Abstract
Background
Prostate cancer remains the most common non-skin cancer malignancy in men. Prostate-specific antigen (PSA) is recognized as a biomarker for the diagnosis, monitoring, and risk prediction of prostate cancer. Its use in the setting of prostate cancer screening has been controversial due to the risk of over diagnosis and over treatment.
Objective
Within Australia, there are inconsistent recommendations surrounding the use of PSA screening in clinical practice. In light of the 2016 PSA-screening guidelines by the major Australian health authorities, the current review aims to highlight the controversies and objectively outline the current recommendations within Australia.
Discussion
Health-care authorities across Australia have issued conflicting guidelines for prostate cancer screening culminating in confusion amongst health care practitioners and members of the public alike. A general consensus is held by other countries across the globe but differences amongst the specific details in how to best employ a PSA screening program still exist.
Keywords: Guidelines, Prostate cancer, Prostate-specific antigen, Screening
1. Introduction
Prostate cancer (PCa) is the most common cancer diagnosed in Australian men after non-melanoma skin cancer and accounts for the second highest number of male cancer deaths.1 PCa differs from many other cancers as the clinical course is highly variable. Often it is indolent and may not significantly affect overall survival but conversely, a small subgroup of PCa may represent highly aggressive disease with metastatic potential and may compromise quality of life and patient survival. Early PCa is asymptomatic, with lower urinary tract symptoms, hematuria, pelvic pain, and bony pain representing advanced disease. Accordingly, many men diagnosed with PCa never know they have the disease unless they are tested.
The rising incidence of PCa has made it an important health issue. In 2012, there were 3,079 deaths from PCa in Australia and it was estimated that this figure would increase to 3,440 deaths in 2015.2 Over the most recent decade of reports on cancer incidence in Australia, PCa diagnosis increased from 11,477 in 2000 to 19,993 in 2011. Recently, however, these figures have been declining with decreased rates in routine screening. In light of factors such as the growing Australian population and increasing life expectancy, the Australian Institute of Health and Welfare predicts that this number will continue to rise to approximately 25,000 and 31,000 in 2020.3
Prostate-specific antigen (PSA) is recognized as a biomarker for the diagnosis, monitoring, and risk prediction of PCa.4, 5, 6 PCa screening is characterized as the systematic examination of asymptomatic men (at risk) and is initiated by health authorities in order to reduce mortality and maintain quality of life.7 This is different to a case-finding whereby men enter into a process with their general practitioners (GPs) after discussion on the potential outcomes of a screening PSA. Routine PSA testing, in conjunction with digital rectal examination (DRE) are the hallmarks of PCa screening. To date, PSA screening has aided in the detection of PCa in millions of men across the world. Globally, the use of total PSA as a screening tool in the diagnosis of PCa has become a topic of much debate amongst healthcare governing bodies. Criticism surrounding its sensitivity and specificity profiles has prompted careful consideration of the benefits and risks of its use as a screening tool, and how it influences treatment decision making. Similarly, the role of DRE in screening programs has been a contentious topic in the guidelines. Subsequently, inconsistent practice has occurred culminating in significant implications for public health.8
1.1. Trends in Australia
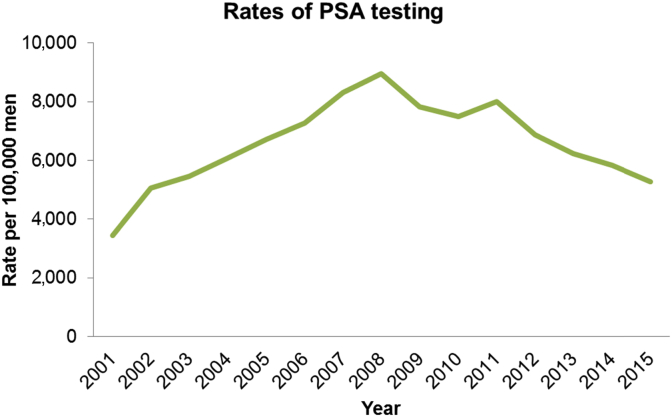
Britt et al reported that nearly 778,500 PSA tests were performed in Australia in 2012 with 80% of these tests for men aged 45–74 years. The GP management rate of PCa increased by 57% from 1998–2000 to 2009–2010, with an estimated 23 to 37 per 10,000 encounters. Similarly, the rate of pathology referrals for PSA tests increased significantly from an estimated 47 per 10,000 encounters in 2000–2001 to an estimated 86 per 10,000 encounters in 2007–2008 (see Fig. 1).9 Increased awareness about the risks of PCa and the availability of the PSA test among members of the community may justify this increase in referrals.10 Over the past decade, the increased number of PSA tests and cases of PCa diagnosed may explain the pronounced rate of encounters for the management of PCa in the GP setting.11
Fig. 1.
Trends in prostate-specific antigen testing.
In response to the US preventative task force recommendations against routine PSA screening in 2008, the number of PSA tests has fallen in the past few years by up to 35%.12 This has likely flowed on to a reduction in incidence, although the prevalence of the disease is stable. The impact of such a reduction creating a cohort of men presenting at a higher stage and with metastases is yet to be determined.
2. Controversies of PSA screening for PCa
2.1. Proposed benefits of PSA screening
Early detection of PCa through screening may allow for early disease stratification, prognosis, and treatment prior to disease progression. Freely available PSA testing in men aged 45–75 years in Austria conferred a notable shift to lower stages of PCa, as seen in one of the largest trials in this field to date.13 Similarly, data from the European Randomised Study of Screening for PCa (ERSPC) Rotterdam section14 revealed a statistically significant transition to improved histological grades and clinical stages on biopsy in the screening arm compared with the control arm. As such, there is robust evidence to suggest screening strategies result in earlier diagnosis of PCa.
The resulting earlier diagnosis and treatment of PCa may provide men with an oncological benefit. The ERSPC study spanning follow-up over 13 years demonstrated a significant 21% relative PCa mortality reduction in favor of screening, and the relative risk reduction in men actually screened was 27% after adjustment for selection effects.15 Indeed, the benefit of early treatment for localized PCa was clearly identified by the Scandinavian Prostate Cancer Group Trial 4 (SPCG-4). The SPCG-4 trial, which followed up 700 men showed that, at 15 years, the absolute risk reduction of dying from PCa was 6.1% following randomization to radical prostatectomy compared with watchful waiting.16 These findings were maintained at extended follow-up.17 It should be noted that these findings are somewhat conflicting with those of the Prostate Intervention Versus Observation Trial (PIVOT), which did not identify any statistically significant difference between the intervention and observation cohorts. However, on subgroup analysis, all-cause mortality was reduced in men with PSA >10 ng/mL after radical prostatectomy.18
Improved survival rates were also demonstrated by Roehl et al19; 7-year progression-free survival rates post-radical prostatectomy were higher in patients who underwent screening, compared with physician-referred patients (P = 0.002). These benefits do not account for the psychological benefits of a normal PSA test, especially those with a family history of PCa.
Finally, the impact of PSA testing cannot be ignored. Wherever PSA testing has been introduced, mortality from PCa has fallen.20, 21 Importantly and often not mentioned are the benefits of PSA screening in reducing presentations of men with metastatic disease by around 70%. The morbidity reductions are significant as are reduced costs on the healthcare system. In the same time period, breast cancer screening has reduced mortality but has had no impact at all on presentations with metastatic disease.22
2.2. Issues with PSA screening
The risks incurred by PCa screening, diagnosis, and the resulting treatment are potentially substantial. There is convincing evidence that PSA-based screening leads to substantial overdiagnosis of prostate tumors. Overdiagnosis occurs in men in whom PCa would not have been detected in their lifetime had it not been for screening, culminating in potentially unnecessary morbidity associated to invasive investigations, therapies, and also the mental implications of the cancer diagnosis.23 The estimated mean lead time in one study ranged from 5.4 years to 6.9 years, and overdiagnosis ranged from 23% to 42% of all screening-detected cancers.24 The findings from the Göteborg screening study similarly highlighted considerable overdiagnosis in PCa following organized screening compared to opportunistic PSA testing. This study concluded that opportunistic screening had minimal effect on the relative risk reduction in PCa mortality. Furthermore, this group estimated that almost twice the number of men needed to be diagnosed to save one man from dying from PCa compared to men offered an organized 2-yearly PSA screening.25
Findings from the ERSPC trial26 showed that screening increased PCa incidence by ∼80% through the effect of overdiagnosis. In addition to this, the risk of undergoing radical prostatectomy or radiation therapy was more than twice as high in the screened group than in the control group. Approximately 3% of men screened are diagnosed with aggressive PCa,23 therefore, early detection and attempted curative therapy can be life-saving. Given that the median age of PCa death is 80 years, often other causes of mortality ensue at this time regardless of the PCa detected.27
The benefits of screening were somewhat negated by large-scale USA data suggesting that PCa screening provided no reduction in mortality during the first 7 years of the trial, with similar results after 10 years.28 This trial was criticized as the control arm was contaminated with many patients having PSA testing (so really comparing screened with part screened unlikely to show difference) and has been roundly condemned for it being given the same weight as the ERSPC—a better-conducted trial lacking contamination.
As a result of active treatment, patients are exposed to the psychosocial stressors and morbidity. However, the newer practice of active surveillance for low volume Gleason (3 + 3 = 6) in appropriate patients has helped reduce the implications of overdiagnosis.29
In addition, issues surrounding PSA levels are widely recognized and include other possible influences on PSA levels, which include prostatitis, urinary tract infection, history of transuretheral resection, benign prostatic hyperplasia, and recent prostate biopsy. However, the degree to which these conditions exert an effect on PSA levels remains unclear.30 It is therefore pertinent for the clinician and patient to discuss the clinical relevance of PSA levels in the context of the patient's clinical picture. Furthermore, variation among PSA measurements between laboratories has been identified as a limitation to its accuracy as a screening tool. However, efforts to achieve international standardization of PSA assays exist.
2.3. Current guidelines in Australia
As discussed, there are inconsistencies amongst the majority of current guidelines advocated by healthcare authorities for PCa screening in Australia. These guidelines are freely available to GPs, clinicians, and the general public. Conflicting recommendations instill confusion, anxiety, and lack of confidence from the public.31 In Australia, the current published guidelines originate from the Royal Australasian College of General Practitioners (RACGP), the Cancer Council of Australia (in conjunction with The Prostate Cancer Foundation of Australia) and the Royal College of Pathologists.
The Royal College of Pathologists of Australia recommends that men who seek to assess their risk of PCa should be offered a PSA test and DRE from the age of 40 years as a baseline measure of risk. Men with PSA levels above the age-related median should be tested annually, while those men with PSA levels below the median could be tested less frequently. High-risk PSA levels warrant further investigation with prostate biopsy.32
In January 2016, a new guideline was developed by the Cancer Council of Australia, in conjunction with The Prostate Cancer Foundation of Australia (PCFA) based upon data from the National Health and Medical Research Council (NHMRC). This guideline involved collaboration between urologists and GPs, as well as many stakeholders involved in the care of men with PCa. As of May 2016, the RACGP endorsed the PCFA statement that patients who decide to undergo regular testing for PCa, should be offered PSA testing every 2 years from age 50 years to 69 years. Further investigation is to be offered if the total PSA is > 3.0 ng/mL.33 These recommendations align with the stance of The Urological Society of Australia and New Zealand.34, 35 Furthermore, the RACGP maintain that men aged 50–69 years (without a family history of PCa) should partake in informed decision making about PCa screening. The benefits and harms of prostate screening using the PSA test remain unclear, therefore, the decision to undergo screening is up to the individual to request testing from their GP.36 These recommendations are highlighted in Table 1.
Table 1.
Summary of clinical practice guidelines recommendations within Australia.
| Routine testing recommendations | Eligibility | Outlined regime | |
|---|---|---|---|
| Royal College of Pathology Australia (2016) | Recommended | In men whose life expectancy is > 7 y | Both a PSA test and a DRE from the age of 40 y on an annual basis |
| Prostate Cancer Foundation of Australia (2016) | Recommended | Men who are at average risk of prostate cancer who have been informed of the benefits and harms of testing, excluding men aged ≥ 70 y | PSA testing every 2 y from age 50 y to 69 y. Digital rectal examination is not recommended as a routine addition to PSA testing in the primary care setting |
| Cancer Council Australia (2016) | Recommended | For men at average risk of prostate cancer who have been informed of the benefits and harms of testing and who decide to undergo regular testing for prostate cancer | For men aged < 50 y who are concerned about their risk for prostate cancer, and have been informed of the benefits and harms of testing, and who wish to undergo regular testing for prostate cancer, offer testing every 2 y from age 45 y to age 69 y DRE is not recommended as a routine test |
| Urological Society of Australia and New Zealand (2016) | Recommended | In accordance with PCFA recommendations | PSA every 2 y |
| Royal Australian College of General Practice (2016) | In accordance with PCFA | ||
PCFA, Prostate Cancer Foundation of Australia; PSA, prostate-specific antigen.
2.4. Comparison with international recommendations
The consensus from recommendations from other parts of the world is geared against a routine test for PCa using a PSA test. In general, the view that routine PCa testing is not recommended is held by the American Academy of Family Physicians and The US Preventive Services Task Force. More specifically, The American Urological Association (AUA) recommends against PCa screening in men aged < 40 years and in men aged ≥ 70 years with a life expectancy of < 10 years. Furthermore, the AUA stance on asymptomatic men is that the greatest benefit of routine screening can be found in men aged 55–69 years. Men outside this age group are encouraged to voice their concerns to healthcare professionals in order to gain the necessary information to make an informed and individualized decision. Similarly, according to the European Association of Urology, mass screening of PCa is not indicated. However, early diagnosis on an individual basis is possible based on DRE and PSA testing using a similar approach to the AUA stance.37
3. Final recommendations for Australian men and their health professionals
Men who have a life expectancy of < 7 years should be informed that screening for PCa is not beneficial and has harms because many of the benefits from screening may take > 10 years to ensue. In keeping with this, the new guidelines state that because any mortality benefit from early diagnosis of PCa from PSA testing is not seen within < 6 years from testing, PSA testing is not recommended for men who are unlikely to live another 7 years. Conversely, men with favorable prognosis may be considered for surveillance screening protocols following adequate counseling. Of further relevance is the PSA velocity (PSAV) risk count, which is defined as the number of serial PSAV measurements exceeding 0.4 ng/mL/yr. The use of PSAV can significantly improve the performance characteristics of screening for overall PCa and high-grade disease by reducing unnecessary biopsies and PCa overdiagnosis compared with PSA alone.38
The lowering of the PSA threshold from 4.0 ng/mL to 3.0 ng/mL has been advocated in previous years. Most recently, the NHMRC decided on the lower threshold of 3.0 ng/mL. However, the high false-negative rate associated with this cutoff has real implications at a population level. Hence, it is probably now more appropriate to refer to age-adjusted and median levels provided on PSA tests to guide the most appropriate range for any patient. The necessity for a more flexible approach to threshold values has become apparent and is reflected in the various guidelines. The more recent guidelines offer a sensible pathway for testing to the public and their GPs. The use of PSA as a screening tool should take into account the age at which screening starts, and the use of different thresholds and screening intervals to ensure that the lag time to diagnosis and overtestin” are both minimized.39
Conflicts of interest
There are no disclosures. The corresponding author is not a recipient of a scholarship.
References
- 1.Cancer in Australia 2014: actual incidence data from 1982 to 2011 and mortality data from 1982 to 2012 with projections to 2014. Asia Pac J Clin Oncol. 2015;11:208–220. doi: 10.1111/ajco.12407. [DOI] [PubMed] [Google Scholar]
- 2.Prostate Cancer Statistics: Australian Institute of Health and Welfare; 2015 [Internet]. [cited 2016 Feb 24]. Available from: https://prostate-cancer.canceraustralia.gov.au/statistics.
- 3.Australian Institute of Health and Welfare . AIHW; Canberra: 2014. Cancer in Australia: an overview.http://www.aihw.gov.au/publication-detail/?id=60129550047 Cancer series no. 90 [Internet] Available from: [Google Scholar]
- 4.McGrath S., Christidis D., Perera M., Vela I., Manning T., Lawrentschuk N. Prostate cancer biomarkers: are we hitting the mark? Prostate Int. 2016;4:130–135. doi: 10.1016/j.prnil.2016.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Perera M., Manning T., Finelli A., Lawrentschuk N. Management of men with previous negative prostate biopsy. Curr Opin Urol. 2016;26:481–487. doi: 10.1097/MOU.0000000000000315. [DOI] [PubMed] [Google Scholar]
- 6.Yassaie O., B M., Perera M., Manning T., Lawrentschuk N., Malcolm A. Primary care follow-up of radical prostatectomy patients: a regional New Zealand experience. Prostate Int. 2016;4:136–139. doi: 10.1016/j.prnil.2016.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Devlin N., Parkin D. Does NICE have a cost-effectiveness threshold and what other factors influence its decisions? A binary choice analysis. Health Econ. 2004;13:437–452. doi: 10.1002/hec.864. [DOI] [PubMed] [Google Scholar]
- 8.Roobol M.J., Kirkels W.J., Schroder F.H. Features and preliminary results of the Dutch centre of the ERSPC (Rotterdam, the Netherlands) BJU Int. 2003;92(Suppl 2):48–54. doi: 10.1111/j.1464-410x.2003.04390.x. [DOI] [PubMed] [Google Scholar]
- 9.Britt H.M.G., Charles J., Henderson J., Bayram C., Pan Y., Valenti L. Australian Institute of Health and Welfare; Canberra: 2010. General practice activity in Australia 2009-10. [Google Scholar]
- 10.Let sleeping dogs lie? What men should know before getting tested for prostate cancer. Aust NZ J Public Health. 2011;35:96. [Google Scholar]
- 11.Australian Institute of Health and Welfare . AIHW; Canberra: 2013. Prostate cancer in Australia.http://www.aihw.gov.au/publication-detail/?id=60129545134 Cancer series 79 [Internet] Available from: [Google Scholar]
- 12.Lo J., Papa N., Bolton D., Murph D., Lawrentschuk N. Australian patterns of prostate cancer care: are they evolving. Prostate Int. 2016;4:20–24. doi: 10.1016/j.prnil.2015.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bartsch G., Horninger W., Klocker H., Reissigl A., Oberaigner W., Schonitzer D. Prostate cancer mortality after introduction of prostate-specific antigen mass screening in the Federal State of Tyrol, Austria. Urology. 2001;58:417–424. doi: 10.1016/s0090-4295(01)01264-x. [DOI] [PubMed] [Google Scholar]
- 14.van der Cruijsen-Koeter I.W., Vis A.N., Roobol M.J., Wildhagen M.F., de Koning H.J., van der Kwast T.H. Comparison of screen detected and clinically diagnosed prostate cancer in the European randomized study of screening for prostate cancer, section rotterdam. J Urol. 2005;174:121–125. doi: 10.1097/01.ju.0000162061.40533.0f. [DOI] [PubMed] [Google Scholar]
- 15.Schröder F.H., Hugosson J., Roobol M.J., Tammela T.L.J., Zappa M., Nelen V. Screening and prostate cancer mortality: results of the European Randomised Study of Screening for Prostate Cancer (ERSPC) at 13 years of follow-up. Lancet. 2014;384:2027–2035. doi: 10.1016/S0140-6736(14)60525-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Holmberg L., Bill-Axelson A., Steineck G., Garmo H., Palmgren J., Johansson E. Results from the Scandinavian Prostate Cancer Group Trial Number 4: a randomized controlled trial of radical prostatectomy versus watchful waiting. J Natl Cancer Inst Monogr. 2012;2012:230–233. doi: 10.1093/jncimonographs/lgs025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bill-Axelson A., Holmberg L., Garmo H., Rider J.R., Taari K., Busch C. Radical prostatectomy or watchful waiting in early prostate cancer. New Engl J Med. 2014;370:932–942. doi: 10.1056/NEJMoa1311593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wilt T.J., Brawer M.K., Jones K.M., Barry M.J., Aronson W.J., Fox S. Radical prostatectomy versus observation for localized prostate cancer. New Engl J Med. 2012;367:203–213. doi: 10.1056/NEJMoa1113162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Roehl K.A., Eggener S.E., Loeb S., Smith N.D., Antenor J.A., Catalona W.J. Survival results in patients with screen-detected prostate cancer versus physician-referred patients treated with radical prostatectomy: early results. Urol Oncol. 2006;24:465–471. doi: 10.1016/j.urolonc.2005.11.039. [DOI] [PubMed] [Google Scholar]
- 20.Walsh P.C. Cancer surveillance series: interpreting trends in prostate cancer—part I: evidence of the effects of screening in recent prostate cancer incidence, mortality, and survival rates. J Urol. 2000;163:364–365. doi: 10.1097/00005392-200001000-00095. [DOI] [PubMed] [Google Scholar]
- 21.Merrill R.M., Stephenson R.A. Trends in mortality rates in patients with prostate cancer during the era of prostate specific antigen screening. J Urol. 2000;163:503–510. [PubMed] [Google Scholar]
- 22.Welch H.G., Gorski D.H., Albertsen P.C. Trends in metastatic breast and prostate cancer—lessons in cancer dynamics. New Engl J Med. 2015;373:1685–1687. doi: 10.1056/NEJMp1510443. [DOI] [PubMed] [Google Scholar]
- 23.Barry M.J., Mulley A.J., Jr. Why are a high overdiagnosis probability and a long lead time for prostate cancer screening so important? J Natl Cancer Inst. 2009;101:362–363. doi: 10.1093/jnci/djp028. [DOI] [PubMed] [Google Scholar]
- 24.Draisma G., Etzioni R., Tsodikov A., Mariotto A., Wever E., Gulati R. Lead time and overdiagnosis in prostate-specific antigen screening: importance of methods and context. J Natl Cancer Inst. 2009;101:374–383. doi: 10.1093/jnci/djp001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Arnsrud Godtman R., Holmberg E., Lilja H., Stranne J., Hugosson J. Opportunistic testing versus organized prostate-specific antigen screening: outcome after 18 years in the Goteborg randomized population-based prostate cancer screening trial. Eur Urol. 2015;68:354–360. doi: 10.1016/j.eururo.2014.12.006. [DOI] [PubMed] [Google Scholar]
- 26.Roobol M.J., Kranse R., Bangma C.H., van Leenders A.G., Blijenberg B.G., van Schaik R.H. Screening for prostate cancer: results of the Rotterdam section of the European randomized study of screening for prostate cancer. Eur Urol. 2013;64:530–539. doi: 10.1016/j.eururo.2013.05.030. [DOI] [PubMed] [Google Scholar]
- 27.Ries LAG, Melbert D, Krapcho M, Stinchcomb DG, Howlader N, Horner MJ, et al., eds. SEER Cancer Statistics Review, 1975–2005 [Internet]. Bethesda (MD): National Cancer Institute. Available from: http://seer.cancer.gov/csr/1975_2005/.
- 28.Andriole G.L., Crawford E.D., Grubb R.L., 3rd, Buys S.S., Chia D., Church T.R. Mortality results from a randomized prostate-cancer screening trial. New Engl J Med. 2009;360:1310–1319. doi: 10.1056/NEJMoa0810696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yaxley J., Yaxley J., Gardiner R., Yaxley W. Prostate cancer - active surveillance as a management option. Aust Fam Physician. 2013;42:74–76. [PubMed] [Google Scholar]
- 30.National Health and Medical Research Council. Prostate-specific antigen (PSA) testing in asymptomatic men 2014 [Internet]. [cited 2015 Sep 30]. Available from: https://www.nhmrc.gov.au/_files_nhmrc/publications/attachments/men4d_psa_testing_asymptomatic_men_140304.pdf.
- 31.Medew J. September 22, 2015. Thousands of men abandoning controversial prostate cancer screening tests. Sydney Morning Herald. [Google Scholar]
- 32.RCPA announces position on PSA testing for prostate cancer [press release] The Royal College of Pathologists of Australasia; 2 August 2011. [Google Scholar]
- 33.Cancer Council Australia. Cancer Guidelines Wiki. Clinical practice guidelines PSA testing and early management of test-detected prostate cancer [Internet]. [cited 2016 15 Feb]. Available from: http://wiki.cancer.org.au/australia/Guidelines:PSA_Testing.
- 34.New prostate cancer testing guidelines to improve outcomes for Australian men [press release] Urological Society of Australia and New Zealand; Wednesday, 20 February 2016. [Google Scholar]
- 35.Urologists urge men at risk not to abandon prostate cancer testing [press release] Urological Society of Australia and New Zealand; 23 September 2015. [Google Scholar]
- 36.Prostate cancer screening info sheet. Royal Australian College of General Practitioners; 2015. [Google Scholar]
- 37.Mottet N., Bellmunt J., Briers E., van den Bergh R.C.N., Bolla M., van Casteren N.J. European Association of Urology; 2015. Guidelines on prostate cancer. [Google Scholar]
- 38.Loeb S., Metter E.J., Kan D., Roehl K.A., Catalona W.J. Prostate-specific antigen velocity (PSAV) risk count improves the specificity of screening for clinically significant prostate cancer. BJU Int. 2012;109:508–513. doi: 10.1111/j.1464-410X.2011.10900.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Oefelein M.G., Feng A., Scolieri M.J., Ricchiutti D., Resnick M.I. Reassessment of the definition of castrate levels of testosterone: implications for clinical decision making. Urology. 2000;56:1021–1024. doi: 10.1016/s0090-4295(00)00793-7. [DOI] [PubMed] [Google Scholar]