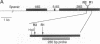Abstract
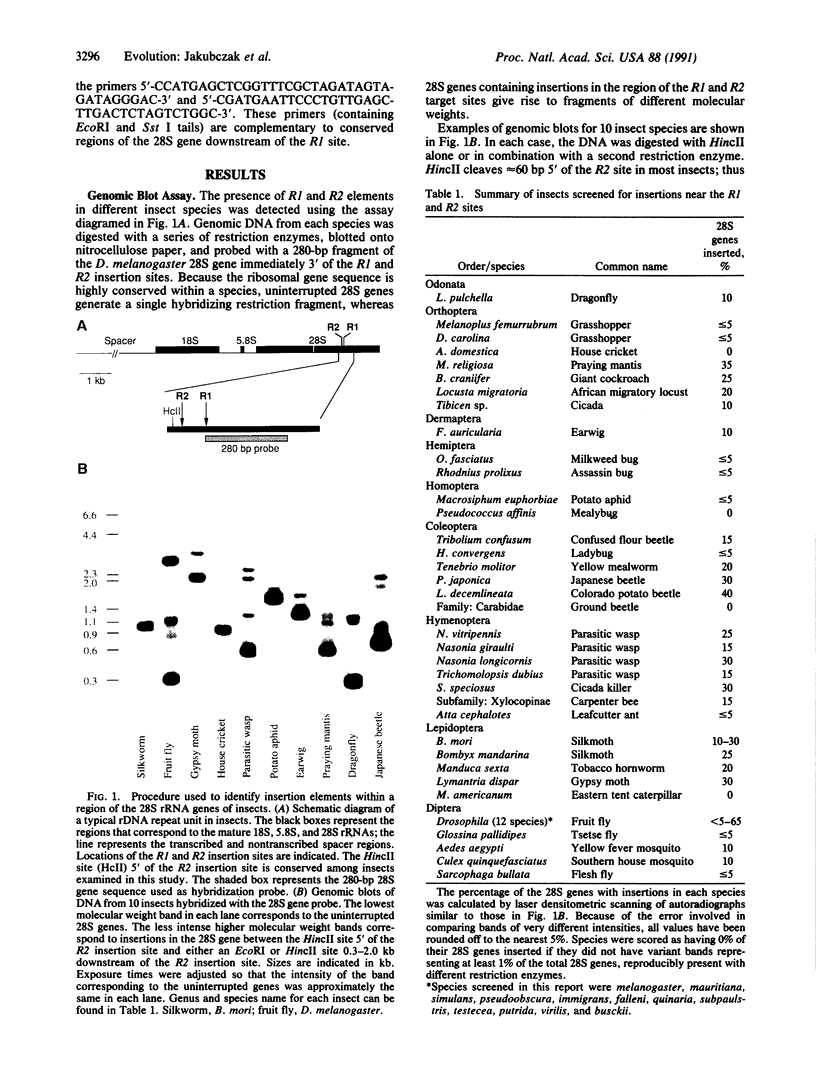
A large number of insect species have been screened for the presence of the retrotransposable elements R1 and R2. These elements integrate independently at specific sites in the 28S rRNA genes. Genomic blots indicated that 43 of 47 insect species from nine orders contained insertions, ranging in frequency from a few percent to greater than 50% of the 28S genes. Sequence analysis of these insertions from 8 species revealed 22 elements, 21 of which corresponded to R1 or R2 elements. Surprisingly, many species appeared to contain highly divergent copies of R1 and R2 elements. For example, a parasitic wasp contained at least four families of R1 elements; the Japanese beetle contained at least five families of R2 elements. The presence of these retrotransposable elements throughout Insecta and the observation that single species can harbor divergent families within its rRNA-encoding DNA loci present interesting questions concerning the age of these elements and the possibility of cross-species transfer.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Back E., Van Meir E., Müller F., Schaller D., Neuhaus H., Aeby P., Tobler H. Intervening sequences in the ribosomal RNA genes of Ascaris lumbricoides: DNA sequences at junctions and genomic organization. EMBO J. 1984 Nov;3(11):2523–2529. doi: 10.1002/j.1460-2075.1984.tb02167.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boeke J. D., Corces V. G. Transcription and reverse transcription of retrotransposons. Annu Rev Microbiol. 1989;43:403–434. doi: 10.1146/annurev.mi.43.100189.002155. [DOI] [PubMed] [Google Scholar]
- Browne M. J., Read C. A., Roiha H., Glover D. M. Site specific insertion of a type I rDNA element into a unique sequence in the Drosophila melanogaster genome. Nucleic Acids Res. 1984 Dec 11;12(23):9111–9122. doi: 10.1093/nar/12.23.9111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burke W. D., Calalang C. C., Eickbush T. H. The site-specific ribosomal insertion element type II of Bombyx mori (R2Bm) contains the coding sequence for a reverse transcriptase-like enzyme. Mol Cell Biol. 1987 Jun;7(6):2221–2230. doi: 10.1128/mcb.7.6.2221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dawid I. B., Rebbert M. L. Nucleotide sequences at the boundaries between gene and insertion regions in the rDNA of Drosophilia melanogaster. Nucleic Acids Res. 1981 Oct 10;9(19):5011–5020. doi: 10.1093/nar/9.19.5011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jakubczak J. L., Xiong Y., Eickbush T. H. Type I (R1) and type II (R2) ribosomal DNA insertions of Drosophila melanogaster are retrotransposable elements closely related to those of Bombyx mori. J Mol Biol. 1990 Mar 5;212(1):37–52. doi: 10.1016/0022-2836(90)90303-4. [DOI] [PubMed] [Google Scholar]
- Jamrich M., Miller O. L., Jr The rare transcripts of interrupted rRNA genes in Drosophila melanogaster are processed or degraded during synthesis. EMBO J. 1984 Jul;3(7):1541–1545. doi: 10.1002/j.1460-2075.1984.tb02008.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kerrebrock A. W., Srivastava R., Gerbi S. A. Isolation and characterization of ribosomal DNA variants from Sciara coprophila. J Mol Biol. 1989 Nov 5;210(1):1–13. doi: 10.1016/0022-2836(89)90286-6. [DOI] [PubMed] [Google Scholar]
- Kidd S. J., Glover D. M. Drosophila melanogaster ribosomal DNA containing type II insertions is variably transcribed in different strains and tissues. J Mol Biol. 1981 Oct 5;151(4):645–662. doi: 10.1016/0022-2836(81)90428-9. [DOI] [PubMed] [Google Scholar]
- Long E. O., Dawid I. B. Expression of ribosomal DNA insertions in Drosophila melanogaster. Cell. 1979 Dec;18(4):1185–1196. doi: 10.1016/0092-8674(79)90231-9. [DOI] [PubMed] [Google Scholar]
- Nur U., Werren J. H., Eickbush D. G., Burke W. D., Eickbush T. H. A "selfish" B chromosome that enhances its transmission by eliminating the paternal genome. Science. 1988 Apr 22;240(4851):512–514. doi: 10.1126/science.3358129. [DOI] [PubMed] [Google Scholar]
- Ochman H., Gerber A. S., Hartl D. L. Genetic applications of an inverse polymerase chain reaction. Genetics. 1988 Nov;120(3):621–623. doi: 10.1093/genetics/120.3.621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paskewitz S. M., Collins F. H. Site-specific ribosomal DNA insertion elements in Anopheles gambiae and A. arabiensis: nucleotide sequence of gene-element boundaries. Nucleic Acids Res. 1989 Oct 25;17(20):8125–8133. doi: 10.1093/nar/17.20.8125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rae P. M., Kohorn B. D., Wade R. P. The 10 kb Drosophila virilis 28S rDNA intervening sequence is flanked by a direct repeat of 14 base pairs of coding sequence. Nucleic Acids Res. 1980 Aug 25;8(16):3491–3504. doi: 10.1093/nar/8.16.3491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roiha H., Miller J. R., Woods L. C., Glover D. M. Arrangements and rearrangements of sequences flanking the two types of rDNA insertion in D. melanogaster. Nature. 1981 Apr 30;290(5809):749–753. doi: 10.1038/290749a0. [DOI] [PubMed] [Google Scholar]
- Smith V. L., Beckingham K. The intron boundaries and flanking rRNA coding sequences of Calliphora erythrocephala rDNA. Nucleic Acids Res. 1984 Feb 10;12(3):1707–1724. doi: 10.1093/nar/12.3.1707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tautz D., Hancock J. M., Webb D. A., Tautz C., Dover G. A. Complete sequences of the rRNA genes of Drosophila melanogaster. Mol Biol Evol. 1988 Jul;5(4):366–376. doi: 10.1093/oxfordjournals.molbev.a040500. [DOI] [PubMed] [Google Scholar]
- Wellauer P. K., Dawid I. B., Tartof K. D. X and Y chromosomal ribosomal DNA of Drosophila: comparison of spacers and insertions. Cell. 1978 Jun;14(2):269–278. doi: 10.1016/0092-8674(78)90113-7. [DOI] [PubMed] [Google Scholar]
- Xiong Y. E., Eickbush T. H. Functional expression of a sequence-specific endonuclease encoded by the retrotransposon R2Bm. Cell. 1988 Oct 21;55(2):235–246. doi: 10.1016/0092-8674(88)90046-3. [DOI] [PubMed] [Google Scholar]
- Xiong Y., Burke W. D., Jakubczak J. L., Eickbush T. H. Ribosomal DNA insertion elements R1Bm and R2Bm can transpose in a sequence specific manner to locations outside the 28S genes. Nucleic Acids Res. 1988 Nov 25;16(22):10561–10573. doi: 10.1093/nar/16.22.10561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiong Y., Eickbush T. H. Origin and evolution of retroelements based upon their reverse transcriptase sequences. EMBO J. 1990 Oct;9(10):3353–3362. doi: 10.1002/j.1460-2075.1990.tb07536.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiong Y., Eickbush T. H. Similarity of reverse transcriptase-like sequences of viruses, transposable elements, and mitochondrial introns. Mol Biol Evol. 1988 Nov;5(6):675–690. doi: 10.1093/oxfordjournals.molbev.a040521. [DOI] [PubMed] [Google Scholar]
- Xiong Y., Eickbush T. H. The site-specific ribosomal DNA insertion element R1Bm belongs to a class of non-long-terminal-repeat retrotransposons. Mol Cell Biol. 1988 Jan;8(1):114–123. doi: 10.1128/mcb.8.1.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanisch-Perron C., Vieira J., Messing J. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene. 1985;33(1):103–119. doi: 10.1016/0378-1119(85)90120-9. [DOI] [PubMed] [Google Scholar]