Abstract
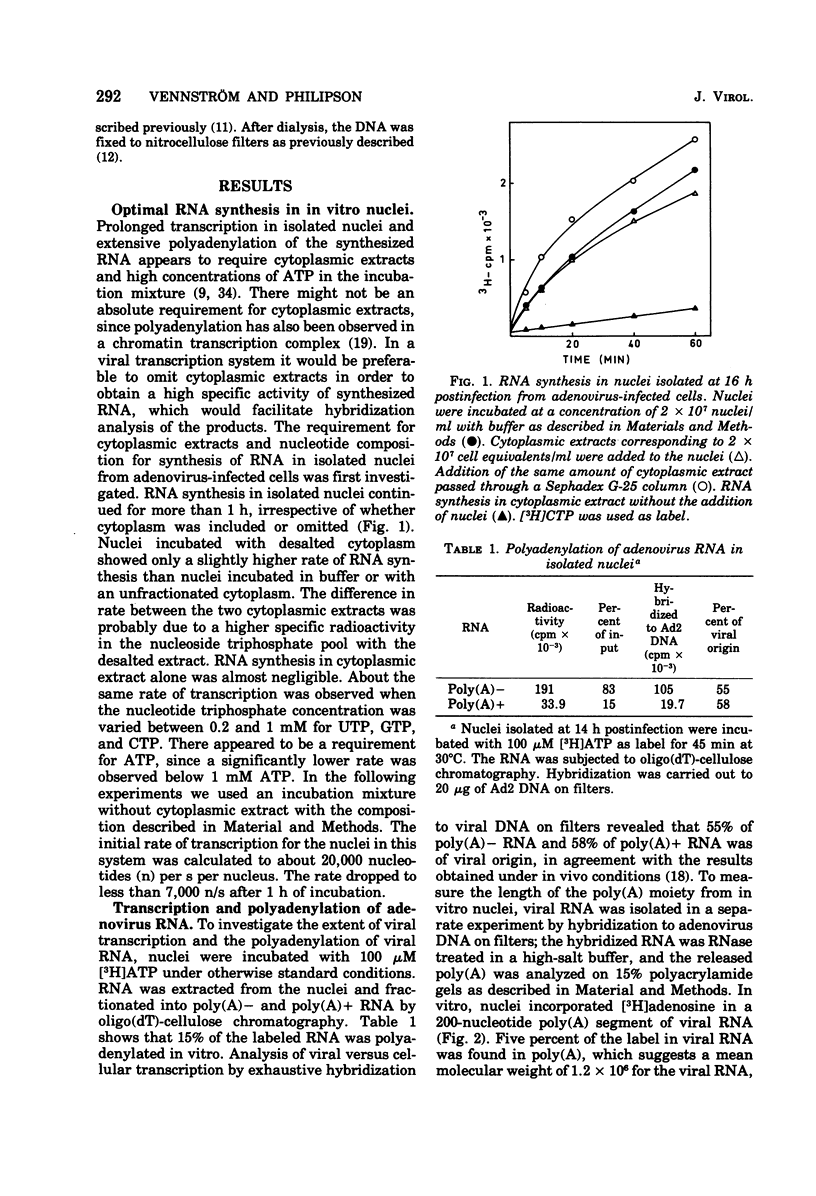
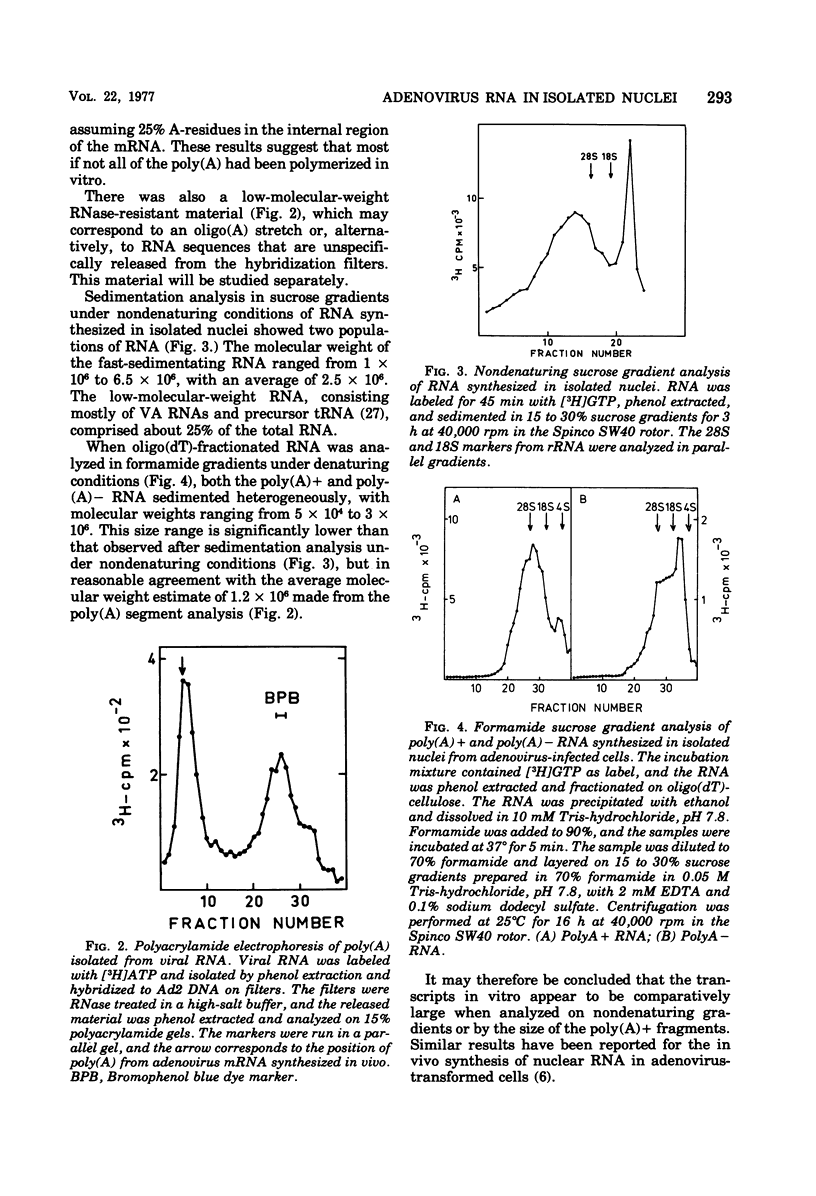
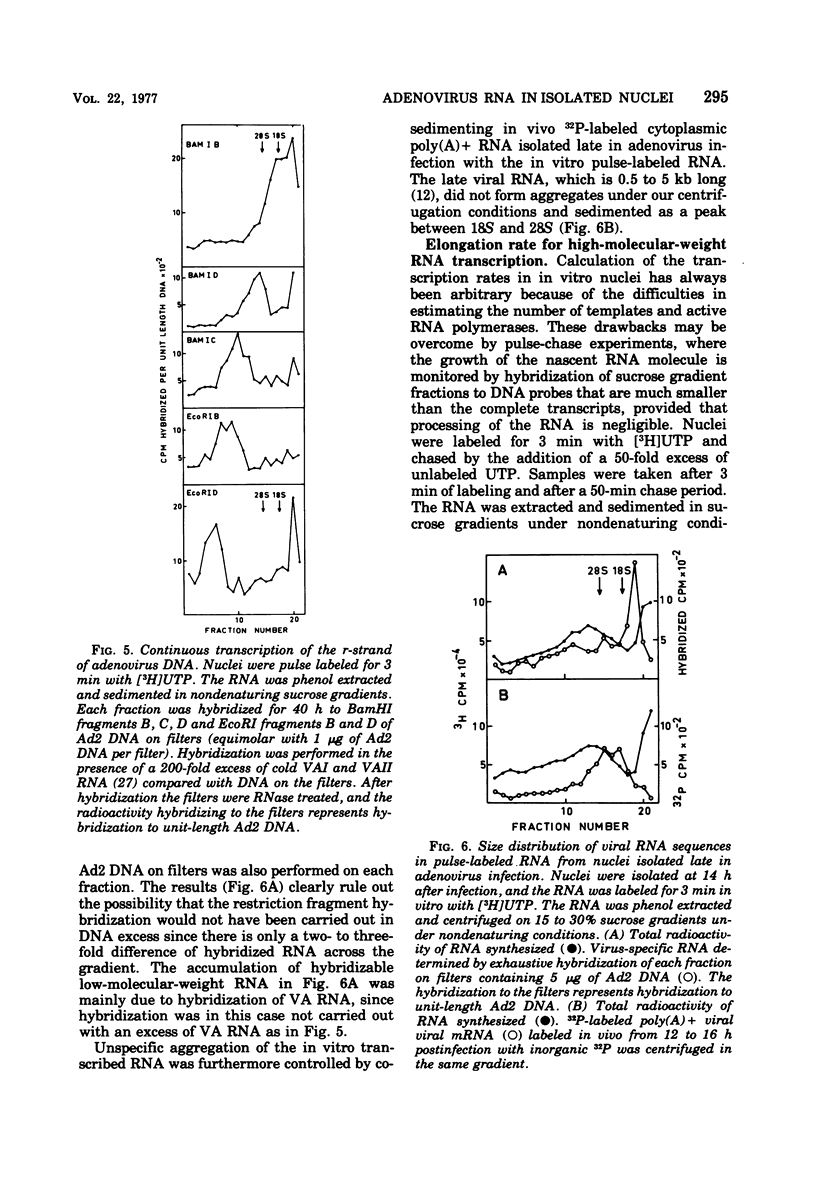
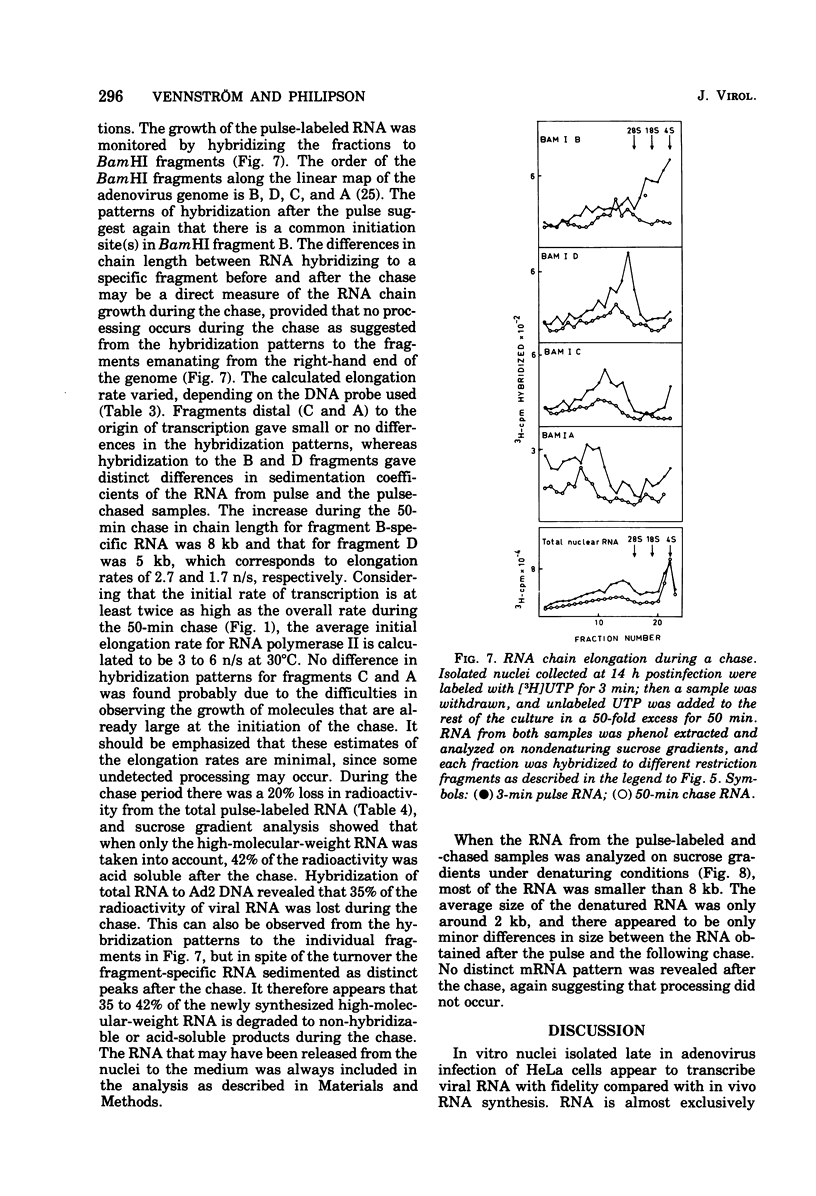
An in vitro nuclear system from adenovirus type 2-infected cells was developed to study transcription of viral RNA. Nuclei isolated from adenovirus-infected HeLa cells late in the infectious cycle synthesized in vitro only RNA from the r-strand of adenovirus DNA. Around 15% of the virus-specific RNA in isolated nuclei was polyadenylated. Short pulse labeling of nascent RNA followbd by hybrization of size-fractionated RNA to specific restriction endonuclease fragments of the genome suggested that the origin(s) for transcription is located on the r-strand in the left 30% of the adenovirus 2 genome at late times in the infectious cycle. Pulse-chase experiments were used to estimate the elongation rate of adenovirus high-molecular-weight RNA in isolated nuclei. An elongation of a least six nucleotides per second was observed in vitro. Viral RNA synthesis in the vitro nuclei showed several similarities to the in vivo system late in the infectious cycle.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bachenheimer S., Darnell J. E. Adenovirus-2 mRNA is transcribed as part of a high-molecular-weight precursor RNA. Proc Natl Acad Sci U S A. 1975 Nov;72(11):4445–4449. doi: 10.1073/pnas.72.11.4445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Busiello E., Di Girolamo M. RNA metabolism in nuclei isolated from HeLa cells. Eur J Biochem. 1975 Jun 16;55(1):61–70. doi: 10.1111/j.1432-1033.1975.tb02138.x. [DOI] [PubMed] [Google Scholar]
- DINTZIS H. M. Assembly of the peptide chains of hemoglobin. Proc Natl Acad Sci U S A. 1961 Mar 15;47:247–261. doi: 10.1073/pnas.47.3.247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Pomerai D. I., Butterworth P. H. Polyadenylation of RNA in vitro in isolated chromatin and nuclei. Eur J Biochem. 1975 Oct 1;58(1):185–192. doi: 10.1111/j.1432-1033.1975.tb02363.x. [DOI] [PubMed] [Google Scholar]
- Flint S. J., Sharp P. A. Adenovirus transcription. V. Quantitation of viral RNA sequences in adenovirus 2-infected and transformed cells. J Mol Biol. 1976 Sep 25;106(3):749–774. doi: 10.1016/0022-2836(76)90263-1. [DOI] [PubMed] [Google Scholar]
- Georgieff M., Bachenheimer S., Darnell J. E. An examination of the nuclear RNA of adenovirus-transformed cells. Cold Spring Harb Symp Quant Biol. 1975;39(Pt 1):475–482. doi: 10.1101/sqb.1974.039.01.059. [DOI] [PubMed] [Google Scholar]
- Holmes D. S., Bonner J. Preparation, molecular weight, base composition, and secondary structure of giant nuclear ribonucleic acid. Biochemistry. 1973 Jun 5;12(12):2330–2338. doi: 10.1021/bi00736a023. [DOI] [PubMed] [Google Scholar]
- Jackson A. H., Sugden B. Inhibition by -amanitin of simian virus 40-specific ribonucleic acid synthesis in nuclei of infected monkey cells. J Virol. 1972 Nov;10(5):1086–1089. doi: 10.1128/jvi.10.5.1086-1089.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jelinek W. R. Poly adenylic acid synthesis in vitro in isolated HeLa cell nuclei and whole cell homogenates,. Cell. 1974 Jul;2(3):197–204. doi: 10.1016/0092-8674(74)90094-4. [DOI] [PubMed] [Google Scholar]
- Landgraf-Leurs M., Green M. DNA strand selection during the transcription of the adenovirus 2 genome in infected and transformed cells. Biochim Biophys Acta. 1973 Jul 27;312(4):667–673. doi: 10.1016/0005-2787(73)90070-1. [DOI] [PubMed] [Google Scholar]
- Lewis J. B., Atkins J. F., Anderson C. W., Baum P. R., Gesteland R. F. Mapping of late adenovirus genes by cell-free translation of RNA selected by hybridization to specific DNA fragments. Proc Natl Acad Sci U S A. 1975 Apr;72(4):1344–1348. doi: 10.1073/pnas.72.4.1344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindberg U., Persson T., Philipson L. Isolation and characterization of adenovirus messenger ribonucleic acid in productive infection. J Virol. 1972 Nov;10(5):909–919. doi: 10.1128/jvi.10.5.909-919.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magnusson G., Winnacker E. L., Eliasson R., Reichard P. Replication of polyoma DNA in isolated nuclei. II. Evidence for semi-conservative replication. J Mol Biol. 1972 Dec 30;72(3):539–552. doi: 10.1016/0022-2836(72)90173-8. [DOI] [PubMed] [Google Scholar]
- Marzluff W. F., Jr, Murphy E. C., Jr, Huang R. C. Transcription of ribonucleic acid in isolated mouse myeloma nuclei. Biochemistry. 1973 Aug 28;12(18):3440–3446. doi: 10.1021/bi00742a013. [DOI] [PubMed] [Google Scholar]
- Marzluff W. F., Jr, Murphy E. C., Jr, Huang R. C. Transcription of the genes for 5S ribosomal RNA and transfer RNA in isolated mouse myeloma cell nuclei. Biochemistry. 1974 Aug 27;13(18):3689–3696. doi: 10.1021/bi00715a011. [DOI] [PubMed] [Google Scholar]
- Parsons J. T., Gardner J., Green M. Biochemical studies on adenovirus multiplication, XIX. Resolution of late viral RNA species in the nucleus and cytoplasm. Proc Natl Acad Sci U S A. 1971 Mar;68(3):557–560. doi: 10.1073/pnas.68.3.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pettersson U., Philipson L. Synthesis of complementary RNA sequences during productive adenovirus infection. Proc Natl Acad Sci U S A. 1974 Dec;71(12):4887–4891. doi: 10.1073/pnas.71.12.4887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Philipson L., Pettersson U., Lindberg U., Tibbetts C., Vennström B., Persson T. RNA synthesis and processing in adenovirus-infected cells. Cold Spring Harb Symp Quant Biol. 1975;39(Pt 1):447–456. doi: 10.1101/sqb.1974.039.01.057. [DOI] [PubMed] [Google Scholar]
- Price R., Penman S. Transcription of the adenovirus genome by an -amanitine-sensitive ribonucleic acid polymerase in HeLa cells. J Virol. 1972 Apr;9(4):621–626. doi: 10.1128/jvi.9.4.621-626.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raskas H. J., Bhaduri S. Poly(adenylic acid) sequences in adenovirus ribonucleic acid released from isolated nuclei. Biochemistry. 1973 Feb 27;12(5):920–925. doi: 10.1021/bi00729a020. [DOI] [PubMed] [Google Scholar]
- Raskas H. J. Release of adenovirus messenger RNA from isolated nuclei. Nat New Biol. 1971 Sep 29;233(39):134–136. doi: 10.1038/newbio233134a0. [DOI] [PubMed] [Google Scholar]
- Reeder R. H., Roeder R. G. Ribosomal RNA synthesis in isolated nuclei. J Mol Biol. 1972 Jun 28;67(3):433–441. doi: 10.1016/0022-2836(72)90461-5. [DOI] [PubMed] [Google Scholar]
- Reichard P., Eliasson R., Söderman G. Initiator RNA in discontinuous polyoma DNA synthesis. Proc Natl Acad Sci U S A. 1974 Dec;71(12):4901–4905. doi: 10.1073/pnas.71.12.4901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- STUDIER F. W. SEDIMENTATION STUDIES OF THE SIZE AND SHAPE OF DNA. J Mol Biol. 1965 Feb;11:373–390. doi: 10.1016/s0022-2836(65)80064-x. [DOI] [PubMed] [Google Scholar]
- Sambrook J., Williams J., Sharp P. A., Grodzicker T. Physical mapping of temperature-sensitive mutations of adenoviruses. J Mol Biol. 1975 Sep 25;97(3):369–390. doi: 10.1016/s0022-2836(75)80046-5. [DOI] [PubMed] [Google Scholar]
- Söderlund H., Pettersson U., Vennström B., Philipson L., Mathews M. B. A new species of virus-coded low molecular weight RNA from cells infected with adenovirus type 2. Cell. 1976 Apr;7(4):585–593. doi: 10.1016/0092-8674(76)90209-9. [DOI] [PubMed] [Google Scholar]
- Tibbetts C., Johansson K., Philipson L. Hydroxyapatite chromatography and formamide denaturation of adenovirus DNA. J Virol. 1973 Aug;12(2):218–225. doi: 10.1128/jvi.12.2.218-225.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tibbetts C., Pettersson U. Complementary strand-specific sequences from unique fragments of adenovirus type 2 DNA for hybridization-mapping experiments. J Mol Biol. 1974 Oct 5;88(4):767–784. doi: 10.1016/0022-2836(74)90398-2. [DOI] [PubMed] [Google Scholar]
- Wall R., Philipson L., Darnell J. E. Processing of adenovirus specific nuclear RNA during virus replication. Virology. 1972 Oct;50(1):27–34. doi: 10.1016/0042-6822(72)90342-x. [DOI] [PubMed] [Google Scholar]
- Wallace R. D., Kates J. State of adenovirus 2 deoxyribonucleic acid in the nucleus and its mode of transcription: studies with isolated viral deoxyribonucleic acid-protein complexes and isolated nuclei. J Virol. 1972 Apr;9(4):627–635. doi: 10.1128/jvi.9.4.627-635.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weinmann R., Raskas H. J., Roeder R. G. Role of DNA-dependent RNA polymerases II and III in transcription of the adenovirus genome late in productive infection. Proc Natl Acad Sci U S A. 1974 Sep;71(9):3426–3439. doi: 10.1073/pnas.71.9.3426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilhelm J., Brison O., Kedinger C., Chambon P. Characterization of adenovirus type 2 transcriptional complexes isolated from infected HeLa cell nuclei. J Virol. 1976 Jul;19(1):61–81. doi: 10.1128/jvi.19.1.61-81.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu G. J., Zubay G. Prolonged transcription in a cell-free system involving nuclei and cytoplasm. Proc Natl Acad Sci U S A. 1974 May;71(5):1803–1807. doi: 10.1073/pnas.71.5.1803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zylber E. A., Penman S. Products of RNA polymerases in HeLa cell nuclei. Proc Natl Acad Sci U S A. 1971 Nov;68(11):2861–2865. doi: 10.1073/pnas.68.11.2861. [DOI] [PMC free article] [PubMed] [Google Scholar]



