Abstract
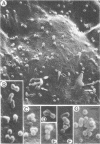
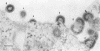
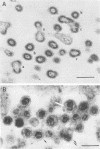
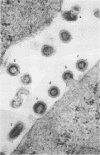
The effect of temperature shiftdown on the assembly of ts3 virions was investigated by both scanning (SEM) and transmission (TEM) electron microscopy. Ts3 is a spontaneous temperature-sensitive mutant of Moloney murine leukemia virus (Mo-MuLV) which previous studies indicated to be defective in assembly or release of the virions. In the present study, both SEM and TEM revealed the following: (i) there were more cell-associated virions in ts3-infected cells grown at the nonpermissive temperature (39°C) than either in cells grown at the permissive temperature (34°C) or in wild-type MuLV-infected cells grown at 39°C; (ii) there were more normal single particles than multiploids (virions with two or more pieces of genomic RNA) in ts3-infected cells grown at the nonpermissive temperature; (iii) there were more multiploids in ts3-infected cells grown at the nonpermissive temperature than either in cells grown at the permissive temperature or in wild-type MuLV-infected cells grown at the nonpermissive temperature; (iv) upon temperature shift from 39 to 34°C, about 90% of the cell-associated virions dissociated from the cell surface. TEM studies also indicated that upon temperature shiftdown, virion assembly rapidly occurred. The above observations suggest that faulty assembly, which results in the production of multiploids, may not be the reason why ts3 virions accumulate on the cell surface at the nonpermissive temperature. The relatively higher proportion of multiploids found in ts3-infected cells grown at 39°C compared with those grown at 34°C may be due to the higher density of budding virions at the cell surface at the nonpermissive temperature, which increases the possibility of two or more particles assembling close to one another. The accumulation of ts3 virions in all stages of assembly at the nonpermissive temperature, together with the fact that rapid assembly and release of ts3 virions occurred on temperature shiftdown, indicates that virion assembly is restricted after it has been initiated. The probable role of altered glycoprotein(s) in restricting virion assembly is discussed.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Blough H. A., Tiffany J. M. Theoretical aspects of structure and assembly of viral envelops. Curr Top Microbiol Immunol. 1975;70:1–30. doi: 10.1007/978-3-642-66101-3_1. [DOI] [PubMed] [Google Scholar]
- Friis R. R., Ogura H., Gelderblom H., Halpern M. S. The defective maturation of viral progeny with a temperature-sensitive mutant of avian sarcoma virus. Virology. 1976 Aug;73(1):259–272. doi: 10.1016/0042-6822(76)90079-9. [DOI] [PubMed] [Google Scholar]
- Hunter E., Hayman M. J., Rongey R. W., Vogt P. K. An avian sarcoma virus mutant that is temperature sensitive for virion assembly. Virology. 1976 Jan;69(1):35–49. doi: 10.1016/0042-6822(76)90192-6. [DOI] [PubMed] [Google Scholar]
- Klenk H. D. Viral envelopes and their relationship to cellular membranes. Curr Top Microbiol Immunol. 1974;(68):29–58. doi: 10.1007/978-3-642-66044-3_2. [DOI] [PubMed] [Google Scholar]
- Sato T., Friend C., De Harven E. Ultrastructural changes in Friend erythroleukemia cells treated with dimethyl sulfoxide. Cancer Res. 1971 Oct;31(10):1402–1417. [PubMed] [Google Scholar]
- Stephenson J. R., Aaronson S. A. Characterization of temperature-sensitive mutants of murine leukemia virus. Virology. 1973 Jul;54(1):53–59. doi: 10.1016/0042-6822(73)90113-x. [DOI] [PubMed] [Google Scholar]
- Wong P. K., MacLeod R. Studies on the budding process of a temperature-sensitive mutant of murine leukemia virus with a scanning electron microscope. J Virol. 1975 Aug;16(2):434–442. doi: 10.1128/jvi.16.2.434-442.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong P. K., McCarter J. A. Studies of two temperature-sensitive mutants of Moloney murine leukemia virus. Virology. 1974 Apr;58(2):396–408. doi: 10.1016/0042-6822(74)90075-0. [DOI] [PubMed] [Google Scholar]
- Wong P. K., Russ L. J., McCarter J. A. Rapid, selective procedure for isolation of spontaneous temperature-sensitive mutants of Moloney leukemia virus. Virology. 1973 Feb;51(2):424–431. doi: 10.1016/0042-6822(73)90441-8. [DOI] [PubMed] [Google Scholar]
- Wong P. K., Yuen P. H., MacLeod R., Chang E. H., Myers M. W., Friedman R. M. The effect of interferon on de novo infection of Moloney murine leukemia virus. Cell. 1977 Feb;10(2):245–252. doi: 10.1016/0092-8674(77)90218-5. [DOI] [PubMed] [Google Scholar]
- Yeger H., Kalnins V. I. Electron microscopy of mammalian type-C RNA viruses: use of conditional lethal mutants in studies on virion maturation and assembly. Virology. 1976 Oct 15;74(2):459–469. [PubMed] [Google Scholar]
- Yuen P. H., Wong P. K. A morphological study on the ultrastructure and assembly of murine leukemia virus using a temperature-sensitive mutant restricted in assembly. Virology. 1977 Jul 15;80(2):260–274. doi: 10.1016/s0042-6822(77)80003-2. [DOI] [PubMed] [Google Scholar]