Abstract
The goals of this work were to characterize progression of osteoarthritic cartilage degeneration in a rabbit medial meniscus destabilization (MMD) model and then to use the model to identify pre-histologic disruptions in chondrocyte metabolism under chronically elevated joint contact stresses in vivo. To characterize PTOA progression, 24 rabbits received either MMD or sham surgery. Limb loading was analyzed preoperatively and at regular postoperative intervals using a Tekscan pressure-sensitive walkway. Animals were euthanized 8 (n=8 MMD; n=8 sham) or 26 weeks (n=8 MMD) postoperatively for histological cartilage evaluation by an objective, semiautomated Mankin scoring routine. To examine pre-histologic pathology, MMD was performed on an additional 20 rabbits, euthanized one (n=9) or four weeks (n=10) postoperatively. Chondrocytes were harvested fresh for measurement of mitochondrial function, an intracellular indicator of pathology after mechanical injury. Both MMD and sham surgery caused slight decreases in limb loading which returned to preoperative levels after 2 weeks. Histologically apparent cartilage damage progressed from 8 to 26 weeks after MMD. Changes in chondrocyte respiration were variable at 1 week, but by 4 weeks postoperatively chondrocyte mitochondrial function was significantly reduced.
Clinical Significance
Many human injuries that lead to PTOA are relatively mild, and the cell-level mechanisms leading to disease remain unclear. We have documented PTOA progression in an animal model of subtle joint injury under continued use, and demonstrated that this model provides a realistic environment for investigation of multi-stage cellular pathology that develops prior to overt tissue degeneration and which could be targeted for disease modifying treatments.
Keywords: Post-Traumatic Osteoarthritis, Rabbit, Mankin Score, Overload, Mitochondria
INTRODUCTION
Post-traumatic osteoarthritis (PTOA) is joint degeneration after injuries ranging in severity and mechanism from high-energy intra-articular fractures and joint impacts accompanying ACL ruptures to chronic low-level mechanical insults such as rotational ankle fractures or ligament sprains that cause subtle instability or persistent abnormal contact mechanics.1; 2 Systematic investigation of PTOA pathology and treatment requires the use of animal models in which the mechanism of injury can be controlled and standardized. This standardization is relatively straightforward in animal models of impact and intra-articular fracture in which the joint or the cartilage surface itself is struck with a known force. Studying disease progression associated with subtle joint injury or persistent biomechanical abnormalities is often challenging in animal models because modest surgical insults often heal or do not progress in small animals, and more severe surgical insults can be so disruptive as to alter animal limb usage and thus disease progression. Animal models of subtle joint injury typically make use of ACL transection, meniscectomy, or a combination, to induce PTOA pathology. A serious disadvantage to this approach has been the relatively unknown extent to which surgical exposure disrupts animal limb usage and mechanics, rendering it unclear what part of the resulting pathology was attributable to the simulated joint injury versus the surgery itself.
Recently, a meniscus destabilization technique initially reported in the mouse3 has been adapted for use in the rabbit via a posterior approach.4; 5 This technique holds great appeal as the posterior approach to the joint minimizes injury to adjacent soft tissues, maximizing the chance that the animal will continue to use and load the limb in a normal fashion. In that previous work in the rabbit stifle (analogous to the human knee), a simple release of the posterior root of the meniscus was shown to increase joint contact stress and decrease contact area in the medial compartment to the same degree as a total meniscectomy. While neither the meniscectomy nor meniscus destabilization cause gross joint laxity, both injuries shift the contact patch laterally and posteriorly onto cartilage potentially unaccustomed to the higher stress. The resulting histological cartilage degeneration was nearly equivalent between the two meniscal injuries.4 This meniscus destabilization model provides a method to create excessive mechanical stress on cartilage with minimal joint instability and soft tissue disruption in an animal model. A pilot series of live animals subjected to a meniscus destabilization indicated that this technique results in reproducible but relatively mild cartilage degeneration in the medial compartment 8 weeks postoperatively.4
Even as meniscal destabilization in the rabbit thus far has demonstrated promise for inducing PTOA after subtle joint injury, joint degeneration in this model has not yet been described as progressing with time, as would be seen in human patients with a chronically overloaded joint. Thus, the first aim of this work was to characterize the progression of the initial stages of cartilage degeneration that developed 8 weeks after meniscus destabilization. It was hypothesized that the animal would use their limb normally after the minimally disruptive surgical procedure, resulting in an increase in the severity of histological cartilage abnormality with time. Once disease progression was established, the second aim of this work was to confirm the presence and determine the time course of changes in mitochondrial respiration that occur in response to mechanical overload. It was hypothesized that a deficiency in chondrocyte mitochondrial function, a measure previously shown to be associated with OA pathology in mice and shown to be time- and overload-dependent in bovine explant studies,6; 7 would manifest prior to tissue-level damage within the pathologic sequence of this rabbit model. This second goal aims to leverage this rabbit model for characterization of subtle changes in chondrocyte biology that occur prior to any appearance of histological OA, and thus present attractive therapeutic targets.
METHODS
Surgical Techniques and In Vivo Monitoring
Under IACUC approval, a total of 44 mature New Zealand white rabbits (males, 7 to 9 months old,8 average weight 3.5 kg) underwent survival experimental surgery. Thirty-six rabbits had medial meniscus destabilization (MMD) as described by Arunakul et al.,4 and the remaining eight received sham surgery. Anesthesia, surgical approach, wound closure, and postoperative care were identical between sham and MMD groups. Briefly, animals were sedated with an intramuscular injection of ketamine, xylazine, and acepromazine, and anesthesia was maintained using isoflurane in oxygen delivered via face mask. The animal was placed in a prone position, and using aseptic technique, instrumentation was passed through the intermuscular plane between the semimembranosus and medial gastrocnemius to make a small (approximately 3 mm) posteromedial arthrotomy on the left stifle. Care was taken to ensure all major muscular and ligamentous structures remained uninjured. For animals receiving an MMD, the posterior root of the medial meniscus was sharply transected from the tibial attachment using a #11 scalpel, while no additional procedures were performed on animals in the sham group. The skin was closed using a bioabsorbable suture. Postoperatively, rabbits were single housed, allowed unrestricted cage activity and an ad libitum diet. Animals were monitored twice daily by veterinary staff.
Characterization of PTOA Progression
Progression of PTOA after MMD was characterized using 24/44 animals in this work. Eight sham surgery animals and 8 MMD animals were sacrificed 8 weeks after MMD surgery, and 8 MMD animals were sacrificed 26 weeks postoperative (respectively designated as the Sham, 8W-MMD, and the 26W-MMD groups, n = 8 for each group).
The 24 animals used to study PTOA progression were operated on over a 2-day period and survival time among the MMD animals was randomized. Changes in experimental limb loading during the postoperative survival period were assessed using a high-resolution (4 sensing elements/cm2) pressure sensing walkway system (HR Walkway® HRV-2, Tekscan Inc., North Boston, MA, USA). From each animal, data were collected preoperatively, as well as 1, 2, 4, 8, 12, 16, and 26 weeks after index surgery. At every data collection time point, each animal completed five to ten hopping trials. Data from trials with continuous hopping motions were analyzed using the Walkway® Ver. 7.01 software, and the average value of at least five successfully measured hops was recorded as the definitive result for the time point for the animal. Peak force, impulse, stance time, stride length, and velocity were studied. All postoperative data points for a given animal were normalized to the individual animal’s preoperative value. Repeated measures two-way ANOVA with Tukey’s multiple comparisons was used to investigate the effects of surgery group and postoperative time on limb loading parameters.
At either 8 or 26 weeks postoperatively, the animals were sacrificed using a lethal injection of Euthasol (120mg/kg). The experimental MMD and the intact contralateral stifles were harvested and prepared for cartilage histological evaluation following OARSI guidelines.9 From each joint, four sagittal sections (one each from the medial and lateral femoral condyles and one each from the medial and lateral tibial plateaus) were prepared and stained with Safranin-O/Fast Green. For each joint surface, a representative histological section was digitized using a stage scanner microscope (Olympus VS110, Olympus America Inc., Center Valley, PA, USA) at a resolution of 322.25 nm/pixel. The central 5 mm anterior-posterior span of the articular cartilage was isolated and exported for histological evaluation in a .tif format at a resolution of 1.61 μm/pixel. The region of interest on the femoral condyles was centered on the caudal (posterior aspect) spherical regions of the joint, where articular cartilage was habitually loaded in the rabbit’s physiological deeply flexed limb orientation.10–13 The region of interest for the tibial plateaus was a central part of the joint surface which is normally uncovered by the menisci.
Analysis of the histological cartilage changes in this work was performed using a rabbit stifle-specific, compartment-specific, semi-automated histology scoring program that objectively assigns Mankin scores based on quantitative data derived from image analysis of the Safranin-O-stained histology slides. This approach limits the subjectivity of Mankin scores given manually by “experts,” which can be highly variable based on experience and knowledge of different species characteristic cartilage appearance.9; 14–16 The analysis program was implemented in MATLAB (MathWorks, Natick, Massachusetts, USA) and objectively identified the number and depth of cartilage cracks and surface defects, quantified cell density based on the number of automatically detected chondrocyte nuclei/lacune per cartilage area, and quantified proteoglycan content based on the numeric intensity of the red pixels in the cartilage.17 These histological features were assessed in 0.5-mm wide subdivisions spanning the anterior/posterior direction of the cartilage. Comparison of these objective quantitative histological data to rabbit and site-specific measures18 for these data allowed for an unbiased assignment of a Mankin score to each evaluated tissue location. To obtain a single value (quantitative or Mankin score) for a given histological section, the values spanning the surface were averaged. To evaluate changes in histological characteristics between Sham and 8W-MMD or between 8W-MMD and 26W-MMD, repeated measures two-way ANOVA with Tukey’s multiple comparisons was used. Significance was set at p < 0.05.
Characterization of Chondrocyte Respiration
Effects of joint overloading after MMD were characterized using 20/44 animals in this work. One animal died intraoperatively from complications associated with anesthesia, leaving 9 MMD animals that were euthanized 1 week after MMD surgery. Ten additional rabbits who received MMD surgery were euthanized 4 weeks postoperatively. No limb loading analysis was performed on these animals.
At the time of euthanasia, full thickness sections of cartilage from both the MMD and contralateral stifles were harvested fresh from loaded portions of the lateral and medial tibial plateau. These are critical loci of disease appearance that also contain sufficient quantities of chondrocytes for analysis. Femoral samples were excluded because of a lack of sufficient cell number and signal strength. Cartilage was then digested in 0.1 mg/ml collagenase/pronase (Sigma-Aldrich, St. Louis, MO) over 16 hours and cells were plated on Seahorse XF96 Analyzer (Seahorse Biosciences, North Billerica, MA, USA) culture plates for analysis. Once cells had recovered from harvest and attached to plates, media were changed to assay media and plates were analyzed according to standard mitochondrial stress test protocols as previously described.7 Briefly, oxygen consumption rates (OCR), were measured in real time in response to a series of injections: 2 μM oligomycin to block complex V, then 0.25 μM carbonyl cyanide-4-(trifluoromethoxy) phenylhydrazone (FCCP) to uncouple the mitochondria, and finally a combination of 2 μM rotenone and 5 μM antimycin A to block electron transport chain activity. This allows determination of basal respiration, maximal respiration (uncoupled respiration), and proton leakage (an expression of that oxygen consumed which does not result in complex V activity). In line with existing mitochondrial stress test literature, proton leakage has been expressed as a percentage of basal OCR. Two-way ANOVA was used to investigate the effects of injury and time on the different respiratory outcome measures. Significance was set at p < 0.05.
RESULTS
Limb Loading
There was no difference in peak contact force between the 8W-MMD group and the first eight weeks of the 26W-MMD rabbit group at any of the postoperative time points (multiplicity adjusted p=0.993, p=0.976, p=0.999, p=0.998 for 1, 2, 4 and 8 weeks postoperatively, respectively). Pooling data from both MMD groups showed that 1 week after surgery, MMD caused an average 9% decrease in peak force passing through the operated limb relative to preoperative loading, however this difference was not significant (multiplicity adjusted p=0.899). This was in comparison to an average 4% decrease in peak force in the intact contralateral limb and an average 8% increase in peak force in the sham animals. There were no significant differences between peak forces in the operated limb and the non-operated contralateral limb for either the sham group or the MMD group (p=0.81). However, there was a significant (p<0.001) 25%–33% increase in peak force relative to preoperative values in all groups at 8 weeks, which persisted through 26 weeks (Figure 1). This was primarily attributed to the approximate 26% increase in body weight relative to preoperative weight at 8 weeks (average 25% - range 17%–30% for 8W-Sham; 27% - range 16%–46% for 8W-MMD), and an average increase in body weight of 46% at 26 weeks (range 8%–69%). There was no significant difference (p=0.716) in peak force between the MMD limb and the intact contralateral in animals out to 26 weeks.
Figure 1.
Peak vertical force (top) and vertical impulse (bottom) in the operated limb (either MMD or sham) and the non-operated contralateral limbs from the MMD and sham groups. Data points are an average and bars are standard deviations. N=8 for all data points except for MMD and MMD Cont at times before 8W when the data were pooled from the 8W-MMD and 26W-MMD groups (n=16). The insets (in gray) show limb loading results on an expanded axis to better illustrate the extremely modest early differences in limb loading associated with surgery and between the MMD and sham groups. Each data series is slightly offset from the others horizontally to allow for visualization of the individual error bars at each time point.
There was an average 40% – 42% increase in vertical impulse in both the intact contralateral and the operated limb in the MMD rabbits one week postoperatively (Figure 1). Similarly, there was a 29% to 32% increase in vertical impulse in the intact contralateral and sham operated limbs one week postoperatively. This increase in impulse was primarily a result of an average 25% to 45% increase in stance time relative to preoperative stance time that occurred in both the sham and the MMD rabbits. After 8 weeks, there was a decrease in stance time in both the operated and non-operated limbs to 51% to 61% of the preoperative stance time. This decrease in stance time corresponded to an 81%–95% increase in limb velocity relative to preoperative values that began 8 weeks postoperatively. As with peak force, there were no significant differences between the MMD limb and the intact contralateral limb in terms of impulse (p=0.716), stance time (p=0.899), or limb velocity (p=0.855) out to 26 weeks postoperatively.
Histological Tissue Appearance
The overall Mankin scores varied significantly by compartment (p<0.001) and by experimental group (p=0.037). Unlike the original report of the model, there was no significant difference in total Mankin scores between the sham and 8W-MMD groups. Values of the reported Mankin scores were modest due to averaging over the entire load-bearing cartilage surface. Mankin scores were typically higher, indicating more severe cartilage degeneration, in the 26W-MMD group than in the 8W-MMD, with greater degeneration occurring on the medial tibial and femoral articular surfaces (Figure 2). Considering the individual components of the Mankin score, specifically structural cartilage damage, changes in cellularity and organization, and proteoglycan (PG) depletion, it appears that the greatest contributor to differences between the sham, 8W-MMD and 26W-MMD groups was the cartilage structure score. Mankin cellularity scores and PG scores showed some trends toward increasing damage with MMD and postoperative time, but these changes were very modest and statistically insignificant (Figure 2).
Figure 2.
Average Mankin scores are plotted on the top left, and individual scoring components contributing to the overall score are plotted in the other three panels. For every plot, open shapes are individual animal data, filled shapes are group averages and vertical bars are standard deviations. Statistical significance between groups as determined by Tukey’s multiple comparisons tests is indicated with horizontal bars and associated multiplicity adjusted p values. Differences in structural scores between the sham, 8W-MMD and 26W-MMD groups appear to be the greatest contributors to the differences in total Mankin scores between the groups.
However, Mankin scores are a very gross assessment of cartilage changes, and in this work Mankin scores were assigned automatically based on objectively quantified data. When considering the quantitive data rather than the Mankin scores, it became apparent that MMD did cause changes in cell density, particularly on the femoral surface and in the more superficial zones of the cartilage (Figure 3). Loss of cell density became statistically significant in the superficial zone in all compartments, but only at 26 weeks postoperatively. Cell density was decreased in both the medial and lateral femoral cartilage throughout the depth of cartilage. Similarly, PG intensity was relatively normal and very comparable between the MMD and sham animals 8 weeks postoperatively, but by 26 weeks postoperatively the PG intensity was significantly decreased throughout the medial tibial cartilage and in the superficial zone in all compartments. It has been reported previously that the average femoral cartilage thickness in immature rabbits is greater than 3 mm, but that thickness decreases to approximately 0.33 mm by approximately 12 weeks and remains nearly constant between 12 weeks and 18 months.19 The average cartilage thickness in the medial femur in our sham animals was 0.34 mm (± 0.05 mm), extremely similar to the values reported for normal rabbits,19 and indicating sham surgery had little effect on the cartilage. In contrast, medial femoral condyle cartilage thickness was 0.47 mm (± 0.09 mm) in the 8W-MMD animals and 0.44 mm (± 0.10 mm) in the 26W-MMD animals, and this same trend of an early increase in cartilage thickness, followed by a slight decrease was evident in all compartments. However, more relevant to PTOA than average cartilage thickness was the development of localized regions of cartilage thinning bordered by average thickness cartilage (Figure 4). These localized thinned areas were considered erosions and were found in none of the sham animals, one of the 8W-MMD animals, and four of the 26W-MMD animals.
Figure 3.
Quantitive histological data for all compartments of the rabbit stifle (knee) provided by the semi-automated Mankin scoring program. For every plot, open shapes are individual animal data, filled shapes are group averages and vertical bars are standard deviations. Statistical significance is indicated with p<0.05 (*), p<0.01 (**), p <0.001 (***). MMD caused progressive cell loss in the superficial zone that appeared to be progressive from 8 to 26 weeks. Cell loss deeper in the cartilage varied with time and compartment. PG loss was initially very similar between MMD and sham animals 8 weeks postoperatively, but was greater 26 weeks postoperatively.
Figure 4.

Representative histology images of the medial tibial plateau from the rabbits comprising the three different study groups used to establish PTOA progression. Images were selected from the n=8 images available per group and oriented with anterior on the left. The least severe damage in each group is on the top row, and the most severe damage is in the bottom row. Rows 2 and 3 are representative intermediate levels of degeneration most representative of the group as a whole. The most notable progression in overall joint damage is localized erosion (**) that began to become apparent in a few animals in the 8W-MMD group, but was more severe and present in more animals at 26 weeks. This feature was not well-captured by the automated histology scoring program, and is best appreciated by qualitative observation.
Chondrocyte Respiration
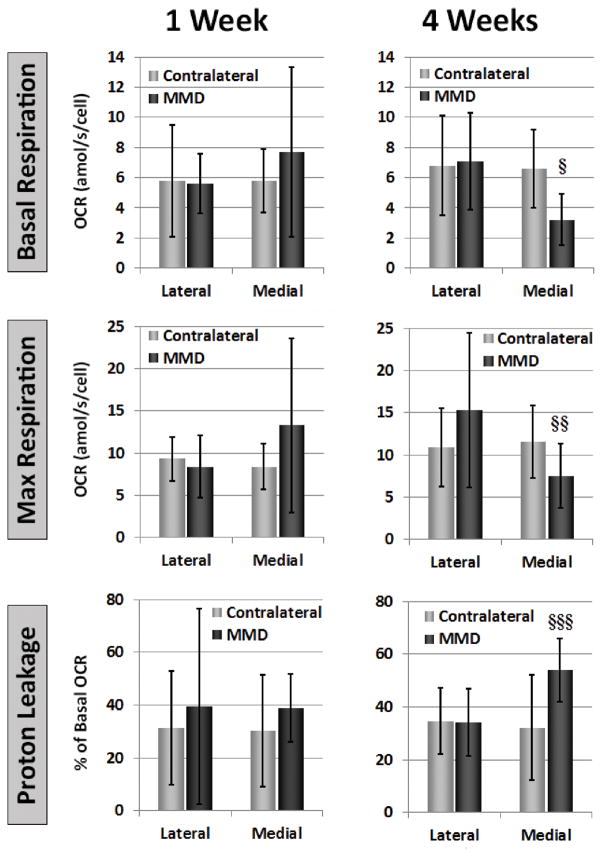
Mitochondrial stress tests from 1 week animals demonstrated a trend towards increased basal and maximum respiration in the medial tibia that did not reach significance given the high variance of the group (Figure 5). Similarly, proton leakage was somewhat elevated 1 week after injury in the MMD group relative to contralateral but this trend did not reach significance. By 4 weeks after injury, the medial surface of the tibia demonstrated significant (pairwise p values ranging 0.020 to 0.045) deficits in basal and maximum OCR (Figure 5) as well as a significant (pairwise p values ranging 0.011 to 0.029) increase in proton leakage by chondrocyte mitochondria. Two-way ANOVA indicated significant effects of compartment (p=0.029) and the interaction between compartment and injury (p=0.048) for basal respiration and significant interaction between compartment and injury (p=0.035) for maximal respiration. Similarly, two-way ANOVA indicated significant effects of injury (p=0.028) and the interaction between compartment and injury (p=0.022) for proton leakage. These differences in the medial tibial plateau surface of MMD stifles were largely not present in the lateral surfaces where disease is less prominent. While there was not a sham group for comparison of chondrocyte respiratory measures, there were no group-related differences in respiration by the contralateral joints, and interestingly, the values of each respiration measurement in the contralateral joints were similar to those made in normal joints of different animal species (data not shown).
Figure 5.
Average chondrocyte respiration measurements. Shaded columns represent group average values and error bars indicate standard deviations. Pairwise statistical comparison indicated differences only at 4 weeks between medial MMD and lateral MMD/lateral contralateral basal respiration (§ indicates p=0.045/0.020), between medial MMD and lateral MMD max respiration (§§ indicates p=0.030), and between medial MMD and medial contralateral/lateral MMD/lateral contralateral proton leakage (§§§ indicates p=0.011/p=0.023/p=0.029).
DISCUSSION
As in the initial report of the effects of medial meniscus destabilization,4 cartilage in the medial compartment of the rabbit stifle began to show degenerative changes 8 weeks after MMD. These degenerative changes progressed over the reported 26 week study period, resulting in significant decreases in cell density and development of cartilage cracks and focal erosions. Notably, the rabbits continued to use their operated limbs normally during the course of this progressive cartilage degeneration. Continued limb usage in combination with previous work demonstrating an increase in cartilage contact stress after MMD would indicate that the progressive cartilage degeneration was a direct result of chronic overloading. This indicates the MMD rabbit model is an appropriate in vivo system in which to investigate therapeutic approaches to PTOA targeting mechanobiological responses of chondrocytes. In agreement with in vitro studies,7 we observed time-dependent losses in chondrocyte mitochondrial function after surgery and were able to temporally locate these changes to several weeks post-injury but prior to tissue degeneration. It was noteworthy that these deficits did not manifest after 1 week while joints remain inflamed; instead they appeared by 4 weeks when joint inflammation had largely subsided, suggesting that chondrocyte mitochondria may be an early cell biological locus for accumulating mechanical damage over time.
While human PTOA patients will verbally complain of joint instability and associated pain, animals often indicate joint dysfunction or pain by changes in limb usage.20–23 Many animal species will load-protect a painful study limb, meaning that changes in cartilage health and appearance result from some combination of injury related factors and disuse factors. We found that early postoperative decreases in limb loading were very minimal, indicating that the animals did not find the surgical procedure highly debilitating or disruptive, which was important for attributing MMD-related cartilage changes to chronic overload. Similarly, over the duration of the study, limb loading characteristics were nearly identical on the operative and non-operative sides, indicating the animals were using their operated limbs in a nearly normal manner. One limitation to this analysis was that the Tekscan methodology did not permit studies of joint kinematics or instability, though this was deemed acceptable given that previous work has indicated MMD would result in minimal changes to joint kinematics.4; 11 Another limitation is that the limb loading data reported are only a measure of the animals’ limb usage when ambulating. This information does not describe the activity of the animal or any increase or decreases associated with surgery or disease progression, although regular veterinary monitoring did not indicate any dramatic changes in animal activity over the course of the study.
Unlike the initial report of the MMD in a rabbit,4 which reported average Mankin scores eight weeks after MMD ranging from 3 to 5 depending on knee compartment, the average Mankin scores in this work for the 8W-MMD group were milder, ranging from 1 to 3. The primary difference between the two studies was animal age at the time of MMD, with the animals in this work being younger than in the previous study.4 This would suggest it is very important to use fully mature animals for this MMD model to ensure adequate onset of more severe PTOA pathology. Although the 8W-MMD Mankin scores in this work were rather mild, there was a clear increase in Mankin scores from 8 to 26 weeks, indicating progression of cartilage damage. Furthermore, evaluation of changes in the quantified data, particularly measures like cell density, confirmed that cartilage was continuing to degenerate with increased postoperative time, while omitting all the shortcomings associated with scoring scales. This work was limited by the omission of a 26W-Sham group, which prevented determination of the effects of animal age and previous surgery on overall cartilage degeneration; although features such as erosions and chronically thickened cartilage would indicate MMD-induced overload was responsible for most of the cartilage changes seen at 26 weeks. Unfortunately, the algorithm for assessing histological changes is based on defining cartilage thickness as the distance between the bone/cartilage interface and the articular surface. There is no mechanism in place to determine if that thickness is normal or represents a region of erosion besides comparisons with normal rabbit cartilage thickness measurements, which vary by individual animal.18 Thus, the semi-automated objective scoring methodology did not allow for capturing grossly apparent changes in cartilage structure/thickness as are evident in Figure 4. So, while the Mankin and quantitative histological data indicate modest cartilage degeneration and progression with time, qualitative evaluation of the histological images indicates noteworthy degenerative changes (erosions).
The relatively prolonged period required to achieve definitive joint degeneration is both a strength and limitation of this model. Animal studies can be expensive, and models which require long-term survival are particularly so. However, like our model, PTOA in a clinical population that results from subtle joint injury causing chronic cartilage overloading requires many years to develop.24–26 So while time required to achieve end-stage disease in our model is long, the relative ease of the MMD procedure, the ability of the rabbits to tolerate it well, and the human-like slow progression of cartilage degeneration under chronic overload make the rabbit MMD model an excellent model for translational investigations of PTOA after subtle joint injury. In addition, the human-like progression of the rabbit MMD model appears particularly well suited to studies focused on developing early diagnosis and therapeutic techniques. Lastly, we have shown the model provides a vehicle for investigations of in vivo cellular behavior prior to the development of any macroscopic cartilage damage.
Studies have already suggested a pathogenic role for dysregulation of mitophagy in murine PTOA6 as well as persistent loss of mitochondrial function after mechanical overload in bovine explants.7 Similar dysfunction to that previously observed was incurred between 1 week and 4 weeks after MMD surgery, suggesting an accumulation of intracellular damage. In particular, increases in proton leakage despite overall deficits in mitochondrial function reveal a devolving mitochondrial environment with a resemblance to end stage OA.6; 27 This supports the hypothesis that mitochondria are a critical early intersection between chondrocyte biology and the cumulative effects of mechanical overloading. Demonstration of latency in these mitochondrial changes after subtle injury suggests a useful therapeutic window for intervention after injury or induction of overload. This is somewhat limited by a reliance on associated intact contralaterals as controls rather than data from another cohort of rabbits harvested 4 weeks after a sham surgical event. However, if induced, these metabolic changes may also provide diagnostic criteria amenable to MRI spectrometry or other emerging imaging modalities applied after the initial phases of severe inflammation have subsided but well prior to radiographic loss of joint space. Further study of the interactions between mitochondria, anabolic/catabolic balance, and the devolving joint environment of PTOA in this well-characterized rabbit model may reveal even more exciting opportunities for therapeutic intervention in this debilitating disease.
In conclusion, we present evidence that a MMD in a rabbit model is minimally disruptive to animal health, providing for a survival animal model of subtle injury that leads to localized chronic cartilage overloading. We confirmed that in a relatively short 8-week survival period, mild degenerative changes to cartilage are histologically evident, and these degenerative changes progress to significant loss of cell density and focal erosions over a 26 week survival period. We believe this evidence establishes the rabbit MMD model as a valid model of PTOA resulting from chronic overload. This presented the opportunity to confirm for the first time in an in vivo system that load-related impairments of mitochondrial function that have been found in vitro are in fact present in a living joint prior to histological appearance of disease. This rabbit MMD model provides a useful system in which to study pre-degenerative changes in cell metabolic health in a joint that has been proven to succumb to PTOA over time and a vehicle in which to investigate biological treatment strategies for interrupting the progressive degeneration of cartilage subjected to chronic overload.
Acknowledgments
This research was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health (P50 AR055533). The content is solely the responsibility of the authors and the sponsor did not have any role in study design, data interpretation, or in the writing of the manuscript.
Footnotes
Author Contributions: Goetz – Performed histological analysis, supervised gait analysis, analyzed and compiled data, wrote the manuscript
Coleman – Performed all short-term cell biological measures, analyzed and compiled data, wrote the manuscript
Fredericks – Assisted in development of MMD methodology, cared for animals, supervised gait analysis, wrote & edited the manuscript
Petersen – Cared for animals, performed gait analysis, wrote & edited the manuscript
Martin – Assisted with histological data interpretation, edited the manuscript
McKinley – Assisted with study design, assisted with development of MMD methodology, edited the manuscript
Tochigi – Designed the study, developed the MMD methodology, wrote the manuscript
All authors have read and approved the final manuscript.
References
- 1.Anderson DD, Chubinskaya S, Guilak F, et al. Post-traumatic osteoarthritis: improved understanding and opportunities for early intervention. J Orthop Res. 2011;29:802–809. doi: 10.1002/jor.21359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Buckwalter JA, Anderson DD, Brown TD, et al. The Roles of Mechanical Stresses in the Pathogenesis of Osteoarthritis: Implications for Treatment of Joint Injuries. Cartilage. 2013;4:286–294. doi: 10.1177/1947603513495889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Glasson SS, Blanchet TJ, Morris EA. The surgical destabilization of the medial meniscus (DMM) model of osteoarthritis in the 129/SvEv mouse. Osteoarthritis Cartilage. 2007;15:1061–1069. doi: 10.1016/j.joca.2007.03.006. [DOI] [PubMed] [Google Scholar]
- 4.Arunakul M, Tochigi Y, Goetz JE, et al. Replication of chronic abnormal cartilage loading by medial meniscus destabilization for modeling osteoarthritis in the rabbit knee in vivo. J Orthop Res. 2013;31:1555–1560. doi: 10.1002/jor.22393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Brophy RH, Martinez M, Borrelli J, Jr, Silva MJ. Effect of combined traumatic impact and radial transection of medial meniscus on knee articular cartilage in a rabbit in vivo model. Arthroscopy. 2012;28:1490–1496. doi: 10.1016/j.arthro.2012.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Carames B, Hasegawa A, Taniguchi N, et al. Autophagy activation by rapamycin reduces severity of experimental osteoarthritis. Ann Rheum Dis. 2012;71:575–581. doi: 10.1136/annrheumdis-2011-200557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Coleman MC, Ramakrishnan PS, Brouillette MJ, Martin JA. Injurious Loading of Articular Cartilage Compromises Chondrocyte Respiratory Function. Arthritis Rheumatol. 2015 doi: 10.1002/art.39460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Masoud I, Shapiro F, Kent R, Moses A. A longitudinal study of the growth of the New Zealand white rabbit: cumulative and biweekly incremental growth rates for body length, body weight, femoral length, and tibial length. J Orthop Res. 1986;4:221–231. doi: 10.1002/jor.1100040211. [DOI] [PubMed] [Google Scholar]
- 9.Laverty S, Girard CA, Williams JM, et al. The OARSI histopathology initiative - recommendations for histological assessments of osteoarthritis in the rabbit. Osteoarthritis Cartilage. 2010;18(Suppl 3):S53–65. doi: 10.1016/j.joca.2010.05.029. [DOI] [PubMed] [Google Scholar]
- 10.Beloozerova IN, Zelenin PV, Popova LB, et al. Postural control in the rabbit maintaining balance on the tilting platform. J Neurophysiol. 2003;90:3783–3793. doi: 10.1152/jn.00590.2003. [DOI] [PubMed] [Google Scholar]
- 11.Mansour JM, Wentorf FA, Degoede KM. In vivo kinematics of the rabbit knee in unstable models of osteoarthrosis. Ann Biomed Eng. 1998;26:353–360. doi: 10.1114/1.133. [DOI] [PubMed] [Google Scholar]
- 12.Gushue DL, Houck J, Lerner AL. Rabbit knee joint biomechanics: motion analysis and modeling of forces during hopping. J Orthop Res. 2005;23:735–742. doi: 10.1016/j.orthres.2005.01.005. [DOI] [PubMed] [Google Scholar]
- 13.Tochigi Y, Buckwalter JA, Brown TD. Toward improved clinical relevance of cartilage insult models in the rabbit knee: surgical access to the habitual weight-bearing region. Iowa Orthop J. 2013;33:196–201. [PMC free article] [PubMed] [Google Scholar]
- 14.van der Sluijs JA, Geesink RG, van der Linden AJ, et al. The reliability of the Mankin score for osteoarthritis. J Orthop Res. 1992;10:58–61. doi: 10.1002/jor.1100100107. [DOI] [PubMed] [Google Scholar]
- 15.Ostergaard K, Andersen CB, Petersen J, et al. Validity of histopathological grading of articular cartilage from osteoarthritic knee joints. Ann Rheum Dis. 1999;58:208–213. doi: 10.1136/ard.58.4.208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ostergaard K, Petersen J, Andersen CB, et al. Histologic/histochemical grading system for osteoarthritic articular cartilage: reproducibility and validity. Arthritis Rheum. 1997;40:1766–1771. doi: 10.1002/art.1780401007. [DOI] [PubMed] [Google Scholar]
- 17.Moussavi-Harami SF, Pedersen DR, Martin JA, et al. Automated objective scoring of histologically apparent cartilage degeneration using a custom image analysis program. J Orthop Res. 2009;27:522–528. doi: 10.1002/jor.20779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pedersen DR, Goetz JE, Kurriger GL, Martin JA. Comparative digital cartilage histology for human and common osteoarthritis models. Orthop Res Rev. 2013;2013:13–20. doi: 10.2147/ORR.S38400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Julkunen P, Harjula T, Iivarinen J, et al. Biomechanical, biochemical and structural correlations in immature and mature rabbit articular cartilage. Osteoarthritis Cartilage. 2009;17:1628–1638. doi: 10.1016/j.joca.2009.07.002. [DOI] [PubMed] [Google Scholar]
- 20.Safayi S, Miller JW, Wilson S, et al. Treadmill measures of ambulation rates in ovine models of spinal cord injury and neuropathic pain. J Med Eng Technol. 2016:1–8. doi: 10.3109/03091902.2015.1132786. [DOI] [PubMed] [Google Scholar]
- 21.Poole R, Blake S, Buschmann M, et al. Recommendations for the use of preclinical models in the study and treatment of osteoarthritis. Osteoarthritis Cartilage. 2010;18(Suppl 3):S10–16. doi: 10.1016/j.joca.2010.05.027. [DOI] [PubMed] [Google Scholar]
- 22.Matsuda K, Orito K, Amagai Y, et al. Swing time ratio, a new parameter of gait disturbance, for the evaluation of the severity of neuropathic pain in a rat model of partial sciatic nerve ligation. J Pharmacol Toxicol Methods. 2015;79:7–14. doi: 10.1016/j.vascn.2015.12.004. [DOI] [PubMed] [Google Scholar]
- 23.Orito K, Kurozumi S, Ishii I, et al. A sensitive gait parameter for quantification of arthritis in rats. J Pharmacol Sci. 2007;103:113–116. doi: 10.1254/jphs.sc0060156. [DOI] [PubMed] [Google Scholar]
- 24.Roos H, Lauren M, Adalberth T, et al. Knee osteoarthritis after meniscectomy: prevalence of radiographic changes after twenty-one years, compared with matched controls. Arthritis Rheum. 1998;41:687–693. doi: 10.1002/1529-0131(199804)41:4<687::AID-ART16>3.0.CO;2-2. [DOI] [PubMed] [Google Scholar]
- 25.Muthuri SG, McWilliams DF, Doherty M, Zhang W. History of knee injuries and knee osteoarthritis: a meta-analysis of observational studies. Osteoarthritis Cartilage. 2011;19:1286–1293. doi: 10.1016/j.joca.2011.07.015. [DOI] [PubMed] [Google Scholar]
- 26.Gelber AC, Hochberg MC, Mead LA, et al. Joint injury in young adults and risk for subsequent knee and hip osteoarthritis. Ann Intern Med. 2000;133:321–328. doi: 10.7326/0003-4819-133-5-200009050-00007. [DOI] [PubMed] [Google Scholar]
- 27.Gavriilidis C, Miwa S, von Zglinicki T, et al. Mitochondrial dysfunction in osteoarthritis is associated with down-regulation of superoxide dismutase 2. Arthritis Rheum. 2013;65:378–387. doi: 10.1002/art.37782. [DOI] [PubMed] [Google Scholar]