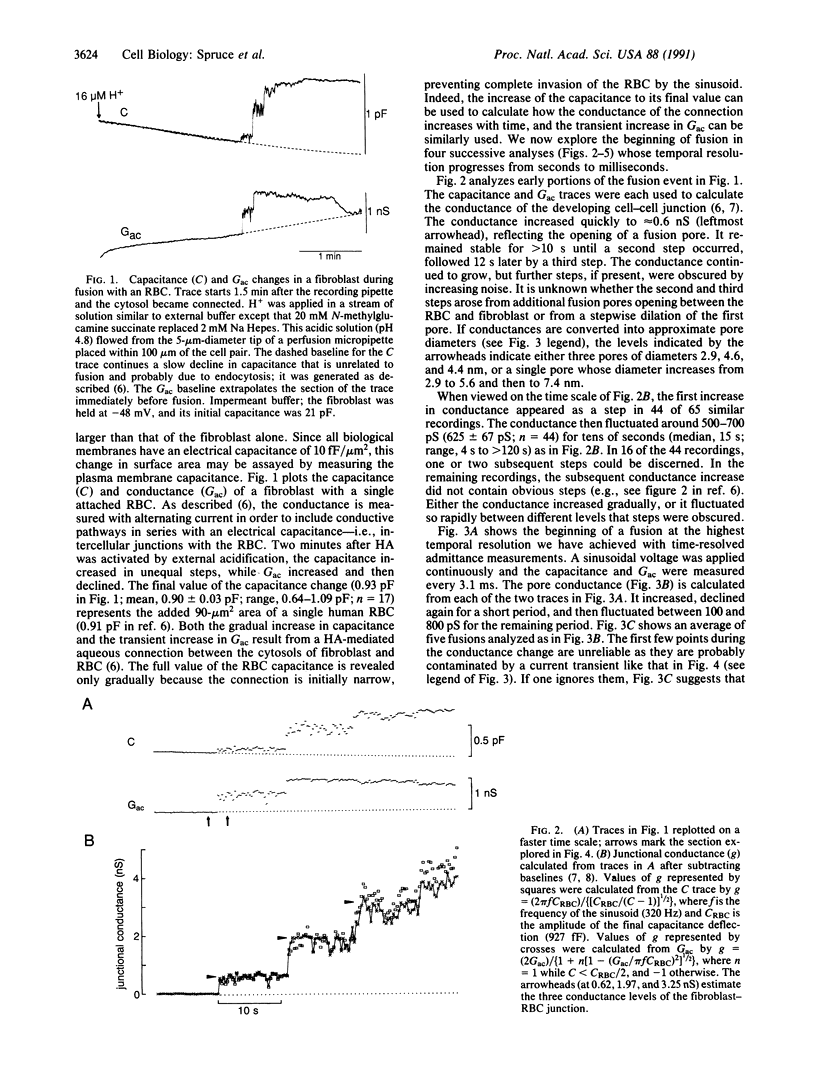
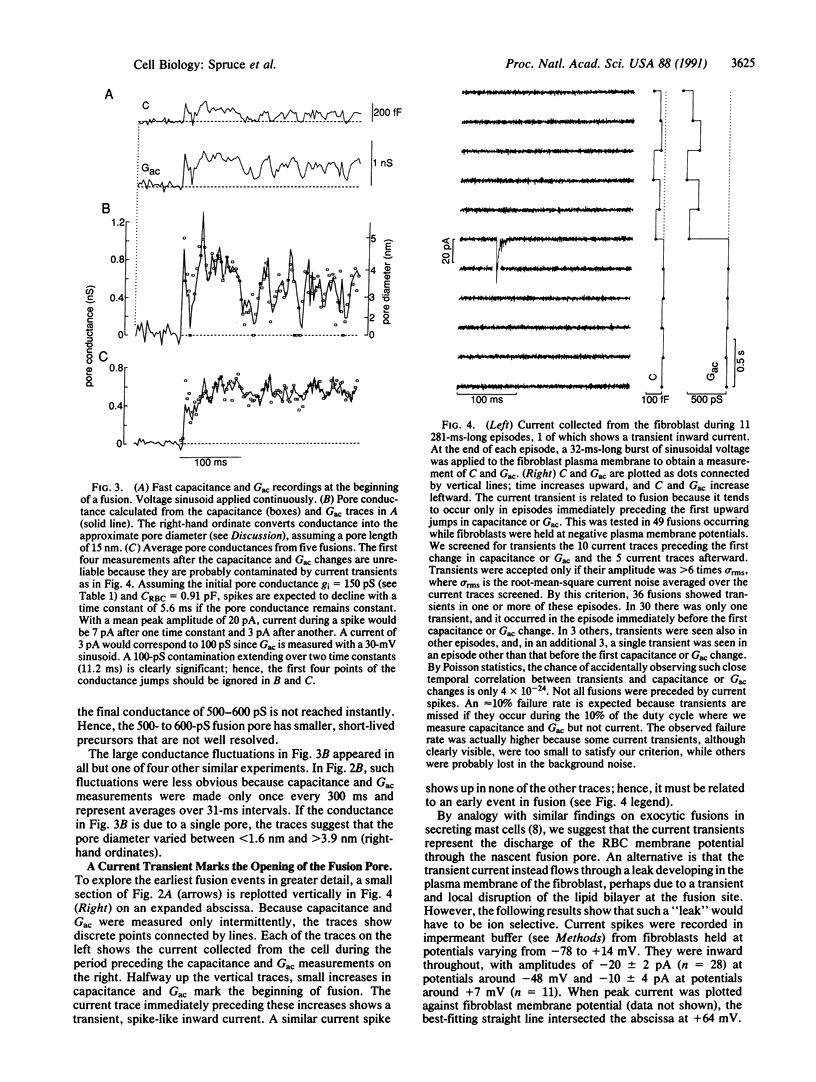
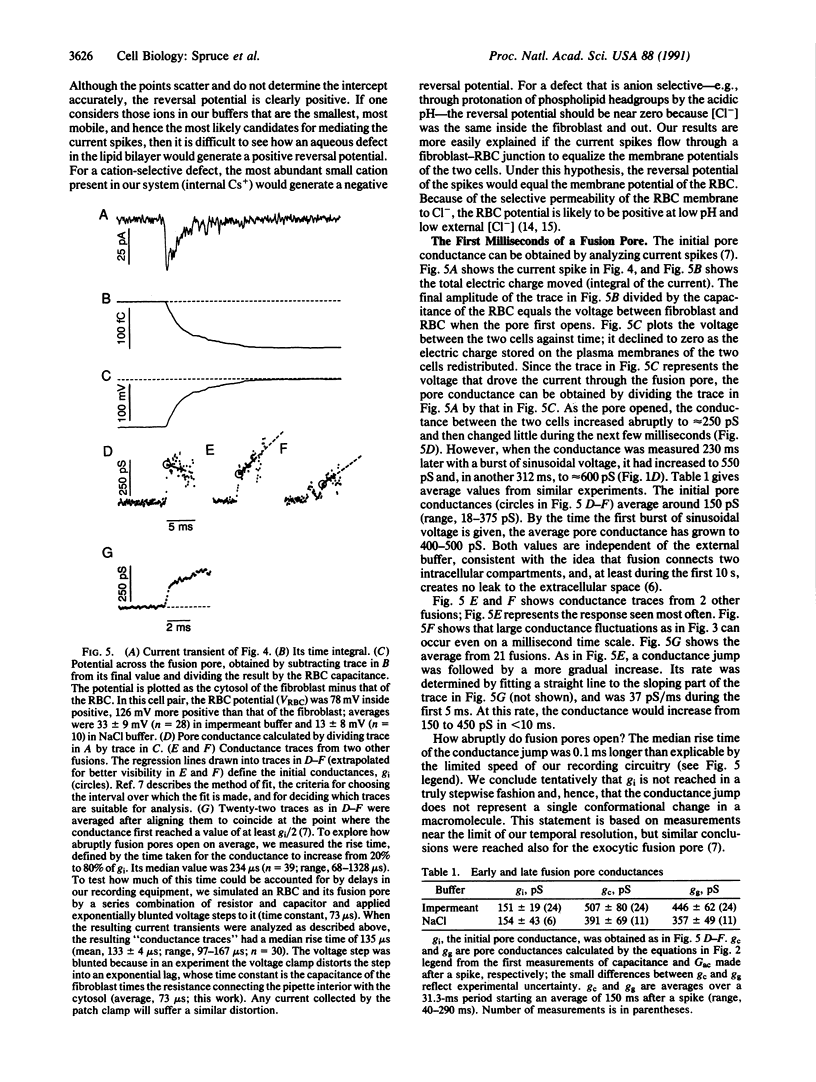
Abstract
Fibroblasts expressing the influenza virus hemagglutinin on their plasma membrane were patch clamped while they fused to erythrocytes. An increase in the fibroblast's membrane capacitance indicated the opening of the "fusion pore," the first aqueous connection between the fusing cells. We show here that the capacitance increase is preceded by a brief current transient, generated as the erythrocyte discharges its membrane potential through the nascent fusion pore. This signal allows one to calculate the pore conductance during the first milliseconds of its existence. The pore conductance jumps from 0 to approximately 150 pS and then grows more gradually over the subsequent tens of milliseconds until growth is arrested. The initial conductance is similar to that of a large ion channel and suggests that the pore is initially only 1-2 nm wide. Hence, we are probably observing events caused by only a small number of hemagglutinin molecules.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Breckenridge L. J., Almers W. Currents through the fusion pore that forms during exocytosis of a secretory vesicle. 1987 Aug 27-Sep 2Nature. 328(6133):814–817. doi: 10.1038/328814a0. [DOI] [PubMed] [Google Scholar]
- Colquhoun D., Sakmann B. Fast events in single-channel currents activated by acetylcholine and its analogues at the frog muscle end-plate. J Physiol. 1985 Dec;369:501–557. doi: 10.1113/jphysiol.1985.sp015912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doxsey S. J., Sambrook J., Helenius A., White J. An efficient method for introducing macromolecules into living cells. J Cell Biol. 1985 Jul;101(1):19–27. doi: 10.1083/jcb.101.1.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellens H., Bentz J., Mason D., Zhang F., White J. M. Fusion of influenza hemagglutinin-expressing fibroblasts with glycophorin-bearing liposomes: role of hemagglutinin surface density. Biochemistry. 1990 Oct 16;29(41):9697–9707. doi: 10.1021/bi00493a027. [DOI] [PubMed] [Google Scholar]
- Hamill O. P., Marty A., Neher E., Sakmann B., Sigworth F. J. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch. 1981 Aug;391(2):85–100. doi: 10.1007/BF00656997. [DOI] [PubMed] [Google Scholar]
- Henderson R., Unwin P. N. Three-dimensional model of purple membrane obtained by electron microscopy. Nature. 1975 Sep 4;257(5521):28–32. doi: 10.1038/257028a0. [DOI] [PubMed] [Google Scholar]
- Lassen U. V., Pape L., Vestergaard-Bogind B. Chloride conductance of the amphiuma red cell membrane. J Membr Biol. 1978 Feb 6;39(1):27–48. doi: 10.1007/BF01872753. [DOI] [PubMed] [Google Scholar]
- Neher E., Marty A. Discrete changes of cell membrane capacitance observed under conditions of enhanced secretion in bovine adrenal chromaffin cells. Proc Natl Acad Sci U S A. 1982 Nov;79(21):6712–6716. doi: 10.1073/pnas.79.21.6712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neyton J., Trautmann A. Physiological modulation of gap junction permeability. J Exp Biol. 1986 Sep;124:993–114. [PubMed] [Google Scholar]
- Neyton J., Trautmann A. Single-channel currents of an intercellular junction. 1985 Sep 26-Oct 2Nature. 317(6035):331–335. doi: 10.1038/317331a0. [DOI] [PubMed] [Google Scholar]
- Ruigrok R. W., Wrigley N. G., Calder L. J., Cusack S., Wharton S. A., Brown E. B., Skehel J. J. Electron microscopy of the low pH structure of influenza virus haemagglutinin. EMBO J. 1986 Jan;5(1):41–49. doi: 10.1002/j.1460-2075.1986.tb04175.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarkar D. P., Morris S. J., Eidelman O., Zimmerberg J., Blumenthal R. Initial stages of influenza hemagglutinin-induced cell fusion monitored simultaneously by two fluorescent events: cytoplasmic continuity and lipid mixing. J Cell Biol. 1989 Jul;109(1):113–122. doi: 10.1083/jcb.109.1.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwarzmann G., Wiegandt H., Rose B., Zimmerman A., Ben-Haim D., Loewenstein W. R. Diameter of the cell-to-cell junctional membrane channels as probed with neutral molecules. Science. 1981 Jul 31;213(4507):551–553. doi: 10.1126/science.7244653. [DOI] [PubMed] [Google Scholar]
- Spruce A. E., Breckenridge L. J., Lee A. K., Almers W. Properties of the fusion pore that forms during exocytosis of a mast cell secretory vesicle. Neuron. 1990 May;4(5):643–654. doi: 10.1016/0896-6273(90)90192-i. [DOI] [PubMed] [Google Scholar]
- Spruce A. E., Iwata A., White J. M., Almers W. Patch clamp studies of single cell-fusion events mediated by a viral fusion protein. Nature. 1989 Nov 30;342(6249):555–558. doi: 10.1038/342555a0. [DOI] [PubMed] [Google Scholar]
- Stegmann T., Doms R. W., Helenius A. Protein-mediated membrane fusion. Annu Rev Biophys Biophys Chem. 1989;18:187–211. doi: 10.1146/annurev.bb.18.060189.001155. [DOI] [PubMed] [Google Scholar]
- Veenstra R. D., DeHaan R. L. Cardiac gap junction channel activity in embryonic chick ventricle cells. Am J Physiol. 1988 Jan;254(1 Pt 2):H170–H180. doi: 10.1152/ajpheart.1988.254.1.H170. [DOI] [PubMed] [Google Scholar]
- White J. M. Viral and cellular membrane fusion proteins. Annu Rev Physiol. 1990;52:675–697. doi: 10.1146/annurev.ph.52.030190.003331. [DOI] [PubMed] [Google Scholar]
- Wiley D. C., Skehel J. J. The structure and function of the hemagglutinin membrane glycoprotein of influenza virus. Annu Rev Biochem. 1987;56:365–394. doi: 10.1146/annurev.bi.56.070187.002053. [DOI] [PubMed] [Google Scholar]



