Abstract
Background
In humans and rodents obesity appears to promote some cancers by increasing incidence, tumor aggressiveness, recurrence, and fatality. However, the relationship between obesity and cancer in dogs has not been thoroughly evaluated.
Hypothesis/Objectives
Whether body condition score (BCS) at the time of lymphoma (LSA) or osteosarcoma (OSA) diagnosis in dogs is predictive of survival time (ST) or progression‐free interval (PFI). We hypothesized that an overweight body state at the time of cancer diagnosis would be associated with negative outcomes.
Animals
Dogs with LSA (n = 270) and OSA (n = 54) diagnosed and treated between 2000 and 2010.
Methods
Retrospective case review. Signalment, body weight, BCS, cancer diagnosis and treatment, relevant clinicopathologic values, and survival data were collected. Dogs were grouped by BCS (underweight, ideal, and overweight) and ST and PFI were compared.
Results
Overall, 5.5% of dogs were underweight, 54.0% were ideal weight, and 40.4% were overweight at diagnosis. Underweight dogs with LSA had shorter ST (P = .017) than ideal or overweight dogs. BCS was not associated with ST for OSA (P = .474). Progression‐free interval did not differ among BCS categories for either cancer.
Conclusions and Clinical Importance
Obesity was not associated with adverse outcomes among dogs with LSA or OSA in this retrospective study; however, being underweight at the time of diagnosis of LSA was associated with shorter survival. More research is needed to elucidate the relationship between excessive body weight and cancer development and progression in dogs.
Keywords: Lymphoma, Nutrition, Obesity, Osteosarcoma
Abbreviations
- BCS
body condition score
- CHOP
cyclophosphamide (C), doxorubicin (H=hydroxydaunorubicin), vincristine (O=Oncovin®), and prednisone (P)
- LSA
lymphoma/lymphosarcoma
- MTR
maximal treatment response
- OSA
osteosarcoma
- PFI
progression‐free interval
- ST
survival time
In people, obesity has been associated with increased overall mortality from cancer,1 increased aggressiveness and decreased response to treatment in some cancers,2 and a higher rate of cancer recurrence.3 Altered concentrations of many hormones and a chronic, low‐grade inflammatory state characterize obesity.4 Many of the biomarkers of obesity—hyperinsulinemia; increased concentrations of insulin‐like growth factor‐1, leptin, sex hormones, and inflammatory cytokines; and decreased concentrations of adiponectin—have been demonstrated to promote tumor growth in people and animal models.5 Conversely, caloric restriction can slow tumor progression and prevent metastasis in rodent models.5 These data suggest a role for nutrition and calorie intake in the pathogenesis and progression of cancer. To date, obesity has been associated with a worse cancer outcome and sometimes a higher rate of cancer recurrence in human cancers of the breast,3 colon,6 and prostate.7 The data for other types of cancers are less clear or lacking.
Spontaneously occurring cancer is widely reported to be the most common cause of death in dogs; lifelong prevalence in some breeds such as the Golden Retriever exceeds 50%.8 Overweight body condition is also common, with 34–59% of all American dogs being overweight or obese.9, 10, 11 Similarly, 15–29% of dogs that presented to the oncology service at 2 veterinary teaching hospitals were obese (>20% over ideal body weight),10, 12 with another 21% overweight.10 Obesity is associated with increased risk of mast cell tumors,10 mammary tumors,13 and transitional cell carcinoma of the bladder in dogs14 in retrospective and epidemiologic studies. However, despite the large numbers of dogs with cancer that are overweight and obese, the associations between obesity and cancer survival and response to treatment have not been thoroughly evaluated in dogs.
Perhaps more intuitive, underweight humans15, 16 and cats17 with cancer generally have shorter survival times than ideal or overweight individuals. This same association in dogs has not been reported in the literature. There are also lingering questions about the “obesity paradox”—where overweight or even obese individuals with certain chronic and acute diseases have longer survival times compared to ideal weight individuals, even when the overweight state is a known risk factor for the initial development of the disease. A well‐known example of this phenomenon can be seen in humans with cardiovascular disease.18 At this time, it is not clear whether this phenomenon also exists for humans or companion animals with cancer, but the preponderance of the data at this time suggests the opposite—that obesity is a risk factor for both the development of many types of cancer and for a poorer prognosis.1, 2, 5
The aims of this study were to determine whether body condition score (BCS) at the time of diagnosis of lymphoma (LSA) and osteosarcoma (OSA) is associated with progression‐free interval (PFI) and survival time (ST). We hypothesized that an overweight or obese body condition would be associated with a shorter ST and PFI compared to ideal weight dogs and that underweight dogs would have shorter ST than ideal weight dogs.
Materials and Methods
Case Identification
Medical records were reviewed retrospectively. Potential cases were identified by searching the Cummings School of Veterinary Medicine at Tufts University medical records database and the oncology service case logs for dogs meeting initial inclusion criteria. These criteria required a histologically or cytologically confirmed diagnosis of LSA or appendicular OSA, or presumptive OSA based on radiographs and malignant clinical behavior (eg, classic lytic bone lesion in a common location for OSA with or without radiographic findings consistent with metastasis). These specific cancer types were chosen based on a review of case logs which suggested that adequate caseload was available. For all dogs, a BCS and body weight recorded within 1 month of diagnosis and survival of at least 1 week postdiagnosis (to exclude cases where euthanasia was elected at time of diagnosis) were required. Another exclusion criterion was history of, or later development of, a second primary malignant tumor, which could have altered prognosis. Dogs with LSA that were categorized as stage 1 extranodal (except for gastrointestinal and cutaneous forms) were also excluded because of their rarity (n = 4) and highly variable survival times compared to other types of LSA based on the authors’ clinical experience. The record search was limited to cases with diagnosis or treatment within the date range of January 1, 2000 to December 31, 2010; this assured the cases were recent enough to reflect currently available diagnostics and treatments (eg, chemotherapy protocols) with records still available for review, but old enough that the vast majority would be deceased for survival analysis.
Study Design
A chart review was performed for each case that met inclusion and exclusion criteria, and data were collected on the dogs’ cancer diagnosis, type and duration of treatment, and survival time. Signalment data, BCS, and body weights (within 1 month of diagnosis and the last available weight and BCS prior to death) were also collected. Specific clinicopathologic parameters known or suspected to correlate with prognosis for LSA (ie, albumin, calcium, and whether hematocrit or PCV was within the reference range) or OSA (ie, alkaline phosphatase) were also recorded. Dogs were considered hypoalbuminemic or hypercalcemic if their albumin or total calcium values were below or above the laboratory reference range at the time of cancer diagnosis, respectively. Likewise, dogs were considered anemic if their packed cell volume or hematocrit was below the laboratory reference range at the time of diagnosis. Cases were categorized by the World Health Organization classification system19 by location (ie, generalized/multicentric including hepatic and splenic, alimentary/gastrointestinal, cutaneous/skin, thymic/mediastinal, other), stage (I‐V), and immunophenotype (B‐ or T‐cell) for LSA and by location (forelimb versus hindlimb) and presence of metastasis at the time of diagnosis for OSA. Substage a or b was assigned to LSA cases either based on record notation at the time of presentation or on the basis of clinical signs at the time of diagnosis—dogs that were presented for weight loss, poor appetite, gastrointestinal signs, etc. were retroactively assigned substage b, whereas those that were clinically ideal except for enlarged lymph nodes or organomegaly were assigned substage a. For LSA, not all dogs underwent full staging (eg, bone marrow aspiration was rare), so the stages recorded represent the minimum stage confirmed in each dog.
The treatment regimen and the date that treatment began were recorded and the date of the first treatment was used to calculate the timing of the maximal treatment response (MTR) for LSA. The most common treatment regimens for canine lymphoma are referred to as “CHOP‐based protocols”. These protocols are combination chemotherapy protocols that include cyclophosphamide (C), doxorubicin (H = hydroxydaunorubicin), vincristine (O=Oncovin®), and prednisone (P). Initial treatment regimens were recorded from the medical records and categorized as CHOP‐based, lomustine, excisional surgery, radiation therapy, corticosteroids alone (palliative), or a combination of treatment modalities (including investigational therapies employed in clinical trials). The MTR was recorded along with the timing of the MTR (whether or not complete remission was achieved within 6 weeks as is typical for chemotherapy‐sensitive LSA cases in our hospital), and indicated the highest degree of treatment response (complete clinical remission, partial remission, stable disease, progressive disease) achieved. For OSA, treatments were categorized as surgery alone, surgery plus chemotherapy, radiation therapy plus chemotherapy, or radiation therapy alone (palliative). The PFI for both cancers was defined as the time from diagnosis until disease progression was recorded. Overall ST was calculated as time from diagnosis to death.
Weight change from diagnosis to death, or from postamputation (the first available weight in the record after amputation, typically in the first 1–2 days postsurgery) to death for dogs with OSA that had an amputation, was calculated as a percentage, and then categorized into the following groups: loss of ≥10% body weight, loss of <10% body weight, no change (within 1% of diagnosis or postamputation weight), gain of <10% body weight, or gain of ≥10% body weight.
After the initial record review, referring veterinarians were contacted to provide any essential missing information. Cases with inadequate data on treatment response and progression were included in the survival analysis only.
Data Analysis
The primary variables of interest were BCS at time of diagnosis, overall ST, and PFI. The MTR and its timing were secondary endpoints for LSA only. For analysis, cases were assigned BCS categories as follows: BCS <4/9 were considered underweight, 4/9 ≤ BCS <6/9 were considered ideal, and BCS ≥6/9 were considered overweight. All analyses were conducted across these BCS categories. For overall survival analysis, the outcome was death caused by any cause. For those subjects still alive during the study, survival time was calculated for the date of last follow‐up and right censored. If the date of death was unknown or the dog was lost to follow‐up, the case was censored based on the last known date that the dog was alive. Survival analysis and analysis of PFI were conducted by Kaplan‐Meier curves and Cox proportional hazards models to determine the effect of individual variables (univariate) on the endpoints. Chi‐square analysis was performed to compare categorical data among subgroups of the study population. Independent variables that were significant at the level of P ≤ .10 were then included in multivariate analyses by linear regression analysis and stepwise (backward) estimation. For the multivariate analyses, the 4 dogs that were confirmed alive were excluded. When the linear regression showed significance for a variable that had more than 2 categories, Kruskal‐Wallis one‐way ANOVA was used along with the Dwass‐Steel‐Critchlow‐Fligner test for posthoc comparison. All analysis was performed with commercial statistical software,1 , 2 with a P < .05 considered statistically significant. All data are presented as median (range) as many of the data were not normally distributed.
Results
LSA
Two hundred and seventy dogs with LSA were included in this study (Table 1). The most common breeds represented were mixed breed (58/270, 21.5%), Golden Retriever (37/270, 13.7%), Labrador Retriever (21/270, 7.8%), and German Shepherd (14/270, 5.2%). LSA types (locations) included 227/270 (84.1%) multicentric, 20/270 (7.4%) mediastinal, 15/270 (5.6%) gastrointestinal, 6/270 (2.2%) cutaneous, and 2/270 (0.7%) other. One dog did not have staging reported in the medical record and review of the record did not allow for retroactive assignment of stage; for the rest of the cases there were 11/269 (4.1%) stage I, 7/269 (2.6%) stage II, 59/269 (21.9%) stage III, 98/269 (36.3%) stage IV, and 94/269 (34.8%) stage V. Immunophenotyping was not available for 132 dogs; of the cases with typing available, 85/138 (61.6%) were B‐cell and 53/138 (38.4%) were T‐cell. At diagnosis, 165/270 (61.1%) dogs showed signs of systemic illness, with 127/270 (47.0%) reporting decreased appetite.
Table 1.
Descriptive data of the populations of dogs with lymphoma and osteosarcoma as well as all dogs combined. Data are presented as median (range)
| Variable | Lymphoma | Osteosarcoma | All Dogs |
|---|---|---|---|
| Number of dogs | 270 | 54 | 324 |
| Age (years) | 7.6 (0.8 to 15.1) | 8.7 (1.3 to 13.6) | 7.8 (0.8 to 15.1) |
| Sex | |||
| Male Intact | 17/270 (6.3%) | 5/54 (9.3%) | 22/324 (6.8%) |
| Male Neutered | 140/270 (51.9%) | 29/54 (53.7%) | 169/324 (52.2%) |
| Female Intact | 5/270 (1.9%) | 0/54 (0%) | 5/324 (1.5%) |
| Female Spayed | 108/270 (40.0%) | 20/54 (37.0%) | 128/324 (39.5%) |
| Initial BCS | 5/9 (1 to 9) | 5.25 (3 to 9) | 5 (1 to 9) |
| Initial BCS Category | |||
| Underweight | 16/270 (5.9%) | 2/54 (3.7%) | 18/324 (5.5%) |
| Ideal weight | 148/270 (54.8%) | 27/54 (50.0%) | 175/324 (54.0%) |
| Overweight | 106/270 (39.3%) | 25/54 (46.3%) | 131/324 (40.4%) |
| Final BCS | 5/9 (1 to 9) | 5/9 (1 to 9) | 5/9 (1 to 9) |
| Final BCS Category | |||
| Underweight | 19/155 (12.3%) | 1/25 (4.0%) | 20/180 (11.1%) |
| Ideal weight | 63/155 (40.6%) | 15/25 (60%) | 78/180 (43.3%) |
| Overweight | 73/155 (47.1%) | 9/25 (36%) | 82/180 (46.6%) |
| Anemia | 82/257 (31.9%) | 10/47 (21.3%) | 92/304 (30.3%) |
| Hypercalcemia | 53/253 (20.9%) | 4/44 (9.1%) | 57/297 (19.2%) |
| Total calcium (mg/dL) | 10.3 (6.0 to 21.0) | 10.4 (9.0 to 12.0) | 10.3 (6.0 to 21.0) |
| Hypoalbuminemia | 51/254 (20.1%) | 3/44 (6.8%) | 54/298 (18.1%) |
| Albumin (mg/dL) | 3.2 (1.1 to 4.6) | 3.4 (2.2 to 4.1) | 3.3 (1.1 to 4.6) |
| Weight change between diagnosis and death (% change) | −2.37 (−30.9 to 37.9) | −1.65 (−27.5 to 26.6) | −2.23 (−30.9 to 37.9) |
| Lost >10% | 50/236 (21.2%) | 7/44 (15.9%) | 57/280 (20.4%) |
| Lost ≤10% | 80/236 (33.9%) | 16/44 (36.4%) | 96/280 (34.3%) |
| No change | 14/236 (5.9%) | 5/44 (11.4%) | 19/280 (6.8%) |
| Gained ≤10% | 53/236 (22.5%) | 11/44 (25.0%) | 64/280 (22.9%) |
| Gained >10% | 39/236 (16.5%) | 5/44 (11.4%) | 44/280 (15.7%) |
| Alkaline phosphatase | N/A | 136 (19 to 767) | N/A |
Key—BCS = body condition score, underweight = <4/9, ideal weight = 4 to <6/9, and overweight = ≥6/9.
The BCS distribution for the dogs at diagnosis was 1 (1 dog), 2–2.5 (3), 3–3.5 (12), 4–4.5 (48), 5–5.5 (100), 6–6.5 (59), 7–7.5 (29), 8 (9), and 9 (9). Over half of the dogs (130/237, 54.9%) lost weight between diagnosis and death, but adequate information was often not available in the medical record to allow more detailed evaluation of the temporal associations of that weight loss with treatment or disease progression; weight change as a percentage of original body weight ranged from −30.9 to 37.9% (median = loss of 2.4%) with 50/236 (21.2%) losing ≥10% of initial body weight. From the first BCS to the final, the proportions of dogs categorized as underweight and overweight increased numerically, whereas those categorized as ideal weight decreased (Table 1).
Two hundred and sixty‐eight dogs (99.3%) received some form of cancer treatment; treatment regimens included CHOP‐based chemotherapy (217/268, 81.0%), lomustine chemotherapy (13/268, 4.9%), palliative steroids alone (6/268, 2.2%), or other [32/268, 11.9%—included radiation therapy, high‐dose cyclophosphamide, MPD‐01 (derived from a Japanese mushroom), therapeutic vaccination, and combinations of the other protocols]. The maximal treatment response recorded was complete remission for 158/259 (61%) dogs, partial remission for 61/259 (23.6%) dogs, stable disease for 28/259 (10.8%) dogs, and progressive disease for 12/259 (4.6%) dogs. These data were not available for 11 dogs. There was no difference in whether dogs achieved complete remission by 5–6 weeks or the MTR among dogs of different BCS categories.
Progression‐Free Interval
Two hundred and seven (76.7%) cases had documented progression of their disease with a median PFI of 132 days (7–1,137 days); data on PFI for the remaining cases were unavailable. For underweight dogs, median PFI was 99 days (35–257 days); for ideal weight dogs, median PFI was 141 days (9–1,137 days); for overweight dogs, median PFI was 143 days (7–1,108 days). Body condition score category at diagnosis was not associated with PFI (P = .150). The presence of anemia (P = .030), hypoalbuminemia (P = .007), and the type of treatment (P < .001) was associated with PFI on univariate analysis, as was substage (P < .001) and immunotype (P = .003). On multivariate analysis, only substage and immunophenotype were independent variables; dogs with substage b (showing clinical illness) had shorter PFI than dogs with substage a (P = .004) and dogs with B‐cell lymphoma had longer PFI than dogs with T‐cell (P = .026). No significant interaction between substage and immunophenotype was identified (P = .06).
Survival Time
Only 4 dogs (1.5%) were known to be alive at the time of analysis. Two hundred and sixteen dogs were known to be deceased; of these, 26 dogs died and 190 dogs were euthanized. Fifty‐four dogs were lost to follow‐up and censored by the last known date that they were alive. The median ST for dogs with LSA was 218 days (7–3,720 days). For underweight dogs, median ST was 176 days (15–623 days); for ideal weight dogs, median ST was 219 days (9–3,720 days); for overweight dogs, median ST was 225 days (range, 7–2,737 days). On univariate analysis, underweight dogs had a significantly shorter ST (P = .017, Fig 1) compared to the ideal and overweight groups, but there was no significant difference in survival between ideal weight and overweight dogs. There was no difference in ST among BCS categories on multivariate analysis.
Figure 1.
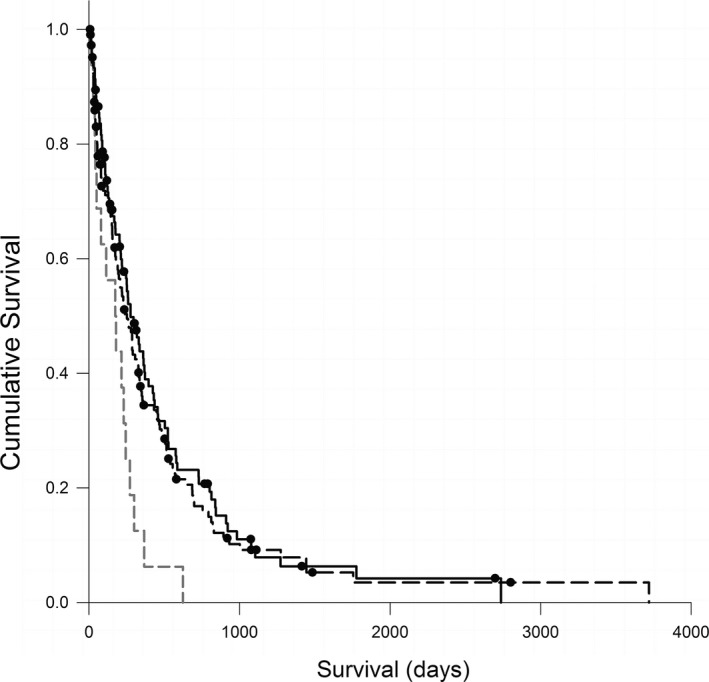
Kaplan‐Meier survival curves comparing survival time (days) for 270 dogs with lymphoma in different body condition score categories at the time of diagnosis—overweight (n = 106, black dashed line); ideal weight (n = 148; solid black line); and underweight (n = 16; gray dashed line), P = .017. Circles represent censored cases.
Stage (P < .001), substage (P < .001), and immunophenotype (P = .010) were associated with ST on univariate analysis; dogs with substage b or T‐cell LSA had shorter survival compared to substage a or B‐cell LSA, respectively. The presence of anemia (P = .004) and hypoalbuminemia (P = .002) at diagnosis was also associated with shorter ST on univariate analysis, but hypercalcemia as assessed by total calcium concentration was not (P = .584). Treatment type was also positively associated with survival (P < .001), with dogs receiving CHOP‐based treatment having the longest survival. The anatomic location of lymphoma was not associated with ST on univariate analysis, but demonstrated a strong trend with multicentric lymphoma having the longest median survival [231 days (7–3,747 days); P = .052]. On multivariate analysis, only change in weight during treatment, anemia, immunophenotype, and stage remained significant—a gain of more than 10% of body weight during treatment was associated with longer survival compared with dogs that maintained weight or lost or gained < 10% of initial body weight (P < .001; Fig 2). Dogs with normal hematocrit and dogs with stage 2 disease lived longer than dogs with anemia (P = .007) or other stages (P = .001), respectively. Dogs with T‐cell lymphoma had shorter ST than dogs with B‐cell (P = .012). Significant interactions were identified between stage and body weight change (P < .001), immunophenotype (P < .001), and anemia (P = .001).
Figure 2.
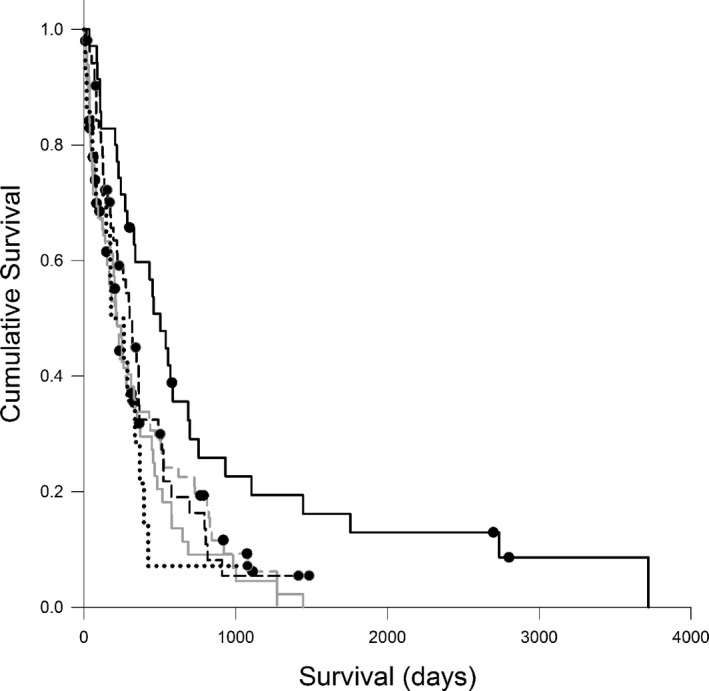
Kaplan‐Meier survival curves comparing survival time (days) for 236 dogs with lymphoma that lost <10% body weight (n = 80; dashed gray line), lost >10% body weight (n = 50; solid gray line), maintained weight (n = 14, dotted black line), gained <10% of body weight (n = 53; dashed black line), or gained >10% body weight (n = 39; solid black line), P = .003. Circles represent censored cases.
OSA
Fifty‐four dogs with OSA were included (Table 1). The most common breeds represented were mixed breeds (15/54, 27.8%), Rottweiler (8/54, 14.8%), Labrador Retrievers (6/54, 11.1%), Golden Retrievers (5/54, 9.3%), and Doberman Pinscher (5/54, 9.3%). The BCS distribution at diagnosis was as follows: 2.5 (1 dog), 3(1), 4–4.5 (8), 5–5.5(19), 6–6.5(19), 7 (3), 8 (2), and 9 (1). About half (23/44, 52.3%) of dogs lost weight between diagnosis and death, after adjusting for decreases in weight secondary to limb amputation when appropriate, whereas 16/44 (36.4%) gained weight. Thirty‐six dogs (66.7%) had a primary tumor in a forelimb versus 18/54 (33.3%) with a tumor in a hindlimb. Metastasis was identified at the time of diagnosis in 4/47 (8.5%) that had staging performed, but it was not associated with ST (P = .802) or PFI (P = .474).
Progression‐Free Interval
The median PFI for dogs with OSA was 150 days (20–1,173 days). Weight change between diagnosis and death was associated with increased PFI on multivariate analysis with dogs gaining more than 10% of body weight living longer than those gaining <10% or losing <10% (both P < .001). There was no significant association detected among BCS category or any other variables and PFI on univariate or multivariate analysis.
Survival Time
No dogs were confirmed alive at the time of analysis. Nine dogs (16.7%) were lost to follow‐up and were censored by the last known date that they were alive. Of the dogs confirmed to be deceased, 2 died, 40 were euthanized, and the cause of death was unknown in 3. Median ST for dogs with OSA was 192.5 days (20–1,271 days) and there was no association with BCS category (P = .474). On univariate survival analysis, anemia (P = .005), hypoalbuminemia (P = .002), and change in body weight after diagnosis (P = .003) were associated with ST. Only weight change remained significant on multivariate analysis with dogs gaining more than 10% of body weight living longer than those gaining <10% or losing <10% (both P < .001).
Discussion
Contrary to one of our hypotheses, obesity was not associated with shorter ST or PFI among dogs with LSA and OSA. For dogs with LSA, there was also no difference among the groups for MTR or whether the dogs achieved clinical remission within 6 weeks of starting treatment among BCS categories. This is in contrast to studies that have shown increased tumor aggressiveness2 along with a higher risk of death caused by cancer1, 7, 20 and shorter survival times21 in overweight humans.
There are several possible reasons why a detrimental association between overweight or obese body condition and outcomes was not observed in this study. The first is that the majority of dogs in the overweight group for both cancers had BCS between 6 and 7 (“overweight”) rather than 8 or 9 (“obese”). It might be that being overweight is not as harmful as being obese. If that is true, then the relative lack of dogs of BCS 8–9 versus 6–7 could have obscured a survival difference between the normal weight and obese dogs. Another possibility is that the effects of obesity might differ with different types of cancer. Indeed, while it is well accepted that obesity increases the risk of development of many types of cancers in people,5 thus far it has only been clearly reported to increase cancer‐related mortality in colorectal cancer and postmenopausal breast cancer.3, 6 Canine LSA is most similar to non‐Hodgkin's lymphoma in humans and the literature is rather inconsistent on the association between obesity and this type of cancer: studies indicating longer, shorter, or no change in ST among overweight or obese people have each been reported.15, 20, 22, 23, 24 Another possibility to explain the lack of association noted in this study is that the effect might be relatively modest and might require a much larger study population to see a difference among BCS categories. Most of the human studies that have demonstrated inverse relationships between body mass index and ST have included considerably higher numbers of cases than did this study.
A similar effect was not seen in dogs with OSA likely because of small numbers of dogs with OSA and the different disease course seen with this cancer compared with LSA. One study reported shorter ST in overweight children with OSA,25 but this study has been contested.26, 27 Overall, there is not nearly as much literature investigating obesity and OSA in people as there is for many other types of cancer, so there is not much consensus. In addition, as OSA typically affects human children, whereas it is most common in older dogs, the mechanisms and associations might differ, and it might be harder to translate findings in humans to those in dogs.
As we hypothesized, median ST was significantly shorter for dogs with LSA categorized as underweight compared to ST for the ideal and overweight groups in the univariate analysis, as occurs in humans with LSA,16, 23 urothelial carcinoma,28 esophageal cancer,29 and gastric cancer,30 and cats with various types of cancers.17 Similar results were not seen for OSA, but there were very few underweight dogs with OSA included in this study. It has been reported that people with OSA who lost more than 4.5 kg (10 pounds) of body weight had a poorer prognosis, but it remains unclear what percentage of body weight loss this represented, whether the patients were at a normal weight at the time of diagnosis or whether the weight of an amputated limb was taken into account;31 therefore, it is hard to draw conclusions on the associations between being underweight and survival in people with OSA.
For dogs with either LSA or OSA, gaining 10% or more body weight during the course of treatment was associated with longer ST than those that gained less than 10%, maintained, or lost weight. A similar situation has been reported in dogs with heart disease, where dogs that gained weight had longer survival compared to those that lost or maintained weight.32 This might be a secondary, rather than a primary effect (ie, dogs that live longer have a longer duration of time to gain weight), but weight loss (and particularly muscle loss) can have a primary role in survival as well. Shorter ST in underweight dogs might be a reflection of more severe disease, loss of lean body mass, or chronic malnutrition, all of which can compromise host defenses and alter treatment protocols. Dog owners might also be more likely to opt for euthanasia for dogs that lose weight compared to dogs that maintain or gain weight during treatment, which can confound survival time. While it is likely that weight loss during treatment reflects a poorer response to treatment, more aggressive disease, or the contribution of comorbid conditions, suboptimal calorie and nutrient intake also can play a role, making careful attention to nutrition important. This strategy can be achieved by a nutritional assessment at every visit, which includes assessment of body weight, BCS, muscle condition score, and diet history.33, 34 In addition, making specific nutritional recommendations and adjusting these recommendations based on patient response can play a role in maintaining optimal nutritional status in dogs with cancer.
Other factors associated with shorter survival for dogs with LSA included anemia and T‐cell LSA. These data agree with previous studies and suggest that hematocrit at time of diagnosis might be a useful prognostic indicator.35, 36, 37, 38
There are some limitations to this study. Body condition score, our main variable of interest, is subjective39 and we had to rely on BCS performed by numerous clinicians across and within cases. It has been the authors’ experience that clinicians tend to underestimate BCS, so it is possible that some overweight animals were included in the ideal weight group, but less likely that the reverse occurred. Coat length and thickness can also complicate estimation of BCS as the authors appreciate that thick‐coated dogs might feel like they have more “fat padding” over their ribs than thin‐coated dogs. Another potential limitation is that the BCS within a month of the time of diagnosis was used to categorize cases into underweight, ideal weight, and overweight; however, this value might not be the most accurate marker of a dog's nutritional status at the time that the cancer first developed. For example, a history of anorexia or weight loss prior to diagnosis could result in a BCS at the time of diagnosis that was not reflective of the typical BCS of the animal prior to the development of cancer. In addition, BCS measurements were not always available on the exact day of diagnosis—some of the BCS measurements were recorded after the diagnosis, others before, and some on the day of diagnosis (although all within 1 month of diagnosis). It has been suggested that the timing of body mass index measurement (ie, prediagnosis versus at the time of diagnosis) in humans might alter interpretation of associations among various body mass index categories and cancer prognosis.40 Moreover, body condition scoring is an estimate of body fat, but changes in BCS do not correlate with loss of muscle until the very lowest scores (1 and 2/9).39 In humans, cachexia, or muscle loss because of systemic illness, regardless of overall body weight and BMI, has been associated with poorer survival.41 Although several different subjective muscle condition scoring systems for dogs have been proposed, these systems have not been well validated and adoption by individual clinicians is quite variable.12, 34 We were thus unable to assess muscle condition reliably from the medical records of dogs in this study. Unlike in studies of rodents and humans, dogs with cancer can be euthanized as a result of owner's financial constraints, progressive disease, treatment failure, or treatment adverse effects, and including euthanasia as a cause of death could confound true assessment of ST. Nearly all of the dogs in this study were euthanized versus dying from their cancer directly. Finally, animals presented to our teaching hospital might not reflect the general population of dogs with LSA and OSA.
The number of LSA dogs with clinical substage b was higher in this study than has been reported in previous studies.35, 38 The retrospective nature of this study made it difficult to assess severity of the systemic signs, so clinical substage was frequently assigned based on systemic signs reported in the record (such as decreased appetite, lethargy, weight loss, vomiting, or diarrhea). It is possible that some of these animals might have been categorized as substage a if evaluated prospectively. The PFI and ST for both LSA and OSA reported here are shorter than those documented in other clinical studies. This is likely as a result of the inclusion of dogs that received palliative care rather than just regular standard‐of‐care treatment in this investigation.
In conclusion, our data do not indicate that overweightedness/obesity as defined as a BCS ≥6/9 at diagnosis adversely affects progression or survival in dogs with LSA or OSA. However, weight gain during treatment was associated with increased survival for both OSA and LSA, whereas anemia, T‐cell phenotype, and higher stage in dogs with LSA were associated with shorter ST. A large, prospective case–control study would enable more definitive conclusions to be drawn about these relationships. We are aware of one such study in golden Retrievers that is ongoing3 which will provide additional data that could help elucidate these complex issues in dogs.
Acknowledgments
The authors acknowledge the Tufts Harrington Oncology program for providing cases and assistance with study design.
Grant Support: This project was funded by the Merial Veterinary Scholars program. The project described was also supported in part by the National Center for Advancing Translational Sciences, National Institutes of Health, Award Number UL1TR000073, and the U.S. Department of Agriculture, under agreement No. 58‐1950‐7‐707. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH, or do any opinions, findings, conclusion, or recommendations expressed in this publication necessarily reflect the view of the U.S. Dept. of Agriculture.
Conflict of Interest Declaration: Authors declare no conflict of interest.
Off‐label Antimicrobial Declaration: Authors declare no off‐label use of antimicrobials.
This project was completed at the Cummings School of Veterinary Medicine at Tufts University. This manuscript was presented as a research report at the 2015 ACVIM Forum in Indianapolis, IN.
Footnotes
Systat, Cranes Software International, Ltd. San Jose, California
SPSS, IBM Corporation, Armonk, New York
Morris Animal Foundation Golden Retriever Lifetime Study http://caninelifetimehealth.org
References
- 1. Calle EE, Rodriguez C, Walker‐Thurmond K, et al. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N Engl J Med 2003;348:1625–1638. [DOI] [PubMed] [Google Scholar]
- 2. Freedland SJ, Aronson WJ, Kane CJ, et al. Impact of obesity on biochemical control after radical prostatectomy for clinically localized prostate cancer: A report by the Shared Equal Access Regional Cancer Hospital database study group. J Clin Oncol 2004;22:446–453. [DOI] [PubMed] [Google Scholar]
- 3. Jain R, Strickler HD, Fine E, et al. Clinical studies examining the impact of obesity on breast cancer risk and prognosis. J Mammary Gland Biol Neoplasia 2013;18:257–266.24221746 [Google Scholar]
- 4. Greenberg AS, Obin MS. Obesity and the role of adipose tissue in inflammation and metabolism. Am J Clin Nutr 2006;83:461S–465S. [DOI] [PubMed] [Google Scholar]
- 5. Harvey AE, Lashinger LM, Hursting SD. The growing challenge of obesity and cancer: An inflammatory issue. Ann N Y Acad Sci 2011;1229:45–52. [DOI] [PubMed] [Google Scholar]
- 6. Wu S, Liu J, Wang X, et al. Association of obesity and overweight with overall survival in colorectal cancer patients: A meta‐analysis of 29 studies. Cancer Causes Control 2014;25:1489–1502. [DOI] [PubMed] [Google Scholar]
- 7. Haque R, Van Den Eeden SK, Wallner LP, et al. Association of body mass index and prostate cancer mortality. Obes Res Clin Pract 2014;8:e374–e381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Craig L. Cause of death in dogs according to breed: A necropsy survey of five breeds. J Am Anim Hosp Assoc 2001;37:438–443. [DOI] [PubMed] [Google Scholar]
- 9. Lund EM, Armstrong PJ, Kirk CA, et al. Prevalence and risk factors for obesity in adult dogs from private US veterinary practices. Int J Appl Res Vet Med 2006;4:177–186. [Google Scholar]
- 10. Weeth LP, Fascetti AJ, Kass PH, et al. Prevalence of obese dogs in a population of dogs with cancer. Am J Vet Res 2007;68:389–398. [DOI] [PubMed] [Google Scholar]
- 11. Courcier EA, Thomson RM, Mellor DJ, et al. An epidemiological study of environmental factors associated with canine obesity. J Small Anim Pract 2010;51:362–367. [DOI] [PubMed] [Google Scholar]
- 12. Michel KE, Sorenmo K, Shofer FS. Evaluation of body condition and weight loss in dogs presented to a veterinary oncology service. J Vet Intern Med 2004;18:692–695. [DOI] [PubMed] [Google Scholar]
- 13. Perez Alenza MD, Pena L, del Castillo N, et al. Factors influencing the incidence and prognosis of canine mammary tumours. J Small Anim Pract 2000;41:287–291. [DOI] [PubMed] [Google Scholar]
- 14. Glickman LT, Schofer FS, McKee LJ, et al. Epidemiologic study of insecticide exposures, obesity, and risk of bladder cancer in household dogs. J Toxicol Environ Health 1989;28:407–414. [DOI] [PubMed] [Google Scholar]
- 15. Carson KR, Bartlett NL, McDonald JR, et al. Increased body mass index is associated with improved survival in United States veterans with diffuse large B‐cell lymphoma. J Clin Oncol 2012;30:3217–3222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Han X, Stevens J, Bradshaw PT. Body mass index, weight change, and survival in non‐Hodgkin lymphoma patients in Connecticut women. Nutr Cancer 2013;65:43–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Baez JL, Michel KE, Sorenmo K, et al. A prospective investigation of the prevalence and prognostic significance of weight loss and changes in body condition in feline cancer patients. J Feline Med Surg 2007;9:411–417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Sharma A, Lavie CJ, Borer JS, et al. Meta‐analysis of the relation of body mass index to all‐cause and cardiovascular mortality and hospitalization in patients with chronic heart failure. Am J Cardiol 2015;115:1428–1434. [DOI] [PubMed] [Google Scholar]
- 19. Owen LN, ed. Histological Classification of Hematopoietic Tumors of Domestic Animals. Geneva: World Health Organization; 1980. [Google Scholar]
- 20. Larsson SC, Wolk A. Obesity and risk of non‐Hodgkin's lymphoma: A meta‐analysis. Int J Cancer 2007;121:1564–1570. [DOI] [PubMed] [Google Scholar]
- 21. Kumar A, Bakkum‐Gamez JN, Weaver AL, et al. Impact of obesity on surgical and oncologic outcomes in ovarian cancer. Gynecol Oncol 2014;135:19–24. [DOI] [PubMed] [Google Scholar]
- 22. Larsson SC, Wolk A. Body mass index and risk of non‐Hodgkin's and Hodgkin's lymphoma: A meta‐analysis of prospective studies. Eur J Cancer 2011;47:2422–2430. [DOI] [PubMed] [Google Scholar]
- 23. Navarro WH, Loberiza FR Jr, Bajorunaite R, et al. Effect of body mass index on mortality of patients with lymphoma undergoing autologous hematopoietic cell transplantation. Biol Blood Marrow Transplant 2006;12:541–551. [DOI] [PubMed] [Google Scholar]
- 24. Leo QJ, Ollberding NJ, Wilkens LR, et al. Obesity and non‐Hodgkin lymphoma survival in an ethnically diverse population: The Multiethnic Cohort study. Cancer Causes Control 2014;25:1449–1459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Altaf S, Enders F, Jeavons E, et al. High‐BMI at diagnosis is associated with inferior survival in patients with osteosarcoma: A report from the Children's Oncology Group. Pediatr Blood Cancer 2013;60:2042–2046. [DOI] [PubMed] [Google Scholar]
- 26. Bielack S, Kevric M. High BMI at diagnosis is not associated with inferior survival in patients with osteosarcoma. A report from the Cooperative Osteosarcoma Study Group. Pediatr Blood Cancer 2014;61:952. [DOI] [PubMed] [Google Scholar]
- 27. Altaf S, Enders F, Krailo M, et al. Response to “High BMI at diagnosis is associated with inferior survival in patients with osteosarcoma”. Pediatr Blood Cancer 2014;61:951. [DOI] [PubMed] [Google Scholar]
- 28. Ishioka J, Masuda H, Kijima T, et al. Bimodal pattern of the impact of body mass index on cancer‐specific survival of upper urinary tract urothelial carcinoma patients. Anticancer Res 2014;34:5683–5688. [PubMed] [Google Scholar]
- 29. Miao L, Chen H, Xiang J, et al. A high body mass index in esophageal cancer patients is not associated with adverse outcomes following esophagectomy. J Cancer Res Clin Oncol 2015;141:941–950. [DOI] [PubMed] [Google Scholar]
- 30. Ejaz A, Spolverato G, Kim Y, et al. Impact of body mass index on perioperative outcomes and survival after resection for gastric cancer. J Surg Res 2015;195:74–82. [DOI] [PubMed] [Google Scholar]
- 31. Taylor WF, Ivins JC, Unni KK, et al. Prognostic variables in osteosarcoma: A multi‐institutional study. J Natl Cancer Inst 1989;81:21–30. [DOI] [PubMed] [Google Scholar]
- 32. Slupe JL, Freeman LM, Rush JE. Association of body weight and body condition with survival in dogs with heart failure. J Vet Intern Med 2008;22:561–565. [DOI] [PubMed] [Google Scholar]
- 33. Baldwin K, Bartges J, Buffington T, et al. AAHA nutritional assessment guidelines for dogs and cats. J Am Anim Hosp Assoc 2010;46:285–296. [DOI] [PubMed] [Google Scholar]
- 34. WSAVA Nutritional Assessment Guidelines Taskforce , Freeman L, Becvarova I, Cave N, MacKay C, Nguyen P, Rama B, Takashima G, Tiffin R, Van Beukelen P, Yathiraj S. 2011 Nutritional assessment guidelines. J Small Anim Pract 2011;52:385–396. [DOI] [PubMed] [Google Scholar]
- 35. Abbo AH, Lucroy MD. Assessment of anemia as an independent predictor of response to chemotherapy and survival in dogs with lymphoma: 96 cases (1993‐2006). J Am Vet Med Assoc 2007;231:1836–1842. [DOI] [PubMed] [Google Scholar]
- 36. Miller AG, Morley PS, Rao S, et al. Anemia is associated with decreased survival time in dogs with lymphoma. J Vet Intern Med 2009;23:116–122. [DOI] [PubMed] [Google Scholar]
- 37. Kiupel M, Teske E, Bostock D. Prognostic factors for treated canine malignant lymphoma. Vet Pathol 1999;36:292–300. [DOI] [PubMed] [Google Scholar]
- 38. Greenlee PG, Filippa DA, Quimby FW, et al. Lymphomas in dogs. A morphologic, immunologic, and clinical study. Cancer 1990;66:480–490. [DOI] [PubMed] [Google Scholar]
- 39. Laflamme D. Development and validation of a body condition score system for dogs. Canine Practice 1997;22:10–15. [Google Scholar]
- 40. Parkin E, O'Reilly DA, Sherlock DJ, et al. Excess adiposity and survival in patients with colorectal cancer: A systematic review. Obes Rev 2014;15:434–451. [DOI] [PubMed] [Google Scholar]
- 41. Suzuki H, Asakawa A, Amitani H, et al. Cancer cachexia–pathophysiology and management. J Gastroenterol 2013;48:574–594. [DOI] [PMC free article] [PubMed] [Google Scholar]


