Abstract
Coronary anatomy has traditionally focused on ventricular circulation. This is largely due to the extent to which coronary artery disease contributes to ischemic heart disease through ventricular myocardial damage. Atrial fibrillation and other tachyarrhythmias that involve the atria, however, remain a major cause of morbidity and mortality. In order to increase mechanistic research and therapeutic interventional procedures for diseases of the atria, an optimal knowledge of atrial anatomy is necessary. While substantial clarity exists regarding the distribution of nerve terminals and the organization of muscle bundles, the anatomy of coronary atrial circulation remains understudied. Historically, the high anatomical variability of atrial coronary branches led to unstandardized nomenclature in the literature. In this review, we delineate the anatomic courses of key atrial coronary branches and their perfusion territories, clarify their nomenclature, and propose unifying anatomical concepts of atrial circulation that we believe to be critical to the success of modern electrophysiologic and surgical procedures.
Introduction
Therapeutic approaches to tachyarrhythmias have improved significantly during the past decade. In atrial fibrillation (AF), for example, treatment strategies have evolved into sophisticated interventional procedures that involve local application of radio-frequency (RF) or other energies.[1,2] These techniques, which hold an excellent safety record,[3] profoundly modify atrial substrate through scar formation with marked success in preventing AF recurrence.[4]
Atrial coronary circulation plays an important role in the safety and efficacy of ablation procedures, as well as in the pathophysiology of atrial fibrillation itself. Atherosclerotic disease in atrial coronary branches serves as a mechanism for AF.[5-7] Consequent application of RF in close proximity to coronary branches may result in coronary vessel injury. This complication is rare, however, most likely due to the ‛heat sink′ effect. Blood flow within a coronary branch located near an RF electrode provides a protective effect through convective cooling and thereby prevents RF energy from substantially heating the vascular endothelium.[8] However, while this ‛heat sink′ is protective to coronary arteries, it may also limit the success of ablation procedures by the phenomenon of the ‛shadow effect′.[9,10] Flow through small intramyocardial vessels is known to disrupt transmural lesion formation, preserve conduction through an RF lesion, and thereby prevent complete conduction block.[11] Atrial coronary perfusion, therefore, plays a significant role in AF pathophysiology, determines the extent of injury to nearby vessels through the ‛heat sink′ effect, and influences the success of lesion formation through the ‛shadow effect′.
As the eminent Mount Sinai Hospital physician, M.A. Kugel, alluded to: “The blood supply to a tissue is often a key to the understanding of pathological processes which may occur in the tissue.”[12]Currently, scarce literature and unstandardized nomenclature present challenges to understanding detailed atrial coronary anatomy and perfusion territories. In general, the sinus nodal artery (SNA) and the atrioventricular nodal artery (AVNA) have been described as major atrial coronary branches. Although they play a significant role in the pathophysiology of atrial fibrillation and other tachyarrhythmias, no unanimous characterization of atrial coronary arteries exists in the cardiology literature.[13] In this light, we present an extensive literature review of atrial circulation for the purpose of clarifying the anatomy and nomenclature of major atrial coronary branches. In addition, we propose unifying principles of atrial coronary arterial circulation relevant to interventional electrophysiologists.
Atrial coronary anatomy, nomenclature
In 1907, London Hospital Medical College anatomist, Arthur Keith, and physiologist, Martin Flack, well-known for their seminal description of the sinus node, were the first to describe atrial coronary branches.[14,15] SNA nomenclature fluctuated over the years. In 1921, the anatomist, Dr. Gross named it the ramus ostii cavae superioris.[16] It was also known as the Keith-Flack artery, named after the British physicians who first described the vessel.[17] Here we use the Baroldi and Scomazzoni nomenclature, which refers to the SNA as the “main atrial branch” emphasizing that it is the largest branch supplying the sinus node irrespective of its side of origin.[18]
In 55% of the population, the SNA arises from the right coronary artery (RCA), usually from the anterior branch. In the remaining 45% of cases, the SNA arises from the left coronary artery (LCA), also usually from the anterior branch. [19] In this review, for best clarity, we have mostly retained Spalteholz classification which is consistent with the majority of studies on atrial coronary anatomy, where branches of the RCA and left circumflex artery (LCX) are named by their site of origin as anterior, intermediate/marginal and posterior branches.[13]
Right atrial branches
Arteries supplying the right atrium (RA) are among the earliest branches of the RCA after the conus artery and originate along the right atrioventricular (AV) groove ([Figure 1]). They are usually classified into the right anterior, intermediate/marginal and posterior atrial branches. The right anterior atrial branch is the first branch arising on the anterior aspect of the RA and ascends posteriorly along the body of the RA to reach the anterior aspect of the interatrial groove. Angiographically, in a right anterior oblique (RAO) projection, the right anterior atrial branch is slender, arises at a variable distance from the conus artery, and courses to the left and superior aspect of the atrium.[20] It gives off branches to both atria and the interatrial septum and terminates by encircling the lower portion of the superior vena cava (SVC). When it is the main atrial branch, it supplies the sinoatrial node (SAN).
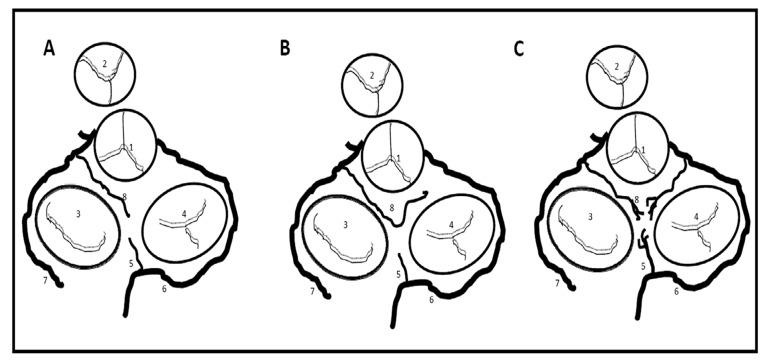
Figure 1. Atrial arterial branches.
Dark brown dotted line represents the outline of the atrial walls. 1. Right coronary artery, 2. Right anterior atrial branch (main atrial branch in this fig), 3. Right intermediate branch, 4. Right posterior branch, 5. Kugel arterial anastomosis, 6. Left circumflex artery, 7. Left anterior atrial branch, 8. Left intermediate atrial branch, 9. Left posterior branch, 10. Left atrial circumflex, 11. Atrioventricular nodal artery.
The right intermediate atrial branch, also called the marginal branch, arises from the acute right margin of the heart, ascends over the anterolateral surface of the RA, and supplies surrounding atrial tissues ([Figure 1]). In only 13.3% of cases it is the main atrial branch that supplies the sinus node.[21,22] The origin of this branch is usually marked by the presence of a small fatty excrescence or by a small cardiac vein that drains into the RA wall.[23] Angiographically, the right intermediate atrial branch is uneasy to visualize due to its small size and variability. For the same reason, the right posterior atrial branch is difficult to locate. When present, the right posterior branch arises on the posterior aspect of the RA and supplies the right posterior atrial wall and left atrial (LA) posterior surface.
Notably, Busquet et al. proposed another classification scheme in which right atrial branches are classified based on cross sectional diameter into two rather than three groups, the major and accessory branches.[23] Major branches are usually found in groups occupying anterior, lateral and posterior positions relative to the tricuspid orifice. Accessory branches arise from the RCA in the AV groove and have an average diameter of less than 0.5 mm and a length not exceeding 10 mm. They are covered by fatty tissue in the AV groove and supply the lower atrial wall and the adjacent right ventricle.
Left atrial branches
Arteries supplying the LA are among the earliest branches of the LCA, usually from the LCX, and originate along the AV groove. Similar to right atrial circulation, left atrial branches are usually classified into the left anterior, intermediate/marginal, and posterior atrial branches. The left anterior atrial branch arises on the anterior aspect of the LA ([Figure 1]). When it is the main atrial branch, it courses upwards along the LA via the anterior interatrial groove to reach the SAN. In RAO projection, it is an early branch which ascends leftwards and upwards.[20] The remaining left atrial branches are variable and feed adjacent atrial tissues along their course over the LA. Another variant is the left atrial circumflex artery (LACX), which may branch early from either the LCX or its main atrial branch ([Figure 1]).[20,21] Early in its course, the LACX ascends through the LA and travels along its lower margin parallel to the left AV groove. It extends around the left heart margin and terminates in the posterior wall of the LA. Sometimes, the LACX extends farther, crossing the crux of the heart along the right AV groove to give off right posterior atrial branches. Rarely, the LACX may supply the SNA itself as the main atrial branch, which in a study of 118 patients, occurred 11% of the time. [21]
Right and left atrial anastomoses and Kugel’s artery
Several anastomoses between atrial coronary arterial systems have been described. Generally, these anastomoses exist as either small intra-atrial or atrioventricular branches, or as a single vessel. Kugel’s artery is the major transatrial pathway that bridges the right and left coronary systems ([Figure 2]). The SNA can also serve as a major anastomotic channel. It is the largest atrial artery and can connect right and left coronary systems throughout its course; it forms a vascular loop around the base of the SVC where it sends tributaries to small branches of the left and right intermediate arteries.[19,24] In other variants, the posterior termination of the LACX can also anastomose with the distal RCA or AVNA.[20]
Figure 2. Schematic representation of the heart shows the three anatomic variations of the arteria anastomotica auricularis magna, superior view.
A. Artery arises from LCX or its branches meet with the vessels at crux, B. Vessel arises from LCX or its branches and takes an abrupt turn to meet the vessels at the RA anterior wall; C. Vessel arises from LCX and fans out to meet with vessels from RCA. 1. Aorta, 2. Pulmonary trunk, 3. Mitral valve, 4. Tricuspid leaflet, 5. AV nodal artery, 6. right coronary artery, 7. Left circumflex artery, 8. Anastomotica auricularis magna
In 1927, Dr. Kugel described a free anastomosis between the right and left coronary arteries formed by a conspicuous vessel that runs in the interatrial septum.[12] He coined the name, arteria anastomotica auricularis magna, acknowledging its relative large size and role in anastomosis. As the major anastomotic channel between left and right atrial coronary systems, Kugel’s artery supplies both the SA and AV nodes. It can therefore function as a backup for collateral flow in the setting of procedural damage to vessels supplying the SA or AV nodes.
Anatomically, Kugel described that the posterior portion of the interatrial septum divides anteriorly forming a “Y” shape where the stem represents the interatrial septum posteriorly and wings to form the anterior walls of both atria. Kugel’s artery lies along the anterior atrial walls and travels into the interatrial septum. [12] In a study of 50 normal hearts, three major variations of Kugel’s artery were proposed ([Figure 2]). The most common, found in 66% of cases, is an artery that originates from either the LCX or its early branches. It passes posteriorly into the LA wall and terminates near the crux of the heart where it connects with either the posterior branches of the distal RCA, posterior descending artery (PDA), or AVNA. Kugel’s artery can therefore serve as an AV anastomotic pathway in addition to a transatrial pathway. In 26% of cases, a branch originates from the LCX, penetrates the interatrial septum, abruptly turns on itself, retraces its entire course though the interatrial septum and terminates in the RA anterior wall. In 8% of cases, the artery diverges from the LCX early, divides into smaller branches and fans out over the LA anterior wall to connect with fine vessels from the proximal RCA in the anterior RA wall.Kugel’s artery also supplies the aortic cusp of the mitral valve (MV), aortic valve (AOV), commissures, and aortic base, implying its role in the pathogenesis of lesions within these structures.[12] Interestingly, in several hearts with atherosclerotic disease, the vessel was notable for its unusually large caliber, evidencing its contribution to collateral circulation in ischemic territories.
Sinus nodal artery: origin, course, termination
PWhile atrial branches vary substantially in their distributions, the SNA is the most preserved artery within the atrial circulation.[19] It arises either from the RCA or LCX. In 55% of human hearts, the RCA supplies the SNA within 2 cm of the coronary ostium and corresponds to the right anterior atrial branch.[16,25] In 45% of cases, it originates within the first few mm of the LCA and usually corresponds to the left anterior atrial branch.19The SNA arises from the proximal LCX in 95% of these left-sided cases, from the left main coronary trunk in 3% of cases, and from the distal portion of the circumflex branch in the remaining 2% of cases.[26] Importantly, several authors suggest that the sinus node region could also be fed by two arteries rather one.[21,25-27]
When the SNA corresponds to the right anterior atrial branch, it arises 0.2–2.2 cm from the right coronary ostium with a cross-sectional diameter of 1–3 mm.[28] When it corresponds to the right intermediate atrial branch, the SNA arises about 3.0-7.5 cm from the right coronary ostium with a diameter of 0.8-2.1 mm.[23] The right SNA travels along the body of the adjacent atrium from the left side to the anterior interatrial myocardial band (Bachmann’s Reviewbundle) to reach the base of the SVC.[28] At the SVC base, the artery circles clockwise or counterclockwise before penetrating the sinus node at the auriculocaval junction. Busquet et al. highlighted the variability in the termination of the SNA relative to the auriculocaval junction as precaval (58%), retrocaval (36%), and pericaval (6%) ([Figure 3]).[23] This variation is also supported by Saremi et al.[29]
Figure 3. Three dimensional reconstruction shows the sinus nodal arteries (superior view).

A. Artery arises from LCX or its branches meet with the vessels at crux, B. Vessel arises from LCX or its branches and takes an abrupt turn to meet the vessels at the RA anterior wall; C. Vessel arises from LCX and fans out to meet with vessels from RCA. 1. Aorta, 2. Pulmonary trunk, 3. Mitral valve, 4. Tricuspid leaflet, 5. AV nodal artery, 6. right coronary artery, 7. Left circumflex artery, 8. Anastomotica auricularis magna.
Relevant to ablationists, the right phrenic nerve is situated anterolateral and in close proximity to the SVC base, around which the SNA circles. This anatomic relationship is not influenced by whether the SNA courses clockwise or counterclockwise at the SVC base. However, during procedures that target areas neighboring the sinus node region, both the phrenic nerve and the SNA may be susceptible to injury. The mean distance from the right phrenic nerve and the SVC-right atrial junction is 5.8-7.8 mm and its mean distance to the anterolateral wall of the SVC ranges 0.3-1.3 mm.30 Therefore, ablation procedures which have the potential to endanger the phrenic nerve in the area of the superior venoatrial junction or the anterolateral wall of the SVC may also impact the SNA.
The sinus node capillary network
An appreciation of the microvascular blood supply within the SAN is also essential to understanding the perfusion of this vital structure. In 70% of postmortem angiographic and histological studies of SAN’s, Verhaeghe et al. showed a fairly uniform arterial distribution pattern.[31] The SNA enters from one pole of the spindle shaped SAN, traverses the central part, sends off several parallel branches, and exits at the opposite pole. The arterial density of the SAN is approximately eight times greater than that of the adjacent free atrial wall, which highlights its teleological significance.[31]
In addition to AF, atrial arterial anatomy is clinically relevant, for example, to SAN dysfunction and sick sinus syndrome (SSS). We now know that the pathobiology of SAN dysfunction involves atherosclerotic disease of the SNA.[32] Initial studies of depressed sinus node function, however, did not find a relationship between SNA atherosclerotic lesions and markers of SAN dysfunction such as prolonged sinus node recovery time (SNRT), heart rate, or advanced age.[32]It was not until
Jordan et al. that sinus-to-atrium conduction time (SACT) was proposed as the electrophysiologic parameter that correlated with SNA atherosclerotic disease.[33] SACT was found to be significantly longer in patients with severe SNA stenosis,[34] which established it as a more sensitive early indicator of SAN dysfunction than SNRT or basal heart rate.[33] The role of SNA atherosclerotic disease in SAN dysfunction and SSS was therefore established and highlighted the need for a greater understanding of atrial coronary circulation.
Procedural importance of the SNA
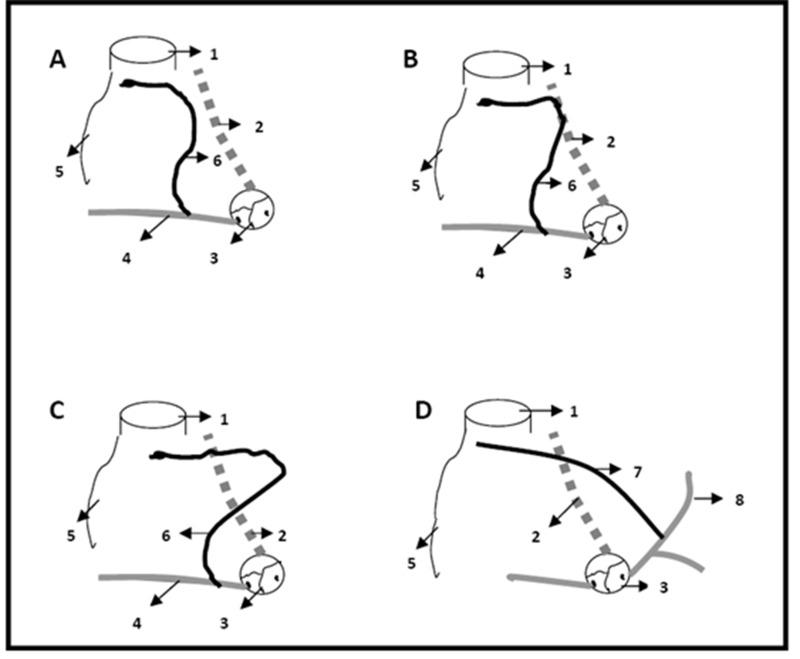
The course of the right and left SNA has important procedural significance. In a study of 50 patients by Berdajs et al. found that the SNA crosses the interatrial septum in all left-sided SNA patients. In right-sided SNA patients, the SNA crosses the interatrial septum when the vessel originates from the anterior atrial branch Figure 4. Consequently, 54% of all SNA arteries in this study crossed the interatrial septum, which is used in the superior
Figure 4. Courses of the right sinus nodal artery.
A) sinus nodal artery does not reach the anterior inter atrial groove, B) sinus nodal artery reaches the anterior inter atrial groove but not beyond, C) sinus nodal artery crosses the anterior inter atrial groove, D) left sinus nodal artery crosses the anterior inter atrial groove. 1. Superior vena cava, 2. Anterior interatrial groove, 3. Aortic sinus, 4. Right coronary artery, 5. Appendage of the right atrium, 6. Right sinus nodal artery, 7. Left sinus nodal artery, 8 Left circumflex artery. (Concept adapted from Sow M L et al. The artery of the sinuatrial node: anatomic considerations based on 45 injection-dissections of the heart. Surg Radiol Anat 1996;18:103-9 with permission of Springer Science+Business Media)
Reviewtransseptal approach to the MV.[35] Therefore, inadvertent damage to the SNA may occur during procedures involving the interatrial septum as well as other regions throughout the SNA course.[21,27,36] Injury to the terminal SNA may occur during RA appendage cannulation or if a ligature is placed too low at the base of the SVC. Right atriotomy may also predispose the SNA to injury, particularly at the anterior and lateral RA wall opposite the proximal RCA. The Fontan procedure often involves the atrial roof, which may also be a site for SNA exposure. Finally, the transseptal approach of Dubost’s procedure, Mustard’s procedure, and Dacron plaque suture, may all threaten the SNA as it courses through the interatrial septum.
S-Shaped Sinus Nodal Artery
In left-sided SNA patients, Nerantzis and Avgoustakis reported the existence of an artery that originates from the LCX below the LA appendage and supplies the SNA ([Figure 5]). This was named the S-shaped sinus nodal artery (SSNA).[37] The SSNA was present in 21.5% of 111 patients studied and appeared either as a branch of the LCX (13.5%), as an upper part of a divided LCX (5.4%), or as a main continuation of the LCX (2.7%). It is larger than the normal SNA and follows the same course to the SAN. This is consistent with findings by Kyriakidis et al. and Saremi et al.[29,38] In addition to feeding the SAN region, the SSNA feeds the LA, a large part of interatrial septum, RA, and part of the AV nodal area. The likelihood of SSNA occlusion is low as atherosclerotic disease often occurs within the first few centimeters of the LCX.[37]
Figure 5. S-shaped sinus nodal artery.

Three-dimensional CT shows S-shaped sinus nodal artery. LAA – left atrial appendage, LSPV – left superior pulmonary vein (arrow), LCX – left circumflex artery, AAo – ascending aorta, LAD – left anterior descending artery, RAA – right atrial appendage, SVC – superior vena cava, LA – left atrium, LAD – left anterior descending artery, Orientation: A - anterior, L- left, P - posterior, R - right. (Adapted from Saremi B et al. Arterial supply to sinuatrial and atrioventricular nodes: Radiology 2008;246:99-107 with permission from RSNA)
Territories of atrial perfusion
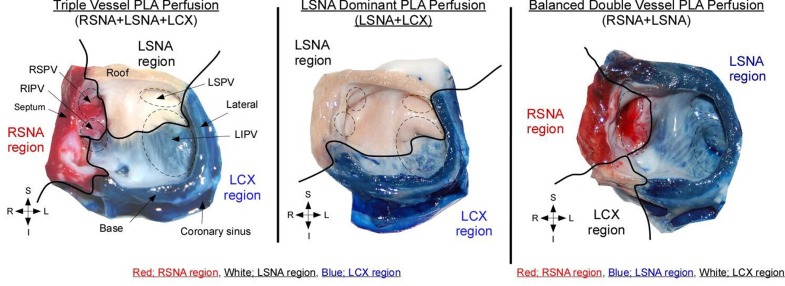
While we have focused on the origin, course and termination of the SNA, we also aim to describe the territories it supplies. A precise anatomic delineation of atrial coronary perfusion territories does not exist in the literature. Perfusion of the posterior LA wall (PLA) and pulmonary veins (PVs), is especially relevant as these are crucial regions for AF maintenance. Recently, our group conducted an investigation in isolated ovine hearts to address this question. Concordant with human descriptions,[39]we reported that in sheep hearts, three atrial coronary branches, left SNA (LSNA), right SNA (RSNA), and atrial branches of the LCX perfuse the PLA including PV regions. We also delineated atrial coronary perfusion territories by selective perfusion of Congo red into the RSNA and Evan’s blue into the LCX ([figure 6]) left and middle panels) or the LSNA (right panel). Both atria, including the PLA and PVs were then dissected for acquisition of photographic snapshots. As shown in ([figure 6]), each atrial branch (RSNA, LSNA, and LCX branches) supplied a well-defined territory it the PLA.
Figure 6. Atrial coronary perfusion territories in sheep.
(left) triple vessel PLA perfusion, (middle) LSNA dominant PLA perfusion, (right) Balanced double vessel PLA perfusion. Black solid line delineates regional perfusion territories. RSNA; right sinus node artery, LSNA; left sinus node artery, RSPV; right superior pulmonary vein, RIPV; right inferior pulmonary vein, LSPV; left superior pulmonary vein, LIPV; left inferior pulmonary vein. (Adapted from Yamazaki M et al. Left atrial coronary perfusion territories in isolated sheep hearts Heart Rhythm 2010;7:1501-1508 with permission from Elsevier)
Interestingly, the contribution of individual arteries to the perfusion of the PLA-PV myocardium varied between specimens. Three anatomical variants were observed with equal frequency: triple vessel PLA perfusion (Figure 6 left, 29.5%), double vessel PLA perfusion (middle, 23.5%) and single vessel PLA perfusion (right, 29.5%). These data suggest that the complexity of atrial vessel origin and course may translate into intricate perfusion and collateral flow to territories well-known to harbor electrical sources of AF initiation and maintenance. Implications in patients with adFigure vanced coronary artery disease remain to be elucidated, as the severity of atherosclerotic burden associated with atrial infarctions may relate to the type of atrial coronary perfusion to the PLA-PV.
Factors of variability: dominance, race, and gender
The origin of the SNA has not been associated with coronary dominance.In regard to race, some authors proposed a higher incidence of right-sided SNA origin in African Americans and Caucasians,[25] but others have shown no association between race and SNA origin.[40] Gender has not been correlated with SNA origin either,[29,40] though some investigators describe a higher incidence of right-sided SNA origin in men.[25,27] Given that preliminary studies show conflicting associations between SNA origin and coronary dominance, race, and gender, larger and more heterogeneous patient populations are needed to definitively answer this question.
AV Nodal Artery: origin, course, termination
The AVNA is well known under this name, but has also been referred to in the literature as the ramus septi fibrosi, which acknowledges that its source also supplies the membranous part of the interventricular septum. This concept was supported by Spirina and Soskin, who named the AVNA, the arteria septi fibrosi.41Its mean length is 0.5-3 cm and mean external diameter at the beginning of its course is 1-3.5 mm.[28] Usually, the artery that crosses the crux of the heart is the artery that feeds the AV node. In 90% of the population, this artery is the RCA and in the remaining 10%, it is the LCX.[19] The AVNA is associated with coronary dominance, where the vessel that gives rise to the PDA also gives rise to the AVNA.
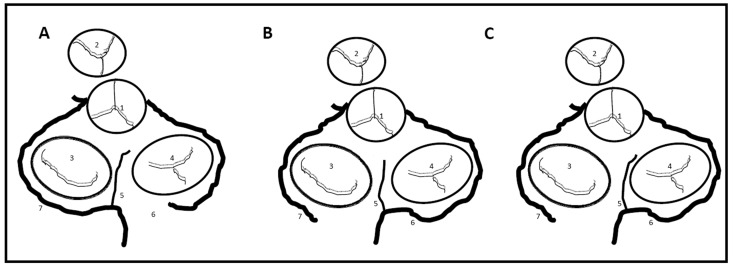
The main vessel that reaches the crux of the heart, be it the RCA or LCX, makes a sharp U-turn under the middle cardiac vein before giving rise to the PDA. This is often used as a landmark during angiogram interpretation.[42] The ‛U′ or ‛V′ shape of this arterial segment has an embryologic origin. In the embryo, the AV node is located in the epicardium, which is retracted into the heart when the myocardium invaginates during the formation of the interatrial septum.[43] The AVNA arises at the apex of this abrupt turn[19,21] and merges into the cardiac wall towards the right fibrous trigone passing closer to the MV ring before entering the AV node.[28] Once it enters the AV node, it usually bends at a right angle beneath the aortic valve and ramifies into the interventricular septum to supply the posterior portion of the left branch of the bundle of His. Three final courses are proposed for the AVNA ([Figure 7]).[44] It may follow close to the tricuspid valve (TV), between the TV and MV, or closer to the MV. The right superior descending artery and Kugel’s artery supply collateral blood to the AV node.[45]
Figure 7. Atrial coronary perfusion territories in sheep.
(left) triple vessel PLA perfusion, (middle) LSNA dominant PLA perfusion, (right) Balanced double vessel PLA perfusion. Black solid line delineates regional perfusion territories. RSNA; right sinus node artery, LSNA; left sinus node artery, RSPV; right superior pulmonary vein, RIPV; right inferior pulmonary vein, LSPV; left superior pulmonary vein, LIPV; left inferior pulmonary vein. (Adapted from Yamazaki M et al. Left atrial coronary perfusion territories in isolated sheep hearts Heart Rhythm 2010;7:1501-1508 with permission from Elsevier)
Any ablation procedure involving the AV node, mainly those for drug-resistant atrial tachyarrhythmias and AV nodal reentrant tachycardias, carry a risk of damaging the AVNA.[46] Specific sites of unintended injury to the AVNA include the crux of the heart, the sinus venosus orifice, or the base of the septal cusp of the TV.[36] Complications can be avoided by setting angiographic landmarks prior to ablation. The distal end of the AVNA serves as a useful landmark to depict the location of the AV node. It also serves as an anatomical landmark within Koch’s triangle to predict the risk of AV block during RFA of AV nodal re-entrant tachycardia. When the distance between the distal end of the AVNA and the ablation target site exceeds 2 mm, AV block rarely occurs.[47]
Atrial venous system
As with the arterial system, atrial venous drainage is understudied and variable but here we describe its anatomy and elucidate overarching concepts. The majority of the venous drainage from the atria, similar to that of the ventricles, occurs through the epicardial venous system, which joins progressively larger vessels that ultimately flow into the RA via the coronary sinus.[48] The remainder of the venous drainage occurs through the Thebesian venous system, where veins drain directly into the cardiac chambers. The left atrial veins are divided into three distinct groups: the posterolateral veins, the posterosuperior veins, and the septal veins.[49,50]The right atrial veins are divided into small, large, short, or long intramyocardial tunnels and small intramural veins.
A prominent vessel of the atrial epicardial venous system is the oblique vein of the left atrium, which is commonly described as the Vein of Marshall. It has an average diameter of 1 mm and rarely exceeds 2-3 cm in length.[51] It descends along the lateral and inferior wall of the LA, passing between the left pulmonary veins and the left atrial appendage. It then joins the great cardiac vein at which point the coronary sinus begins. Clinically, this region is implicated as a substrate for left atrial arrhythmias. Specifically, the ligament of Marshall, which consists of fibrous and muscular bands, nerves and small blood vessels, including the Vein of Marshall, is thought to function as a left atrial arrhythmogenic focus.[52-54] Finally, it should be noted that the vein of Marshall has been suggested to be a conduit by which retrograde ethanol infusion might achieve regional atrial ablation.[55,56]
Future research directions
Our understanding of atrial coronary anatomy is growing, but several research directions are needed to investigate its application to disease pathophysiology. For example, atrial circulation, unlike ventricular circulation, courses upward against gravity and coronary flow occurs only during diastole. Whether this mode of circulation has implications for atrial arrhythmias needs further study. In addition, radiofrequency induced coronary arterial injury though rare, is a well-documented phenomenon with serious complications.[10] New ablation strategies that take atrial coronary anatomy of the posterior atrial wall and SAN into consideration need further elaboration to mitigate this problem. Finally, our understanding of coronary anatomy is limited by our technology, which calls for continued development of smaller catheter tips capable of accessing narrower arteries.
References
- 1.Kühne Michael, Schaer Beat, Ammann Peter, Suter Yves, Osswald Stefan, Sticherling Christian. Cryoballoon ablation for pulmonary vein isolation in patients with paroxysmal atrial fibrillation. Swiss Med Wkly. 2010 Apr 17;140 (15-16):214–21. doi: 10.4414/smw.2010.12977. [DOI] [PubMed] [Google Scholar]
- 2.Lábrová R, Spinar J, Honzíková N. Radiofrequency ablation in treatment of atrial fibrillation. Physiol Res. 2010;59 Suppl 1 ():S43–9. doi: 10.33549/physiolres.932007. [DOI] [PubMed] [Google Scholar]
- 3.Cappato Riccardo, Calkins Hugh, Chen Shih-Ann, Davies Wyn, Iesaka Yoshito, Kalman Jonathan, Kim You-Ho, Klein George, Natale Andrea, Packer Douglas, Skanes Allan, Ambrogi Federico, Biganzoli Elia. Updated worldwide survey on the methods, efficacy, and safety of catheter ablation for human atrial fibrillation. Circ Arrhythm Electrophysiol. 2010 Feb;3 (1):32–8. doi: 10.1161/CIRCEP.109.859116. [DOI] [PubMed] [Google Scholar]
- 4.Letsas Konstantinos P, Efremidis Michael, Charalampous Charalampos, Tsikrikas Spyros, Sideris Antonios. Current ablation strategies for persistent and long-standing persistent atrial fibrillation. Cardiol Res Pract. 2011 Mar 03;2011 () doi: 10.4061/2011/376969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sinno Hani, Derakhchan Katayoun, Libersan Danielle, Merhi Yahye, Leung Tack Ki, Nattel Stanley. Atrial ischemia promotes atrial fibrillation in dogs. Circulation. 2003 Apr 15;107 (14):1930–6. doi: 10.1161/01.CIR.0000058743.15215.03. [DOI] [PubMed] [Google Scholar]
- 6.Nishida Kunihiro, Qi Xiao Yan, Wakili Reza, Comtois Philippe, Chartier Denis, Harada Masahide, Iwasaki Yu-ki, Romeo Philippe, Maguy Ange, Dobrev Dobromir, Michael Georghia, Talajic Mario, Nattel Stanley. Mechanisms of atrial tachyarrhythmias associated with coronary artery occlusion in a chronic canine model. Circulation. 2011 Jan 18;123 (2):137–46. doi: 10.1161/CIRCULATIONAHA.110.972778. [DOI] [PubMed] [Google Scholar]
- 7.Kannel W B, Abbott R D, Savage D D, McNamara P M. Coronary heart disease and atrial fibrillation: the Framingham Study. Am. Heart J. 1983 Aug;106 (2):389–96. doi: 10.1016/0002-8703(83)90208-9. [DOI] [PubMed] [Google Scholar]
- 8.Chatelain P, Zimmermann M, Weber R, Campanini C, Adamec R. Acute coronary occlusion secondary to radiofrequency catheter ablation of a left lateral accessory pathway. Eur. Heart J. 1995 Jun;16 (6):859–61. doi: 10.1093/oxfordjournals.eurheartj.a061007. [DOI] [PubMed] [Google Scholar]
- 9.Viles-Gonzalez Juan F, de Castro Miranda Reynaldo, Scanavacca Mauricio, Sosa Eduardo, d'Avila Andre. Acute and chronic effects of epicardial radiofrequency applications delivered on epicardial coronary arteries. Circ Arrhythm Electrophysiol. 2011 Aug;4 (4):526–31. doi: 10.1161/CIRCEP.110.961508. [DOI] [PubMed] [Google Scholar]
- 10.Castaño Adam, Crawford Thomas, Yamazaki Masatoshi, Avula Uma Mahesh R, Kalifa Jérôme. Coronary artery pathophysiology after radiofrequency catheter ablation: review and perspectives. Heart Rhythm. 2011 Dec;8 (12):1975–80. doi: 10.1016/j.hrthm.2011.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fuller Ithiel A, Wood Mark A. Intramural coronary vasculature prevents transmural radiofrequency lesion formation: implications for linear ablation. Circulation. 2003 Apr 08;107 (13):1797–803. doi: 10.1161/01.CIR.0000058705.97823.F4. [DOI] [PubMed] [Google Scholar]
- 12.M KUGEL . . Anatomical studies on the coronary arteries and their branches. I. Arteries anastomotica auricularis magna (abstract). Am Heart J . 1927;0:0–0. [Google Scholar]
- 13.Spalteholz W , Hirzel S . Leipzig () Boca Raton, FL: CRC Press. 1924;0:0–0. [Google Scholar]
- 14.Silverman Mark E, Hollman Arthur. Discovery of the sinus node by Keith and Flack: on the centennial of their 1907 publication. Heart. 2007 Oct;93 (10):1184–7. doi: 10.1136/hrt.2006.105049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Keith A, Flack M. The Form and Nature of the Muscular Connections between the Primary Divisions of the Vertebrate Heart. J Anat Physiol. 1907 Apr;41 (Pt 3):172–89. [PMC free article] [PubMed] [Google Scholar]
- 16.L Gross. The blood supply to the heart. In: New york . Paul b. Hoeber. 1921;0:0–0. [Google Scholar]
- 17.A CRAINICIANU . . Anatomisehe studien uber die coronararterien und experimentelle untersuchungen uber ihre durchgangigkeit. . Virchows Arch. 1922;0:0–0. [Google Scholar]
- 18.G. S Baroldi, GB Dijksterhuis. . Coronary circulation in the normal and the pathologic heart. . In: Washington,d.C: . 1967;0:0–0. [Google Scholar]
- 19.JAMES T N, BURCH G E. The atrial coronary arteries in man. Circulation. 1958 Jan;17 (1):90–8. doi: 10.1161/01.cir.17.1.90. [DOI] [PubMed] [Google Scholar]
- 20. R Homby, AA Licata. Clinical - anatomical correlates in coronary artery disease. In: Mount kisco, ed. , . Futura publishing company. 1979;0:0–0. [Google Scholar]
- 21.Vieweg W V, Alpert J S, Hagan A D. Origin of the sinoatrial node and atrioventricular node arteries in right, mixed, and left inferior emphasis systems. Cathet Cardiovasc Diagn. 1975;1 (4):361–73. doi: 10.1002/ccd.1810010405. [DOI] [PubMed] [Google Scholar]
- 22.Krupa U. Studies on the coronary vascularization of atria of the heart. Folia Morphol. (Warsz) 1982;41 (2):207–15. [PubMed] [Google Scholar]
- 23.Busquet J, Fontan F, Anderson R H, Ho S Y, Davies M J. The surgical significance of the atrial branches of the coronary arteries. Int. J. Cardiol. 1984 Aug;6 (2):223–36. doi: 10.1016/0167-5273(84)90357-7. [DOI] [PubMed] [Google Scholar]
- 24.James T N. The delivery and distribution of coronary collateral circulation. Chest. 1970 Sep;58 (3):183–203. doi: 10.1378/chest.58.3.183. [DOI] [PubMed] [Google Scholar]
- 25.Romhilt D W, Hackel D B, Estes E H. Origin of blood supply to sinoauricular and atrioventricular node. Am. Heart J. 1968 Feb;75 (2):279–80. doi: 10.1016/s0002-8703(68)90075-6. [DOI] [PubMed] [Google Scholar]
- 26.Futami C, Tanuma K, Tanuma Y, Saito T. The arterial blood supply of the conducting system in normal human hearts. Surg Radiol Anat. 2003 Apr;25 (1):42–9. doi: 10.1007/s00276-002-0085-7. [DOI] [PubMed] [Google Scholar]
- 27.Sow M L, Ndoye J M, Lô E A. The artery of the sinuatrial node: anatomic considerations based on 45 injection-dissections of the heart. Surg Radiol Anat. 1996;18 (2):103–9. doi: 10.1007/BF01795227. [DOI] [PubMed] [Google Scholar]
- 28.Pejković B, Krajnc I, Anderhuber F, Kosutić D. Anatomical aspects of the arterial blood supply to the sinoatrial and atrioventricular nodes of the human heart. J. Int. Med. Res. 2008 Jul 26;36 (4):691–8. doi: 10.1177/147323000803600410. [DOI] [PubMed] [Google Scholar]
- 29.Saremi Farhood, Abolhoda Amir, Ashikyan Oganes, Milliken Jeffrey C, Narula Jagat, Gurudevan Swaminatha V, Kaushal Khushboo, Raney Aidan. Arterial supply to sinuatrial and atrioventricular nodes: imaging with multidetector CT. Radiology. 2008 Jan;246 (1):99–107. doi: 10.1148/radiol.2461070030. [DOI] [PubMed] [Google Scholar]
- 30.Sánchez-Quintana Damian, Cabrera José Angel, Climent Vicente, Farré Jerónimo, Weiglein Andreas, Ho Siew Yen. How close are the phrenic nerves to cardiac structures? Implications for cardiac interventionalists. J. Cardiovasc. Electrophysiol. 2005 Mar;16 (3):309–13. doi: 10.1046/j.1540-8167.2005.40759.x. [DOI] [PubMed] [Google Scholar]
- 31.Verhaeghe L, Van Der Hauwaert L. Arterial blood supply of the human sinus node. Br Heart J. 1967 Sep;29 (5):801–6. doi: 10.1136/hrt.29.5.801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Engel T R, Meister S G, Feitosa G S, Fischer H A, Frankl W S. Appraisal of sinus node artery disease. Circulation. 1975 Aug;52 (2):286–91. doi: 10.1161/01.cir.52.2.286. [DOI] [PubMed] [Google Scholar]
- 33.Jordan J, Yamaguchi I, Mandel W J. Characteristics of sinoatrial conduction in patients with coronary artery disease. Circulation. 1977 Apr;55 (4):569–74. doi: 10.1161/01.cir.55.4.569. [DOI] [PubMed] [Google Scholar]
- 34.Alboni P, Baggioni G F, Scarfò S, Cappato R, Percoco G F, Paparella N, Antonioli G E. Role of sinus node artery disease in sick sinus syndrome in inferior wall acute myocardial infarction. Am. J. Cardiol. 1991 Jun 01;67 (15):1180–4. doi: 10.1016/0002-9149(91)90923-9. [DOI] [PubMed] [Google Scholar]
- 35.Bokeriya L A, Mikhailin S I, Revishvili A S. Anatomical variants of sinoatrial and atrioventricular node arteries. Cor Vasa. 1984;26 (3):220–8. [PubMed] [Google Scholar]
- 36.Nerantzis C, Avgoustakis D. An S-shaped atrial artery supplying the sinus node area. An anatomical study. Chest. 1980 Aug;78 (2):274–8. doi: 10.1378/chest.78.2.274. [DOI] [PubMed] [Google Scholar]
- 37.Kyriakidis M K, Kourouklis C B, Papaioannou J T, Christakos S G, Spanos G P, Avgoustakis D G. Sinus node coronary arteries studied with angiography. Am. J. Cardiol. 1983 Mar 01;51 (5):749–50. doi: 10.1016/s0002-9149(83)80126-x. [DOI] [PubMed] [Google Scholar]
- 38.Kyriakidis M K, Kourouklis C B, Papaioannou J T, Christakos S G, Spanos G P, Avgoustakis D G. Sinus node coronary arteries studied with angiography. Am. J. Cardiol. 1983 Mar 01;51 (5):749–50. doi: 10.1016/s0002-9149(83)80126-x. [DOI] [PubMed] [Google Scholar]
- 39.Yamazaki Masatoshi, Morgenstern Sherry, Klos Matthew, Campbell Katherine, Buerkel Daniel, Kalifa Jérôme. Left atrial coronary perfusion territories in isolated sheep hearts: implications for atrial fibrillation maintenance. Heart Rhythm. 2010 Oct;7 (10):1501–8. doi: 10.1016/j.hrthm.2010.06.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.DiDio L J, Lopes A C, Caetano A C, Prates J C. Variations of the origin of the artery of the sinoatrial node in normal human hearts. Surg Radiol Anat. 1995;17 (1):19–26. doi: 10.1007/BF01629494. [DOI] [PubMed] [Google Scholar]
- 41.Spirina G A. Topography and blood supply of atrioventricular node of the human heart. Cor Vasa. 1983;25 (1):42–8. [PubMed] [Google Scholar]
- 42.JAMES T N. A useful landmark for interpreting angiocardiograms. Radiology. 1962 Nov;79 ():804–6. doi: 10.1148/79.5.804. [DOI] [PubMed] [Google Scholar]
- 43.JAMES T N, BURCH G E. Blood supply of the human interventricular septum. Circulation. 1958 Mar;17 (3):391–6. doi: 10.1161/01.cir.17.3.391. [DOI] [PubMed] [Google Scholar]
- 44.Berdajs Denis, Künzli Andreas, Shurr Ulrich, Zünd Gregor, Turina Marko I, Genonni Michele. Clinical anatomy of the atrioventricular node artery. J. Heart Valve Dis. 2006 Mar;15 (2):225–9. [PubMed] [Google Scholar]
- 45.Abuin G, Nieponice A. New findings on the origin of the blood supply to the atrioventricular node. Clinical and surgical significance. Tex Heart Inst J. 1998;25 (2):113–7. [PMC free article] [PubMed] [Google Scholar]
- 46.El-Maasarany Shirley H, Elazab Eman E B, Jensen Steen, Henein Michael Y. A-V nodal artery anatomy and relations to the posterior septal space and its contents. Int. J. Cardiol. 2010 May 14;141 (1):92–8. doi: 10.1016/j.ijcard.2008.11.205. [DOI] [PubMed] [Google Scholar]
- 47.Lin J L, Huang S K, Lai L P, Lin L J, Chen J H, Tseng Y Z, Lien W P. Distal end of the atrioventricular nodal artery predicts the risk of atrioventricular block during slow pathway catheter ablation of atrioventricular nodal re-entrant tachycardia. Heart. 2000 May;83 (5):543–50. doi: 10.1136/heart.83.5.543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Jain Ajay K, Smith Elliot J, Rothman Martin T. the coronary venous system: an alternative route of access to the myocardium. J Invasive Cardiol. 2006 Nov;18 (11):563–8. [PubMed] [Google Scholar]
- 49.von Lüdinghausen M. The venous drainage of the human myocardium. Adv Anat Embryol Cell Biol. 2003;168 ():I–VIII, 1-104. doi: 10.1007/978-3-642-55623-4. [DOI] [PubMed] [Google Scholar]
- 50.von Lüdinghausen M, Ohmachi N, Besch S, Mettenleiter A. Atrial veins of the human heart. Clin Anat. 1995;8 (3):169–89. doi: 10.1002/ca.980080302. [DOI] [PubMed] [Google Scholar]
- 51.Loukas Marios, Bilinsky Samuel, Bilinsky Esther, el-Sedfy Abraham, Anderson Robert H. Cardiac veins: a review of the literature. Clin Anat. 2009 Jan;22 (1):129–45. doi: 10.1002/ca.20745. [DOI] [PubMed] [Google Scholar]
- 52.de Oliveira Italo Martins, Scanavacca Maurício Ibrahim, Correia Aristides Tadeu, Sosa Eduardo Argentino, Aiello Vera Demarchi. Anatomic relations of the Marshall vein: importance for catheterization of the coronary sinus in ablation procedures. Europace. 2007 Oct;9 (10):915–9. doi: 10.1093/europace/eum175. [DOI] [PubMed] [Google Scholar]
- 53.Kamanu Santosh, Tan Alex Y, Peter C Thomas, Hwang Chun, Chen Peng-Sheng. Vein of Marshall activity during sustained atrial fibrillation. J. Cardiovasc. Electrophysiol. 2006 Aug;17 (8):839–46. doi: 10.1111/j.1540-8167.2006.00516.x. [DOI] [PubMed] [Google Scholar]
- 54.Okuyama Yuji, Miyauchi Yasushi, Park Angela M, Hamabe Akira, Zhou Shengmei, Hayashi Hideki, Miyauchi Mizuho, Omichi Chikaya, Pak Hui Nam, Brodsky Lauren A, Mandel William J, Fishbein Michael C, Karagueuzian Hrayr S, Chen Peng Sheng. High resolution mapping of the pulmonary vein and the vein of Marshall during induced atrial fibrillation and atrial tachycardia in a canine model of pacing-induced congestive heart failure. J. Am. Coll. Cardiol. 2003 Jul 16;42 (2):348–60. doi: 10.1016/s0735-1097(03)00586-2. [DOI] [PubMed] [Google Scholar]
- 55.Valderrábano Miguel, Chen Harvey R, Sidhu Jasvinder, Rao Liyun, Ling Yuesheng, Khoury Dirar S. Retrograde ethanol infusion in the vein of Marshall: regional left atrial ablation, vagal denervation and feasibility in humans. Circ Arrhythm Electrophysiol. 2009 Feb;2 (1):50–6. doi: 10.1161/CIRCEP.108.818427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Valderrábano Miguel, Liu Xiushi, Sasaridis Christine, Sidhu Jasvinder, Little Stephen, Khoury Dirar S. Ethanol infusion in the vein of Marshall: Adjunctive effects during ablation of atrial fibrillation. Heart Rhythm. 2009 Nov;6 (11):1552–8. doi: 10.1016/j.hrthm.2009.07.036. [DOI] [PMC free article] [PubMed] [Google Scholar]