Abstract
Although banned in most countries, dichlorodiphenyl-trichloroethane (DDT) continues to be used for vector control in some malaria endemic areas. Previous findings from the Center for the Health Assessment of Mothers and Children of Salinas (CHAMACOS) cohort study found increased prenatal levels of DDT and its breakdown product dichlorodiphenyl-dichloroethylene (DDE) to be associated with altered neurodevelopment in children at 1 and 2 years of age. In this study, we combined the measured maternal DDT/E concentrations during pregnancy obtained for the prospective birth cohort with predicted prenatal DDT and DDE levels estimated for a retrospective birth cohort. Using generalized estimating equation (GEE) and linear regression models, we evaluated the relationship of prenatal maternal DDT and DDE serum concentrations with children’s cognition at ages 7 and 10.5 years as assessed using the Full Scale Intelligence Quotient (IQ) and 4 subtest scores (Working Memory, Perceptual Reasoning, Verbal Comprehension, and Processing Speed) of the Wechsler Intelligence Scale for Children (WISC). In GEE analyses incorporating both age 7 and 10.5 scores (n = 619), we found prenatal DDT and DDE levels were not associated with Full Scale IQ or any of the WISC subscales (p-value >0.05). In linear regression analyses assessing each time point separately, prenatal DDT levels were inversely associated with Processing Speed at age 7 years (n = 316), but prenatal DDT and DDE levels were not associated with Full Scale IQ or any of the WISC subscales at age 10.5 years (n = 595). We found evidence for effect modification by sex. In girls, but not boys, prenatal DDE levels were inversely associated with Full Scale IQ and Processing Speed at age 7 years. We conclude that prenatal DDT levels may be associated with delayed Processing Speed in children at age 7 years and the relationship between prenatal DDE levels and children’s cognitive development may be modified by sex, with girls being more adversely affected.
1. Introduction
Dichlorodiphenyl-trichloroethane (DDT) is an organochlorine insecticide which was used worldwide in agriculture and vector control efforts until concerns about its environmental persistence and toxic effects on wildlife and humans led to usage restrictions and prohibitions (ATSDR, 2002; Rosenberg, 2004). In the United States, DDT was banned in 1972 except for use in emergency disease control (ATSDR, 2002). Agricultural DDT use ended in the mid-1970s in central Mexico, but in coastal regions DDT use continued until the 1990s for domestic food production (NACEC, 2001) and until 2000 for malaria control (Chanon et al., 2003). DDT was banned under the 2001 Stockholm Convention on Persistent Organic Pollutants for all uses except disease control (Rosenberg, 2004). In 2006, the World Health Organization endorsed the increased use of DDT for vector control in malaria endemic areas (World Health Organization, 2006). DDT is currently used for malaria control in at least 10 countries including Botswana, Democratic Republic of Congo, Gambia, India, Mozambique, Namibia, South Africa, Swaziland, Zambia, and Zimbabwe (World Health Organization, 2014).
Animal studies have shown that DDT and its breakdown product, dichlorodiphenyl-dichloroethylene (DDE), are neurodevelopmental toxicants (Craig and Ogilvie, 1974; Eriksson et al., 1990; Eriksson and Nordberg, 1986; Johansson et al., 1996; U.S. DHHS, 2002). DDT levels timed to sensitive periods of prenatal (Craig and Ogilvie, 1974) and neonatal (Eriksson et al., 1990; Eriksson and Nordberg, 1986; Johansson et al., 1996) nervous system development have been shown in mice to cause behavioral and neurochemical changes into adulthood.
Most studies conducted in humans have focused primarily on the neurodevelopmental toxicity of DDE rather than DDT exposure and results have been inconsistent. For example, no adverse associations were found between transplacental (scaled average of cord blood, placenta, and maternal blood) (Rogan et al., 1987) or breast milk DDE levels with performance on the Bayley Scales of Infant Development (BSID) at 6 to 24 months of age (Gladen et al., 1988; Rogan and Gladen, 1991) or on the McCarthy Scales of Children’s Abilities (MCSA) at ages 3, 4, and 5 years (Gladen and Rogan, 1991) in a large North Carolina birth cohort study recruited in the 1980s (n = 802), nor in a study of cord blood DDE levels and performance on the Neonatal Behavioral Assessment Scale (Stewart et al., 2000) or the Fagan Test of Infant Intelligence at 6 and 12 months (Darvill et al., 2000) in 141 newborns born in Oswego, New York between 1991 and 1994. However, other studies have found significant adverse associations, including in a small Spanish study of 13-month olds (n = 92) with relatively low cord serum DDE levels that reported inverse relationships with Mental Development Index (MDI), Psychomotor Development Index (PDI), and social development index on the BSID and Griffith Scales of Infant Development (Ribas-Fito et al., 2003), and in a Mexican study (n = 244) from the State of Morelos where DDT was used until 1998 that reported a relationship between prenatal DDE levels and the BSID PDI in infants up to 12-months old (Torres-Sanchez et al., 2007) and with general cognitive index, quantitative, verbal, and memory domains of the MSCA at 3.5 to 5 years old (Torres-Sánchez et al., 2013), although not with PDI or MDI at 30 months (Torres-Sánchez et al., 2009).
Only a few studies have measured serum concentrations of DDT in addition to DDE (DDT/E). In a large United States (US) pregnancy cohort study from 1959–1965 (n = 1100) during the time of peak DDT usage no adverse association was found between prenatal maternal DDT nor DDE levels and children’s BSID MDI or PDI scores at 8 months or Full-Scale Intelligence Quotient (IQ) on the Wechsler Intelligence Scale for Children (WISC) at 7 years of age (Jusko et al., 2012). In a cohort from Ribera d’Ebre and Menorca, Spain (n = 475), DDT levels measured in cord blood samples, but not DDE levels, were associated with poorer performance in general cognitive, quantitative, verbal, memory, and executive function domains of the MCSA (Ribas-Fito et al., 2006). Similarly, in our previous work from the Center for the Health Assessment of Mothers and Children of Salinas (CHAMACOS) cohort study (n = 360), comprised primarily of California women who had recently immigrated from Mexico, we reported significant inverse associations between prenatal DDT levels and PDI at 6 and 12 months and MDI at 12 and 24 months (Eskenazi et al., 2006) but also no association of prenatal DDE concentrations beyond 6 months (and then only on the PDI)(Eskenazi et al., 2006).
In the present study, we examined the relationship of prenatal maternal DDT and DDE serum concentrations and the cognitive development of CHAMACOS children at 7- and 10.5-years of age. Given that DDT and DDE are well-known endocrine disrupting chemicals with estrogenic and antiandrogenic properties, respectively (ATSDR, 2002), we examined whether associations with neurodevelopment differ by sex.
2. Methods
2.1. Study Population
Between 1999–2000, 601 pregnant women living in the agricultural Salinas Valley, California were enrolled in the initial CHAMACOS cohort (CHAM1) to investigate the health effects of pesticides and other environmental pollutants on pregnant women and their children. Women were recruited from Salinas Valley clinics providing prenatal care to low-income residents. Per eligibility criteria, CHAM1 recruitment was limited to women who were ≥18 years of age, <20 weeks of gestation, English- or Spanish-speaking, eligible for Medi-Cal (subsidized health care), and planning to deliver at Natividad Medical Center (NMC), the local county hospital. Of the 601 initially enrolled women, 526 both remained in the study at delivery and bore live-born singletons. Details of the CHAM1 cohort have been described previously (Eskenazi et al., 2003; Eskenazi et al., 2004).
In 2009–2011, when the CHAM1 children were 9 years old, an additional 309 mothers and their 9-year-old children (CHAM2) were enrolled into the CHAMACOS cohort. Eligibility criteria for CHAM2 participants mirrored those for CHAM1; namely, participant mothers needed to speak English or Spanish, and to have resided in Salinas Valley during their pregnancy with their singleton index child, qualified for Medi-Cal during pregnancy, received prenatal care, and been ≥18 years of age at time of delivery. CHAM2 participants were recruited via local schools, churches, libraries, food banks, newspaper/radio advertisements, and friends/relatives of CHAM1 participants.
2.2. Data Collection
Trained bilingual psychometricians assessed child neurodevelopment with the WISC, 4th edition at age 7 for CHAM1 children (mean age = 7.11 years, standard deviation (SD) = 0.23 years) and at age 10.5 for CHAM1 and CHAM2 children (mean age = 10.61 years, SD = 0.18 years). Children were assessed in their dominant language (English or Spanish), as ascertained via direct assessment. WISC tests were conducted in the CHAMACOS field office for the large majority of children, or in a quiet area of the child’s home for the minority of families who had moved outside the Salinas Valley. Children completed the following WISC subtests: Block Design, Digit Span, Coding, Letter-Number Sequencing, Matrix Reasoning, Symbol Search, Similarities, and Vocabulary. Subtest results were used to calculate Full Scale IQ scores as well as scaled scores for Verbal Comprehension, Perceptual Reasoning, Processing Speed, and Working Memory (mean = 100, SD = 15) (Wechsler, 2003). A single psychometrician administered all 7-year neurodevelopmental assessments and three psychometricians administered the 10.5-year assessments.
Maternal cognitive functioning was assessed when children were aged 6 months (CHAM1) and/or 9 years (CHAM1 and CHAM2) using the Peabody Picture Vocabulary Test (PPVT) – Revised (standardized in Spanish and English, mean = 100, SD = 15)(Dunn and Dunn, 1981). The home learning environment and quality of parent-child relationships was assessed with the Home Observation for Measurement of the Environment (HOME)(Caldwell and Bradley, 1984) at the 7- and 10.5-year visits. The test included maternal responses to parenting questions and observed parent-child interactions. Maternal depression was assessed using the Center for Epidemiological Studies Depression scale (CES-D)(Radloff, 1977) at the 7- and 9-year visits.
2.3. Serum Collection and DDT/E Analysis
For the majority of CHAM1 mothers, blood samples were collected via venipuncture in the 2nd trimester of pregnancy and, if unavailable, at delivery (Bradman et al., 2007). For the majority of CHAM2 (n=293) and a subset of CHAM1 participants (n=226), blood samples were also collected via venipuncture from mother and/or children approximately 9 years after delivery. Samples were frozen and shipped to the Center for Disease Control and Prevention (CDC) to be analyzed by gas chromatography-high resolution mass spectrometry for p,p′-DDT (hereafter, DDT) and p,p′-DDE (hereafter, DDE) (Sjödin et al., 2004). Serum levels of triglycerides and total cholesterol were measured using standard enzymatic methods (Roche Chemicals, Indianapolis, IN). DDT and DDE levels were expressed on a serum-lipid basis (nanograms per gram lipid) based on the total lipid levels using the summation method described by Phillips et al. (1989). DDT and DDE concentrations measured during pregnancy were above the limit of detection (LOD) in 99.6 and 100% of the samples, respectively. DDT serum values below the LOD (mean LOD = 1.9 ng/g-lipid) were assigned the LOD/√2.(Hornung and Reed, 1990) For two mothers with measured DDE but not DDT levels during pregnancy, values for DDT were imputed using a linear regression model with DDE as the independent variable.
2.4. Prediction of CHAM2 Prenatal Maternal DDT/E Serum Concentrations
ForCHAM2 mothers (n = 293) and a subset of CHAM1 mothers (n = 60) without measured maternal DDT/E concentrations during pregnancy, log10 prenatal DDT/E levels were predicted using log10 DDT/E concentrations in the mother and/or child measured when the child was 9 years old and using additional questionnaire data (e.g., years in the US, country of birth, maternal parity, and breastfeeding history). Prediction models were built with a subsample of CHAM1 mother/child pairs (n= 166) who had both measured maternal DDT/E concentrations during pregnancy and measured maternal and/or child levels when the child was 9-years old along with questionnaire data. To build the prediction models, we used the Super Learner algorithm, which is an ensemble machine learning technique that utilizes a weighted combination of algorithms to return a prediction function that minimized 10-fold cross-validated mean squared error (MSE) (van der Laan et al., 2007). In the subset of participants who had measured maternal DDT/E levels during pregnancy and repeated DDT/E measures in either the mother and child when the child was 9 years old, the Super Learner algorithm showed strong predictive ability with root MSEs ranging from 0.09 to 0.30 log ng/g and R2s ranging from 0.86 to 0.97 between measured and predicted log10-transformed prenatal DDT and DDE concentrations. Methods to back-extrapolate used in this analysis are described in Verner et al. (2015); however, to speed up computational time, we did not use the Deletion/Substitution/Addition (DSA) algorithm as a candidate Super Learner algorithm in this analysis. As the missing maternal DDT/E concentrations during delivery can be conceptualized as a missing data problem, we performed multiple imputations of the predicted DDT/E levels to capture the contribution of the variability in the DDT/E prediction on our assessment of the association between prenatal DDT/E and neurodevelopment. To accomplish this, we performed 100 bootstrap samples (sampling with replacement) of the CHAM1 data (n = 166) and built 100 Super Learner models in each of these datasets (Graham et al., 2007). Each of these 100 prediction models was used to predict the missing prenatal DDT/E levels resulting in 100 datasets containing both measured and predicted levels. We then evaluated the relationship between prenatal maternal DDT/E levels and children’s neurodevelopment in each of these datasets. Lastly, we combined the multiple imputation summary statistics (beta coefficients and standard errors) to calculate a single metric of association between prenatal DDT/E levels and child neurodevelopment based on methods described in Rubin (1987).
2.5. Data Analysis
DDT/E concentrations were log10-transformed to stabilize variance. Using chi-squared (categorical variables) and t-tests (continuous variables), we tested for differences in demographic characteristics and log10 DDT/E levels between: 1) the CHAM1 children who were followed and those lost to follow-up; and 2) the CHAM1 (followed) and CHAM2 study populations. Directed acyclic graphs were used to identify confounders and we adjusted all regression models for the following covariates: parity, maternal education at delivery, time lived in the U.S. at the time of the index child’s birth, country of birth, and age at delivery. Maternal intelligence and depression (dichotomized as CES-D score ≥ or < 16) (Lewinsohn et al., 1997), language of neurodevelopmental assessment, poverty status at the time of assessment (above or below poverty thresholds set by U.S. census bureau), child’s age and sex, and HOME-derived z-score at the time of assessment were also adjusted for because they are important determinants of child WISC scores (Ellis and Hennelly, 1980; Luster and Dubow, 1992; Neisser et al., 1996; Petterson and Albers, 2001; Yeates et al., 1983). The maternal PPVT score at the 6-months visit was used for the 7-year analysis, and the PPVT score at the 9-year visit was used for 10.5-year analysis. However, when a mother’s PPVT score from the preferred age point was missing, it was substituted with the score from the other age point (n≤8 such substitutions per analysis). Missing confounder data (<5%) was imputed based on each variable’s observed probability distribution.
We used generalized estimating equations (GEE) models with robust standard errors to assess associations between prenatal DDT/E levels and 7- and 10.5-year WISC scores simultaneously. We also used multiple linear regression models to test age-specific associations between prenatal DDT/E levels and WISC scores at 7 years and 10.5 years (i.e., with separate models for each age). Associations of DDT and DDE with WISC scores were assessed in separate models due to high collinearity of these two compounds (variable inflation factor > 5). To check the assumption that linear models were appropriate, we compared generalized additive models (GAM) with cubic splines to ordinary least square models using chi-square tests. GAM models did not fit the data better than linear models; therefore, only results from linear regression models are presented for age-specific analyses.
Insensitivity analyses, we explored the potential confounding effect of prenatal maternal levels to additional environmental neurotoxicants including organophosphate pesticides (measured as dialkyl phosphate (DAP) metabolites) (Bouchard et al., 2011; Engel et al., 2011), polybrominated diphenyl ethers (PBDEs) (Eskenazi et al., 2013; Herbstman et al., 2010), lead (Lanphear et al., 2005; Schnaas et al., 2006), and polychlorinated biphenyls (PCB) (Jacobson and Jacobson, 1996; Patandin et al., 1999). Each toxicant was included in separate models and also tested for interaction with DDT and DDE, but none of these toxicants were retained in the final model as the magnitude of the coefficients for DDT/E concentrations were not changed by >10% and the interaction terms were not significant (p-interaction > 0.10). We ran additional models including cross-product terms to test for effect modification attributable to:1) measured versus predicted prenatal DDT/E levels; 2) membership in the CHAM1 versus CHAM2 cohort; and 3) child sex. Main effects were considered statistically significant if the p-values were below < 0.05 based on a two-tailed test. All analyses were performed using the statistical program R, version 3.1.3 (R Core Team, 2013).
3. Results
Of the 316 children who completed the neurodevelopmental assessment at 7 years (all CHAM1), prenatal maternal DDT/E concentrations were measured in 256 and predicted in 60. Of the 595 completing the 10.5-year assessment (CHAM1 n = 302, CHAM2 n = 293), prenatal maternal DDT/E levels were measured in 244 and predicted in 351. Mothers (n = 619) of these children were predominantly Mexican-born (86%) and multiparous (67%); most were < 30 years old (71%) and had not completed high school (76%) at the time of delivery.
Compared to CHAM1 children who were lost to follow-up, CHAM1 children included in this analysis were more likely to be female, to have been born to older mothers, and to have breastfed longer (data not shown). However, prenatal DDT/E levels; maternal birthplace, time in U.S., parity, PPVT scores, and depression; household per person income; and HOME scores did not significantly differ between those followed and those not followed (results not shown). No differences in outcome or covariate characteristics were observed between CHAM1 and CHAM2 (Table 1), except that a higher percentage of CHAM2 mothers had “at risk” depression scores compared to CHAM1 mothers (p-value = 0.07).
Table 1.
Comparison between CHAM1 (n=302) and CHAM2 (n=292) demographic statisticsa
| Cohort Characteristics | CHAM1 n (%) |
CHAM2 n (%) |
p-valueb |
|---|---|---|---|
| Child intelligence (full-scale WISC score)c | 0.14 | ||
|
| |||
| ≤ 74 | 25 (8.3) | 24 (8.2) | |
| 75–99 | 211 (69.9) | 216 (74.0) | |
| ≥ 100 | 66 (21.9) | 52 (17.8) | |
|
| |||
| Maternal PPVT score | 0.17 | ||
|
| |||
| ≤ 74 | 61 (20.2) | 75 (25.6) | |
| 75–99 | 101 (33.4) | 92 (31.4) | |
| ≥ 100 | 140 (46.4) | 126 (43.0) | |
|
| |||
| Maternal education | 0.22 | ||
|
| |||
| ≤ 6th grade | 129 (42.7) | 121 (41.3) | |
| 7–12th grade | 109 (36.1) | 93 (31.7) | |
| ≥ high school graduate | 64 (21.2) | 79 (27.0) | |
|
| |||
| Maternal depression (CES-D ≥ 16)d,e | 0.07 | ||
|
| |||
| No | 229 (75.8) | 202 (68.9) | |
| Yes | 73 (24.2) | 91 (31.1) | |
|
| |||
| Maternal age at delivery (years) | 0.95 | ||
|
| |||
| 18–24 | 118 (39.1) | 119 (40.9) | |
| 25–29 | 105 (34.8) | 82 (28.2) | |
| 30–34 | 50 (16.6) | 58 (19.9) | |
| 35–45 | 29 (9.6) | 32 (11.0) | |
|
| |||
| Maternal country of birth | 0.81 | ||
|
| |||
| Mexico | 264 (87.4) | 259 (88.4) | |
| Other | 38 (12.6) | 34 (11.6) | |
|
| |||
| Maternal years in US prior to birth | 0.58 | ||
|
| |||
| ≤ 1 | 63 (20.9) | 56 (19.1) | |
| 2–5 | 83 (27.5) | 82 (28.0) | |
| 6–10 | 77 (25.5) | 68 (23.2) | |
| ≥ 11 | 48 (15.9) | 61 (20.8) | |
| Entire life | 31 (10.3) | 26 (8.9) | |
|
| |||
| Family income at 10.5 years | 0.42 | ||
|
| |||
| < poverty level | 86 (28.5) | 74 (25.3) | |
| > poverty level | 216 (71.5) | 219 (74.7) | |
|
| |||
| Parity prior to index child | 0.13 | ||
|
| |||
| 0 | 91 (30.1) | 106 (36.2) | |
| ≥1 | 211 (69.9) | 187 (63.8) | |
|
| |||
| Breastfeeding duration of index child | 0.68 | ||
|
| |||
| ≤ 2 months | 73 (24.2) | 71 (24.2) | |
| 2–12 months | 128 (42.4) | 115 (39.2) | |
| ≥ 12 months | 101 (33.4) | 107 (36.5) | |
|
| |||
| Sex of index child | 0.20 | ||
|
| |||
| Female | 140 (46.4) | 152 (51.9) | |
| Male | 162 (53.6) | 141 (48.1) | |
Participants who completed a neurodevelopmental assessment at the 10.5-year visit
p-value from t-test (variables = child intelligence, maternal PPVT scores, maternal age, and HOME z-score) or chi-square test between CHAM1 and CHAM2 study populations
Assessed at 10.5-year visit
Assessed at 9-year visit
Yes = CES-D score≥16
Distributions did not significantly differ (p-value > 0.05) between prenatal DDT/E concentrations that were measured (DDT/E geometric mean (GM) = 21.4 and 606.4 ng/g-lipid, respectively) versus predicted (DDT/E GM = 20.2 and 607.3 ng/g-lipid, respectively) (Table S1). Similarly, distributions of prenatal DDT/E concentrations did not differ significantly between CHAM1 (DDT/E GM = 21.3 and 604.5 ng/g-lipid, respectively) and CHAM2 (DDT/E GM = 20.1 and 609.6 ng/g-lipid, respectively). We found no evidence for effect modification (p-interaction > 0.1) based on whether prenatal DDT/E was measured or predicted or on membership in the CHAM1 versus CHAM2 cohort. Therefore, estimations using measured and predicted values from both cohorts are reported in the main text, but GEE and linear regression model results using only measured prenatal DDT/E levels are presented in the Supplemental Material.
In GEE analyses including both CHAM1 and CHAM2 children and WISC scores for both time points, prenatal DDT and DDE levels were not significantly associated with Full Scale IQ or WISC subscales (Table 2). In age-specific linear regression models using only the 7-year visit CHAM1 data, we found consistent inverse associations of prenatal DDT levels with Full Scale IQ and WISC subscales; however, only the associations for Processing Speed (β = −2.4; 95% CI: −4.5,−0.2; p = 0.03) and Verbal Comprehension (β = −1.9; 95% CI: −4.0,0.2; p = 0.08) reached or approached statistical significance. We found null associations between prenatal DDE levels and Full Scale IQ or any WISC subscales; however, in all cases except for Working Memory, the direction of the association was negative. In linear regression models using the 10.5-year data from both CHAM1 and CHAM2 children, we observed no associations of prenatal DDT or DDE levels with Full Scale IQ or WISC subscales.
Table 2.
Change in cognitive scores in children tested at 7 and/or 10.5-years for each 10-fold increase in prenatal DDT/E exposure using GEE and linear regression modelsa
| Cognitive test |
7 and/or 10.5-Years of Age (CHAM1 and CHAM2, n=619b)
|
||||
|---|---|---|---|---|---|
| DDT
|
DDE
|
||||
| Mean (SD)c | β (95% CI)d | p-value | β (95% CI) | p-value | |
|
|
|
||||
| WISC-IV scale | |||||
|
| |||||
| Working memory | 95.3 (12.0) | −1.0 (−2.5,0.5) | 0.19 | −0.5 (−2.3,1.3) | 0.56 |
|
| |||||
| Perceptual reasoning | 95.8 (15.7) | 0.0 (−1.7,1.8) | 0.97 | 0.2 (−1.8,2.3) | 0.82 |
|
| |||||
| Verbal comprehension | 92.3 (17.1) | 0.0 (−1.5,1.6) | 0.99 | 0.2 (−1.6,2.0) | 0.79 |
|
| |||||
| Processing speed | 101.5 (13.4) | −1.1 (−2.7,0.4) | 0.16 | −1.5 (−3.3,0.3) | 0.10 |
|
| |||||
| Full-scale IQ | 94.3 (13.8) | −0.5 (−2.0,1.0) | 0.54 | −0.3 (−2.1,1.5) | 0.75 |
|
| |||||
| Cognitive test | 7-Years of Age (CHAM1, n=316b) | ||||
|
| |||||
|
DDT
|
DDE
|
||||
| Mean (SD) | β (95% CI) | p-value | β (95% CI) | p-value | |
|
|
|
||||
| WISC-IV scale | |||||
|
| |||||
| Working memory | 93.8 (13.4) | −1.1 (−3.3,1.1) | 0.32 | 0.1 (−2.4,2.6) | 0.94 |
|
| |||||
| Perceptual reasoning | 102.9 (16.1) | −0.3 (−2.7,2.0) | 0.78 | −0.3 (−3.0,2.3) | 0.80 |
|
| |||||
| Verbal comprehension | 106.8 (16.7) | −1.9 (−4.0,0.2) | 0.08 | −1.8 (−4.2,0.6) | 0.14 |
|
| |||||
| Processing speed | 108.3 (13.2) | −2.4 (−4.5,−0.2) | 0.03 | −1.4 (−3.9,1.1) | 0.27 |
|
| |||||
| Full-scale IQ | 104.1 (14.2) | −1.7 (−3.9,0.5) | 0.14 | −1.2 (−3.8,1.3) | 0.33 |
|
| |||||
| Cognitive test | 10.5-Years of Age (CHAM1 and CHAM2, n=595b) | ||||
|
| |||||
|
DDT
|
DDE
|
||||
| Mean (SD) | β (95% CI) | p-value | β (95% CI) | p-value | |
|
|
|
||||
| WISC-IV scale | |||||
|
| |||||
| Working memory | 96.1 (11.1) | −1.0 (−2.6,0.6) | 0.21 | −0.9 (−2.8,0.9) | 0.33 |
|
| |||||
| Perceptual reasoning | 92.1 (14.2) | 0.2 (−1.6,2.1) | 0.79 | 0.6 (−1.5,2.8) | 0.56 |
|
| |||||
| Verbal comprehension | 84.6 (11.4) | 0.5 (−1.1,2.1) | 0.53 | 0.9 (−0.9,2.8) | 0.32 |
|
| |||||
| Processing speed | 98.2 (12.2) | −0.8 (−2.5,0.9) | 0.38 | −1.6 (−3.5,0.4) | 0.11 |
|
| |||||
| Full-scale IQ | 89.6 (10.8) | −0.2 (−1.8,1.4) | 0.81 | −0.1 (−1.9,1.8) | 0.95 |
Adjusted for maternal education, age, parity, PPVT scores, CES-D scores, birth country, and years in the U.S prior to delivery; HOME z-score; language of WISC testing; child sex and age at WISC testing; and household poverty.
Depending on WISC index, n ranged from 284–316 for 7-year analysis, 594–595 for the 10.5-year analysis, and 616–619 for the GEE analysis
Mean and standard deviation (SD) of WISC indexes. For GEE, average of 7- and 10.5-year visits.
β= beta coefficient on regression models, CI= confidence interval
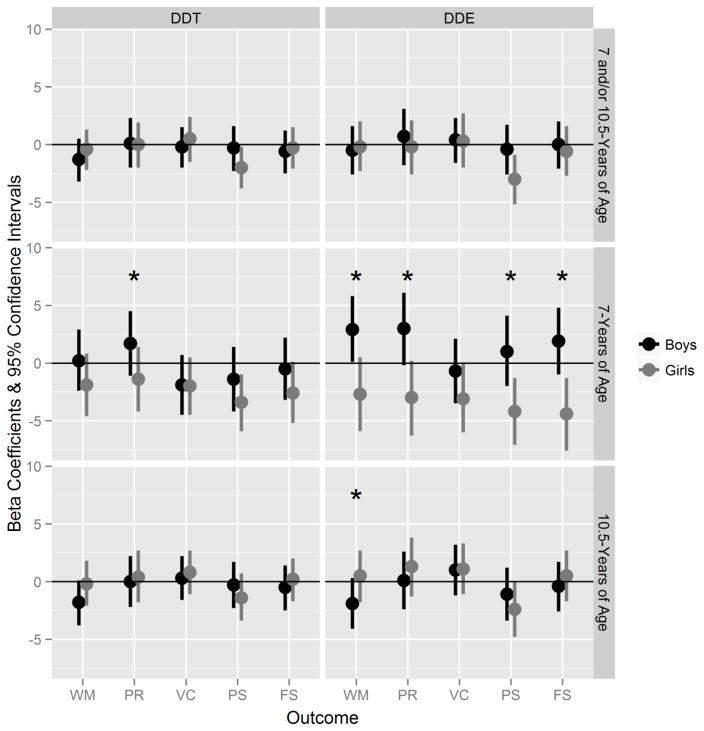
In GEE analyses, we observed no effect modification by sex on associations between prenatal DDT or DDE levels and any outcomes (Figure 1 and Table S3). In age-specific regression analyses, we found that the association between prenatal DDT levels and Perceptual Reasoning at age 7 differed significantly between boys and girls (p-interaction = 0.08, Table S4). More specifically, prenatal DDT levels showed a negative association with Perceptual Reasoning scores in girls (β = −1.4; 95% CI: −4.2, 1.4, p = 0.32), but a positive association in boys (β = 1.7; 95% CI: −1.1, 4.5, p = 0.24); however, neither association was significant. We found that the association between prenatal DDE levels and Working Memory, Perceptual Reasoning, Processing Speed and Full Scale IQ at age 7 differed significantly between boys and girls (p-interaction < 0.10, Table S4). Specifically, when we stratified by sex, we observed significant inverse associations between prenatal DDE levels and Verbal Comprehension (β = −3.1; 95% CI: −6.0,−0.1, p = 0.04), Processing Speed (β = −4.2; 95% CI: −7.1,−1.3, p < 0.01) and Full Scale IQ (β = −4.4; 95% CI: −7.6,−1.3, p = 0.01) in girls but not in boys. We also observed non-significant inverse associations between prenatal DDE levels and Working Memory and Perceptual Reasoning in girls but positive associations in boys. At age 10.5 years, associations between prenatal DDE levels and Working Memory scores were significantly different between boys and girls (p-interaction = 0.03) (Table S5). In contrast to the 7-year results, we found borderline-significant inverse associations of prenatal DDT/E levels with Working Memory at 10.5 years for boys only (DDT: β = −1.8; 95% CI: −3.8, 0.1, p = 0.06; DDE: β = −1.9; 95% CI: −4.1, 0.3, p = 0.10).
Figure 1.
Change in cognitive scores for each 10-fold increase in prenatal DDT/E serum concentrations in children tested at 7 and/or 10.5-years, stratified by sex. Models adjusted for maternal education, age, parity, PPVT scores, CES-D scores, birth country, and years in the U.S prior to delivery; HOME z-score; language of WISC testing; child age at WISC testing; and household poverty. Vertical lines with middle dots indicate 95% confidence intervals and beta coefficients, respectively, for the GEE or linear models. Asterisks (*) indicate the p-value for the interaction term is <0.10. Outcome key: WM=Working Memory, PR = Perceptual Reasoning, VC = Verbal Comprehension, PS = Processing Speed, FS = Full Scale IQ.
4. Discussion
In this study, we combined data from two similar cohorts, one prospective and one retrospective, to investigate the relationship between prenatal maternal DDT and DDE levels and cognitive development in children at ages 7 and 10.5 years as assessed by the WISC IV. In analyses including all children and cognitive outcomes from both time points (GEE analyses), we observed null associations between prenatal DDT and DDE levels and Full Scale IQ and any of the WISC subscales. In age-specific analyses, prenatal DDT levels were inversely associated with Processing Speed at age 7, but neither prenatal DDT nor DDE levels were associated with Full Scale IQ or any of the WISC subscales at age 10.5. We found evidence for effect modification by sex at age 7 with girls exhibiting stronger inverse associations than boys between prenatal DDE levels and all intelligence metrics except Verbal Comprehension.
Only one previous study has measured prenatal levels of both DDT and DDE and followed the neurodevelopmental functioning of children until grade school. Similar to our results, Jusko et al. (2012) found prenatal DDT and DDE levels to not be associated with Full Scale IQ scores at 7 years in a highly exposed historical cohort (median serum DDT = 1,100 and DDE = 3,000 ng/g-lipid). However, the associations between DDT/E and WISC subscales were not evaluated.
In the present study, we observed an inverse association between prenatal DDE levels and Processing Speed at age 7 years. Impaired Processing Speed may be a risk factor for attention and learning problems (Calhoun and Mayes, 2005; Shanahan et al., 2006) and the associations that we observed between prenatal DDE levels and reduced Processing Speed are in accordance with previous research showing prenatal DDE levels to be associated with attention problems (Sage et al., 2010; Siren et al., 2013). For example, Sage et al. (2010) found higher risk for attention-deficit/hyperactivity disorder-like behaviors in 7–11 year old children (n=607) with higher prenatal maternal levels of DDE.
We found evidence for effect modification by sex as prenatal DDE levels were inversely associated with Processing Speed and Full Scale IQ at age 7 in girls, but not boys. Of the four previous studies that examined effect modification by sex on prenatal levels to DDT or DDE and cognitive development in children (Eskenazi et al., 2006; Ribas-Fito et al., 2006; Torres-Sánchez et al., 2009; Torres-Sánchez et al., 2013), only two reported a significant interaction (Eskenazi et al., 2006; Ribas-Fito et al., 2006). Notably, the later two studies reported effect modification with DDT isomers, not DDE. Although we did not previously find sex modified the relationship between p,p′-DDT and cognitive development in CHAMACOS children up to 24-months old, we found a significant inverse association between prenatal o,p′-DDT levels and PDI scores at 12-months in boys but not girls in the CHAMACOS cohort (Eskenazi et al., 2006). In contrast to our previous findings but supporting our current study’s results of girls more adversely affected by prenatal exposure, Ribas-Fito et al. (2006) observed stronger inverse associations between p,p′-DDT and General Cognition, Verbal Memory, and Working Memory on the MSCA in girls than boys at 4 years of age. As sex modified the relationship between prenatal DDE, not DDT, levels and WISC scores, our findings support the hypothesis of sexually dimorphic effects of androgen receptor antagonists on the developing brain (Parent et al., 2011; Zhang et al., 2008). Nevertheless, given the inconsistent findings of effect modification by sex between our study and previous studies, further research is needed.
Although most associations were not significant, we found consistent inverse associations of prenatal DDT/E levels with most IQ metrics assessed at age 7, but we did not observe consistent associations in the 10.5-year data. This “washing out” of the association may be due to the effect of puberty on IQ, as pubertal development has been associated with differences in IQ (Galatzer et al., 1984; Judith Semon et al., 1991; Karlsson, 1990). Because timing of puberty may be on the causal pathway of DDT effects on IQ (Den Hond and Schoeters, 2006; Roy et al., 2009), we did not adjust for puberty in our analyses. Alternatively, the lack of associations at age 10.5 may indicate that the effects of prenatal DDT/E levels on cognitive development are not long lasting and could have diminished by the time the children reach age 10.5. When we examined only those with measured rather than predicted prenatal DDT/E levels, the finding of no associations at age 10.5 suggests that the lack of association is not due to selection bias or measurement error induced by including the CHAM2 participants or those with predicted levels.
We used a novel approach to predict the prenatal DDT and DDE levels of half the cohort. This is the first study to predict prenatal DDT levels as an exposure or assess IQ as an outcome of the prediction. Karmaus et al. (2004) conducted the only previous study to back-extrapolate prenatal DDE levels (Karmaus et al., 2004). In this study, repeated PCB blood measurements in non-pregnant women sampled ~10 years apart were used to develop linear regression models to predict past and future DDE serum levels (Karmaus et al., 2004). Using these prediction models, researchers found prenatal DDE exposure to be associated with decreased initiation/duration of breastfeeding, decreased age of menarche, and increased weight gain and body mass index in the offspring 20–50 years after birth (Karmaus et al., 2005; Karmaus et al., 2009; Vasiliu et al., 2004). We believe that our back-extrapolation method has potentially less prediction error than Karmaus et al.’s methods because our models were built based on: 1) repeated DDT/E levels; 2) blood collected from pregnant women; and 3) cross-validation techniques to prevent overfitting of the model.
Strengths of the present study include its longitudinal design with the WISC administered at two time points. Important confounders were also assessed at multiple time points and adjusted for in the models. The innovative use of back-extrapolation of prenatal DDT/E levels allowed for increased sample size to investigate health effects from early life exposures. CHAM1 and CHAM2 had similar demographic and exposure characteristics, giving strength to the assumption that both cohorts came from the same target population and can reasonably be combined. In addition, we were able to investigate for potential confounding by additional suspected neurotoxicants (DAPs, PBDEs, lead, and PCBs). Limitations include not having measured prenatal DDT/E levels on all study participants. In addition, for the 10.5-year analysis, more predicted (n = 351) than measured (n = 244) prenatal DDT/E levels were used in the analyses. However, the Super Learner algorithm showed strong predictive ability (RMSEs 0.09–0.30 log ng/g; R2s 0.86–0.97) and we accounted for the uncertainty of the prediction of prenatal levels with multiple imputations (Graham et al., 2007). Despite this, error in the prediction may have influenced the study results. Finally, we investigated the relationship of DDT and DDE concentrations with IQ metrics in separate models due to collinearity, which makes evaluating the true effect of each compound difficult to elucidate.
5. Conclusion
We examined prenatal maternal DDT and DDE blood concentrations in relation to children’s neurodevelopment at 7 and 10.5 years. We found evidence that prenatal exposure to DDT may be associated with delayed Processing Speed in children at age 7 years. We also found evidence that the relationship between prenatal DDE exposure and cognitive development may be modified by sex, with girls being more adversely affected.
Supplementary Material
Highlights.
We combined a prospective and retrospective birth-cohort
Prenatal DDT and DDE levels were both measured and predicted
We assessed children’s intelligence at two time points
We found an association between prenatal DDT levels and Processing Speed at age 7 years
Acknowledgments
We thank the CHAMACOS staff, students, community partners, and participants and families, as well as Nina Holland PhD and biorepository staff for their assistance in specimen management and Asa Bradman PhD for his contribution in formulating the study design and methods. Support for this research was provided by the National Institutes of Health (National Institute of Environmental Health Sciences) grant numbers:P01 ES0009605, R01ES017054, and R01ES015572; and by the U.S. Environmental Protection Agency (EPA) grant numbers: R82670901, RD83171001, and RD83451301. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the funders.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- ATSDR. Public Health. 2002. Toxicological Profile For DDT, DDE, and DDD. [PubMed] [Google Scholar]
- Bouchard MF, Chevrier J, Harley KG, Kogut K, Vedar M, Calderon N, et al. Prenatal exposure to organophosphate pesticides and IQ in 7-year-old children. Environ Health Perspect. 2011;119:1189–95. doi: 10.1289/ehp.1003185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradman AS, Schwartz JM, Fenster L, Barr DB, Holland NT, Eskenazi B. Factors predicting organochlorine pesticide levels in pregnant Latina women living in a United States agricultural area. J Expo Sci Environ Epidemiol. 2007;17:388–99. doi: 10.1038/sj.jes.7500525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caldwell B, Bradley R. Home observation for measurement of the environment. Little Rock, AR: University of Arkansas; 1984. [Google Scholar]
- Calhoun SL, Mayes SD. Processing speed in children with clinical disorders. Psychology in the Schools. 2005;42:333–43. [Google Scholar]
- Chanon KE, Mendez-Galvan JF, Galindo-Jaramillo JM, Olguin-Bernal H, Borja-Aburto VH. Cooperative actions to achieve malaria control without the use of DDT. International journal of hygiene and environmental health. 2003;206:387–94. doi: 10.1078/1438-4639-00235. [DOI] [PubMed] [Google Scholar]
- Craig GR, Ogilvie DM. Alteration of t-maze performance in mice exposed to DDT during pregnancy and lactation. Environ Physiol Biochem. 1974;4:189–99. [PubMed] [Google Scholar]
- Darvill T, Lonky E, Reihman J, Stewart P, Pagano J. Prenatal exposure to PCBs and infant performance on the fagan test of infant intelligence. Neurotoxicology. 2000;21:1029–38. [PubMed] [Google Scholar]
- Den Hond E, Schoeters G. Endocrine disrupters and human puberty. Int J Androl. 2006;29:264–71. doi: 10.1111/j.1365-2605.2005.00561.x. discussion 86–90. [DOI] [PubMed] [Google Scholar]
- Dunn LM, Dunn LM. Peabody Picture Vocabulary Test-Revised: PPVT-R. American Guidance Service; 1981. [Google Scholar]
- Ellis NC, Hennelly RA. A bilingual word-length effect: Implications for intelligence testing and the relative ease of mental calculation in Welsh and English. British Journal of Psychology. 1980;71:43–51. [Google Scholar]
- Engel SM, Wetmur J, Chen J, Zhu C, Barr DB, Canfield RL, et al. Prenatal exposure to organophosphates, paraoxonase 1, and cognitive development in childhood. Environ Health Perspect. 2011;119:1182–8. doi: 10.1289/ehp.1003183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eriksson P, Nilsson-Hakansson L, Nordberg A, Aspberg A, Fredriksson A. Neonatal exposure to DDT and its fatty acid conjugate: effects on cholinergic and behavioural variables in the adult mouse. Neurotoxicology. 1990;11:345–54. [PubMed] [Google Scholar]
- Eriksson P, Nordberg A. The effects of DDT, DDOH-palmitic acid, and a chlorinated paraffin on muscarinic receptors and the sodium-dependent choline uptake in the central nervous system of immature mice. Toxicol Appl Pharmacol. 1986;85:121–7. doi: 10.1016/0041-008x(86)90105-5. [DOI] [PubMed] [Google Scholar]
- Eskenazi B, Bradman A, Gladstone EA, Jaramillo S, Birch K, Holland N. CHAMACOS, A Longitudinal Birth Cohort Study: Lessons from the Fields. Journal of Children’s Health. 2003;1:3–27. [Google Scholar]
- Eskenazi B, Chevrier J, Rauch SA, Kogut K, Harley KG, Johnson C, et al. In utero and childhood polybrominated diphenyl ether (PBDE) exposures and neurodevelopment in the CHAMACOS study. Environ Health Perspect. 2013;121:257–62. doi: 10.1289/ehp.1205597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eskenazi B, Harley K, Bradman A, Weltzien E, Jewell NP, Barr DB, et al. Association of in utero organophosphate pesticide exposure and fetal growth and length of gestation in an agricultural population. Environ Health Perspect. 2004;112:1116–24. doi: 10.1289/ehp.6789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eskenazi B, Marks AR, Bradman A, Fenster L, Johnson C, Barr D, et al. In utero exposure to dichlorodiphenyltrichloroethane (DDT) and dichlorodiphenyldichloroethylene (DDE) and neurodevelopment among young Mexican American children. Pediatrics. 2006;118:233–41. doi: 10.1542/peds.2005-3117. [DOI] [PubMed] [Google Scholar]
- Galatzer A, Beth-Halachmi N, Kauli R, Laron Z. Intellectual function of girls with precocious puberty. Pediatrics. 1984;74:246–9. [PubMed] [Google Scholar]
- Gladen BC, Rogan WJ. Effects of perinatal polychlorinated biphenyls and dichlorodiphenyl dichloroethene on later development. J Pediatr. 1991;119:58–63. doi: 10.1016/s0022-3476(05)81039-x. [DOI] [PubMed] [Google Scholar]
- Gladen BC, Rogan WJ, Hardy P, Thullen J, Tingelstad J, Tully M. Development after exposure to polychlorinated biphenyls and dichlorodiphenyl dichloroethene transplacentally and through human milk. J Pediatr. 1988;113:991–5. doi: 10.1016/s0022-3476(88)80569-9. [DOI] [PubMed] [Google Scholar]
- Graham JW, Olchowski AE, Gilreath TD. How many imputations are really needed? Some practical clarifications of multiple imputation theory. Prevention science: the official journal of the Society for Prevention Research. 2007;8:206–13. doi: 10.1007/s11121-007-0070-9. [DOI] [PubMed] [Google Scholar]
- Herbstman JB, Sjodin A, Kurzon M, Lederman SA, Jones RS, Rauh V, et al. Prenatal exposure to PBDEs and neurodevelopment. Environ Health Perspect. 2010;118:712–9. doi: 10.1289/ehp.0901340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hornung RW, Reed LD. Estimation of Average Concentration in the Presence of Nondetectable Values. Applied Occupational and Environmental Hygiene. 1990;5:46–51. [Google Scholar]
- Jacobson JL, Jacobson SW. Intellectual impairment in children exposed to polychlorinated biphenyls in utero. New England Journal of Medicine. 1996;335:783–9. doi: 10.1056/NEJM199609123351104. [DOI] [PubMed] [Google Scholar]
- Johansson U, Fredriksson A, Erickson LL. Low-dose effects of paraoxon in adult mice exposed neonatally to DDT: changes in behavioral and cholinergic receptor variables. Environmental Toxicology and Pharmacology. 1996;2:307–14. doi: 10.1016/s1382-6689(96)00062-2. [DOI] [PubMed] [Google Scholar]
- Judith Semon D, Graber JA, Petersen AC. The Effects of Pubertal Development on Achievement during Adolescence. American Journal of Education. 1991;99:444–60. [Google Scholar]
- Jusko TA, Klebanoff MA, Brock JW, Longnecker MP. In-utero exposure to dichlorodiphenyltrichloroethane and cognitive development among infants and school-aged children. Epidemiology. 2012;23:689–98. doi: 10.1097/EDE.0b013e31825fb61d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karlsson J. Time of Puberty Onset and Intellectual and Neuropsychological Functioning. In: Holmes C, editor. Psychoneuroendocrinology. Springer; New York: 1990. [Google Scholar]
- Karmaus W, Davis S, Fussman C, Brooks K. Maternal concentration of dichlorodiphenyl dichloroethylene (DDE) and initiation and duration of breast feeding. Paediatric and perinatal epidemiology. 2005;19:388–98. doi: 10.1111/j.1365-3016.2005.00658.x. [DOI] [PubMed] [Google Scholar]
- Karmaus W, Fussman C, Muttineni J, Zhu X. Backward estimation of exposure to organochlorines using repeated measurements. Environ Health Perspect. 2004;112:710–6. doi: 10.1289/ehp.6761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karmaus W, Osuch JR, Eneli I, Mudd LM, Zhang J, Mikucki D, et al. Maternal levels of dichlorodiphenyl-dichloroethylene (DDE) may increase weight and body mass index in adult female offspring. Occupational and environmental medicine. 2009;66:143–9. doi: 10.1136/oem.2008.041921. [DOI] [PubMed] [Google Scholar]
- Lanphear BP, Hornung R, Khoury J, Yolton K, Baghurst P, Bellinger DC, et al. Low-level environmental lead exposure and children’s intellectual function: an international pooled analysis. Environmental Health Perspectives. 2005;113:894. doi: 10.1289/ehp.7688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewinsohn PM, Seeley JR, Roberts RE, Allen NB. Center for Epidemiologic Studies Depression Scale (CES-D) as a screening instrument for depression among community-residing older adults. Psychology and Aging. 1997;12:277–87. doi: 10.1037//0882-7974.12.2.277. [DOI] [PubMed] [Google Scholar]
- Luster T, Dubow E. Home Environment and Maternal Intelligence as Predictors of Verbal Intelligence: A Comparison of Preschool and School-Age Children. Merrill-Palmer Quarterly (1982-) 1992;38:151–75. [Google Scholar]
- NACEC. History of DDT in North America to 1997. Montreal: North American Commission for Environmental Cooperation; 2001. [Google Scholar]
- Neisser U, Boodoo G, Bouchard TJ, Jr, Boykin AW, Brody N, Ceci SJ, et al. Intelligence: knowns and unknowns. American psychologist. 1996;51:77. [Google Scholar]
- Parent A-S, Naveau E, Gerard A, Bourguignon J-P, Westbrook GL. Early developmental actions of endocrine disruptors on the hypothalamus, hippocampus, and cerebral cortex. Journal of Toxicology and Environmental Health, Part B. 2011;14:328–45. doi: 10.1080/10937404.2011.578556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patandin S, Lanting CI, Mulder PGH, Boersma ER, Sauer PJJ, Weisglas-Kuperus N. Effects of environmental exposure to polychlorinated biphenyls and dioxins on cognitive abilities in Dutch children at 42 months of age. The Journal of pediatrics. 1999;134:33–41. doi: 10.1016/s0022-3476(99)70369-0. [DOI] [PubMed] [Google Scholar]
- Petterson SM, Albers AB. Effects of Poverty and Maternal Depression on Early Child Development. Child Development. 2001;72:1794–813. doi: 10.1111/1467-8624.00379. [DOI] [PubMed] [Google Scholar]
- Phillips DL, Pirkle JL, Burse VW, Bernert JT, Jr, Henderson LO, Needham LL. Chlorinated hydrocarbon levels in human serum: effects of fasting and feeding. Arch Environ Contam Toxicol. 1989;18:495–500. doi: 10.1007/BF01055015. [DOI] [PubMed] [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; Vienna, Austria: 2013. http://www.R-project.org/ [Google Scholar]
- Radloff L. The CES-D Scale: A Self-Report Depression Scale for Research in the General Population. Applied Psycological Measurement. 1977;1:385–401. [Google Scholar]
- Ribas-Fito N, Cardo E, Sala M, Eulalia de Muga M, Mazon C, Verdu A, et al. Breastfeeding, exposure to organochlorine compounds, and neurodevelopment in infants. Pediatrics. 2003;111:e580–5. doi: 10.1542/peds.111.5.e580. [DOI] [PubMed] [Google Scholar]
- Ribas-Fito N, Torrent M, Carrizo D, Munoz-Ortiz L, Julvez J, Grimalt JO, et al. In utero exposure to background concentrations of DDT and cognitive functioning among preschoolers. Am J Epidemiol. 2006;164:955–62. doi: 10.1093/aje/kwj299. [DOI] [PubMed] [Google Scholar]
- Rogan W, Gladen B. PCBs, DDE, and child development at 18 and 24 months. Ann Epidemiol. 1991;1:407–13. doi: 10.1016/1047-2797(91)90010-a. [DOI] [PubMed] [Google Scholar]
- Rogan WJ, Gladen BC, McKinney JD, Carreras N, Hardy P, Thullen J, et al. Polychlorinated biphenyls (PCBs) and dichlorodiphenyl dichloroethene (DDE) in human milk: effects on growth, morbidity, and duration of lactation. American journal of public health. 1987;77:1294–7. doi: 10.2105/ajph.77.10.1294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenberg T. New York Times Magazine. New York City: 2004. What the World Needs Now is DDT. [Google Scholar]
- Roy JR, Chakraborty S, Chakraborty TR. Estrogen-like endocrine disrupting chemicals affecting puberty in humans--a review. Medical science monitor: international medical journal of experimental and clinical research. 2009;15:RA137–45. [PubMed] [Google Scholar]
- Rubin DB. Multiple Imputations for Nonresponse in Surveys. New York: John Wiley & Sons; 1987. [Google Scholar]
- Sagiv SK, Thurston SW, Bellinger DC, Tolbert PE, Altshul LM, Korrick SA. Prenatal Organochlorine Exposure and Behaviors Associated With Attention Deficit Hyperactivity Disorder in School-Aged Children. American Journal of Epidemiology. 2010;171:593–601. doi: 10.1093/aje/kwp427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schnaas L, Rothenberg SJ, Flores M-F, Martinez S, Hernandez C, Osorio E, et al. Reduced Intellectual Development in Children with Prenatal Lead Exposure. Environmental Health Perspectives. 2006;114:791–7. doi: 10.1289/ehp.8552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shanahan M, Pennington B, Yerys B, Scott A, Boada R, Willcutt E, et al. Processing Speed Deficits in Attention Deficit/Hyperactivity Disorder and Reading Disability. J Abnorm Child Psychol. 2006;34:584–601. doi: 10.1007/s10802-006-9037-8. [DOI] [PubMed] [Google Scholar]
- Sioen I, Den Hond E, Nelen V, Van de Mieroop E, Croes K, Van Larebeke N, et al. Prenatal exposure to environmental contaminants and behavioural problems at age 7–8 years. Environ Int. 2013;59:225–31. doi: 10.1016/j.envint.2013.06.014. [DOI] [PubMed] [Google Scholar]
- Sjödin A, Jones RS, Lapeza CR, Focant J-F, McGahee EE, Patterson DG. Semiautomated high-throughput extraction and cleanup method for the measurement of polybrominated diphenyl ethers, polybrominated biphenyls, and polychlorinated biphenyls in human serum. Analytical chemistry. 2004;76:1921–7. doi: 10.1021/ac030381+. [DOI] [PubMed] [Google Scholar]
- Stewart P, Reihman J, Lonky E, Darvill T, Pagano J. Prenatal PCB exposure and neonatal behavioral assessment scale (NBAS) performance. Neurotoxicology and Teratology. 2000;22:21–9. doi: 10.1016/s0892-0362(99)00056-2. [DOI] [PubMed] [Google Scholar]
- Torres-Sanchez L, Rothenberg SJ, Schnaas L, Cebrian ME, Osorio E, Del Carmen Hernandez M, et al. In utero p,p′-DDE exposure and infant neurodevelopment: a perinatal cohort in Mexico. Environ Health Perspect. 2007;115:435–9. doi: 10.1289/ehp.9566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres-Sánchez L, Schnaas L, Cebrián ME, Hernández MdC, Valencia EO, García Hernández RM, et al. Prenatal dichlorodiphenyldichloroethylene (DDE) exposure and neurodevelopment: A follow-up from 12 to 30 months of age. Neuro Toxicology. 2009;30:1162–5. doi: 10.1016/j.neuro.2009.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres-Sánchez L, Schnaas L, Rothenberg SJ, Cebrián ME, Osorio-Valencia E, del Carmen Hernández M, et al. Prenatal p, p-DDE Exposure and Neurodevelopment among Children 3.5–5 Years of Age. Environmental Health Perspectives. 2013;121:263. doi: 10.1289/ehp.1205034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- U.S. DHHS. Toxicological profile for DDT, DDE, and DDD. Atlanta, GA: U.S. Dept. of Health and Human Services Public Health Service, Agency for Toxic Substances and Disease Registry; 2002. [PubMed] [Google Scholar]
- van der Laan MJ, Polley EC, Hubbard AE. Super learner. Statistical Applications in Genetics and Molecular Biology. 2007;6:1–21. doi: 10.2202/1544-6115.1309. [DOI] [PubMed] [Google Scholar]
- Vasiliu O, Muttineni J, Karmaus W. In utero exposure to organochlorines and age at menarche. Human Reproduction. 2004;19:1506–12. doi: 10.1093/humrep/deh292. [DOI] [PubMed] [Google Scholar]
- Verner MA, Gaspar FW, Chevrier J, Gunier RB, Sjodin A, Bradman A, et al. Increasing sample size in prospective birth cohorts: back-extrapolating prenatal levels of persistent organic pollutants in newly enrolled children. Environ Sci Technol. 2015;49:3940–8. doi: 10.1021/acs.est.5b00322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wechsler D. Wechsler Intelligence Scale for Children - Fourth Edition (WISC-IV) Administration and Scoring Manual. San Antonio, TX: Harcourt Assessment Incorporated; 2003. [Google Scholar]
- World Health Organization. Indoor residual spraying: Use of indoor residual spraying for scaling up global malaria control and elimination: WHO Position Statement. 2006. [Google Scholar]
- World Health Organization. World Malaria Report. Geneva, Switzerland: 2014. [Google Scholar]
- Yeates KO, MacPhee D, Campbell FA, Ramey CT. Maternal IQ and home environment as determinants of early childhood intellectual competence: A developmental analysis. Developmental Psychology. 1983;19:731–9. [Google Scholar]
- Zhang JM, Konkle AT, Zup SL, McCarthy MM. Impact of sex and hormones on new cells in the developing rat hippocampus: a novel source of sex dimorphism? The European journal of neuroscience. 2008;27:791–800. doi: 10.1111/j.1460-9568.2008.06073.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.