Abstract
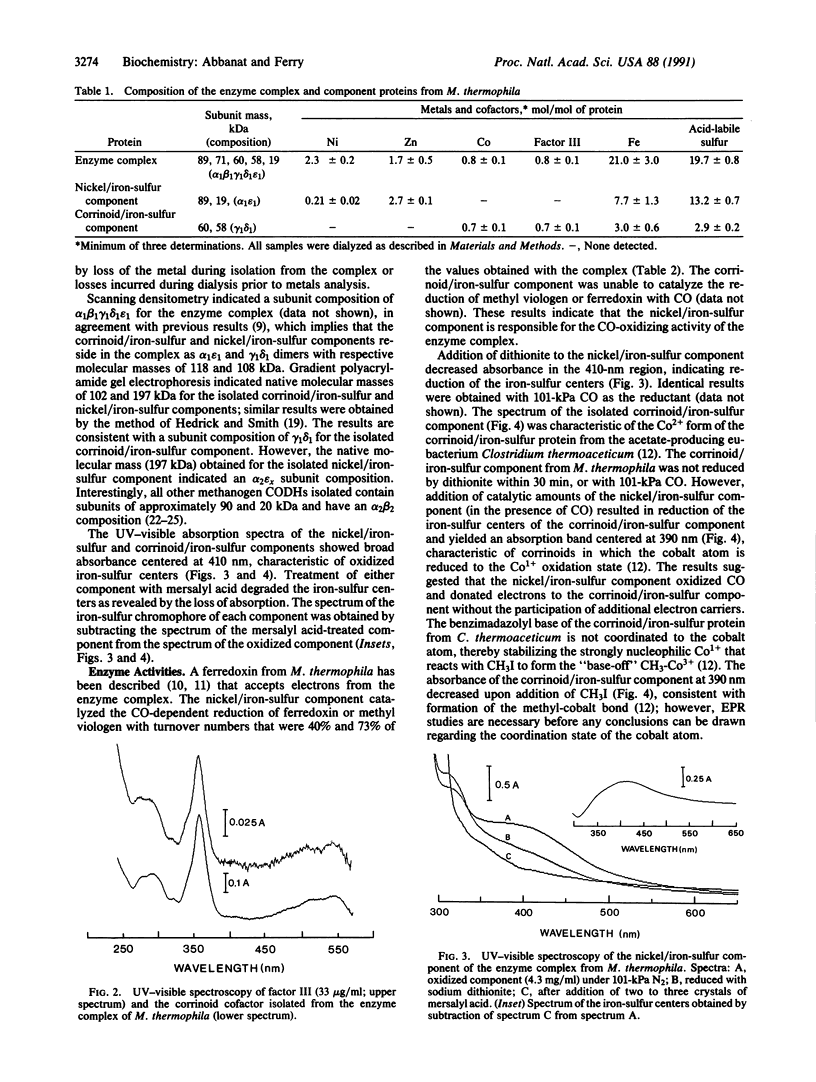
An enzyme complex was isolated from acetate-grown Methanosarcina thermophila that oxidized CO and catalyzed the synthesis or cleavage of acetyl-CoA. The complex consisted of five subunits (alpha1beta1gamma1delta1epsilon1) of 89, 71, 60, 58, and 19 kDa. The complex contained nickel, iron, acid-labile sulfide, and cobalt in a corrinoid cofactor. Two components were resolved by anion-exchange chromatography of the complex in the presence of dodecyltrimethylammonium bromide and Triton X-100: a 200-kDa nickel/iron-sulfur protein with the 89- and 19-kDa (alpha2epsilonx) subunits and a 100-kDa corrinoid/iron-sulfur protein with the 60- and 58-kDa subunits (gamma1delta1). The nickel/iron-sulfur component contained 0.21 Ni, 2.7 Zn, 7.7 Fe, and 13.2 acid-labile sulfide (per alpha1epsilon1). The corrinoid/iron-sulfur component contained 0.7 Co, 0.7 factor III [Coalpha-[alpha-(5-hydroxybenzimidazolyl)]-Cobeta-cyanocobamide], 3.0 Fe, and 2.9 acid-labile sulfide (gamma1delta1). Both components contained iron-sulfur centers. The nickel/iron-sulfur component oxidized CO and reduced methyl viologen or a ferredoxin isolated from M. thermophila. The nickel/iron-sulfur component also oxidized CO and transferred electrons to the corrinoid/iron-sulfur component, reducing the iron-sulfur and Co centers. UV-visible spectroscopy indicated that the reduced corrinoid/iron-sulfur component could be methylated with CH3I. The results suggest that the enzyme complex from M. thermophila contained at least two enzyme components, each with a specific function. The properties of the component enzymes support a mechanism proposed for acetyl-CoA synthesis (or cleavage) by the enzyme complex.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abbanat D. R., Ferry J. G. Synthesis of acetyl coenzyme A by carbon monoxide dehydrogenase complex from acetate-grown Methanosarcina thermophila. J Bacteriol. 1990 Dec;172(12):7145–7150. doi: 10.1128/jb.172.12.7145-7150.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beinert H. Semi-micro methods for analysis of labile sulfide and of labile sulfide plus sulfane sulfur in unusually stable iron-sulfur proteins. Anal Biochem. 1983 Jun;131(2):373–378. doi: 10.1016/0003-2697(83)90186-0. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- DeMoll E., Grahame D. A., Harnly J. M., Tsai L., Stadtman T. C. Purification and properties of carbon monoxide dehydrogenase from Methanococcus vannielii. J Bacteriol. 1987 Sep;169(9):3916–3920. doi: 10.1128/jb.169.9.3916-3920.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DiMarco A. A., Bobik T. A., Wolfe R. S. Unusual coenzymes of methanogenesis. Annu Rev Biochem. 1990;59:355–394. doi: 10.1146/annurev.bi.59.070190.002035. [DOI] [PubMed] [Google Scholar]
- Grahame D. A., Stadtman T. C. Carbon monoxide dehydrogenase from Methanosarcina barkeri. Disaggregation, purification, and physicochemical properties of the enzyme. J Biol Chem. 1987 Mar 15;262(8):3706–3712. [PubMed] [Google Scholar]
- Harder S. R., Lu W. P., Feinberg B. A., Ragsdale S. W. Spectroelectrochemical studies of the corrinoid/iron-sulfur protein involved in acetyl coenzyme A synthesis by Clostridium thermoaceticum. Biochemistry. 1989 Nov 14;28(23):9080–9087. doi: 10.1021/bi00449a019. [DOI] [PubMed] [Google Scholar]
- Hedrick J. L., Smith A. J. Size and charge isomer separation and estimation of molecular weights of proteins by disc gel electrophoresis. Arch Biochem Biophys. 1968 Jul;126(1):155–164. doi: 10.1016/0003-9861(68)90569-9. [DOI] [PubMed] [Google Scholar]
- Jetten M. S., Stams A. J., Zehnder A. J. Purification and characterization of an oxygen-stable carbon monoxide dehydrogenase of Methanothrix soehngenii. Eur J Biochem. 1989 May 1;181(2):437–441. doi: 10.1111/j.1432-1033.1989.tb14744.x. [DOI] [PubMed] [Google Scholar]
- Krzycki J. A., Zeikus J. G. Characterization and purification of carbon monoxide dehydrogenase from Methanosarcina barkeri. J Bacteriol. 1984 Apr;158(1):231–237. doi: 10.1128/jb.158.1.231-237.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ladapo J., Whitman W. B. Method for isolation of auxotrophs in the methanogenic archaebacteria: role of the acetyl-CoA pathway of autotrophic CO2 fixation in Methanococcus maripaludis. Proc Natl Acad Sci U S A. 1990 Aug;87(15):5598–5602. doi: 10.1073/pnas.87.15.5598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lovley D. R., White R. H., Ferry J. G. Identification of methyl coenzyme M as an intermediate in methanogenesis from acetate in Methanosarcina spp. J Bacteriol. 1984 Nov;160(2):521–525. doi: 10.1128/jb.160.2.521-525.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu W. P., Harder S. R., Ragsdale S. W. Controlled potential enzymology of methyl transfer reactions involved in acetyl-CoA synthesis by CO dehydrogenase and the corrinoid/iron-sulfur protein from Clostridium thermoaceticum. J Biol Chem. 1990 Feb 25;265(6):3124–3133. [PubMed] [Google Scholar]
- Nelson M. J., Ferry J. G. Carbon monoxide-dependent methyl coenzyme M methylreductase in acetotrophic Methosarcina spp. J Bacteriol. 1984 Nov;160(2):526–532. doi: 10.1128/jb.160.2.526-532.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ragsdale S. W., Clark J. E., Ljungdahl L. G., Lundie L. L., Drake H. L. Properties of purified carbon monoxide dehydrogenase from Clostridium thermoaceticum, a nickel, iron-sulfur protein. J Biol Chem. 1983 Feb 25;258(4):2364–2369. [PubMed] [Google Scholar]
- Ragsdale S. W., Lindahl P. A., Münck E. Mössbauer, EPR, and optical studies of the corrinoid/iron-sulfur protein involved in the synthesis of acetyl coenzyme A by Clostridium thermoaceticum. J Biol Chem. 1987 Oct 15;262(29):14289–14297. [PubMed] [Google Scholar]
- Ragsdale S. W., Wood H. G., Antholine W. E. Evidence that an iron-nickel-carbon complex is formed by reaction of CO with the CO dehydrogenase from Clostridium thermoaceticum. Proc Natl Acad Sci U S A. 1985 Oct;82(20):6811–6814. doi: 10.1073/pnas.82.20.6811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raybuck S. A., Bastian N. R., Orme-Johnson W. H., Walsh C. T. Kinetic characterization of the carbon monoxide-acetyl-CoA (carbonyl group) exchange activity of the acetyl-CoA synthesizing CO dehydrogenase from Clostridium thermoaceticum. Biochemistry. 1988 Oct 4;27(20):7698–7702. doi: 10.1021/bi00420a019. [DOI] [PubMed] [Google Scholar]
- Raybuck S. A., Ramer S. E., Abbanat D. R., Peters J. W., Orme-Johnson W. H., Ferry J. G., Walsh C. T. Demonstration of carbon-carbon bond cleavage of acetyl coenzyme A by using isotopic exchange catalyzed by the CO dehydrogenase complex from acetate-grown Methanosarcina thermophila. J Bacteriol. 1991 Jan;173(2):929–932. doi: 10.1128/jb.173.2.929-932.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stupperich E., Steiner I., Rühlemann M. Isolation and analysis of bacterial cobamides by high-performance liquid chromatography. Anal Biochem. 1986 Jun;155(2):365–370. doi: 10.1016/0003-2697(86)90447-1. [DOI] [PubMed] [Google Scholar]
- Terlesky K. C., Barber M. J., Aceti D. J., Ferry J. G. EPR properties of the Ni-Fe-C center in an enzyme complex with carbon monoxide dehydrogenase activity from acetate-grown Methanosarcina thermophila. Evidence that acetyl-CoA is a physiological substrate. J Biol Chem. 1987 Nov 15;262(32):15392–15395. [PubMed] [Google Scholar]
- Terlesky K. C., Ferry J. G. Ferredoxin requirement for electron transport from the carbon monoxide dehydrogenase complex to a membrane-bound hydrogenase in acetate-grown Methanosarcina thermophila. J Biol Chem. 1988 Mar 25;263(9):4075–4079. [PubMed] [Google Scholar]
- Terlesky K. C., Ferry J. G. Purification and characterization of a ferredoxin from acetate-grown Methanosarcina thermophila. J Biol Chem. 1988 Mar 25;263(9):4080–4082. [PubMed] [Google Scholar]
- Terlesky K. C., Nelson M. J., Ferry J. G. Isolation of an enzyme complex with carbon monoxide dehydrogenase activity containing corrinoid and nickel from acetate-grown Methanosarcina thermophila. J Bacteriol. 1986 Dec;168(3):1053–1058. doi: 10.1128/jb.168.3.1053-1058.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woese C. R. Bacterial evolution. Microbiol Rev. 1987 Jun;51(2):221–271. doi: 10.1128/mr.51.2.221-271.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]