Abstract
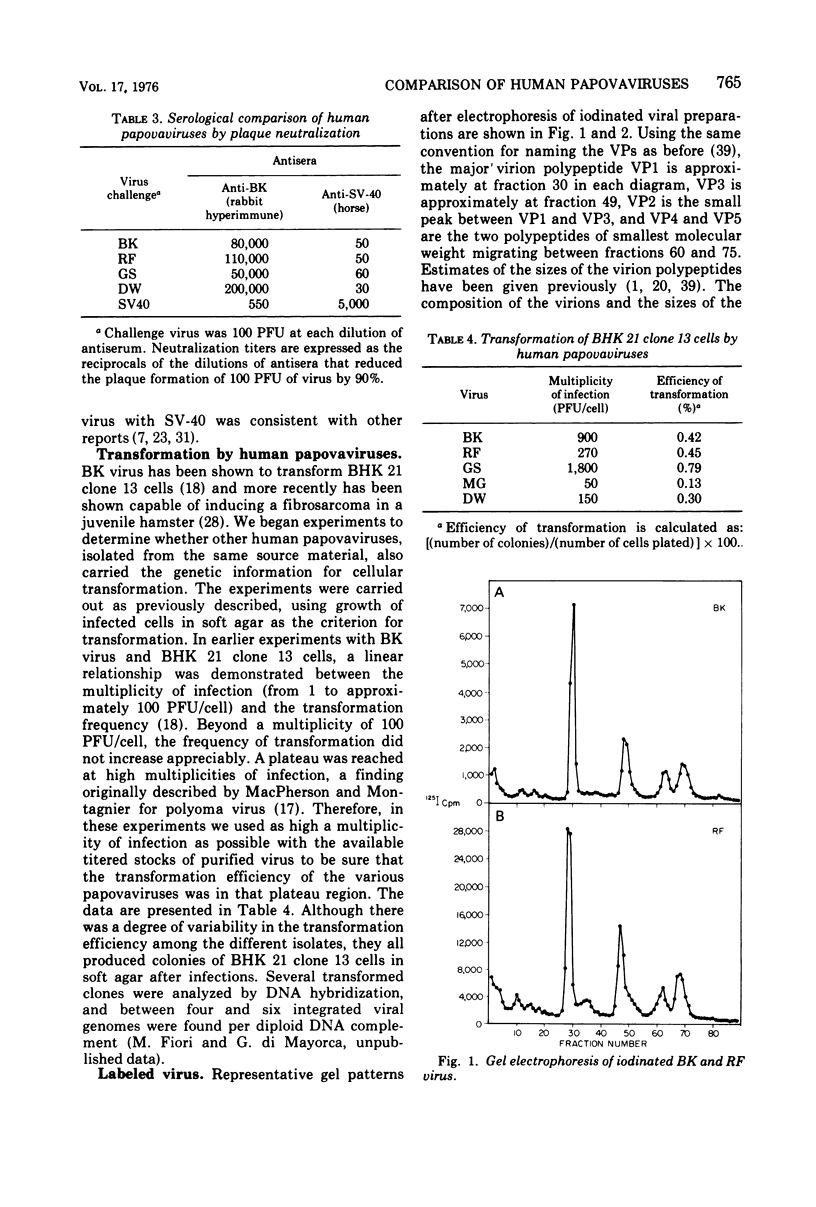
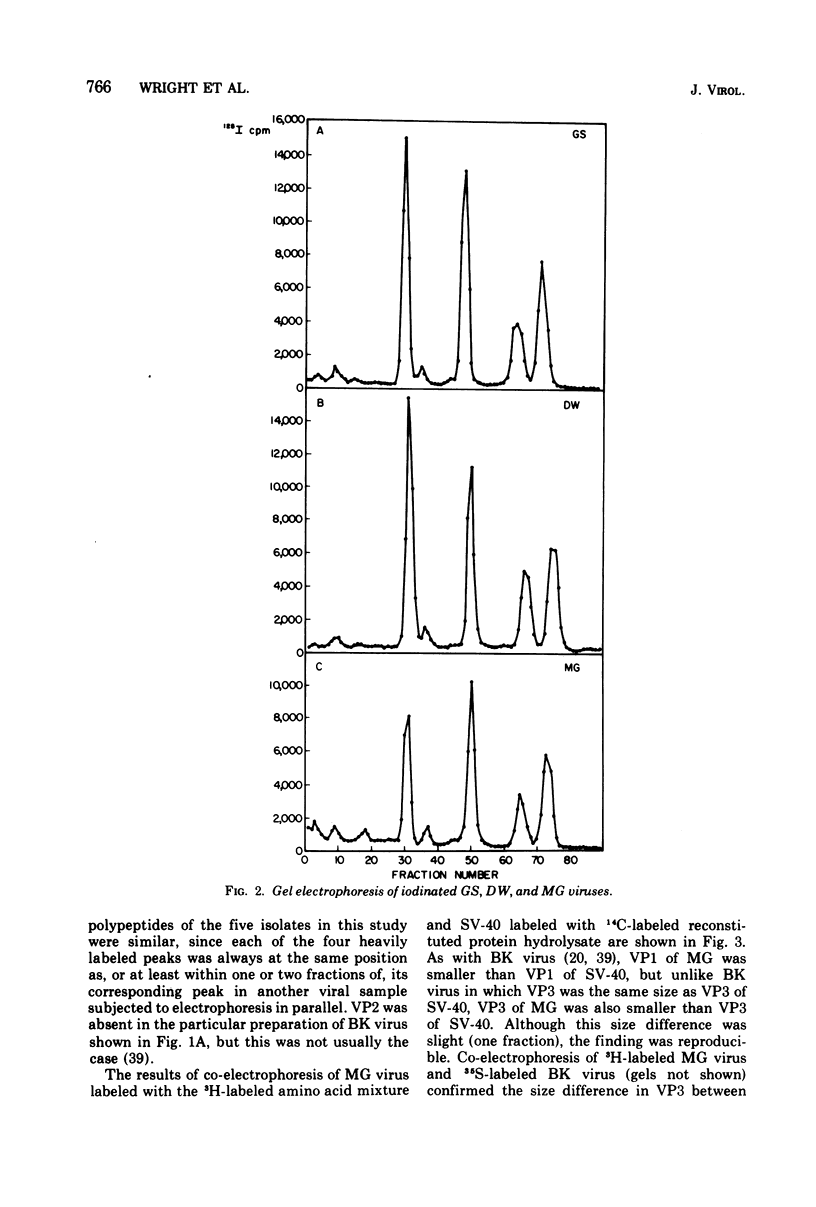
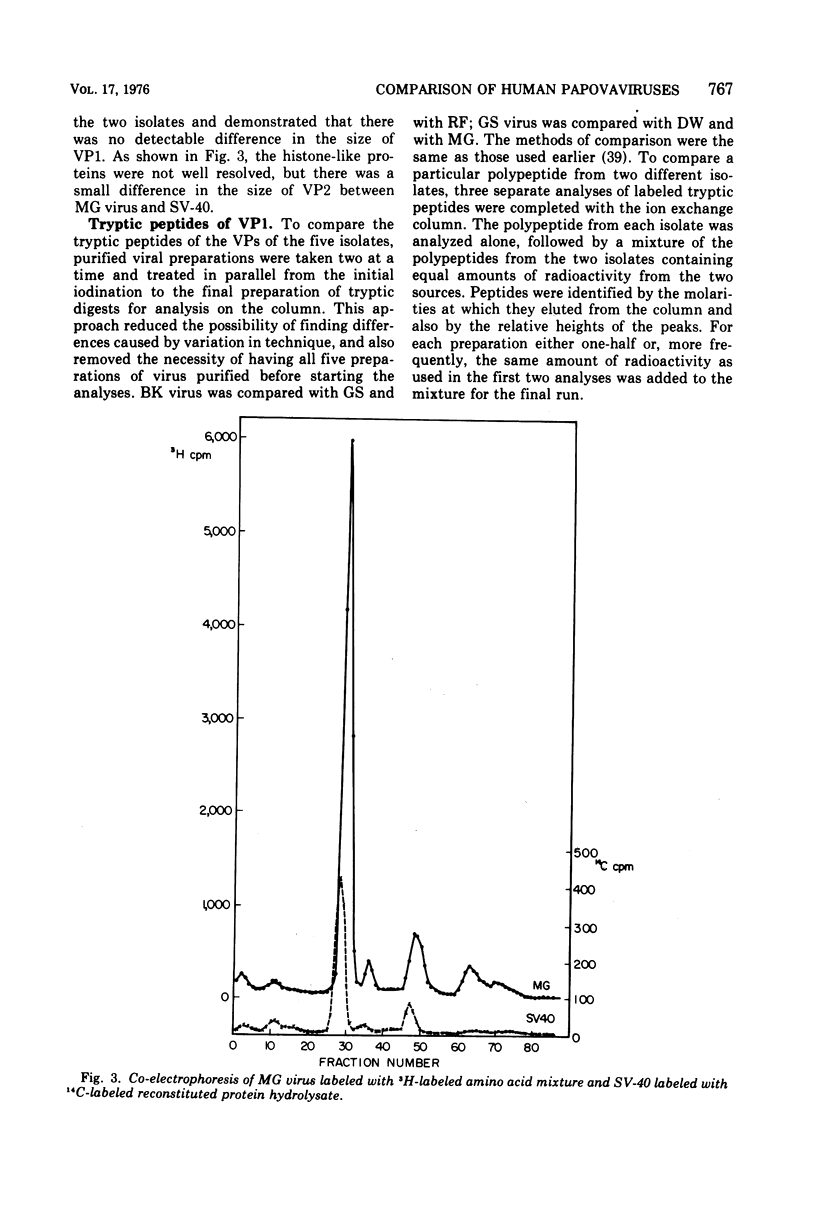
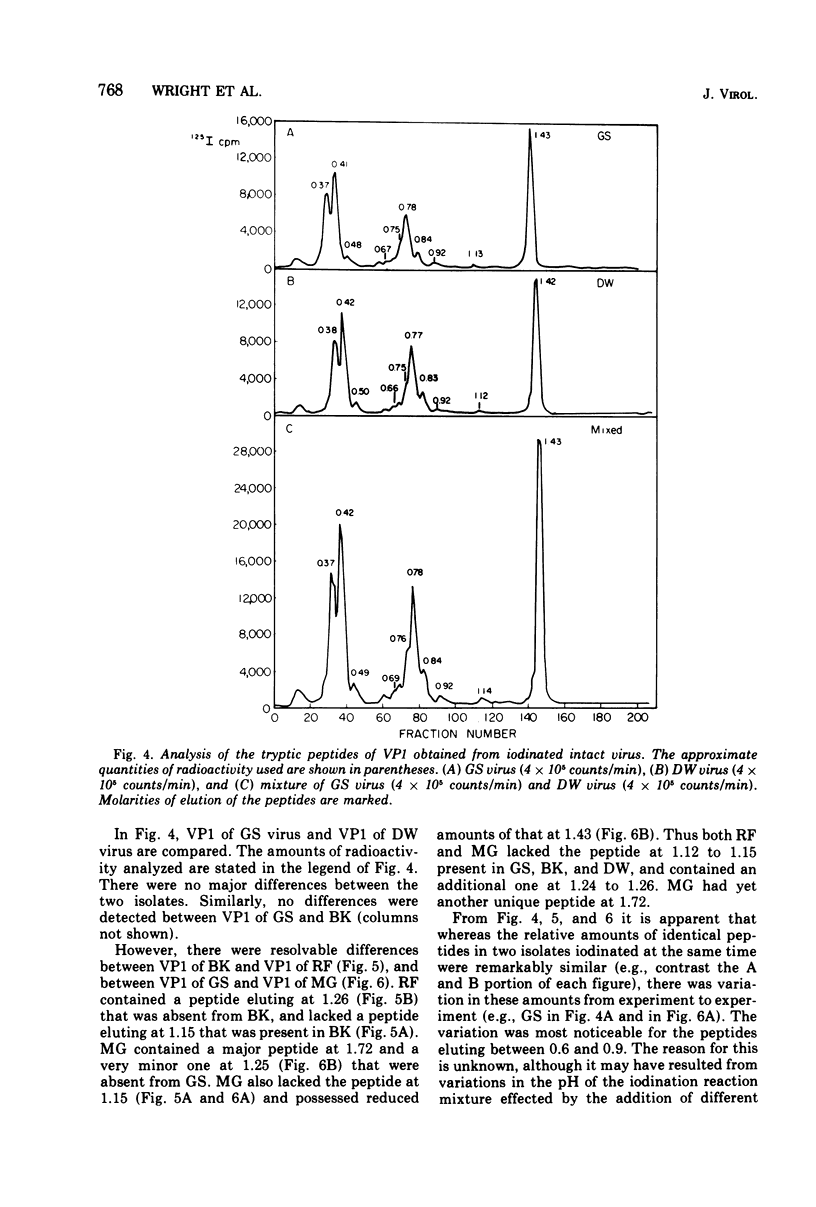
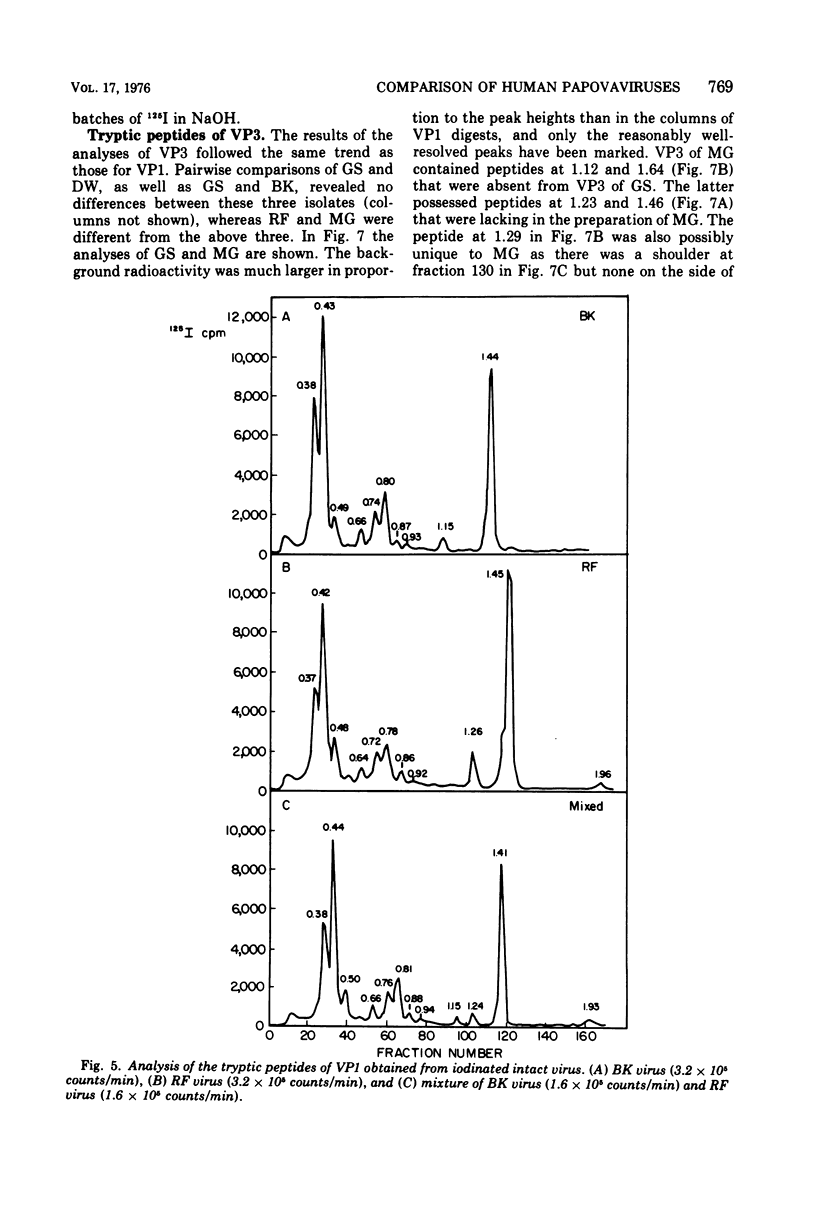
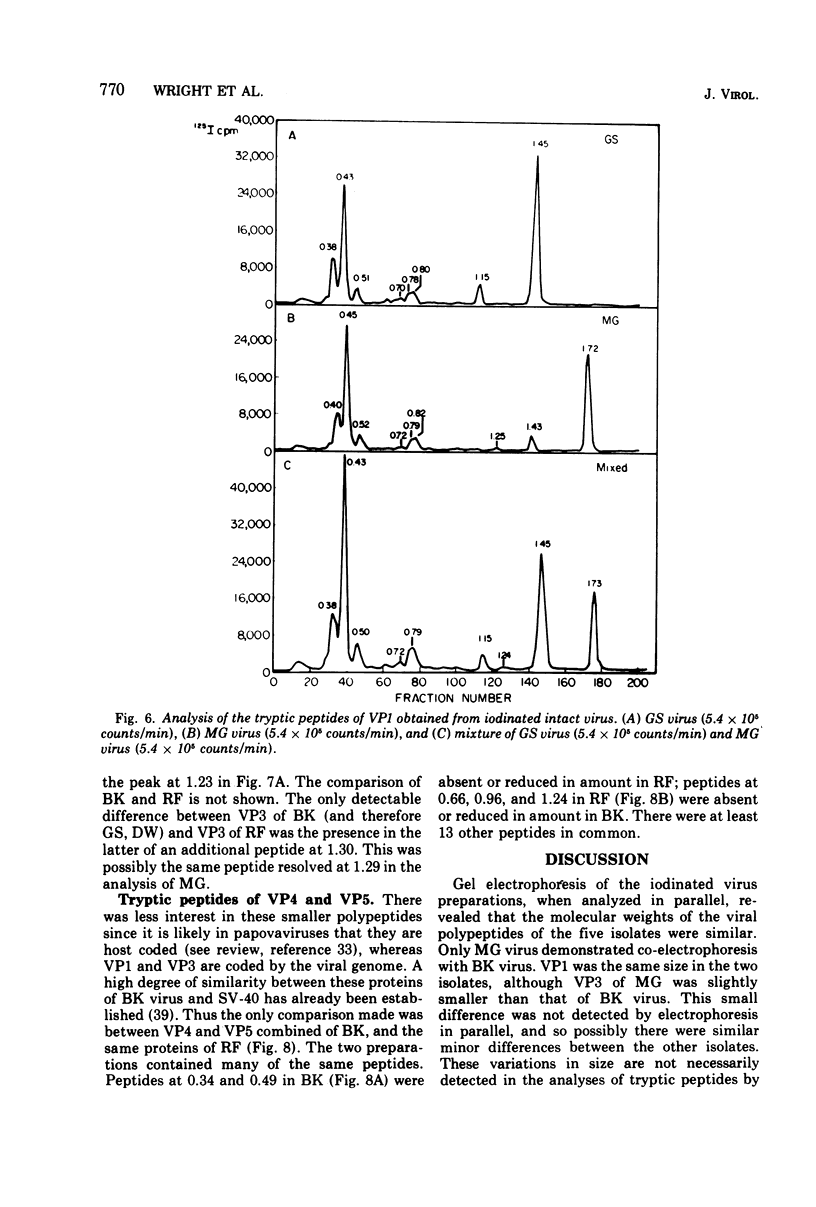
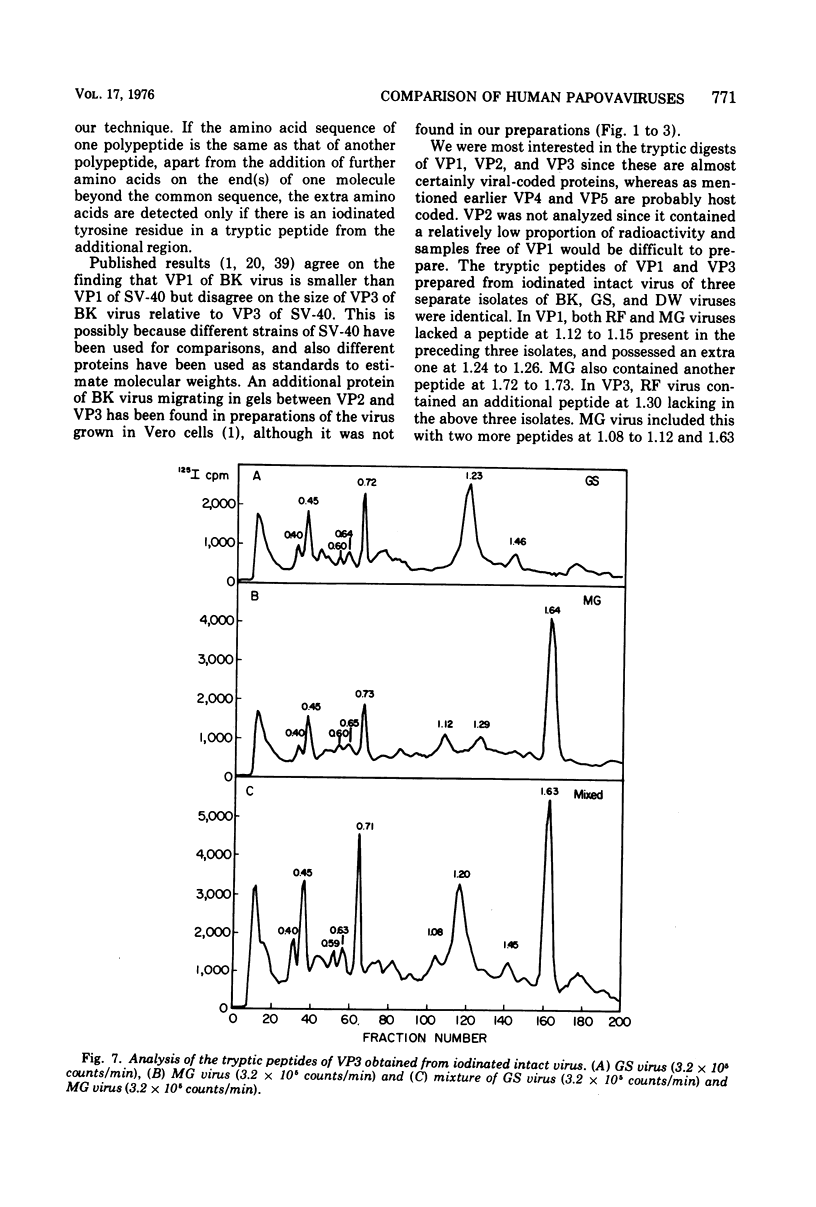
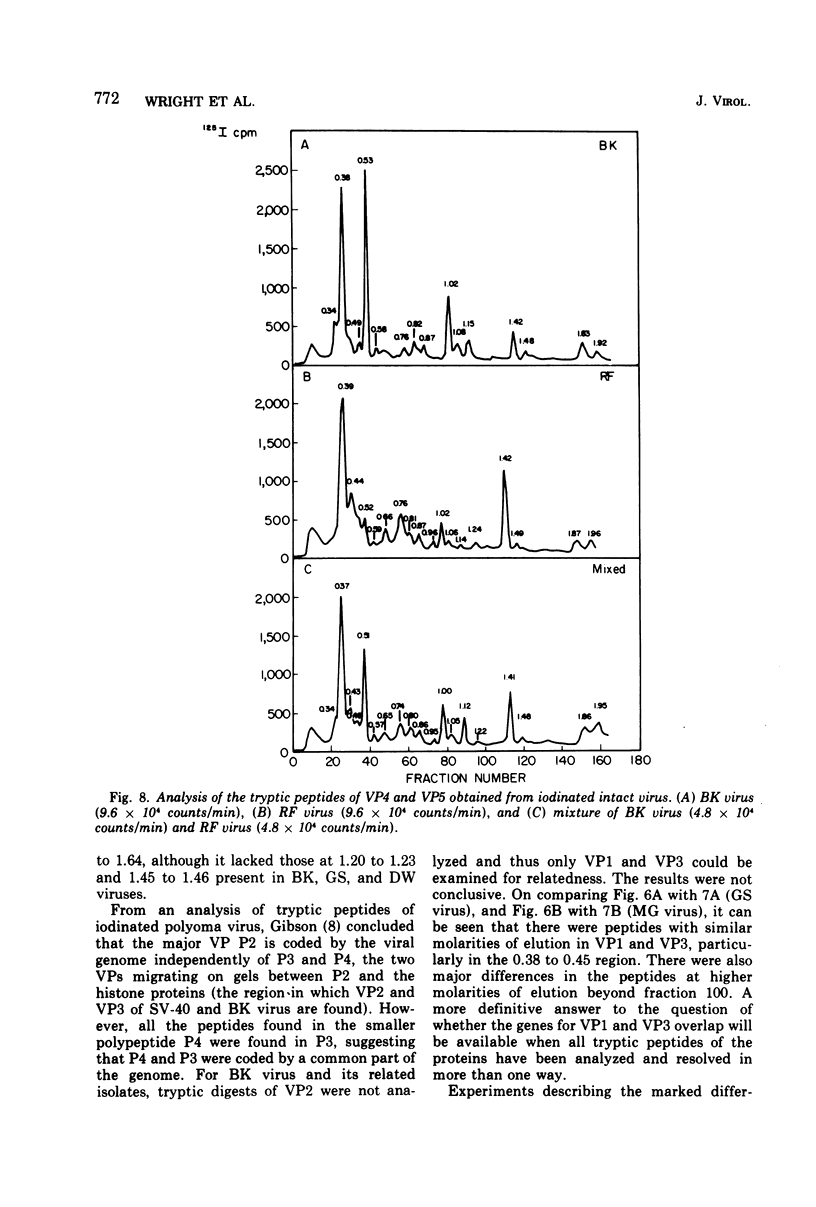
Four isolates of human papovaviruses (RF, GS, DW, and MG viruses) obtained from the urine specimens of renal allograft recipients in widely separated locations were compared with BK virus. Hemagglutination inhibition tests and plaque neutralization assays showed that all were antigenically related to BK virus. All isolates transformed baby hamster kidney cells, transformation being determined by the ability of the cells to plate in soft agar. Purified preparations of each isolate were iodinated with chloramine T and the polypeptide compositions were compared by electrophoresis of disrupted viruses in polyacrylamide gels containing sodium dodecyl sulfate. The gel patterns of all isolates were similar to that of BK virus. The tryptic digests of two major iodinated virion proteins, VP1 and VP3, were analyzed on an ion exchange column. The peptide patterns of GS, DW, and BK virus were identical; those of RF and MG virus closely resembled the patterns of the above three with only minor differences in some peptides. The results show that the four isolates and BK virus are antigenically closely related, have similar onocogenic potential, and are distinguishable from simian virus 40 and JC virus.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barbanti-Brodano G., Minelli G. P., Portolani M., Lambertini L., Toppini M. Structural proteins of a human papovavirus (BK virus): a comparison with the structural proteins of simian virus 40. Virology. 1975 Mar;64(1):269–271. doi: 10.1016/0042-6822(75)90098-7. [DOI] [PubMed] [Google Scholar]
- Blaese R. M., Strober W., Brown R. S., Waldmann T. A. The Wiskott-Aldrich syndrome. A disorder with a possible defect in antigen processing or recognition. Lancet. 1968 May 18;1(7551):1056–1061. doi: 10.1016/s0140-6736(68)91411-6. [DOI] [PubMed] [Google Scholar]
- Coleman D. V., Gardner S. D., Field A. M. Human polyomavirus infection in renal allograft recipients. Br Med J. 1973 Aug 18;3(5876):371–375. doi: 10.1136/bmj.3.5876.371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dougherty R. M., DiStefano H. S. Isolation and characterization of a papovavirus from human urine. Proc Soc Exp Biol Med. 1974 Jun;146(2):481–487. doi: 10.3181/00379727-146-38131. [DOI] [PubMed] [Google Scholar]
- GREENWOOD F. C., HUNTER W. M., GLOVER J. S. THE PREPARATION OF I-131-LABELLED HUMAN GROWTH HORMONE OF HIGH SPECIFIC RADIOACTIVITY. Biochem J. 1963 Oct;89:114–123. doi: 10.1042/bj0890114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardner S. D., Field A. M., Coleman D. V., Hulme B. New human papovavirus (B.K.) isolated from urine after renal transplantation. Lancet. 1971 Jun 19;1(7712):1253–1257. doi: 10.1016/s0140-6736(71)91776-4. [DOI] [PubMed] [Google Scholar]
- Gardner S. D. Prevalence in England of antibody to human polyomavirus (B.k.). Br Med J. 1973 Jan 13;1(5845):77–78. doi: 10.1136/bmj.1.5845.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson W. Polyoma virus proteins: a description of the structural proteins of the virion based on polyacrylamide gel electrophoresis and peptide analysis. Virology. 1974 Dec;62(2):319–336. doi: 10.1016/0042-6822(74)90395-x. [DOI] [PubMed] [Google Scholar]
- Howley P. M., Mullarkey M. F., Takemoto K. K., Martin M. A. Characterization of human papovavirus BK DNA. J Virol. 1975 Jan;15(1):173–181. doi: 10.1128/jvi.15.1.173-181.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobson M. F., Asso J., Baltimore D. Further evidence on the formation of poliovirus proteins. J Mol Biol. 1970 May 14;49(3):657–669. doi: 10.1016/0022-2836(70)90289-5. [DOI] [PubMed] [Google Scholar]
- Jung M., Krech U., Price P. C., Pyndiah M. N. Evidence of chronic persistent infections with polyomaviruses (BK type) in renal transplant recipients. Arch Virol. 1975;47(1):39–46. doi: 10.1007/BF01315591. [DOI] [PubMed] [Google Scholar]
- Lavi S., Winocour E. Acquisition of sequences homologous to host deoxyribonucleic acid by closed circular simian virus 40 deoxyribonucleic acid. J Virol. 1972 Feb;9(2):309–316. doi: 10.1128/jvi.9.2.309-316.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lecatsas G., Prozesky O. W. Excretion of morphological variants of human polyoma virus. Arch Virol. 1975;47(4):393–397. doi: 10.1007/BF01347981. [DOI] [PubMed] [Google Scholar]
- Lecatsas G., Prozesky O. W., van Wyk J., Els H. J. Papova virus in urine after renal transplantation. Nature. 1973 Feb 2;241(5388):343–344. doi: 10.1038/241343a0. [DOI] [PubMed] [Google Scholar]
- MACPHERSON I., MONTAGNIER L. AGAR SUSPENSION CULTURE FOR THE SELECTIVE ASSAY OF CELLS TRANSFORMED BY POLYOMA VIRUS. Virology. 1964 Jun;23:291–294. doi: 10.1016/0042-6822(64)90301-0. [DOI] [PubMed] [Google Scholar]
- Major E. O., Di Mayorca G. Malignant transformation of BHK21 clone 13 cells by BK virus--a human papovavirus. Proc Natl Acad Sci U S A. 1973 Nov;70(11):3210–3212. doi: 10.1073/pnas.70.11.3210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manz H. J., Dinsdale H. B., Morrin P. A. Progressive multifocal leukoencephalopathy after renal transplantation. Demonstration of Papova-like virions. Ann Intern Med. 1971 Jul;75(1):77–81. doi: 10.7326/0003-4819-75-1-77. [DOI] [PubMed] [Google Scholar]
- Mullarkey M. F., Hruska J. F., Takemoto K. K. Comparison of two human papovaviruses with simian virus 40 by structural protein and antigenic analysis. J Virol. 1974 May;13(5):1014–1019. doi: 10.1128/jvi.13.5.1014-1019.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Osborn J. E., Robertson S. M., Padgett B. L., ZuRhein G. M., Walker D. L., Weisblum B. Comparison of JC and BK human papovaviruses with simian virus 40: restriction endonuclease digestion and gel electrophoresis of resultant fragments. J Virol. 1974 Mar;13(3):614–622. doi: 10.1128/jvi.13.3.614-622.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Padgett B. L., Walker D. L., ZuRhein G. M., Eckroade R. J., Dessel B. H. Cultivation of papova-like virus from human brain with progressive multifocal leucoencephalopathy. Lancet. 1971 Jun 19;1(7712):1257–1260. doi: 10.1016/s0140-6736(71)91777-6. [DOI] [PubMed] [Google Scholar]
- Penney J. B., Jr, Narayan O. Studies of the antigenic relationships of the new human papovaviruses by electron microscopy agglutination. Infect Immun. 1973 Aug;8(2):299–300. doi: 10.1128/iai.8.2.299-300.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plummer G., Masterson J. G. Herpes simplex virus and cancer of the cervix. Am J Obstet Gynecol. 1971 Sep;111(1):81–84. doi: 10.1016/0002-9378(71)90929-x. [DOI] [PubMed] [Google Scholar]
- Portolani M., Barbanti-Brodano G., Placa M. L. Malignant transformation of hamster kidney cells by BK virus. J Virol. 1975 Feb;15(2):420–422. doi: 10.1128/jvi.15.2.420-422.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reese J. M., Reissing M., Daniel R. W., Shah K. V. Occurrence of BK virus and BK virus-specific antibodies in the urine of patients receiving chemotherapy for malignancy. Infect Immun. 1975 Jun;11(6):1375–1381. doi: 10.1128/iai.11.6.1375-1381.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sack G. H., Jr, Narayan O., Danna K. J., Weiner L. P., Nathans D. The nucleic acid of an SV40-like virus isolated from a patient with progressive multifocal leukoencephalopathy. Virology. 1973 Feb;51(2):345–350. doi: 10.1016/0042-6822(73)90433-9. [DOI] [PubMed] [Google Scholar]
- Shah K. V., Daniel R. W., Strandberg J. D. Sarcoma in a hamster inoculated with BK virus, a human papovavirus. J Natl Cancer Inst. 1975 Apr;54(4):945–950. [PubMed] [Google Scholar]
- Shah K. V., Daniel R. W., Warszawski R. M. High prevalence of antibodies to BK virus, an SV40-related papovavirus, in residents of Maryland. J Infect Dis. 1973 Dec;128(6):784–787. doi: 10.1093/infdis/128.6.784. [DOI] [PubMed] [Google Scholar]
- Tai H. T., Smith C. A., Sharp P. A., Vinograd J. Sequence heterogeneity in closed simian virus 40 deoxyribonucleic acid. J Virol. 1972 Feb;9(2):317–325. doi: 10.1128/jvi.9.2.317-325.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takemoto K. K., Mullarkey M. F. Human papovavirus, BK strain: biological studies including antigenic relationship to simian virus 40. J Virol. 1973 Sep;12(3):625–631. doi: 10.1128/jvi.12.3.625-631.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takemoto K. K., Rabson A. S., Mullarkey M. F., Blaese R. M., Garon C. F., Nelson D. Isolation of papovavirus from brain tumor and urine of a patient with Wiskott-Aldrich syndrome. J Natl Cancer Inst. 1974 Nov;53(5):1205–1207. doi: 10.1093/jnci/53.5.1205. [DOI] [PubMed] [Google Scholar]
- Walker D. L., Padgett B. L., ZuRhein G. M., Albert A. E., Marsh R. F. Human papovavirus (JC): induction of brain tumors in hamsters. Science. 1973 Aug 17;181(4100):674–676. doi: 10.1126/science.181.4100.674. [DOI] [PubMed] [Google Scholar]
- Weiner L. P., Herndon R. M., Narayan O., Johnson R. T., Shah K., Rubinstein L. J., Preziosi T. J., Conley F. K. Isolation of virus related to SV40 from patients with progressive multifocal leukoencephalopathy. N Engl J Med. 1972 Feb 24;286(8):385–390. doi: 10.1056/NEJM197202242860801. [DOI] [PubMed] [Google Scholar]
- Weiner L. P., Narayan O., Penney J. B., Jr, Herndon R. M., Feringa E. R., Tourtellotte W. W., Johnson R. T. Papovavirus of JC type in progressive multifocal leukoencephalopathy. Rapid identification and subsequent isolation. Arch Neurol. 1973 Jul;29(1):1–3. doi: 10.1001/archneur.1973.00490250019001. [DOI] [PubMed] [Google Scholar]
- Weiner L. P., Narayan O. Virologic studies of progressive multifocal leukoencephalopathy. Prog Med Virol. 1974;18(0):229–240. [PubMed] [Google Scholar]
- Wright P. J., Di Mayorca G. Virion polypeptide composition of the human papovavirus BK: comparison with simian virus 40 and polyoma virus. J Virol. 1975 Apr;15(4):828–835. doi: 10.1128/jvi.15.4.828-835.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]



