Abstract
White adipose tissue expansion is associated with both hypertrophy and hyperplasia of adipocytes. Exercise training results in adipocyte hypotrophy by activating lipolysis, but it is poorly understood whether exercise regulates adipogenesis by altering adipogenic gene expression. The purpose of this study was to evaluate the effect of a single bout of swimming exercise on adipogenic gene expression in white adipose tissue (WAT). Male C57BL/6J mice were divided into two groups: a sedentary control group and a 120-minute swimming exercise group. Immediately after acute exercise, adipogenic gene expression in WAT was analysed by RT-PCR, and tdTomato positive cells in WAT from UCP1-cre-tdTomato mice were observed under a confocal microscope. In epididymal white adipose tissue (eWAT), PPARγ2 and C/EBPα expression at the mRNA level was significantly decreased with high induction of Wnt10b and KLFs (KLF2, KLF3, KLF7, KLF6, KLF9 and KLF15), whereas PPARγ2, not C/EBPα, was decreased with high induction of Wnt6 and KLFs (KLF2, KLF3, KLF7, KLF6 and KLF9) in inguinal white adipose tissue (iWAT) after acute exercise. The expression of C/EBPβ and C/EBPδ was upregulated in both WATs with a high level of PGC-1α expression. Expression level of UCP1 was increased only in adipocytes of eWAT, while beige cell specific gene expression was comparable between groups and tdTomato positive cells were not found in WAT of UCP1-cre-tdTomato reporter mouse immediately after acute exercise. These results suggest that acute exercise suppresses adipogenic gene expression and may regulate thermogenesis by activating C/EBPβ, PGC-1α and UCP1 in WAT.
Keywords: Acute exercise, PPARγ2, C/EBPs, Wnts, KLFs, White adipose tissue
INTRODUCTION
White adipose tissue (WAT) is a specialized organ for lipid storage and regulates whole body metabolism as a major endocrine organ secreting adipokines [1]. WAT has high plasticity since it expands its mass by increasing the size of existing adipocytes (hypertrophy) and/or forming new adipocytes (hyperplasia) in response to the demand for additional lipid storage [2, 3]. In mouse, epididymal white adipose tissue (eWAT) expansion under a high fat diet condition, which contributes to the development of metabolic syndromes such as obesity, insulin resistance, type 2 diabetes and cardiovascular disease [1], is preferentially associated with adipocyte hyperplasia, whereas inguinal white adipose tissue (iWAT) expansion mainly depends on adipocyte hypertrophy [4], supporting that inhibition of adipogenesis is critical for the treatment of metabolic syndromes.
Peroxisome proliferator-activated receptor γ (PPARγ) and CCAAT/enhancer-binding proteins (C/EBPs) have received much attention as master regulators for adipogenesis [5]. PPARγ is a member of the nuclear receptor superfamily and is expressed as two isoforms, PPARγ1 and PPARγ2, depending on different promoter usage and alternative splicing [6, 7]. While PPARγ1 is expressed in multiple tissues, PPARγ2 expression is restricted in adipose tissue [8, 9]. PPAR2 deficient mouse shows impairment of adipose tissue development with adipocyte death and deterioration of insulin sensitivity [10, 11], whereas treatment with PPARγ activators results in the development of a large number of small adipocytes in obese rat WAT with increased fat mass [12, 13].
C/EBPs belong to the large family of basic leucine zipper transcription factors and have six members (C/EBPα, C/EBPβ, C/EBPγ, C/EBPδ, C/EBPε and CHOP) that form hetero- or homo-dimers to bind the same C/EBP consensus sequence. Three of them (C/EBPα, C/EBPβ and C/EBPδ) are expressed in both WAT and brown adipose tissue (BAT) [5, 14]. C/EBPα, either alone or in combination with PPARγ, promotes adipogenesis in mouse fibroblastic cells since they regulate each other’s expression [15, 16]. Additionally, C/EBP null mice have a perinatal lethal phenotype due to defective liver gluconeogenesis and subsequent hypoglycaemia [17]. Restoration of hepatic C/EBPα level in this mouse causes an absence of WAT except the inguinal mammary gland [18]. C/EBPα and PPARγ2 are induced by C/EBPβ alone or with C/EBP in 3T3-L1 preadipocytes to promote adipogenesis [19, 20]. However, C/EBPα expression is attenuated only in the fibroblast from C/EBPβ and C/EBPδ double knockout (KO) mice, not from C/EBPβ or C/EBPδ null mice [21]. In addition, PPARγ expression is markedly decreased in the fibroblast from C/EBPβ and C/EBPδ double KO and C/EBP null mice. In contrast, the expression of C/EBPα and PPARγ in the WAT of three genotypes is compatible, although WAT weight is significantly reduced only in C/EBPβ and C/EBPδ double KO mice, not in C/EBPβ or C/EBPδ null mice [21]. These results indicate that C/EBPβ or C/EBPδ is not indispensable for adipogenesis, but C/EBPβ and C/EBPδ act synergistically to induce adipogenesis.
Adipogenesis is also regulated by Wingless-type MMTV integration site family members (WNTs), Krüppel-like factors (KLFs), GATA binding protein (GATA) 2, GATA3 and sterol regulatory element binding transcription factor 1 (SREBP1c). WNTs are secreted glycoproteins that act though autocrine and paracrine mechanisms to regulate the development of many cell types. Wnt signalling has been reported to inhibit adipogenesis by suppressing PPARγ and C/EBPα expression [22, 23]. The Krüppel-like factors (KLFs) are a large family of C2H2 zinc-finger proteins and play an important role in adipogenesis and obesity as positive or negative regulators [24]. In particular, KLF6, KLF9 and KLF15 promote adipogenesis by regulating PPARγ and/or C/EBPs expression [25–27]. In contrast, KLF2, KLF3 and KLF7 are anti-adipogenic factors. KLF3, associated with C/EBPα, inhibits adipocyte differentiation, and KLF3 deficient mouse has a significant reduction in WAT mass [28]. Overexpression of KLF2 or KLF7 inhibits adipogenesis, with a reduction of PPARγ and C/EBPα expression [29, 30]. Forced expression of GATA binding protein (GATA) 2 and GATA3 in preadipocytes inhibits adipogenesis by suppressing PPARγ and forming a protein complex with C/EBPα and C/EBPβ [31, 32]. SREBP1c, as a pro-adipogenic transcriptional factor, induces lipid biosynthesis by insulin [33, 34]. Although SREBP1c null mouse has a normal fat amount [35], adipose tissue specific SREBP1c overexpression induces severe lipodystrophy [36].
Endurance exercise training is well known to reduce WAT mass in obesity by using fatty acid (FA) from adipose tissue as fuel [37, 38]. FA mobilization from WAT is regulated by the activity of hormone-sensitive lipase (HSL), monoglyceride lipase (MGL) and adipose triglyceride lipase (ATGL) [39]. Since exercise promotes lipolysis from adipocytes by activating HSL and ATGL [40, 41], exercise-induced hypotrophy of adipocytes is, at least in part, responsible for the reduction of WAT mass in obese individuals. However, it remains uncertain whether endurance exercise regulates adipogenesis by altering adipogenic gene expression. Therefore, we evaluated the effect of a single bout of swimming exercise on the alteration of adipogenic gene expression in WAT. In this study, we show that acute exercise regulates expression levels of PPARγ2, C/EBPs, Wnts and KLFs in WAT.
MATERIALS AND METHODS
Mice
C57BL/6J male mice were purchased from Vital River Laboratory Animal Technology. Co. Ltd, (Beijing, China). UCP1-cre (stock number 024670) and tdTomato reporter mice (stock number 007909) were from Jackson Laboratory (Bar Harbor, ME). UCP1-cre mouse was crossed with tdTomato mouse that has a loxP-flanked stop cassette. Mice were housed in an environmentally controlled facility certified by the Office of Laboratory Animal Welfare with a 12 hour light/dark cycle and had free access to food and water. After sacrifice, eWAT and iWAT were snap frozen in liquid nitrogen and stored at −80°C for molecular analysis. Animal studies were approved by the Institutional Animal Care and Use Committee of the Institute of Zoology, Chinese Academy of Sciences.
Acute exercise protocol
At 10 weeks of age, C57BL/6J male mice were randomly divided into two groups: a sedentary control group (Sed) and a swimming exercise group (Exe). Mice in Exe (N=8) were adapted to swimming for 10 min for 2 days in a row to avoid water stress and then 1 week later were subjected to swimming exercise with lead fish sinkers (5% of body weight) on the tail for 2 h in plastic barrels filled with water (32+1°C) [42, 43]. Mice in Sed (N=8) were kept in barrels without water for 2 h. After exercise, mice were sacrificed immediately.
Adipocytes and stromal vascular fraction isolation
Adipocytes and the stromal vascular fraction (SVF) were isolated from WATs as described previously [44]. WATs in DMEM/F-12 media (Invitrogen) containing 1.0% BSA were chopped with surgical scissors then digested with 0.2% collagenase type 2 (Invitrogen) for 25 min at 37°C. After filtering the mixture through 100 μM mesh to remove undigested fragments, the filtrate was centrifuged at 300 x g rpm for 5 min at 4°C. Separated adipocytes were collected with a disposable transfer pipette, and washed 3 times with DMEM/F-12 media. The SVF pellet was resuspended and washed 3 times with DMEM/F-12 media. After isolation, samples were snap frozen in liquid nitrogen for RNA isolation.
Cold exposure
C57BL/6J male mice (10 weeks old) were placed in a cold room (4°C) for 2 h without food to analyse thermogenic gene expression in WATs (N=8). Control mice were placed at room temperature (RT) and fasted for 2 h (N=8). After sacrifice, eWAT and iWAT were removed and snap frozen in liquid nitrogen for RNA isolation. For identifying UCP1 expressing cells in WAT, 10-week old UCP1-cre-tdTomato reporter mice were randomly divided into three groups; Sed, Exe, and a cold exposure group (8°C for 72 h) as a positive control (N=3 in each group). After sacrifice, eWAT and iWAT were removed then formalin fixed for histological analyses.
RT-PCR
Total RNA was extracted from eWAT, iWAT, adipocytes and SVF samples collected from Con and Exe using TRIzol isolation reagent (Invitrogen, Carlsbad, CA) according to the manufacturer’s instructions. RNA concentration was spectrophotometrically determined using NanoDrop (Thermo Scientific). Two micrograms of RNA from WATs or SVF and 150 ng of RNA from adipocytes were reverse transcribed using murine leukaemia virus reverse transcriptase and oligo (dT)16 primer. The resulting cDNAs from tissue samples were assayed in duplicate. qRT-PCR was conducted using 2X SYBR green PCR master mix on a real-time PCR system (Applied Biosystems). Gene expression data were normalized to the housekeeping gene cyclophilin A and analysed using the delta delta cycle threshold method (ΔΔCt) [45]. Primer sets are described in Table 1.
TABLE 1.
Sequences of primers for RT-PCR.
| Transcript | Primer sequence (5’-3’) |
|---|---|
| PPARγ2 | F: TCGCTGATGCACTGCCTATG R: GAGAGGTCCACAGAGCTGATT |
| C/EBPα | F: GCGGGAACGCAACAACATC R: GTCACTGGTCAACTCCAGCAC |
| C/EBPβ | F: TGACGCAACACACGTGTAACTG R: AACAACCCCGCAGGAACAT |
| C/EBPδ | F: CGTTGCCTCTACTTTCCTCAA R: GGTTAAGCCCGCAAACATTAC |
| Wnt6 | F: GCGGAGACGATGTGGACTTC R: ATGCACGGATATCTCCACGG |
| Wnt10a | F: ACTCCGACCTGGTCTACTTT R: ACCCGTGCTGCTCTTATTG |
| Wnt10b | F: GGCTGTAACCACGACAT R: GGCTGTAACCACGACAT |
| KLF2 | F: AATGACTCTGCCACCAGTTC R: GACCCGAGGGAAATAAGTCAAT |
| KLF3 | F: CTACACAGGAAACGCCACAT R: GGAGAGAGAGAGAGAGAAAGAGAG |
| KLF4 | F: TCAACAGAGCCAACTCCTAATC R: AAGCAGATACCCTCTCAAAGC |
| KLF5 | F: CTGCCACTCTGCCAGTTAAT R: GAAGTGGATACGTCGCTTCTC |
| KLF6 | F: GGAGAAGAAGGAGGAATCAGAAC R: CAGAGTTCAGGCTATTGGTCTC |
| KLF7 | F: CACAGGTGAGAAGCCTTACAA R: ACCTGTGTGTTTCCTGTAGTG |
| KLF9 | F: GCTGACTCCGACTTCCAATAC R: GCTCCCAGTCCCTAATGTTAAA |
| KLF15 | F: GCAGTGGAGGTATTGGAGATAG R: AGAAATTCAGGGAAGCAGAAATG |
| GATA2 | F: GGAGAAAGGAGTAGGCAAGAAG R: CCCAAGAACACAAATAGCACAC |
| GATA3 | F: AGCTGCCAGATAGCATGAAG R: TAGGGCGGATAGGTGGTAAT |
| SREBP1c | F: CCTGCTTGGCTCTTCTCTTT R: GTCAGCTTGTTTGCGATGTC |
| PRDM16 | F: CAGCACGGTGAAGCCATTC R: GCGTGCATCCGCTTGTG |
| PGC-1a | F: GTCAACAGCAAAAGCCACAA R: TCTGGGGTCAGAGGAAGAGA |
| UCP1 | F: GAGGTCGTGAAGGTCAGAATG R: AAGCTTTCTGTGGTGGCTATAA |
| TMEM26 | F: ACCCTGTCATCCCACAGAG R: TGTTTGGTGGAGTCCTAAGGTC |
| TH | F: AGGGGCCTTTCCCAAAGTTC R: ATCAAAGGGTCCAGCCACAC |
| Dio2 | F: GCCATGCCATGCAGTTAGGA R: TTGGGAATTCGGGGCTACAG |
| CD137 | F: CGTGCAGAACTCCTGTGATAAC R: GTCCACCTATGCTGGAGAAGG |
| HOXC8 | F: CTTCGTCAACCCCCTGTTTTC R: GTCTTGGACGTGGTGCGAG |
| TBX1 | F: GGCAGGCAGACGAATGTTC R: TTGTCATCTACGGGCACAAAG |
| Cyclophilin A | F: TCCAAAGACAGCAGAAAACTTTCG R: TCTTCTTGCTGGTCTTGCCATTCC |
Immunohistochemistry
Both eWAT and iWAT were removed from UCP1-cre-tdTomato reporter mouse, fixed with 1% formalin for 12 h at 4°C, and then washed three times. WATs were minced into pieces (~50 mm) using a scalpel, permeabilized with 1% Triton-X 100 for 6 h at 4°C, blocked with 5% goat serum for 1 h at RT and then incubated with 2 μg of Bodipy (Invitrogen, Carlsbad, CA) for 2 h at RT. After three washes with PBS, images were acquired with a Zeiss LSM780 laser scanning confocal microscope (Carl Zeiss).
Statistical analysis
All results are expressed as means ± standard deviation (SD). To test normality, the Shapiro-Wilk test was performed and then, depending its outcome, data were analysed using Student’s t test or the Mann-Whitney U test. Statistical significance was set at P<0.05.
RESULTS
Acute exercise modulates PPARγ2 and C/EBPs expression in WAT
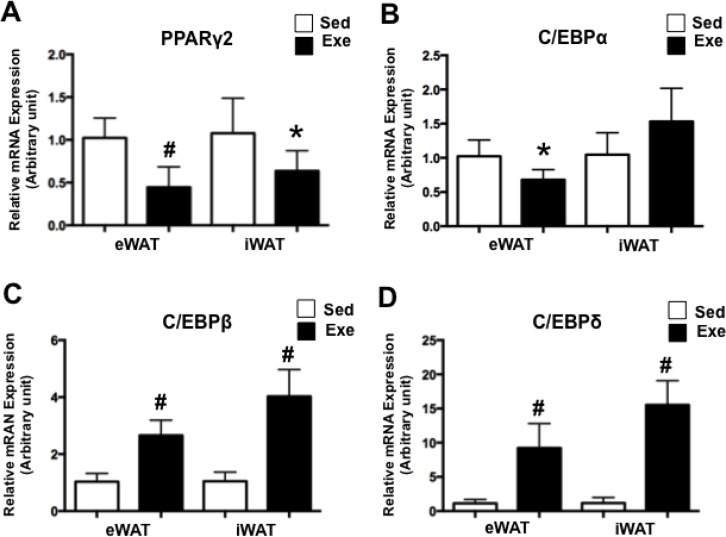
To determine whether acute exercise regulates adipogenic gene expression, we analysed expression levels of PPARγ2 and C/EBPs in WATs of Sed and Exe mice. Acute exercise markedly reduced PPARγ2 expression in eWAT (P<0.0001) and iWAT (P<0.05) (Fig. 1A). Additionally, C/EBPα expression was significantly attenuated in eWAT (P<0.05) but was unchanged in iWAT after acute exercise (Fig. 1B). Conversely, the expression levels of C/EBPβ and C/EBPδ, known to stimulate PPARγ and C/EBPα expression, were highly upregulated in both WATs after acute exercise (P<0.0001) (Fig. 1C-D). Our results indicate that acute exercise contributes to regulating adipogenic gene expression in WAT.
FIG. 1.
Expression of PPARγ2 and C/EBPs after acute exercise.
Note: The expression of PPARγ2 (A), C/EBPα (B), C/EBPβ(C) and C/EBPδ (D) was assessed by RT-PCR. Each value was normalized to cyclophilin A. Bars represent the means and error bars represent SD (N=8). *P<0.05, #P<0.001 vs. Sed.
Acute exercise regulates Wnt6 and Wnt10b expression in a fat depot-specific manner
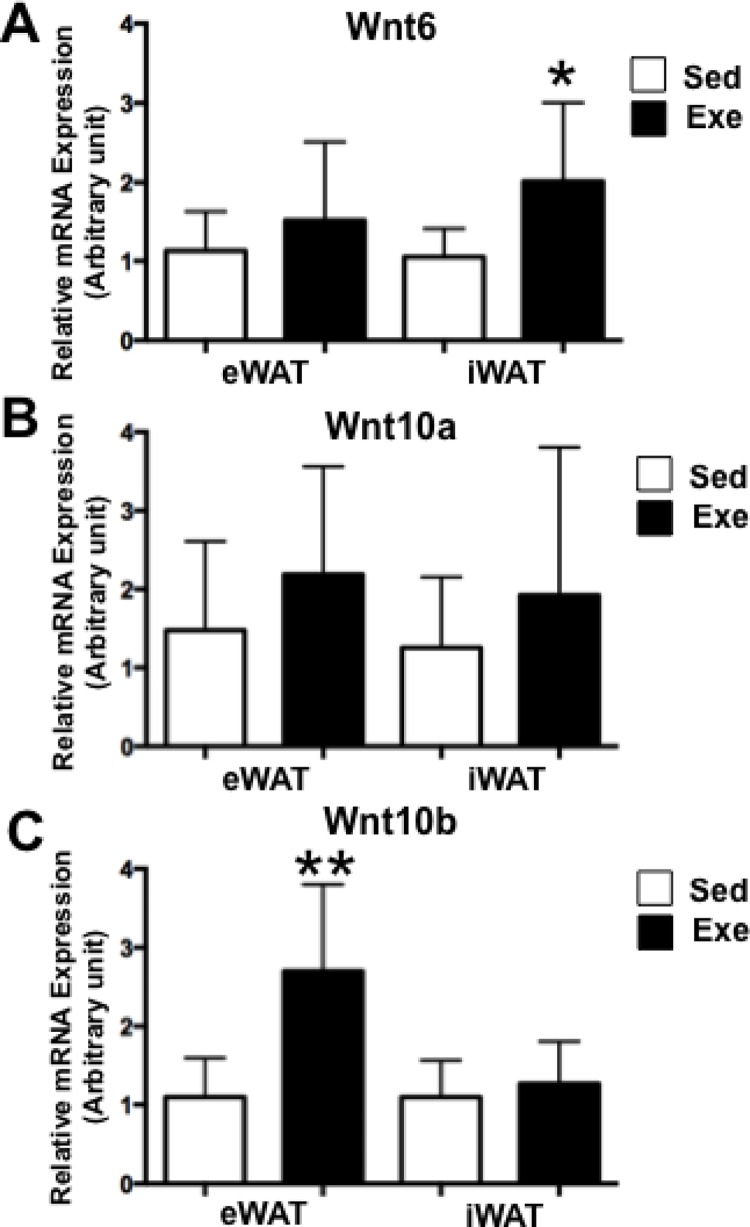
Since PPARγ2 expression was downregulated in WAT after acute exercise, we evaluated whether acute exercise leads to increased expression levels of Wnts. After acute exercise, Wnt6 expression was higher in iWAT (P<0.05) (Fig. 2A), whereas Wnt10b expression was significantly increased in eWAT (P<0.01) (Fig. 2C). However, Wnt10a expression was comparable in both WATs between groups (Fig. 2B). These results suggest that Wnt6 and Wnt10b expression is regulated by acute exercise in a fat depot-specific manner.
FIG. 2.
Wnts expression after acute exercise.
The expression of Wnt6 (A), Wnt10a (B) and Wnt10b (C) was assessed by RT-PCR. Each value was normalized to cyclophilin A. Bars represent the means and error bars represent SD (N=8). *P<0.05, **P<0.01 vs. Sed.
Acute exercise leads to alteration of KLF expression
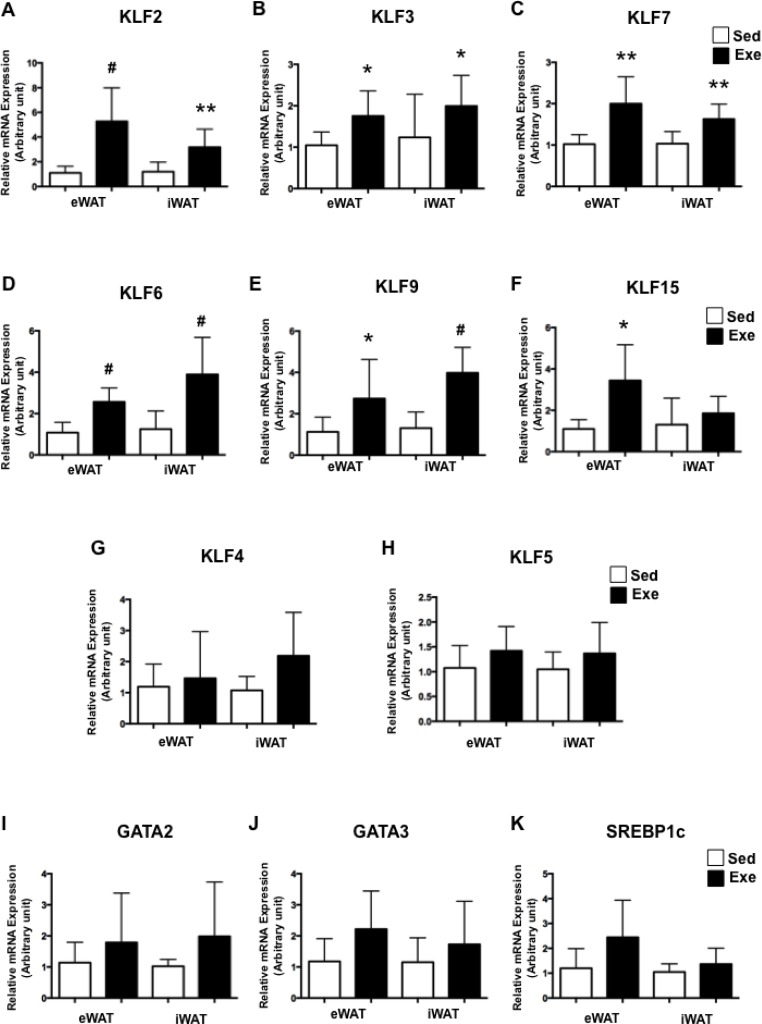
Other factors regulated during adipogenesis include KLFs, GATA2, GATA3 and SREBP1c. Immediately after swimming exercise, the expression levels of KLF2, KLF3 and KLF7, known as anti-adipogenic factors, were significantly higher in both WATs (Fig. 3A-C). Interestingly, the expression levels of pro-adipogenic KLFs such as KLF6, KLF9 and KLF15 were also significantly upregulated in WAT from Exe (Fig. 3D-F), whereas KLF4 and KLF6 expression levels were not altered after acute exercise (Fig. 3G-H). Also, the expression levels of GATA2, GATA3 and SREBP1c in WATs were not altered after acute exercise (Fig. 3I-K). These results suggest that acute exercise regulates both pro- and anti-adipogenic KLF expression.
FIG. 3.
KLFs expression after acute exercise.
The expression of KLFs (A-H), GATA2 (I) GATA3 (J) and SREBP1c (K) was assessed by RT-PCR. Each value was normalized to cyclophilin A. Bars represent the means and error bars represent SD (N=8). *P<0.05, **P<0.01, #P<0.001 vs. Sed.
The expression level of UCP1 is increased in eWAT after acute exercise
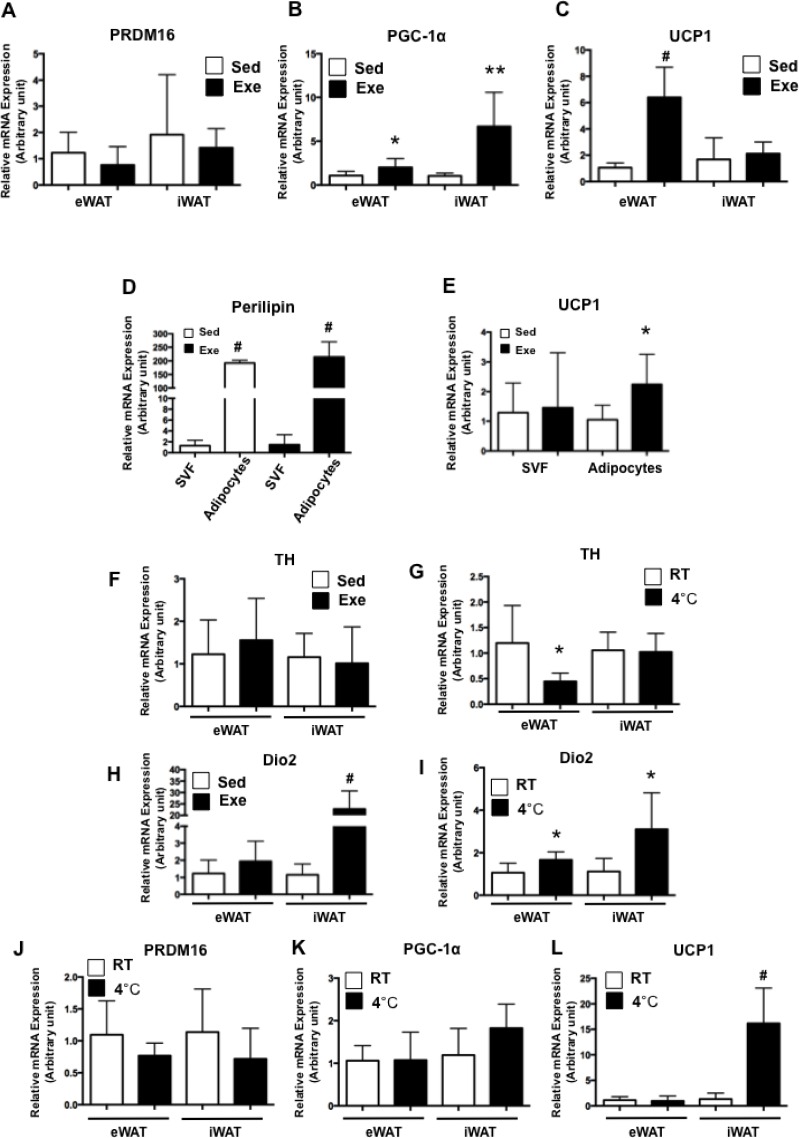
Acute exercise increased the expression level of C/EBPβ, which forms complexes with PRD1-BF-1-RIZ1 homologous domain 16 (PRDM16) and then regulates peroxisome proliferator-activated receptor-γ coactivator-1α (PGC-1α) and uncoupling protein 1 (UCP1) expression [46]. Thus, we next examined whether acute exercise induces PGC-1α and UCP1 expression in WAT. Although there was no difference in PRDM16 expression (Fig. 4A), the expression level of PGC-1α was significantly increased in both eWAT (P<0.05) and iWAT (P<0.01) after acute exercise (Fig. 4B). Interestingly, acute exercise induced expression levels of C/EBPβ and PGC-1α to a higher extent in iWAT than in eWAT; however, UCP1 expression was upregulated only in eWAT (P<0.001), not in iWAT (Fig. 4C). Next, we evaluated UCP1 expression in adipocytes and SVF from eWAT of both groups. The expression level of perilipin, a mature adipocyte marker, was significantly higher in adipocytes (Fig. 4D), indicating that adipocytes and SVF were successfully isolated. Interestingly, UCP1 upregulation was found in adipocytes (Fig. 4E), indicating that acute swimming exercise increased UCP1 expression in adipocytes of eWAT. It is suggested that swimming exercise even in warm water could activate non-shivering thermogenesis (NST) to counteract the heat loss due to high conductivity of water [47]. NST is mainly regulated by the sympathetic nervous system and thyroid hormone [48, 49]. Therefore, mice were placed in a cold room (4°C) for 2 h and then we analysed the expression levels of tyrosine hydroxylase (TH), which converts phenylalanine into dopamine and iodothyronine deiodinase type 2 (dio2), which is the enzyme responsible for the conversion of T4 to T3. After cold exposure, TH expression was significantly downregulated in eWAT; however, there was no alteration in eWAT after swimming exercise (Fig. 4F-G). The induction of dio2 was observed in both WATs after cold exposure, whereas it was upregulated only in iWAT after swimming exercise to a greater extent than after cold exposure (Fig. 4H-I). Although higher induction of dio2 after swimming exercise than cold exposure could result from combined effects of swimming exercise and NST, more than a 20-fold increase of dio2 expression after swimming exercise may suggest that its induction could be caused more by exercise than NST, since acute exercise also increases serum T3 immediately after exercise [50]. In addition, the expression level of PRDM16 and PGC-1α was not altered in WATs (Fig. 4J-K), and UCP1 expression was highly upregulated only in iWAT (Fig. 4L) after 2 h of cold exposure. These different gene expression patterns in TH, dio2, PGC-1α and UCP1 between swimming exercise and cold exposure suggest that activation of C/EBPβ mediated PGC-1α and UCP1 in eWAT after swimming exercise could be mainly the result of exercise rather than NST.
FIG. 4.
PRDM16, PGC-1α and UCP1 expression after acute exercise.
Total RNA from eWAT and iWAT was isolated and the expression of PRDM16 (A), PGC-1α (B), UCP1 (C), TH (F) and Dio2 (H) was assessed by RT-PCR. Total RNA from SVF and adipocytes from eWAT were isolated and the expression of perilipin (D) and UCP1 (E) was assessed by RT-PCR. *P<0.05, **P<0.01, #P<0.001 vs. Sed. After 2 h of cold exposure (4°C), total RNA from eWAT and iWAT was isolated and the expression of TH (G), Dio2 (I), PRDM16 (J), PGC-1α (K) and UCP1 (L) was assessed by RT-PCR. Each value was normalized to cyclophilin A. Bars represent the means and error bars represent SD (N=8). *P<0.05, #P<0.001 vs. RT.
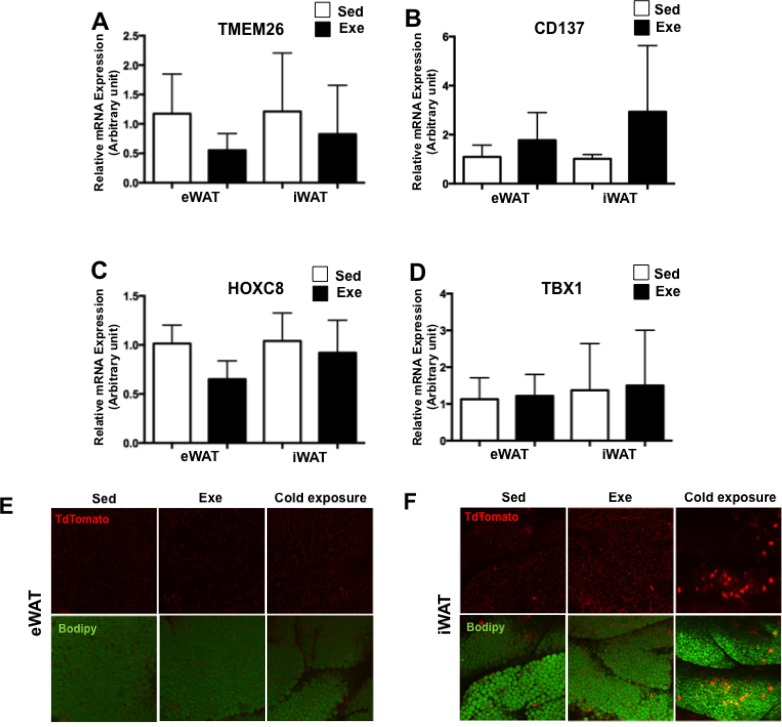
Beige cells are not found in eWAT immediately after acute exercise
UCP1 expressing cells in WAT, known as beige cells, are identified by specific markers such as transmembrane protein 26 (TMEM26), CD137, homeobox C8 (HOXC8) and t-box1 transcription factor c (TBX1) [51]. Therefore, we measured the expression levels of beige cell specific markers via qPCR to investigate whether UCP1 upregulation accompanies the recruitment of beige cells in eWAT after acute exercise. However, expression levels of TMEM26, CD137, HOXC8 and TBX1 were not altered in either WAT after acute exercise (Fig. 5A-D). For further investigation, we analysed UCP1 expression in WAT from UCP1-cre-tdTomato reporter mice after acute exercise. WATs from cold-exposed UCP1-cre-tdTomato reporter mice were used as a positive control. Consistent with mRNA expression levels of beige cell specific markers, tdTomato positive cells were not found in either WAT from UCP1-cre-tdTomato mice after acute exercise. However, tdTomato positive cells were dramatically increased in iWAT from UCP1-cre-tdTomato reporter mice at 3 days after cold exposure (Fig. 5E-F). These results indicate that beige cells are not detected in eWAT at least immediately after 2 h of single swimming exercise.
FIG. 5.
Beige cell recruitment in WATs after acute exercise.
The expression of TMEM26 (A), CD137 (B), HOXC8 (C) and TBX1 (D) was assessed by RT-PCR. Each value was normalized to cyclophilin A. Bars represent the means and error bars represent SD (N=8). *P<0.05, **P<0.01, #P<0.001 vs. Sed. tdTomato expression was detected from eWAT and iWAT of UCP1-cre-tdTomato mouse after acute exercise or cold exposure (E-F).
DISCUSSION
Exercise training is known to cause numerous beneficial effects on adipose tissue biology; however, it is not fully understood whether acute exercise contributes to regulating adipogenic gene expression. To address this, we analysed adipogenic gene expression in mouse eWAT and iWAT after a single bout of swimming exercise. The findings of the present study demonstrate that acute exercise regulated the expression levels of PPARγ2, C/EBPs, Wnts and KLFs in WAT.
The intensity, mode and duration of exercise may play a role in regulating PPARγ expression at the mRNA level. Previous studies have shown that exercise training does not increase PPARγ protein expression in WAT [52, 53]. Since exercise increases plasma FA that could function as a ligand for PPARα and PPARγ [54], the lack of changes in the PPAR expression may suggest that exercise could activate rather than express PPARs, as noted by Petridou et at [53]. However, exercise training downregulates PPARγ mRNA expression in the stromal-vascular fraction from adipose tissue [55]. We found that acute exercise also attenuated the expression level of PPARγ2 in both WATs in this study. On the other hand, C/EBPα expression was markedly decreased only in eWAT, not iWAT, after acute exercise in our study. C/EBPα may not be a critical factor for adipogenesis at least in iWAT since restoration of hepatic C/EBPα level in C/EBPα null mice does not effect the development of iWAT [18].
Wnt6, Wnt10a and Wnt10b act as negative regulators of adipogenesis [22, 23, 56, 57]. Although resistance training has been observed to increase Wnt expression in muscle and serum [58, 59], it has not been studied whether acute exercise regulates Wnt expression in WAT. Interestingly, acute exercise increased Wnt10b expression in eWAT and Wnt6 expression in iWAT, indicating that acute exercise regulates Wnts expression in a fat-depot specific manner. Future studies with genetically modified mice models are necessary to elucidate fat-depot specific Wnt function in adipogenesis. KLFs regulate adipogenesis and obesity as positive or negative regulators [24], and it is not clear whether acute exercise regulates expression of KLFs in WAT. In our study, acute exercise significantly increased the expression levels of anti-adipogenic KLFs (KLF2, KLF3 and KLF7) in both WATs. However, the expression levels of three pro-adipogenic KLFs (KLF6, KLF9 and KLF15) were also upregulated in WAT after acute exercise. Since these three KLFs regulate adipogenesis by activating PPAR and/or C/EBPs, induction of these three genes may result from the downregulation of PPARγ and C/EBPα mRNA expression as a compensatory response.
Collectively, our results suggest that acute exercise may contribute to inhibiting adipogenesis by regulating the induction of adipogenic genes such as PPARγ2, C/EBPα, Wnts and anti-adipogenic KLFs (KLF2, KLF3 and KLF7) in WAT.
In contrast with PPARγ and C/EBPα, acute exercise led to higher expression levels of C/EBPβ and C/EBPδ in both WATs. Induction of these genes in response to acute exercise may result from an acute compensatory mechanism for the reduction of PPARγ and/or C/EBPα expression, since C/EBPβ and C/EBPδ are known to induce the C/EBPα and PPARγ2 genes in preadipocytes for the development of adipogenesis [19, 20]. Another possible explanation is their various biological functions other than adipogenesis in WAT. In particular, it was recently found that PRDM16-C/EBPβ complex synergistically enhances the promoter activity of PGC-1α [46, 60]. PGC-1α plays an important role in adipogenesis and adaptive thermogenesis by regulating mitochondrial biogenesis via activating UCP1 expression and fatty acid oxidation enzymes [46, 61–64]. Our finding that acute exercise markedly increased PGC-1α expression in both WATs is consistent with a previous study showing that 2 h swimming exercise increases PGC-1 mRNA expression in visceral WAT in rat [65]. It is noteworthy that UCP1 expression in WAT is mediated by PGC-1α after acute exercise or exercise training [66]. Ringholm et al. found that UCP1 mRNA expression in eWAT peaked immediately after acute exercise and then decreased gradually to the rest level at 10 hours after exercise, whereas mRNA and protein levels of UCP1 in iWAT were upregulated only at 6 hours after acute exercise [66]. In this study, we also observed that acute exercise increased UCP1 mRNA expression in eWAT, not iWAT, immediately after acute exercise. Nonetheless, expression levels of beige cell markers, such as TMEM26, CD137, HOXC8 and TBX1, were not changed and UCP1 positive cells were not detected in eWAT of UCP1-cre-tdTomato mouse at least after acute exercise. It seems that long-term adaptation to exercise may be required to recruit UCP1 positive cells in eWAT, since beige cells in WAT are found after a week of endurance exercise training [67]. Also it may require a long time between acute exercise and sacrifice to identify UCP1 positive cells after acute exercise, as it is well established that UCP1 positive cells are recruited in visceral fat in animals subjected to exercise training. Taken together, our results show that C/EBPβ expression in response to acute exercise may be associated with PGC-1α and UCP1 expression in eWAT.
Swimming exercise even in warm water could activate non-shivering thermogenesis to counteract the heat loss due to the high conductivity of water [47]. In our observations, the expression patterns of TH, dio2, PGC-1α and UCP1 in eWATs after 2 h of swimming exercise were not identical to those in eWAT after 2 h of cold exposure. Also, induction of dio2 in iWAT was dramatically higher after acute exercise than after cold exposure, and this is consistent with a previous study showing that a significant increase in serum T3 occurs immediately after acute exercise [50]. These results indicate that the activation of C/EBPβ mediated PGC-1α and UCP1 in eWAT may be caused mainly by swimming exercise rather than NST.
Since changes in gene expression at the mRNA level do not reflect changes at the protein level, the protein levels of adipogenic genes after acute exercise need to be analysed in future studies. Also, mice in Sed that were staying in air at RT rather than at 32+1°C for 2 h may be considered to be a weakness of this study, as the difference in temperature between groups could affect gene expression. Therefore, future study with treadmill exercise is required to confirm the effects of acute exercise on adipogenic gene expression at mRNA and protein levels.
CONCLUSIONS
In summary, we demonstrated for the first time that acute exercise could regulate adipogenic gene expression in WAT. We made a novel observation that even a single swimming exercise resulted in attenuations of PPARγ2 and C/EBPα expression at the mRNA level in eWAT, with high induction of Wnt10b and anti-adipogenic KLFs (KLF2, KLF3 and KLF7). In addition, C/EBPβ and C/EBPγ expression was upregulated, in parallel with expression levels of PGC-1α and UCP1. In iWAT, acute exercise attenuated the expression of PPARγ2, with high induction of Wnt6 and anti-adipogenic KLFs (KLF2, KLF3 and KLF7). While expression of C/EBP, C/EBPβ and PGC-1γ was also increased in iWAT, UCP1 expression was not changed. These results suggest that acute exercise inhibits adipogenic gene expression and may regulate thermogenesis by activating PGC-1α and UCP1 in WATs.
Conflict of interests
the authors declared no conflict of interests regarding the publication of this manuscript.
REFERENCES
- 1.Guilherme A, Virbasius JV, Puri V, Czech MP. Adipocyte dysfunctions linking obesity to insulin resistance and type 2 diabetes. Nat Rev Mol Cell Biol. 2008;9:367–377. doi: 10.1038/nrm2391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Garaulet M, Hernandez-Morante JJ, Lujan J, Tebar FJ, Zamora S. Relationship between fat cell size and number and fatty acid composition in adipose tissue from different fat depots in overweight/obese humans. Int J Obes (Lond). 2006;30:899–905. doi: 10.1038/sj.ijo.0803219. [DOI] [PubMed] [Google Scholar]
- 3.Marques BG, Hausman DB, Martin RJ. Association of fat cell size and paracrine growth factors in development of hyperplastic obesity. Am J Physiol. 1998;275:R1898–1908. doi: 10.1152/ajpregu.1998.275.6.R1898. [DOI] [PubMed] [Google Scholar]
- 4.Wang QA, Tao C, Gupta RK, Scherer PE. Tracking adipogenesis during white adipose tissue development, expansion and regeneration. Nat Med. 2013;19:1338–1344. doi: 10.1038/nm.3324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rosen ED, MacDougald OA. Adipocyte differentiation from the inside out. Nat Rev Mol Cell Biol. 2006;7:885–896. doi: 10.1038/nrm2066. [DOI] [PubMed] [Google Scholar]
- 6.Zhu Y, Qi C, Korenberg JR, Chen XN, Noya D, Rao MS, Reddy JK. Structural organization of mouse peroxisome proliferator-activated receptor gamma (mPPAR gamma) gene: alternative promoter use and different splicing yield two mPPAR gamma isoforms. Proc Natl Acad Sci U S A. 1995;92:7921–7925. doi: 10.1073/pnas.92.17.7921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fajas L, Auboeuf D, Raspe E, Schoonjans K, Lefebvre AM, Saladin R, Najib J, Laville M, Fruchart JC, Deeb S, Vidal-Puig A, Flier J, Briggs MR, Staels B, Vidal H, Auwerx J. The organization, promoter analysis, and expression of the human PPARgamma gene. J Biol Chem. 1997;272:18779–18789. doi: 10.1074/jbc.272.30.18779. [DOI] [PubMed] [Google Scholar]
- 8.Braissant O, Foufelle F, Scotto C, Dauca M, Wahli W. Differential expression of peroxisome proliferator-activated receptors (PPARs): tissue distribution of PPAR-alpha, -beta, and -gamma in the adult rat. Endocrinology. 1996;137:354–366. doi: 10.1210/endo.137.1.8536636. [DOI] [PubMed] [Google Scholar]
- 9.Tontonoz P, Hu E, Graves RA, Budavari AI, Spiegelman BM. mPPAR gamma 2: tissue-specific regulator of an adipocyte enhancer. Genes Dev. 1994;8:1224–1234. doi: 10.1101/gad.8.10.1224. [DOI] [PubMed] [Google Scholar]
- 10.Zhang J, Fu M, Cui T, Xiong C, Xu K, Zhong W, Xiao Y, Floyd D, Liang J, Li E, Song Q, Chen YE. Selective disruption of PPARgamma 2 impairs the development of adipose tissue and insulin sensitivity. Proc Natl Acad Sci U S A. 2004;101:10703–10708. doi: 10.1073/pnas.0403652101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Medina-Gomez G, Virtue S, Lelliott C, Boiani R, Campbell M, Christodoulides C, Perrin C, Jimenez-Linan M, Blount M, Dixon J, Zahn D, Thresher RR, Aparicio S, Carlton M, Colledge WH, Kettunen MI, Seppanen-Laakso T, Sethi JK, O’Rahilly S, Brindle K, Cinti S, Oresic M, Burcelin R, Vidal-Puig A. The link between nutritional status and insulin sensitivity is dependent on the adipocyte-specific peroxisome proliferator-activated receptor-gamma2 isoform. Diabetes. 2005;54:1706–1716. doi: 10.2337/diabetes.54.6.1706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Okuno A, Tamemoto H, Tobe K, Ueki K, Mori Y, Iwamoto K, Umesono K, Akanuma Y, Fujiwara T, Horikoshi H, Yazaki Y, Kadowaki T. Troglitazone increases the number of small adipocytes without the change of white adipose tissue mass in obese Zucker rats. J Clin Invest. 1998;101:1354–1361. doi: 10.1172/JCI1235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.de Souza CJ, Eckhardt M, Gagen K, Dong M, Chen W, Laurent D, Burkey BF. Effects of pioglitazone on adipose tissue remodeling within the setting of obesity and insulin resistance. Diabetes. 2001;50:1863–1871. doi: 10.2337/diabetes.50.8.1863. [DOI] [PubMed] [Google Scholar]
- 14.Landschulz WH, Johnson PF, McKnight SL. The DNA binding domain of the rat liver nuclear protein C/EBP is bipartite. Science. 1989;243:1681–1688. doi: 10.1126/science.2494700. [DOI] [PubMed] [Google Scholar]
- 15.Freytag SO, Paielli DL, Gilbert JD. Ectopic expression of the CCAAT/enhancer-binding protein alpha promotes the adipogenic program in a variety of mouse fibroblastic cells. Genes Dev. 1994;8:1654–1663. doi: 10.1101/gad.8.14.1654. [DOI] [PubMed] [Google Scholar]
- 16.Rosen ED, Hsu CH, Wang X, Sakai S, Freeman MW, Gonzalez FJ, Spiegelman BM. C/EBPalpha induces adipogenesis through PPARgamma: a unified pathway. Genes Dev. 2002;16:22–26. doi: 10.1101/gad.948702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wang ND, Finegold MJ, Bradley A, Ou CN, Abdelsayed SV, Wilde MD, Taylor LR, Wilson DR, Darlington GJ. Impaired energy homeostasis in C/EBP alpha knockout mice. Science. 1995;269:1108–1112. doi: 10.1126/science.7652557. [DOI] [PubMed] [Google Scholar]
- 18.Linhart HG, Ishimura-Oka K, DeMayo F, Kibe T, Repka D, Poindexter B, Bick RJ, Darlington GJ. C/EBPalpha is required for differentiation of white, but not brown, adipose tissue. Proc Natl Acad Sci U S A. 2001;98:12532–12537. doi: 10.1073/pnas.211416898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yeh WC, Cao Z, Classon M, McKnight SL. Cascade regulation of terminal adipocyte differentiation by three members of the C/EBP family of leucine zipper proteins. Genes Dev. 1995;9:168–181. doi: 10.1101/gad.9.2.168. [DOI] [PubMed] [Google Scholar]
- 20.Wu Z, Bucher NL, Farmer SR . Induction of peroxisome proliferator-activated receptor gamma during the conversion of 3T3 fibroblasts into adipocytes is mediated by C/EBPbeta, C/EBPdelta, and glucocorticoids. Mol Cell Biol. 1996;16:4128–4136. doi: 10.1128/mcb.16.8.4128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tanaka T, Yoshida N, Kishimoto T, Akira S. Defective adipocyte differentiation in mice lacking the C/EBPbeta and/or C/EBPdelta gene. EMBO J. 1997;16:7432–7443. doi: 10.1093/emboj/16.24.7432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ross SE, Hemati N, Longo KA, Bennett CN, Lucas PC, Erickson RL, MacDougald OA. Inhibition of adipogenesis by Wnt signaling. Science. 2000;289:950–953. doi: 10.1126/science.289.5481.950. [DOI] [PubMed] [Google Scholar]
- 23.Cawthorn WP, Bree AJ, Yao Y, Du B, Hemati N, Martinez-Santibanez G, MacDougald OA. Wnt6, Wnt10a and Wnt10b inhibit adipogenesis and stimulate osteoblastogenesis through a beta-catenin-dependent mechanism. Bone. 2012;50:477–489. doi: 10.1016/j.bone.2011.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wu Z, Wang S. Role of kruppel-like transcription factors in adipogenesis. Dev Biol. 2013;373:235–243. doi: 10.1016/j.ydbio.2012.10.031. [DOI] [PubMed] [Google Scholar]
- 25.Mori T, Sakaue H, Iguchi H, Gomi H, Okada Y, Takashima Y, Nakamura K, Nakamura T, Yamauchi T, Kubota N, Kadowaki T, Matsuki Y, Ogawa W, Hiramatsu R, Kasuga M. Role of Kruppel-like factor 15 (KLF15) in transcriptional regulation of adipogenesis. J Biol Chem. 2005;280:12867–12875. doi: 10.1074/jbc.M410515200. [DOI] [PubMed] [Google Scholar]
- 26.Pei H, Yao Y, Yang Y, Liao K, Wu JR. Kruppel-like factor KLF9 regulates PPARgamma transactivation at the middle stage of adipogenesis. Cell Death Differ. 2011;18:315–327. doi: 10.1038/cdd.2010.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li D, Yea S, Li S, Chen Z, Narla G, Banck M, Laborda J, Tan S, Friedman JM, Friedman SL, Walsh MJ. Kruppel-like factor-6 promotes preadipocyte differentiation through histone deacetylase 3-dependent repression of DLK1. J Biol Chem. 2005;280:26941–26952. doi: 10.1074/jbc.M500463200. [DOI] [PubMed] [Google Scholar]
- 28.Sue N, Jack BH, Eaton SA, Pearson RC, Funnell AP, Turner J, Czolij R, Denyer G, Bao S, Molero-Navajas JC, Perkins A, Fujiwara Y, Orkin SH, Bell-Anderson K, Crossley M. Targeted disruption of the basic Kruppel-like factor gene (Klf3) reveals a role in adipogenesis. Mol Cell Biol. 2008;28:3967–3978. doi: 10.1128/MCB.01942-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kawamura Y, Tanaka Y, Kawamori R, Maeda S. Overexpression of Kruppel-like factor 7 regulates adipocytokine gene expressions in human adipocytes and inhibits glucose-induced insulin secretion in pancreatic beta-cell line. Mol Endocrinol. 2006;20:844–856. doi: 10.1210/me.2005-0138. [DOI] [PubMed] [Google Scholar]
- 30.Banerjee SS, Feinberg MW, Watanabe M, Gray S, Haspel RL, Denkinger DJ, Kawahara R, Hauner H, Jain MK. The Kruppel-like factor KLF2 inhibits peroxisome proliferator-activated receptor-gamma expression and adipogenesis. J Biol Chem. 2003;278:2581–2584. doi: 10.1074/jbc.M210859200. [DOI] [PubMed] [Google Scholar]
- 31.Tong Q, Dalgin G, Xu H, Ting CN, Leiden JM, Hotamisligil GS. Function of GATA transcription factors in preadipocyte-adipocyte transition. Science. 2000;290:134–138. doi: 10.1126/science.290.5489.134. [DOI] [PubMed] [Google Scholar]
- 32.Tong Q, Tsai J, Tan G, Dalgin G, Hotamisligil GS. Interaction between GATA and the C/EBP family of transcription factors is critical in GATA-mediated suppression of adipocyte differentiation. Mol Cell Biol. 2005;25:706–715. doi: 10.1128/MCB.25.2.706-715.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kim JB, Spiegelman BM. ADD1/SREBP1 promotes adipocyte differentiation and gene expression linked to fatty acid metabolism. Genes Dev. 1996;10:1096–1107. doi: 10.1101/gad.10.9.1096. [DOI] [PubMed] [Google Scholar]
- 34.Kim JB, Sarraf P, Wright M, Yao KM, Mueller E, Solanes G, Lowell BB, Spiegelman BM. Nutritional and insulin regulation of fatty acid synthetase and leptin gene expression through ADD1/SREBP1. J Clin Invest. 1998;101:1–9. doi: 10.1172/JCI1411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Shimano H, Shimomura I, Hammer RE, Herz J, Goldstein JL, Brown MS, Horton JD. Elevated levels of SREBP-2 and cholesterol synthesis in livers of mice homozygous for a targeted disruption of the SREBP-1 gene. J Clin Invest. 1997;100:2115–2124. doi: 10.1172/JCI119746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Shimomura I, Hammer RE, Richardson JA, Ikemoto S, Bashmakov Y, Goldstein JL, Brown MS. Insulin resistance and diabetes mellitus in transgenic mice expressing nuclear SREBP-1c in adipose tissue: model for congenital generalized lipodystrophy. Genes Dev. 1998;12:3182–3194. doi: 10.1101/gad.12.20.3182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sakurai T, Ogasawara J, Kizaki T, Sato S, Ishibashi Y, Takahashi M, Kobayashi O, Oh-Ishi S, Nagasawa J, Takahashi K, Ishida H, Izawa T, Ohno H. The effects of exercise training on obesity-induced dysregulated expression of adipokines in white adipose tissue. Int J Endocrinol. 2013;2013:801743. doi: 10.1155/2013/801743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Horowitz JF. Fatty acid mobilization from adipose tissue during exercise. Trends Endocrinol Metab. 2003;14:386–392. doi: 10.1016/s1043-2760(03)00143-7. [DOI] [PubMed] [Google Scholar]
- 39.Zechner R, Kienesberger PC, Haemmerle G, Zimmermann R, Lass A. Adipose triglyceride lipase and the lipolytic catabolism of cellular fat stores. J Lipid Res. 2009;50:3–21. doi: 10.1194/jlr.R800031-JLR200. [DOI] [PubMed] [Google Scholar]
- 40.Ogasawara J, Nomura S, Rahman N, Sakurai T, Kizaki T, Izawa T, Ishida H, Haga S, Ohno H. Hormone-sensitive lipase is critical mediators of acute exercise-induced regulation of lipolysis in rat adipocytes. Biochem Biophys Res Commun. 2010;400:134–139. doi: 10.1016/j.bbrc.2010.08.026. [DOI] [PubMed] [Google Scholar]
- 41.Ogasawara J, Sakurai T, Kizaki T, Ishibashi Y, Izawa T, Sumitani Y, Ishida H, Radak Z, Haga S, Ohno H. Higher levels of ATGL are associated with exercise-induced enhancement of lipolysis in rat epididymal adipocytes. PLoS One. 2012;7:e40876. doi: 10.1371/journal.pone.0040876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lukaszuk B, Bialuk I, Gorski J, Zajaczkiewicz M, Winnicka MM, Chabowski A. A single bout of exercise increases the expression of glucose but not fatty acid transporters in skeletal muscle of IL-6 KO mice. Lipids. 2012;47:763–772. doi: 10.1007/s11745-012-3678-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Borghi SM, Pinho-Ribeiro FA, Zarpelon AC, Cunha TM, Alves-Filho JC, Ferreira SH, Cunha FQ, Casagrande R, Verri WA., Jr Interleukin-10 limits intense acute swimming-induced muscle mechanical hyperalgesia in mice. Exp Physiol. 2015;100:531–544. doi: 10.1113/EP085026. [DOI] [PubMed] [Google Scholar]
- 44.Motoshima H, Wu X, Sinha MK, Hardy VE, Rosato EL, Barbot DJ, Rosato FE, Goldstein BJ. Differential regulation of adiponectin secretion from cultured human omental and subcutaneous adipocytes: effects of insulin and rosiglitazone. J Clin Endocrinol Metab. 2002;87:5662–5667. doi: 10.1210/jc.2002-020635. [DOI] [PubMed] [Google Scholar]
- 45.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 46.Kajimura S, Seale P, Kubota K, Lunsford E, Frangioni JV, Gygi SP, Spiegelman BM. Initiation of myoblast to brown fat switch by a PRDM16-C/EBP-beta transcriptional complex. Nature. 2009;460:1154–1158. doi: 10.1038/nature08262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Cannon B, Nedergaard J. Brown adipose tissue: function and physiological significance. Physiol Rev. 2004;84:277–359. doi: 10.1152/physrev.00015.2003. [DOI] [PubMed] [Google Scholar]
- 48.Himms-Hagen J. Nonshivering thermogenesis. Brain Res Bull. 1984;12:151–160. doi: 10.1016/0361-9230(84)90183-7. [DOI] [PubMed] [Google Scholar]
- 49.Ribeiro MO, Carvalho SD, Schultz JJ, Chiellini G, Scanlan TS, Bianco AC, Brent GA. Thyroid hormone--sympathetic interaction and adaptive thermogenesis are thyroid hormone receptor isoform--specific. J Clin Invest. 2001;108:97–105. doi: 10.1172/JCI12584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Fortunato RS, Ignacio DL, Padron AS, Pecanha R, Marassi MP, Rosenthal D, Werneck-de-Castro JP, Carvalho DP. The effect of acute exercise session on thyroid hormone economy in rats. J Endocrinol. 2008;198:347–353. doi: 10.1677/JOE-08-0174. [DOI] [PubMed] [Google Scholar]
- 51.Wu J, Bostrom P, Sparks LM, Ye L, Choi JH, Giang AH, Khandekar M, Virtanen KA, Nuutila P, Schaart G, Huang K, Tu H, van Marken Lichtenbelt WD, Hoeks J, Enerback S, Schrauwen P, Spiegelman BM. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell. 2012;150:366–376. doi: 10.1016/j.cell.2012.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kawamura T, Yoshida K, Sugawara A, Nagasaka M, Mori N, Takeuchi K, Kohzuki M. Regulation of skeletal muscle peroxisome proliferator-activated receptor gamma expression by exercise and angiotensin-converting enzyme inhibition in fructose-fed hypertensive rats. Hypertens Res. 2004;27:61–70. doi: 10.1291/hypres.27.61. [DOI] [PubMed] [Google Scholar]
- 53.Petridou A, Tsalouhidou S, Tsalis G, Schulz T, Michna H, Mougios V. Long-term exercise increases the DNA binding activity of peroxisome proliferator-activated receptor gamma in rat adipose tissue. Metabolism. 2007;56:1029–1036. doi: 10.1016/j.metabol.2007.03.011. [DOI] [PubMed] [Google Scholar]
- 54.Kliewer SA, Sundseth SS, Jones SA, Brown PJ, Wisely GB, Koble CS, Devchand P, Wahli W, Willson TM, Lenhard JM, Lehmann JM. Fatty acids and eicosanoids regulate gene expression through direct interactions with peroxisome proliferator-activated receptors alpha and gamma. Proc Natl Acad Sci U S A. 1997;94:4318–4323. doi: 10.1073/pnas.94.9.4318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Sakurai T, Endo S, Hatano D, Ogasawara J, Kizaki T, Oh-ishi S, Izawa T, Ishida H, Ohno H. Effects of exercise training on adipogenesis of stromal-vascular fraction cells in rat epididymal white adipose tissue. Acta Physiol (Oxf). 2010;200:325–338. doi: 10.1111/j.1748-1708.2010.02159.x. [DOI] [PubMed] [Google Scholar]
- 56.Longo KA, Wright WS, Kang S, Gerin I, Chiang SH, Lucas PC, Opp MR, MacDougald OA. Wnt10b inhibits development of white and brown adipose tissues. J Biol Chem. 2004;279:35503–35509. doi: 10.1074/jbc.M402937200. [DOI] [PubMed] [Google Scholar]
- 57.Wright WS, Longo KA, Dolinsky VW, Gerin I, Kang S, Bennett CN, Chiang SH, Prestwich TC, Gress C, Burant CF, Susulic VS, MacDougald OA. Wnt10b inhibits obesity in ob/ob and agouti mice. Diabetes. 2007;56:295–303. doi: 10.2337/db06-1339. [DOI] [PubMed] [Google Scholar]
- 58.Leal ML, Lamas L, Aoki MS, Ugrinowitsch C, Ramos MS, Tricoli V, Moriscot AS. Effect of different resistance-training regimens on the WNT-signaling pathway. Eur J Appl Physiol. 2011;111:2535–2545. doi: 10.1007/s00421-011-1874-7. [DOI] [PubMed] [Google Scholar]
- 59.Spillane M, Schwarz N, Willoughby DS. Upper-body resistance exercise augments vastus lateralis androgen receptor-DNA binding and canonical Wnt/beta-catenin signaling compared to lower-body resistance exercise in resistance-trained men without an acute increase in serum testosterone. Steroids. 2015;98:63–71. doi: 10.1016/j.steroids.2015.02.019. [DOI] [PubMed] [Google Scholar]
- 60.Wang H, Peiris TH, Mowery A, Le Lay J, Gao Y, Greenbaum LE. CCAAT/enhancer binding protein-beta is a transcriptional regulator of peroxisome-proliferator-activated receptor-gamma coactivator-1alpha in the regenerating liver. Mol Endocrinol. 2008;22:1596–1605. doi: 10.1210/me.2007-0388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Puigserver P, Wu Z, Park CW, Graves R, Wright M, Spiegelman BM. A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell. 1998;92:829–839. doi: 10.1016/s0092-8674(00)81410-5. [DOI] [PubMed] [Google Scholar]
- 62.Puigserver P, Spiegelman BM. Peroxisome proliferator-activated receptor-gamma coactivator 1 alpha (PGC-1 alpha): transcriptional coactivator and metabolic regulator. Endocr Rev. 2003;24:78–90. doi: 10.1210/er.2002-0012. [DOI] [PubMed] [Google Scholar]
- 63.Seale P, Kajimura S, Yang W, Chin S, Rohas LM, Uldry M, Tavernier G, Langin D, Spiegelman BM. Transcriptional control of brown fat determination by PRDM16. Cell Metab. 2007;6:38–54. doi: 10.1016/j.cmet.2007.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wu Z, Puigserver P, Andersson U, Zhang C, Adelmant G, Mootha V, Troy A, Cinti S, Lowell B, Scarpulla RC, Spiegelman BM. Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1. Cell. 1999;98:115–124. doi: 10.1016/S0092-8674(00)80611-X. [DOI] [PubMed] [Google Scholar]
- 65.Sutherland LN, Bomhof MR, Capozzi LC, Basaraba SA, Wright DC. Exercise and adrenaline increase PGC-1{alpha} mRNA expression in rat adipose tissue. J Physiol. 2009;587:1607–1617. doi: 10.1113/jphysiol.2008.165464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Ringholm S, Grunnet Knudsen J, Leick L, Lundgaard A, Munk Nielsen M, Pilegaard H. PGC-1alpha is required for exercise- and exercise training-induced UCP1 up-regulation in mouse white adipose tissue. PLoS One. 2013;8:e64123. doi: 10.1371/journal.pone.0064123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.De Matteis R, Lucertini F, Guescini M, Polidori E, Zeppa S, Stocchi V, Cinti S, Cuppini R. Exercise as a new physiological stimulus for brown adipose tissue activity. Nutr Metab Cardiovasc Dis. 2013;23:582–590. doi: 10.1016/j.numecd.2012.01.013. [DOI] [PubMed] [Google Scholar]