and trial exclusions. Most of the scatters tended to concentrate in the vicinity of the identity line, i.e., a y = x line, suggesting agreement between the AB and CB approaches. But scatters from four networks, i.e. Bansback 2009 [17], Macfadyen 2005 [23], Wang 2010 [29] and Yu 2006 [30], were found to deviate from the identity line and marked with colored points. The right panel excerpts scatter plots for these four networks individually. For Bansback 2009 [17] and Yu 2006 [30], omission of trials had larger impact from the AB approach than from the CB approach; while for Macfadyen 2005 [23] and Wang 2010 [29], CB approach was more sensitive to excluding trials. However, only small numbers of the points in the scatter plots were away from the identity line; more specifically, 22 out of 616 (i.e., 3.6%) in Bansback 2009 [17], 6 out of 78 (i.e., 7.7%) in Macfadyen 2005 [23], 5 out of 210 (i.e., 2.4%) in Wang 2010 [29] and 24 out of 1548 (i.e., 1.6%) in Yu 2006 [30]. These points are circled in their individual scatter plots in the right panel of Fig 2.
Fig 2. Scatter plots of logOR changes after and before excluding trials for the 20 networks.
The x-axis labels changes obtained from the AB approach, i.e., , while the y-axis labels changes obtained from the CB approach, i.e., . Left panel pools results from the 20 networks with scatters deviating from the identity line in color. Right panel excerpts colored scatters.
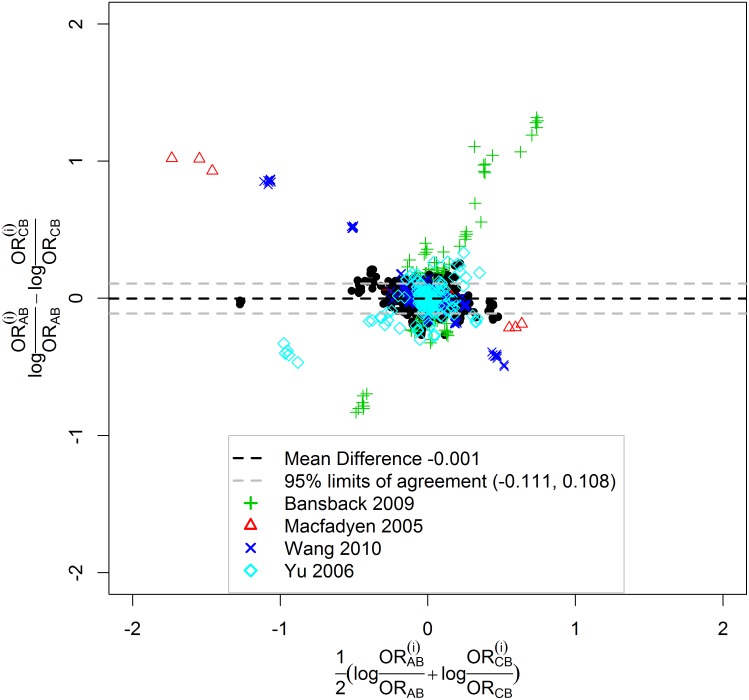
The Bland-Altman plot in Fig 3 further consolidates the agreement between these two approaches on the impact of excluding trials. The differences pooled from all networks including all pairwise comparisons and trial exclusions were plotted against the means . The mean of these differences (i.e., mean of ) was equal to -0.001 and was drawn in black dashed line in Fig 3. The standard deviation (SD) of the differences was 0.055 and the width of the 95% limits of agreement (drawn in grey dashed lines) was 0.219. The narrow range of the 95% limits of agreement showed good agreement. In addition, 98.2% (15597/15878) of the differences were contained in the 95% limits of agreement. Thus we conclude that the CB approach agrees well with the AB approach in terms of the impact of excluding trials. Note that the 4 excerpted networks are also highlighted in color in the Bland-Altman plot.
Fig 3. Bland-Altman plot.
The differences between the AB and CB approaches in terms of log OR changes after omission of trials, i.e., , is drawn agaist the mean, i.e., . The mean difference and 95% limits of agreement are shown in dashed lines. Four networks are highlighted in color.
Statistical testing was also conducted to compare the AB and CB approaches in addition to the graphical exploration. We let ηAB and ηBC denote the true mean and mean of all pairwise comparisons and trial exclusions from all networks for the AB and CB approaches, respectively. The estimates for ηAB and ηBC based on the current data were 0.021 and 0.021. Using the 10,000 bootstrap samples, 95% CIs for ηAB and ηBC were estimated to be (0.014, 0.040) and (0.013, 0.038), respectively. For the hypothesis testing H0:ηAB = ηBC versus HA: ηAB ≠ ηBC, the p-value was calculated based on another 10,000 bootstrap samples under the null hypothesis. It turned out that p-value = 0.156. Therefore the absolute log OR changes under the AB approach were not statistically significantly different from those under the CB approach.
Impact on treatment ranks and inconsistency based on the AB approach
Table 3 shows changes in the best treatment and Pbest after omission of trials. Networks whose outcomes have negative interpretations are listed in italics. The best treatment in thirteen networks after omission of trials remains the same. The Pbest of that treatment is also provided for both the full and reduced networks. For example, in Ara 2009 [14], ATO 80 ranks as the best treatment in both the full (with Pbest = 0.880) and reduced networks (with Pbest ranging from 0.778 to 0.878). The rest seven networks show changes in the best treatment. For Baker 2009 [15], BUD + FOR is the best treatment in the full network with Pbest = 0.463, while TIO is the best treatment after omission of Trials 11, 16, 17, 22, 26 or 34 with Pbest = 0.470, 0.551, 0.448, 0.479, 0.514 and 0.445 respectively, and BUD is the best treatment after omission of Trial 18. For Ballesteros 2005 [16], MAOI is the best treatment with Pbest = 0.496 in the full network, but SSRI becomes the best treatment with Pbest = 0.529 after omission of Trial 18. For Lu 2009 [2], PPI-D is the best treatment in the full network with Pbest = 0.567, but PPI becomes the best after omission of Trials 19, 22, 36 or 39 with Pbest = 0.538, 0.523, 0.584 and 0.538 respectively. For Puhan 2009 [26], AC is the best treatment in the full network with Pbest = 0.545 and CT is the best after omission of Trials 9, 19 or 33 with Pbest = 0.343, 0.405, 0.344 respectively. For Wang 2010 [29], MI is the best treatment with Pbest = 0.619 in the full network but CHSS+ becomes the best with Pbest = 0.518 after omission of Trial 37. Finally for Yu 2006 [30], SEV is the best treatment with Pbest = 0.673 in the full network while DES becomes the best with Pbest = 0.723 after omission of Trial 13.
Table 3. Impact on treatment ranks, probability of the best treatment, and inconsistency using the AB approach.
Note: For networks in italics, the treatment with the lowest event rate is the best treatment; for the other networks, the treatment with the highest event rate is the best treatment. -----represents that inconsistency cannot be assessed.
| Network | Change in the best treatment | Change in probability of being the best treatment | Change in inconsistency | |
|---|---|---|---|---|
| Full | Reduced | |||
| Ara 2009 [14] | None | ATO 80 (0.880) | ATO 80 (0.778–0.878) | None |
| Baker 2009 [15] | Top three switch | BUD+FOR (0.463) | BUD + FOR (0.437–0.546); TIO (0.470, 0.551, 0.448, 0.479, 0.514, 0.445 after omission of Trials 11, 16, 17, 22, 26, 34); BUD (0.796 after omission of Trial 18) | None |
| Ballesteros 2005 [16] | Top two switch | MAOI (0.496) | MAOI (0.476–0.674); SSRI (0.529 after omission of Trial 6) | ----- |
| Bansback 2009 [17] | None | INF (0.971) | INF (0.815–0.979) | ----- |
| Bucher 1997 [18] | None | TMP-SMX (0.996) | TMP-SMX (0.961–0.998) | None |
| Cipriani 2009 [19] | None | MIR (0.541) | MIR (0.381–0.639) | None |
| Eisenberg 2008 [20] | None | VAR (0.974) | VAR (0.942–0.987) | Yes (inconsistency between BUP and VAR observed after omission of Trial 61) |
| Elliott 2007 [21] | None | TD (0.698) | TD (0.535–0.858) | None |
| Govan 2009 [22] | None | AW (0.987) | AW (0.881–0.993) | None |
| Lu 2006 [3] | None | GC (0.760) | GC (0.554–0.907) | None |
| Lu 2009 [2] | Top two switch | PPI-D (0.567) | PPI-D (0.500–0.829); PPI (0.538, 0.523, 0.584, 0.538 after omission of Trials 19, 22, 36, 39) | None |
| Macfayden 2005 [23] | None | TQA (0.946) | TQA (0.855–0.967) | None |
| Middleton 2010 [24] | Top two switch | MIR (0.458) | MIR (0.438–0.849); FG (0.465, 0.556, 0.414, 0.492 after omission of Trials 16, 17, 18, 20) | None |
| Mills 2009 [25] | None | VAR (0.994) | VAR (0.978–0.997) | None |
| Picard 2000 [7] | None | LID+TOU (0.870) | LID + TOU (0.713–0.939) | None |
| Puhan 2009 [26] | Top two switch | AC (0.545) | AC (0.469–0.675); CT (0.343, 0.405, 0.344 after omission of Trials 9, 19, 33) | None |
| Thijs 2008 [27] | None | ASA+DP (0.715) | ASA+DP (0.582–0.802) | Yes (inconsistency between Placebo and ASA observed after omission of Trial 4) |
| Trikalinos 2009 [28] | None | DES (0.700) | DES (0.581–0.820) | Yes (Inconsistency between PTCA and BMS disappear after omission of Trials 7, 10, 17, 46, 50, 51, 53, 57, 62) |
| Wang 2010 [29] | Top two switch | MI (0.619) | MI (0.430–0.852); CHSS+ (0.518 after omission of Trial 37) | None |
| Yu 2006 [30] | Top two switch | SEV (0.673) | SEV (0.487–0.789); DES (0.723 after omission of Trial 13) | ----- |
Changes in inconsistency are also presented in Table 3. Three networks are not assessed because omission of some trials in these networks loses information of group (i), (ii) or (iii) for all pairs of treatments and thus disables the detection of inconsistency. For the rest seventeen networks, one eligible pair for each network is assessed. Omission of trials does not change the status of inconsistency in most networks except three (Eisenberg 2008 [20], Thijs 2009 [27] and Trikalinos 2009 [28]). In Eisenberg 2008 [20], inconsistency between BUP and VAR is observed after omission of Trial 61. In Thijs 2009 [27], inconsistency between Placebo and ASA appears after omission of Trial 4. In Trikalinos 2009 [28], inconsistency between PTCA and BMS disappears after omission of Trials 7, 10, 17, 46, 50, 51, 53, 57 or 62.
Discussion
It is common for NMAs to exclude specific trials and treatment arms based on diverse criteria [8], some limitations and preferences. The impact of exclusion of treatments arms was investigated in Mills et al. [8] and Lin et al. [9] empirically and substantial influence was found, whereas the impact of exclusion of trials has not been explored before. In this paper we empirically studied this impact using 20 published networks and documented that exclusion of trials can sometimes affect the estimation of treatment effects substantially.
We also found that exclusion of trials, which contain larger sample sizes compared with the other trials in the comparison of treatments with sparse information and which report different crude event rates from the rest, tend to result in larger changes in the estimation, which is as expected. Broadly network geometry including the abundance of trials, randomized patients for different trials and gaps of evidence in the treatment network should be taken seriously. In addition, the changes in treatment ranks and inconsistency are not correlated with changes in treatment effects.
Although the AB approach focuses on reporting population-averaged absolute risks and the CB approach focuses on estimating ORs, they both are sensitive to excluding trials. Our empirical study suggested that the two approaches generally agreed on the magnitude of changes in log OR (i.e.,), though some small disagreement were observed in 4 of the 20 networks. This work also contributes to the call for more empirical comparison of the AB and CB approaches [33, 34].
It has been discussed in the literature on how eligibility criteria may influence the results and the conclusions of traditional pairwise meta-analysis [40–44]. These findings suggest that in meta-analysis comparing multiple treatments, it is also very important to develop a rigorous systematic review protocol with logically considered inclusion and exclusion criteria and study selection process, such that the results from NMAs are robust and generalizable.
There are some limitations in our analysis. First, we used a selection criterion requiring each treatment to be studied in at least two studies. The literature has no well-established criterion serving this purpose. Second, though we did check the changes in evidence consistency, inconsistency detection in NMA is still an open question, has problems under both AB and CB framework, and awaits improvements [3, 35]. Third, we did not check outlying trials in this empirical study. Methods may need to be tailored to downweight outlyingness if needed [31].
Turning to future work, we are interested in exploring better inclusion and exclusion criteria for NMAs such as the minimum number of trials required to include a treatment arm in the NMA, and how to account for study quality in NMAs. Sufficient number of trials for each treatment arm is required to ensure sufficient statistical power to make robust conclusion, whereas outlying or low-quality trials should be deleted or down-weighted at the same time [31]. These have the potential to serve as supplement to the guidance for future conduct of NMAs and contribute to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) Extension Statement [45].
Supporting Information
(PDF)
Acknowledgments
J.Z. was supported in part by the NIAID AI103012 and a start-up fund from the University of Maryland. H.C. is supported in part by the US NIAID AI103012, NIDCR R03DE024750, NLM R21LM012197, NCI P30CA077598, NIMHD U54-MD008620, and NIDDK U01DK106786. Partial funding for open access provided by the UMD Libraries’ Open Access Publishing Fund.
Data Availability
We have included the datasets and codes as supplementary materials to this paper.
Funding Statement
J.Z. was supported in part by the NIAID AI103012 and a start-up fund from the University of Maryland. H.C. is supported in part by the US NIAID AI103012, NIDCR R03DE024750, NLM R21LM012197, NCI P30CA077598, NIMHD U54-MD008620, and NIDDK U01DK106786. Partial funding for open access provided by the UMD Libraries’ Open Access Publishing Fund. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Zhang J, Carlin BP, Neaton JD, Soon GG, Nie L, Kane R, et al. Network meta-analysis of randomized clinical trials: Reporting the proper summaries. Clinical Trials. 2014;11(2):246–62. 10.1177/1740774513498322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lu G, Ades A. Modeling between-trial variance structure in mixed treatment comparisons. Biostatistics. 2009;10(4):792–805. 10.1093/biostatistics/kxp032 [DOI] [PubMed] [Google Scholar]
- 3.Lu G, Ades A. Assessing evidence inconsistency in mixed treatment comparisons. Journal of the American Statistical Association. 2006;101(474):447–59. [Google Scholar]
- 4.Zhang J, Chu H, Hong H, Beth VA, Carlin BP. Bayesian hierarchical models for network meta-analysis incorporating nonignorable missingness. Statistical Methods in Medical Research. 2016;Forthcoming. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Li T, Puhan MA, Vedula SS, Singh S, Dickersin K. Network meta-analysis-highly attractive but more methodological research is needed. BMC medicine. 2011;9(1):79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Turner EH, Matthews AM, Linardatos E, Tell RA, Rosenthal R. Selective publication of antidepressant trials and its influence on apparent efficacy. New England Journal of Medicine. 2008;358(3):252–60. 10.1056/NEJMsa065779 [DOI] [PubMed] [Google Scholar]
- 7.Picard P, Tramer MR. Prevention of pain on injection with propofol: a quantitative systematic review. Anesthesia & Analgesia. 2000;90(4):963–9. [DOI] [PubMed] [Google Scholar]
- 8.Mills EJ, Kanters S, Thorlund K, Chaimani A, Veroniki AA, Ioannidis JPA. The effects of excluding treatments from network meta-analyses: survey. BMJ. 2013;347(f5195). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lin L, Chu H, Hodges JS. Sensitivity to excluding treatments in network meta-analysis. Epidemiology. 2016;27(4):562–9. 10.1097/EDE.0000000000000482 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Veroniki AA, Vasiliadis HS, Higgins JPT, Salanti G. Evaluation of inconsistency in networks of interventions. International Journal of Epidemiology. 2013;42(1):332–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Brown TJ, Hooper L, Elliott R, Payne K, Webb R, Roberts C, et al. A comparison of the cost-effectiveness of five strategies for the prevention of non-steroidal anti-inflammatory drug-induced gastrointestinal toxicity: a systematic review with economic modelling. Health Technol Assess. 2006;10 (iii—iv, xi—xiii):1–183. [DOI] [PubMed] [Google Scholar]
- 12.Bhaumik DK, Amatya A, Normand S-LT, Greenhouse J, Kaizar E, Neelon B, et al. Meta-analysis of rare binary adverse event data. Journal of the American Statistical Association. 2012;107(498):555–67. 10.1080/01621459.2012.664484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ma Y, Chu H, Mazumdar M. Meta-analysis of Proportions of Rare Events—A Comparison of Exact Likelihood Methods with Robust Variance Estimation. Communications in Statistics-Simulation and Computation. 2014; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ara R, Pandor A, Stevens J, Rees A, Rafia R. Early high-dose lipid-lowering therapy to avoid cardiac events: a systematic review and economic evaluation. Health Technol Assess. 2009;13:1–118. [DOI] [PubMed] [Google Scholar]
- 15.Baker WL, Baker EL, Coleman CI. Pharmacologic Treatments for Chronic Obstructive Pulmonary Disease: A Mixed-Treatment Comparison Meta-analysis. Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy. 2009;29(8):891–905. [DOI] [PubMed] [Google Scholar]
- 16.Ballesteros J. Orphan comparisons and indirect meta-analysis: A case study on antidepressant efficacy in dysthymia comparing tricyclic antidepressants, selective serotonin reuptake inhibitors, and monoamine oxidase inhibitors by using general linear models. Journal of Clinical Psychopharmacology. 2005;25(2):127–31. [DOI] [PubMed] [Google Scholar]
- 17.Bansback N, Sizto S, Sun H, Feldman S, Willian MK, Anis A. Efficacy of systemic treatments for moderate to severe plaque psoriasis: systematic review and meta-analysis. Dermatology. 2009;219(3):209–18. 10.1159/000233234 [DOI] [PubMed] [Google Scholar]
- 18.Bucher HC, Guyatt GH, Griffith LE, Walter SD. The results of direct and indirect treatment comparisons in meta-analysis of randomized controlled trials. Journal of Clinical Epidemiology. 1997;50(6):683–91. [DOI] [PubMed] [Google Scholar]
- 19.Cipriani A, Furukawa TA, Salanti G, Geddes JR, Higgins J, Churchill R, et al. Comparative efficacy and acceptability of 12 new-generation antidepressants: a multiple-treatments meta-analysis. The Lancet. 2009;373(9665):746–58. [DOI] [PubMed] [Google Scholar]
- 20.Eisenberg MJ, Filion KB, Yavin D, Bélisle P, Mottillo S, Joseph L, et al. Pharmacotherapies for smoking cessation: a meta-analysis of randomized controlled trials. Canadian Medical Association Journal. 2008;179(2):135–44. 10.1503/cmaj.070256 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Elliott WJ, Meyer PM. Incident diabetes in clinical trials of antihypertensive drugs: a network meta-analysis. The Lancet. 2007;369:201–7. [DOI] [PubMed] [Google Scholar]
- 22.Govan L, Ades A, Weir C, Welton N, Langhorne P. Controlling ecological bias in evidence synthesis of trials reporting on collapsed and overlapping covariate categories. Statistics in Medicine. 2010;29(12):1340–56. 10.1002/sim.3869 [DOI] [PubMed] [Google Scholar]
- 23.Macfadyen CA, Acuin JM, Gamble C. Topical antibiotics without steroids for chronically discharging ears with underlying eardrum perforations. Cochrane Database Systematic Reviews (Online). 2005;CD004618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Middleton L, Champaneria R, Daniels J, Bhattacharya S, Cooper K, Hilken N, et al. Hysterectomy, endometrial destruction, and levonorgestrel releasing intrauterine system (Mirena) for heavy menstrual bleeding: systematic review and meta-analysis of data from individual patients. BMJ. 2010;341:c3929 10.1136/bmj.c3929 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mills EJ, Wu P, Spurden D, Ebbert JO, Wilson K. Efficacy of pharmacotherapies for short-term smoking abstinance: a systematic review and meta-analysis. Harm Reduct J. 2009;6(25):1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Puhan MA, Bachmann LM, Kleijnen J, ter Riet G, Kessels AG. Inhaled drugs to reduce exacerbations in patients with chronic obstructive pulmonary disease: a network meta-analysis. BMC medicine. 2009;7(1):2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Thijs V, Lemmens R, Fieuws S. Network meta-analysis: simultaneous meta-analysis of common antiplatelet regimens after transient ischaemic attack or stroke. European Heart Journal. 2008;29(9):1086–92. 10.1093/eurheartj/ehn106 [DOI] [PubMed] [Google Scholar]
- 28.Trikalinos TA, Alsheikh-Ali AA, Tatsioni A, Nallamothu BK, Kent DM. Percutaneous coronary interventions for non-acute coronary artery disease: a quantitative 20-year synopsis and a network meta-analysis. The Lancet. 2009;373(9667):911–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wang H, Huang T, Jing J, Jin J, Wang P, Yang M, et al. Effectiveness of different central venous catheters for catheter-related infections: a network meta-analysis. Journal of Hospital Infection. 2010;76(1):1–11. 10.1016/j.jhin.2010.04.025 [DOI] [PubMed] [Google Scholar]
- 30.Yu CH, Beattie WS. The effects of volatile anesthetics on cardiac ischemic complications and mortality in CABG: a meta-analysis. Canadian Journal of Anesthesia. 2006;53(9):906–18. 10.1007/BF03022834 [DOI] [PubMed] [Google Scholar]
- 31.Zhang J, Fu H, Carlin BP. Detecting outlying trials in network meta-analysis. Statistics in medicine. 2015;34(19):2695–707. 10.1002/sim.6509 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hong H, Chu H, Zhang J, Carlin BP. A Bayesian missing data framework for generalized multiple outcome mixed treatment comparisons. Research synthesis methods. 2016;7(1):6–22. 10.1002/jrsm.1153 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Dias S, Ades A. Absolute or relative effects? Arm-based synthesis of trial data. Research Synthesis Methods. 2015; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hong H, Chu H, Zhang J, Carlin BP. Rejoinder to the discussion of “a Bayesian missing data framework for generalized multiple outcome mixed treatment comparisons,” by S. Dias and A.E. Ades. Research Synthesis Methods. 2015; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhao H, Hodges JS, Ma H, Jiang Q, Carlin BP. Hierarchical Bayesian approaches for detecting inconsistency in network meta-analysis. Statistics in Medicine. 2016; [DOI] [PubMed] [Google Scholar]
- 36.Efron B, Tibshirani RJ. An introduction to the bootstrap: CRC press; 1994. [Google Scholar]
- 37.Plummer M, editor JAGS: A program for analysis of Bayesian graphical models using Gibbs sampling; Proceedings of the 3rd international workshop on distributed statistical computing; Vienna, Austria 2003: Vienna.
- 38.Plummer M, editor rjags: Bayesian graphical models using MCMC. Rpackage version 2.1.0–10. http://CRAN.R-project.org/package=rjags2011.
- 39.Gelman A, Rubin DB. Inference from iterative simulation using multiple sequences. Statistical Science. 1992;7(4):457–72. [Google Scholar]
- 40.Cook DJ, Reeve BK, Guyatt GH, Heyland DK, Griffith LE, Buckingham L, et al. Stress Ulcer Prophylaxis in Critically III Patients: Resolving Discordant Meta-analyses. JAMA. 1996;275(4):308–14. [PubMed] [Google Scholar]
- 41.Jadad AR, Cook DJ, Browman GP. A guide to interpreting discordant systematic reviews. Canadian Medical Association Journal. 1997;156(10):1411–6. [PMC free article] [PubMed] [Google Scholar]
- 42.Linde K, Willich SN. How objective are systematic reviews? Differences between reviews on complementary medicine. Journal of the Royal Society of Medicine. 2003;96(1):17–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Peinemann F, McGauran N, Sauerland S, Lange S. Disagreement in primary study selection between systematic reviews on negative pressure wound therapy. BMC medical research methodology. 2008;8(1):41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Poolman RW, Abouali JA, Conter HJ, Bhandari M. Overlapping systematic reviews of anterior cruciate ligament reconstruction comparing hamstring autograft with bone-patellar tendon-bone autograft: why are they different? The Journal of Bone & Joint Surgery. 2007;89(7):1542–52. [DOI] [PubMed] [Google Scholar]
- 45.Hutton B, Salanti G, Caldwell DM, Chaimani A, Schmid CH, Cameron C, et al. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations. Annals of internal medicine. 2015;162(11):777–84. 10.7326/M14-2385 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(PDF)
Data Availability Statement
We have included the datasets and codes as supplementary materials to this paper.